Recovery of Cembratrien-Diols from Waste Tobacco (Nicotiana tabacum L.) Flowers by Microwave-Assisted Deep Eutectic Solvent Extraction: Optimization, Separation, and In Vitro Bioactivity
Abstract
1. Introduction
2. Results and Discussion
2.1. Evaluation of DES Extraction Efficiency
2.2. Optimization of DES Extraction Using Single-Factor Experiments
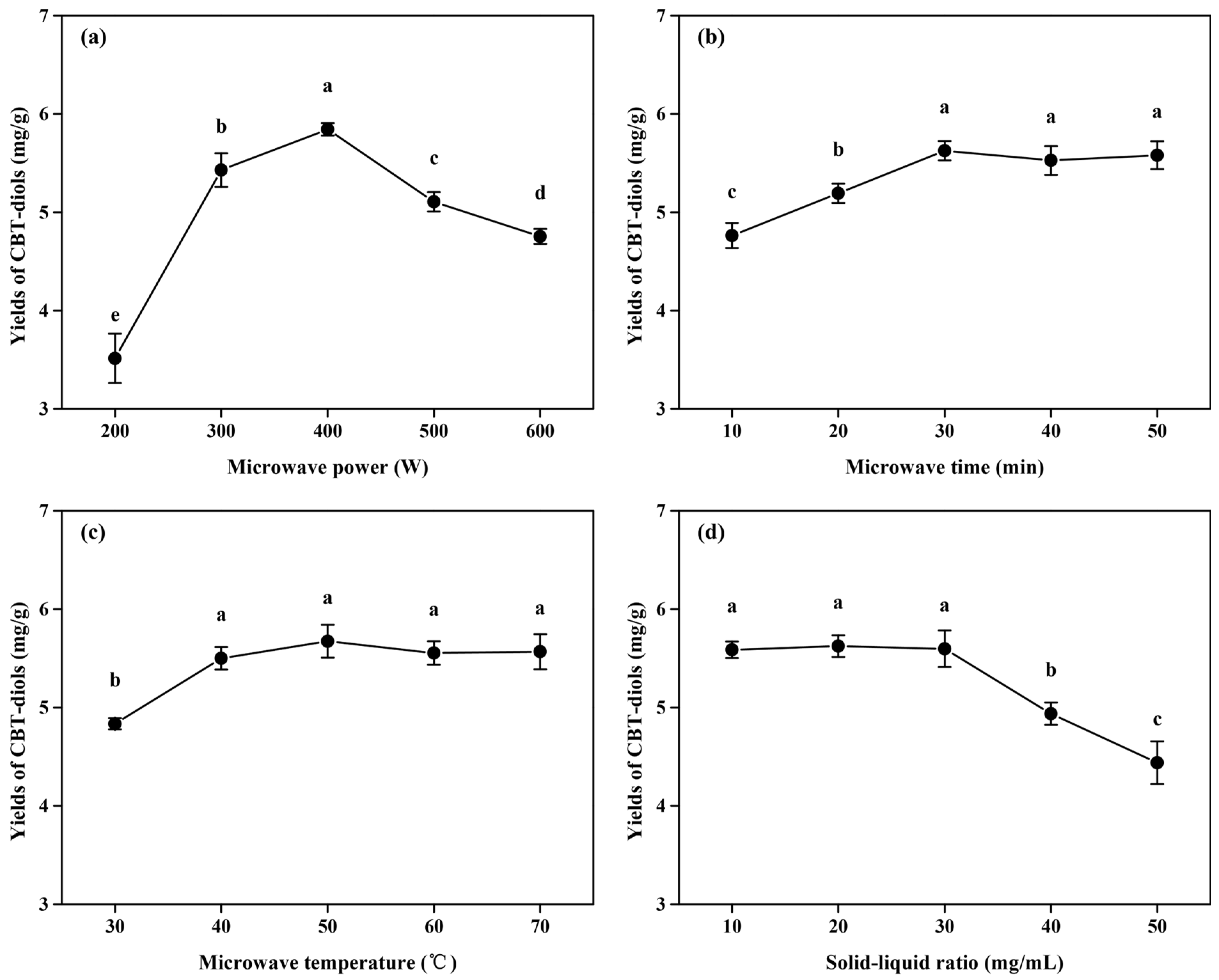
2.2.1. Effect of Microwave Power on the Yields of CBT-Diols from WTFs
2.2.2. Effect of Microwave Time on the Yields of CBT-Diols from WTFs
2.2.3. Effect of Microwave Temperature on the Yields of CBT-Diols from WTFs
2.2.4. Effect of Solid/Liquid Ratio on the Yields of CBT-Diols from WTFs
2.3. Optimization of DES Extraction Using the Response Surface Methodology
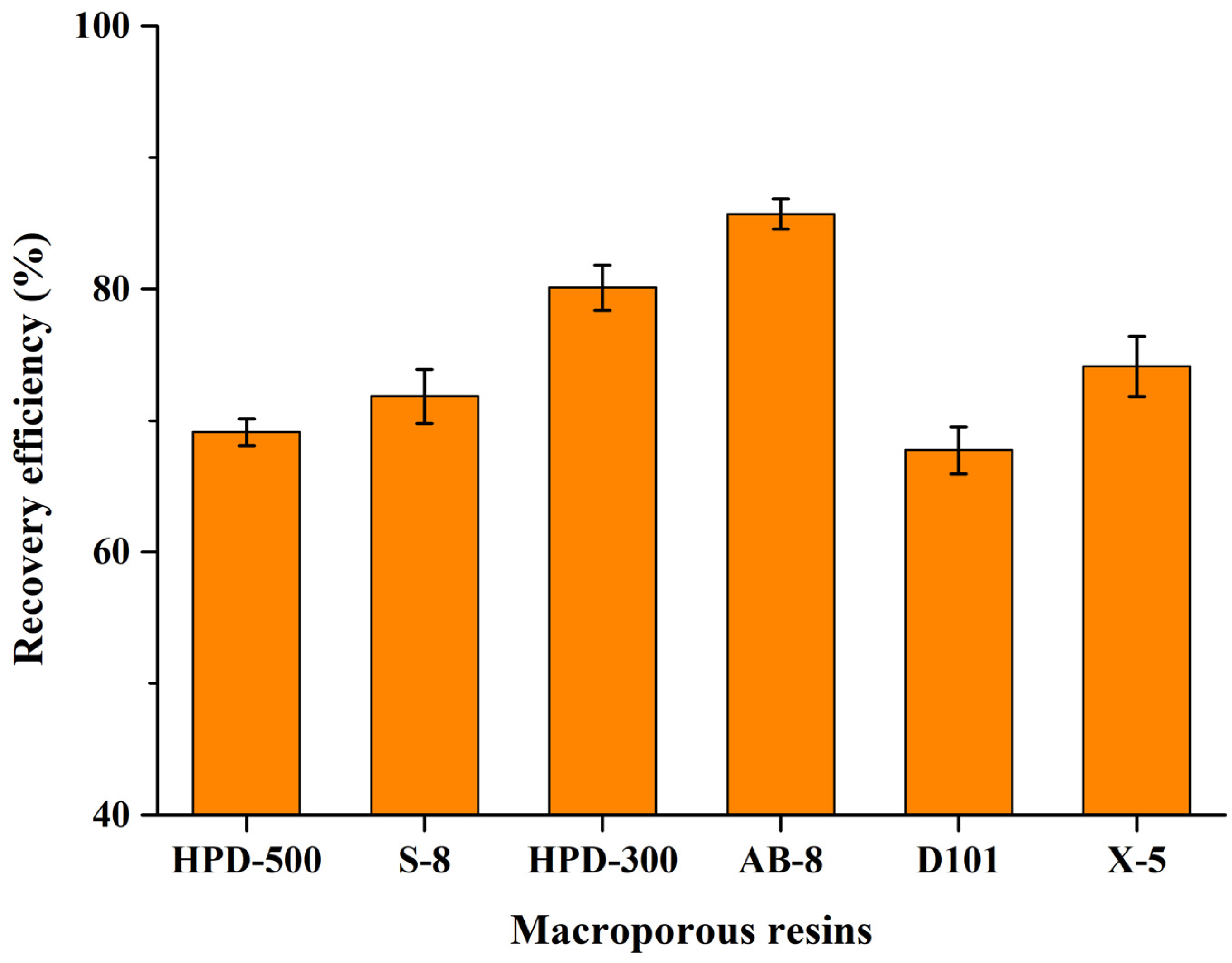
2.4. Recovery of CBT-Diols from DES Extraction Systems
2.5. In Vitro Bioactivity of CBT-Diols Extracted from WTFs Using DESs
2.5.1. Antimicrobial Activity
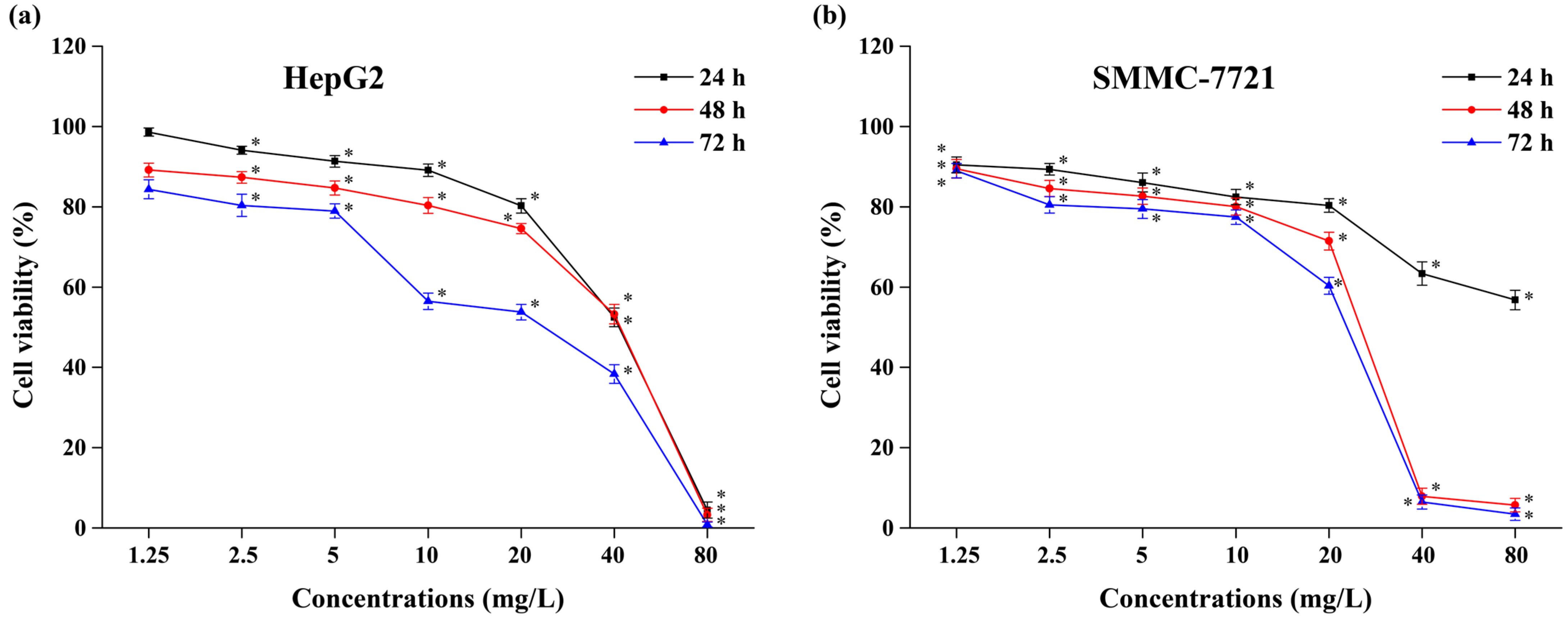
2.5.2. Antitumor Activity
3. Materials and Methods
3.1. Materials and Reagents
3.2. Preparation and Characterization of DESs
3.3. Screening of DESs for the Microwave-Assisted Extraction of CBT-Diols from WTFs
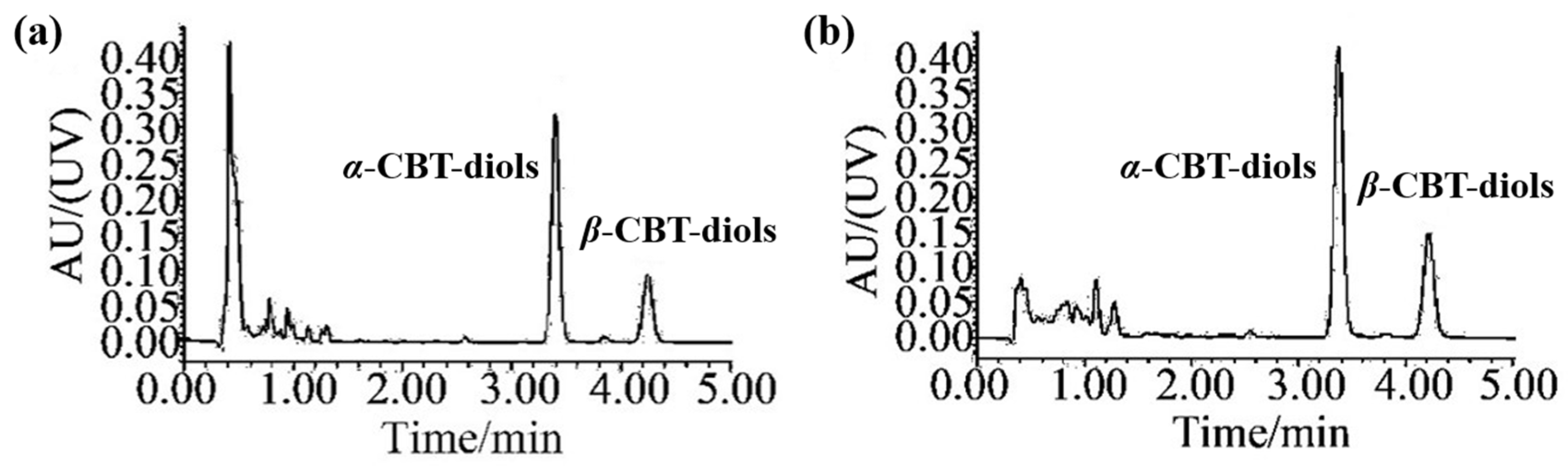
3.4. Quantification of CBT-Diols Extracted from WTFs Using HPLC-UV Analysis
3.5. Single-Factor Optimization for the Extraction of CBT-Diols from WTFs
3.6. Response Surface Methodology Optimization of the Extraction of CBT-Diols from WTFs
3.7. Recovery of CBT-Diols from DES Extracts Using Macroporous Resins
3.8. Evaluation of In Vitro Bioactivity of CBT-Diols Extracted from WTFs Using DESs
3.8.1. Evaluation of Antimicrobial Activity
3.8.2. Evaluation of Antitumor Activity
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Menetrez, M.L.; Spurr, H.W.; Danehower, D.A.; Lawson, D.R. Influence of tobacco leaf surface chemicals on germination of Peronospora tabacina adam sporangia. J. Chem. Ecol. 1990, 16, 1565–1576. [Google Scholar] [CrossRef]
- Yang, Q.; Wang, J.; Zhang, P.; Xie, S.; Yuan, X.; Hou, X.; Yan, N.; Fang, Y.; Du, Y. In vitro and in vivo antifungal activity and preliminary mechanism of cembratrien-diols against Botrytis cinerea. Ind. Crops Prod. 2020, 154, 112745. [Google Scholar] [CrossRef]
