Experimental and Theoretical Investigation of External Electric-Field-Induced Crystallization of TKX-50 from Solution by Finite-Temperature String with Order Parameters as Collective Variables for Ionic Crystals
Abstract
1. Introduction
2. Theory
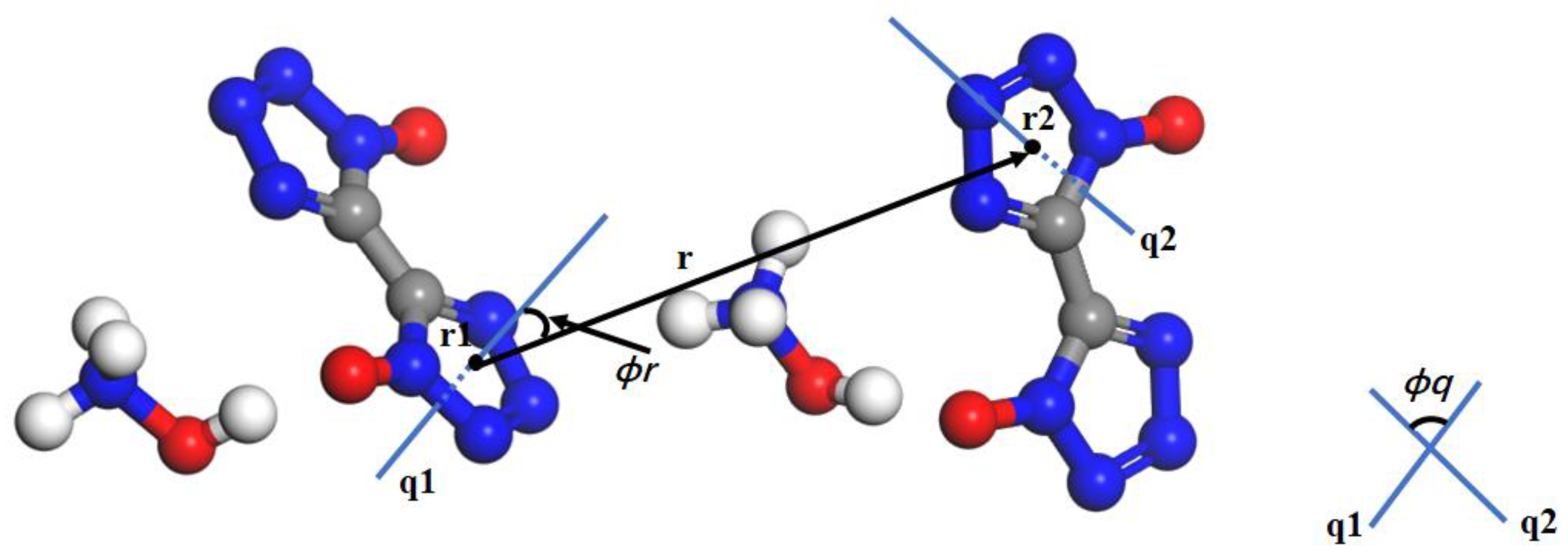
2.1. Order Parameters
2.2. Finite-Temperature String
2.3. Minimum-Free-Energy Path from Finite-Temperature String
3. Results and Discussion
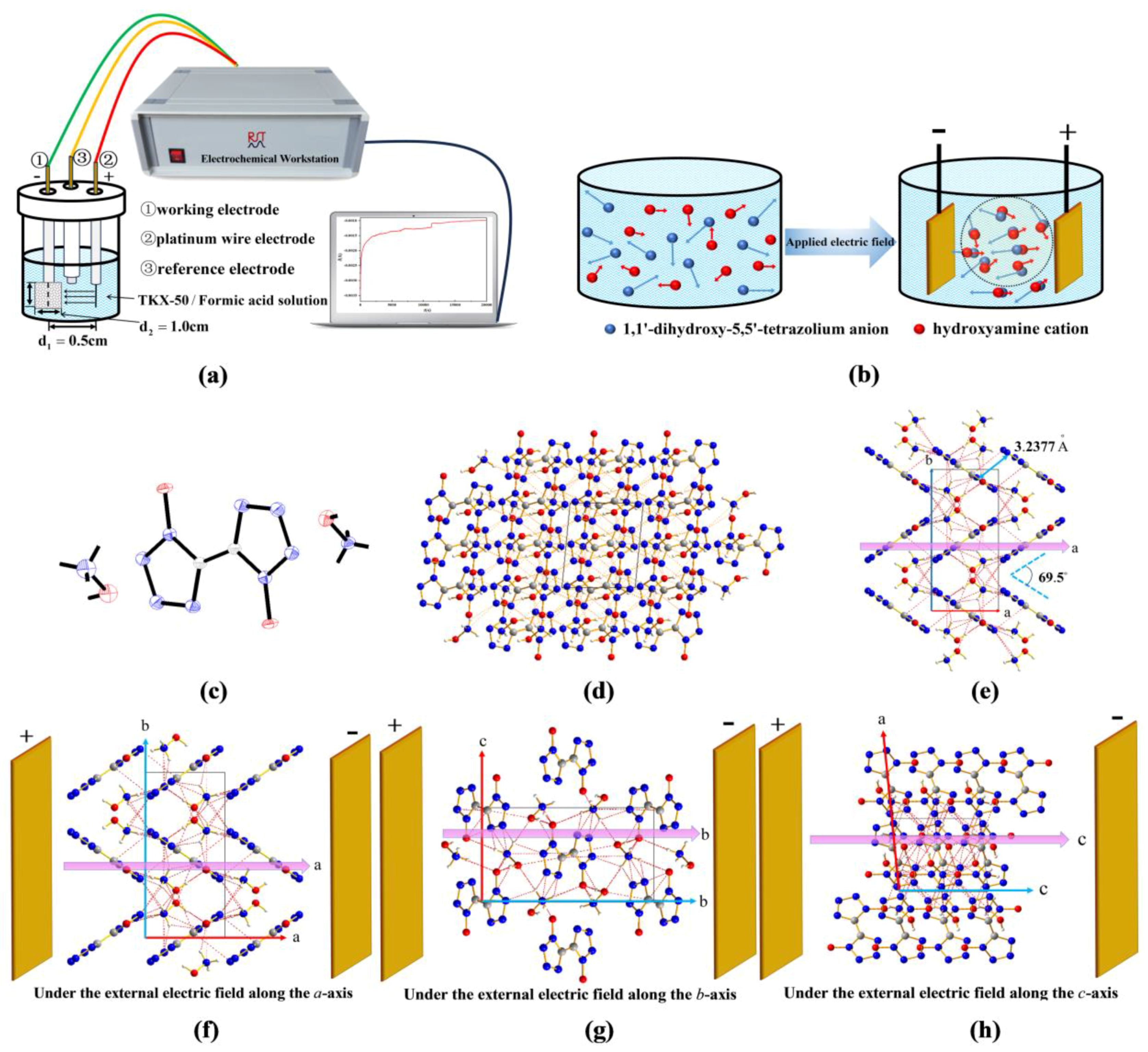
3.1. Crystallization of TKX-50 by DC External Electric Field
3.2. MD Simulation on Crystallization of TKX-50 without External Electric Field
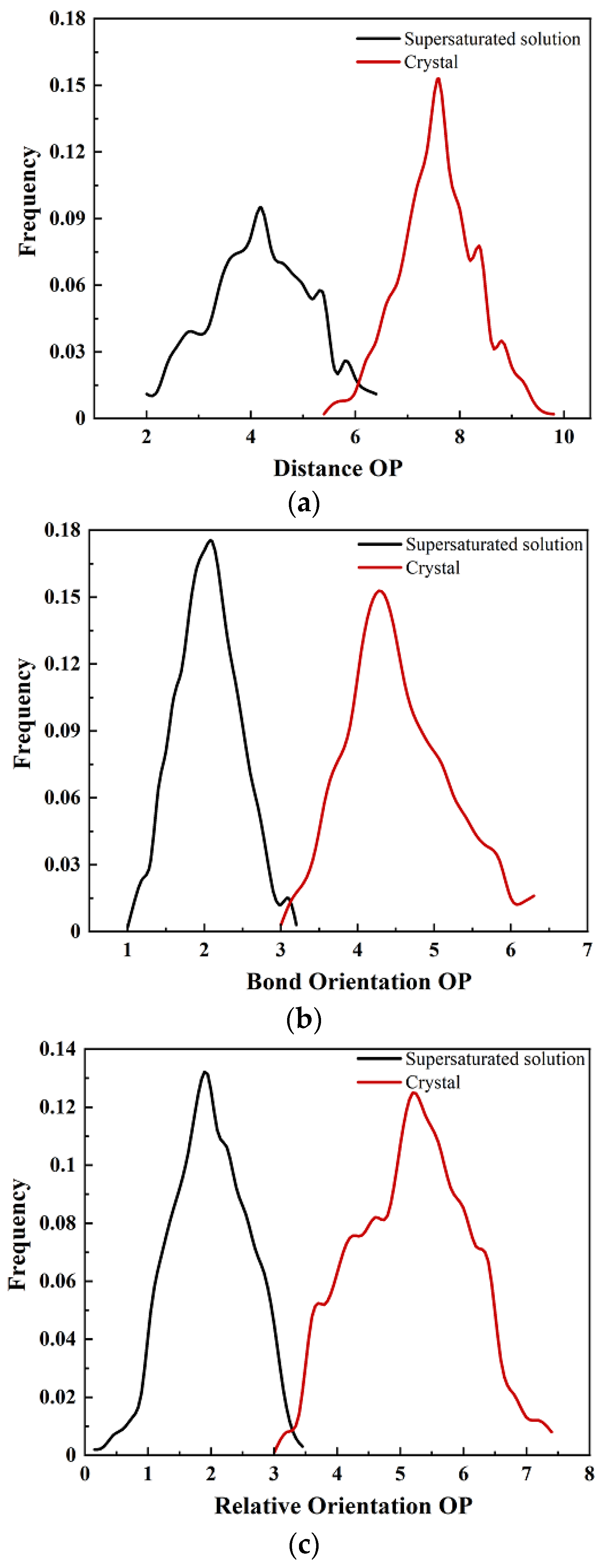
3.2.1. Peaks in Pair Distribution Function
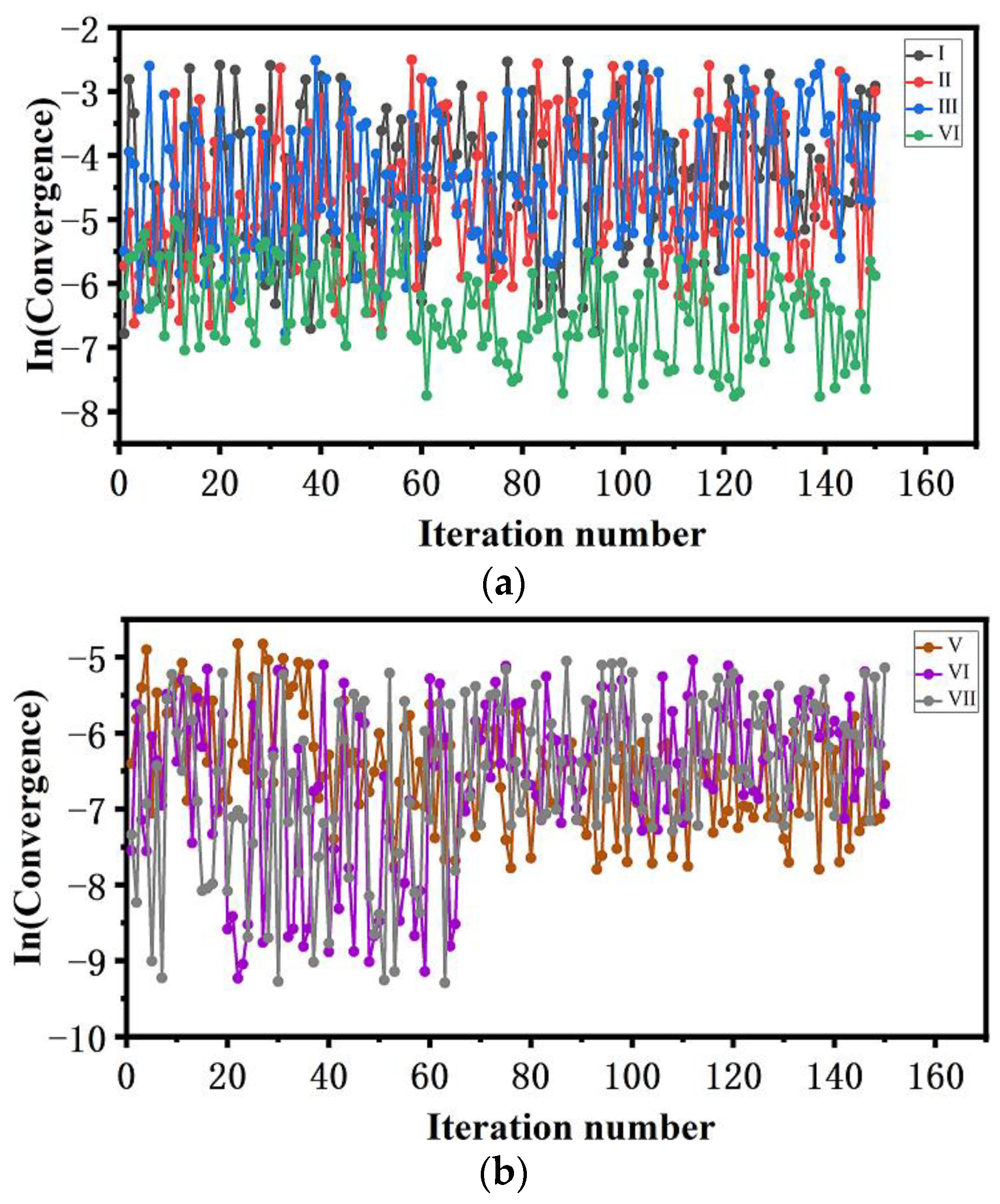
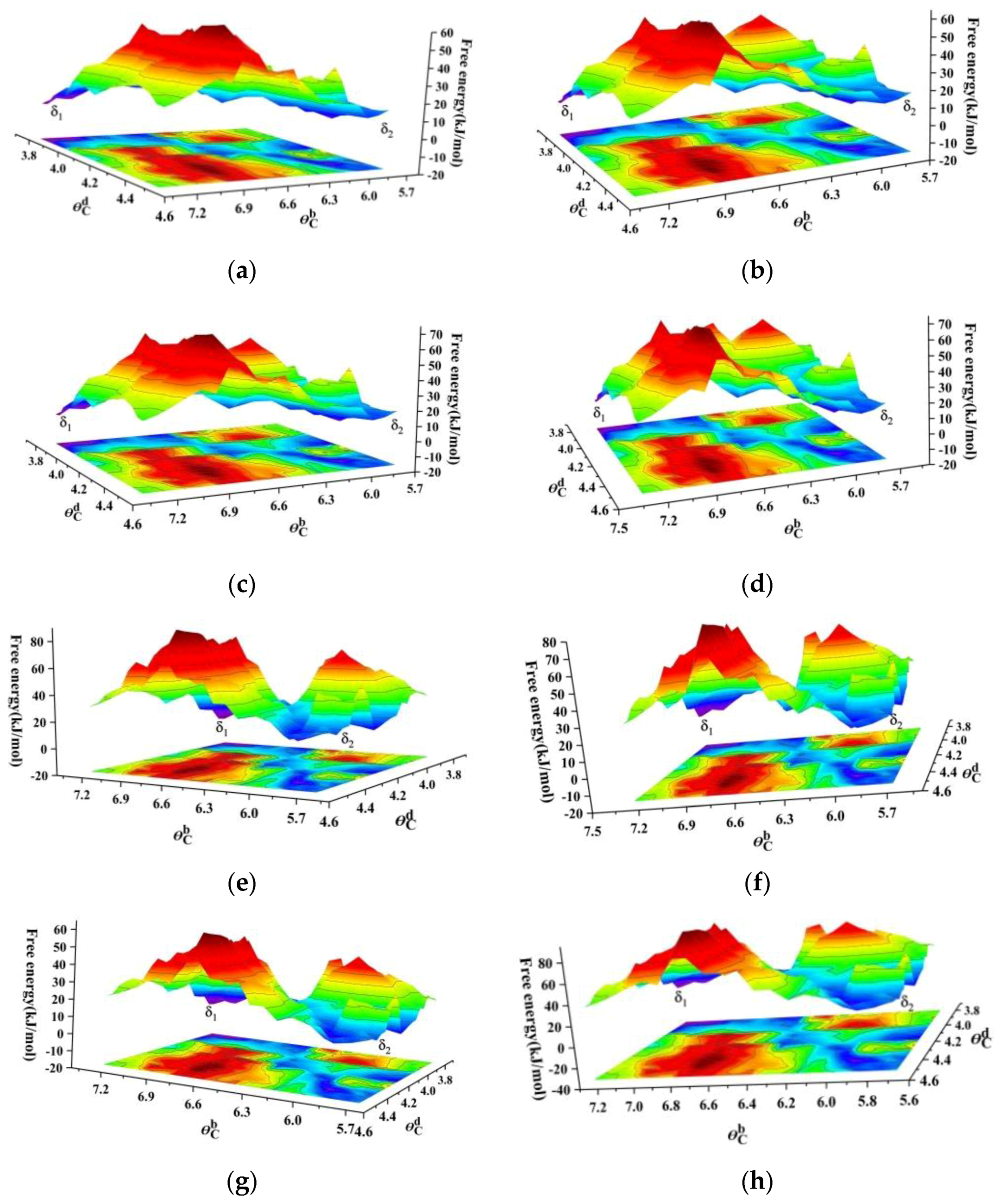
3.2.2. Convergence of FTS and K-Means Clustering
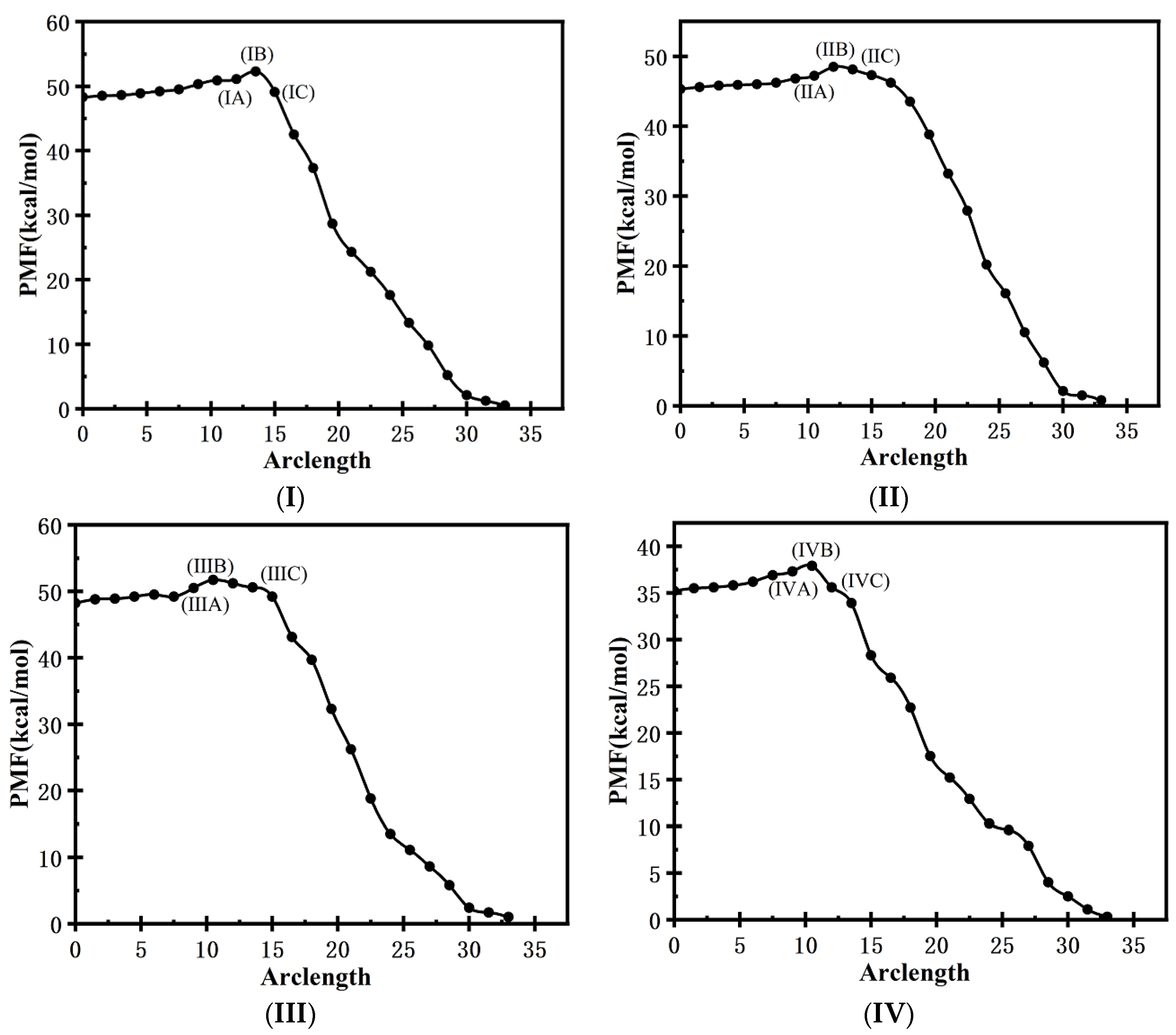
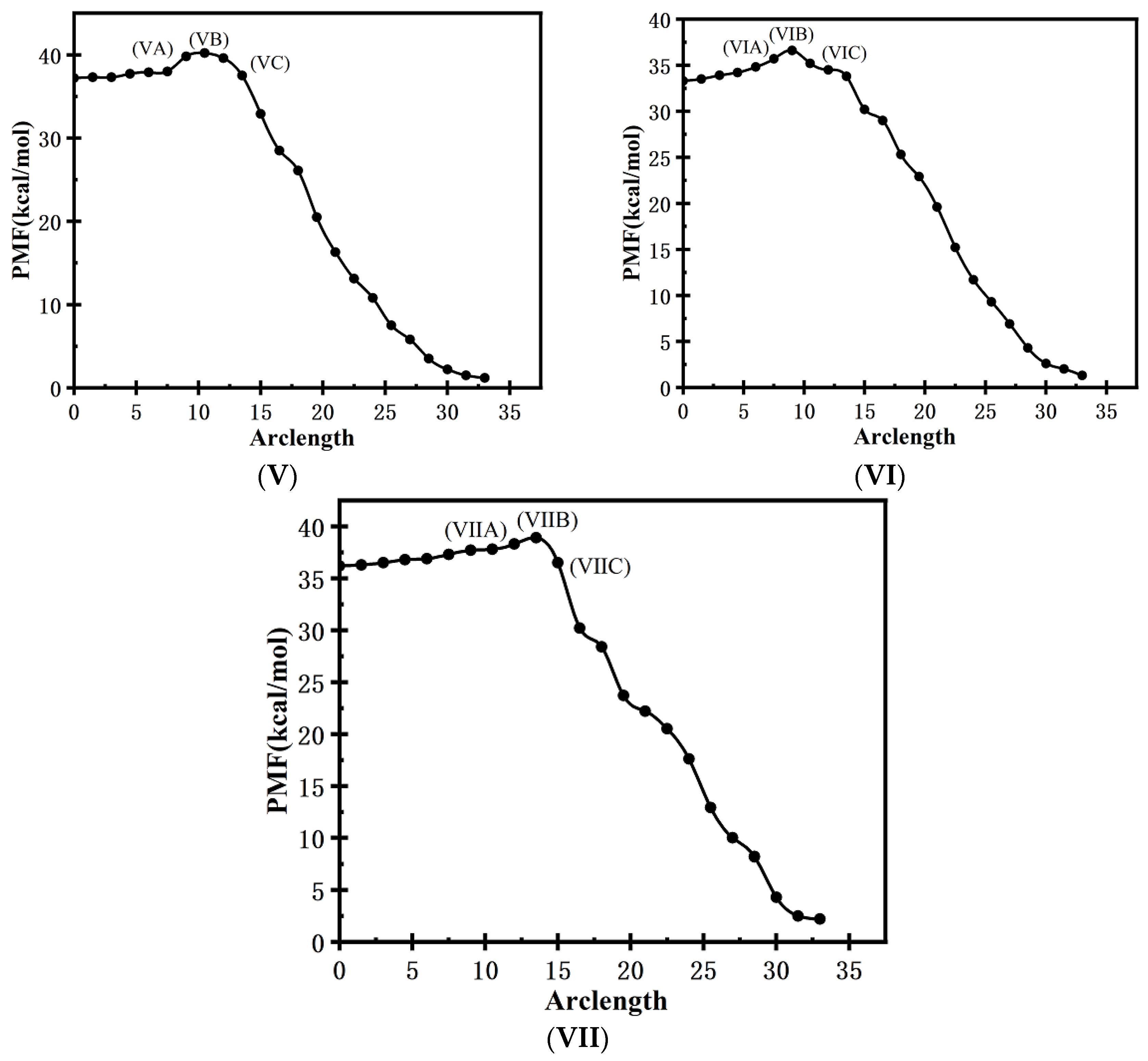
3.2.3. Minimum-Free-Energy Path
3.3. MD Simulation of Crystallization of TKX-50 under the External Electric Fields
3.4. Detonation Performance
4. Experiment and MD Simulation Details
4.1. Experiment
4.2. MD Simulation Details
4.3. Detonation Performance Calculations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, H.; Ren, D.; Cheng, X. The theoretical investigation of the β-crystobalite structure under the effect of electric field. Comp. Mater. Sci. 2015, 96, 306–311. [Google Scholar] [CrossRef]
- Simura, R.; Nakamura, K.; Uda, S. Change of melting temperature of non-doped and Mg-doped lithium niobate underan external electric field. J. Cryst. Growth 2008, 310, 3873–3877. [Google Scholar] [CrossRef]
- Roland, C.M.; Fragiadakis, D.; Bogoslovov, R.; Urban, S.; Dabrowski, R.; Tykarska, M.; Osiecka, N.; Czub, J. Volumetric, dielectric, calorimetric and X-ray studies of smectogenic 10PBO8 at atmospheric and elevated pressures. Liq. Cryst. Rev. 2012, 39, 993–1001. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Rahman, M. Electric-field induced isotropic to smectic-Cphase transition. J. Mol. Liq. 2014, 196, 204–207. [Google Scholar] [CrossRef]
- Mahmood, A.; Chan, M.S.Y.; Saleemi, A.S.; Guo, J.; Lee, S.L. Synergic effect: Temperature-assisted electric-field-induced supramolecular phase transitions at the liquid/solid Interface. Langmuir 2019, 35, 8031–8037. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Zhang, P.; Zhang, Q.; Qiao, R.; He, Q.; Li, H.B.; Wang, Y.; Guo, J.; Zhang, D.; Duan, Z.; et al. Electric-field control of tri-statephase transformation with a selective dual-ion switch. Nature 2017, 546, 124–128. [Google Scholar] [CrossRef]
