Mycotransformation of Commercial Grade Cypermethrin Dispersion by Aspergillus terreus PDB-B Strain Isolated from Lake Sediments of Kulamangalam, Madurai
Abstract
1. Introduction
1.1. Cypermethrin as a Pollutant
1.2. Mycobiodegradation as a Solution
1.3. Current Studies
2. Results and Discussion
2.1. Physicochemical Properties of Soil
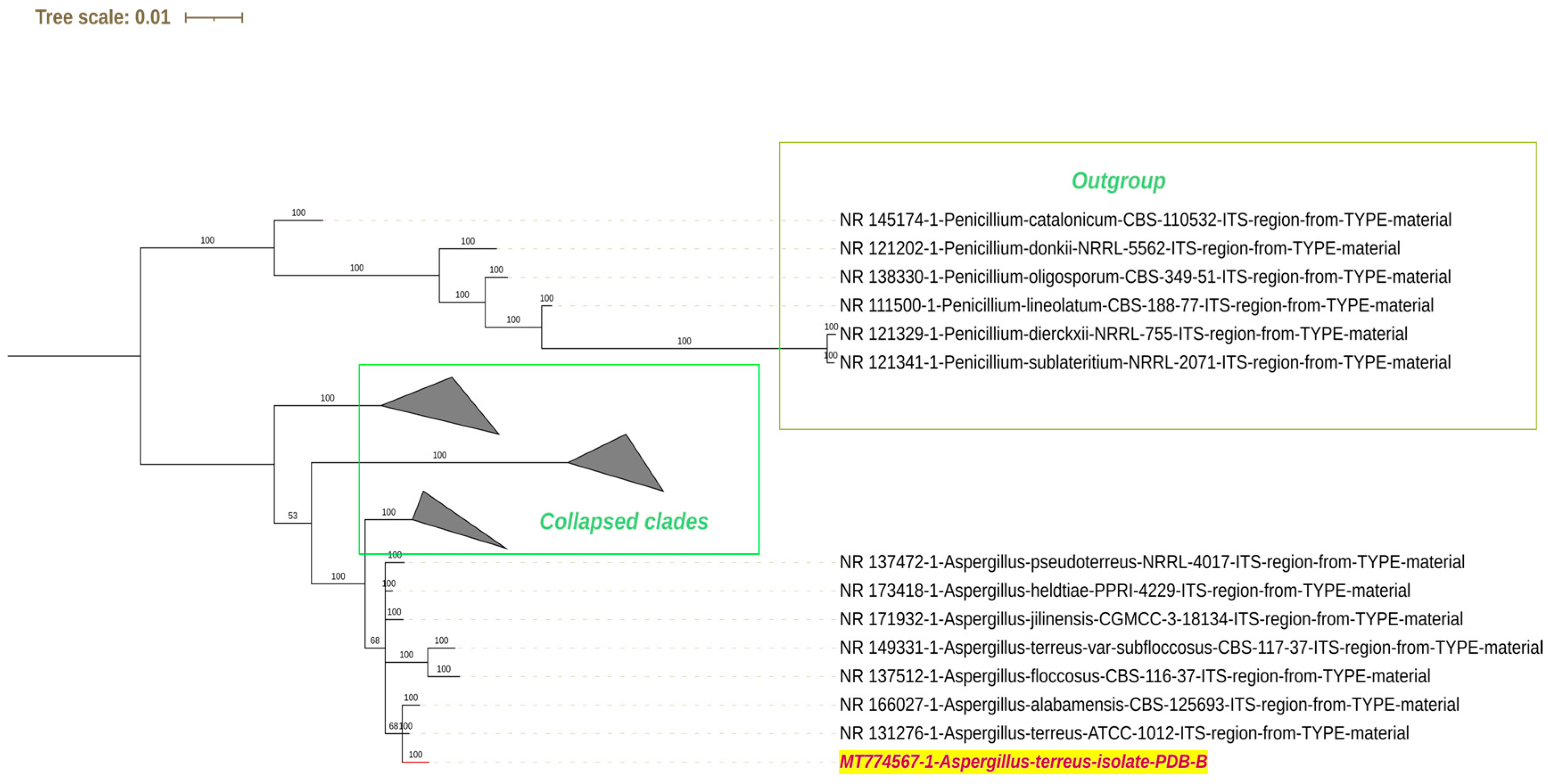
2.2. Isolation and Identification of Fungi
2.3. Screening for Cypermethrin Resistance
2.4. Screening for Extracellular Enzymes Involved in Cypermethrin Degradation
Lignin-Degrading Enzymes
2.5. Studies on Biodegradation of Cypermethrin
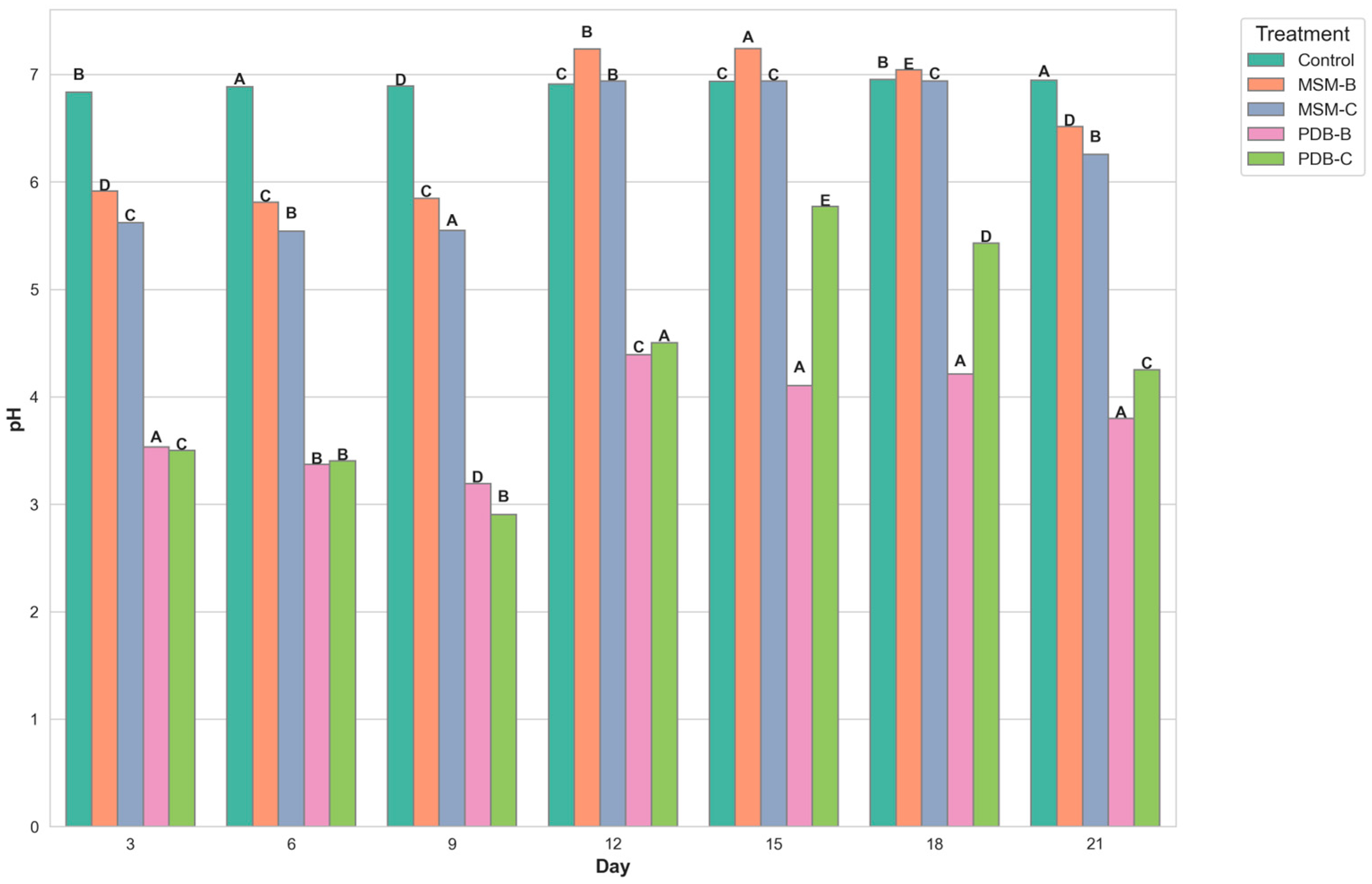
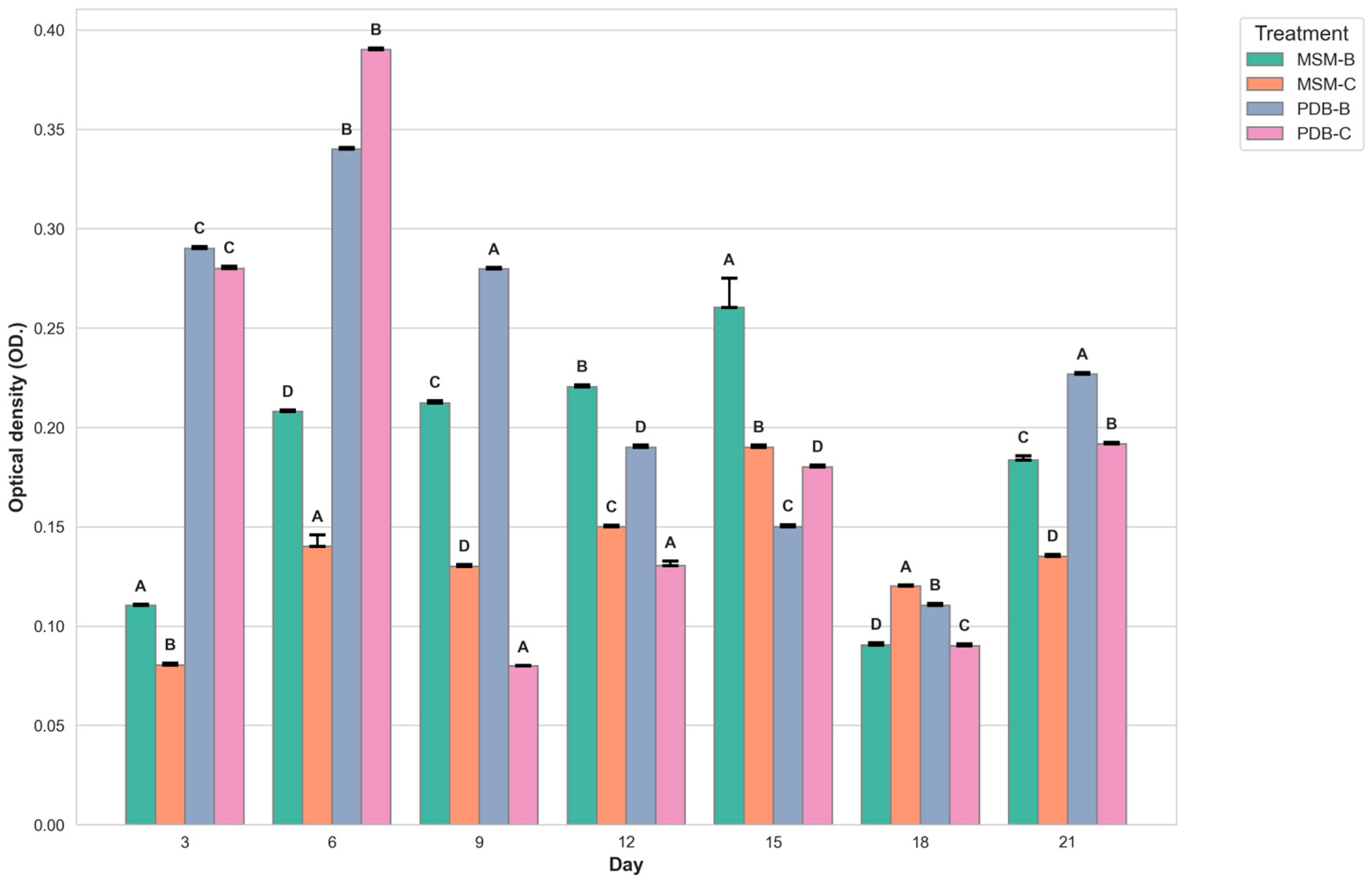
2.5.1. Studies on Change in pH, Dry Mycelial Weight, and Optical Density
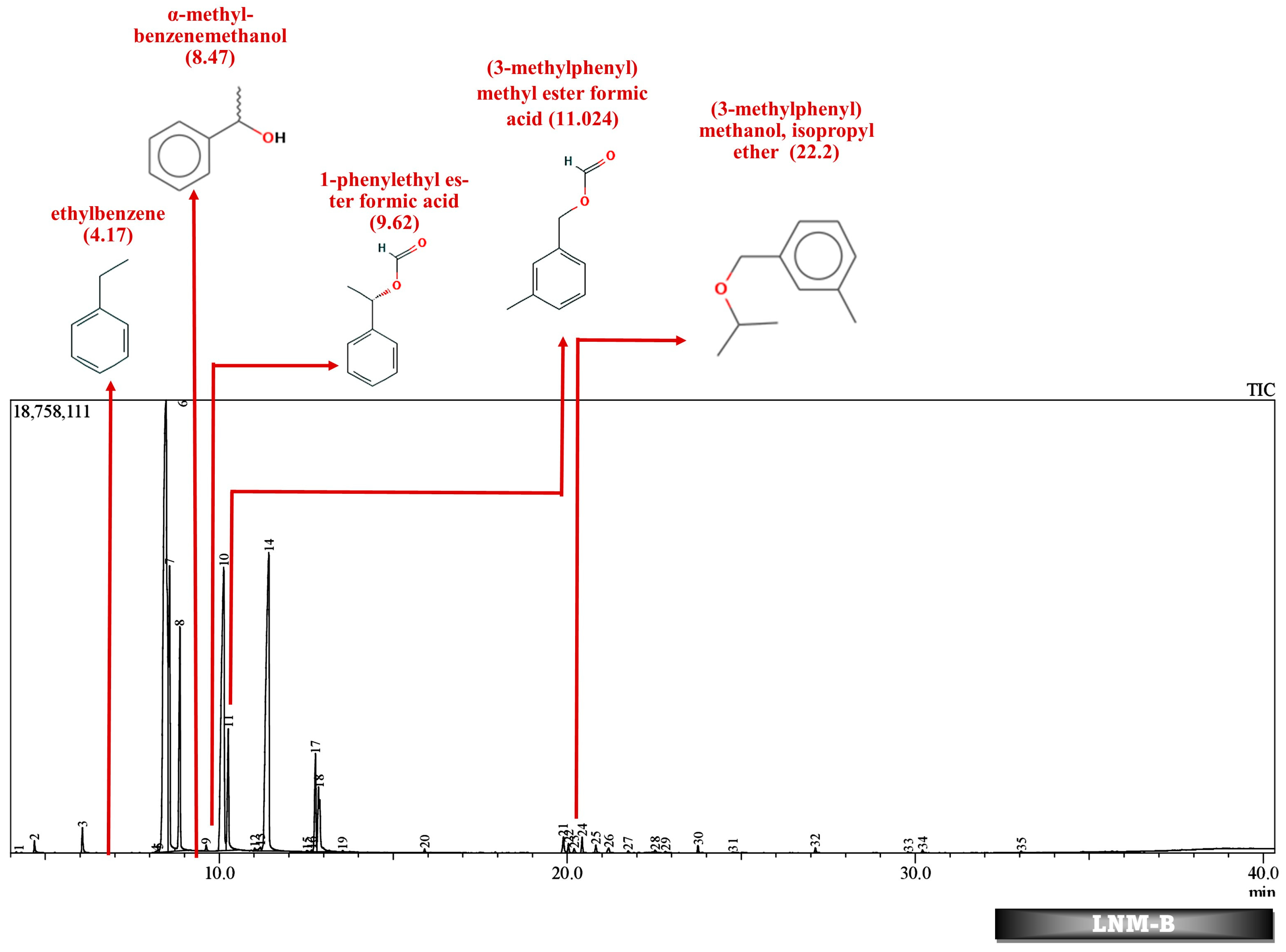
2.5.2. Analysis of Degradation Efficiency and Products by GC-MS
2.6. Soil Microcosm Studies
Brine Shrimp Lethality Assay (BSLP)
3. Materials and Methods
3.1. Collection of Samples
3.2. Determination of the Physicochemical Properties of Soil
3.3. Isolation and Identification of Fungi
3.4. Screening for Resistance to Cypermethrin
3.5. Screening for Extracellular Enzymes Involved in Cypermethrin Degradation
3.6. Qualitative Assay for Lignin-Degrading Enzymes
3.7. Biodegradation of Cypermethrin
3.7.1. Changes in pH and Biomass
3.7.2. Microcosm Studies
3.8. Sample Extraction and Analysis by GC-MS
3.9. Prediction and Data Processing of Transformation Products (TPs)
3.10. Brine Shrimp Lethality Assay (BSLP)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Deutsch, C.A.; Tewksbury, J.J.; Tigchelaar, M.; Battisti, D.S.; Merrill, S.C.; Huey, R.B.; Naylor, R.L. Increase in crop losses to insect pests in a warming climate. Science 2018, 361, 916–919. [Google Scholar] [CrossRef]
- Tang, F.H.; Lenzen, M.; McBratney, A.; Maggi, F. Risk of pesticide pollution at the global scale. Nat. Geosci. 2021, 14, 206–210. [Google Scholar] [CrossRef]
- Hassaan, M.A.; El Nemr, A. Pesticides pollution: Classifications, human health impact, extraction and treatment techniques. Egypt. J. Aquat. Res. 2020, 46, 207–220. [Google Scholar] [CrossRef]
- Gangola, S.; Sharma, A.; Bhatt, P.; Khati, P.; Chaudhary, P. Presence of esterase and laccase in Bacillus subtilis facilitates biodegradation and detoxification of cypermethrin. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ullah, S.; Zuberi, A.; Alagawany, M.; Farag, M.R.; Dadar, M.; Karthik, K.; Tiwari, R.; Dhama, K.; Iqbal, H.M. Cypermethrin induced toxicities in fish and adverse health outcomes: Its prevention and control measure adaptation. J. Environ. Manag. 2018, 206, 863–871. [Google Scholar] [CrossRef]
- Farag, M.R.; Alagawany, M.; Bilal, R.M.; Gewida, A.G.; Dhama, K.; Abdel-Latif, H.M.; Amer, M.S.; Rivero-Perez, N.; Zaragoza-Bastida, A.; Binnaser, Y.S.; et al. An overview on the potential hazards of pyrethroid insecticides in fish, with special emphasis on cypermethrin toxicity. Animals 2021, 11, 1880. [Google Scholar] [CrossRef]
- Bhatt, P.; Bhatt, K.; Huang, Y.; Lin, Z.; Chen, S. Esterase is a powerful tool for the biodegradation of pyrethroid insecticides. Chemosphere 2020, 244, 125507. [Google Scholar] [CrossRef]
