Synthesis of BODIPY@MOFs as Hybrid Materials for Emerging Applications: A Review
Abstract
1. Introduction
1.1. BODIPYs and Their Applications
1.2. Metal–Organic Frameworks: General Considerations and Applications
2. BODIPYs as Ligands
2.1. Monodentate Ligands
2.2. Bidentate Ligands: MOFs Containing Exclusively BODIPY Ligands
2.3. Bidentate Ligands: MOFs Containing BODIPY Ligands and Auxiliary Ligands
2.4. Tridentate Ligands
3. BODIPYs Attached to MOFs by Organic Reactions
4. Non-Coordinating BODIPYs
5. Conclusions and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AA | Accelerated Ageing |
| ACQ | Aggregation-Caused Quenching |
| AIE | Aggregation-Induced Emission |
| BODIPY | Boron-Dipyrromethene |
| CCDC | Cambridge Crystallographic Data Centre |
| CES1 | Carboxylesterase 1 |
| CuAAC | Copper-catalyzed Azide-Alkyne Cycloaddition |
| DOX | Doxorubicin |
| ECL | Electrochemiluminescence |
| EDX | Energy-Dispersive X-ray Spectroscopy |
| FRET | Förster Resonance Energy Transfer |
| ICP-MS | Inductively Coupled Plasma Mass Spectrometry |
| ILAG | Ion-and Liquid-Assisted Grinding |
| IR | Infrared Spectroscopy |
| IRET | Intrareticular Energy Transfer |
| ITO | Indium Tin Oxide |
| LED | Light-Emitting Diode |
| MIL | Matériaux de l’Institut Lavoisier |
| MOF | Metal–Organic Frameworks |
| NIR | Near-Infrared |
| NMR | Nuclear Magnetic Resonance |
| NLO | Nonlinear Optics |
| NTR | Nitroreductase |
| NU | Northwestern University |
| OER | Oxygen Evolution Reactions |
| PCN | Porous Coordination Networks |
| PDT | Photodynamic Therapy |
| PET | Photoinduced Electron Transfer |
| PXRD | Powder X-ray Diffraction |
| ROS | Reactive Oxygen Species |
| SALE | Solvent-Assisted Ligand Exchange |
| SALI | Solvent-Assisted Ligand Incorporation |
| SCXRD | Single-Crystal X-ray Diffraction |
| SEM | Scanning Electron Microscopy |
| SPDT | Sonophotodynamic Therapy |
| TEM | Transmission Electron Microscopy |
| TGA | Thermogravimetric Analysis |
| TDC | 2,5-thiophenedicarboxylate |
| UiO | University of Oslo |
| UV-vis | Ultraviolet-Visible Spectroscopy |
| VEGF | Vascular Endothelial Growth Factor |
| XPS | X-ray Photoelectron Spectroscopy |
| ZIF | Zeolitic Imidazolate Framework |
References
- Baudron, S.A. Luminescent Metal-Organic Frameworks Based on Dipyrromethene Metal Complexes and BODIPYs. CrystEngComm 2016, 18, 4671–4680. [Google Scholar] [CrossRef]
- Gupta, G.; Sun, Y.; Das, A.; Stang, P.J.; Yeon Lee, C. BODIPY Based Metal-Organic Macrocycles and Frameworks: Recent Therapeutic Developments. Coord. Chem. Rev. 2022, 452, 214308. [Google Scholar] [CrossRef]
- Xia, Q.; Zhang, J.; Chen, X.; Cheng, C.; Chu, D.; Tang, X.; Li, H.; Cui, Y. Synthesis, Structure and Property of Boron-Based Metal–Organic Materials. Coord. Chem. Rev. 2021, 435, 213783. [Google Scholar] [CrossRef]
- Kowada, T.; Maeda, H.; Kikuchi, K. BODIPY-Based Probes for the Fluorescence Imaging of Biomolecules in Living Cells. Chem. Soc. Rev. 2015, 44, 4953–4972. [Google Scholar] [CrossRef]
- Lu, H.; MacK, J.; Yang, Y.; Shen, Z. Structural Modification Strategies for the Rational Design of Red/NIR Region BODIPYs. Chem. Soc. Rev. 2014, 43, 4778–4823. [Google Scholar] [CrossRef] [PubMed]
- Loudet, A.; Burgess, K. BODIPY Dyes and Their Derivatives: Syntheses and Spectroscopic Properties. Chem. Rev. 2007, 107, 4891–4932. [Google Scholar] [CrossRef]
- Choi, S.; Bouffard, J.; Kim, Y. Aggregation-Induced Emission Enhancement of a Meso-Trifluoromethyl BODIPY via J-Aggregation. Chem. Sci. 2013, 5, 751–755. [Google Scholar] [CrossRef]
- Sharma, P.; Bouffard, J.; Kim, Y. A Reflection on ‘Aggregation-Induced Emission Enhancement of a Meso-Trifluoromethyl BODIPY via J-Aggregation’: From Fundamental Discovery to Versatile Sensing Platforms. Chem. Sci. 2025, 16, 21147–21151. [Google Scholar] [CrossRef]
- Kamkaew, A.; Lim, S.H.; Lee, H.B.; Kiew, L.V.; Chung, L.Y.; Burgess, K. BODIPY Dyes in Photodynamic Therapy. Chem. Soc. Rev. 2012, 42, 77–88. [Google Scholar] [CrossRef]