- Jeong, J.; Aetukuri, N.; Graf, T.; Schladt, T.D.; Samant, M.G.; Parkin, S.S.P. Suppression of metal-insulator transition in VO2 by electric field-induced oxygen vacancy formation. Science 2013, 339, 1402–1405. [Google Scholar] [CrossRef]
- Alexander, L.F.; Radacsi, N. Application of electric fields for controlling crystallization. CrystEngComm 2019, 21, 5014–5031. [Google Scholar] [CrossRef]
- Radacsi, N. Process Intensification in Crystallization: Submicron Particle Generation Using Alternative Energy Forms. Ph.D. Thesis, University of Debrecen, Hungary, Germany, 2012. Available online: https://www.academia.edu/10747704/Process_Intensification_in_Crystallization_Submicron_Particle_Generation_using_Alternative_Energy_Forms (accessed on 1 October 2012). [CrossRef]
- Flores-Hernández, E.; Stojanoff, V.; Arreguín-Espinosa, R.; Moreno, A.; Sánchez-Puig, N.J. An electrically assisted device for protein crystallization in a vapor-diffusion setup. J. Appl. Crystallogr. 2013, 46, 832–834. [Google Scholar] [CrossRef]
- Walter, T.K.; Ferreira, C.F.D.G.; Iulek, J.; Benelli, E.M. Use of Protein Thin Film Organized by External Electric Field as a Template for Protein Crystallization. ACS Omega 2018, 3, 8683–8690. [Google Scholar] [CrossRef]
- Martínez-Caballero, S.; Cuéllar-Cruz, M.; Demitri, N.; Polentarutti, M.; Rodríguez-Romero, A.; Rodríguez-Romero, A. Glucose lsomerase Polymorphs Obtained Using an Ad Hoc Protein Crystallization Temperature Device and a Growth Cell Applying an Electric Field. Cryst. Growth Des. 2016, 16, 1679–1686. [Google Scholar] [CrossRef]
- Rodríguez-Romero, A.; Esturau-Escofet, N.; Pareja-Rivera, C.; Moreno, A. Crystal growth of high-quality protein crystals under the presence of an alternant electric field in pulse-wave mode, and a strong magnetic field with radio frequency pulses characterized by x-ray diffraction. Crystals 2017, 7, 179. [Google Scholar] [CrossRef]
- Sazaki, G.; Moreno, A.; Nakajima, K. Novel coupling effects of the magnetic and electric fields on protein crystallization. J. Cryst. Growth 2004, 262, 499–502. [Google Scholar] [CrossRef]
- Koizumi, H.; Uda, S.; Fujiwara, K.; Okada, J.; Nozawa, J. Effect of an External Electric Field on the Kinetics of Dislocation-Free Growth of Tetragonal Hen Egg White Lysozyme Crystals. Crystals 2017, 7, 170. [Google Scholar] [CrossRef]
- Li, F.; Lakerveld, R. Influence of Alternating Electric Fields on Protein Crystallization in Microfluidic Devices with Patterned Electrodes in a Parallel-Plate Configuration. Cryst. Growth Des. 2017, 17, 3062–3070. [Google Scholar] [CrossRef]