- Ceci, A.; Pinzari, F.; Russo, F.; Persiani, A.M.; Gadd, G.M. Roles of saprotrophic fungi in biodegradation or transformation of organic and inorganic pollutants in co-contaminated sites. Appl. Microbiol. Biotechnol. 2019, 103, 53–68. [Google Scholar] [CrossRef]
- Das, S. (Ed.) Microbial Biodegradation and Bioremediation; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Kaur, P.; Sharma, A.; Parihar, L. In vitro study of mycoremediation of cypermethrin-contaminated soils in different regions of Punjab. Ann. Microbiol. 2015, 65, 1949–1959. [Google Scholar] [CrossRef]
- Hu, K.; Deng, W.; Zhu, Y.; Yao, K.; Li, J.; Liu, A.; Ao, X.; Zou, L.; Zhou, K.; He, L.; et al. Simultaneous degradation of β-cypermethrin and 3-phenoxybenzoic acid by Eurotium cristatum ET1, a novel “golden flower fungus” strain isolated from Fu Brick Tea. MicrobiologyOpen 2019, 8, e00776. [Google Scholar] [CrossRef]
- Aguila-Torres, P.; Maldonado, J.; Gaete, A.; Figueroa, J.; González, A.; Miranda, R.; González-Stegmaier, R.; Martin, C.; González, M. Biochemical and genomic characterization of the cypermethrin-degrading and biosurfactant-producing bacterial strains isolated from marine sediments of the Chilean Northern Patagonia. Mar. Drugs 2020, 18, 252. [Google Scholar] [CrossRef]
- Zhang, M.; Ming, Y.; Guo, H.; Zhu, Y.; Yang, Y.; Chen, S.; He, L.; Ao, X.; Liu, A.; Zhou, K.; et al. Screening of lactic acid bacteria for their capacity to bind cypermethrin in vitro and the binding characteristics and its application. Food Chem. 2021, 347, 129000. [Google Scholar] [CrossRef]
- Whangchai, K.; Van Hung, T.; Al-Rashed, S.; Narayanan, M.; Kandasamy, S.; Pugazhendhi, A. Biodegradation competence of Streptomyces toxytricini D2 isolated from leaves surface of the hybrid cotton crop against β cypermethrin. Chemosphere 2021, 276, 130152. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, P.; Rene, E.R.; Huang, Y.; Wu, X.; Zhou, Z.; Li, J.; Kumar, A.J.; Sharma, A.; Chen, S. Indigenous bacterial consortium-mediated cypermethrin degradation in the presence of organic amendments and Zea mays plants. Environ. Res. 2022, 212, 113137. [Google Scholar] [CrossRef]
- Gur Ozdal, O.; Algur, O.F. Biodegradation α-endosulfan and α-cypermethrin by Acinetobacter schindleri B7 isolated from the microflora of grasshopper (Poecilimon tauricola). Arch. Microbiol. 2022, 204, 1–8. [Google Scholar] [CrossRef]
- Zhao, J.; Jiang, Y.; Gong, L.; Chen, X.; Xie, Q.; Jin, Y.; Du, J.; Wang, S.; Liu, G. Mechanism of β-cypermethrin metabolism by Bacillus cereus GW-01. Chem. Eng. J. 2022, 430, 132961. [Google Scholar] [CrossRef]
- Liu, F.; Bai, J.; Huang, W.; Li, F.; Ke, W.; Zhang, Y.; Xie, D.; Zhang, B.; Guo, X. Characterization of a novel beta-cypermethrin-degrading strain of Lactobacillus pentosus 3–27 and its effects on bioremediation and the bacterial community of contaminated alfalfa silage. J. Hazard. Mater. 2022, 423, 127101. [Google Scholar] [CrossRef] [PubMed]
- Muthabathula, P.; Biruduganti, S. Analysis of Biodegradation of the Synthetic Pyrethroid Cypermethrin by Beauveria bassiana. Curr. Microbiol. 2022, 79, 1–9. [Google Scholar] [CrossRef]
- Narayanan, M.; Murugan, J.M.; Kandasamy, G.; Kandasamy, S.; Nasif, O.; Rajendran, M.; Pugazhendhi, A. The biotransformation potential of Bacillus cereus on β-cypermethrin to protect the earthworm (Perionyx excavatus) on insecticide-contaminated soil. Arch. Agron. Soil Sci. 2022, 68, 944–955. [Google Scholar] [CrossRef]