- Gurubasavaraj, P.M.; Sajjan, V.P.; Muñoz-Flores, B.M.; Jiménez Pérez, V.M.; Hosmane, N.S. Recent Advances in BODIPY Compounds: Synthetic Methods, Optical and Nonlinear Optical Properties, and Their Medical Applications. Molecules 2022, 2022, 1877. [Google Scholar] [CrossRef] [PubMed]
- Rana, P.; Singh, N.; Majumdar, P.; Prakash Singh, S. Evolution of BODIPY/Aza-BODIPY Dyes for Organic Photoredox/Energy Transfer Catalysis. Coord. Chem. Rev. 2022, 470, 214698. [Google Scholar] [CrossRef]
- Ziessel, R.; Ulrich, G.; Harriman, A. The Chemistry of Bodipy: A New El Dorado for Fluorescence Tools. New J. Chem. 2007, 31, 496–501. [Google Scholar] [CrossRef]
- Zhao, J.; Xu, K.; Yang, W.; Wang, Z.; Zhong, F. The Triplet Excited State of Bodipy: Formation, Modulation and Application. Chem. Soc. Rev. 2015, 44, 8904–8939. [Google Scholar] [CrossRef]
- Hassanain, H.; Davies, E.S.; Lewis, W.; Kays, D.L.; Champness, N.R. Structural Characterization and Optical Properties of Two Copper(I)-Iodide BODIPY Coordination Polymers. CrystEngComm 2019, 21, 4551–4556. [Google Scholar] [CrossRef]
- Mulliken, R.S. Lewis Acids and Bases and Molecular Complexes. J. Chem. Phys. 1951, 19, 514–515. [Google Scholar] [CrossRef]
- Zhou, H.C.J.; Kitagawa, S. Metal-Organic Frameworks (MOFs). Chem. Soc. Rev. 2014, 43, 5415–5418. [Google Scholar] [CrossRef]
- Morris, W.; Volosskiy, B.; Demir, S.; Gándara, F.; McGrier, P.L.; Furukawa, H.; Cascio, D.; Stoddart, J.F.; Yaghi, O.M. Synthesis, Structure, and Metalation of Two New Highly Porous Zirconium Metal-Organic Frameworks. Inorg. Chem. 2012, 51, 6443–6445. [Google Scholar] [CrossRef]
- Yusuf, V.F.; Malek, N.I.; Kailasa, S.K. Review on Metal-Organic Framework Classification, Synthetic Approaches, and Influencing Factors: Applications in Energy, Drug Delivery, and Wastewater Treatment. ACS Omega 2022, 7, 44507–44531. [Google Scholar] [CrossRef]
- Li, H.; Eddaoudi, M.; O’Keefe, M.; Yaghi, O.M. Design and Synthesis of an Exceptionally Stable and Highly Porous Metal-Organic Framework. Nature 1999, 402, 276–279. [Google Scholar] [CrossRef]
- Cavka, J.H.; Jakobsen, S.; Olsbye, U.; Guillou, N.; Lamberti, C.; Bordiga, S.; Lillerud, K.P. A New Zirconium Inorganic Building Brick Forming Metal Organic Frameworks with Exceptional Stability. J. Am. Chem. Soc. 2008, 130, 13850–13851. [Google Scholar] [CrossRef]
- Park, K.S.; Ni, Z.; Côté, A.P.; Choi, J.Y.; Huang, R.; Uribe-Romo, F.J.; Chae, H.K.; O’Keeffe, M.; Yaghi, O.M. Exceptional Chemical and Thermal Stability of Zeolitic Imidazolate Frameworks. Proc. Acad. Nat. Sci. USA 2006, 103, 10186–10191. [Google Scholar] [CrossRef]
- Eddaoudi, M.; Li, H.; Reineke, T.; Fehr, M.; Kelley, D.; Groy, T.L.; Yaghi, O.M. Design and Synthesis of Metal-Carboxylate Frameworks with Permanent Microporosity. Top. Catal. 1999, 9, 105–111. [Google Scholar] [CrossRef]
- MacRae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From Visualization to Analysis, Design and Prediction. J. Appl. Crystallogr. 2020, 53, 226–235. [Google Scholar] [CrossRef]
- Kukkar, P.; Kukkar, D. Recent Breakthroughs in the Solvothermal Assisted Synthesis of Luminescent Metal-Organic Frameworks. Inorg. Chem. Commun. 2025, 178, 114631. [Google Scholar] [CrossRef]
- Huang, C.W.; Nguyen, V.H.; Zhou, S.R.; Hsu, S.Y.; Tan, J.X.; Wu, K.C.W. Metal-Organic Frameworks: Preparation and Applications in Highly Efficient Heterogeneous Photocatalysis. Sustain. Energy Fuels 2020, 4, 504–521. [Google Scholar] [CrossRef]
- Tranchemontagne, D.J.; Hunt, J.R.; Yaghi, O.M. Room Temperature Synthesis of Metal-Organic Frameworks: MOF-5, MOF-74, MOF-177, MOF-199, and IRMOF-0. Tetrahedron 2008, 64, 8553–8557. [Google Scholar] [CrossRef]
- Dai, S.; Simms, C.; Dovgaliuk, I.; Patriarche, G.; Tissot, A.; Parac-Vogt, T.N.; Serre, C. Monodispersed MOF-808 Nanocrystals Synthesized via a Scalable Room-Temperature Approach for Efficient Heterogeneous Peptide Bond Hydrolysis. Chem. Mater. 2021, 33, 7057–7066. [Google Scholar] [CrossRef]
