Multifunctional Applications of Ionic Liquids in Polymer Materials: A Brief Review
Abstract
1. Introduction
2. Filler Modification by ILs
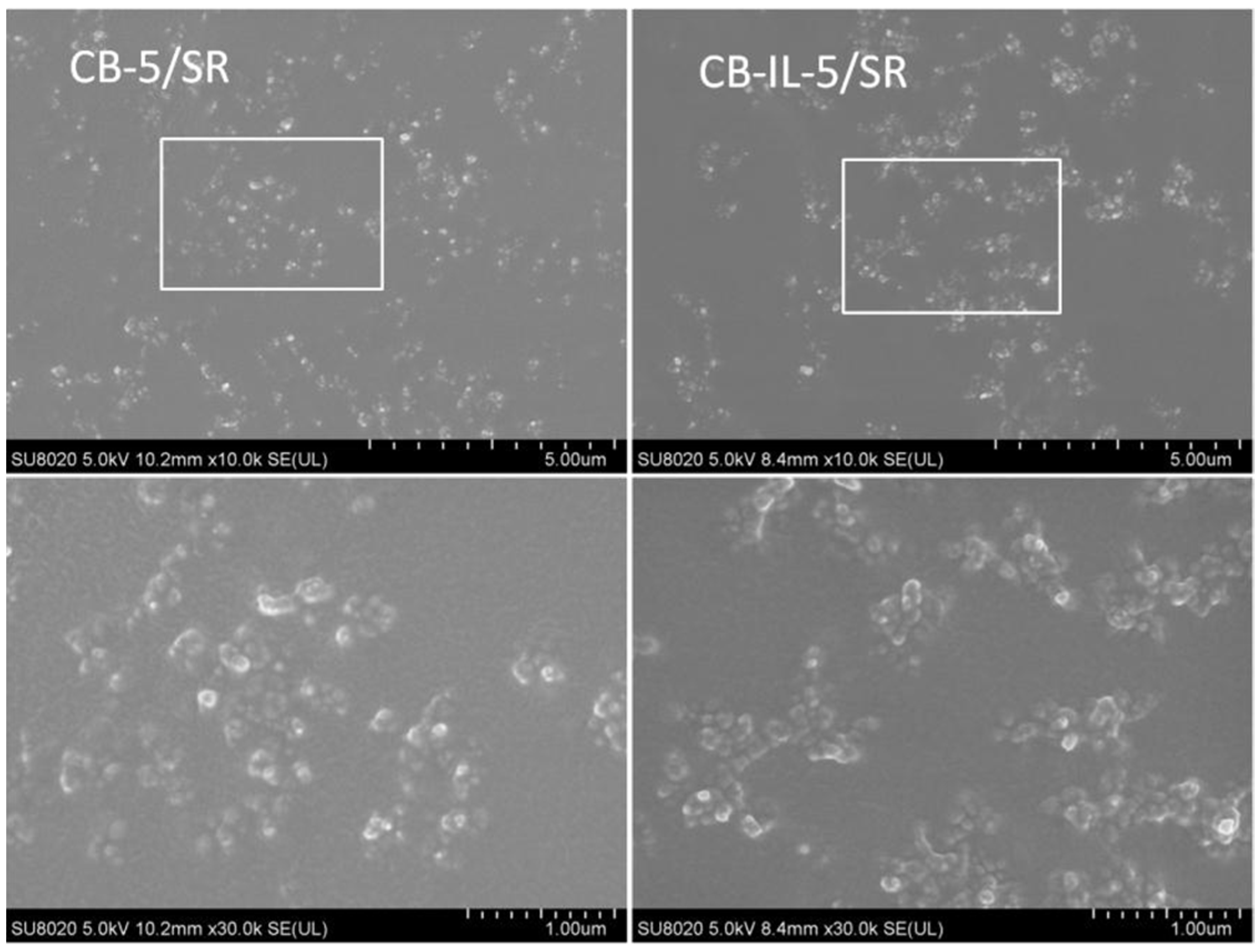
2.1. Carbon Black
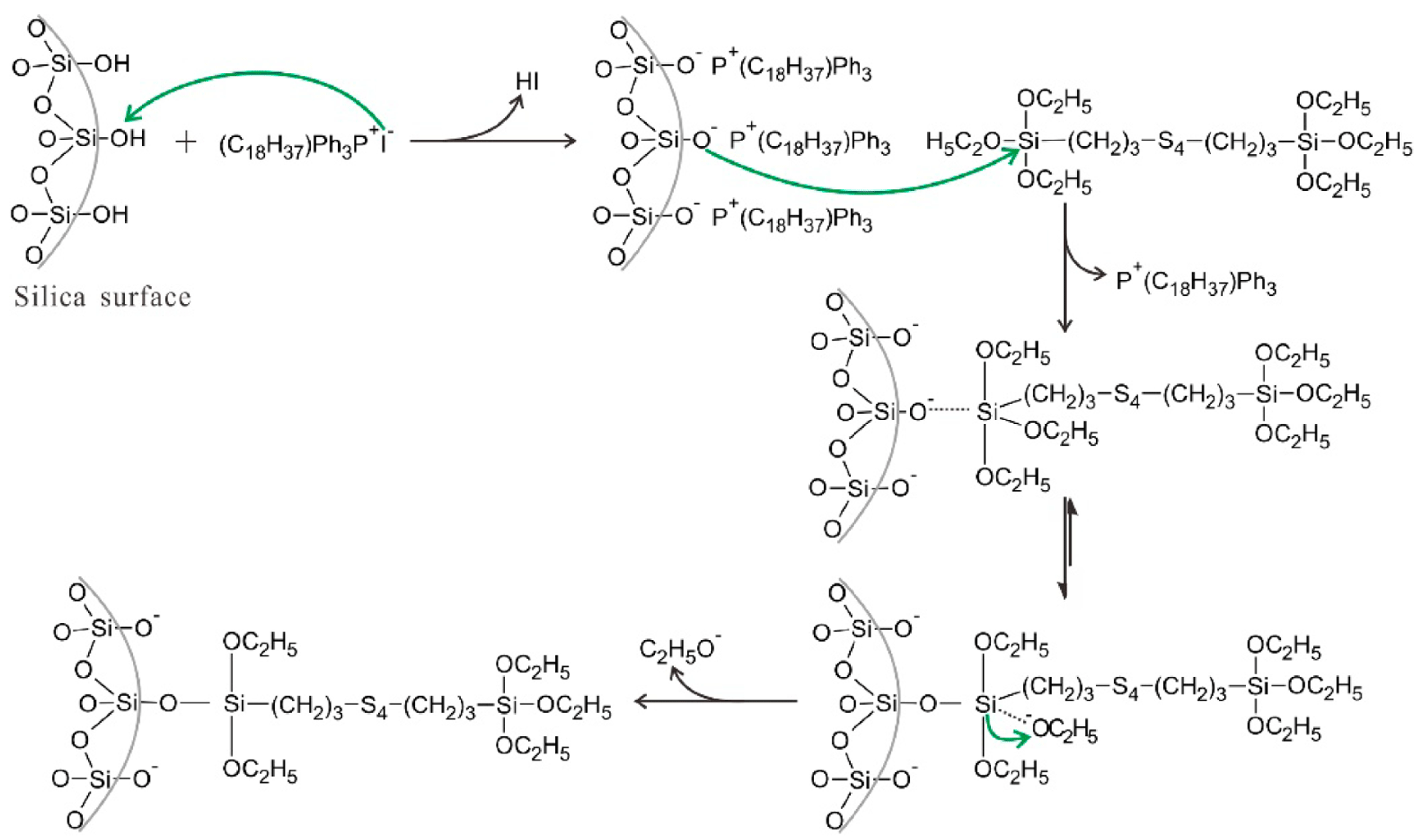
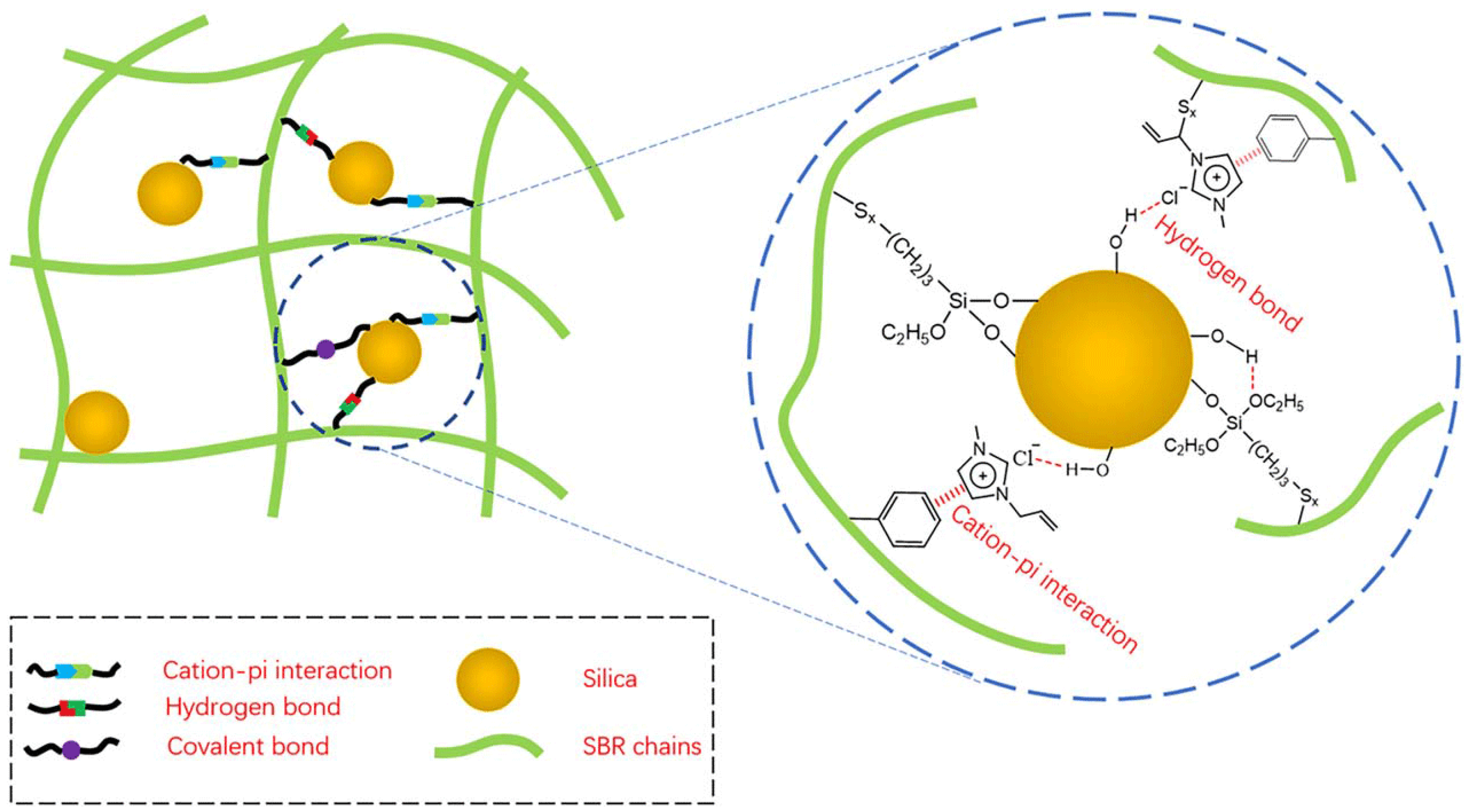
2.2. Silica
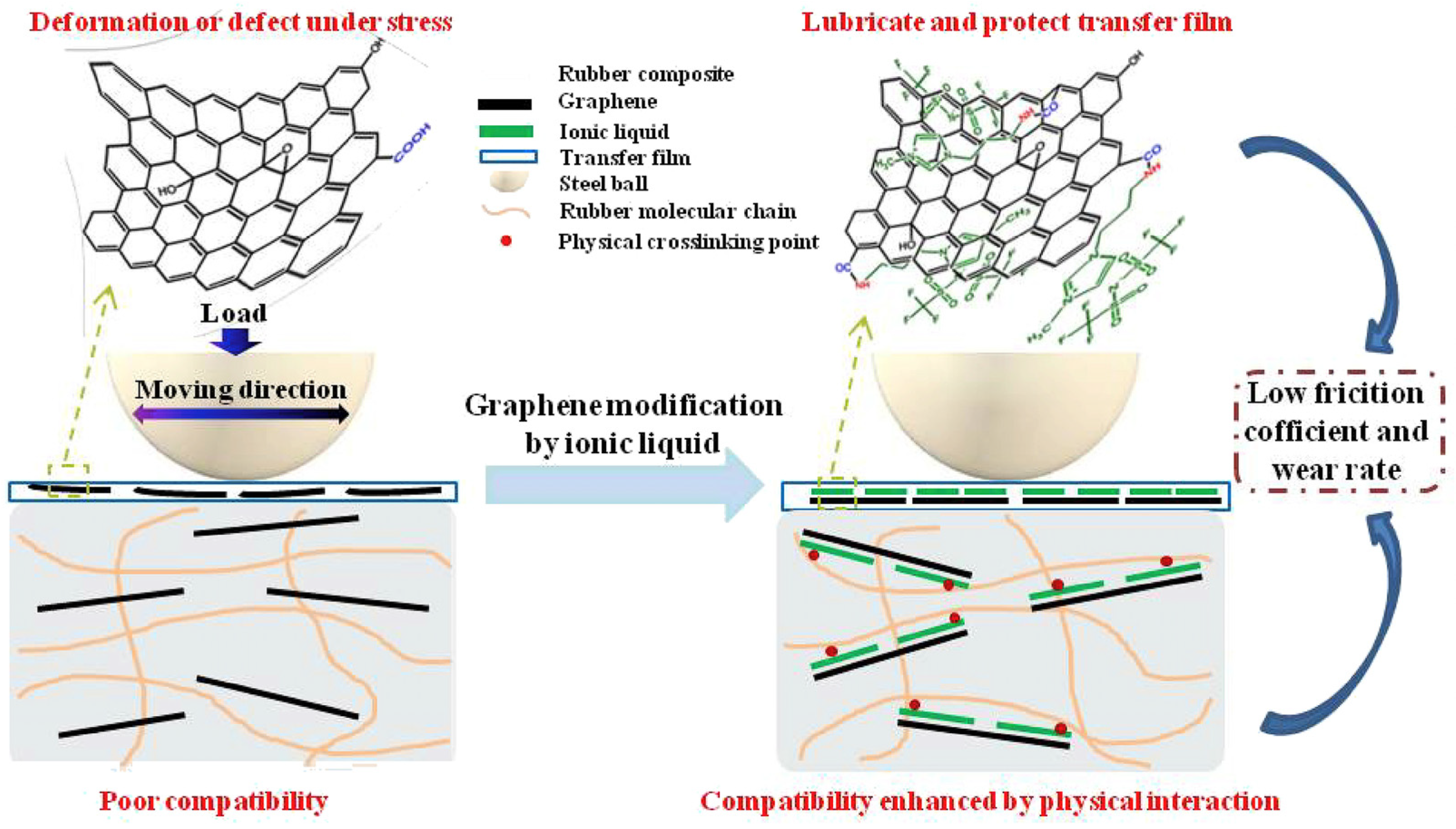
2.3. Graphene Oxide
2.4. Multi-Walled Carbon Nanotubes
2.5. Other Fillers
3. Application of ILs in Polymer Materials
3.1. Multiple Functions of ILs in Rubber Vulcanization
3.2. Applications in the Thermal Stability of Rubber
3.3. Applications in the Thermal Conductivity of Rubber
3.4. Applications in Electrical Conductivity of Rubber
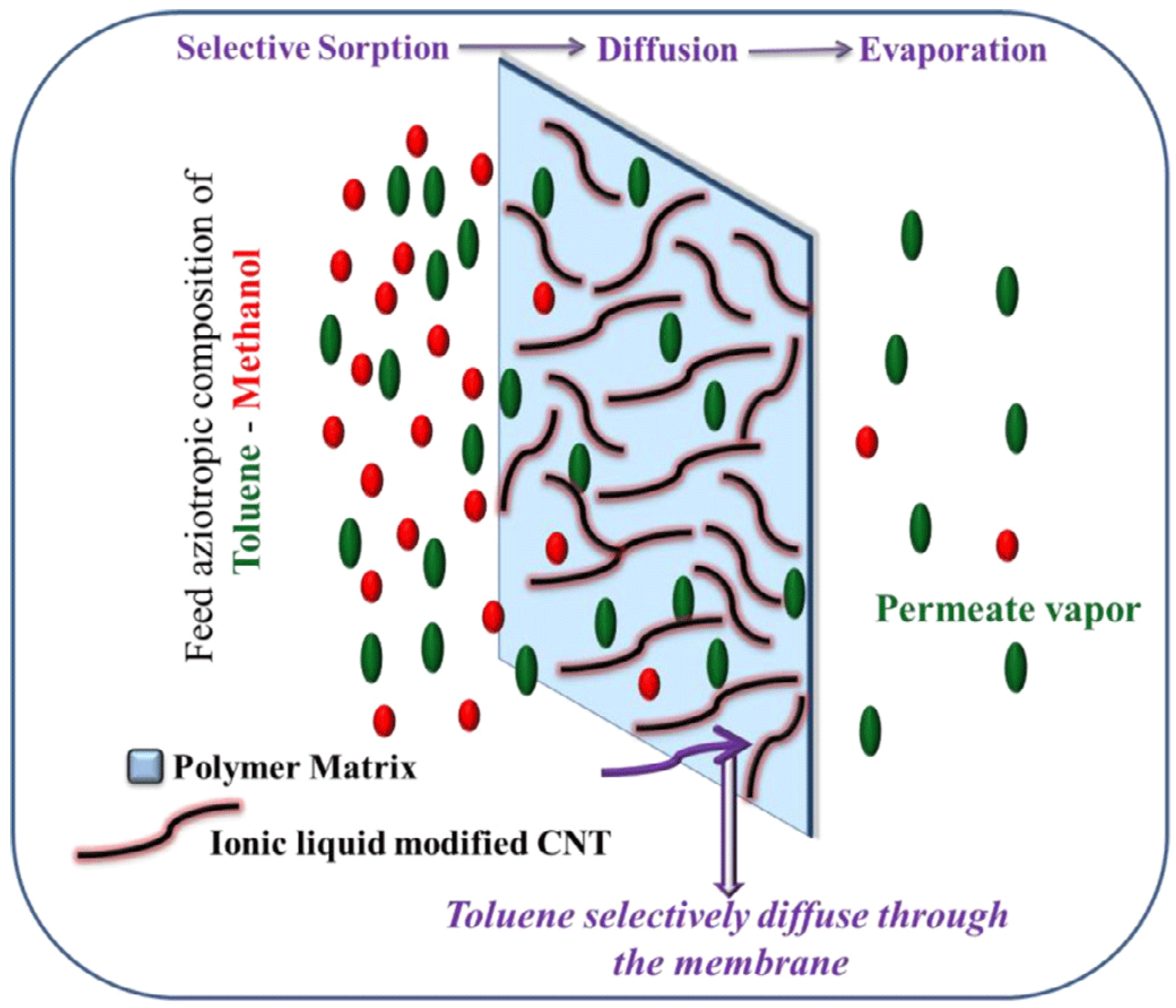
3.5. Applications in Polymeric Selectity and Permeability
3.6. Application of ILs in Electromagnetic Shielding
3.7. Application of ILs in Piezoresistive Sensitivity
3.8. Multiple Functions of ILs in Energy
4. Summary and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Osada, I.; de Vries, H.; Scrosati, B.; Passerini, S. Ionic-liquid-based polymer electrolytes for battery applications. Angew. Chem. Int. Ed. Engl. 2016, 55, 500–513. [Google Scholar] [CrossRef] [PubMed]
- Xue, B.; Wu, X.; Guo, Y.; Zhang, C.; Qian, W.; Zhang, L. Ionic liquids applications in flow batteries. J. Electrochem. Soc. 2022, 169, 080501. [Google Scholar] [CrossRef]
- Hassanshahi, N.; Hu, G.; Li, J. Application of Ionic liquids for chemical demulsification: A review. Molecules 2020, 25, 4915–4941. [Google Scholar] [CrossRef] [PubMed]
- Gholinejad, M.; Zareh, F.; Sheibani, H.; Nájera, C.; Yus, M. Magnetic ionic liquids as catalysts in organic reactions. J. Mol. Liq. 2022, 367, 120395–120441. [Google Scholar] [CrossRef]
- Singh, S.K.; Savoy, A.W. Ionic liquids synthesis and applications: An overview. J. Mol. Liq. 2020, 297, 112038–1122060. [Google Scholar] [CrossRef]
- Tamate, R.; Hashimoto, K.; Ueki, T.; Watanabe, M. Block copolymer self-assembly in ionic liquids. PCCP 2018, 20, 25123–25139. [Google Scholar] [CrossRef]
- Wang, W.X.; Kong, Y.; Peng, J.J.; Li, B.; Xu, H.F. Probing the mechanism of green solvent solubilization of hemicellulose based on molecular dynamics simulations. Ind. Crop. Prod. 2022, 186, 115159–115168. [Google Scholar] [CrossRef]
- Kaur, G.; Kumar, H.; Singla, M. Diverse applications of ionic liquids: A comprehensive review. J. Mol. Liq. 2022, 351, 118556–118574. [Google Scholar] [CrossRef]