- Rubin, E.; Owen, C.; Stojanoff, V. Crystallization under an External Electric Field: A Case Study of Glucose lsomerase. Crystals 2017, 7, 206. [Google Scholar] [CrossRef]
- Taleb, M.; Didierjean, C.; Jelsch, C.; Mangeot, J.P.; Capelle, B.; Aubry, A. Crystallization of proteins under an external electric field. J. Cryst. Growth 1999, 200, 575–582. [Google Scholar] [CrossRef]
- Nanev, C.N.; Penkova, A. Nucleation of lysozyme crystals under external electric andultrasonic fields. J. Cryst. Growth 2001, 232, 285–293. [Google Scholar] [CrossRef]
- Nanev, C.N.; Penkova, A. Nucleation and growth of lysozyme crystals under external electric field. Colloids. Surf. A 2002, 209, 139–145. [Google Scholar] [CrossRef]
- Mirkin, N.; Frontana-Uribe, B.A.; Rodríguez-Romero, A.; Hernández-Santoyo, A.; Moreno, A. The influence of an internal electric field upon protein crystallization using the gel-acupuncture method. Acta Crystallogr. Sect. D Biol. Crystallogr. 2003, 59, 1533–1538. [Google Scholar] [CrossRef]
- Profio, G.D.; Reijonen, M.T.; Caliandro, R.; Guagliardi, A.; Curcio, E.; Drioli, E. Insights into the polymorphism of glycine: Membrane crystallization in an electric field. Phys. Chem. Chem. Phys. 2013, 15, 9271. [Google Scholar] [CrossRef]
- Moreno, A.; Sazaki, G. The use of a new ad hoc growth cell with parallel electrodes for the nucleation control of lysozyme. J. Cryst. Growth 2004, 264, 438–444. [Google Scholar] [CrossRef]
- Hammadi, Z.; Astier, J.P.; Morin, R.; Veesler, S. Protein Crystallization Induced by a Localized Voltage. Cryst. Growth. Des. 2007, 7, 1472–1475. [Google Scholar] [CrossRef]
- Nieto-Mendoza, E.; Frontana-Uribe, B.A.; Sazaki, G.; Moreno, A. Investigations on electromigration phenomena for protein crystallization using crystal growth cells with multiple electrodes: Effect of the potential control. J. Cryst. Growth 2005, 275, 1437–1446. [Google Scholar] [CrossRef]
- Chen, C.-J.; Zhu, W.-B.; Chao, J.-H.; Shang, A.; Lee, Y.G.; Liu, R.-J.; Yin, S.S.; Mark, D.; Hoffman, R.C. Study of thermal and spatial dependent electric field-induced phase transition in relaxor ferroelectric crystals using Raman spectroscopy. J. Alloys Compd. 2019, 804, 35–41. [Google Scholar] [CrossRef]
- Chen, Y.-L.; Wang, Z.-W.; Chen, S.; Ren, H.; Li, B.-W.; Yan, W.-S.; Zhang, G.-B.; Jiang, J.; Zou, C.-W. Electric-field Control of Li-Doping Induced Phase Transition in VO2 Film with Crystal Facet-Dependence. Nano. Energy 2018, 51, 300–307. [Google Scholar] [CrossRef]