- Li, H.; Eddaoudi, M.; Groy, T.L.; Yaghi, O.M. Establishing Microporosity in Open Metal-Organic: Gas Sorption Isotherms for Zn(BDC) (BDC = 1,4-Benzenedicarboxylate). J. Am. Chem. Soc. 1998, 120, 8571–8572. [Google Scholar] [CrossRef]
- Ma, X.; Chai, Y.; Li, P.; Wang, B. Metal-Organic Framework Films and Their Potential Applications in Environmental Pollution Control. Acc. Chem. Res. 2019, 52, 1461–1470. [Google Scholar] [CrossRef]
- Golbaghi, A.; Pourbabaki, R.; Zarandi, A.F.; Sadeghian, M.; Damiri, Z. Sampling of Air Pollutants by MOFs, COFs, and POFs in Needle Traps: A Systematic Review. Environ. Health Eng. Manag. 2025, 12, 1546. [Google Scholar] [CrossRef]
- Ma, W.; Yu, L.; Kang, P.; Chu, Z.; Li, Y. Modifications and Applications of Metal-Organic-Framework-Based Materials for Photocatalysis. Molecules 2024, 29, 5834. [Google Scholar] [CrossRef]
- Huang, T.; Yang, H.; Xu, W.; Sun, Y.; Pang, H. Research Progress of MOF-Based Materials in Photocatalytic Reduction of CO2 and N2. Chem. Catalysis 2024, 4, 100929. [Google Scholar] [CrossRef]
- Hao, Y.; Chen, S.; Zhou, Y.; Zhang, Y.; Xu, M. Recent Progress in Metal–Organic Framework (MOF) Based Luminescent Chemodosimeters. Nanomaterials 2019, 9, 974. [Google Scholar] [CrossRef]
- Gong, D.; Li, X.; Chen, Y.; Jiao, T. Recent Progress in MOF-Based Flexible Sensors: A Review. Sustain. Mater. Technol. 2025, 44, e01429. [Google Scholar] [CrossRef]
- Elenkova, D.; Dimitrova, Y.; Tsvetkov, M.; Morgenstern, B.; Milanova, M.; Todorovsky, D.; Zaharieva, J. Investigation of the Sensing Properties of Lanthanoid Metal–Organic Frameworks (Ln-MOFs) with Terephthalic Acid. Molecules 2024, 29, 3713. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Dong, T.; Bian, Z.; Liu, H.; Liu, X.; Liu, A. Metal-Organic Frameworks Based Fluorescent Sensing: Mechanisms and Detection Applications. Coord. Chem. Rev. 2025, 529, 216470. [Google Scholar] [CrossRef]
- Fu, H.; Xu, Z.; Hou, H.; Luo, R.; Ju, H.; Lei, J. Framework-Enhanced Electrochemiluminescence in Biosensing. Chemosensors 2023, 11, 422. [Google Scholar] [CrossRef]
- Asad, M.; Anwar, M.I.; Miao, B.; Abbas, A.; Majeed, S.; Mir, I.A.; Rabbani, M.S.; Hussain, S.; Xu, S.; Al-Tahan, M.A.; et al. Recent Advances in Luminescent Metal-Organic Frameworks (L-MOFs) as Sustainable Materials for Sensing of Potentially Toxic Environmental Ubiquitous Explosive Contaminants. Sustain. Mater. Technol. 2024, 42, e01155. [Google Scholar] [CrossRef]
- Cui, Y.; Yue, Y.; Qian, G.; Chen, B. Luminescent Functional Metal-Organic Frameworks. Chem. Rev. 2012, 112, 1126–1162. [Google Scholar] [CrossRef]
- Anwar, M.I.; Asad, M.; Ma, L.; Zhang, W.; Abbas, A.; Khan, M.Y.; Zeeshan, M.; Khatoon, A.; Gao, R.; Manzoor, S.; et al. Nitrogenous MOFs and Their Composites as High-Performance Electrode Material for Supercapacitors: Recent Advances and Perspectives. Coord. Chem. Rev. 2023, 478, 214967. [Google Scholar] [CrossRef]
- Zulfiqar, A.; Miao, B.; Khan, F.; Ali, N.; Ahmed, S.; Rehman, W.; Asad, M.; Nawaz, M.A.; Mir, I.A.; Rasheed, L. Metal-Organic Framework (MOF)-Based Catalysts for Sustainable Energy Technologies: A Review. Langmuir 2025, 41, 24049–24077. [Google Scholar] [CrossRef]
- Qin, J.S.; Yuan, S.; Lollar, C.; Pang, J.; Alsalme, A.; Zhou, H.C. Stable Metal-Organic Frameworks as a Host Platform for Catalysis and Biomimetics. Chem. Commun. 2018, 54, 4231–4249. [Google Scholar] [CrossRef]
- Wang, W.; Wang, L.; Li, Z.; Xie, Z. BODIPY-Containing Nanoscale Metal-Organic Frameworks for Photodynamic Therapy. Chem. Commun. 2016, 52, 5402–5405. [Google Scholar] [CrossRef]
- Oh, J.S.; You, Y.; Park, K.C.; Gupta, G.; Kang, D.-K.; Lee, C.Y. Toward an Efficient Photosensitizer for Photodynamic Therapy: Incorporating BODIPY into Porphyrinic Nanoscale MOFs through the Solvent-Assisted Ligand Incorporation. Dyes Pigments 2019, 170, 107576. [Google Scholar] [CrossRef]