- Silva, W.; Zanatta, M.; Ferreira, A.S.; Corvo, M.C.; Cabrita, E.J. Revisiting ionic liquid structure-property relationship: A critical analysis. Int. J. Mol. Sci. 2020, 21, 7745. [Google Scholar] [CrossRef]
- Zhang, S.; Sun, N.; He, X.; Lu, X.; Zhang, X. Physical properties of ionic liquids: Database and evaluation. J. Phys. Chem. Ref. Data 2006, 35, 1475–1517. [Google Scholar] [CrossRef]
- Zhang, S.; Sun, N.; Zhang, X.; Lv, X. Periodicity and map for discovery of new ionic liquids. Sci. China Ser. B 2006, 49, 103–115. [Google Scholar] [CrossRef]
- Narongthong, J.; Das, A.; Le, H.H.; Wiessner, S.; Sirisinha, C. An efficient highly flexible strain sensor: Enhanced electrical conductivity, piezoresistivity and flexibility of a strongly piezoresistive composite based on conductive carbon black and an ionic liquid. Compos. Part A 2018, 113, 330–338. [Google Scholar] [CrossRef]
- Abraham, J.; Sisanth, K.S.; Zachariah, A.K.; Mariya, H.J.; George, S.C.; Kalarikkal, N.; Thomas, S. Transport and solvent sensing characteristics of styrene butadiene rubber nanocomposites containing imidazolium ionic liquid modified carbon nanotubes. J. Appl. Polym. Sci. 2020, 137, 49429–49438. [Google Scholar] [CrossRef]
- Szadkowski, B.; Marzec, A.; Zaborski, M. Use of carbon black as a reinforcing nano-filler in conductivity-reversible elastomer composites. Polym. Test. 2020, 81, 106222–106251. [Google Scholar] [CrossRef]
- Zhang, D.; Shi, Y.; An, J.; Yang, S.; Li, B. Triallyl cyanurate copolymerization delivered nonflammable and fast ion conducting elastic polymer electrolytes. J. Mater. Chem. A 2022, 10, 23095–23102. [Google Scholar] [CrossRef]
- Lewandowski, A. New composite solid electrolytes based on a polymer and ionic liquids. Solid State Ionics 2004, 169, 21–24. [Google Scholar] [CrossRef]
- Li, Y.; Shao, W.; Ma, Z.; Zheng, M.; Song, H. Performance Analysis of a HT-PEMFC System with 6FPBI Membranes Doped with Cross-Linkable Polymeric Ionic Liquid. Int. J. Mol. Sci. 2022, 23, 9618. [Google Scholar] [CrossRef]
- Klapiszewski, L.; Szalaty, T.J.; Kurc, B.; Stanisz, M.; Skrzypczak, A.; Jesionowski, T. Functional hybrid materials based on manganese dioxide and lignin activated by ionic liquids and their application in the production of lithium ion batteries. Int. J. Mol. Sci. 2017, 18, 1509. [Google Scholar] [CrossRef]