- Sayed, K.A.; Sylvester, P.W. Biocatalytic and semisynthetic studies of the anticancer tobacco cembranoids. Expert Opin. Investig. Drugs 2007, 16, 877–887. [Google Scholar] [CrossRef]
- Yan, N.; Du, Y.; Liu, X.; Zhang, H.; Liu, Y.; Zhang, P.; Gong, D.; Zhang, Z. Chemical structures, biosynthesis, bioactivities, biocatalysis and semisynthesis of tobacco cembranoids: An overview. Ind. Crops Prod. 2016, 83, 66–80. [Google Scholar] [CrossRef]
- Hou, P.; Zeng, Y.; Ma, B.; Bi, K.; Chen, X. A new cytotoxic cembrane diterpene from the roots of Euphorbia pekinensis Rupr. Fitoterapia 2013, 90, 10–13. [Google Scholar] [CrossRef]
- Ferchmin, P.A.; Lukas, R.J.; Hann, R.M.; Fryer, J.D.; Eaton, J.B.; Pagán, O.R.; Rodríguez, A.D.; Nicolau, Y.; Rosado, M.; Cortés, S.; et al. Tobacco cembranoids block behavioral sensitization to nicotine and inhibit neuronal acetylcholine receptor function. J. Neurosci. Res. 2001, 64, 18–25. [Google Scholar] [CrossRef]
- Hany, N.B.; Mohammad, A.K.; John, C.W.; Emad, M.; Khalid, A. Bioactive natural, biocatalytic, and semisynthetic tobacco cembranoids. Planta Medica 2011, 77, 467–476. [Google Scholar] [CrossRef]
- Ferchmin, P.A.; Myrna, A.; Rebeca, R.S.; Janaina, A.; Joyce, V.-R.; Brenda, C.; Marimeé, C.; Wilmarie, T.-R.; Annabell, S.; Antonio, H.M.; et al. 4R-cembranoid protects against diisopropylfluorophosphate-mediated neurodegeneration. NeuroToxicology 2014, 44, 80–90. [Google Scholar] [CrossRef]
- Alena, S.; Šeregelj, V.; Bernardo, D.R.; Lato, P.; Cvetanović, A.; Mišan, A.; Marrucho, I. Recovery of β-carotene from pumpkin using switchable natural deep eutectic solvents. Ultrason. Sonochem. 2021, 76, 105638. [Google Scholar] [CrossRef]
- You, T.; Sun, D.; Zhu, Z. Development of natural deep eutectic solvents (DESs) as anti-freezing agents for the frozen food industry: Water-tailoring effects, anti-freezing mechanisms and applications. Food Chem. 2022, 371, 131150. [Google Scholar] [CrossRef]
- Alexandre, P.; Rita, C.; Ivo, A.; Marta, M.; Rui, L.R.; Ana, R.C.D. Natural deep eutectic solvents-solvents for the 21st Century. ACS Sustain. Chem. Eng. 2014, 5, 1063–1071. [Google Scholar] [CrossRef]