- Liu, L.; Dai, J.; Ji, Y.; Shen, B.; Zhang, X.; Linhardt, R.J. Detection of Protamine and Heparin Using a Promising Metal Organic Frameworks Based Fluorescent Molecular Device BZA-BOD@ZIF-90. Sens. Actuators B Chem. 2021, 341, 130006. [Google Scholar] [CrossRef]
- Shen, Z.Q.; Qu, L.L.; Kan, X.L.; Chen, Q.Y.; He, G.Q.; Sun, M. Construction of BODIPY Functional ZIF-8 with Improved Visible Light-Induced Antibacterial Activity. Colloids Surf. A Physicochem. Eng. Asp. 2022, 644, 128835. [Google Scholar] [CrossRef]
- Atilgan, A.; Islamoglu, T.; Howarth, A.J.; Hupp, J.T.; Farha, O.K. Detoxification of a Sulfur Mustard Simulant Using a BODIPY-Functionalized Zirconium-Based Metal–Organic Framework. ACS Appl. Mater. Interfaces 2017, 9, 24555–24560. [Google Scholar] [CrossRef]
- Seo, C.; Kim, M.; Lee, J.; Lee, C.Y.; Kim, J. Spectroscopic Evidence of Energy Transfer in Bodipy-Incorporated Nano-Porphyrinic Metal-Organic Frameworks. Nanomaterials 2020, 10, 1925. [Google Scholar] [CrossRef] [PubMed]
- Tran, A.; Leroux, M.; Michelin, C.; Réveret, F.; Boyer, D.; Cisnetti, F. Carboxylate BODIPY Integrated in MOF-5: Easy Preparation and Solid-State Luminescence. J. Mater. Chem. C 2023, 11, 14896–14905. [Google Scholar] [CrossRef]
- Mondloch, J.E.; Bury, W.; Fairen-Jimenez, D.; Kwon, S.; Demarco, E.J.; Weston, M.H.; Sarjeant, A.A.; Nguyen, S.T.; Stair, P.C.; Snurr, R.Q.; et al. Vapor-Phase Metalation by Atomic Layer Deposition in a Metal-Organic Framework. J. Am. Chem. Soc. 2013, 135, 10294–10297. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Pan, J.; Gong, Z.; Meng, S. Near-Infrared-Responsive Metal–Organic Framework@NaLnF4 Upconversion Nanoparticle Heterostructures for Advanced Photodynamic Theranostics. ACS Appl. Mater. Interfaces 2025, 17, 41816–41824. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Jiang, X.; Gong, Z.; Li, J.; Du, X.; Meng, S. Formulating the Relationship Between Intermolecular Interactions and Photodynamic Effects Based on the Optical Regularity Exhibited by Rare Earth-BODIPY Crystalline Frameworks System. Adv. Funct. Mater. 2025, 35, 2415925. [Google Scholar] [CrossRef]
- Chen, X.; Mendes, B.B.; Zhuang, Y.; Conniot, J.; Mercado Argandona, S.; Melle, F.; Sousa, D.P.; Perl, D.; Chivu, A.; Patra, H.K.; et al. A Fluorinated BODIPY-Based Zirconium Metal-Organic Framework for In Vivo Enhanced Photodynamic Therapy. J. Am. Chem. Soc. 2024, 146, 1644–1656. [Google Scholar] [CrossRef]
- Lin, X.C.; Wang, Y.M.; Chen, X.; You, P.Y.; Mo, K.M.; Ning, G.H.; Li, D. A Photosensitizing Metal–Organic Framework as a Tandem Reaction Catalyst for Primary Alcohols from Terminal Alkenes and Alkynes. Angew. Chem. Int. Ed. 2023, 62, e202306497. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Song, W.; Liang, Y.; Liu, Y.; Li, B.; Li, H.; Zhang, L.; Ma, Y.; Ye, R.; Tang, B.Z.; et al. Direct Synthesis of Topology-Controlled BODIPY and CO2-Based Zirconium Metal-Organic Frameworks for Efficient Photocatalytic CO2 Reduction. Angew. Chem. Int. Ed. 2025, 64, e202421248. [Google Scholar] [CrossRef]
- Liu, B.; Chen, X.; Yang, Y.; Alizadeh Kiapi, M.R.; Menon, D.; Zhao, Q.; Yuan, G.; Keenan, L.L.; Fairen-Jimenez, D.; Xia, Q. Engineering Bodipy-Based Metal–Organic Frameworks for Efficient Full-Spectrum Photocatalysis in Amide Synthesis. Angew. Chem. Int. Ed. 2025, 64, e202505405. [Google Scholar] [CrossRef]
- Lippke, J.; Brosent, B.; Von Zons, T.; Virmani, E.; Lilienthal, S.; Preuße, T.; Hulsmann, M.; Schneider, A.M.; Wuttke, S.; Behrens, P.; et al. Expanding the Group of Porous Interpenetrated Zr-Organic Frameworks (PIZOFs) with Linkers of Different Lengths. Inorg. Chem. 2017, 56, 748–761. [Google Scholar] [CrossRef]
- Lee, C.Y.; Farha, O.K.; Hong, B.J.; Sarjeant, A.A.; Nguyen, S.T.; Hupp, J.T. Light-Harvesting Metal-Organic Frameworks (MOFs): Efficient Strut-to-Strut Energy Transfer in Bodipy and Porphyrin-Based MOFs. J. Am. Chem. Soc. 2011, 133, 15858–15861. [Google Scholar] [CrossRef]