- Chen, Y.; Tao, J.; Deng, L.; Li, L.; Li, J.; Yang, Y.; Khashab, N.M. Polyetherimide/bucky gels nanocomposites with superior conductivity and thermal stability. ACS. Appl. Mater. Interfaces 2013, 5, 7478–7484. [Google Scholar] [CrossRef]
- Therattil, J.A.; Pothan, L.A.; Maria, H.J.; Kalarikal, N.; Thomas, S. Natural rubber/carbon nanotube/Ionic liquid composite membranes: Vapor permeation and gas permeability properties. Macromol. Symp. 2020, 398, 2000222–2000232. [Google Scholar] [CrossRef]
- Qiu, M.; Zhang, B.; Wu, H.; Cao, L.; He, X.Y.; Li, Y.; Li, J.Z.; Xu, M.Z.; Jiang, Z.Y. Preparation of anion exchange membrane with enhanced conductivity and alkaline stability by incorporating ionic liquid modified carbon nanotubes. J. Membr. Sci. 2019, 573, 1–10. [Google Scholar] [CrossRef]
- Rhyu, S.Y.; Cho, Y.; Kang, S.W. Nanocomposite membranes consisting of poly(ethylene oxide)/ionic liquid/ZnO for CO2 separation. J. Ind. Eng. Chem. 2020, 85, 75–80. [Google Scholar] [CrossRef]
- Huang, G.J.; Isfahani, A.P.; Muchtar, A.; Sakurai, K.; Shrestha, B.B.; Qin, D.T.; Yamaguch, D.; Sivaniah, E.; Ghalei, B. Pebax/ionic liquid modified graphene oxide mixed matrix membranes for enhanced CO2 capture. J. Membr. Sci. 2018, 565, 370–379. [Google Scholar] [CrossRef]
- Zhang, R.; Ke, Q.; Zhang, Z.; Zhou, B.; Cui, G.; Lu, H. Tuning functionalized ionic liquids for CO2 capture. Int. J. Mol. Sci. 2022, 23, 11401. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Shang, Y.; Zhou, Y.; Guo, J.; Yan, F. Enabling antibacterial and antifouling coating via grafting of a nitric oxide-releasing ionic liquid on silicone rubber. Biomacromolecules 2022, 23, 2329–2341. [Google Scholar] [CrossRef]
- Maciejewska, M.; Sowinska-Baranowska, A. Bromide and chloride ionic liquids applied to enhance the vulcanization and performance of natural rubber biocomposites filled with nanosized silica. Nanomaterials 2022, 12, 1209–1231. [Google Scholar] [CrossRef]
- Sowinska, A.; Maciejewska, M.; Grajewska, A. Bis (trifluoromethylsulfonyl) imide ionic liquids applied for fine-tuning the cure characteristics and performance of natural rubber composites. Int. J. Mol. Sci. 2021, 22, 3678. [Google Scholar] [CrossRef]
- Caldas, C.M.; Soares, B.G.; Indrusiak, T.; Barra, G.M.O. Ionic liquids as dispersing agents of graphene nanoplatelets in poly(methyl methacrylate) composites with microwave absorbing properties. J. Appl. Polym. Sci. 2020, 138, 49814–49831. [Google Scholar] [CrossRef]