- Saied, E.; Fouda, A.; Alemam, A.M.; Sultan, M.H.; Barghoth, M.G.; Radwan, A.A.; Desouky, S.G.; Azab, I.H.E.; Nahhas, N.E.; Hassan, S.E.D. Evaluate the Toxicity of Pyrethroid Insecticide Cypermethrin before and after Biodegradation by Lysinibacillus cresolivuorans Strain HIS7. Plants 2021, 10, 1903. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Balomajumder, C. Effective mycoremediation coupled with bioaugmentation studies: An advanced study on newly isolated Aspergillus sp. in Type-II pyrethroid-contaminated soil. Environ. Pollut. 2020, 261, 114073. [Google Scholar] [CrossRef]
- Bhatt, P.; Huang, Y.; Zhang, W.; Sharma, A.; Chen, S. Enhanced cypermethrin degradation kinetics and metabolic pathway in Bacillus thuringiensis strain SG4. Microorganisms 2020, 8, 223. [Google Scholar] [CrossRef]
- Rao, D.L.N.; Ghai, S.K. Urease and dehydrogenase activity of alkali and reclaimed soils. Soil Res. 1985, 23, 661–665. [Google Scholar] [CrossRef]
- Mackay, E.M.; Pateman, J.A. The regulation of urease activity in Aspergillus nidulans. Biochem. Genet. 1982, 20, 763–776. [Google Scholar] [CrossRef]
- Singh, G.; Verma, A.K.; Kumar, V. Catalytic properties, functional attributes and industrial applications of β-glucosidases. 3 Biotech 2016, 6, 3. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.Y.; Xia, W.; Zhang, X.X.; Chen, S.; Nie, X.Z.; Qian, L.C. Characterization of β-glucosidase from Aspergillus terreus and its application in the hydrolysis of soybean isoflavones. J. Zhejiang Univ. Sci. B 2016, 17, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Junior, A.B.; Borges, D.G.; Tardioli, P.W.; Farinas, C.S. Characterization of β-glucosidase produced by Aspergillus niger under solid-state fermentation and partially purified using MANAE-Agarose. Biotechnol. Res. Int. 2014, 2014, 317092. [Google Scholar]
- Narasimha, G.; Sridevi, A.; Ramanjaneyulu, G.; Rajasekhar Reddy, B. Purification and Characterization of β-Glucosidase from Aspergillus niger. Int. J. Food Prop. 2016, 19, 652–661. [Google Scholar] [CrossRef]
- Ma, S.J.; Leng, B.; Xu, X.Q.; Zhu, X.Z.; Shi, Y.; Tao, Y.M.; Chen, Q.X. Purification and characterization of b-1, 4-glucosidase from Aspergillus glaucus. Afr. J. Biotechnol. 2011, 10, 19607–19614. [Google Scholar]
- Faulds, C.B.; Williamson, G. Purification and characterization of a ferulic acid esterase (FAE-III) from Aspergillus niger: Specificity for the phenolic moiety and binding to microcrystalline cellulose. Microbiology 1994, 140, 779–787. [Google Scholar] [CrossRef]
- Williamson, G.; Kroon, P.A.; Faulds, C.B. Hairy plant polysaccharides: A close shave with microbial esterases. Microbiology 1998, 144, 2011–2023. [Google Scholar] [CrossRef]
- De Vries, R.P.; Michelsen, B.; Poulsen, C.H.; Kroon, P.A.; Van Den Heuvel, R.H.; Faulds, C.B.; Visser, J. The faeA genes from Aspergillus niger and Aspergillus tubingensis encode ferulic acid esterases involved in degradation of complex cell wall polysaccharides. Appl. Environ. Microbiol. 1997, 63, 4638–4644. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, H.; Yan, Q.; Yang, S.; Duan, X.; Jiang, Z. Biochemical characterization of a first fungal esterase from Rhizomucor miehei showing high efficiency of ester synthesis. PLoS ONE 2013, 8, e77856. [Google Scholar] [CrossRef]