- Zhou, L.; Xue, Y.S.; Xu, Y.; Zhang, J.; Du, H. Bin Two Photoluminescent Metal-Organic Frameworks Based on a BODIPY-Derived Bipyridine Ligand. CrystEngComm 2013, 15, 7315–7320. [Google Scholar] [CrossRef]
- Li, M.; Yao, Y.; Ding, J.; Liu, L.; Qin, J.; Zhao, Y.; Hou, H.; Fan, Y. Spectroscopic and Crystallographic Investigations of Novel Bodipy-Derived Metal-Organic Frameworks. Inorg. Chem. 2015, 54, 1346–1353. [Google Scholar] [CrossRef]
- Kong, W.; Liu, T.; Meng, X.; Dong, P.; Lei, J.; Ju, H. Intrareticular Energy Transfer in Dual-Photosensitized Metal-Organic Frameworks to Enhance Photoelectrochemical Performance. J. Am. Chem. Soc. 2025, 147, 24976–24983. [Google Scholar] [CrossRef]
- Xu, Z.; Wu, F.; Zhu, D.; Fu, H.; Shen, Z.; Lei, J. BODIPY-Based Metal-Organic Frameworks as Efficient Electrochemiluminescence Emitters for Telomerase Detection. Chem. Commun. 2022, 58, 11515–11518. [Google Scholar] [CrossRef]
- Gupta, G.; Gusmão, F.; Paul, A.; Šljukić, B.; Santos, D.M.F.; Lee, J.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L.; Lee, C.Y. A Mixed-Ligand Co Metal-Organic Framework and Its Carbon Composites as Excellent Electrocatalysts for the Oxygen Evolution Reaction in Green-Energy Devices. Dalton Trans. 2023, 53, 5001–5009. [Google Scholar] [CrossRef]
- Meng, Y.; Du, Y.; Lin, Y.; Su, Y.; Li, R.; Feng, Y.; Meng, S. A Two-Fold Interpenetration Pillar-Layered Metal-Organic Frameworks Based on BODIPY for Chemo-Photodynamic Therapy. Dyes Pigments 2021, 188, 109174. [Google Scholar] [CrossRef]
- Gupta, G.; Kim, M.; Lee, J.; Lee, C.Y. Zinc-Based Metal Organic Framework Derived From Anthracene and BODIPY Chromophores: Synthesis and Photophysical Properties. Bull. Korean Chem. Soc. 2021, 42, 645–648. [Google Scholar] [CrossRef]
- Yang, H.; Wang, J.; Ma, J.; Yang, H.; Zhang, J.; Lv, K.; Wen, L.; Peng, T. A Novel BODIPY-Based MOF Photocatalyst for Efficient Visible-Light-Driven Hydrogen Evolution. J. Mater. Chem. A Mater. 2019, 7, 10439–10445. [Google Scholar] [CrossRef]
- Kang, Q.; Wang, S.D.; Guo, J.; Mo, K.L.; Lv, K.-L.; Wen, L.L. Novel BODIPY-Based MOFs toward High-Efficiency Photocatalytic Oxidation of Sulfides and Arylboronic Acids. Inorg. Chem. Front. 2024, 11, 4731–4739. [Google Scholar] [CrossRef]
- Nie, C.Y.; Xue, L.J.; Wang, S.D.; Li, M.Y.; Kang, Q.; Wen, L.L. Isomorphic BODIPY-Based Metal-Organic Frameworks for High-Efficiency Photoredox Organic Transformations. Inorg. Chem. Front. 2025, 12, 3521–3530. [Google Scholar] [CrossRef]
- Qi, H.; Teesdale, J.J.; Pupillo, R.C.; Rosenthal, J.; Bard, A.J. Synthesis, Electrochemistry, and Electrogenerated Chemiluminescence of Two BODIPY-Appended Bipyridine Homologues. J. Am. Chem. Soc. 2013, 135, 13558–13566. [Google Scholar] [CrossRef]
- Zhang, M.; Cao, M.; Sun, Z.B.; Han, Z.; Wang, Z.Y.; Wang, Q.Y.; Zang, S.Q. Modulating BODIPY-Based Silver Chalcogenide Cluster-Based Metal-Organic Frameworks for Real-Time Decontamination of a Gaseous Sulfur Mustard Simulant. Chem. Mater. 2023, 35, 10238–10246. [Google Scholar] [CrossRef]
- Li, H.; Xu, H.; Wang, G.; Chen, J.; Ji, D.; Huang, Y.; Cui, G.; He, H.; Guo, Z. Rational Design of Mesoporous Coordination Polymer Nanophotosensitizers for Photodynamic Tumor Ablation. ACS Appl. Mater. Interfaces 2023, 15, 21746–21753. [Google Scholar] [CrossRef]
- Taylor-Pashow, K.M.L.; Della Rocca, J.; Xie, Z.; Tran, S.; Lin, W. Postsynthetic Modifications of Iron-Carboxylate Nanoscale Metal-Organic Frameworks for Imaging and Drug Delivery. J. Am. Chem. Soc. 2009, 131, 14261–14263. [Google Scholar] [CrossRef]
- Quan, Y.; Li, Q.Y.; Zhang, Q.; Zhang, W.Q.; Lu, H.; Yu, J.H.; Chen, J.; Zhao, X.; Wang, X.J. A Diiodo-BODIPY Postmodified Metal-Organic Framework for Efficient Heterogeneous Organo-Photocatalysis. RSC Adv. 2016, 6, 23995–23999. [Google Scholar] [CrossRef]
- Liu, W.; Chen, L.; Wang, L.; Xie, Z. Photoactive Metal–Organic Framework@Porous Organic Polymer Nanocomposites with pH-Triggered Type I Photodynamic Therapy. Adv. Mater. Interfaces 2020, 7, 2000504. [Google Scholar] [CrossRef]