- Okamura, Y.; Kagawa, F.; Seki, S.; Tokura, Y. Transition to and from the skyrmion lattice phase by electric fields in a magnetoelectric compound. Nat. Commun. 2016, 7, 12669. [Google Scholar] [CrossRef] [PubMed]
- Zaragoza, A.; Espinosa, J.; Ramos, R.; Cobos, J.A.; Aragones, J.L.; Vega, C.; Sanz, E.; Ramirez, J.; Valeriani, C. Phase boundaries, nucleation rates and speed of crystal growth of the water-to-ice transition under an electric field: A simulation study. J. Phy-Condens. Mat. 2018, 30, 174002. [Google Scholar] [CrossRef]
- Deng, J.-K.; Chang, Z.-Y.; Zhao, T.; Ding, X.-D.; Sun, J.; Liu, J.Z. Electric Field Induced Reversible Phase Transition in Li Doped Phosphorene: Shape Memory Effect and Superelasticity. J. Am. Chem. Soc. 2016, 138, 4772–4778. [Google Scholar] [CrossRef]
- Sawahata, H.; Yamaguchi, N.Y.; Fumiyuki, I. Electric-field-induced Z2 topological phase transition in strained single bilayer Bi(111). Appl. Phys. Express 2019, 12, 075009. [Google Scholar] [CrossRef]
- Sawahata, H.; Yamaguchi, N.Y.; Kotaka, H.; Fumiyuki, I. First-principles study of electric-field-induced topological phase transition in one-bilayer Bi(111). Jpn. J. Appl. Phys. 2018, 57, 030309. [Google Scholar] [CrossRef]
- Collins, J.L.; Tadich, A.; Wu, W.; Gomes, L.C.; Rodrigues, J.N.B.; Liu, C.; Hellerstedt, J.; Ryu, H.; Tang, S.; Mo, S.-K.; et al. Electric-field-tuned topological phase transition in ultrathin Na3Bi. Nature 2018, 564, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Matvija, P.; Rozbořil, F.; Sobotík, P.; Ošťádal, I.; Pieczyrak, B.; Jurczyszyn, L.; Kocán, P. Electric-field-controlled phase transition in a 2D molecular layer. Sci. Rep. 2017, 7, 7357. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D.S.; Thomas, R.; Katiyar, R.S.; Scott, J.F.; Kohlstedt, H.; Petraru, A.; Hwang, C.S. Emerging memories: Resistive switching mechanisms and current status. Rep. Prog. Phys. 2012, 28, 076502. [Google Scholar] [CrossRef] [PubMed]
- Mallah, T.; Cavallini, M. Surfaces, thin films and patterning of spin crossover compounds. C. R. Chim. 2018, 21, 1270–1286. [Google Scholar] [CrossRef]
- Dickson, A.; Warmflash, A.; Dinner, A.R. Nonequilibrium umbrella sampling in spaces of many order parameters. J. Chem. Phys. 2009, 130, 074104. [Google Scholar] [CrossRef] [PubMed]
- Faradjian, A.K.; Elber, R. Computing time scales from reaction coordinates by milestoning. J. Chem. Phys. 2004, 120, 10880–10889. [Google Scholar] [CrossRef]
- Santiso, E.E.; Trout, B.L. A general set of order parameters for molecular crystals. J. Chem. Phys. 2011, 134, 064109. [Google Scholar] [CrossRef] [PubMed]