- Novotný, Č.; Svobodová, K.; Erbanová, P.; Cajthaml, T.; Kasinath, A.; Lang, E.; Šašek, V. Ligninolytic fungi in bioremediation: Extracellular enzyme production and degradation rate. Soil Biol. Biochem. 2004, 36, 1545–1551. [Google Scholar] [CrossRef]
- Bilal, M.; Iqbal, H. Ligninolytic enzymes mediated ligninolysis: An untapped biocatalytic potential to deconstruct lignocellulosic molecules in a sustainable manner. Catal. Lett. 2020, 150, 524–543. [Google Scholar] [CrossRef]
- Scherer, M.; Fischer, R. Molecular characterization of a blue-copper laccase, TILA, of Aspergillus nidulans. FEMS Microbiol. Lett. 2001, 199, 207–213. [Google Scholar] [CrossRef]
- Clutterbuck, A.J. Absence of laccase from yellow-spored mutants of Aspergillus nidulans. Microbiology 1972, 70, 423–435. [Google Scholar] [CrossRef]
- Bonugli-Santos, R.C.; Durrant, L.R.; Da Silva, M.; Sette, L.D. Production of laccase, manganese peroxidase and lignin peroxidase by Brazilian marine-derived fungi. Enzym. Microb. Technol. 2010, 46, 32–37. [Google Scholar] [CrossRef]
- Gajendiran, A.; Abraham, J. Biomineralisation of fipronil and its major metabolite, fipronil sulfone, by Aspergillus glaucus strain AJAG1 with enzymes studies and bioformulation. 3 Biotech 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Nikolaivits, E.; Dimarogona, M.; Karagiannaki, I.; Chalima, A.; Fishman, A.; Topakas, E. Versatile fungal polyphenol oxidase with chlorophenol bioremediation potential: Characterization and protein engineering. Appl. Environ. Microbiol. 2018, 84, e01628-18. [Google Scholar] [CrossRef]
- Roberts, T.R.; Standen, M.E. Degradation of the pyrethroid cypermethrin NRDC 149 (±)-α-cyano-3-phenoxybenzyl (±)-cis, trans-3-(2, 2-dichlorovinyl)-2, 2-dimethylcyclopropanecarboxylate and the respective cis-(NRDC 160) and trans-(NRDC 159) isomers in soils. Pestic. Sci. 1977, 8, 305–319. [Google Scholar] [CrossRef]
- Shono, T.; Ohsawa, K.; Casida, J.E. Metabolism of trans-and cis-permethrin, trans-and cis-cypermethrin, and decamethrin by microsomal enzymes. J. Agric. Food Chem. 1979, 27, 316–325. [Google Scholar] [CrossRef]
- Prenafeta-Boldú, F.X.; Ballerstedt, H.; Gerritse, J.; Grotenhuis, J.T.C. Bioremediation of BTEX hydrocarbons: Effect of soil inoculation with the toluene-growing fungus Cladophialophora sp. strain T1. Biodegradation 2004, 15, 59–65. [Google Scholar] [CrossRef]
- Nagaki, M.; Sato, R.; Tanabe, S.; Sato, T.; Hasui, Y.; Chounan, Y.; Tanaka, K.; Harada, Y. Biotransformation of acetophenone to 1-phenylethanol by fungi. Trans. Mater. Res. Soc. Jpn. 2016, 41, 247–250. [Google Scholar] [CrossRef][Green Version]
- Holland, H.L.; Bergen, E.J.; Chenchaiah, P.C.; Khan, S.H.; Munoz, B.; Ninniss, R.W.; Richards, D. Side chain hydroxylation of aromatic compounds by fungi.: 1. Products and stereochemistry. Can. J. Chem. 1987, 65, 502–507. [Google Scholar] [CrossRef]
- Hofrichter, M.; Ullrich, R. Oxidations catalyzed by fungal peroxygenases. Curr. Opin. Chem. Biol. 2014, 19, 116–125. [Google Scholar] [CrossRef]