- Li, Y.; Li, J.J.; Zhang, Q.; Zhang, J.Y.; Zhang, N.; Fang, Y.Z.; Yan, J.; Ke, Q. The Multifunctional BODIPY@Eu-MOF Nanosheets as Bioimaging Platform: A Ratiometric Fluorescencent Sensor for Highly Efficient Detection of F-, H2O2 and Glucose. Sens. Actuators B Chem. 2022, 354, 131140. [Google Scholar] [CrossRef]
- Li, B.; Lu, X.; Sun, X.; Zhou, H.; Tian, Y.; Hai, Z.; Li, D. Fluorescence Lifetime Imaging-Guided Photodynamic Therapy over Two-Photon Responsive Metal-Organic Frameworks. Inorg. Chem. Front. 2025, 12, 1850–1856. [Google Scholar] [CrossRef]
- Guo, G.C.; Zhao, J.P.; Guo, S.; Shi, W.X.; Liu, F.C.; Lu, T.B.; Zhang, Z.M. Building Co16-N3-Based UiO-MOF to Expand Design Parameters for MOF Photosensitization. Angew. Chem. Int. Ed. 2024, 63, e202402374. [Google Scholar] [CrossRef]
- Li, B.; Lu, X.; Tian, Y.; Li, D. Embedding Multiphoton Active Units within Metal–Organic Frameworks for Turning on High-Order Multiphoton Excited Fluorescence for Bioimaging. Angew. Chem. Int. Ed. 2022, 61, e202206755. [Google Scholar] [CrossRef] [PubMed]
- Lyu, J.; Zhang, X.; Otake, K.I.; Wang, X.; Li, P.; Li, Z.; Chen, Z.; Zhang, Y.; Wasson, M.C.; Yang, Y.; et al. Topology and Porosity Control of Metal-Organic Frameworks through Linker Functionalization. Chem. Sci. 2019, 10, 1186–1192. [Google Scholar] [CrossRef]
- Wang, R.; Bukowski, B.C.; Duan, J.; Zhang, K.; Snurr, R.Q.; Hupp, J.T. Geometry and Chemistry: Influence of Pore Functionalization on Molecular Transport and Diffusion in Solvent-Filled Zirconium Metal-Organic Frameworks. ACS Appl. Mater. Interfaces 2023, 15, 51854–51862. [Google Scholar] [CrossRef]
- Glembockyte, V.; Frenette, M.; Mottillo, C.; Durantini, A.M.; Gostick, J.; Štrukil, V.; Friščić, T.; Cosa, G. Highly Photostable and Fluorescent Microporous Solids Prepared via Solid-State Entrapment of Boron Dipyrromethene Dyes in a Nascent Metal-Organic Framework. J. Am. Chem. Soc. 2018, 140, 16882–16887. [Google Scholar] [CrossRef]
- Fetzer, M.N.A.; Vieten, M.; Limon, A.; Janiak, C. Encapsulation of a Highly Acid-Stable Dicyano-Bodipy in Zr-Based Metal–Organic Frameworks with Increased Fluorescence Lifetime and Quantum Yield Within the Solid Solution Concept. Molecules 2025, 30, 4151. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.; Ma, C.; Ji, Y.; Dai, J.; Li, B.; Zhang, X.; Huang, H. Detection of Carboxylesterase 1 and Chlorpyrifos with ZIF-8 Metal–Organic Frameworks Using a Red Emission BODIPY-Based Probe. ACS Appl. Mater. Interfaces 2021, 13, 8718–8726. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Wang, F.; Lin, J.; Yang, W.; Kang, W.; Jiang, H.; Adade, S.Y.S.S.; Cai, J.; Xue, Z.; Chen, Q. Detection of Wheat Toxigenic Aspergillus Flavus Based on Nano-Composite Colorimetric Sensing Technology. Food Chem. 2023, 405, 134803. [Google Scholar] [CrossRef]
- Wu, J.; Zhai, J.; Wang, B.; Ding, W.; Ding, R.; Li, K.; Muhammad, S.; Zhang, X.; He, P.; Shen, B. Fabrication of a Zeolite Imidazole Framework-8-Based Red Emitting Nanocomposite for Sensitive Detection of Nitro Reductase. Dyes Pigments 2022, 202, 110220. [Google Scholar] [CrossRef]
- Guan, Q.; Zhou, L.-L.; Li, Y.A.; Dong, Y.B. Diiodo-Bodipy-Encapsulated Nanoscale Metal-Organic Framework for pH-Driven Selective and Mitochondria Targeted Photodynamic Therapy. Inorg. Chem. 2018, 57, 10137–10145. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Zhang, L.; Ju, M.; Liu, L.; Ma, C.; Huang, Y.; Wang, B.; Ding, W.; Luan, X.; Shen, B. Rational Modulation of BODIPY Photosensitizers to Design Metal-Organic Framework-Based NIR Nanocomposites for High-Efficiency Photodynamic Therapy in a Hypoxic Environment. ACS Appl. Mater. Interfaces 2022, 14, 46262–46272. [Google Scholar] [CrossRef]
- Friščić, T. New Opportunities for Materials Synthesis Using Mechanochemistry. J. Mater. Chem. 2010, 20, 7599–7605. [Google Scholar] [CrossRef]
