Semisynthesis of Betaxanthins from Purified Betacyanin of Opuntia dillenii sp.: Color Stability and Antiradical Capacity
Abstract
1. Introduction
2. Results and Discussion
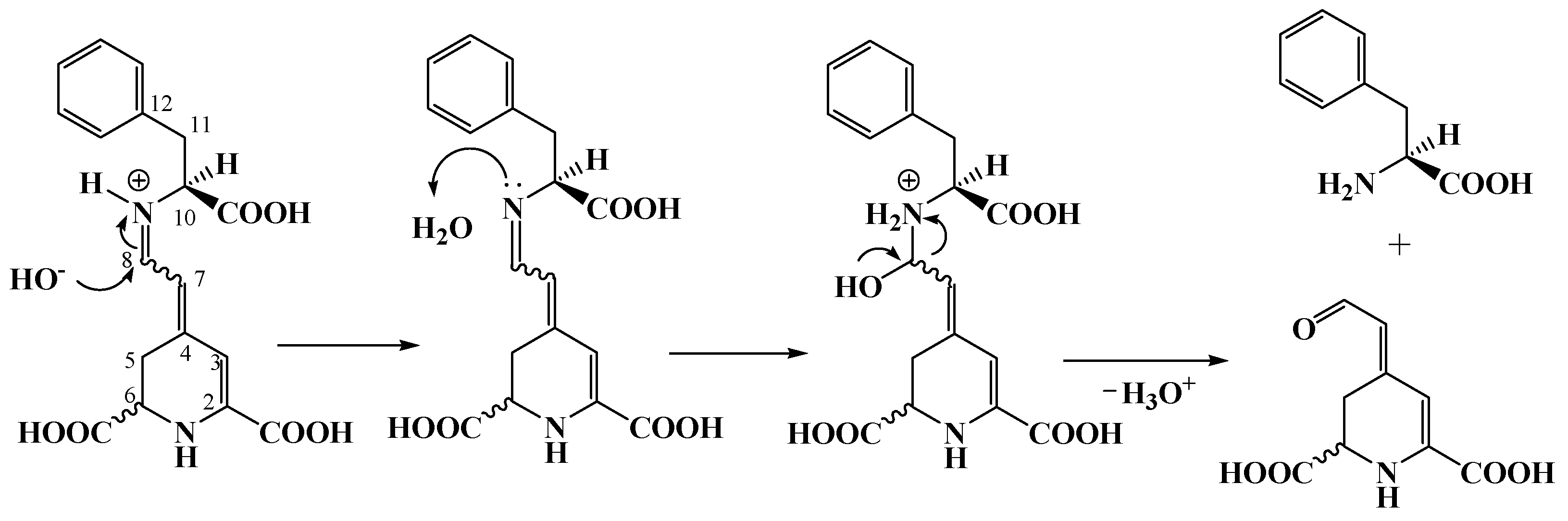
2.1. Isolation of the Purified Extract from the Fruit of Opuntia Dilleni and Obtaining Betalamic Acid
2.2. Semisynthesis of ʟ−Amino Acid-Betaxanthins
2.3. Tentative Characterization of Betaxanthins by UV-Vis and HPLC-ESI-MS
2.4. Determination of the Antioxidant Capacity
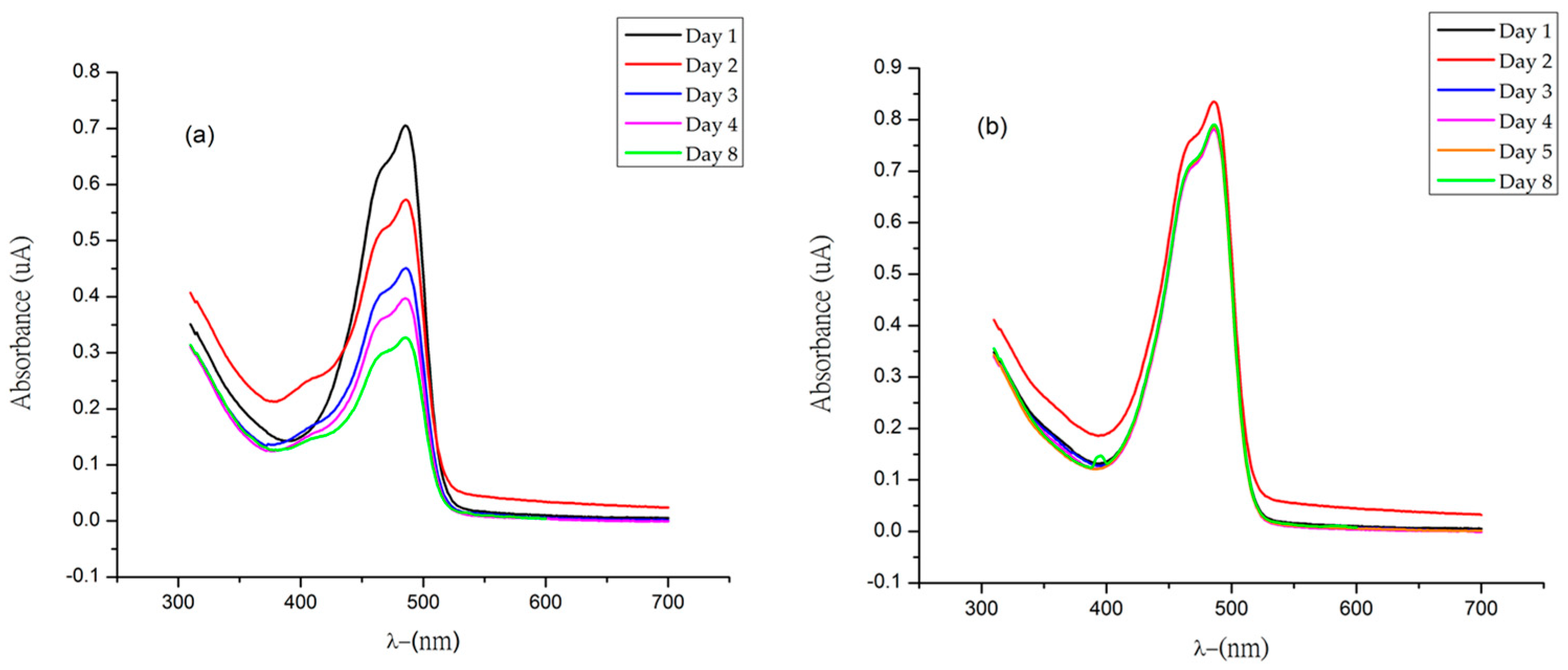
2.5. Stability Evaluation of Semisynthetic ʟ−Amino Acid-Betaxanthins by Tristimulus Colorimetry
3. Materials and Methods
3.1. Reagents and Solvents
3.2. Plant Material
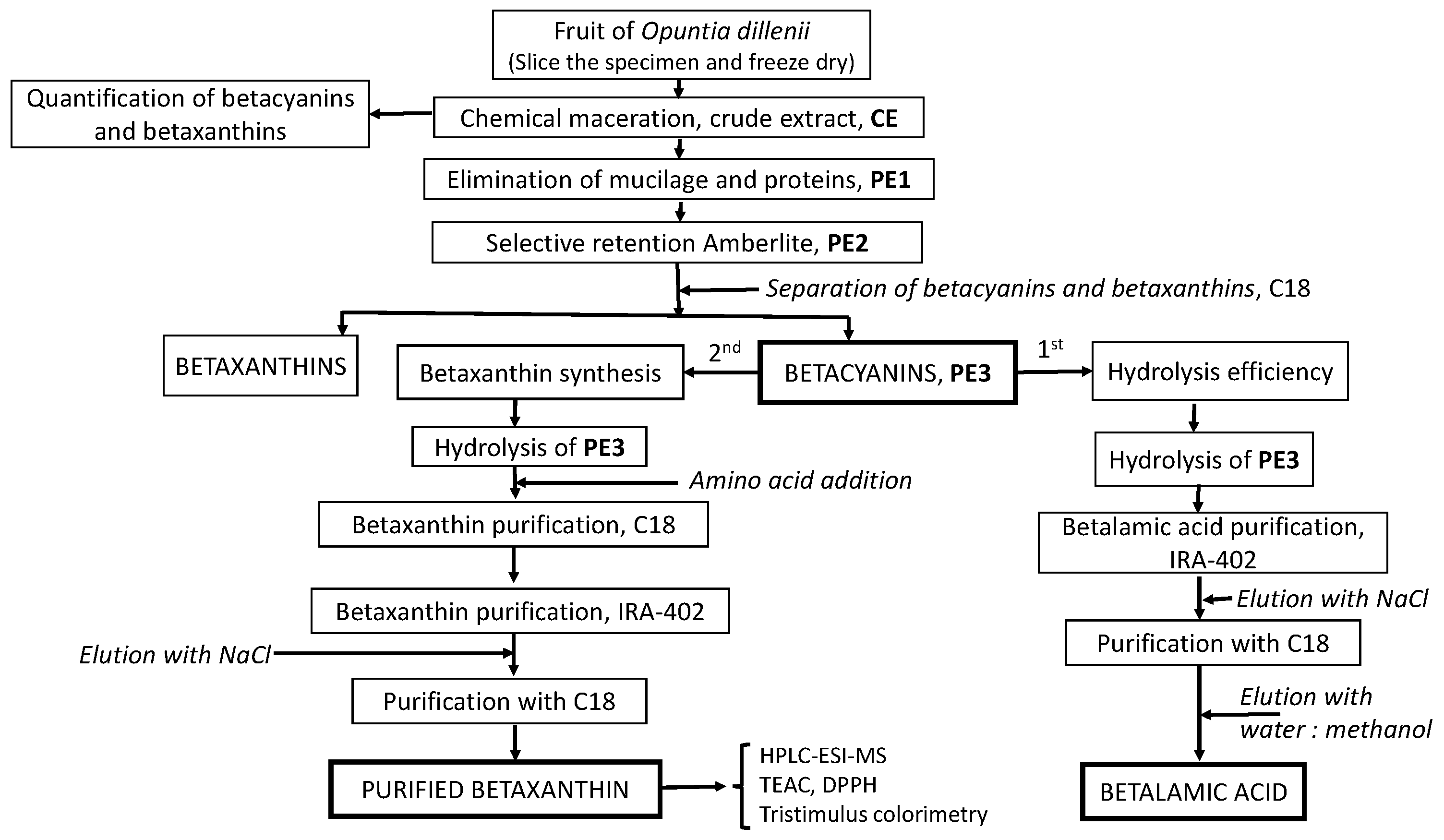
3.3. Preparation of Crude Extract (CE) and Purified Extract (PE1)
3.4. Separation of Polyphenols with Ambertite XAD-7 from PE1
3.5. Isolation of Betacyanins from PE2
3.6. Quantification of Betacyanins and Betaxanthins
3.7. Obtaining Betalamic Acid
3.8. Semisynthesis of ʟ−Amino Acid-Betaxanthins
3.9. Betaxanthins Purification
3.10. Analysis of Betaxanthins by UHPLC–ESI–MS
3.11. Determination of Antioxidant Capacity Equivalent to Trolox (TEAC)
3.12. Determination of Antioxidant Activity by 2,2-Diphenyl-1-Picrylhydrazyl (DPPH)
3.13. Colorimetric Measurements at Different pH
3.14. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Delgado-Vargas, F.; Jimenez, A.R.; Paredes-Lopez, O. Natural pigments: Carotenoids, anthocyanins, and betalains—Characteristics, biosynthesis, processing, and stability. Crit. Rev. Food Sci. 2000, 40, 173–289. [Google Scholar] [CrossRef]