- Borges, K.B.; de Souza Borges, W.; Durán-Patrón, R.; Pupo, M.T.; Bonato, P.S.; Collado, I.G. Stereoselective biotransformations using fungi as biocatalysts. Tetrahedron Asymmetry 2009, 20, 385–397. [Google Scholar] [CrossRef]
- Zilbeyaz, K.; Taskin, M.; Kurbanoglu, E.B.; Kurbanoglu, N.I.; Kilic, H. Production of (R)-1-phenylethanols through bioreduction of acetophenones by a new fungus isolate Trichothecium roseum. Chirality Pharmacol. Biol. Chem. Conseq. Mol. Asymmetry 2010, 22, 543–547. [Google Scholar] [CrossRef]
- Doi, M.; Matsui, M.; Kanayama, T.; Shuto, Y.; Kinoshita, Y. Asymmetric reduction of acetophenone by Aspergillus species and their possible contribution to katsuobushi flavor. Biosci. Biotechnol. Biochem. 1992, 56, 958–960. [Google Scholar] [CrossRef][Green Version]
- Andrade, L.H.; Keppler, A.F.; Schoenlein-Crusius, I.H.; Porto, A.L.; Comasseto, J.V. Evaluation of acetophenone monooxygenase and alcohol dehydrogenase activities in different fungal strains by biotransformation of acetophenone derivatives. J. Mol. Catal. B Enzym. 2004, 31, 129–135. [Google Scholar] [CrossRef]
- Jin, H.; Li, Z.Y.; Dong, X.W. Enantioselective hydrolysis of various substituted styrene oxides with Aspergillus niger CGMCC 0496. Org. Biomol. Chem. 2004, 2, 408–414. [Google Scholar] [CrossRef]
- Chen, S.; Hu, Q.; Hu, M.; Luo, J.; Weng, Q.; Lai, K. Isolation and characterization of a fungus able to degrade pyrethroids and 3-phenoxybenzaldehyde. Bioresour. Technol. 2011, 102, 8110–8116. [Google Scholar] [CrossRef]
- Chen, S.; Deng, Y.; Chang, C.; Lee, J.; Cheng, Y.; Cui, Z.; Zhang, L.H. Pathway and kinetics of cyhalothrin biodegradation by Bacillus thuringiensis strain ZS-19. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef]
- Saikia, N.; Gopal, M. Biodegradation of β-cyfluthrin by fungi. J. Agric. Food Chem. 2004, 52, 1220–1223. [Google Scholar] [CrossRef]
- Zhang, C.; Jia, L.; Wang, S.; Qu, J.; Li, K.; Xu, L.; Yan, Y. Biodegradation of beta-cypermethrin by two Serratia spp. with different cell surface hydrophobicity. Bioresour. Technol. 2010, 101, 3423–3429. [Google Scholar] [CrossRef]
- Zhao, H.; Geng, Y.; Chen, L.; Tao, K.; Hou, T. Biodegradation of cypermethrin by a novel Catellibacterium sp. strain CC-5 isolated from contaminated soil. Can. J. Microbiol. 2013, 59, 311–317. [Google Scholar] [CrossRef]
- Gu, X.-Z.; Zhang, G.-Y.; Chen, L.; Dai, R.-L.; Yu, Y.-C. Persistence and dissipation of synthetic pyrethroid pesticides in red soils from the Yangtze River delta area. Environ. Geochem. Health 2008, 30, 67–77. [Google Scholar] [CrossRef]
- Xu, Z.; Shen, X.; Zhang, X.C.; Liu, W.; Yang, F. Microbial degradation of alpha-cypermethrin in soil by compound-specific stable isotope analysis. J. Hazard. Mater. 2015, 295, 37–42. [Google Scholar] [CrossRef]
- Margesin, R.; Schinner, F. (Eds.) Manual for Soil Analysis-Monitoring and Assessing Soil Bioremediation; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2005; Volume 5. [Google Scholar]
- Tsui, C.K.; Woodhall, J.; Chen, W.; Lévesque, C.A.; Lau, A.; Schoen, C.D.; Baschien, C.; Najafzadeh, M.J.; de Hoog, G.S. Molecular techniques for pathogen identification and fungus detection in the environment. IMA Fungus 2011, 2, 177–189. [Google Scholar] [CrossRef]
