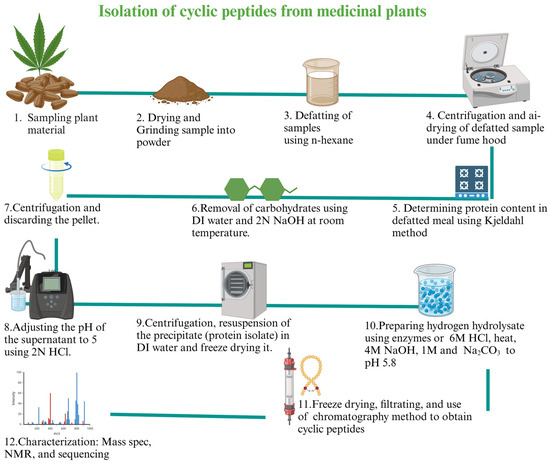
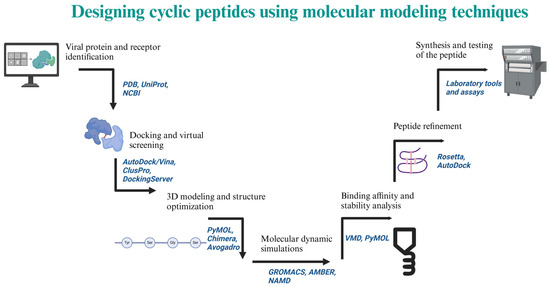
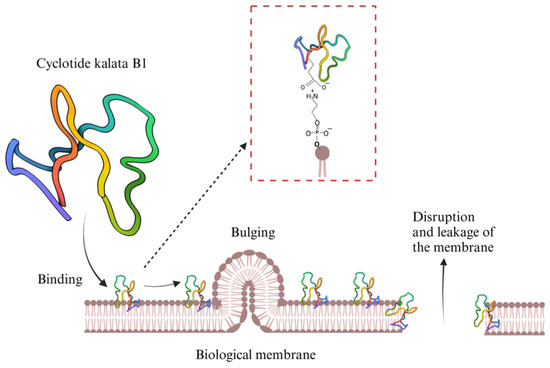
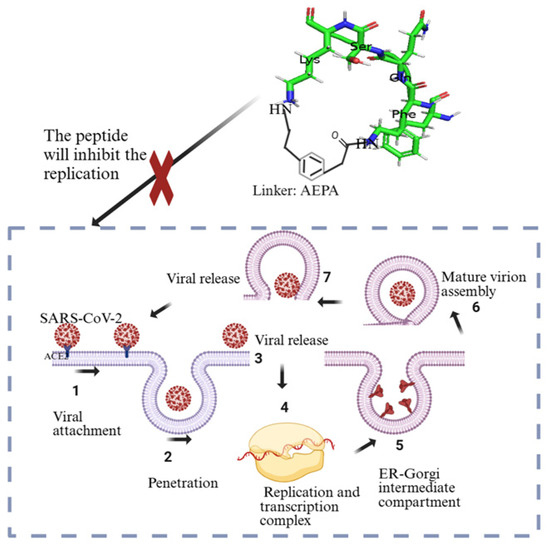
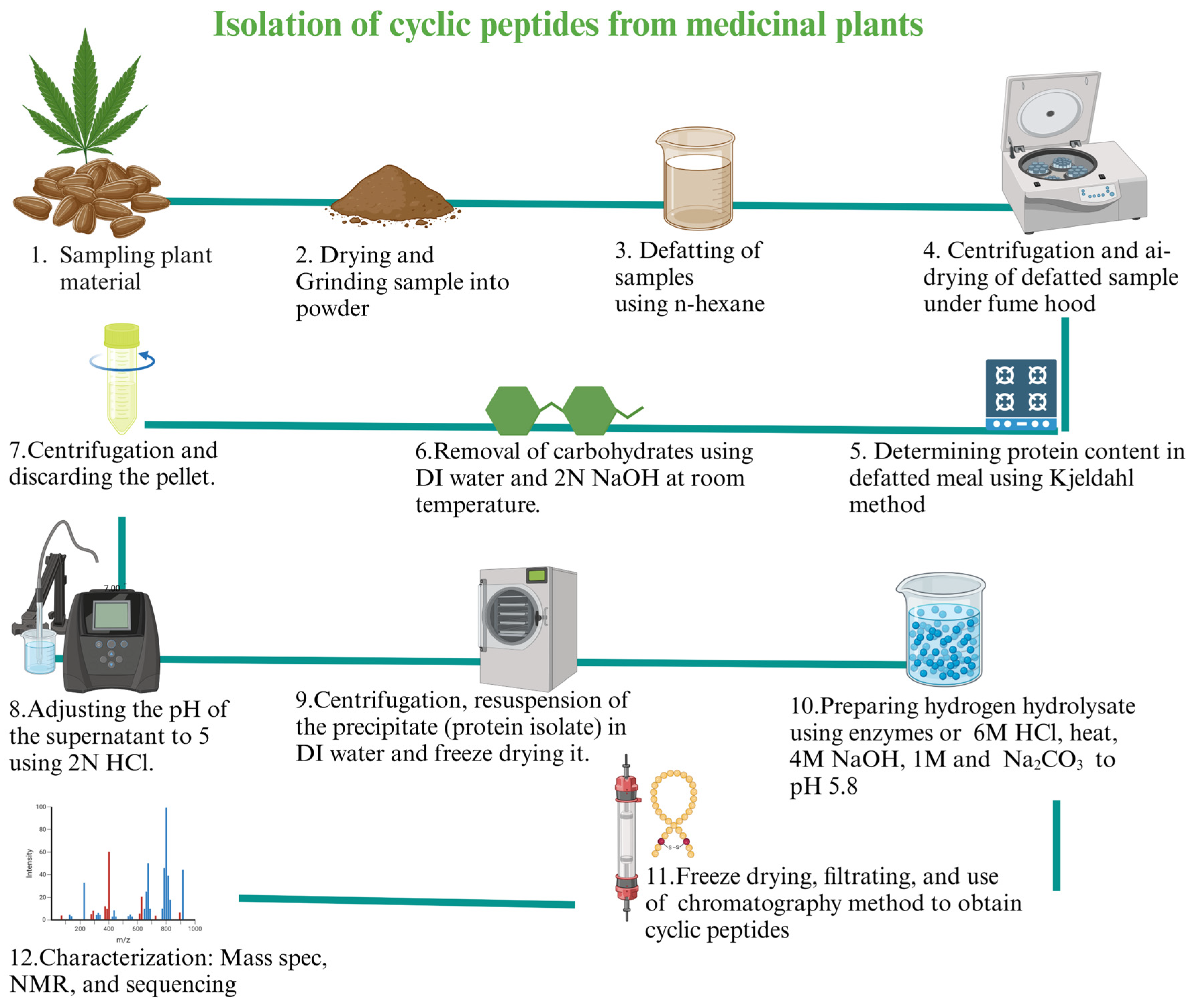
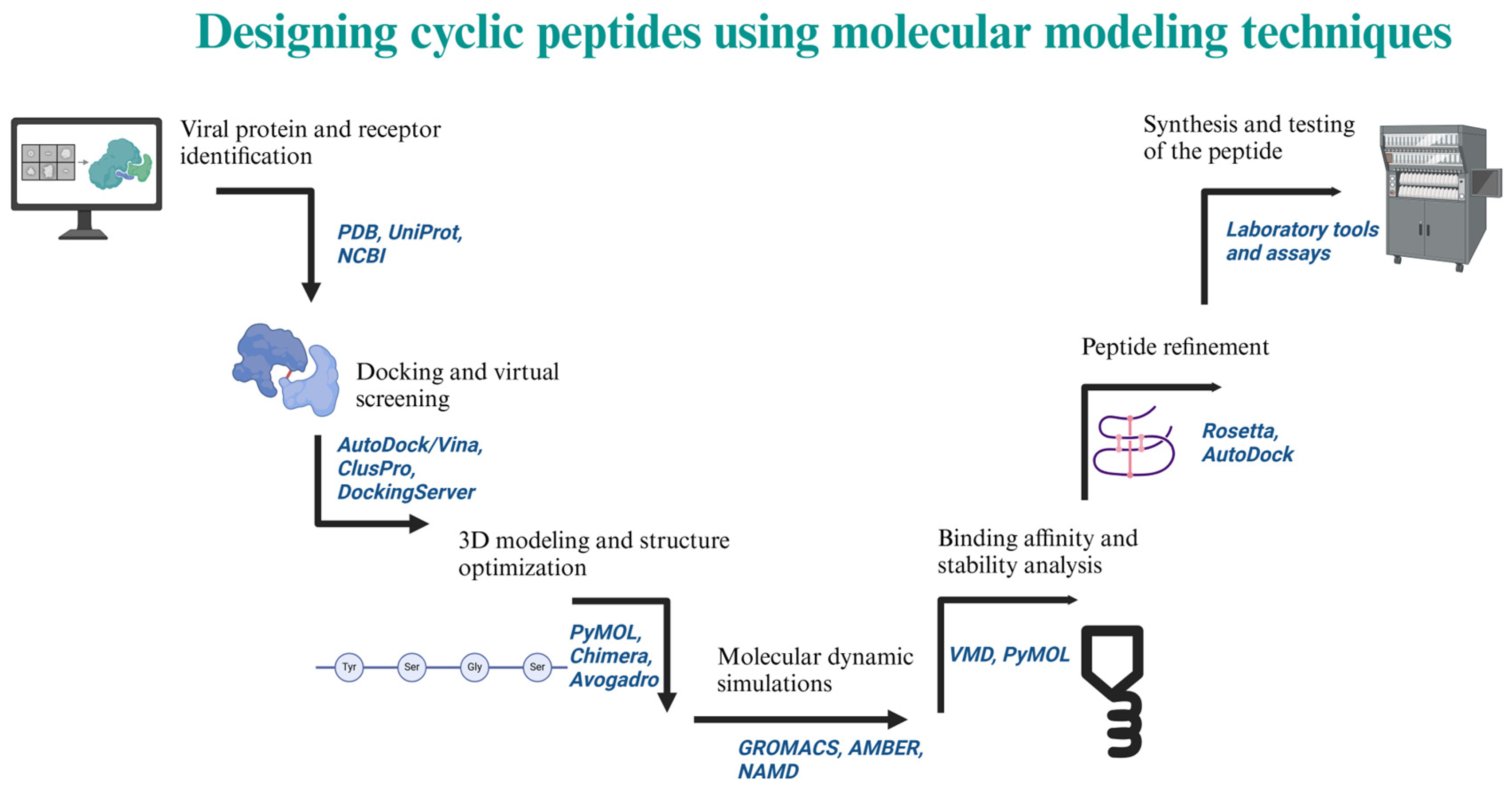
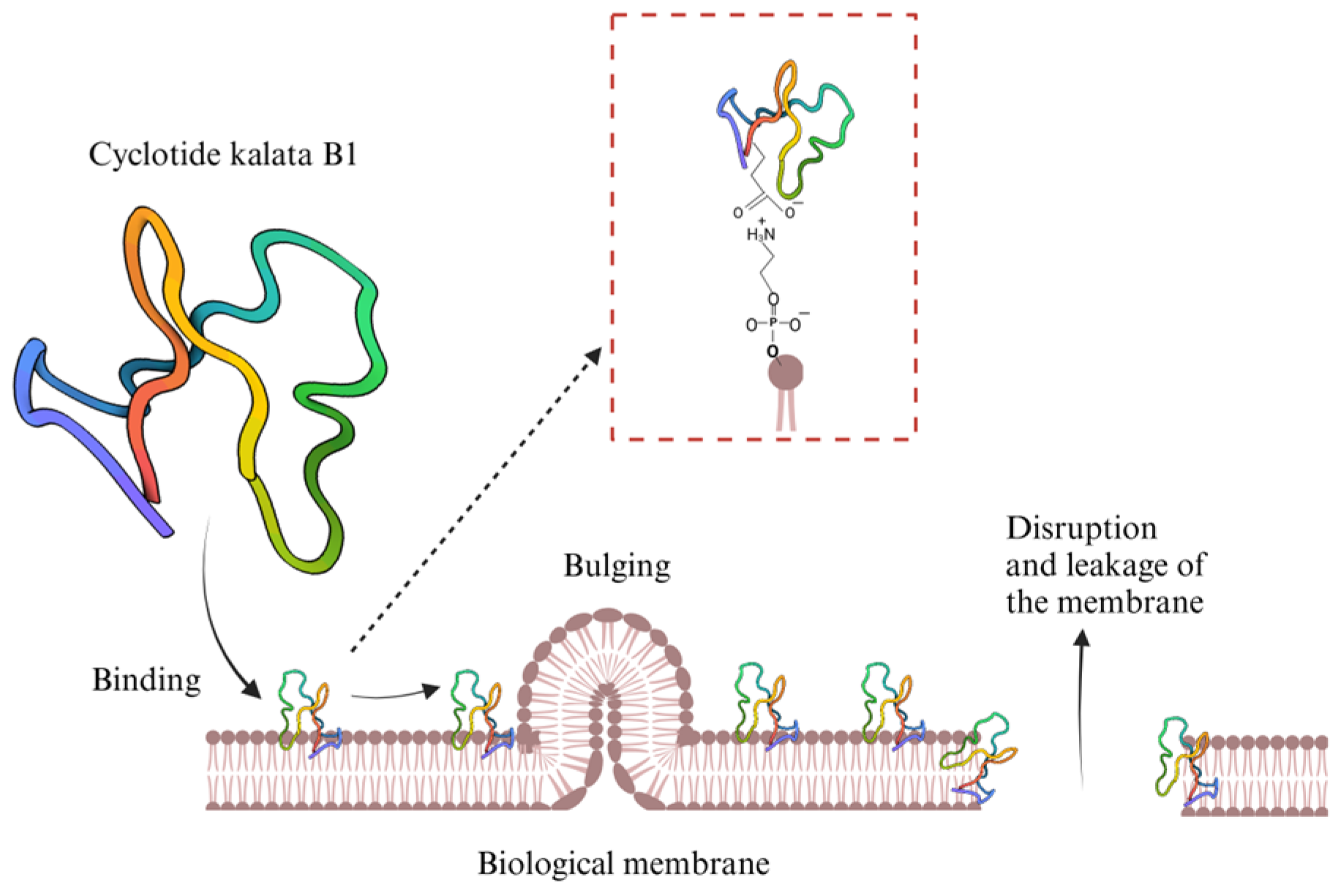
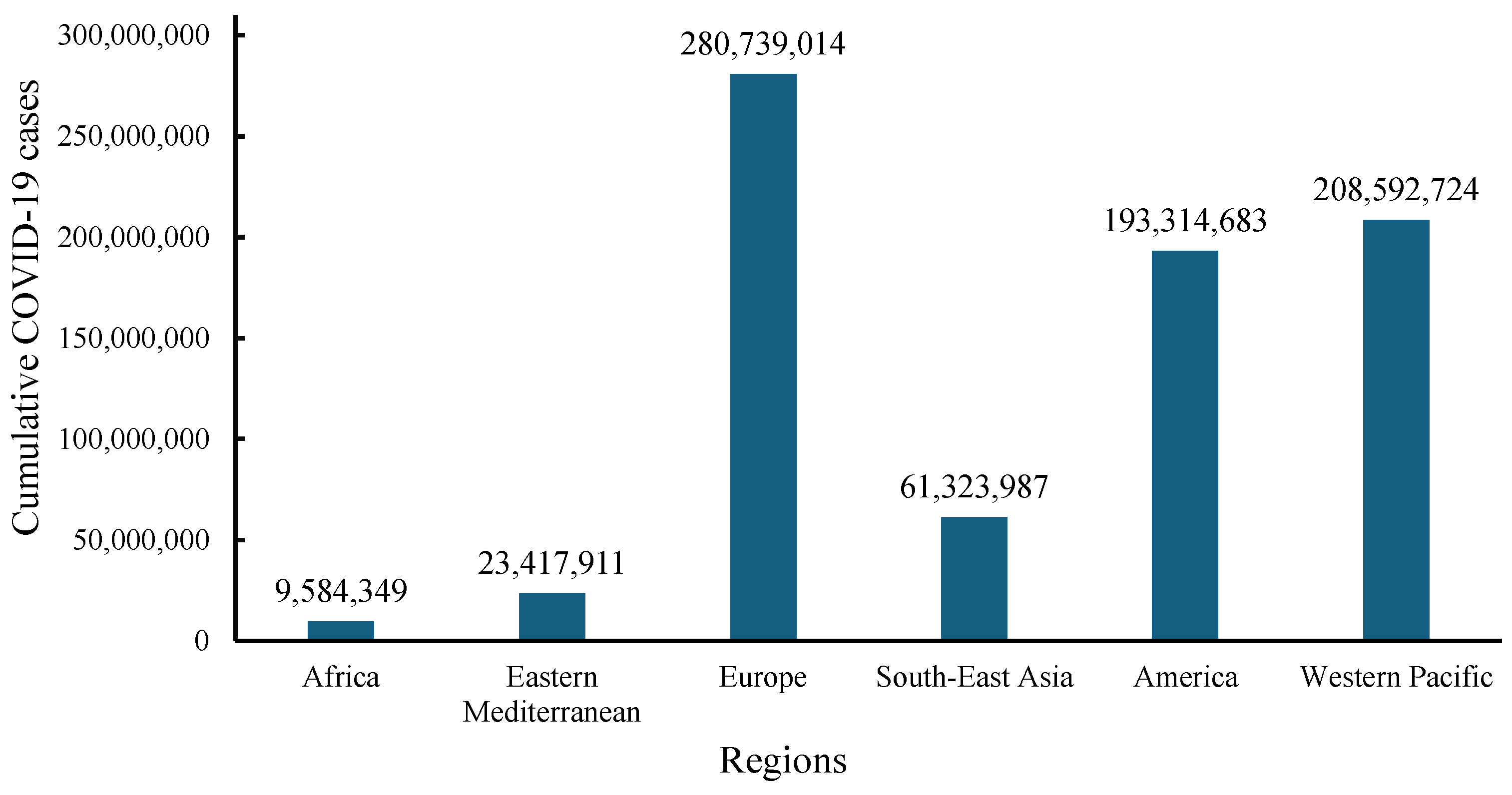Abstract
The COVID-19 pandemic has underscored the need for effective and affordable antiviral drugs. Anthropogenic activities have increased interactions among humans, animals, and wildlife, contributing to the emergence of new and re-emerging viral diseases. RNA viruses pose significant challenges due to their rapid mutation rates, high transmissibility, and ability to adapt to host immune responses and antiviral treatments. The World Health Organization has identified several diseases (COVID-19, Ebola, Marburg, Zika, and others), all caused by RNA viruses, designated as being of priority concern as potential causes of future pandemics. Despite advances in antiviral treatments, many viruses lack specific therapeutic options, and more importantly, there is a paucity of broad-spectrum antiviral drugs. Additionally, the high costs of current treatments such as Remdesivir and Paxlovid highlight the need for more affordable antiviral drugs. Cyclic peptides from natural sources or designed through molecular modeling have shown promise as antiviral drugs with stability, low toxicity, high target specificity, and low antiviral resistance properties. This review emphasizes the urgent need to develop specific and broad-spectrum antiviral drugs and highlights cyclic peptides as a sustainable solution to combat future pandemics. Further research into these compounds could provide a new weapon to combat RNA viruses and address the gaps in current antiviral drug development.
1. Introduction
Globalization, trade, and other anthropogenic activities increase contact among humans, animals, and other wildlife populations [1]. These interactions contribute to new and reemerging viral disease outbreaks that pose great public health challenges [1,2,3]. Among the potential pathogens that cause interspecies diseases, RNA viruses are of special concern [2,4]. RNA viruses have received much attention due to significant differences compared with DNA viruses in terms of their evolution, rapid transmission, mutation, and life-threatening nature [5,6]. RNA viruses pose a greater threat than DNA viruses because of their high mutation rate and rapid adaptation to new environments, including the host immune response and antiviral drugs [7]. This is due to the error-prone nature of RNA-dependent polymerases, which lack the ability to proofread misincorporated nucleotides during RNA-dependent RNA replication. Consequently, this leads to the formation of diverse variants. In contrast, DNA viruses have more robust proofreading mechanisms [7,8].
The World Health Organization (WHO) has identified several diseases caused by RNA viruses that have the potential to cause future deadly outbreaks and pandemics. These include COVID-19, Middle East respiratory syndrome (MERS), severe acute respiratory syndrome (SARS), Crimean–Congo hemorrhagic fever, Ebola virus disease, Marburg virus disease, Lassa fever, Nipah and other henipaviral diseases, Rift Valley fever, Zika, and Disease X [9]. RNA viruses responsible for causing these diseases originate from reservoir species, often mammals, and jump to humans to cause syndromes of varying forms and severity [10]. Furthermore, despite the discoveries made and the attempts to combat viral diseases, specific treatments for many viruses are lacking because of viral escape mutants [11].
Previous outbreaks and pandemics have not only overwhelmed medical and public health capacity but also created enormous strain on economic, scientific, and political systems [12]. If we are wise enough to heed their warnings, recent pandemics, especially those in the twentieth and twenty-first centuries, provide valuable insights into designing and implementing more effective strategies to combat future pandemics.
The world was unprepared for the COVID-19 pandemic and had to combat the threat through ill-conceived wellness protocols, experimental vaccines, and repurposed antiviral drugs. For example, hand sanitizers, face masks, social distancing, lockdowns, mRNA vaccines, and off-label antiviral drugs were extensively deployed to control the COVID-19/SARS-CoV-2 outbreak.