- Lu, W.C.; Chiu, C.S.; Chan, Y.J.; Mulio, A.T.; Li, P.H. Recent research on different parts and extracts of Opuntia dillenii and its bioactive components, functional properties, and applications. Nutrients 2023, 15, 2962. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Rodríguez, P.; Guerrero-Rubio, M.A.; Henarejos-Escudero, P.; García-Carmona, F.; Gandía-Herrero, F. Health-promoting potential of betalains in vivo and their relevance as functional ingredients: A review. Trends Food Sci. Technol. 2022, 122, 66–82. [Google Scholar] [CrossRef]
- Shirazinia, R.; Rahimi, V.B.; Kehkhaie, A.R.; Sahebkar, A.; Rakhshandeh, H.; Askari, V.R. Opuntia dillenii: A forgotten plant with promising pharmacological properties. J. Pharmacopunct. 2019, 22, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Gandía-Herrero, F.; García-Carmona, F. Biosynthesis of betalains: Yellow and violet plant pigments. Trends Plant Sci. 2013, 18, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Betancourt, C.; Cejudo-Bastante, M.J.; Heredia, F.J.; Hurtado, N. Pigment composition and antioxidant capacity of betacyanins and betaxanthins fractions of Opuntia dillenii (Ker gawl) haw cactus fruit. Food Res. Int. 2017, 101, 173–179. [Google Scholar] [CrossRef]
- Sravan Kumar, S.; Singh Chauhan, A.; Giridhar, P. Nanoliposomal encapsulation mediated enhancement of betalain stability: Characterisation, storage stability and antioxidant activity of Basella rubra L. fruits for its applications in vegan gummy candies. Food Chem. 2020, 333, 127442. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, M.; Gamian, A.; Sroka, Z. Antiradical activity of beetroot (Beta vulgaris L.) betalains. Molecules 2021, 26, 2439. [Google Scholar] [CrossRef]
- Fernando, G.S.N.; Sergeeva, N.N.; Frutos, M.J.; Marshall, L.J.; Boesch, C. Novel approach for purification of major betalains using flash chromatography and comparison of radical scavenging and antioxidant activities. Food Chem. 2022, 385, 132632. [Google Scholar] [CrossRef]