- Dai, Y.; van Spronsen, J.; Witkamp, G.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef]
- Shang, X.C.; Dou, Y.; Zhang, Y.; Tan, J.; Liu, X.; Zhang, Z. Tailor-made natural deep eutectic solvents for green extraction of isoflavones from chickpea (Cicer arietinum L.) sprouts. Ind. Crops Prod. 2019, 140, 111724. [Google Scholar] [CrossRef]
- Dai, Y.; Witkamp, G.; Verpoorte, R.; Choi, Y.H. Tailoring properties of natural deep eutectic solvents with water to facilitate their applications. Food Chem. 2015, 187, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Oscar, Z.; Ilkay, K. Greener extraction of anthocyanins and antioxidant activity from blackberry (Rubus spp.) using natural deep eutectic solvents. LWT 2022, 158, 113184. [Google Scholar] [CrossRef]
- Fu, X.; Wang, D.; Belwal, T.; Xie, J.; Xu, Y.; Li, L.; Zou, L.; Zhang, L.; Luo, Z. Natural deep eutectic solvent enhanced pulse-ultrasonication assisted extraction as a multi-stability protective and efficient green strategy to extract anthocyanin from blueberry pomace. LWT 2021, 144, 111220. [Google Scholar] [CrossRef]
- Rubiya, R.; Sajad, M.W.; Shaziya, M.; Masoodi, F.A.; Mohd, M.D. Green extraction of bioactive compounds from apple pomace by ultrasound assisted natural deep eutectic solvent extraction: Optimisation, comparison and bioactivity. Food Chem. 2023, 398, 133871. [Google Scholar] [CrossRef]
- Dai, Y.; Witkamp, G.J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as a new extraction media for phenolic metabolites in Carthamus tinctorius L. Anal. Chem. 2013, 85, 6272–6278. [Google Scholar] [CrossRef]
- Guo, N.; Kou, P.; Jiang, Y.; Wang, L.; Niu, L.J.; Liu, Z.M.; Fu, Y.J. Natural deep eutectic solvents couple with integrative extraction technique as an effective approach for mulberry anthocyanin extraction. Food Chem. 2019, 296, 78–85. [Google Scholar] [CrossRef]
- Liu, Y.; Li, J.; Fu, R.; Zhang, L.; Wang, D.; Wang, S. Enhanced extraction of natural pigments from Curcuma longa L. using natural deep eutectic solvents. Ind. Crops Prod. 2019, 140, 111620. [Google Scholar] [CrossRef]