All three of these approaches to combating viral pandemics (wellness protocols, vaccines, and antiviral drugs) are crucial to ultimate success. However, all these approaches require continual research and development. While each approach has its own limitations and risks, all these approaches are vital and deserve equal funding. The COVID-19 pandemic sadly demonstrated what happens when one approach (vaccines) is favored while another approach (antiviral drugs) is neglected.
During the COVID-19 pandemic, wellness protocols included the use of personal protective equipment (PPE) such as face masks to prevent the spread of SARS-CoV-2, with different types of PPE available for public and healthcare settings [13]. However, masks had limitations including discomfort during prolonged use, breathing difficulties, and variable filtration [13]. Chemical disinfectants and sanitizers such as detergents, bleach, and alcohols were also widely used to target the outer lipid layer of the coronavirus and deactivate the viral particle [14], but their excessive use led to respiratory issues, irritation, inflammation, swelling, and problems affecting the central nervous system and gastrointestinal tract [15]. Social distancing measures, including lockdowns and stay-at-home orders, significantly impacted mental health, leading to increased anxiety, depression, stress, suicide attempts, violence, substance abuse, and domestic abuse [16]. In addition, economic hardships such as job losses and food insecurity were the direct consequences of shortsighted government responses to the pandemic [17]. Furthermore, school closures led to loss of teaching and socialization as well as loss of access to free meals and physical activity programs for children [18].
Vaccines became the preferred approach to combating the COVID-19 pandemic even though there were no approved vaccines when it started. By January 2025, the WHO had granted emergency approval for numerous vaccines to target SARS-CoV-2 variants, including mRNA vaccines (Pfizer/BioNTech Comirnaty and Moderna Spikevax) [19], non-replicating viral vector vaccines (AstraZeneca Vaxzevria, Janssen (Johnson & Johnson) Jcovden, Serum Institute of India Covishield (Oxford/AstraZeneca formulation) [19], and CanSino Convidecia [20]), inactivated vaccines (Sinopharm (Beijing) Covilo, Sinovac CoronaVac, and Bharat Biotech Covaxin) [21], and protein subunit vaccines (Novavax Nuvaxovid, Serum Institute of India COVOVAX (Novavax formulation), Corbevax [22], and SK Bioscience Co Ltd. SKYCovione [23]).
However, to adequately assess the effectiveness and safety of these new vaccines, human trials had to be conducted over significant time frames. Cases of liver damage (hepatotoxicity) were reported following vaccination against COVID-19 [24]. Another study found that adenovirus-based vaccines were associated with increased risk of blood clots (thrombosis) and low platelet levels (thrombocytopenia), while mRNA vaccines were linked to a higher risk of heart inflammation (myocarditis), with a fatality rate of 1–2 per 200 cases [25]. There is also evidence of how vaccines can trigger immune reactions leading to serious neurological harm [25]. Many countries, including the United States, Germany, France, United Kingdom, Canada, Australia, Japan, Taiwan, Singapore, and Thailand have implemented compensation programs for vaccine-related injuries and death [26]. However, such compensation cannot bring back lost lives or even reverse the harm. These previously reported findings highlight the need for continued vigilance, as similar adverse effects may occur in individuals receiving vaccines in the future.
It is even more alarming that the long-term safety of experimental vaccines is completely unknown because of the compressed time taken for their approval. It is well known that some vaccines have shown significant side effects long after the initial vaccination. For example, a study of the tetravalent dengue vaccine (CYD-TDV) in participants from 2 to 16 years of age found that vaccinated individuals were more frequently hospitalized (3%) than the control group (1.8%) [27]. This raised concerns that the vaccine increased the incidence of dengue disease in people [28]. The long-term effects of the experimental COVID-19 vaccines are completely unknown.