- Pedreño, M.A.; Escribano, J. Correlation between antiradical activity and stability of betanine from Beta vulgaris L. roots under different pH, temperature and light conditions. J. Sci. Food Agric. 2001, 81, 627–631. [Google Scholar] [CrossRef]
- Escribano, J.; Pedreño, M.A.; García-Carmona, F.; Muñoz, R. Characterization of the antiradical activity of betalains from Beta Vulgaris L. roots. Phytochem. Anal. Int. J. Plant Chem. Biochem. Tech. 1998, 9, 124–127. [Google Scholar] [CrossRef]
- Gandía-Herrero, F.; Escribano, J.; García-Carmona, F. Biological activities of plant pigments betalains. Crit. Rev. Food Sci. Nutr. 2016, 56, 937–945. [Google Scholar] [CrossRef]
- Swarna, J.; Lokeswari, T.S.; Smita, M.; Ravindhran, R. Characterisation and determination of in vitro antioxidant potential of betalains from Talinum triangulare (jacq.) willd. Food Chem. 2013, 141, 4382–4390. [Google Scholar] [CrossRef]
- Carreón-Hidalgo, J.P.; Franco-Vásquez, D.C.; Gómez-Linton, D.R.; Pérez-Flores, L.J. Betalain plant sources, biosynthesis, extraction, stability enhancement methods, bioactivity, and applications. Food Res. Int. 2022, 151, 110821. [Google Scholar] [CrossRef] [PubMed]
- Abedi-Firoozjah, R.; Parandi, E.; Heydari, M.; Kolahdouz-Nasiri, A.; Bahraminejad, M.; Mohammadi, R.; Rouhi, M.; Garavand, F. Betalains as promising natural colorants in smart/active food packaging. Food Chem. 2023, 424, 136408. [Google Scholar] [CrossRef]
- Mosquera, N.; Cejudo-Bastante, M.J.; Heredia, F.J.; Hurtado, N. Identification of new betalains in separated betacyanin and betaxanthin fractions from Ulluco (Ullucus tuberosus Caldas) by HPLC-DAD-ESI-MS. Plant Foods Hum. Nutr. 2020, 75, 434–440. [Google Scholar] [CrossRef]