- Wang, J.; Jing, W.; Tian, H.; Liu, M.; Yan, H.; Bi, W.; David, D.Y.C. Investigation of deep eutectic solvent-based microwave-assisted extraction and efficient recovery of natural products. ACS Sustain. Chem. Eng. 2020, 8, 12080–12088. [Google Scholar] [CrossRef]
- Tong, X.; Yang, J.; Zhao, Y.; Wan, H.; He, Y.; Zhang, L.; Wan, H.; Li, C. Greener extraction process and enhanced in vivo bioavailability of bioactive components from Carthamus tinctorius L. by natural deep eutectic solvents. Food Chem. 2021, 348, 129090. [Google Scholar] [CrossRef] [PubMed]
- Zuo, J.; Ma, P.; Geng, S.; Kong, Y.; Li, X.; Fan, Z.; Zhang, Y.; Dong, A.; Zhou, Q. Optimization of the extraction process of flavonoids from Trollius ledebouri with natural deep eutectic solvents. J. Sep. Sci. 2022, 45, 717–727. [Google Scholar] [CrossRef]
- Jurić, T.; Mićić, N.; Potkonjak, A.; Milanov, D.; Dodić, J.; Trivunović, Z.; Popović, B.M. The evaluation of phenolic content, in vitro antioxidant and antibacterial activity of Mentha piperita extracts obtained by natural deep eutectic solvents. Food Chem. 2021, 362, 130226. [Google Scholar] [CrossRef]
- Mohammed, A.; Yavuz, Y.; Gu, L. Use natural deep eutectic solvents as efficient green reagents to extract procyanidins and anthocyanins from cranberry pomace and predictive modeling by RSM and artificial neural networking. Sep. Purif. Technol. 2021, 255, 117720. [Google Scholar] [CrossRef]
- Choi, Y.H.; Verpoorte, R. Green solvents for the extraction of bioactive compounds from natural products using ionic liquids and deep eutectic solvents. Curr. Opin. Food Sci. 2019, 26, 87–93. [Google Scholar] [CrossRef]
- Chen, Z.; Wu, K.; Zhu, W.; Wang, Y.; Su, C.; Yi, F. Chemical compositions and bioactivities of essential oil from perilla leaf (Perillae Folium) obtained by ultrasonic-assisted hydro-distillation with natural deep eutectic solvents. Food Chem. 2022, 375, 131834. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.C.; Chu, D.; Zhang, J.; Zheng, Y.; Li, Y. Microwave-assisted extraction, partial purification and biological activity in vitro of polysaccharides from bladder-wrack (Fucus vesiculosus) by using deep eutectic solvents. Sep. Purif. Technol. 2021, 259, 118169. [Google Scholar] [CrossRef]