The third approach for fighting viral pandemics involves the use of antiviral drugs; as yet, however, there are very few specific antiviral drugs and no broad-spectrum antiviral drugs. As a 2021 editorial in Nature noted, “when the COVID-19 pandemic hit, the medicine cabinet was all but empty” [29]. During the pandemic, the US Food and Drug Administration (FDA) approved the use of Remdesivir to treat hospitalized patients with severe symptoms of COVID-19 [30]; unfortunately, this drug was limited and costly (>USD 3100 per patient) [31]. Some of the other drugs used to treat the disease included monoclonal antibodies such as casirivimab and imdevimab (administered together) or bamlanivimab and etesevimab (administered together)] [30]. Furthermore, in 2023, the FDA approved Paxlovid, which contains nirmatrelvir and ritonavir, for the treatment of mild to moderate COVID-19 in adults at high risk of developing severe illness [32,33]; however, the medication can cause liver problems, allergic reactions, high blood pressure, and other serious side effects [33]. The average cost of Paxlovid for 5 days of treatment is USD 1634.38 [34]. These examples highlight the need to develop more antiviral drugs to reduce treatment costs.
Many antiviral compounds have significant toxicity; for example, ribavirin used against RNA viruses and 7-deazaadenosine nucleosides (especially those with ethynyl or small hetaryl groups) showed activity against dengue and SARS-CoV-2 but exhibited significant cytotoxicity [35,36]. In addition, lethal mutagenetic agents such as Favipiravir and Molnupiravir can target RNA viruses but also cause genotoxicity and potential carcinogenic effects [36].
Antiviral drug development has long been neglected compared with vaccine development. During the COVID-19 pandemic, repurposed drugs were used extensively to fill the gap. Thus, effective and safe antiviral drugs to efficiently treat future viral threats need to be developed. Phytocompounds can contribute to the development of new antiviral drugs as they are a huge source of bioactive compounds including antivirals. Phytochemicals with antiviral activities include peptides (both linear and cyclic) and other compounds. Peptides have been found to influence membrane permeability, viral replication, and cellular functions. Therefore, they are attractive targets for antiviral research and development [37]. Specifically, cyclic peptides have attracted attention as antiviral candidates due to their stability, enzymatic hydrolysis resistance, and selectivity to receptors [38,39]. These peptides have demonstrated therapeutic potential in treating cancer and various diseases, including resistant and non-resistant bacterial, viral, and fungal infections [40]. Linear peptides are susceptible to peptidase enzyme degradation, pose off-target effects, and require routine dosing; thus, they are unstable and ineffective [41]. Furthermore, cyclic peptides offer low toxicity [40,42,43], compared with other antiviral compounds that target RNA viruses, along with high effectiveness, but they pose serious risks. The growing interest in both natural and synthetic (designed) cyclic peptide drugs can be attributed to their diverse chemical structures and biological activities, improvements in methods for their synthesis, and increased understanding of protein–protein interactions [44,45].
Overall, there is an urgent need to develop specific and broad-spectrum antiviral drugs that are effective and have low cytotoxicity in humans. This perspective review discusses the role that cyclic peptides could play as a solution to these issues, as they have low toxicity, can be easily modified to match the target, and are stable. This review explores the scientific literature to identify gaps in antiviral drug development, specifically to understand and harness the potential of natural and designed cyclic peptides to target the most recent coronavirus, SARS-CoV-2, and other coronaviruses expected to emerge in the future.
2. Health, Social, and Economic Consequences of the COVID-19 Pandemic
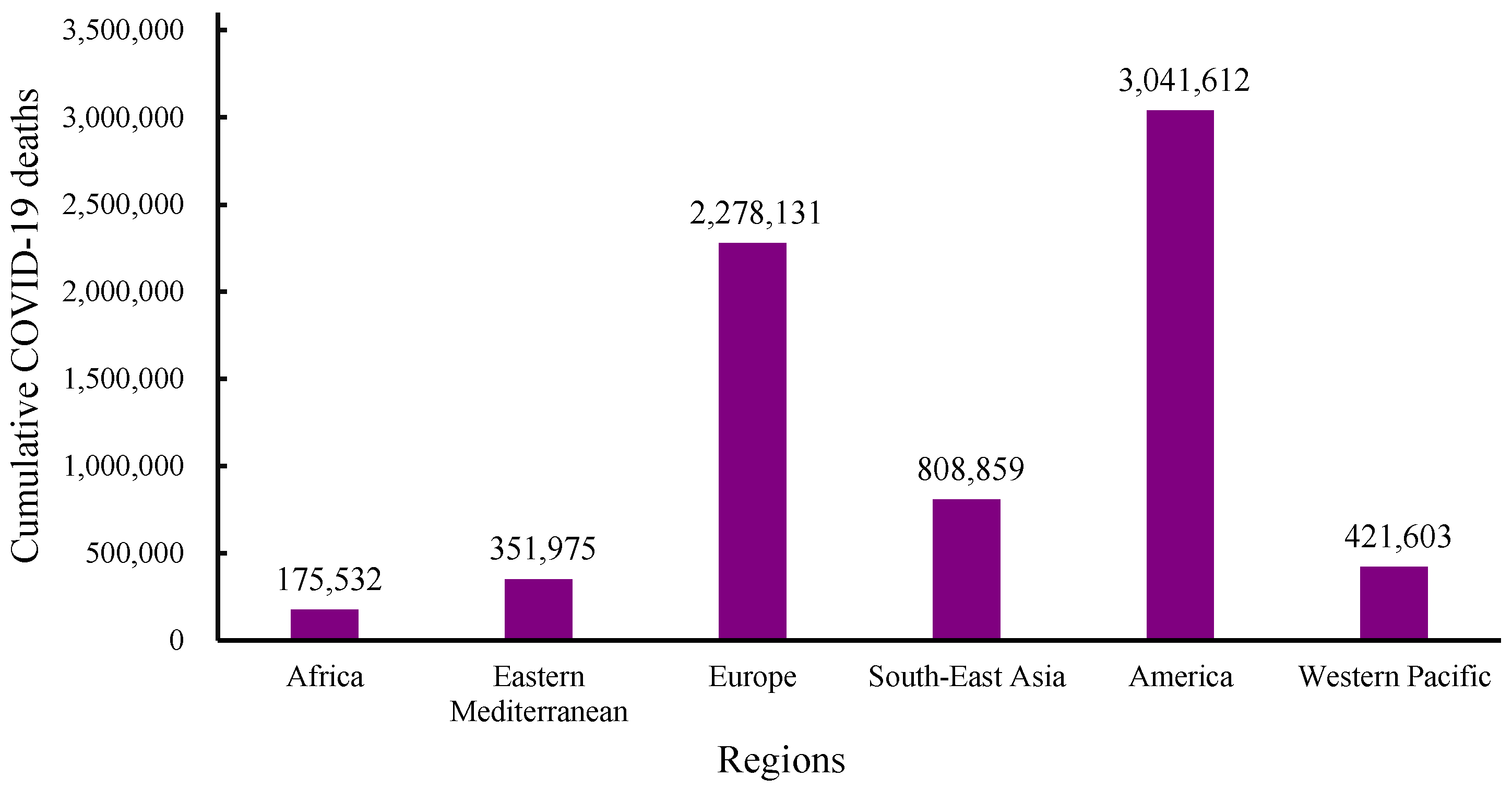
Despite efforts made in viral prevention and control, the emergence of the COVID-19 pandemic brought emotional, health, and economic shocks to the lives of many. The WHO dashboard reported that from December 2019 to 1 December 2024, total COVID-19 infections numbered 776,973,432 globally, with 7,077,725 deaths (Figure 1 and Figure 2) [46]. In December 2024, there were more than 47,230 cases globally and 521 deaths from COVID-19 within the previous 7 days. Europe had the highest cumulative infection rates, with 0.326 from 2019 to 2023 and 0.377 from 2019 to 2024 [46,47,48]. In contrast, Africa had the lowest rates, with 0.011 from 2019 to 2023 and 0.006 from 2019 to 2024 [46,47,49]. Furthermore, high- and middle-income countries experienced higher infection rates compared with low-income countries [46,47].
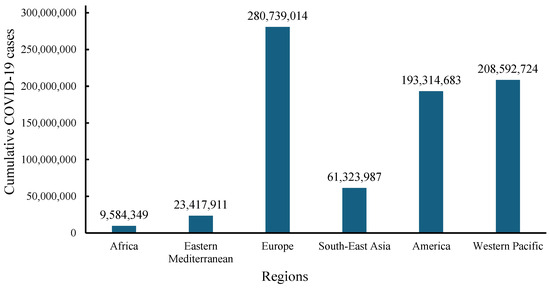
Figure 1.
WHO data for cumulative numbers of COVID-19 cases across continents from December 2019 to 1 December 2024 [46].
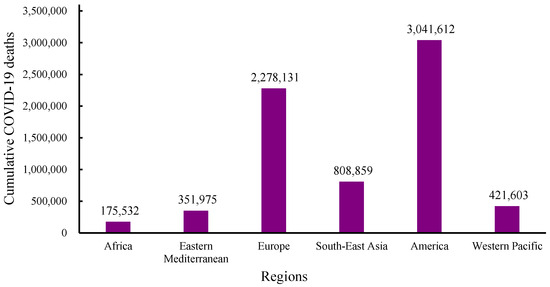
Figure 2.
WHO data for cumulative numbers of COVID-19 deaths across continents from December 2019 to 1 December 2024 [46].
Table 1 shows that vaccination rates decreased with increases in dosage and/or numbers of boosters, and this trend applied across all continents. Some of the reasons may have been psychological hesitancy due to the population’s lack of confidence in vaccines, perceptions of government measures, and perceptions of the information provided, as well as the lack of convincing information [50,51]. Another interesting observation is that while Africa had the lowest vaccination rate, it also had the lowest death rate.

Table 1.
Percentages of the total population vaccinated with at least one dose, complete primary series, and at least one booster, as of 1 December 2024 [46].
Africa’s low death rate while having the lowest vaccination rate might be attributable to several factors but one interesting possibility could be their extensive use of traditional medicine. Africans have a long history of using traditional plants to treat illnesses, even COVID-19, including illnesses known to be caused by viruses [52]. Research has demonstrated that plant extracts can be effective against coronaviruses, suggesting their potential as anti-SARS-CoV-2 agents [53]. Medicinal plants such as Bryophyllum pinnatum, Aframomum melegueta, Garcinia kola, Sphenocentrum jollyanum, Adansonia digitata, Sutherlandia frutescens, Hibiscus sabdariffa, Moringa oleifera, and Nigella sativa have shown combinations of antiviral, immunomodulatory, anti-inflammatory, and anti-COVID-19 symptom activity [52]. Methanol extracts from Adansonia digitata L. (Baobab) inhibited pro-inflammatory iNOS, which is an enzyme that promotes inflammation and causes the degradation of IκBα, a protein that helps in controlling inflammation. This extract also restricted the movement of NF-κB from the cytosol to the nucleus when murine macrophage RAW264.7 cells were induced with LPS (lipopolysaccharide), thereby reducing the inflammatory response in those cells [54]. Natural antiviral compounds from medicinal plants include ribosome-inactivating proteins (RIPs), terpenoids (glycyrrhizin and other specific terpenoids and lignoids), flavonoids (myricetin, scutellarein, quercetin, herbacetin, rhoifolin, pectolinarin, epigallocatechin gallate, gallocatechin gallate), phenolic compounds, etc. [11,55]. Another factor might have been that during the outbreak, several African governments acted swiftly to mitigate the spread of the virus before their healthcare systems were overwhelmed. Lockdowns, border closures, and curfews were implemented early, limiting the transmission of the virus [56]. Rwanda, Uganda, and Senegal enforced strict containment measures, including widespread mask mandates and contact tracing [56]. Also, Africa’s warmer climate may have contributed to the continent’s relatively low COVID-19 death rate. Studies in various African countries have indicated that abundant sunlight, high temperatures, higher relative humidity, and low wind speed may reduce the survivability of respiratory viruses, including SARS-CoV-2 [57].