- Cejudo-Bastante, M.J.; Hurtado, N.; Heredia, F.J. Potential use of new Colombian sources of betalains. Colorimetric study of red prickly pear (Opuntia dillenii) extracts under different technological conditions. Food Res. Int. 2015, 71, 91–99. [Google Scholar] [CrossRef]
- Nestora, S.; Merlier, F.; Prost, E.; Haupt, K.; Rossi, C.; Tse Sum Bui, B. Solid-phase extraction of betanin and isobetanin from beetroot extracts using a dipicolinic acid molecularly imprinted polymer. J. Chromatogr. A 2016, 1465, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Rubio, M.A.; López-Llorca, R.; Henarejos-Escudero, P.; García-Carmona, F.; Gandía-Herrero, F. Scaled-up biotechnological production of individual betalains in a microbial system. Microb. Biotechnol. 2019, 12, 993–1002. [Google Scholar] [CrossRef] [PubMed]
- Schliemann, W.; Kobayashi, N.; Strack, D. The decisive step in betaxanthin biosynthesis is a spontaneous reaction. Plant Physiol. 1999, 119, 1217–1232. [Google Scholar] [CrossRef]
- Gandía-Herrero, F.; García-Carmona, F.; Escribano, J. Development of a protocol for the semisynthesis and purification of betaxanthins. Phytochem. Anal. 2006, 17, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Cabanes, J.; Gandía-Herrero, F.; Escribano, J.; García-Carmona, F.; Jiménez-Atiénzar, M. Fluorescent bioinspired protein labeling with betalamic acid. Derivatization and characterization of novel protein-betaxanthins. Dye. Pigment. 2016, 133, 458–466. [Google Scholar] [CrossRef]
- Cabanes, J.; Gandía-Herrero, F.; Escribano, J.; García-Carmona, F.; Jiménez-Atiénzar, M. One-step synthesis of betalains using a novel betalamic acid derivatized support. J. Agric. Food Chem. 2014, 62, 3776–3782. [Google Scholar] [CrossRef] [PubMed]