- Cliffe, M.J.; Mottillo, C.; Stein, R.S.; Bučar, D.K.; Friščić, T. Accelerated Aging: A Low Energy, Solvent-Free Alternative to Solvothermal and Mechanochemical Synthesis of Metal-Organic Materials. Chem. Sci. 2012, 3, 2495–2500. [Google Scholar] [CrossRef]
- Khatua, S.; Goswami, S.; Biswas, S.; Tomar, K.; Jena, H.S.; Konar, S. Stable Multiresponsive Luminescent MOF for Colorimetric Detection of Small Molecules in Selective and Reversible Manner. Chem. Mater. 2015, 27, 5349–5360. [Google Scholar] [CrossRef]
- Ding, Z.; Wang, C.; Wang, S.; Wu, L.; Zhang, X. Light-Harvesting Metal-Organic Framework Nanoprobes for Ratiometric Fluorescence Energy Transfer-Based Determination of pH Values and Temperature. Microchim. Acta 2019, 186, 476. [Google Scholar] [CrossRef] [PubMed]







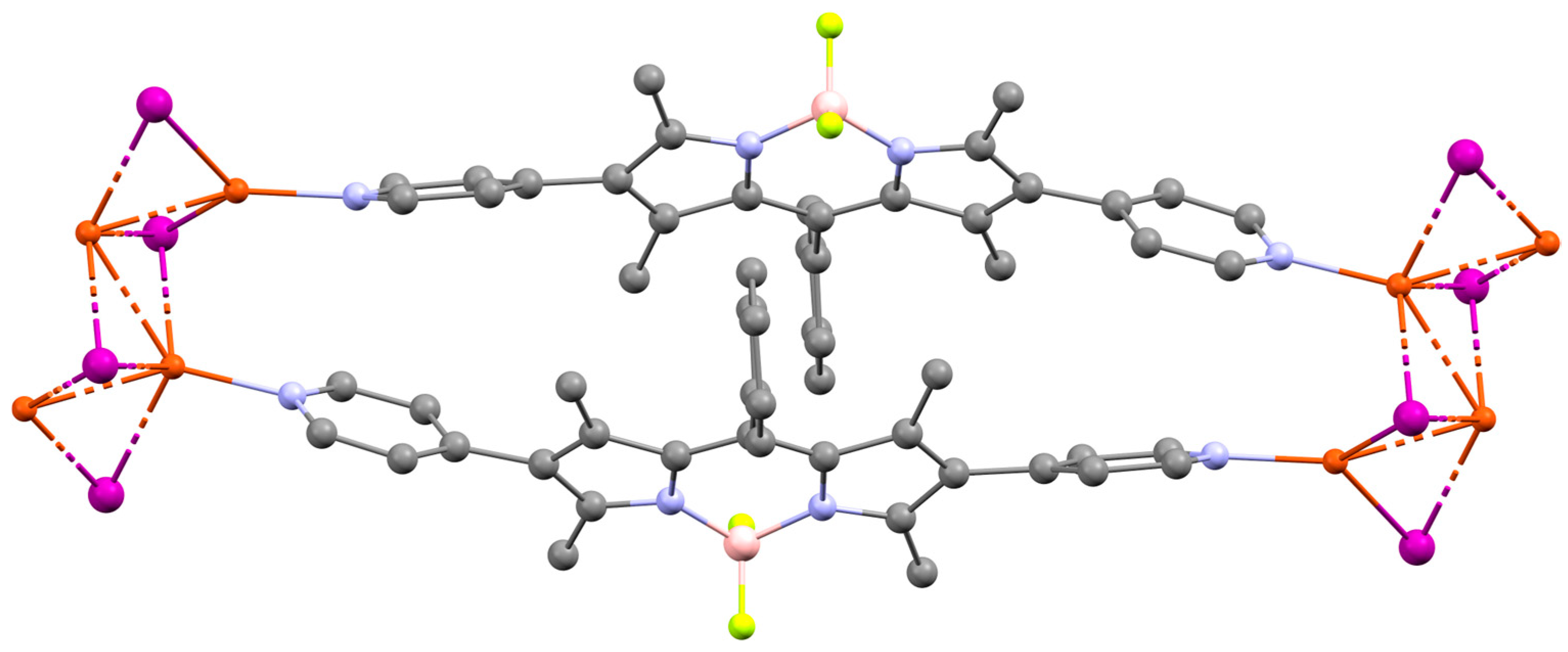


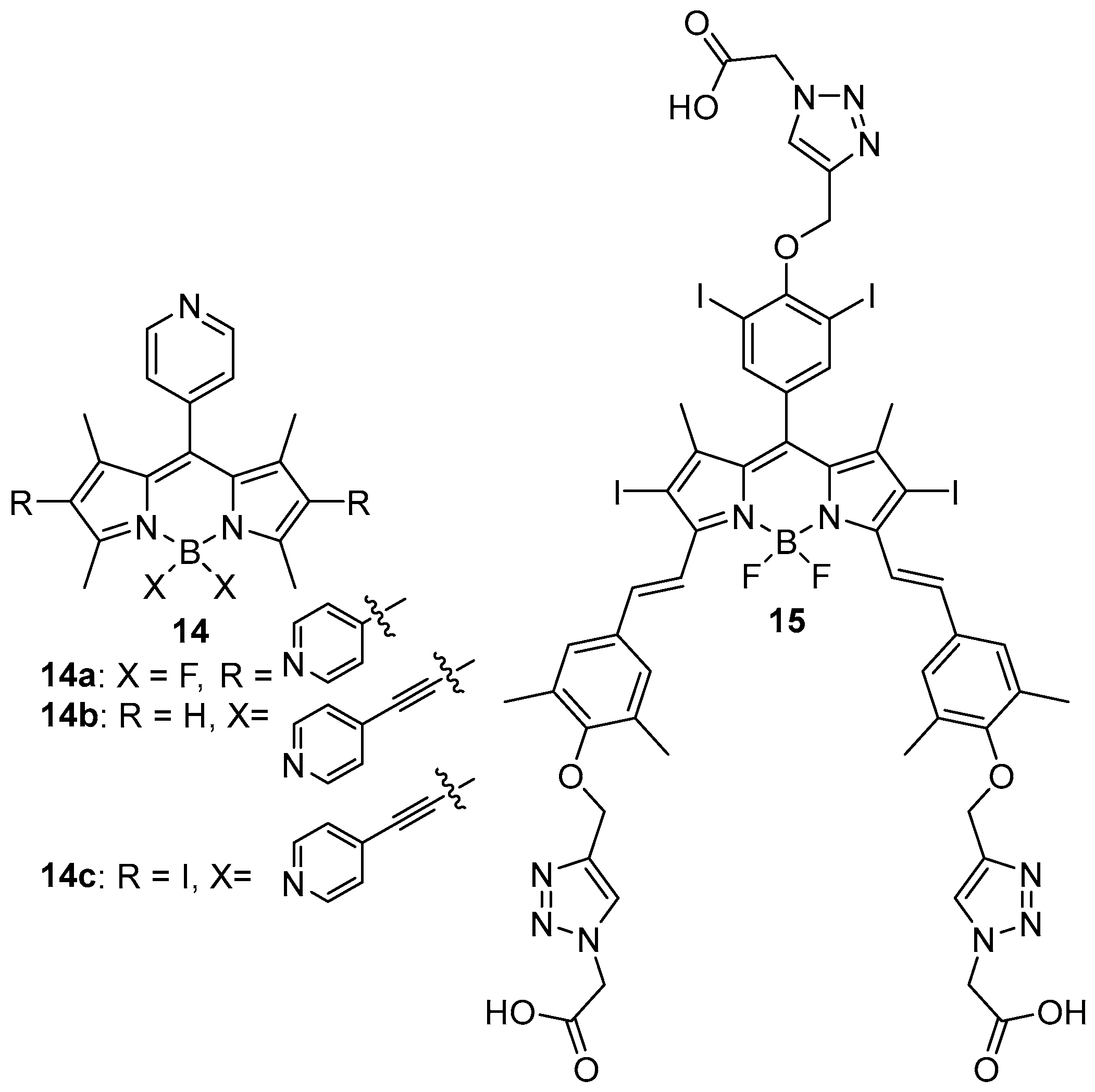


| MOF | Formula | General Synthetic Method | Reaction Medium | Temperature (°C) | Time (h) | Reference |
|---|---|---|---|---|---|---|
| MOF-5 | Zn4O(C8H6O4)3 | Slow vapour diffusion | Et3N/ Toluene/DMF | RT | NR 1 | [28] |
| UiO-66 | Zr6O4(OH)4(C8H4O4)6 | Solvothermal | DMF | 120 | 24 | [20] |
| ZIF-8 | Zn(C4H6N2)2 | DMF | 140 | 24 | [21] |
| Ligand, MOF | Approach (Synthetic Conditions) | Quantification of BODIPY in the MOF | Application | Reference |
|---|---|---|---|---|
| 2, UiO-66 | SALE (DMF 65 °C, 24 h) | 27.9% (ICP-MS, UV-Vis) | PDT | [43] |
| 3, PCN-222 | SALI (MeCN/DMSO 3:2, 60 °C, 24 h) | 44% relative to the porphyrin ligand of PCN-222 (Liquid NMR after decomposition) | PDT | [44] |
| 4, ZIF-90 | One-pot (H2O, RT, overnight) | ND (0.3 wt-% relative to imidazole ligand used) | Detection of analytes | [45] |
| 5, ZIF-8 | One-pot (DMF, RT, 12 h addition of BODIPY delayed by 2 h) | ND (2.5 mol-% relative to the imidazole ligand used) | Antibacterial agent | [46] |
| 6, NU-1000 | SALI (MeCN, 60°C, 24 h) | 0.75/Zr6 node (X-ray fluorescence) | Photocatalysis (detoxification of sulphur mustard) | [47] |
| 3 or 4, nPCN-222 | SALI (MeCN/DMSO 3:2, 60 °C, 24 h) | 3.5–3.6/Zr6 node (Liquid NMR after decomposition) | Light harvesting/FRET | [48] |
| 6 or 7, MOF-5 | One-pot (DMF/NEt3, RT, 2.5 h) | Up to 17.7 wt-% (Spectrophotometric assay of washings) | Luminescent materials | [49] |
| Ligand, Cluster | Approach (Synthetic Conditions) | Proof of Structure | Application | Reference(s) |
|---|---|---|---|---|
| 1a/Cu4I4 1b/Cu2I2 | One-pot (MeCN, 55 °C, 24 h) | SCXRD compared with PXRD | Luminescent materials 1 | [14] |
| 8/RE ions 2 | One-pot (DMF/Acetate/Water, 60 °C, 24 h) | SCXRD compared with PXRD | PDT | [51,52] |
| 9/Zr6O8 | SALE (DMF, 70 °C, 96 h) 3 | SCXRD compared with PXRD before and after SALE 1H NMR (100% of exchange) | PDT | [53] |
| 10/Cu3L3 | One-pot (nBu-OH/BnOH, 120 °C, 72 h) | PXRD compared with the predicted model 4 | Photocatalysis (Organic synthesis) | [54] |
| 11/Zr6O8 11/Zr12O22 | One-pot (Formic acid, 70 °C, 72 h) | Elemental analysis SCXRD compared with PXRD | Photocatalysis (CO2 reduction) | [55] |
| 12/Zr6O8 | One-pot (DMF, 120 °C, 24 h) | Elemental analysis SCXRD compared with PXRD | Photocatalysis (Organic synthesis) | [56] |
| Ligand/Metal/ Aux. Ligand | Synthetic Conditions 1 | Proof of Structure | Application | Reference |
|---|---|---|---|---|
| 1a/Zn2+/L2 or L3 | DMF, 80 °C, 2 h addition of BODIPY delayed by 20 h with HNO3/EtOH | SCXRD compared with PXRD | FRET | [58] |
| 1a/Zn2+/L4 or L3 | DMF, HNO3/EtOH, 80 °C, 48 h | SCXRD compared with PXRD | Luminescent Materials | [59] |
| 12a/Zn2+ or Cd2+/L6 | DMF, HNO3/EtOH, 80 °C, 48 h | SCXRD compared with PXRD | Luminescent Materials | [60] |
| 1a/Cd2+/L3 | DMA/HNO3, 90 °C, 72 h | SCXRD compared with PXRD | Detection of analytes | [61] |
| 1a/Zn2+/L5 | DMF, HNO3/EtOH, 85 °C, 48 h | SCXRD compared with PXRD | Detection of analytes | [62] |
| 1a/Co2+/L4 | DMF/water, 85 °C, 72 h 2 | SCXRD compared with PXRD | Electrocatalysis (OER catalyst) | [63] |
| 13/Cd2+/L8 | DMF/EtOH, HNO3, 80 °C, 72 h | SCXRD compared with PXRD | PDT | [64] |