In the United States, the advent of the COVID-19 pandemic highlighted health disparities that had been observed in earlier influenza outbreaks [58,59]. Shockingly, in the throes of the pandemic, approximately 98 out of every 100,000 African Americans died of COVID-19, exhibiting a mortality rate one-third higher than that of Latinos (65 per 100,000) and more than double the rates for both Whites (47 per 100,000) and Asians (40 per 100,000) [60]. The disproportionate representation of African Americans in confirmed COVID-19 cases and fatalities highlights the pandemic’s role in intensifying existing disparities related to race, socioeconomic status, and healthcare accessibility [60,61]. In addition, preexisting conditions such as diabetes, obesity, cardiovascular diseases, and kidney disease were significant risk factors for COVID-19 [62]. Non-Hispanic black and Hispanic/Latino adults experienced high rates of hospitalization and mortality due to COVID-19, as well as increased risk of non-COVID-19-related mortality through causes such as cardiovascular disease [63]. Throughout the COVID-19 pandemic, the non-Hispanic Black population, particularly those of advanced age, experienced challenges in securing appointments and faced affordability issues associated with obtaining medication [64]. After US Food and Drug Administration (FDA) approval, Remdesivir was the recommended treatment for patients with severe COVID-19 during the pandemic; however, it was in short supply and costly (>USD 3100 per patient) [30,31]. In the face of infection, African Americans exhibited double the hospitalization rate compared with their White counterparts [65]. Other drugs used to treat COVID-19 included monoclonal antibodies such as casirivimab and imdevimab (administered together) or bamlanivimab and etesevimab (administered together) [30], all of which were costly and in short supply. All these factors placed a disproportionate burden on African Americans. Similarly, African Americans were vaccinated at significantly lower rates compared with other groups [66]. As of 30 November 2021, race and ethnicity data had been reported for 70% of individuals who received at least one dose of vaccine. Within this group, 58% identified as White, 19% as Hispanic, 10% as African American, 6% as Asian, and 6% as multiracial, while the remaining individuals were American Indian, Alaskan Native, or Native Hawaiian or other Pacific Islanders [67]. This highlights the need for strategies that resonate with African American communities. The emergence of SARS-CoV-2 and subsequent variants has refocused attention on existing antiviral medications, especially broad-spectrum antivirals that are effective and affordable [68]. Unfortunately, there are currently only 100 approved antiviral drugs that are limited to treating only 10 out of over 220 known viruses that affect humans [69,70]. COVID-19 has underscored the urgent demand for swiftly deployable compounds to treat newly emerging or re-emerging viral diseases, particularly in situations where vaccine development is in progress [70]. Numerous antiviral agents have seen limited use in clinical treatment due to issues of ineffectiveness and resistance [71]. Minority groups, including African Americans, embrace the use of medicinal plants, being drawn to their innate therapeutic qualities and maintaining a steadfast belief in their safety [72]. Natural products characterized by potentially lower toxicity and the ability to target multiple aspects offer a promising source of compounds for effectively combating viral infections in the future [71].
4. Interaction Between SARS-CoV-2 and Host Cells
One of the most important factors required in order to prevent anticipated infection by SARS-CoV-2 is a clear understanding of the molecular mechanism that lies behind its entry into the host cell. The first step is understanding the interaction between the viral infectious component and its binding receptor on the host cell.
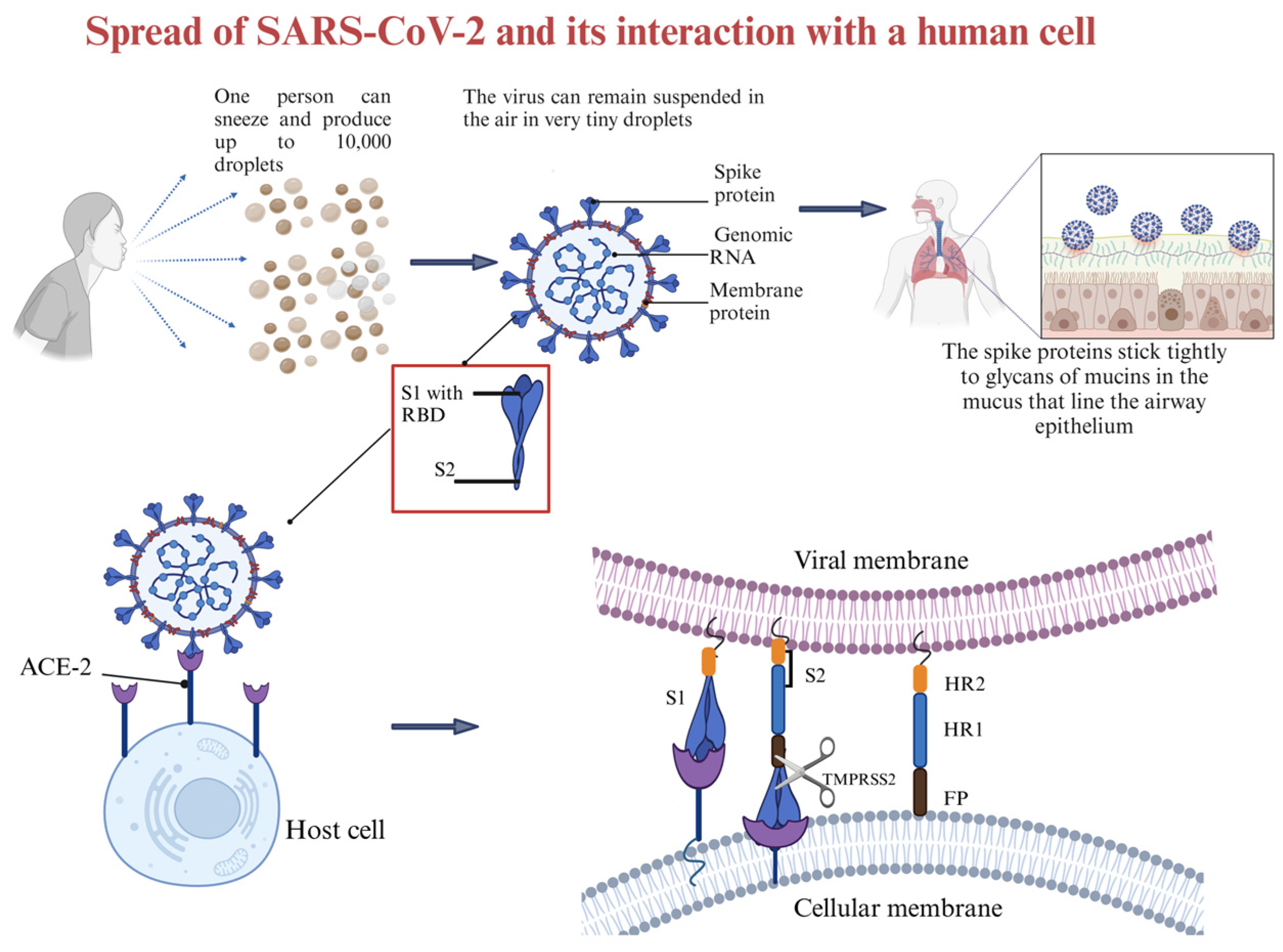
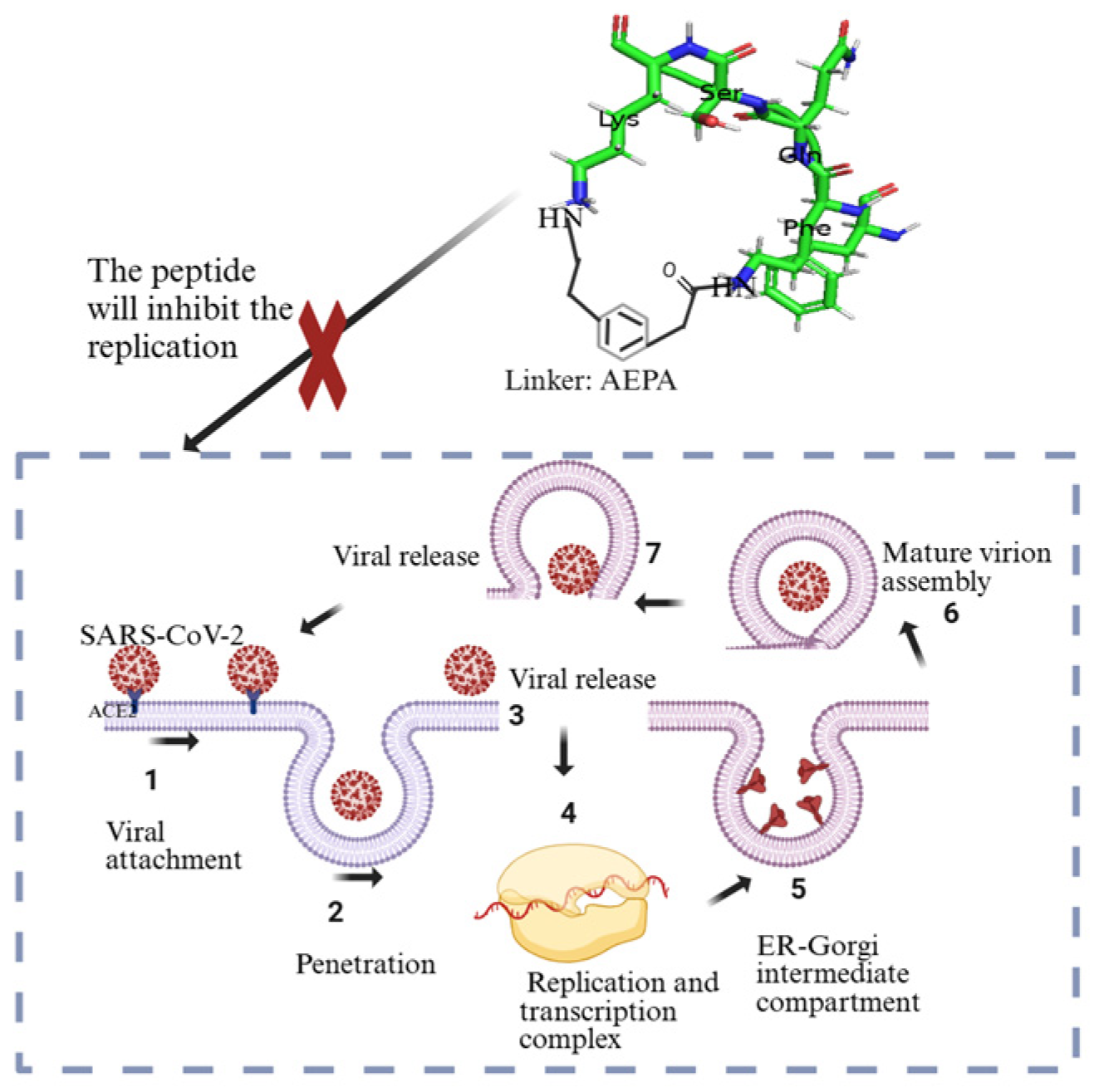
The current understanding is that coronaviruses enter host cells through one of three ways: receptor-mediated plasma membrane fusion, receptor-mediated endocytosis, or antibody-dependent viral entry [86]. In receptor-mediated plasma membrane fusion (Figure 3), SARS-CoV-2 spike protein binds with AC2 on the host cell’s membrane, triggering fusion and allowing the virus to enter and release its genetic material [87]. This was evidenced in the research conducted by Lu and Sun (2020) using assays to study the binding affinity between SARS-CoV-2 and ACE2 [88], and by Engler et al. (2023) [89] that demonstrated how SARS-CoV-2 interacts with ACE2 and EGFR (epidermal growth factor receptor). Using assays like immunoblotting analyses and siRNA-mediated knockdown, the research identified ACE2 as the primary receptor for viral entry. EGFR served as a cofactor, activating the EGFR-MAPK signaling pathway and forming a complex with ACE2, which increased SARS-CoV-2 infection in that study [89]. In receptor-mediated endocytosis, the virus binds to the host cell receptor, and the cell engulfs the virus in a vesicle, where it is internalized and releases its genetic material [90]. Ojha et al. (2024) found that without transmembrane protease serine subtype 2 (TMPRSS2), SARS-CoV-2 infects ACE2-expressing mouse embryonic fibroblasts mainly through dynamin- and actin-dependent endocytosis instead of plasma membrane fusion [91]. In antibody-dependent viral entry, antibodies specific to the virus bind to the spike protein, enhancing viral entry by facilitating fusion or promoting endocytosis [92,93]. Research conducted by Liu et al. (2021) [94] evaluated a range of anti-spike monoclonal antibodies derived from COVID-19 patients. The researchers found that certain antibodies targeting the N-terminal domain (NTD) of the spike protein promoted the open conformation of the receptor-binding domain (RBD), thereby increasing the spike protein’s binding affinity to ACE2 and enhancing SARS-CoV-2 infectivity [94].
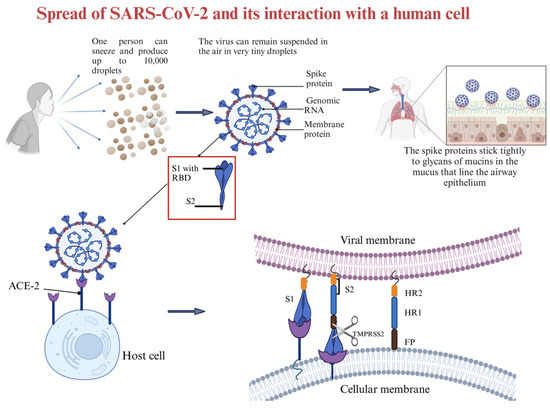
Figure 3.
Diagram of how SARS-CoV-2 is spread and how it interacts with the host cell. Created in BioRender. Collier, W. (2025) https://BioRender.com/t31u631 (accessed 21 March 2025).
The existence of proteins on the surface of the host cell plays an important role in maintaining the attachment of viruses to the host cell for both endocytosis and fusion [86]. The knowledge gained by studying the interaction between SARS-CoV-2 and its main receptor led to an understanding of how to block the virus from entering the host cell, which may support the development of new and promising strategies for clinical treatment and prevention [95].