- Członka, S.; Strąkowska, A.; Strzelec, K.; Kairytė, A.; Vaitkus, S. Composites of rigid polyurethane foams and silica powder filler enhanced with ionic liquid. Polym. Test. 2019, 75, 12–25. [Google Scholar] [CrossRef]
- Mondal, T.; Basak, S.; Bhowmick, A.K. Ionic liquid modification of graphene oxide and its role towards controlling the porosity, and mechanical robustness of polyurethane foam. Polymer 2017, 127, 106–118. [Google Scholar] [CrossRef]
- Sidorov, O.I.; Vygodskii, Y.S.; Lozinskaya, E.I.; Milekhin, Y.M.; Matveev, A.A.; Poisova, T.P.; Ferapontov, F.V.; Sokolov, V.V. Ionic liquids as curing catalysts for epoxide-containing compositions. Polym. Sci. Ser. D 2017, 10, 134–142. [Google Scholar] [CrossRef]
- Nguyen, T.K.L.; Soares, B.G.; Duchet-Rumeau, J.; Livi, S. Dual functions of ILs in the core-shell particle reinforced epoxy networks: Curing agent vs dispersion aids. Compos. Sci. Technol. 2017, 140, 30–38. [Google Scholar] [CrossRef]
- Shi, K.; Luo, J.T.; Huan, X.H.; Lin, S.; Liu, X.; Jia, X.L.; Zu, L.; Cai, Q.; Yang, X.P. Ionic liquid-graphene oxide for strengthening microwave curing epoxy composites. ACS Appl. Nano Mater. 2020, 3, 11955–11969. [Google Scholar] [CrossRef]
- Liu, C.; Yu, C.Y.; Sang, G.L.; Xu, P.; Ding, Y.S. Improvement in EMI shielding properties of silicone rubber/POE blends containing ILs modified with carbon black and MWCNTs. Appl. Sci. 2019, 9, 1774. [Google Scholar] [CrossRef]
- Liu, C.; Wang, P.; Hu, J.P.; Cheng, S.; Pan, S.C.; Xu, P.; Ding, Y.S. Effect of phase morphology on electromagnetic interference shielding performance of silicone rubber/POE blends containing ILs modified MWCNTs. Synth. Met. 2019, 256, 116140–116145. [Google Scholar] [CrossRef]
- Shamsuri, A.A.; Abdan, K.; Kaneko, T. A concise review on the physicochemical properties of biopolymer blends prepared in ionic liquids. Molecules 2021, 26, 216–233. [Google Scholar] [CrossRef]
- Chu, L.; Kan, M.; Jerrams, S.; Zhang, R.; Xu, Z.; Liu, L.; Wen, S.; Zhang, L. Constructing chemical interface layers by using ionic liquid in graphene oxide/rubber composites to achieve high-wear resistance in environmental-friendly green tires. ACS Appl. Mater. Interfaces 2022, 14, 5995–6004. [Google Scholar] [CrossRef]
- Sahu, G.; Tripathy, J.; Sahoo, B.P. Significant enhancement of dielectric properties of graphene oxide filled polyvinyl alcohol nanocomposites: Effect of ionic liquid and temperature. Polym. Compos. 2020, 41, 4411–4430. [Google Scholar] [CrossRef]