| 1a/Zn2+/L9 | DMF/EtOH, HNO3, 90 °C, 18 h | SCXRD compared with PXRD | Photocatalysis (Organic synthesis) | [65] |
| 1a/Zn2+/L5 | DMA/CH3OH, HNO3, 90 °C, 48 h | SCXRD compared with PXRD | Photocatalysis (Hydrogen evolution reaction) | [66] |
| 1a/Zn2+ or Cd2+/L10 | DMA/EtOH, HNO3, 90 °C, 24 h | SCXRD compared with PXRD | Photocatalysis (Organic synthesis) | [67] |
| 1a/Zn2+ or Co2+/L11 | DMF/MeOH, 70 °C, 48 h | SCXRD compared with PXRD | Photocatalysis (Organic synthesis) | [68] |
| Ligand/ Cluster | Synthetic Conditions | Proof of Structure | Application | Reference |
|---|---|---|---|---|
| 14/Ag12(SR)6(CF3COO)6 R = tBu, iPr | Ethanol/Acetonitrile (1:1), PhPO3H2 (2–5 d) | SCXRD compared with PXRD | Photocatalysis (decontamination) | [70] |
| 15/Zr6O8 | DMF, RT, 6 h | SCXRD compared with PXRD | PDT | [71] |
| BODIPY, MOF | Reaction (Synthetic Condition) | Quantification of BODIPY in the MOF | Application | Reference |
|---|---|---|---|---|
| 16, MIL-101(Fe) | Nucleophilic Substitution (excess BODIPY, THF, RT, 2 days) | ~10% (UV-Vis) | Biologically targeted dye delivery | [72] |
| 17, UiO-68-NH2 | Amine acylation (THF, RT, 1 day) | 11% of the organic ligands functionalized (XRF) | Synthetic photocatalysis | [73] |
| 18, UiO-66-NH2 | Imine formation (DMF, TFA, TP, RT, overnight) | 15% wt-% (TGA, total added organic content 1) | PDT | [74] |
| 19, EuIII-L13 MOF | Adduct formation on the boronic acid 2 (DMF/Ethylene glycol 7:3, autoclave, 120°) | ND (up to 9.5 wt-% loading) | Detection of analytes | [75] |
| 20, ZrTc 3 | Azide-alkyne cycloaddition 4 | ND | PDT and biological imaging | [76] |
| 21 or 22, Co16 MOF 5 | Azide-alkyne cycloaddition 2 (DMF/MeOH 1:1, autoclave, 140 °C, cat.: CuSO4/ascorbic acid) | ND (proof of grafting by control experiments) | Synthetic photocatalysis | [77] |
| BODIPY, MOF | Approach (Conditions) | Quantification of BODIPY in the MOF | Application | Reference |
|---|---|---|---|---|
| 24, ZIF-8 | Mechanochemistry 1 | Up to 100% encapsulation of up to 5 wt-% BODIPY (UV-Vis assay of washings) | Luminescent Materials | [81] |
| 26, UiO-66, MOF-808, DUT-67 and MIP-206 | Post-synthetic soaking of the MOF in a DCM solution of the BODIPY (RT, 5 days) OR One-pot (DMF, 80 °C, 12 h) | UV-vis assay after MOF digestion in KOH.Post-synthetic: 0.3–0.57 wt-%One-pot 1.9–7.2 wt-% | Luminescent Materials | [82] |
| 27, ZIF-8 | One-pot encapsulation during MOF synthesis (Water, RT, 12 h) | 76% integration of 0.3 wt-% BODIPY (UV-Vis assay of washings) | Detection of analytes | [83] |
| 28, copper-based terpyridine MOF | Post-synthetic soaking of the MOF in DMF solution of BODIPY | ND (1% wt-% of BODIPY used) | Detection of analytes | [84] |
| 29, ZIF-8 | One-pot encapsulation during MOF synthesis (Water, RT, overnight) | ND (<0.15 mol% of BODIPY used) | Detection of analytes | [85] |
| 30, ZIF-90 | One-pot encapsulation during MOF synthesis (DMF, trioctylamine, <0 °C, 1 min) | 25.4 wt-% (ICP) 2 | PDT | [86] |
| 31, ZIF-8 | One-pot encapsulation during MOF synthesis (Water, RT, 12 h) | ND (0.2 wt-% of BODIPY used) | PDT | [87] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bureller, L.; Michelin, C.; Cisnetti, F. Synthesis of BODIPY@MOFs as Hybrid Materials for Emerging Applications: A Review. Molecules 2025, 30, 4790. https://doi.org/10.3390/molecules30244790
Bureller L, Michelin C, Cisnetti F. Synthesis of BODIPY@MOFs as Hybrid Materials for Emerging Applications: A Review. Molecules. 2025; 30(24):4790. https://doi.org/10.3390/molecules30244790
Chicago/Turabian StyleBureller, Louise, Clément Michelin, and Federico Cisnetti. 2025. "Synthesis of BODIPY@MOFs as Hybrid Materials for Emerging Applications: A Review" Molecules 30, no. 24: 4790. https://doi.org/10.3390/molecules30244790
APA StyleBureller, L., Michelin, C., & Cisnetti, F. (2025). Synthesis of BODIPY@MOFs as Hybrid Materials for Emerging Applications: A Review. Molecules, 30(24), 4790. https://doi.org/10.3390/molecules30244790