The infectious component of SARS-CoV-2 is the spike protein, which consists of S1 and S2 subunits. S1 contains the RBD responsible for binding to the host cell receptor. The major cell entry receptor for SARS-CoV-2 is ACE2 [96]. ACE2 receptor expression is most prominent in the small intestine, testes, kidney, heart muscle, colon, and thyroid gland [97]. The lungs have low mRNA and protein expression levels of ACE, and blood cells do not express any [97]. S2 is made of a fusion peptide (FP), heptad repeat 1 (HR1), heptad repeat 2 (HR2), and a transmembrane domain (TM), with cytoplasmic domain fusion (CP), which is responsible for mediating viral fusion and entry [98]. The S2 subunit is highly conserved, and it is 99% homologous to human SARS-like coronavirus and the bat coronaviruses SL-CoV ZXC21 and ZC45 [98]. During viral infection, the S1 subunit is degraded and S2 is inserted into the host cell, thereby exposing three pairs of HR1 and HR2 domains that form a six-helix bundle structure that brings the cellular lipid bilayer and the viral lipid bilayer into fusion (Figure 3). TMPRSS2 is an enzyme that cleaves the spike protein into S1 and S2 subunits (Figure 3). In the respiratory tract, this enzyme is located on the cell surface, especially in the airway epithelium. After the degradation of S1 and formation of the six-helix bundle structure, the fusion process is in the pre-hairpin stage (intermediate conformation) [98]. Thus, a peptide might be designed to target HR1 and prevent conformation of the S2 subunit [98].
The mutations listed in Table 2 presented challenges relating to the discovery, use, and acceptability of vaccines. In this context, antiviral drugs, especially broad-spectrum antivirals, could have been effective for saving lives during the pandemic. However, no such broad-spectrum antiviral drug has yet been approved, which poses a big challenge in treating new and re-emerging viruses, particularly in the current context where approval is lacking for broad-spectrum antiviral drugs. Furthermore, the advent of highly pathogenic coronaviruses like SARS-CoV and MERS-CoV, along with the potential for future outbreaks caused by unknown RNA viruses, highlights the need for broad-spectrum antiviral drugs [99,100]. Traditional antiviral development has focused on targeting specific viruses, leaving us vulnerable to new or mutated strains. However, broad-spectrum antivirals offer a proactive approach to pandemic preparedness by targeting conserved mechanisms shared by various viral families [100,101]. This approach is particularly important for RNA viruses like SARS-CoV-2, which have a high mutation rate and pose a significant threat due to their rapid replication and transmissibility [101]. For example, favipiravir, an RNA-dependent RNA polymerase inhibitor, has demonstrated efficacy against a range of RNA viruses including Ebola, Lassa fever, and influenza [102,103,104]. It is important to note that both natural and synthetic cyclic peptides show potential as components of broad-spectrum antivirals [100]. These peptides can disrupt the viral envelope, inhibiting viral entry and replication. Investing in research and the development of broad-spectrum antivirals, including cyclic peptides, is essential in order to establish a robust defense against future viral threats and ensure global health security [101].

Table 2.
SARS-CoV-2 types and variants of concern, their country of origin, and key spike protein mutations in relation to the Wuhan strain.
6. Future Directions
Future research on the use of natural and designed cyclic peptides as antiviral drugs holds significant promise for combating future coronavirus outbreaks. These peptides, which can be obtained from natural sources or designed through molecular modeling, offer several advantages such as high specificity, stability, and the ability to target viral proteins with minimal toxicity to human cells. Additionally, the versatility of cyclic peptides permits the design of broad-spectrum antivirals that can target multiple variants of coronaviruses, addressing the challenges of viral mutation and resistance. Moving forward, a combination of high-throughput screening, computational modeling, and structural biology will be essential to optimize the efficacy and delivery of these peptides. Collaborative efforts between virologists, pharmacologists, and chemists will play a critical role in translating these promising antiviral candidates into safe and effective treatments for future coronavirus pandemics.
7. Conclusions
RNA viruses, including SARS-CoV-2 and others, have caused social, economic, and biological challenges. The pandemic caused widespread illness and increased mortality, created a major challenge for researchers, and raised concerns globally about potential future pandemics. Africa had the lowest number of cases, the fewest deaths, and the lowest percentage of vaccination coverage, while Europe had the highest cases, America had the most deaths, and the Western Pacific had the highest vaccination coverage. RNA viruses undergo rapid mutations due to a lack of proofreading mechanisms in their nucleotides, making it very challenging to provide sustainable treatments. Various approved antiviral drugs are effective; however, some of them cause high cytotoxicity. Additionally, there is a gap in the approval of broad-spectrum antivirals that can target all RNA viruses, which would help control future pandemics. Cyclic peptides from natural sources such as plants, bacteria, and fungi, as well as those designed through molecular modeling techniques, are potential candidates for sustainable and broad-spectrum antiviral drugs as they are stable, easily modified, and have low toxicity and resistance. These cyclic peptides can prevent interaction between the ACE2 receptor and the spike protein of SARS-CoV-2 by binding with either the receptor or the spike protein. It is crucial that continual antiviral drug development programs are initiated and funded on par with vaccine development, to allow researchers to keep exploring the potential of antiviral cyclic peptides. The development of antiviral cyclic peptides could be a major contribution to a long-term solution for tackling new and re-emerging viral outbreaks and pandemics around the world.
Author Contributions
Conceptualization, H.U., W.E.C., T.O.N. and J.M.J.; writing—original draft, H.U., W.E.C., T.O.N., J.F.R.I., C.H., D.T. and U.N.R.; writing—review and editing, H.U., W.E.C., T.O.N., J.M.J., D.G.M., C.G.D., G.G.K., E.F.A., J.F.R.I., C.H., D.T., U.N.R., Y.A.D. and K.L.R.; visualization, H.U. and W.E.C.; supervision, W.E.C. and T.O.N.; funding acquisition, W.E.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Tuskegee University Center for Biomedical Research/Research Centers in Minority Institutions (TU CBR/RCMI), grant number 5454MD007585.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
We acknowledge the support from Mfon Uboh (Prairie View A&M University), as well as the Tuskegee University Center for Biomedical Research (TU CBR)/Research Centers in Minority Institutions (RCMI), Integrative Biosciences PhD Program, Department of Chemistry, College of Agriculture, Environment and Nutrition Sciences, George Washington Carver Agricultural Experiment Station, Tuskegee University.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global Trends in Emerging Infectious Diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Hernandez, R.; Jácome, R.; Vidal, Y.L.; de León, S.P. Are RNA Viruses Candidate Agents for the next Global Pandemic? A Review. ILAR J. 2017, 58, 343–358. [Google Scholar] [CrossRef]
- Baize, S.; Pannetier, D.; Oestereich, L.; Rieger, T.; Koivogui, L.; Magassouba, N.; Soropogui, B.; Sow, M.S.; Keïta, S.; De Clerck, H.; et al. Emergence of Zaire Ebola Virus Disease in Guinea. N. Engl. J. Med. 2014, 371, 1418–1425. [Google Scholar] [CrossRef]
- Bray, M. Highly Pathogenic RNA Viral Infections: Challenges for Antiviral Research. Antivir. Res. 2008, 78, 1–8. [Google Scholar]
- Holmes, E.C. Evolution and Emergence of RNA Viruses; Oxford University Press Inc.: New York, NY, USA, 2009; ISBN 978-0-19-921112-8. [Google Scholar]
- Markiewicz, L.; Drazkowska, K.; Sikorski, P.J. Tricks and Threats of RNA Viruses–towards Understanding the Fate of Viral RNA. RNA Biol. 2021, 18, 669–687. [Google Scholar]
- Figlerowicz, M.; Alejska, M.; Kurzyńska-Kokorniak, A.; Figlerowicz, M. Genetic Variability: The Key Problem in the Prevention and Therapy of RNA-Based Virus Infections. Med. Res. Rev. 2003, 23, 488–518. [Google Scholar] [CrossRef] [PubMed]
- Villa, T.G.; Abril, A.G.; Sánchez, S.; de Miguel, T.; Sánchez-Pérez, A. Animal and Human RNA Viruses: Genetic Variability and Ability to Overcome Vaccines. Arch. Microbiol. 2021, 203, 443–464. [Google Scholar] [PubMed]
- World Health Organization (WHO). WHO to Identify Pathogens That Could Cause Future Outbreaks and Pandemics. Available online: https://www.who.int/news/item/21-11-2022-who-to-identify-pathogens-that-could-cause-future-outbreaks-and-pandemics (accessed on 6 December 2024).
- Grubaugh, N.D.; Ladner, J.T.; Lemey, P.; Pybus, O.G.; Rambaut, A.; Holmes, E.C.; Andersen, K.G. Tracking Virus Outbreaks in the Twenty-First Century. Nat. Microbiol. 2019, 4, 10–19. [Google Scholar] [CrossRef]
- Ali, S.I.; Sheikh, W.M.; Rather, M.A.; Venkatesalu, V.; Bashir, S.M.; Nabi, S.U. Medicinal Plants: Treasure for Antiviral Drug Discovery. Phytother. Res. 2021, 35, 3447–3483. [Google Scholar]
- McCloskey, B.; Heymann, D.L. SARS to Novel Coronavirus-Old Lessons and New Lessons. Epidemiol. Infect. 2020, 148, e22. [Google Scholar] [CrossRef]
- Gupta, D.; AK, S.; Bansal, A.; Gupta, N.; Patki, V.; Sood, A.K.; Sachdev, A.; Parekh, B.J. Use of personal protective during COVID-19 in resource limited-the barest minimum. Indian J. Pract. Pediatr. 2020, 22, 195. [Google Scholar]
- Choi, H.; Chatterjee, P.; Lichtfouse, E.; Martel, J.A.; Hwang, M.; Jinadatha, C.; Sharma, V.K. Classical and Alternative Disinfection Strategies to Control the COVID-19 Virus in Healthcare Facilities: A Review. Environ. Chem. Lett. 2021, 19, 1945–1951. [Google Scholar] [PubMed]
- Dhama, K.; Patel, S.K.; Kumar, R.; Masand, R.; Rana, J.; Yatoo, M.I.; Tiwari, R.; Sharun, K.; Mohapatra, R.K.; Natesan, S.; et al. The Role of Disinfectants and Sanitizers during COVID-19 Pandemic: Advantages and Deleterious Effects on Humans and the Environment. Environ. Sci. Pollut. Res. 2021, 28, 34211–34228. [Google Scholar] [CrossRef]
- de Araújo, L.A.; Veloso, C.F.; de Campos Souza, M.; de Azevedo, J.M.C.; Tarro, G. The Potential Impact of the COVID-19 Pandemic on Child Growth and Development: A Systematic Review. J. Pediatr. 2021, 97, 369–377. [Google Scholar]
- Usher, K.; Jackson, D.; Durkin, J.; Gyamfi, N.; Bhullar, N. Pandemic-Related Behaviours and Psychological Outcomes; A Rapid Literature Review to Explain COVID-19 Behaviours. Int. J. Ment. Health Nurs. 2020, 29, 1018–1034. [Google Scholar] [CrossRef]
- Viner, R.M.; Russell, S.J.; Croker, H.; Packer, J.; Ward, J.; Stansfield, C.; Mytton, O.; Bonell, C.; Booy, R. School Closure and Management Practices during Coronavirus Outbreaks Including COVID-19: A Rapid Systematic Review. Lancet Child Adolesc. Health 2020, 4, 397–404. [Google Scholar]
- Altmann, D.M.; Boyton, R.J. COVID-19 Vaccination: The Road Ahead. Science 2022, 375, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The CanSino Biologics Ad5-NCoV-S [Recombinant] COVID-19 Vaccine: What You Need to Know. Available online: https://www.who.int/news-room/feature-stories/detail/the--cansino-biologics-ad5-ncov-s--recombinant---covid-19-vaccine--what-you-need-to-know (accessed on 2 February 2025).
- Hotez, P.J.; Bottazzi, M.E. Whole Inactivated Virus and Protein-Based COVID-19 Vaccines. Annu. Rev. Med. 2022, 73, 55–64. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. COVID-19 Vaccines with WHO Emergency Use Listing. Available online: https://extranet.who.int/prequal/vaccines/covid-19-vaccines-who-emergency-use-listing (accessed on 3 February 2025).
- International Vaccine Institute. SK Bioscience’s COVID-19 Vaccine Makes WHO’s Emergency Use Listing. Available online: https://www.ivi.int/sk-biosciences-covid-19-vaccine-makes-whos-emergency-use-listing/ (accessed on 3 February 2025).