- Carter, E.; Ciccotti, G.; Hynes, J.; Kapral, R. Constrained reaction coordinate dynamics for the simulation of rare events. Chem. Phys. Lett. 1989, 156, 472–477. [Google Scholar] [CrossRef]
- Anwar, J.; Zahn, D. Uncovering molecular processes in crystal nucleation and growth by using molecular simulation. Angew. Chem. Int. Ed. 2011, 50, 1996–2013. [Google Scholar] [CrossRef]
- Zinovjev, K.; Tuñón, I. Adaptive finite temperature string method in collective variables. J. Phys. Chem. A 2017, 121, 9764–9772. [Google Scholar] [CrossRef]
- Dickson, B.M.; Huang, H.; Post, C.B. Unrestrained computation of free energy along a path. J. Phys. Chem. B 2012, 116, 11046–11055. [Google Scholar] [CrossRef]
- Díaz, L.G.; Ensing, B. Path finding on high-dimensional free energy landscapes. Phys. Rev. Lett. 2012, 109, 020601. [Google Scholar] [CrossRef]
- Maragliano, L.; Roux, B.; Vanden-Eijnden, E. Comparison between mean forces and swarms-of-trajectories string methods. J. Chem. Theory Comput. 2014, 10, 524–533. [Google Scholar] [CrossRef]
- Song, H.-D.; Zhu, F.-Q. Finite temperature string method with umbrella sampling: Application on a side chain flipping in Mhp1 transporter. J. Phys. Chem. B 2017, 121, 3376–3386. [Google Scholar] [CrossRef]
- Cao, L.R.; Lv, C.; Yang, W. Hidden conformation events in DNA Base extrusions: A generalized-ensemble path optimization and equilibrium simulation study. J. Chem. Theory. Comput. 2013, 9, 3756–3768. [Google Scholar] [CrossRef]
- Weinan, E.; Ren, W.; Vanden-Eijnden, E. String method for the study of rare events. Phys. Rev. B Condens. Matter Mater. Phys. 2002, 66, 052301. [Google Scholar]
- Vanden-Eijnden, E.; Venturoli, M. Revisiting the finite temperature string method for thecalculation of reaction tubes and free energies. J. Chem. Phys. 2009, 130, 194103. [Google Scholar] [CrossRef] [PubMed]
- Bellucci, M.A.; Trout, B.L. Bezier curve string method for the study of rare events in complex chemical systems. J. Chem. Phys. 2014, 141, 074110. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.; Santiso, E.E.; Trout, B.L. Computer simulations of homogeneous nucleation of benzene from the melt. J. Phys. Chem. B 2011, 115, 10400–10412. [Google Scholar] [CrossRef]
- Gobbo, G.; Bellucci, M.A.; Tribello, G.A.; Ciccotti, G.; Trout, B.L. Nucleation of molecular crystals driven by relative information entropy. J. Chem. Theory Comput. 2018, 14, 959–972. [Google Scholar] [CrossRef]