- Esteves, L.C.; Machado, C.O.; Gonçalves, L.C.P.; Cavalcante, V.F.; Obeid, G.; Correra, T.C.; Bastos, E.L. Structural effects on the antioxidant properties of amino acid betaxanthins. Antioxidants 2022, 11, 2259. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, K.K.; Bastos, E.L. Rationale on the high radical scavenging capacity of betalains. Antioxidants 2019, 8, 222. [Google Scholar] [CrossRef] [PubMed]
- Wawrzkiewicz, M.; Hubicki, Z. Anion exchange resins as effective sorbents for removal of acid, reactive, and direct dyes from textile wastewaters. In Ion Exchange—Studies and Applications; InTech: Houston, TX, USA, 2015; pp. 37–72. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Gandía-Herrero, F.; Escribano, J.; García-Carmona, F. Structural implications on color, fluorescence, and antiradical activity in betalains. Planta 2010, 232, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Rubio, M.A.; Hernández-García, S.; Escribano, J.; Jiménez-Atiénzar, M.; Cabanes, J.; García-Carmona, F.; Gandía-Herrero, F. Betalain health-promoting effects after ingestion in Caenorhabditis Elegans are mediated by DAF-16/FOXO and SKN-1/Nrf2 transcription factors. Food Chem. 2020, 330, 127228. [Google Scholar] [CrossRef] [PubMed]
- Herbach, K.M.; Stintzing, F.C.; Carle, R. Betalain stability and degradation—Structural and chromatic aspects. J. Food Sci. 2006, 71, R41–R50. [Google Scholar] [CrossRef]
- Fernández-Vázquez, R.; Stinco, C.M.; Hernanz, D.; Heredia, F.J.; Vicario, I.M. Colour training and colour differences thresholds in orange juice. Food Qual. Prefer. 2013, 30, 320–327. [Google Scholar] [CrossRef]
- Abu-Eittah, R.; Obaid, A.; Basahl, S.; Diefallah, E. Molecular orbital treatment of some amino acids. Bull. Chem. Soc. Jpn. 1988, 61, 2609–2613. [Google Scholar] [CrossRef]