- Sylwia, B.; Jakub, A. Evaluation of new natural deep eutectic solvents for the extraction of isoflavones from soy products. Talanta 2017, 168, 329–335. [Google Scholar] [CrossRef]
- Kristina, R.; Natka, Ć.; Višnja, G.; Marina, C.; Marina, T.; Karin, K.; Ivana, R. Natural deep eutectic solvents as beneficial extractants for enhancement of plant extracts bioactivity. LWT 2016, 73, 45–51. [Google Scholar] [CrossRef]
- Hossein, M.; Seyed, M.S.; Fatemeh, E.; Ali, A. Optimization of ultrasound-assisted extraction of Moringa peregrina oil with response surface methodology and comparison with Soxhlet method. Ind. Crops Prod. 2019, 131, 106–116. [Google Scholar] [CrossRef]
- José, C.M.; Beatriz, G.; Inmaculada, R.; Encarnación, R.; Mladen, B.; Šic Žlabur, J.; Eulogio, C. Optimization of ultrasound-assisted extraction of biomass from olive trees using response surface methodology. Ultrason. Sonochem. 2019, 51, 487–495. [Google Scholar] [CrossRef]
- Puntipa, P.; Satoshi, I.; Ubonrat, S. Response surface methodology for optimization of cinnamon essential oil nanoemulsion with improved stability and antifungal activity. Ultrason. Sonochem. 2020, 60, 104604. [Google Scholar] [CrossRef]
- Jalil, J.; Kamyar, Y. Optimization of heavy metal biosorption onto freshwater algae (Chlorella coloniales) using response surface methodology (RSM). Chemosphere 2019, 217, 447–455. [Google Scholar] [CrossRef]
- Chen, Y.; Xue, Y. Optimization of microwave assisted extraction, chemical characterization and antitumor activities of polysaccharides from porphyra haitanensis. Carbohydr. Polym. 2019, 206, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Dong, Y.; Chen, G.; Hu, Q. Extraction, purification, characterization and antitumor activity of polysaccharides from Ganoderma lucidum. Carbohydr. Polym. 2010, 80, 783–789. [Google Scholar] [CrossRef]
- Zhang, B.; Li, Y.; Zhang, F.; Robert, J.L.; Zeng, G.; Zhang, A. Extraction, structure and bioactivities of the polysaccharides from Pleurotus eryngii: A review. Int. J. Biol. Macromol. 2020, 150, 1342–1347. [Google Scholar] [CrossRef] [PubMed]