- Johnson, M.; Zaretskaya, I.; Raytselis, Y.; Merezhuk, Y.; McGinnis, S.; Madden, T.L. NCBI BLAST: A better web interface. Nucleic Acids Res. 2008, 36 (Suppl. S2), W5–W9. [Google Scholar] [CrossRef]
- Dereeper, A.; Guignon, V.; Blanc, G.; Audic, S.; Buffet, S.; Chevenet, F.; Dufayard, J.F.; Guindon, S.; Lefort, V.; Lescot, M.; et al. Phylogeny. fr: Robust phylogenetic analysis for the non-specialist. Nucleic Acids Res. 2008, 36 (Suppl. S2), W465–W469. [Google Scholar] [CrossRef]
- Lemoine, F.; Correia, D.; Lefort, V.; Doppelt-Azeroual, O.; Mareuil, F.; Cohen-Boulakia, S.; Gascuel, O. NGPhylogeny. fr: New generation phylogenetic services for non-specialists. Nucleic Acids Res. 2019, 47, W260–W265. [Google Scholar] [CrossRef]
- Letunic, I.; Bork, P. Interactive Tree Of Life (iTOL) v5: An online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021, 49, W293–W296. [Google Scholar] [CrossRef]
- Sangeetha, J.; Thangadurai, D. Staining techniques and biochemical methods for the identification of fungi. In Laboratory Protocols in Fungal Biology; Springer: New York, NY, USA, 2013; pp. 237–257. [Google Scholar]
- Pointing, S.B. Qualitative methods for the determination of lignocellulolytic enzyme production by tropical fungi. Fungal Divers. 1999, 2, 17–33. [Google Scholar]
- Morris, S.; Nicholls, J. An evaluation of optical density to estimate fungal spore concentrations in water suspensions. Strain 1978, 1, 1240–1242. [Google Scholar] [CrossRef]
- Garoiaz, H.; Berrabah, M.; Elidrissi, A.; Hammouti, B.; Ríos, A. Analysis of cypermethrin residues and its main degradation products in soil and formulation samples by gas chromatography-electron impact-mass spectrometry in the selective ion monitoring mode. Int. J. Environ. Anal. Chem. 2012, 92, 1378–1388. [Google Scholar] [CrossRef]
- Wicker, J.; Lorsbach, T.; Gütlein, M.; Schmid, E.; Latino, D.; Kramer, S.; Fenner, K. enviPath–The environmental contaminant biotransformation pathway resource. Nucleic Acids Res. 2016, 44, D502–D508. [Google Scholar] [CrossRef]
- Trostel, L.; Coll, C.; Fenner, K.; Hafner, J. Combining predictive and analytical methods to elucidate pharmaceutical biotransformation in activated sludge. Environ. Sci. Process. Impacts 2023, 25, 1322–1336. [Google Scholar] [CrossRef]
- Zhang, Y.; Mu, J.; Han, J.; Gu, X. An improved brine shrimp larvae lethality microwell test method. Toxicol. Mech. Methods 2012, 22, 23–30. [Google Scholar] [CrossRef]

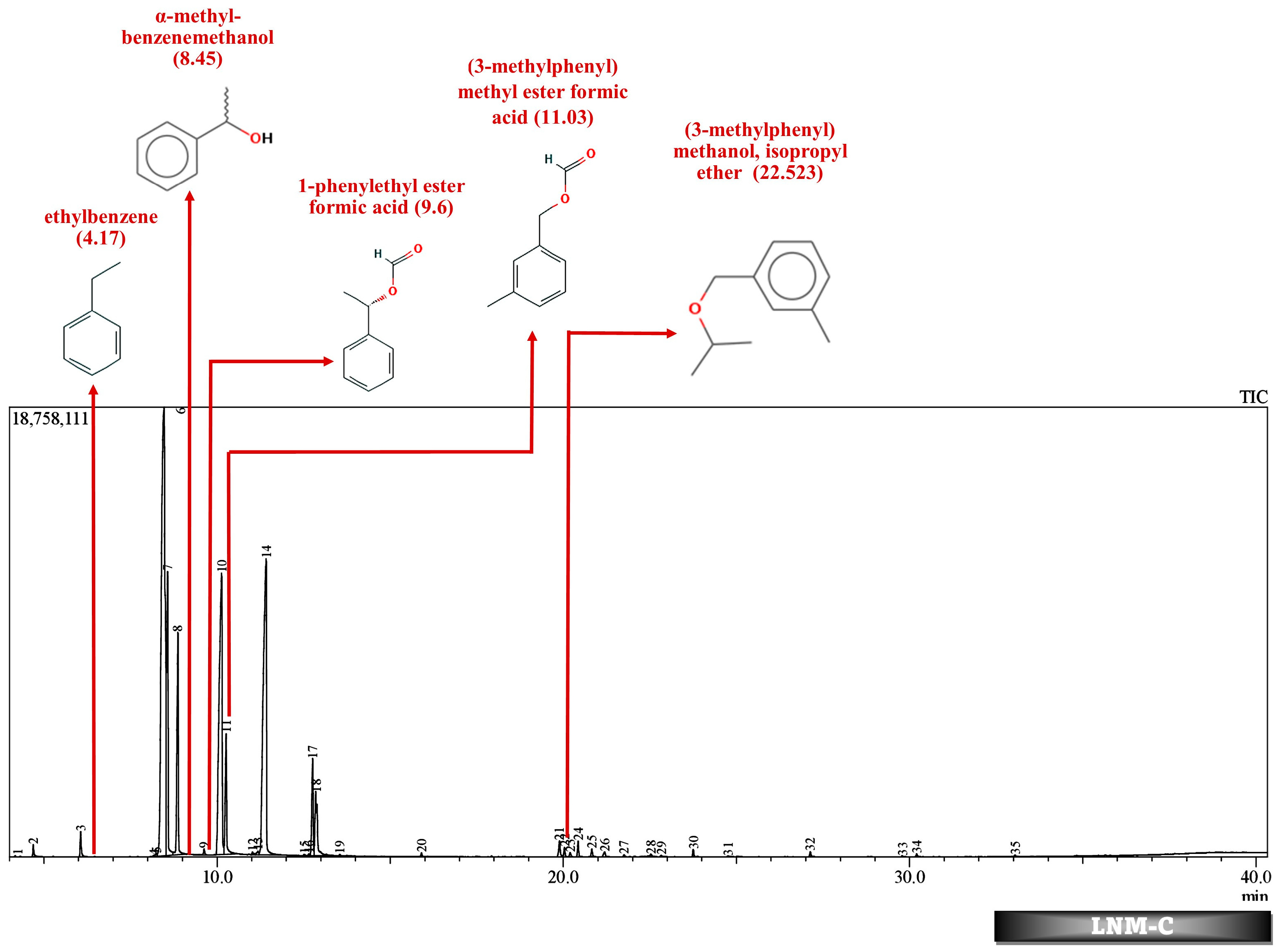
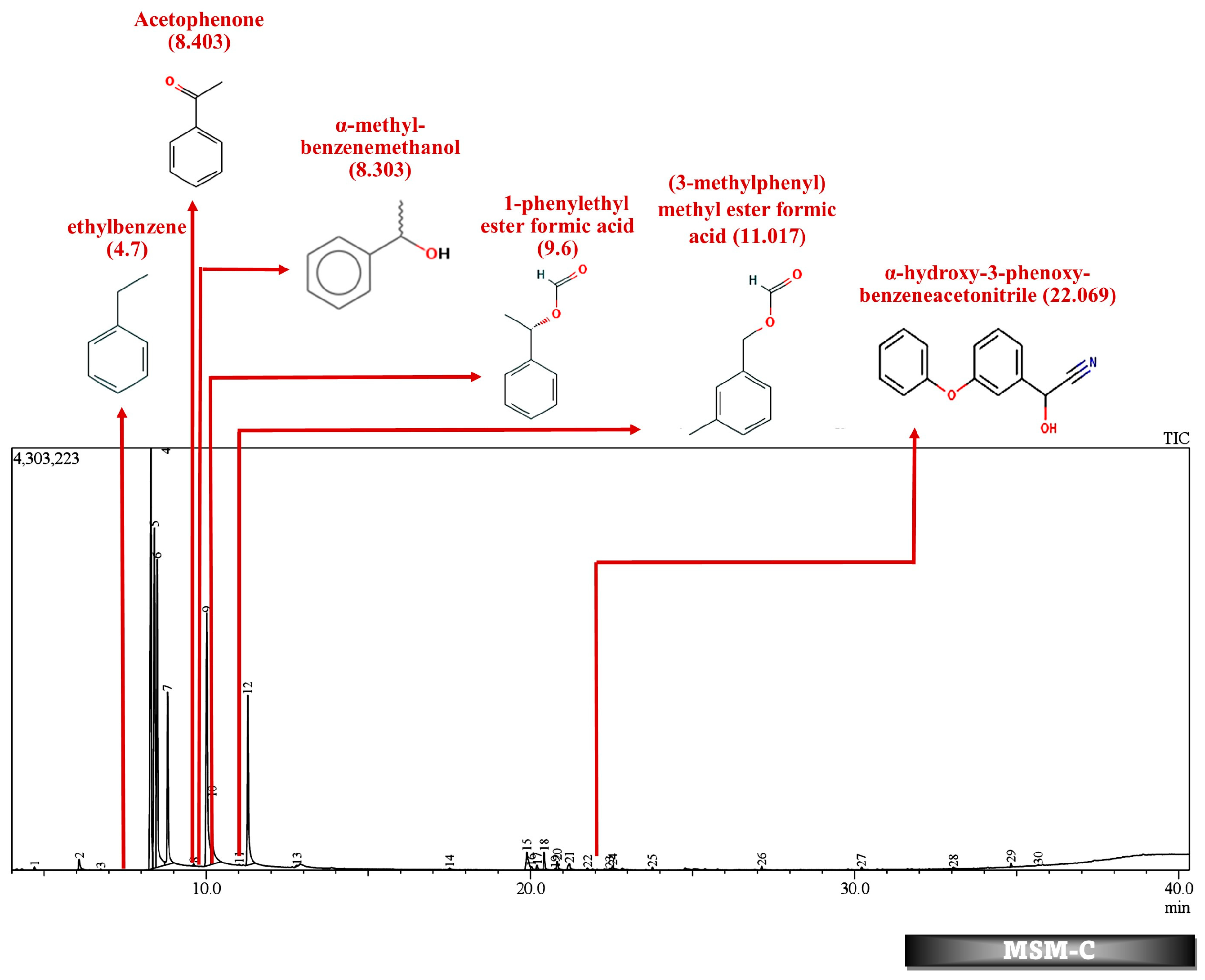


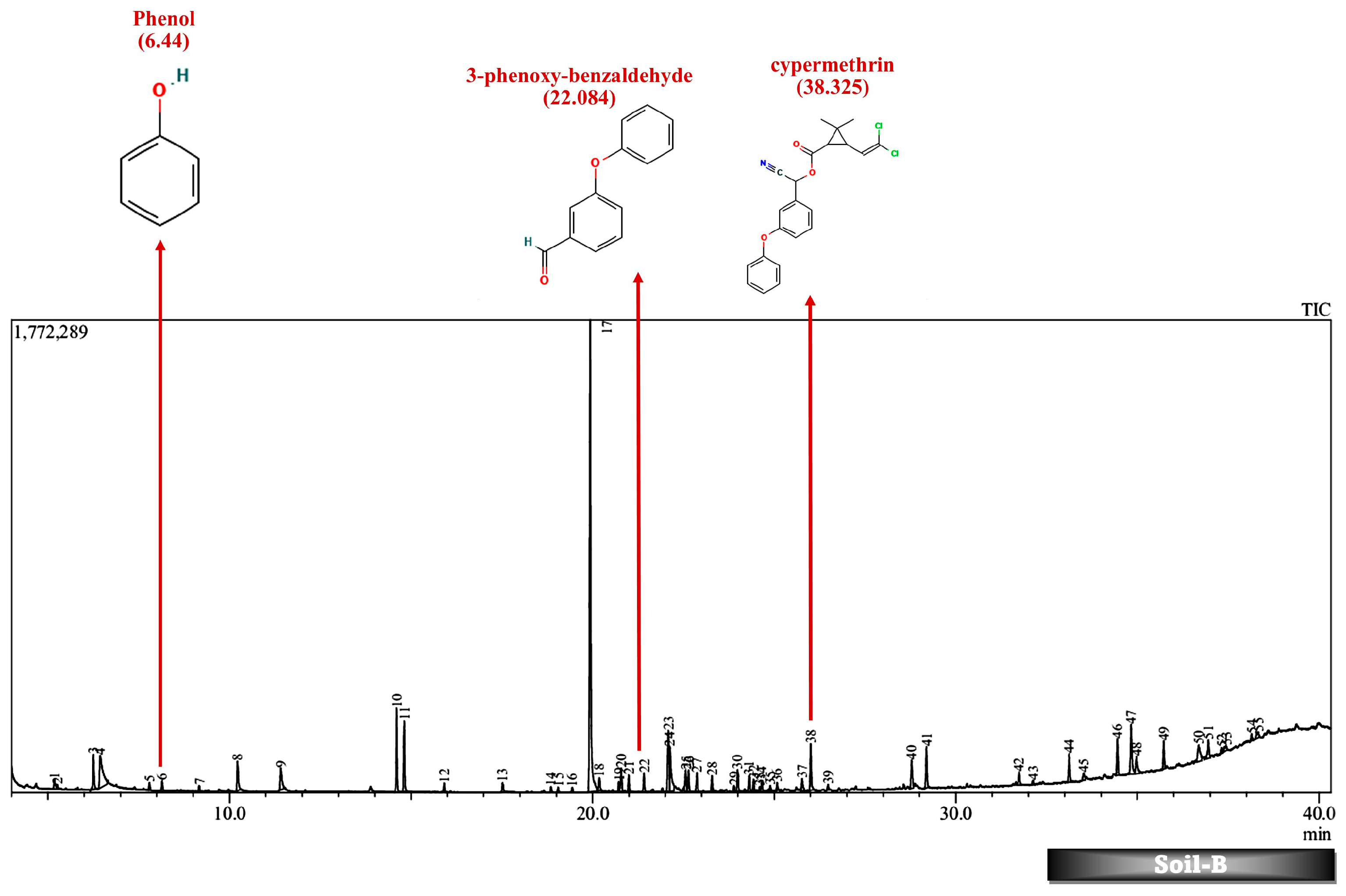
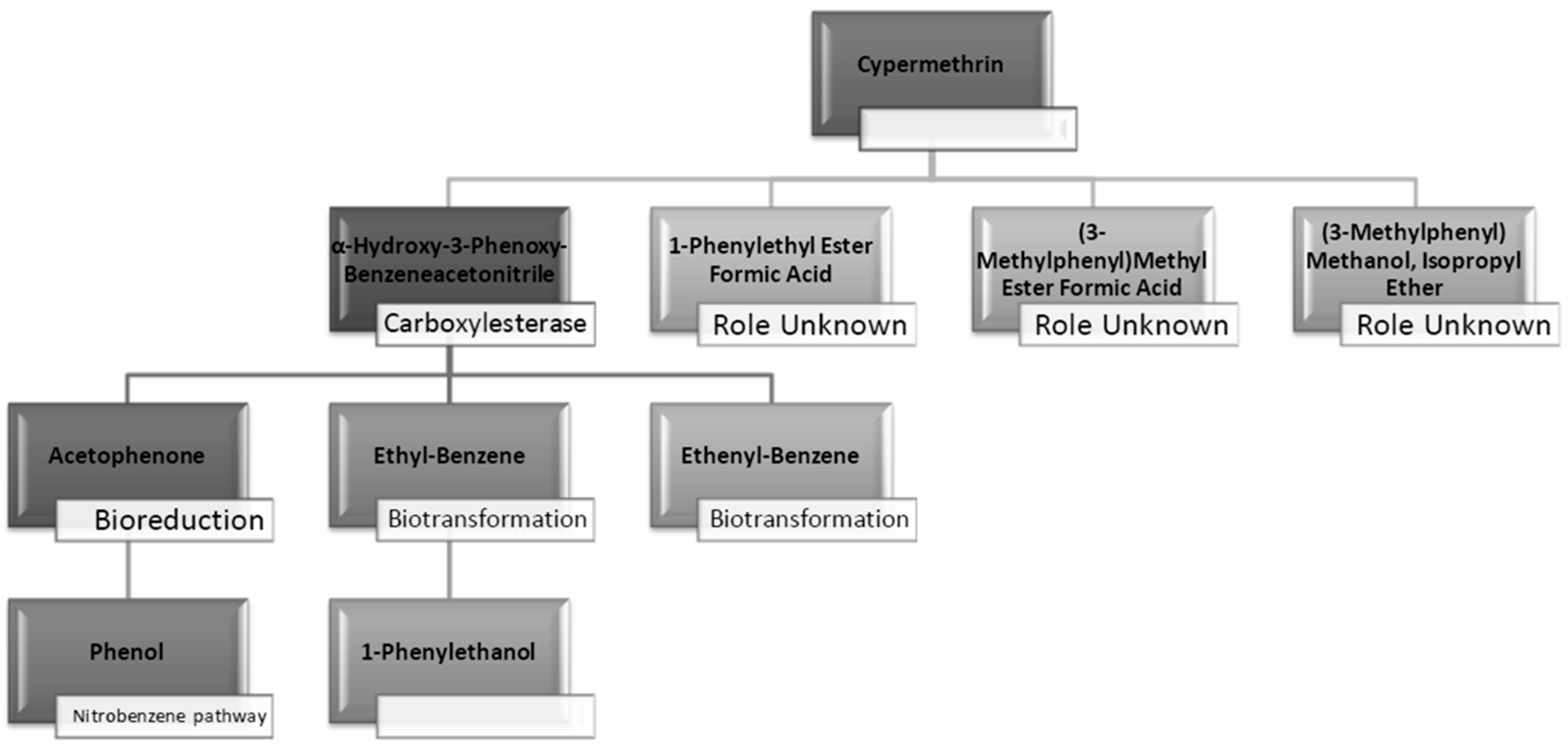
| No. | Compound | Strain | Source of Isolate | Derived Metabolites | Conc. (mg/L) | % Degrad. | References |
|---|---|---|---|---|---|---|---|
| 1. | Cypermethrin (technical grade) | Bacillus thuringiensis strain SG4 and strain SG2 | Soil | 3-phenoxybenzoic acid (3-PBA), 3-IAA, DCMU, Sulfosulfuron, Allethrin I | NA | 91.3 | [15] |
| 2. | α-cypermethrin | Acinetobacter schindleri | grasshopper (Poecilimon tauricola) | 3-phenoxybenzaldehyde, 3-PBA, phenol, muconic acid | 100 | 68.4 | [16] |
| 3. | β-cypermethrin | Bacillus cereus GW-01 | Sheep’s rumen chyme | 3-PBA, phenol, catechol | 100 | ∼60 | [17] |
| 4. | β-cypermethrin | Lactobacillus pentosus 3–27 | β-CYP contaminated silage | 3-PBA | 50 | 96 | [18] |