- Mac, Q.J. Some methods for classification and analysis of multivariate observations. In Proceedings of the Fifth Berkeley Symposium on Mathematical Statistics and Probability; Berkeley Symposium on Mathematical Statistics and Probability: Berkeley, CA, USA, 1967. [Google Scholar]
- Abo-Elnaga, Y.; Nasr, S. K-means cluster interactive algorithm-based evolutionary approach for solving bilevel multi-objective programming problems. Alex. Eng. J. 2021, 61, 811–827. [Google Scholar] [CrossRef]
- Rong, H.; Ramirez-Serrano, A.; Guan, L.; Gao, Y. lmage object extraction based on semantic detection and improved K-Means agorithm. IEEE Access 2020, 8, 171129–171139. [Google Scholar] [CrossRef]
- Ren, F.-D.; Liu, Y.-Z.; Ding, K.-W.; Chang, L.-L.; Cao, D.-L.; Liu, S.-B. Finite temperature string by K-means clustering sampling with order parameter as collective variables for molecular crystal: Application to polymorphic transformation between β-CL-20 and ε-CL-20. Phys. Chem. Chem. Phys. 2024, 26, 3500–3515. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Rohani, S.; Gong, J.; Wang, J. Recent Developments in the Crystallization Process: Toward the Pharmaceutical Industry. Engineering 2017, 3, 343–353. [Google Scholar] [CrossRef]
- Aber, J.E.; Arnold, S.; Garetz, B.A.; Myerson, A.S. Strong dc Electric Field Applied to Supersaturated Aqueous Glycine Solution Induces Nucleation of the γ Polymorph. Phys. Rev. Lett. 2005, 94, 145503. [Google Scholar] [CrossRef] [PubMed]
- Jha, P.K.; Sadot, M.; Vino, S.A.; Jury, V.; Curet-Ploquin, S.; Rouaud, O.; Havet, M.; Le-Bail, A. A review on effect of DC voltage on crystallization process in food systems. Innov. Food. Sci. Emerg. 2017, 42, 204–219. [Google Scholar] [CrossRef]
- Panda, S. The Internet of Things: Breakthroughs in Research and Practice; IGI Global: Pennsylvania, PA, USA, 2017. [Google Scholar]
- Myerson, A.S.; Ginde, R. Industrial Crystallization of Melts; CRC Press: Boca Raton, FL, USA, 2004; pp. 183–240. [Google Scholar]
- Chen, F.; Zhou, T.; Li, J.; Wang, X.; Cao, D.; Wang, J.; Yang, Z. Crystal morphology of dihydroxylammonium 5,5′-bistetrazole-1,1′-diolate (TKX-50) under solvents system with different polarity using molecular dynamics. Comp. Mater. Sci. 2019, 168, 48–57. [Google Scholar] [CrossRef]
- Xiong, S.-L.; Chen, S.-S.; Jin, S.-H.; Li, L.-J. Additives effects on crystal morphology of dihydroxylammonium 5,5’-bistetrazole-1,1’-diolate by molecular dynamics simulations. J. Energ. Mater. 2016, 34, 384–394. [Google Scholar] [CrossRef]
- Xu, X.; Chen, D.; Li, H.; Xu, R.; Zhao, H. Crystal Morphology Modification of 5, 5′-Bisthiazole-1, 1′-dioxyhydroxyammonium Salt. ChemistrySelect 2020, 5, 1919–1924. [Google Scholar] [CrossRef]