- Mann, R.; Sekhon, S.; Sekhon, S. Drug-Induced Liver Injury After COVID-19 Vaccine. Cureus 2021, 13, e16491. [Google Scholar] [CrossRef]
- Gøtzsche, P.C.; Demasi, M. Serious Harms of the COVID-19 Vaccines: A Systematic Review. MedRxiv 2022. [Google Scholar] [CrossRef]
- Kang, C.R.; Choe, Y.J.; Yoon, S.J. COVID-19 Vaccine Injury Compensation Program: Lessons Learned from a Review of 10 Implementing Countries. J. Korean Med. Sci. 2024, 39, e121. [Google Scholar] [PubMed]
- Sridhar, S.; Luedtke, A.; Langevin, E.; Zhu, M.; Bonaparte, M.; Machabert, T.; Savarino, S.; Zambrano, B.; Moureau, A.; Khromava, A.; et al. Effect of Dengue Serostatus on Dengue Vaccine Safety and Efficacy. N. Engl. J. Med. 2018, 379, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Vannice, K.S.; Wilder-Smith, A.; Barrett, A.D.T.; Carrijo, K.; Cavaleri, M.; de Silva, A.; Durbin, A.P.; Endy, T.; Harris, E.; Innis, B.L.; et al. Clinical Development and Regulatory Points for Consideration for Second-Generation Live Attenuated Dengue Vaccines. Vaccine 2018, 36, 3411–3417. [Google Scholar] [CrossRef]
- Springer Nature Limited. Funders, now is the time to invest big in COVID drugs. Nature 2021, 592, 326. [Google Scholar] [CrossRef] [PubMed]
- Whittington, M.D.; Pearson, S.D.; Rind, D.M.; Campbell, J.D. The Cost-Effectiveness of Remdesivir for Hospitalized Patients With COVID-19. Value Health 2022, 25, 744–750. [Google Scholar] [CrossRef]
- Carethers, J.M. Rectifying COVID-19 Disparities with Treatment and Vaccination. JCI Insight 2021, 6, e147800. [Google Scholar] [CrossRef]
- U.S Food & Drug Administration. FDA Approves First Oral Antiviral for Treatment of COVID-19 in Adults. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-first-oral-antiviral-treatment-covid-19-adults (accessed on 27 January 2025).
- Pfizer If It’s COVID, PAXLOVID. Available online: https://www.paxlovid.com/ (accessed on 27 January 2025).
- Aungst, C. How to Find the COVID-19 Pills Paxlovid and Lagevrio. Available online: https://www.goodrx.com/conditions/covid-19/covid-pill-cost-availability (accessed on 27 January 2025).
- Milisavljevic, N.; Konkolová, E.; Kozák, J.; Hodek, J.; Veselovská, L.; Sýkorová, V.; Čížek, K.; Pohl, R.; Eyer, L.; Svoboda, P.; et al. Antiviral Activity of 7-Substituted 7-Deazapurine Ribonucleosides, Monophosphate Prodrugs, and Triphoshates against Emerging RNA Viruses. ACS Infect. Dis. 2021, 7, 471–478. [Google Scholar] [CrossRef]
- Hassine, I.H.; Ben M’Hadheb, M.; Menéndez-Arias, L. Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity. Viruses 2022, 14, 841. [Google Scholar] [CrossRef]
- Musarra-Pizzo, M.; Pennisi, R.; Ben-Amor, I.; Mandalari, G.; Sciortino, M.T. Antiviral Activity Exerted by Natural Products against Human Viruses. Viruses 2021, 13, 828. [Google Scholar] [CrossRef]
- Mammari, N.; Krier, Y.; Albert, Q.; Devocelle, M.; Varbanov, M. Plant-Derived Antimicrobial Peptides as Potential Antiviral Agents in Systemic Viral Infections. Pharmaceuticals 2021, 14, 774. [Google Scholar] [CrossRef]
- Heydari, H.; Golmohammadi, R.; Mirnejad, R.; Tebyanian, H.; Fasihi-Ramandi, M.; Moghaddam, M.M. Antiviral Peptides against Coronaviridae Family: A Review. Peptides 2021, 139, 170526. [Google Scholar] [PubMed]
- Bera, S.; Mondal, D. Natural Cyclic Peptides as Clinical and Future Therapeutics. Curr. Org. Chem. 2019, 23, 38–75. [Google Scholar] [CrossRef]
- Chia, L.Y.; Kumar, P.V.; Maki, M.A.A.; Ravichandran, G.; Thilagar, S. A Review: The Antiviral Activity of Cyclic Peptides. Int. J. Pept. Res. Ther. 2023, 29, 7. [Google Scholar]
- Ke, J.; Zhang, J.; Li, J.; Liu, J.; Guan, S. Design of Cyclic Peptide-Based Nanospheres and the Delivery of SiRNA. Int. J. Mol. Sci. 2022, 23, 12071. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yang, L.; Cao, S.; Gao, Z.; Yang, B.; Zhang, G.; Zhu, R.; Wu, D. CyclicPepedia: A Knowledge Base of Natural and Synthetic Cyclic Peptides. Brief. Bioinform. 2024, 25, bbae190. [Google Scholar] [CrossRef]
- El Sayed, K.A. Natural Products as Antiviral Agents. Stud. Nat. Prod. Chem. 2000, 24, 473–572. [Google Scholar]
- Cardote, T.A.F.; Ciulli, A. Cyclic and Macrocyclic Peptides as Chemical Tools To Recognise Protein Surfaces and Probe Protein-Protein Interactions. ChemMedChem 2015, 11, 787–794. [Google Scholar] [CrossRef]
- World Health Organization (WHO). WHO COVID-19 Dashboard. Available online: https://data.who.int/dashboards/covid19/cases?n=c (accessed on 11 December 2024).
- Du, J.; Lang, H.M.; Ma, Y.; Chen, A.W.; Qin, Y.Y.; Zhang, X.P.; Huang, C.Q. Global Trends in COVID-19 Incidence and Case Fatality Rates (2019–2023): A Retrospective Analysis. Front. Public Health 2024, 12, 1355097. [Google Scholar] [CrossRef]
- Wordometer Europe Population (LIVE). Available online: https://www.worldometers.info/world-population/europe-population/ (accessed on 28 January 2025).
- Worldometer Africa Population (LIVE). Available online: https://www.worldometers.info/world-population/africa-population/ (accessed on 28 January 2025).
- Soares, P.; Rocha, J.V.; Moniz, M.; Gama, A.; Laires, P.A.; Pedro, A.R.; Dias, S.; Leite, A.; Nunes, C. Factors Associated with COVID-19 Vaccine Hesitancy. Vaccines 2021, 9, 300. [Google Scholar] [CrossRef]
- Hornsey, M.J. Reasons Why People May Refuse COVID-19 Vaccination (and What Can Be Done about It). World Psychiatry 2022, 21, 217–218. [Google Scholar] [CrossRef]
- Akindele, A.J.; Sowemimo, A.; Agunbiade, F.O.; Sofidiya, M.O.; Awodele, O.; Ade-Ademilua, O.; Orabueze, I.; Ishola, I.O.; Ayolabi, C.I.; Salu, O.B.; et al. Bioprospecting for Anti-COVID-19 Interventions From African Medicinal Plants: A Review. Nat. Prod. Commun. 2022, 17, 1934578X221096968. [Google Scholar] [CrossRef]
- Vellingiri, B.; Jayaramayya, K.; Iyer, M.; Narayanasamy, A.; Govindasamy, V.; Giridharan, B.; Ganesan, S.; Venugopal, A.; Venkatesan, D.; Ganesan, H.; et al. COVID-19: A Promising Cure for the Global Panic. Sci. Total. Environ. 2020, 725, 138277. [Google Scholar] [PubMed]
- Ayele, Y.; Kim, J.-A.; Park, E.; Kim, Y.-J.; Retta, N.; Dessie, G.; Rhee, S.-K.; Koh, K.; Nam, K.-W.; Kim, H.S. A Methanol Extract of Adansonia Digitata L. Leaves Inhibits Pro-Inflammatory INOS Possibly via the Inhibition of NF-ΚB Activation. Biomol. Ther. 2013, 21, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Khan, T.; Khan, M.A.; Mashwani, Z.u.R.; Ullah, N.; Nadhman, A. Therapeutic Potential of Medicinal Plants against COVID-19: The Role of Antiviral Medicinal Metabolites. Biocatal. Agric. Biotechnol. 2021, 31, 101890. [Google Scholar]
- Haider, N.; Osman, A.Y.; Gadzekpo, A.; Akipede, G.O.; Asogun, D.; Ansumana, R.; Lessells, R.J.; Khan, P.; Hamid, M.M.A.; Yeboah-Manu, D.; et al. Lockdown Measures in Response to COVID-19 in Nine Sub-Saharan African Countries. BMJ Glob. Health 2020, 5, e003319. [Google Scholar] [CrossRef]
- Mwiinde, A.M.; Siankwilimba, E.; Sakala, M.; Banda, F.; Michelo, C. Climatic and Environmental Factors Influencing COVID-19 Transmission—An African Perspective. Trop. Med. Infect. Dis. 2022, 7, 433. [Google Scholar] [CrossRef]
- Roberts, J.D.; Tehrani, S.O. Environments, Behaviors, and Inequalities: Reflecting on the Impacts of the Influenza and Coronavirus Pandemics in the United States. Int. J. Environ. Res. Public Health 2020, 17, 4484. [Google Scholar] [CrossRef]
- Quinn, S.C.; Kumar, S.; Freimuth, V.S.; Musa, D.; Casteneda-Angarita, N.; Kidwell, K. Racial Disparities in Exposure, Susceptibility, and Access to Health Care in the US H1N1 Influenza Pandemic. Am. J. Public Health 2011, 101, 285–293. [Google Scholar] [CrossRef]
- Reyes, M.V. The Disproportional Impact of COVID-19 on African Americans. Health Hum. Rights 2020, 22, 299–307. [Google Scholar]
- Snowden, L.R.; Graaf, G. COVID-19, Social Determinants Past, Present, and Future, and African Americans’ Health. J. Racial Ethn. Health Disparities 2021, 8, 12–20. [Google Scholar] [CrossRef]
- Carethers, J.M. Insights into Disparities Observed with COVID-19. J. Intern. Med. 2021, 289, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Bunsawat, K.; Grosicki, G.J.; Jeong, S.; Robinson, A.T. Racial and Ethnic Disparities in Cardiometabolic Disease and COVID-19 Outcomes in White, Black/African American, and Latinx Populations: Physiological Underpinnings. Prog. Cardiovasc. Dis. 2022, 71, 11–19. [Google Scholar] [CrossRef]
- Clay, S.L.; Woodson, M.J.; Mazurek, K.; Antonio, B. Racial Disparities and COVID-19: Exploring the Relationship Between Race/Ethnicity, Personal Factors, Health Access/Affordability, and Conditions Associated with an Increased Severity of COVID-19. Race Soc. Probl. 2021, 13, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Price-Haywood, E.G.; Burton, J.; Fort, D.; Seoane, L. Hospitalization and Mortality among Black Patients and White Patients with COVID-19. N. Engl. J. Med. 2020, 382, 2534–2543. [Google Scholar] [CrossRef]
- Sharma, M.; Batra, K.; Batra, R. A Theory-Based Analysis of COVID-19 Vaccine Hesitancy among African Americans in the United States: A Recent Evidence. Healthcare 2021, 9, 1273. [Google Scholar] [CrossRef] [PubMed]
- Majee, W.; Anakwe, A.; Onyeaka, K.; Harvey, I.S. The Past Is so Present: Understanding COVID-19 Vaccine Hesitancy Among African American Adults Using Qualitative Data. J. Racial Ethn. Health Disparities 2023, 10, 462–474. [Google Scholar] [CrossRef]
- Leneva, I.; Kartashova, N.; Poromov, A.; Gracheva, A.; Korchevaya, E.; Glubokova, E.; Borisova, O.; Shtro, A.; Loginova, S.; Shchukina, V.; et al. Antiviral Activity of Umifenovir in Vitro against a Broad Spectrum of Coronaviruses, Including the Novel SARS-CoV-2 Virus. Viruses 2021, 13, 1665. [Google Scholar] [CrossRef]
- Du, R.; Achi, J.G.; Cui, Q.; Rong, L. Paving New Roads toward the Advancement of Broad-Spectrum Antiviral Agents. J. Med. Virol. 2024, 96, e29369. [Google Scholar] [CrossRef]
- Adamson, C.S.; Chibale, K.; Goss, R.J.M.; Jaspars, M.; Newman, D.J.; Dorrington, R.A. Antiviral Drug Discovery: Preparing for the next Pandemic. Chem. Soc. Rev. 2021, 50, 3647–3655. [Google Scholar] [CrossRef]
- Tian, W.J.; Wang, X.J. Broad-Spectrum Antivirals Derived from Natural Products. Viruses 2023, 15, 1100. [Google Scholar] [CrossRef]
- Gardiner, P.; Whelan, J.; White, L.F.; Filippelli, A.C.; Bharmal, N.; Kaptchuk, T.J. A Systematic Review of the Prevalence of Herb Usage Among Racial/Ethnic Minorities in the United States. J. Immigr. Minor. Health 2013, 15, 817–828. [Google Scholar] [PubMed]
- Burrell, C.J.; Howard, C.R.; Murphy, F.A. Coronaviruses. In Fenner and White’s Medical Virology; Elsevier: Amsterdam, The Netherlands, 2017; pp. 437–446. [Google Scholar]
- Kahn, J.S.; McIntosh, K. History and Recent Advances in Coronavirus Discovery. Pediatr. Infect. Dis. J. 2005, 24, S223–S227. [Google Scholar] [CrossRef] [PubMed]
- Rabaan, A.A.; Al-Ahmed, S.H.; Haque, S.; Sah, R.; Tiwari, R.; Malik, Y.S.; Dhama, K.; Yatoo, M.I.; Bonilla-Aldana, D.K.; Rodriguez-Morales, A.J. SARS-CoV-2, SARS-CoV, and MERS-CoV: A Comparative Overview. Infez. Med. 2020, 28, 174–184. [Google Scholar]
- Shahrajabian, M.H.; Sun, W.; Cheng, Q. Product of Natural Evolution (SARS, MERS, and SARS-CoV-2); Deadly Diseases, from SARS to SARS-CoV-2. Hum. Vaccin. Immunother. 2021, 17, 62–83. [Google Scholar] [PubMed]
- Zhou, Y.; Yang, Y.; Huang, J.; Jiang, S.; Du, L. Advances in MERS-CoV Vaccines and Therapeutics Based on the Receptor-Binding Domain. Viruses 2019, 11, 60. [Google Scholar] [CrossRef]
- Zeidler, A.; Karpinski, T.M. SARS-CoV, MERS-COV, SARS-CoV-2 Comparison of Three Emerging Coronaviruses. Jundishapur J. Microbiol. 2020, 13, e103744. [Google Scholar]
- Johns Hopkins BLOOMBERG SCHOOL of PUBLIC HEALTH and Center for Health Security. Coronaviruses: SARS, MERS, and 2019-NCoV. Nat. Med. 2020, 26, 450–452. [Google Scholar]
- Meo, S.A.; Alhowikan, A.M.; Al-Khlaiwi, T.; Meo, I.M.; Halepoto, D.M.; Iqbal, M.; Usmani, A.M.; Hajjar, W.; Ahmed, N. Novel Coronavirus 2019-NCoV: Prevalence, Biological and Clinical Characteristics Comparison with SARS-CoV and MERS-CoV. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 2012–2019. [Google Scholar]
- World Health Organization. WHO-Convened Global Study of Origins of SARS-CoV-2: China Part; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Knight, D. COVID-19 Pandemic Origins: Bioweapons and the History of Laboratory Leaks. South. Med. J. 2021, 114, 465–467. [Google Scholar] [CrossRef]
- Baker, N. The Lab-Leak Hypothesis. N. Y. Mag. 2021, 4, 1–32. [Google Scholar]
- Kormann, C. The Mysterious Case of the COVID-19 Lab-Leak Theory—Did the Virus Spring from Nature or from Human Error? New Yorker 2021, 12, 1–12. [Google Scholar]
- Center for Disease Control and Prevention Symptoms of COVID-19. Available online: https://www.cdc.gov/covid/signs-symptoms/index.html (accessed on 20 January 2025).