- Liao, D.; Cheng, C.; Liu, J.; Zhao, L.; Huang, D.; Chen, G. Characterization and antitumor activities of polysaccharides obtained from ginger (Zingiber officinale) by different extraction methods. Int. J. Biol. Macromol. 2020, 152, 894–903. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, S.; Dang, S.; Han, S.; Yun, C.; Wang, W.; Wang, H. Optimized ultrasound-assisted extraction of total polyphenols from Empetrum nigrum and its bioactivities. J. Chromatogr. B 2021, 1173, 122699. [Google Scholar] [CrossRef]
- Saito, Y.; Takizawa, H.; Konishi, S. Identification of cembratriene-4,6-diol as antitumor-promoting agent from cigarette smoke condensate. Carcinogenesis 1985, 6, 1189–1194. [Google Scholar] [CrossRef]
- Palos-Hernández, A.; Yolanda, G.F.M.; José, E.B.; José, L.P.I.; Ana, M.G.P. Obtaining green extracts rich in phenolic compounds from underexploited food by-products using natural deep eutectic solvents. Opportunities and challenges. Sustain. Chem. Pharm. 2022, 29, 100773. [Google Scholar] [CrossRef]
- Shang, X.C.; Zhang, M.; Hu, J.; Zhang, Y.; Yang, L.; Hou, X. Chemical compositions, extraction optimizations, and in vitro bioactivities of flavonoids from perilla leaves (Perillae folium) by microwave-assisted natural deep eutectic solvents. Antioxidants 2023, 12, 104. [Google Scholar] [CrossRef] [PubMed]


| Run | Variables | Yields (mg/g) | |||
|---|---|---|---|---|---|
| A (W) | B (min) | C (°C) | D (mg/mL) | ||
| 1 | 400 | 30 | 40 | 30 | 5.99 |
| 2 | 400 | 40 | 50 | 30 | 5.49 |
| 3 | 300 | 30 | 50 | 30 | 4.91 |
| 4 | 300 | 40 | 40 | 30 | 5.09 |
| 5 | 500 | 30 | 30 | 30 | 4.94 |
| 6 | 400 | 40 | 40 | 40 | 5.77 |
| 7 | 400 | 30 | 40 | 30 | 6.06 |
| 8 | 400 | 20 | 30 | 30 | 5.53 |
| 9 | 400 | 30 | 40 | 30 | 6.02 |
| 10 | 400 | 30 | 30 | 20 | 5.97 |
| 11 | 500 | 30 | 40 | 20 | 5.88 |
| 12 | 400 | 20 | 40 | 20 | 5.87 |
| 13 | 400 | 30 | 40 | 30 | 5.97 |
| 14 | 500 | 20 | 40 | 30 | 5.56 |
| 15 | 400 | 30 | 50 | 20 | 5.98 |
| 16 | 300 | 30 | 30 | 30 | 4.91 |
| 17 | 500 | 30 | 50 | 30 | 5.08 |
| 18 | 400 | 20 | 40 | 40 | 5.79 |
| 19 | 400 | 20 | 50 | 30 | 5.93 |
| 20 | 300 | 30 | 40 | 20 | 4.91 |
| 21 | 400 | 40 | 30 | 30 | 5.73 |
| 22 | 400 | 30 | 50 | 40 | 5.72 |