- Xiao, L.; Guo, S.; Su, H.; Gou, B.; Liu, Q.; Hao, G.; Hu, Y.; Wang, X.; Jiang, W. Preparation and characteristics of a novel PETN/TKX-50 co-crystal by a solvent/non-solvent method. RSC Adv. 2019, 9, 9204–9210. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-J.; Su, Q.; Chen, S.-S. Synthesis of cumulative nitrogen rich compound of dihydroxylammonium 5,5-bistetrazole -1,1-diolate (TKX-50). Initiators Pyrotech. 2014, 3, 38–41. [Google Scholar]
- Maragliano, L.; Fischer, A.; Vanden-Eijnden, E.; Ciccotti, G. String method in collective variables: Minimum free energypaths and isocommittor surfaces. J. Chem. Phys. 2006, 125, 024106. [Google Scholar] [CrossRef] [PubMed]
- Beckham, G.T.; Peters, B.; Starbuck, C.; Variankaval, N.; Trout, B.L. Surface-mediated nucleation in the solid-state polymorph transformation of terephthalic acid. J. Am. Chem. Soc. 2007, 129, 4714–4723. [Google Scholar] [CrossRef] [PubMed]
- Santiso, E.E.; Trout, B.L. A general method for molecular modeling of nucleation from the melt. J. Chem. Phys. 2015, 143, 174109. [Google Scholar] [CrossRef]
- Maragliano, L.; Vanden-Eijnden, E. On-the-fly string method for minimum free energy paths calculation. Chem. Phys. Lett. 2007, 446, 182–190. [Google Scholar] [CrossRef]
- He, X.; Shen, Y.; Hung, F.R.; Santiso, E.E. Molecular simulation of homogeneous nucleation of crystals of an ionic liquid from the melt. J. Chem. Phys. 2015, 143, 124506. [Google Scholar] [CrossRef]
- Beckham, G.T.; Peters, B.; Trout, B.L. Evidence for a size dependent nucleation mechanism in solid state polymorph transformations. J. Phys. Chem. B 2008, 112, 7460–7466. [Google Scholar] [CrossRef]
- Koizumi, H.; Fujiwara, K.; Uda, S. Control of Nucleation Rate for Tetragonal Hen-Egg White Lysozyme Crystals by Application of an Electric Field withVariable Frequencies. Cryst. Growth Des. 2009, 9, 2420–2424. [Google Scholar] [CrossRef]
- Zhu, Z.-S.; Jiang, Z.-M.; Wang, P.-C.; Lu, M.; Shu, Q.; Yu, X.-H. Synthesis and Properties of Dihydroxylammonium 5,5’-Bistetrazole-1,1’-diolate. Chin. J. Energ. Mater. 2014, 3, 332–336. [Google Scholar]
- Darden, T.; York, D.; Pedersen, L. Particle mesh Ewald: An N.log(N) method for Ewald sums in large systems. J. Chem. Phys. 1993, 98, 10089–10092. [Google Scholar] [CrossRef]
- Hess, B.; Bekker, H.; Berendsen, H.J.C.; Fraaije, J. LINCS: A linear constraint solver for molecular simulations. J. Comput. Chem. 1997, 18, 1463–1472. [Google Scholar] [CrossRef]