- Wu, Q.; Fu, X.; Chen, Z.; Wang, H.; Wang, J.; Zhu, Z.; Zhu, G. Composition, Color Stability and Antioxidant Properties of Betalain-Based Extracts from Bracts of Bougainvillea. Molecules 2022, 27, 5120. [Google Scholar] [CrossRef] [PubMed]
- Solarte, N.; Cejudo-Bastante, M.J.; Hurtado, N.; Heredia, F.J. First accurate profiling of antioxidant anthocyanins and flavonols of Tibouchina urvilleana and Tibouchina mollis edible flowers aided by fractionation with amberlite XAD-7. Int. J. Food Sci. Technol. 2022, 57, 2416–2423. [Google Scholar] [CrossRef]
- Gandía-Herrero, F.; Escribano, J.; García-Carmona, F. Purification and antiradical properties of the structural unit of betalains. J. Nat. Prod. 2012, 75, 1030–1036. [Google Scholar] [CrossRef]
- Cai, Y.-Z.; Sun, M.; Corke, H. Characterization and application of betalain pigments from plants of the Amaranthaceae. Trends Food Sci. Technol. 2005, 16, 370–376. [Google Scholar] [CrossRef]
- Heredia, F.J.; Álvarez, C.; González-Miret, M.L.; Ramírez, A. CromaLab, Análisis de Color; Registro General de la Propiedad Intelectual: Sevilla, Spain, 2004.
- CIE 1964 10; Technical Report Colorimetry. Commission Internationale de L’Eclairage Central Bureau: Vienna, Austria, 2004.
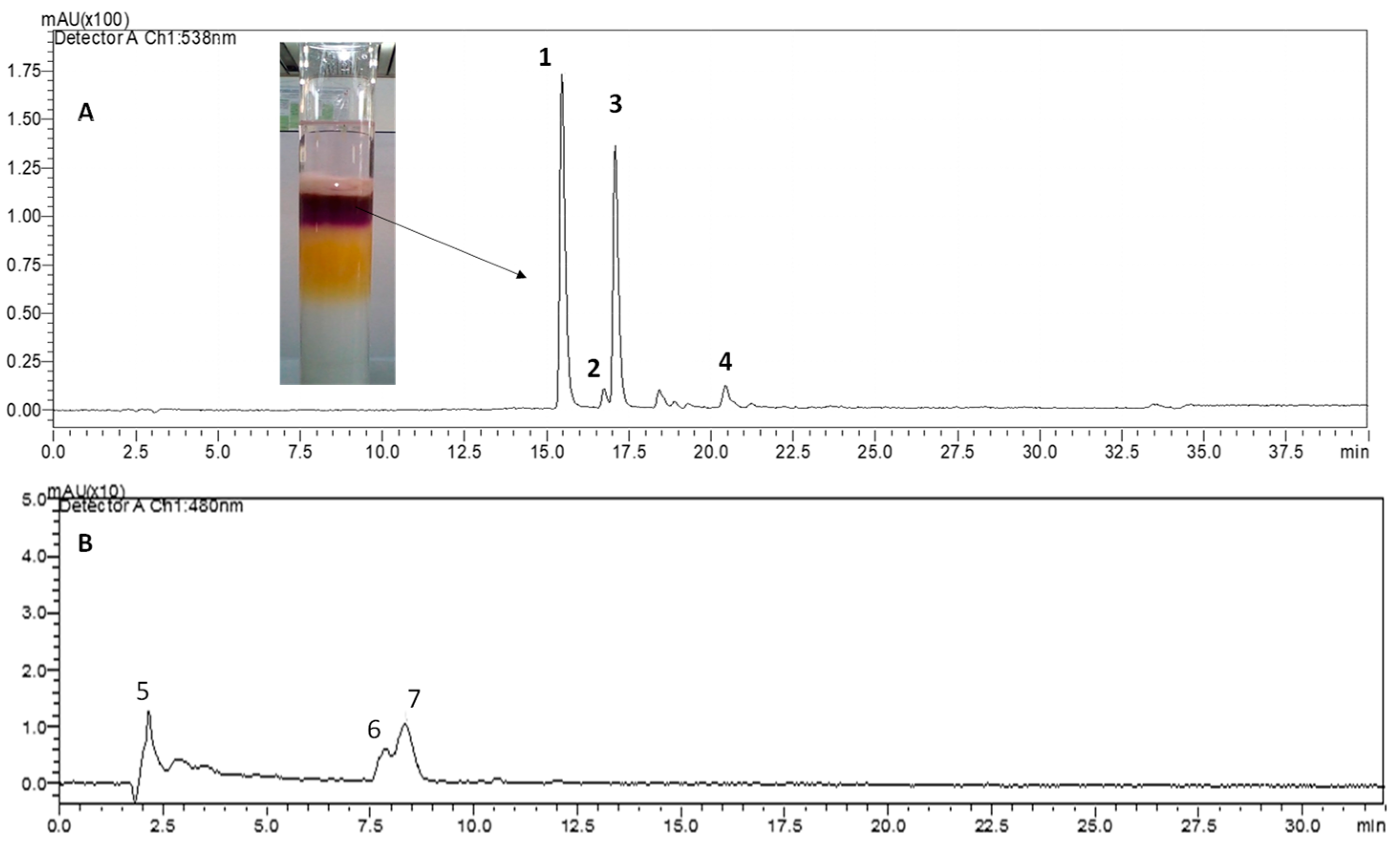
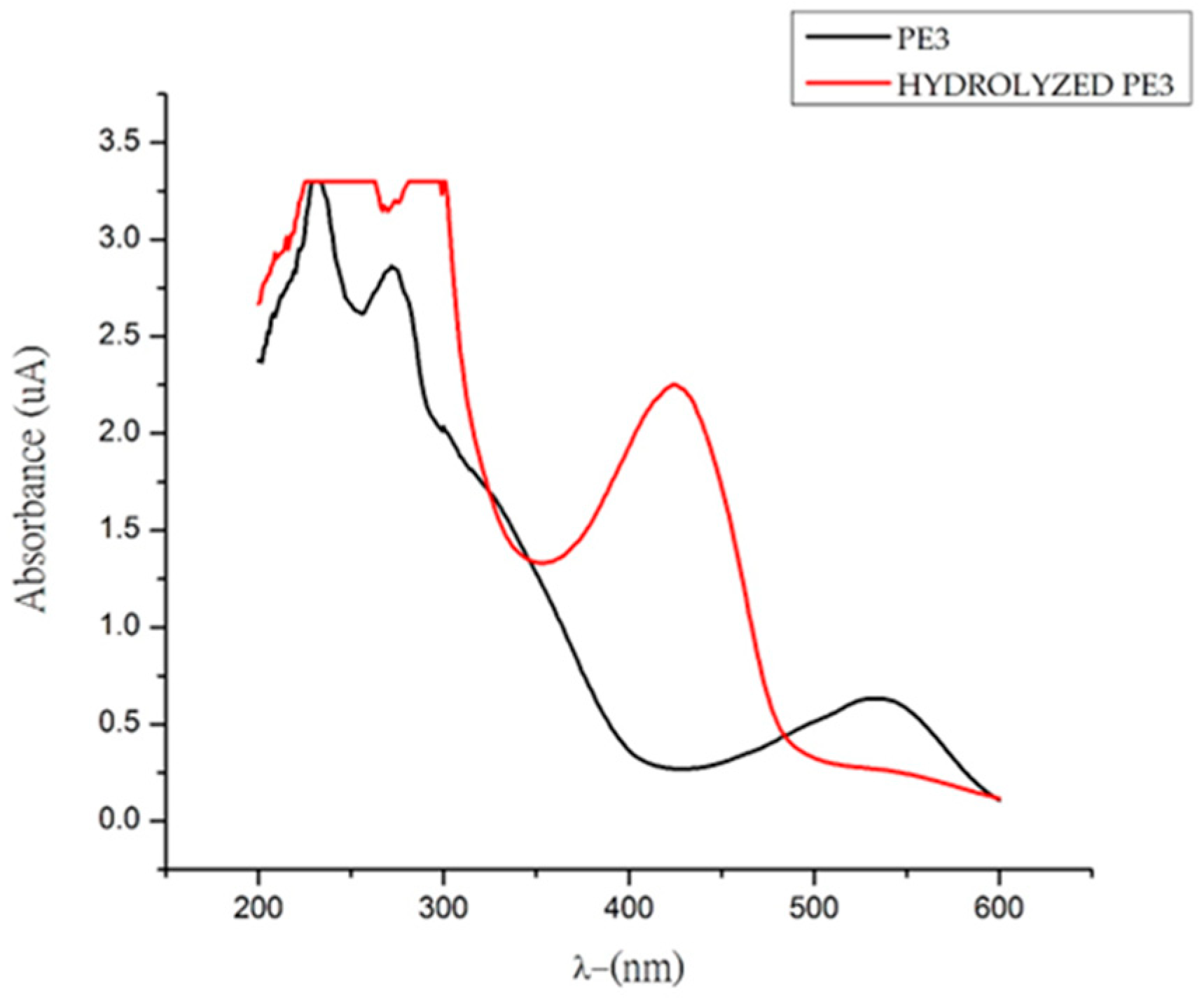
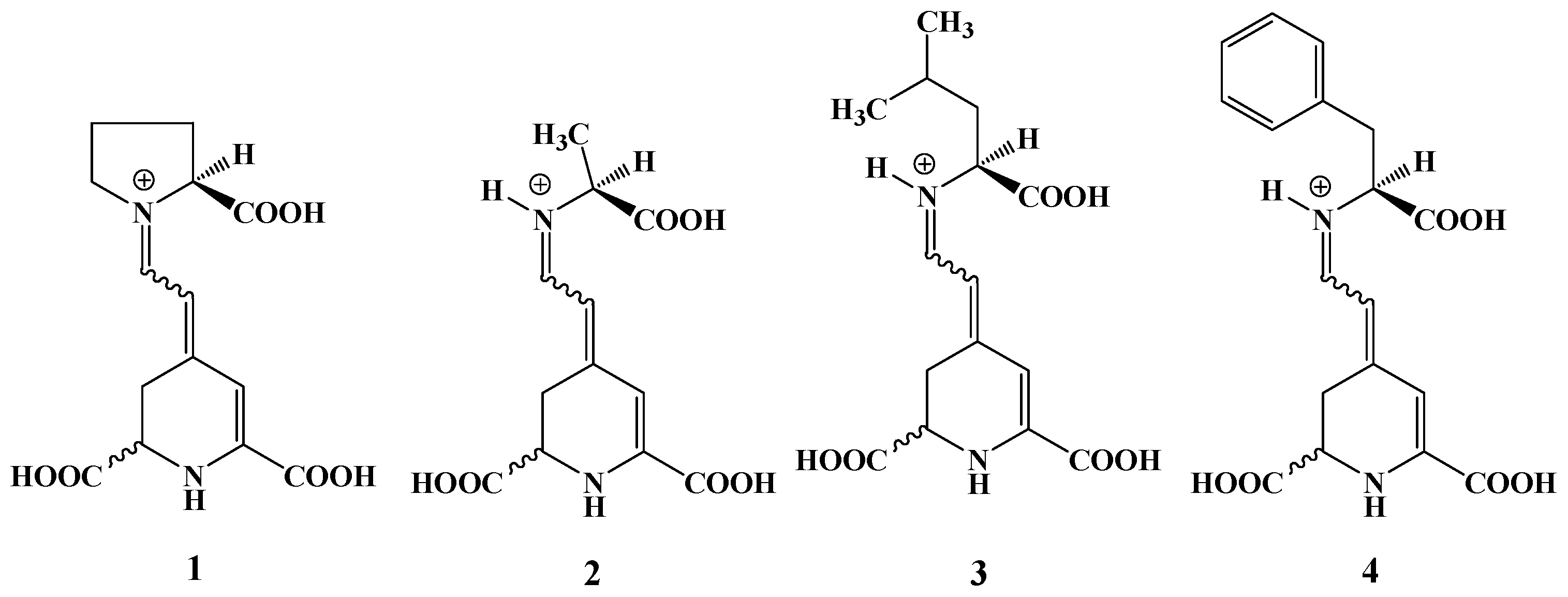

| Betaxanthin | λmax | Ratio Experimental/Theoretical | Yield (%) | % Area (HPLC) S,S/S,R |
|---|---|---|---|---|
| ʟ−ProBX | 484 | 0.275/0.361 | 76.2 | 66.4/33.6 |
| ʟ−AlaBx | 475 | 0.149/0.198 | 75.3 | 47.8/45.2 |
| ʟ−LeuBX | 475 | 0.267/0.379 | 70.1 | 72.7/27.3 |
| ʟ−PheBX | 477 | 0.482/0.697 | 69.2 | 86.3/13.7 |