- Zhang, Q.; Xiang, R.; Huo, S.; Zhou, Y.; Jiang, S.; Wang, Q.; Yu, F. Molecular Mechanism of Interaction between SARS-CoV-2 and Host Cells and Interventional Therapy. Signal Transduct. Target. Ther. 2021, 6, 233. [Google Scholar] [PubMed]
- Shang, J.; Wan, Y.; Luo, C.; Ye, G.; Geng, Q.; Auerbach, A.; Li, F. Cell Entry Mechanisms of SARS-CoV-2. Proc. Natl. Acad. Sci. USA 2020, 117, 11727–11734. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Sun, P.D. High Affinity Binding of SARS-CoV-2 Spike Protein Enhances ACE2 Carboxypeptidase Activity. J. Biol. Chem. 2020, 295, 18579–18588. [Google Scholar] [CrossRef]
- Engler, M.; Albers, D.; Von Maltitz, P.; Groß, R.; Münch, J.; Cirstea, I.C. ACE2-EGFR-MAPK Signaling Contributes to SARS-CoV-2 Infection. Life Sci. Alliance 2023, 9, e202201880. [Google Scholar] [CrossRef]
- Wang, K.; Chen, W.; Zhang, Z.; Deng, Y.; Lian, J.Q.; Du, P.; Wei, D.; Zhang, Y.; Sun, X.X.; Gong, L.; et al. CD147-Spike Protein Is a Novel Route for SARS-CoV-2 Infection to Host Cells. Signal Transduct. Target. Ther. 2020, 5, 283. [Google Scholar] [CrossRef]
- Ojha, R.; Jiang, A.; Mäntylä, E.; Quirin, T.; Modhira, N.; Witte, R.; Gaudin, A.; De Zanetti, L.; Gormal, R.S.; Vihinen-Ranta, M.; et al. Dynamin Independent Endocytosis Is an Alternative Cell Entry Mechanism for Multiple Animal Viruses. PLoS Pathog. 2024, 20, e1012690. [Google Scholar] [CrossRef]
- Eroshenko, N.; Gill, T.; Keaveney, M.K.; Church, G.M.; Trevejo, J.M.; Rajaniemi, H. Implications of Antibody-Dependent Enhancement of Infection for SARS-CoV-2 Countermeasures. Nat. Biotechnol. 2020, 38, 789–791. [Google Scholar] [CrossRef]
- Iwasaki, A.; Yang, Y. The Potential Danger of Suboptimal Antibody Responses in COVID-19. Nat. Rev. Immunol. 2020, 20, 339–341. [Google Scholar]
- Liu, Y.; Soh, W.T.; Kishikawa, J.-i.; Hirose, M.; Nakayama, E.E.; Li, S.; Sasai, M.; Suzuki, T.; Tada, A.; Arakawa, A.; et al. An Infectivity-Enhancing Site on the SARS-CoV-2 Spike Protein Targeted by Antibodies. Cell 2021, 184, 3452–3466.e18. [Google Scholar] [CrossRef]
- Sivaraman, H.; Er, S.Y.; Choong, Y.K.; Gavor, E.; Sivaraman, J. Structural Basis of SARS-CoV-2-and SARS-CoV-Receptor Binding and Small-Molecule Blockers as Potential Therapeutics. Annu. Rev. Pharmacol. Toxicol. 2021, 61, 465–493. [Google Scholar] [CrossRef] [PubMed]
- Scialo, F.; Daniele, A.; Amato, F.; Pastore, L.; Matera, M.G.; Cazzola, M.; Castaldo, G.; Bianco, A. ACE2: The Major Cell Entry Receptor for SARS-CoV-2. Lung 2020, 198, 867–877. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Y.; Luo, W.; Huang, L.; Xiao, J.; Li, F.; Qin, S.; Song, X.; Wu, Y.; Zeng, Q.; et al. A Comprehensive Investigation of the Mrna and Protein Level of Ace2, the Putative Receptor of SARS-CoV-2, in Human Tissues and Blood Cells. Int. J. Med. Sci. 2020, 17, 1522–1531. [Google Scholar] [CrossRef]
- Ling, R.; Dai, Y.; Huang, B.; Huang, W.; Yu, J.; Lu, X.; Jiang, Y. In Silico Design of Antiviral Peptides Targeting the Spike Protein of SARS-CoV-2. Peptides 2020, 130, 170328. [Google Scholar] [CrossRef] [PubMed]
- Totura, A.L.; Bavari, S. Broad-Spectrum Coronavirus Antiviral Drug Discovery. Expert Opin. Drug Discov. 2019, 14, 397–412. [Google Scholar] [CrossRef]
- Karim, M.; Lo, C.W.; Einav, S. Preparing for the next Viral Threat with Broad-Spectrum Antivirals. J. Clin. Investig. 2023, 133, e170236. [Google Scholar] [CrossRef]
- Adalja, A.; Inglesby, T. Broad-Spectrum Antiviral Agents: A Crucial Pandemic Tool. Expert Rev. Anti. Infect. Ther. 2019, 17, 467–470. [Google Scholar] [CrossRef]
- Rosenke, K.; Feldmann, H.; Westover, J.B.; Hanley, P.W.; Martellaro, C.; Feldmann, F.; Saturday, G.; Lovaglio, J.; Scott, D.P.; Furuta, Y.; et al. Use of Favipiravir to Treat Lassa Virus Infection in Macaques. Emerg. Infect. Dis. 2018, 24, 1696–1699. [Google Scholar] [CrossRef]
- Raabe, V.N.; Kann, G.; Ribner, B.S.; Andres, A.M.; Varkey, J.B.; Mehta, A.K.; Lyon, G.M.; Vanairsdale, S.; Faber, K.; Becker, S.; et al. Favipiravir and Ribavirin Treatment of Epidemiologically Linked Cases of Lassa Fever. Clin. Infect. Dis. 2017, 65, 855–859. [Google Scholar] [CrossRef]
- Sissoko, D.; Laouenan, C.; Folkesson, E.; M’Lebing, A.B.; Beavogui, A.H.; Baize, S.; Camara, A.M.; Maes, P.; Shepherd, S.; Danel, C.; et al. Experimental Treatment with Favipiravir for Ebola Virus Disease (the JIKI Trial): A Historically Controlled, Single-Arm Proof-of-Concept Trial in Guinea. PLoS Med. 2016, 13, e1001967. [Google Scholar] [CrossRef]
- Sanches, P.R.S.; Charlie-Silva, I.; Braz, H.L.B.; Bittar, C.; Calmon, M.F.; Rahal, P.; Cilli, E.M. Recent Advances in SARS-CoV-2 Spike Protein and RBD Mutations Comparison between New Variants Alpha (B.1.1.7, United Kingdom), Beta (B.1.351, South Africa), Gamma (P.1, Brazil) and Delta (B.1.617.2, India). J. Virus Erad. 2021, 7, 100054. [Google Scholar] [CrossRef]
- Meng, B.; Kemp, S.A.; Papa, G.; Datir, R.; Ferreira, I.A.T.M.; Marelli, S.; Harvey, W.T.; Lytras, S.; Mohamed, A.; Gallo, G.; et al. Recurrent Emergence of SARS-CoV-2 Spike Deletion H69/V70 and Its Role in the Alpha Variant B.1.1.7. Cell Rep. 2021, 35, 109292. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Hong, W.; Pan, X.; Lu, G.; Wei, X. SARS-CoV-2 Omicron Variant: Characteristics and Prevention. MedComm 2021, 2, 838–845. [Google Scholar] [PubMed]
- CDC; IDSA. COVID-19 Variant Update. Available online: https://www.idsociety.org/covid-19-real-time-learning-network/diagnostics/covid-19-variant-update/#/+/0/publishedDate_na_dt/desc/ (accessed on 22 January 2025).
- Orio, L.P.; Boschin, G.; Recca, T.; Morelli, C.F.; Ragona, L.; Francescato, P.; Arnoldi, A.; Speranza, G. New ACE-Inhibitory Peptides from Hemp Seed (Cannabis sativa L.) Proteins. J. Agric. Food Chem. 2017, 65, 10482–10488. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.; Enan, G.; Al-Mohammadi, A.R.; Abdel-Shafi, S.; Abdel-Hameid, S.; Sitohy, M.Z.; El-Gazzar, N. Antibacterial Peptides Produced by Alcalase from Cowpea Seed Proteins. Antibiotics 2021, 10, 870. [Google Scholar] [CrossRef]
- Weidmann, J.; Craik, D.J. Discovery, Structure, Function, and Applications of Cyclotides: Circular Proteins from Plants. J. Exp. Bot. 2016, 67, 4801–4812. [Google Scholar] [CrossRef]
- Grover, T.; Mishra, R.; Bushra; Gulati, P.; Mohanty, A. An Insight into Biological Activities of Native Cyclotides for Potential Applications in Agriculture and Pharmaceutics. Peptides 2021, 135, 170430. [Google Scholar] [CrossRef]
- Saether, O.; Craik, D.J.; Campbell, I.D.; Sletten, K.; Juul, J.; Norman, D.G. Elucidation of the Primary and Three-Dimensional Structure of the Uterotonic Polypeptide Kalata B1. Biochemistry 1995, 34, 4147–4158. [Google Scholar] [CrossRef]
- RCSB PDB (Protein Data Bank). REFINED STRUCTURE AND DISULFIDE PAIRING OF THE KALATA B1 PEPTIDE. Available online: https://www.rcsb.org/structure/1K48 (accessed on 18 March 2025).
- Panya, A.; Yongpitakwattana, P.; Budchart, P.; Sawasdee, N.; Krobthong, S.; Paemanee, A.; Roytrakul, S.; Rattanabunyong, S.; Choowongkomon, K.; Yenchitsomanus, P. Novel Bioactive Peptides Demonstrating Anti-Dengue Virus Activity Isolated from the Asian Medicinal Plant Acacia Catechu. Chem. Biol. Drug Des. 2019, 93, 100–109. [Google Scholar] [CrossRef]
- Castilla, V.; Barquero, A.A.; Mersich, S.E.; Coto, C.E. In Vitro Anti-Junin Virus Activity of a Peptide Isolated from Melia azedarach L. Leaves. Int. J. Antimicrob. Agents 1998, 10, 67–75. [Google Scholar] [CrossRef]
- Nguyen, P.Q.T.; Ooi, J.S.G.; Nguyen, N.T.K.; Wang, S.; Huang, M.; Liu, D.X.; Tam, J.P. Antiviral Cystine Knot α-Amylase Inhibitors from Alstonia Scholaris. J. Biol. Chem. 2015, 290, 31138–31150. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Nguyen, Q.; Tam, J. 2MM6 | Pdb_00002mm6 Solution Structure of Alpha Amylase Inhibitor Peptide AS1 from Allatide Scholaris. RCSB PDB. Available online: https://www.rcsb.org/structure/2MM6#entity-1 (accessed on 19 March 2025).