- Phillips, J.C.; Braun, R.; Wang, W.; Gumbart, J.C.; Tajkhorshid, E.; Villa, E.; Chipot, C.; Skeel, R.D.; Kale, L.V.; Schulten, K. Scalable molecular dynamics with NAMD. J. Comput. Chem. 2005, 26, 1781–1802. [Google Scholar] [CrossRef] [PubMed]
- Kamlet, M.J.; Jacobs, S.J. Chemistry of detonations. I. A simple method for calculating detonation properties of C-H-N-O explosives. J. Chem. Phys. 1968, 48, 23–35. [Google Scholar] [CrossRef]


| r (Å) | 1/σ2 (Å−1) | (°) | (°) | ||
|---|---|---|---|---|---|
| 5.53 (5.46) | 28.56 | 11.53 (12.68) | 31.25 | 75.31 (69.68) | 11.26 |
| 7.56 (7.62) | 5.19 | 69.82 (67.53) | 28.62 | 43.26 (44.52) | 23.18 |
| 8.69 (8.43) | 16.40 | 78.93 (83.26) | 10.63 | 14.83 (16.83) | 17.53 |
| r (Å) | 1/σ2 (Å−1) | (°) | (°) | ||
|---|---|---|---|---|---|
| 5.62 a (5.50) b 6.03 c | 18.69 (26.50) 15.83 | 42.82 (61.29) 68.66 | 32.83 (18.39) 30.26 | 25.69 (27.57) 19.68 | 11.53 (12.63) 9.10 |
| 7.56 (7.16) 6.98 | 29.02 (10.31) 6.28 | 69.82 (66.53) 70.15 | 25.72 (26.17) 36.01 | 32.55 (11.53) 28.62 | 20.12 (16.93) 22.26 |
| 8.69 (8.53) 8.55 | 15.62 (23.19) 21.51 | 78.18 (82.31) 80.66 | 15.70 (18.63) 19.25 | 16.76 (15.92) 17.39 | 23.17 (9.13) 16.52 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, F.; Wang, X.; Zhang, Q.; Wang, X.; Chang, L.; Zhang, Z. Experimental and Theoretical Investigation of External Electric-Field-Induced Crystallization of TKX-50 from Solution by Finite-Temperature String with Order Parameters as Collective Variables for Ionic Crystals. Molecules 2024, 29, 1159. https://doi.org/10.3390/molecules29051159
Ren F, Wang X, Zhang Q, Wang X, Chang L, Zhang Z. Experimental and Theoretical Investigation of External Electric-Field-Induced Crystallization of TKX-50 from Solution by Finite-Temperature String with Order Parameters as Collective Variables for Ionic Crystals. Molecules. 2024; 29(5):1159. https://doi.org/10.3390/molecules29051159
Chicago/Turabian StyleRen, Fude, Xiaolei Wang, Qing Zhang, Xiaojun Wang, Lingling Chang, and Zhiteng Zhang. 2024. "Experimental and Theoretical Investigation of External Electric-Field-Induced Crystallization of TKX-50 from Solution by Finite-Temperature String with Order Parameters as Collective Variables for Ionic Crystals" Molecules 29, no. 5: 1159. https://doi.org/10.3390/molecules29051159
APA StyleRen, F., Wang, X., Zhang, Q., Wang, X., Chang, L., & Zhang, Z. (2024). Experimental and Theoretical Investigation of External Electric-Field-Induced Crystallization of TKX-50 from Solution by Finite-Temperature String with Order Parameters as Collective Variables for Ionic Crystals. Molecules, 29(5), 1159. https://doi.org/10.3390/molecules29051159