- Qian, Z.; Peng, Z. Reinforcing styrene-butadiene rubber composites by constructing multiple interaction between rubber and silica. Polym. Compos. 2018, 40, 1740–1747. [Google Scholar] [CrossRef]
- Zhang, X.; Xue, X.; Jia, H.; Wang, J.; Ji, Q.; Xu, Z. Influence of ionic liquid on the polymer-filler coupling and mechanical properties of nano-silica filled elastomer. J. Appl. Polym. Sci. 2017, 134, 44478–44487. [Google Scholar] [CrossRef]
- Das, A.; Leuteritz, A.; Nagar, P.K.; Adhikari, B.; Stöckelhuber, K.; Jurk, R.; Heinrich, G. Improved gas barrier properties of composites based on ionic liquid integrated graphene nanoplatelets and bromobutyl rubber. Int. Polym. Sci. Technol. 2016, 43, 1–8. [Google Scholar] [CrossRef]
- Wang, J.; Jia, H.; Ding, L.; Xiong, X.; Gong, X. The mechanism of carbon-silica dual phase filler modified by ionic liquid and its reinforcing on natural rubber. Polym. Compos. 2015, 36, 1721–1730. [Google Scholar] [CrossRef]
- Fleck, F.; Froltsov, V.; Kluppel, M. Polymer-filler interphase dynamics and reinforcement of elastomer nanocomposites. Soft Mater. 2014, 12, 121–134. [Google Scholar] [CrossRef]
- Kreyenschulte, H.; Richter, S.; Gotze, T.; Fischer, D.; Steinhauser, D.; Kluppel, M.; Heinrich, G. Interaction of 1-allyl-3-methyl-imidazolium chloride and carbon black and its influence on carbon black filled rubbers. Carbon 2012, 50, 3649–3658. [Google Scholar] [CrossRef]
- Narongthong, J.; Wiessner, S.; Hait, S.; Sirisinha, C.; Stockelhuber, K.W. Strain-rate independent small-strain-sensor: Enhanced responsiveness of carbon black filled conductive rubber composites at slow deformation by using an ionic liquid. Compos. Sci. Technol. 2020, 188, 107972–107982. [Google Scholar] [CrossRef]
- Narongthong, J.; Le, H.H.; Das, A.; Sirisinha, C.; Wiessner, S. Ionic liquid enabled electrical-strain tuning capability of carbon black based conductive polymer composites for small-strain sensors and stretchable conductors. Compos. Sci. Technol. 2019, 174, 202–211. [Google Scholar] [CrossRef]
- Ge, Y.; Zhang, Q.Y.; Zhang, Y.D.; Liu, F.S.; Han, J.J.; Wu, C.F. High-performance natural rubber latex composites developed by a green approach using ionic liquid-modified multiwalled carbon nanotubes. J. Appl. Polym. Sci. 2018, 135, 46588–46594. [Google Scholar] [CrossRef]