| Betaxanthin | Molecular Formula | Rt (min) (S,S/S,R) | m/z [M + H]+ |
|---|---|---|---|
| ʟ−ProBX | C14H17N2O6 | 8.6/7.9 | 310.02547 |
| ʟ−AlaBx | C12H15N2O6 | 5.4/5.1 | 284.16047 |
| ʟ−LeuBX | C15H21N2O6 | 28.8/26.4 | 326.80460 |
| ʟ−PheBX | C18H19N2O6 | 32.0/29.5 | 360.12592 |
| Betaxanthin | TEAC (mmol Tx/mmol BX) | DPPH (% Inhibition) |
|---|---|---|
| ʟ−ProBX | 1.17 ± 0.07 a | 9.85 ± 1.48 a |
| ʟ−AlaBx | 2.27 ± 0.04 b | 16.22 ± 1.49 b |
| ʟ−LeuBX | 1.61 ± 0.03 c | 12.54 ± 1.36 c |
| ʟ−PheBX | 1.27 ± 0.03 a | 11.32 ± 1.97 c |
| Betalamic acid | 2.15 ± 0.62 b | 8.73 ± 1.39 a |
| Betaxanthins | pH | Storage | Visual Color | L* | a* | b* | C*ab | hab | ∆L* | ∆C*ab | ∆hab | ∆E*ab |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ʟ−Ala-Bx | 4 | BS | 93.8 ± 0.22 | −2.1 ± 0.03 | 37.2 ± 0.07 | 37.3 ± 0.10 | 93.2 ± 0.37 | 3.4 | 20.6 | 0.35 | 20.9 | |
| AS | 97.2 ± 0.38 | −1.0 ± 0.01 | 16.7 ± 0.04 | 16.7 ± 0.05 | 93.5 ± 0.28 | |||||||
| 6 | BS | 92.9 ± 0.15 | −1.2 ± 0.03 | 50.9 ± 0.11 | 51.0 ± 0.13 | 91.4 ± 0.18 | 1.5 | 7.9 | 0.50 | 8.1 | ||
| AS | 94.3 ± 0.09 | −1.4 ± 0.05 | 43.0 ± 0.08 | 43.1 ± 0.07 | 91.8 ± 0.13 | |||||||
| ʟ−Leu-Bx | 4 | BS | 94.2 ± 0.13 | −1.5 ± 0.02 | 35.4 ± 0.07 | 35.5 ± 0.09 | 92.4 ± 0.19 | 2.9 | 25.5 | 5.6 | 25.7 | |
| AS | 97.0 ± 0.11 | 0.55 ± 0.00 | 10.0 ± 0.04 | 10.0 ± 0.08 | 86.9 ± 0.16 | |||||||
| 6 | BS | 93.1 ± 0.08 | −0.7 ± 0.01 | 42.2 ± 0.11 | 42.2 ± 0.11 | 91.0 ± 0.13 | 2.0 | 15.3 | 1.1 | 15.4 | ||
| AS | 95.1 ± 0.21 | −1.0 ± 0.01 | 26.9 ± 0.06 | 26.9 ± 0.04 | 92.1 ± 0.11 | |||||||
| ʟ−Phe-Bx | 4 | BS | 97.3 ± 0.19 | −1.2 ± 0.02 | 16.3 ± 0.08 | 16.4 ± 0.05 | 94.4 ± 0.10 | 1.3 | 10.5 | 2.0 | 10.6 | |
| AS | 98.6 ± 0.27 | −0.7 ± 0.00 | 5.9 ± 0.05 | 5.9 ± 0.04 | 96.3 ± 0.21 | |||||||