| 23 | 400 | 30 | 30 | 40 | 5.56 |
| 24 | 300 | 30 | 40 | 40 | 5.04 |
| 25 | 300 | 20 | 40 | 30 | 5.38 |
| 26 | 400 | 30 | 40 | 30 | 6.09 |
| 27 | 500 | 40 | 40 | 30 | 5.24 |
| 28 | 400 | 40 | 40 | 20 | 6.13 |
| 29 | 500 | 30 | 40 | 40 | 5.11 |
| Source | Sum of Squares | Degrees of Freedom | Mean Square | F Value | p-Value |
|---|---|---|---|---|---|
| Model | 4.67 | 14 | 0.3334 | 15.8 | <0.0001 |
| A | 0.2054 | 1 | 0.2054 | 9.74 | 0.0075 ** |
| B | 0.031 | 1 | 0.031 | 1.47 | 0.2455 |
| C | 0.0184 | 1 | 0.0184 | 0.8725 | 0.3661 |
| D | 0.2552 | 1 | 0.2552 | 12.1 | 0.0037 ** |
| AB | 0.0002 | 1 | 0.0002 | 0.0107 | 0.9192 |
| AC | 0.0049 | 1 | 0.0049 | 0.2322 | 0.6373 |
| AD | 0.2025 | 1 | 0.2025 | 9.6 | 0.0079 ** |
| BC | 0.1024 | 1 | 0.1024 | 4.85 | 0.0448 * |
| BD | 0.0196 | 1 | 0.0196 | 0.929 | 0.3515 |
| CD | 0.0056 | 1 | 0.0056 | 0.2666 | 0.6137 |
| A2 | 3.52 | 1 | 3.52 | 166.87 | <0.0001 ** |
| B2 | 0.0191 | 1 | 0.0191 | 0.9048 | 0.3576 |
| C2 | 0.4879 | 1 | 0.4879 | 23.12 | 0.0003 ** |
| D2 | 0.0046 | 1 | 0.0046 | 0.22 | 0.6463 |
| Residual | 0.2954 | 14 | 0.0211 | ||
| Lack of Fit | 0.2857 | 10 | 0.0286 | 11.76 | 0.149 |
| Pure error | 0.0097 | 4 | 0.0024 | ||
| Corr. total | 4.96 | 28 |
| Samples | Diameter of Inhibition Zone (mm) | ||||
|---|---|---|---|---|---|
| Salmonella | S. aureus | E. coli | B. subtilis | P. aeruginosa | |
| CBT-diols | 8.35 ± 0.57 | 28.63 ± 0.55 | 14.55 ± 0.69 | 15.02 ± 0.30 | 21.28 ± 0.63 |
| Solvent control | - | - | - | - | - |
| Positive control | 17.08 ± 0.77 | 32.17 ± 0.96 | 24.89 ± 0.52 | 24.37 ± 0.51 | 24.12 ± 0.65 |
| Abbreviation | Composition | Molar Ratio | Density a (g/cm3) | Viscosity a (mPa/s) | |
|---|---|---|---|---|---|
| Component 1 | Component 2 | ||||
| DES-1 | Choline chloride | Glycerol | 1:2 | 1.18 ± 0.03 | 271.3 ± 1.8 |
| DES-2 | Choline chloride | Urea | 1:2 | 1.08 ± 0.05 | 153.3 ± 0.9 |
| DES-3 | Choline chloride | Lactic acid | 1:3 | 1.14 ± 0.07 | 105.5 ± 1.6 |
| DES-4 | Choline chloride | Citric acid | 1:1 | 1.29 ± 0.04 | 128.9 ± 1.1 |
| DES-5 | Choline chloride | d-(+)-Glucose | 3:2 | 1.28 ± 0.06 | 164.8 ± 1.4 |
| DES-6 | Urea | Glycerol | 1:2 | 1.13 ± 0.03 | 143.6 ± 1.7 |