- Le, H.H.; Wießner, S.; Das, A.; Fischer, D.; auf der Landwehr, M.; Do, Q.K.; Stöckelhuber, K.W.; Heinrich, G.; Radusch, H.J. Selective wetting of carbon nanotubes in rubber compounds—Effect of the ionic liquid as dispersing and coupling agent. Eur. Polym. J. 2016, 75, 13–24. [Google Scholar] [CrossRef]
- Shi, F.M.; Ma, Y.X.; Ma, J.; Wang, P.P.; Sun, W.X. Preparation and characterization of PVDF/TiO2 hybrid membranes with ionic liquid modified nano-TiO2 particles. J. Membr. Sci. 2013, 427, 259–269. [Google Scholar] [CrossRef]
- Sowinska-Baranowska, A.; Maciejewska, M.; Duda, P. The potential application of starch and walnut shells as biofillers for natural rubber (NR) composites. Int. J. Mol. Sci. 2022, 23, 7968. [Google Scholar] [CrossRef]
- Chen, R.; Hempelmann, R. Ionic liquid-mediated aqueous redox flow batteries for high voltage applications. Electrochem. Commun. 2016, 70, 56–59. [Google Scholar] [CrossRef]
- Zhang, Y.; Ye, R.; Henkensmeier, D.; Hempelmann, R.; Chen, R. “Water-in-ionic liquid” solutions towards wide electrochemical stability windows for aqueous rechargeable batteries. Electrochim. Acta 2018, 263, 47–52. [Google Scholar] [CrossRef]
- Sowinska-Baranowska, A.; Maciejewska, M. Influence of the silica specific surface area and ionic liquids on the curing characteristics and performance of styrene-butadiene rubber composites. Materials 2021, 14, 5302. [Google Scholar] [CrossRef] [PubMed]
- Maciejewska, M.; Sowinska, A. Influence of fillers and ionic liquids on the crosslinking and performance of natural rubber biocomposites. Polymers 2021, 13, 1656. [Google Scholar] [CrossRef] [PubMed]
- Chumnum, K.; Kalkornsurapranee, E.; Johns, J.; Sengloyluan, K.; Nakaramontri, Y. Combination of self-healing butyl rubber and natural rubber composites for improving the stability. Polymers 2021, 13, 443. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Fan, Y.; Kamran, U.; Park, S.J. Improved thermal conductivity and mechanical property of mercapto group-activated boron nitride/elastomer composites for thermal management. Compos. Part A 2022, 156, 106869–106877. [Google Scholar] [CrossRef]
- Wang, R.; Sun, Y.W.; Hou, Y.B.; Tian, Y.; Zhang, Y.D.; Liu, F.S.; Han, J.J. Effect of ionic liquid modified carbon nanotubes on the properties of nitrile butadiene rubber and nitrile butadiene latex nanocomposites. J. Appl. Polym. Sci. 2022, 139, e53020. [Google Scholar] [CrossRef]