| 6 | BS | 92.3 ± 0.15 | −0.2 ± 0.00 | 43.5 ± 0.09 | 43.5 ± 0.09 | 90.3 ± 0.14 | 42.4 | 30.8 | 1.6 | 52.4 | ||
| AS | 49.9 ± 0.08 | 0.29 ± 0.01 | 12.7 ± 0.10 | 12.7 ± 0.03 | 88.7 ± 0.21 | |||||||
| ʟ−Pro-Bx | 4 | BS | 93.6 ± 0.09 | 3.3 ± 0.03 | 43.9 ± 0.15 | 44.1 ± 0.09 | 85.7 ± 0.21 | 2.6 | 18.8 | 0.72 | 18.9 | |
| AS | 96.2 ± 0.20 | 1.6 ± 0.00 | 25.3 ± 0.07 | 25.3 ± 0.03 | 86.4 ± 0.09 | |||||||
| 6 | BS | 93.3 ± 0.16 | 3.7 ± 0.02 | 47.4 ± 0.08 | 47.5 ± 0.07 | 85.5 ± 0.07 | 0.27 | 0.44 | 0.46 | 0.64 | ||
| AS | 93.6 ± 0.11 | 4.1 ± 0.04 | 47.8 ± 0.08 | 48.0 ± 0.08 | 85.1 ± 0.19 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruz, S.; Checa, N.; Tovar, H.; Cejudo-Bastante, M.J.; Heredia, F.J.; Hurtado, N. Semisynthesis of Betaxanthins from Purified Betacyanin of Opuntia dillenii sp.: Color Stability and Antiradical Capacity. Molecules 2024, 29, 2116. https://doi.org/10.3390/molecules29092116
Cruz S, Checa N, Tovar H, Cejudo-Bastante MJ, Heredia FJ, Hurtado N. Semisynthesis of Betaxanthins from Purified Betacyanin of Opuntia dillenii sp.: Color Stability and Antiradical Capacity. Molecules. 2024; 29(9):2116. https://doi.org/10.3390/molecules29092116
Chicago/Turabian StyleCruz, Silvia, Neyder Checa, Hugo Tovar, María Jesús Cejudo-Bastante, Francisco J. Heredia, and Nelson Hurtado. 2024. "Semisynthesis of Betaxanthins from Purified Betacyanin of Opuntia dillenii sp.: Color Stability and Antiradical Capacity" Molecules 29, no. 9: 2116. https://doi.org/10.3390/molecules29092116
APA StyleCruz, S., Checa, N., Tovar, H., Cejudo-Bastante, M. J., Heredia, F. J., & Hurtado, N. (2024). Semisynthesis of Betaxanthins from Purified Betacyanin of Opuntia dillenii sp.: Color Stability and Antiradical Capacity. Molecules, 29(9), 2116. https://doi.org/10.3390/molecules29092116