| 5. | Cypermethrin (technical grade) | Beauveria bassiana (ITCC 913) | Indian Type Culture Collection (ITCC), India | 4-hydroxybenzoate, cypermethrin, 2-(4-hydroxy phenoxy) benzoic acid methyl ester, 3,5,-dihydroxybenzoic acid, 3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropane carboxylate, 3,5,-dimethoxy phenol, and phenol | 0.12 | NA * | [19] |
| 6. | β-cypermethrin | Bacillus cereus | BT cotton cultivated and long-time pesticide-exposed soil | 1-(2-acetoxyethyl)-3,6-diazahomoadamantan-9-one, benzene, 2 (dimethylamino)-, 1-ethyl-3-methyl, ethanethiol, silane, 1-(3-hydroxy-3-methylbutyl)-3, fumaric acid, 9-anthracenyltrimethyl- | 100 | NA * | [20] |
| 7. | Cypermethrin (technical grade) | Lysinibacillus cresolivuorans his7 | pesticide-contaminated soil | 1H-purine-2,6-dione,3,7-dihydro 1,3,7 trimethy; benzene ethanamine, à-methyl-3-[4-methylphenyloxy]; 9-octadecenamide; 1,2-Benzenedicarboxylicacid-3-nitro; acetic acid (4-chloro-2-methylphenoxy) | 2500 | 86.9 | [21] |
| 8. | β-cypermethrin | Streptomyces toxytricini D2 | pesticide-exposed surface of cotton leaves | 3-PBA, methyl salicylate, phthalic acid, phenol, and 3-phenoxy benzaldehyde | 6% solution | 80.71 ± 1.17 | [14] |
| 9. | α-cypermethrin (≥97%) | Aspergillus sp. PYR-P2 | pesticide-contaminated soil | 3-phenoxybenzaldehyde; α-cyano-3-phenoxybenzyl-3-(2,2-dichlorovinyl)-2,2-dimethyl cyclopropane carboxylate; 3-phenoxybenzoic acid | 500 | 91.56 | [22] |
| 10. | Cypermethrin (technical grade) | Bacillus thuringiensis strain SG4 | pesticide-contaminated soil from agricultural fields | 3-phenoxybenzaldehyde; 2-hydroxy-2(3-phenoxyphenyl) acetonitrile; 3-(2,2-dichloroethenyl)-2,2-dimethyl cyclopropanecarboxylate; 2-hydroxy-2 (3-phenoxyphenyl) acetonitrile | 50 | 80 | [23] |
| CONTROL | LNM-C | LNM-B | MSM-B | MSM-C | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RT (min) | A% | Name | RT (min) | A% | Name | RT (min) | A% | Name | RT (min) | A% | Name | RT (min) | A% | Name |
| 38.34 | 0.06 | cypermethrin | ||||||||||||
| 22.523 | 0.08 | (3-methylphenyl) methanol, isopropyl ether | 22.2 | 0.11 | (3-methylphenyl) methanol, isopropyl ether | |||||||||
| 22.069 | 0.17 | α-hydroxy-3-phenoxy-benzeneacetonitrile | ||||||||||||
| 11.03 | 0.06 | (3-methylphenyl) methyl ester formic acid | 11.024 | 0.08 | (3-methylphenyl) methyl ester formic acid | 11.038 | 0.12 | (3-methylphenyl) methyl ester formic acid | 11.017 | 0.03 | (3-methylphenyl) methyl ester formic acid | |||
| 9.6 | 0.13 | 1-phenylethyl ester formic acid | 9.626 | 0.14 | 1-phenylethyl ester formic acid | 9.647 | 0.14 | 1-phenylethyl ester formic acid | 9.6 | 0.1 | 1-phenylethyl ester formic acid | |||
| 8.5 | 32.52 | α-methyl-benzenemethanol | 8.476 | 35.51 | α-methyl-benzenemethanol | 8.572 | 32.65 | α-methyl-benzenemethanol | 8.303 | 21.77 | α-methyl-benzenemethanol | |||
| 8.403 | 19.56 | 1-phenyl-ethanone | ||||||||||||
| 6.526 | 0.13 | phenol | ||||||||||||
| 4.171 | 0.04 | ethylbenzene | 4.17 | 0.02 | ethylbenzene | 4.176 | 0.17 | ethylbenzene | 4.7 | 0.15 | ethenylbenzene | |||
| Soil-C | Soil-B | ||||
|---|---|---|---|---|---|
| Retention Time (min) | AREA% | Name | Retention Time (min) | AREA% | Name |
| 38.604 | 0.44 | cypermethrin | 38.325 | 0.65 | cypermethrin |