- Członka, S.; Strąkowska, A.; Strzelec, K.; Kairytė, A.; Kremensas, A. Melamine, silica, and ionic liquid as a novel flame retardant for rigid polyurethane foams with enhanced flame retardancy and mechanical properties. Polym. Test. 2020, 87, 106511–106538. [Google Scholar] [CrossRef]
- Matchawet, S.; Kaesaman, A.; Vennemann, N.; Kummerlowe, C.; Nakason, C. Optimization of electrical conductivity, dielectric properties, and stress relaxation behavior of conductive thermoplastic vulcanizates based on ENR/COPA blends by adjusting mixing method and ionic liquid loading. Ind. Eng. Chem. Res. 2017, 56, 3629–3639. [Google Scholar] [CrossRef]
- Therattil, J.; Anil, K.S.; Pothan, L.A.; Rouxel, D.; Maria, H.J.; Kalarikal, N.; Thomas, S. Cure acceleration and plasticizing effect of imidazolium ionic liquid on the properties of natural rubber/carbon nanotube composites. Funct. Compos. Struct. 2020, 2, 035003–0350017. [Google Scholar] [CrossRef]
- Ke, K.; Potschke, P.; Gao, S.; Voit, B. An ionic liquid as interface linker for tuning piezoresistive sensitivity and toughness in poly(vinylidene fluoride)/carbon nanotube composites. ACS Appl. Mater. Interfaces 2017, 9, 5437–5446. [Google Scholar] [CrossRef]
- Yaragalla, S.; Sindam, B.; Abraham, J.; Raju, K.C.J.; Kalarikkal, N.; Thomas, S. Fabrication of graphite-graphene-ionic liquid modified carbon nanotubes filled natural rubber thin films for microwave and energy storage applications. J. Polym. Res. 2015, 22, 1–10. [Google Scholar]
- Abraham, J.; Jose, T.; George, S.C.; Kalarikkal, N.K.N.; Thomas, S. Mechanics and pervaporation performance of ionic liquid modified CNT based SBR membranes-a case study for the separation of toluene/heptane mixtures. Int. J. Membr. Sci. Technol. 2015, 2, 30–38. [Google Scholar]
- Abraham, J.; Zachariah, A.K.; Wilson, R.; Ibarra-Gomez, R.; Muller, R.; George, S.C.; Kalarikkal, N.; Thomas, S. Effect of ionic liquid modified MWCNT on the rheological and microstructural developments in styrene butadiene rubber nanocomposites. Rubber Chem. Technol. 2019, 92, 531–545. [Google Scholar] [CrossRef]
- Abraham, J.; Arif, M.; Xavier, P.; Bose, S.; George, S.C.; Kalarikkal, N.; Thomas, S. Investigation into dielectric behaviour and electromagnetic interference shielding effectiveness of conducting styrene butadiene rubber composites containing ionic liquid modified MWCNT. Polymer 2017, 112, 102–115. [Google Scholar] [CrossRef]