| DES-7 | Tartaric acid | d-(+)-Glucose | 1:1 | 1.03 ± 0.05 | 129.6 ± 1.0 |
| Variables | Symbols | Coded Levels | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| Microwave power (W) | A | 300 | 400 | 500 |
| Microwave time (min) | B | 20 | 30 | 40 |
| Microwave temperature (°C) | C | 30 | 40 | 50 |
| Solid/liquid ratio (mg/mL) | D | 20 | 30 | 40 |
| Macroporous Resins | Polarity | Specific Saturated Adsorption Capacity (mg/g) | Specific Surface Area (m2/g) | Average Aperture (nm) |
|---|---|---|---|---|
| HPD-500 | High polarity | 22.40 ± 0.0022 | 500–550 | 10–12 |
| S-8 | High polarity | 21.57 ± 0.0028 | 100–120 | 28–30 |
| HPD-300 | Low polarity | 41.16 ± 0.0075 | 800–870 | 5–5.5 |
| AB-8 | Low polarity | 48.31 ± 0.0015 | 480–520 | 9–10 |
| D101 | Nonpolar | 34.46 ± 0.0027 | 550–600 | 9–10 |
| X-5 | Nonpolar | 32.19 ± 0.0070 | 650–700 | 9–10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, T.; Yang, L.; Shang, X.; Bian, S. Recovery of Cembratrien-Diols from Waste Tobacco (Nicotiana tabacum L.) Flowers by Microwave-Assisted Deep Eutectic Solvent Extraction: Optimization, Separation, and In Vitro Bioactivity. Molecules 2024, 29, 1563. https://doi.org/10.3390/molecules29071563
Yu T, Yang L, Shang X, Bian S. Recovery of Cembratrien-Diols from Waste Tobacco (Nicotiana tabacum L.) Flowers by Microwave-Assisted Deep Eutectic Solvent Extraction: Optimization, Separation, and In Vitro Bioactivity. Molecules. 2024; 29(7):1563. https://doi.org/10.3390/molecules29071563
Chicago/Turabian StyleYu, Tao, Long Yang, Xianchao Shang, and Shiquan Bian. 2024. "Recovery of Cembratrien-Diols from Waste Tobacco (Nicotiana tabacum L.) Flowers by Microwave-Assisted Deep Eutectic Solvent Extraction: Optimization, Separation, and In Vitro Bioactivity" Molecules 29, no. 7: 1563. https://doi.org/10.3390/molecules29071563
APA StyleYu, T., Yang, L., Shang, X., & Bian, S. (2024). Recovery of Cembratrien-Diols from Waste Tobacco (Nicotiana tabacum L.) Flowers by Microwave-Assisted Deep Eutectic Solvent Extraction: Optimization, Separation, and In Vitro Bioactivity. Molecules, 29(7), 1563. https://doi.org/10.3390/molecules29071563