| 6.44 | 5.03 | phenol | |||
| 22.089 | 4.48 | 3-phenoxybenzaldehyde | 22.084 | 4.46 | 3-phenoxybenzaldehyde |
| SMILES | Name |
|---|---|
| CC1(C)C(C=C(Cl)Cl)C1C([O-)=O | 3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate |
| OC(CN)c1ccc(Oc2cccc2)c1 | 2-hydroxy-2-(3-phenoxyphenyl)acetonitrile |
| CC1(CO)C(C=C(Cl)Cl)C1C([O-])=O | 3-(2,2-dichloroethenyl)-2-(hydroxymethyl)-2-methylcyclopropane-1-carboxylic acid |
| OC(C([O-])=O)c1ccc(Oc2cccc2)c1 | 3-Phenoxy-4-Hydroxyphenylacetic Acid |
| O=Cc1cccc(Oc2cccc2)c1 | 3-phenoxybenzaldehyde |
| CC1(C)C(C=C(Cl)Cl)C1C(=O)OC(C([O-])=O)c1ccc(Oc2cccc2)c1 | 2-[3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropanecarbonyl]oxy-2-(3-phenoxyphenyl)acetic acid |
| CC1(C=O)C(C=C(Cl)Cl)C1C([O-])=O | |
| OC(C([O-])=O)c1cccc(O)c1O | 2-(2,3-dihydroxyphenyl)-2-hydroxyacetate |
| Oc1ccccc1 | Phenol |
| OC(C([O-])=O)c1ccc(O)c(O)c1 | 2-(3,4-dihydroxyphenyl)-2-hydroxyacetate |
| OC(C([O-])=O)c1ccc(Oc2ccc(O)c2O)c1 | methyl 3,5-dihydroxy-4phenylmethoxybenzoate |
| [O-]C(=O)C(=O)c1ccc(Oc2cccc2)c1 | (3-acetylphenyl)benzoate |
| OC(C([O-])=O)c1cccc(O)c1 | 2-hydroxy-2-(3-hydroxyphenyl)acetate |
| Oc1ccccc1O | 2-hydroxyphenoxy |
| [O-]C(=O)c1ccc(Oc2cccc2)c1 | 3-phenoxybenzoate |
| Sample Name | Lethality (%) of Artemia nauplii |
|---|---|
| Control | 100 |
| Negative Control | 73.33 |
| Dilution between sample and ASW 5:5 | |
| PDB-B | 100 |
| PDB-C | 100 |
| MSM-B | 30 |
| MSM-C | 36.66 |
| Dilution between sample and ASW 7:3 | |
| PDB-B | 100 |
| PDB-C | 100 |
| MSM-B | 30 |
| MSM-C | 33 |
| Sample Types | Media | Microorganisms | Sample Name |
|---|---|---|---|
| Liquid | Potato Dextrose Broth (PDB) | Fungi PDB-B | LNM-B |
| LNM-C | |||
| Mineral Salt Medium (MSM) | Fungal Consortium (A, PDB-B, J, UN2, M1, and SM108) | MSM-B | |
| MSM-C | |||
| Soil | - | Fungi PDB-B | Soil-B |
| - | Fungal Consortium (A, PDB-B, J, UN2, M1, and SM108) | Soil-C |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kannan, P.; Baskaran, H.; Juliana Selvaraj, J.B.; Saeid, A.; Kiruba Nester, J.M. Mycotransformation of Commercial Grade Cypermethrin Dispersion by Aspergillus terreus PDB-B Strain Isolated from Lake Sediments of Kulamangalam, Madurai. Molecules 2024, 29, 1446. https://doi.org/10.3390/molecules29071446
Kannan P, Baskaran H, Juliana Selvaraj JB, Saeid A, Kiruba Nester JM. Mycotransformation of Commercial Grade Cypermethrin Dispersion by Aspergillus terreus PDB-B Strain Isolated from Lake Sediments of Kulamangalam, Madurai. Molecules. 2024; 29(7):1446. https://doi.org/10.3390/molecules29071446
Chicago/Turabian StyleKannan, Priyadharshini, Hidayah Baskaran, Jemima Balaselvi Juliana Selvaraj, Agnieszka Saeid, and Jennifer Michellin Kiruba Nester. 2024. "Mycotransformation of Commercial Grade Cypermethrin Dispersion by Aspergillus terreus PDB-B Strain Isolated from Lake Sediments of Kulamangalam, Madurai" Molecules 29, no. 7: 1446. https://doi.org/10.3390/molecules29071446
APA StyleKannan, P., Baskaran, H., Juliana Selvaraj, J. B., Saeid, A., & Kiruba Nester, J. M. (2024). Mycotransformation of Commercial Grade Cypermethrin Dispersion by Aspergillus terreus PDB-B Strain Isolated from Lake Sediments of Kulamangalam, Madurai. Molecules, 29(7), 1446. https://doi.org/10.3390/molecules29071446