- Wang, S.; Nguyen, Q.; Tam, J. 2MM5 | Pdb_00002mm5 Solution Structure of Alpha-Amylase Inhibitor Peptide AS4 from Allatide Scholaris. RCSB PDB. Available online: https://www.rcsb.org/structure/2MM5#entity-1 (accessed on 19 March 2025).
- Singh, S.; Chauhan, P.; Sharma, V.; Rao, A.; Kumbhar, B.V.; Prajapati, V.K. Identification of Multi-Targeting Natural Antiviral Peptides to Impede SARS-CoV-2 Infection. Struct. Chem. 2023, 34, 1743–1758. [Google Scholar] [CrossRef]
- Yang, X.; Feng, P.; Yin, Y.; Bushley, K.; Spatafora, J.W.; Wang, C. Cyclosporine Biosynthesis in Tolypocladium inflatum Benefits Fungal Adaptation to the Environment. mBio 2018, 9, e01211-18. [Google Scholar] [CrossRef]
- Ciesek, S.; Steinmann, E.; Wedemeyer, H.; Manns, M.P.; Neyts, J.; Tautz, N.; Madan, V.; Bartenschlager, R.; Von Hahn, T.; Pietschmann, T. Cyclosporine A Inhibits Hepatitis C Virus Nonstructural Protein 2 through Cyclophilin A. Hepatology 2009, 50, 1638–1645. [Google Scholar] [CrossRef] [PubMed]
- Ianevski, A.; Zusinaite, E.; Kuivanen, S.; Strand, M.; Lysvand, H.; Teppor, M.; Kakkola, L.; Paavilainen, H.; Laajala, M.; Kallio-Kokko, H.; et al. Novel Activities of Safe-in-Human Broad-Spectrum Antiviral Agents. Antivir. Res. 2018, 154, 174–182. [Google Scholar] [CrossRef]
- Glowacka, P.; Rudnicka, L.; Warszawik-Hendzel, O.; Sikora, M.; Goldust, M.; Gajda, P.; Stochmal, A.; Blicharz, L.; Rakowska, A.; Olszewska, M. The Antiviral Properties of Cyclosporine. Focus on Coronavirus, Hepatitis C Virus, Influenza Virus, and Human Immunodeficiency Virus Infections. Biology 2020, 9, 192. [Google Scholar] [CrossRef]
- Rautenbach, M.; Vlok, N.M.; Stander, M.; Hoppe, H.C. Inhibition of Malaria Parasite Blood Stages by Tyrocidines, Membrane-Active Cyclic Peptide Antibiotics from Bacillus brevis. Biochim. Biophys. Acta Biomembr. 2007, 1768, 1488–1497. [Google Scholar] [CrossRef]
- Pärn, K.; Eriste, E.; Langel, Ü. The Antimicrobial and Antiviral Applications of Cell-Penetrating Peptides. In Cell-Penetrating Peptides; Humana Press: New York, NY, USA, 2015; pp. 223–245. [Google Scholar]
- Rautenbach, M.; Vosloo, J.A.; Van Rensburg, W.; Engelbrecht, Y. Natural Antimicrobial Peptides as Green Microbicides in Agriculture: A Proof of Concept Study on the Tyrocidines from Soil Bacteria; Green Economy Research Report; Green Fund, Development Bank of Southern Africa, Midrand: Midrand, South Africa, 2016. [Google Scholar] [CrossRef]
- Wenzel, M.; Rautenbach, M.; Vosloo, J.A.; Siersma, T.; Aisenbrey, C.H.M.; Zaitseva, E.; Laubscher, W.E.; van Rensburg, W.; Behrends, J.C.; Bechinger, B.; et al. The Multifaceted Antibacterial Mechanisms of the Pioneering Peptide Antibiotics Tyrocidine and Gramicidin S. mBio 2018, 9, e00802-18. [Google Scholar] [CrossRef] [PubMed]
- Tally, F.P.; DeBruin, M.F. Development of Daptomycin for Gram-Positive Infections. J. Antimicrob. Chemother. 2000, 46, 523–526. [Google Scholar] [CrossRef]
- Cao, X.; Huang, L.; Tang, M.; Liang, Y.; Liu, X.; Hou, H.; Liang, S. Antibiotics Daptomycin Interacts with S Protein of SARS-CoV-2 to Promote Cell Invasion of Omicron (B1.1.529) Pseudovirus. Virulence 2024, 15, 2339703. [Google Scholar] [CrossRef]
- Norman, A.; Franck, C.; Christie, M.; Hawkins, P.M.E.; Patel, K.; Ashhurst, A.S.; Aggarwal, A.; Low, J.K.K.; Siddiquee, R.; Ashley, C.L.; et al. Discovery of Cyclic Peptide Ligands to the SARS-CoV-2 Spike Protein Using MRNA Display. ACS Cent. Sci. 2021, 7, 1001–1008. [Google Scholar] [CrossRef]
- Kreutzer, A.G.; Krumberger, M.; Diessner, E.M.; Parrocha, C.M.T.; Morris, M.A.; Guaglianone, G.; Butts, C.T.; Nowick, J.S. A Cyclic Peptide Inhibitor of the SARS-CoV-2 Main Protease. Eur. J. Med. Chem. 2021, 221, 113530. [Google Scholar] [CrossRef] [PubMed]
- Johansen-Leete, J.; Ullrich, S.; Fry, S.E.; Frkic, R.; Bedding, M.J.; Aggarwal, A.; Ashhurst, A.S.; Ekanayake, K.B.; Mahawaththa, M.C.; Sasi, V.M.; et al. Antiviral Cyclic Peptides Targeting the Main Protease of SARS-CoV-2. Chem. Sci. 2022, 13, 3826–3836. [Google Scholar] [CrossRef] [PubMed]
- Staufer, O.; Gantner, G.; Platzman, I.; Tanner, K.; Berger, I.; Spatz, J.P. Bottom-up Assembly of Viral Replication Cycles. Nat. Commun. 2022, 13, 6530. [Google Scholar] [CrossRef]
- Johnson, N.; Pattinson, C.; Burgoyne, K.; Hijazi, K.; Houssen, W.E.; Milne, B.F. SARS-CoV-2 Spike Protein-Derived Cyclic Peptides as Modulators of Spike Interaction with GRP78. ChemBioChem 2024, 25, e202300789. [Google Scholar] [CrossRef] [PubMed]
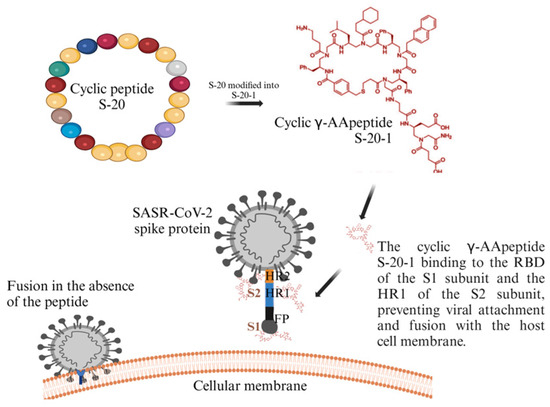
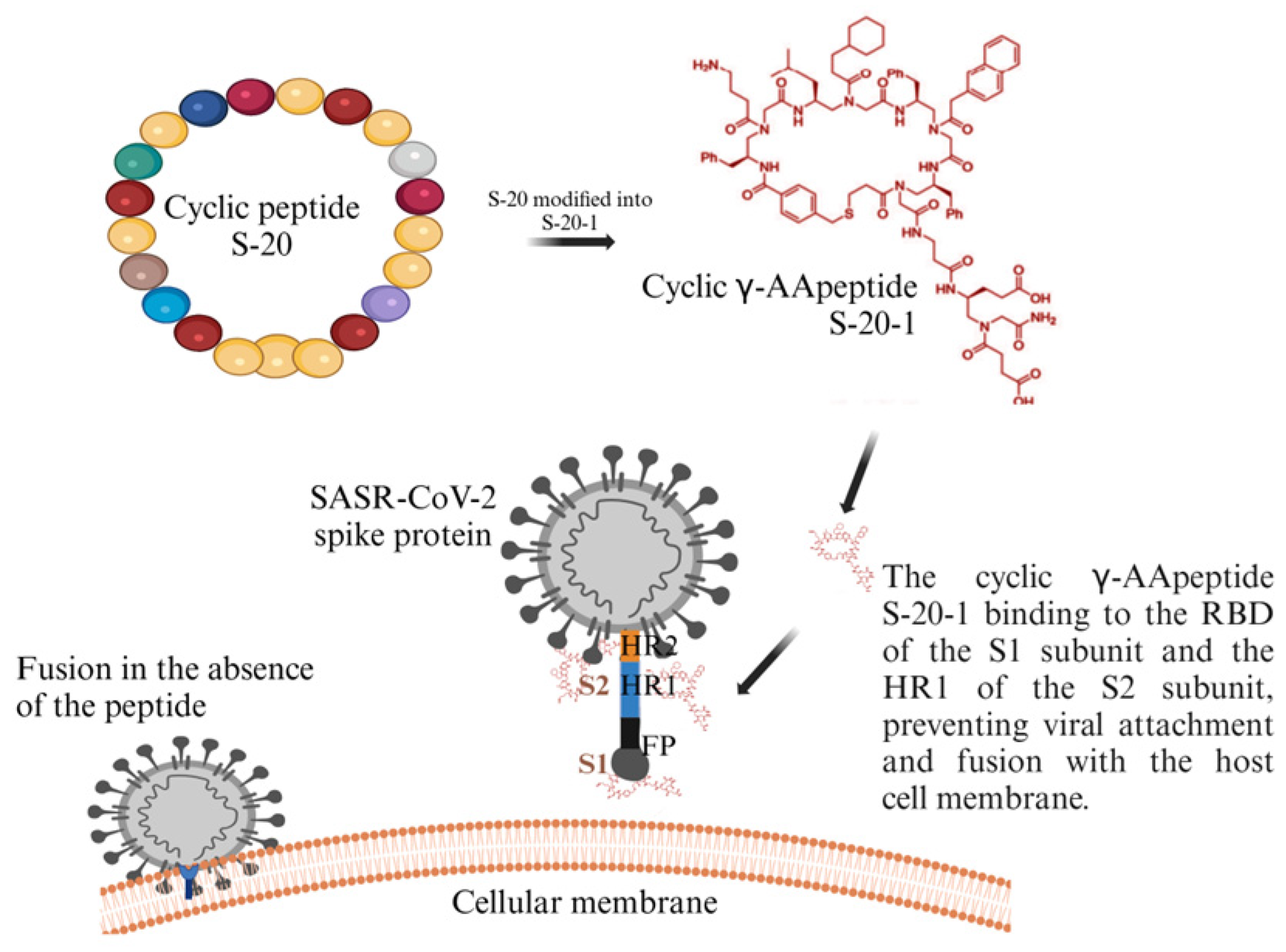
- Xue, S.; Wang, X.; Wang, L.; Xu, W.; Xia, S.; Sun, L.; Wang, S.; Shen, N.; Yang, Z.; Huang, B.; et al. A Novel Cyclic γ-AApeptide-Based Long-Acting Pan-Coronavirus Fusion Inhibitor with Potential Oral Bioavailability by Targeting Two Sites in Spike Protein. Cell Discov. 2022, 8, 88. [Google Scholar] [CrossRef]
- Iannuzzelli, J.A.; Bonn, R.; Hong, A.S.; Anitha, A.S.; Jenkins, J.L.; Wedekind, J.E.; Fasan, R. Cyclic Peptides Targeting the SARS-CoV-2 Programmed Ribosomal Frameshifting RNA from a Multiplexed Phage Display Library. Chem. Sci. 2024, 15, 19520–19533. [Google Scholar] [CrossRef]
- Freitas, E.D.; Bataglioli, R.A.; Oshodi, J.; Beppu, M.M. Antimicrobial Peptides and Their Potential Application in Antiviral Coating Agents. Colloids Surf B Biointerfaces 2022, 217, 112693. [Google Scholar]
- Ellingson, K.D.; Pogreba-Brown, K.; Gerba, C.P.; Elliott, S.P. Impact of a Novel Antimicrobial Surface Coating on Health Care-Associated Infections and Environmental Bioburden at 2 Urban Hospitals. Clin. Infect. Dis. 2020, 71, 1807–1813. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).