- Pingot, M.; Szadkowski, B.; Zaborski, M. Effect of carbon nanofibers on mechanical and electrical behaviors of acrylonitrile-butadiene rubber composites. Polym. Adv. Technol. 2018, 29, 1661–1669. [Google Scholar] [CrossRef]
- Soares, B.G.; Alves, F.F. Nanostructured epoxy-rubber network modified with MWCNT and ionic liquid: Electrical, dynamic-mechanical, and adhesion properties. Polym. Compos. 2018, 39 (S4), E2584–E2594. [Google Scholar] [CrossRef]
- Ruan, H.; Zhang, Q.; Liao, W.Q.; Li, Y.Q.; Huang, X.H.; Xu, X.; Lu, S.R. Enhancing tribological, mechanical, and thermal properties of polyimide composites by the synergistic effect between graphene and ionic liquid. Mater. Des. 2020, 189, 108527–108536. [Google Scholar] [CrossRef]
- Lopes Pereira, E.C.; Soares, B.G. Conducting epoxy networks modified with non-covalently functionalized multi-walled carbon nanotube with imidazolium-based ionic liquid. J. Appl. Polym. Sci. 2016, 133, 43976–43984. [Google Scholar] [CrossRef]
- Cao, X.; Jin, M.; Liang, Y.; Li, Y. Synergistic effects of two types of ionic liquids on the dispersion of multi-walled carbon nanotubes in ethylene-vinyl acetate elastomer: Preparation and characterization of flexible conductive composites. Polym. Int. 2017, 66, 1708–1715. [Google Scholar] [CrossRef]
- Tabaczynska, A.; Dabrowska, A.; Maslowski, M.; Strakowska, A. Mechanical and electrical performance of flexible polymer film designed for a textile electrically-conductive path. Materials 2021, 14, 2169. [Google Scholar] [CrossRef]
- Fang, D.; Zhou, C.T.; Liu, G.; Luo, G.J.; Gong, P.J.; Yang, Q.; Niu, Y.H.; Li, G.X. Effects of ionic liquids and thermal annealing on the rheological behavior and electrical properties of poly(methyl methacrylate)/carbon nanotubes composites. Polymer 2018, 148, 68–78. [Google Scholar] [CrossRef]
- Xiong, X.G.; Wang, J.Y.; Jia, H.B.; Fang, E.Y.; Ding, L.F. Structure, thermal conductivity, and thermal stability of bromobutyl rubber nanocomposites with ionic liquid modified graphene oxide. Polym. Degrad. Stab. 2013, 98, 2208–2214. [Google Scholar] [CrossRef]
- Lei, Y.; Guo, B.; Liu, X.; Jia, D. Reinforced rubber with ionic liquid modifued carbon black. Polym. Polym. Compos. 2011, 19, 593–602. [Google Scholar] [CrossRef]
- Guo, B.C.; Liu, X.L.; Zhou, W.Y.; Lei, Y.D.; Jia, D.M. Adsorption of ionic liquid onto halloysite nanotubes: Mechanism and reinforcement of the modified clay to rubber. J. Macromol. Sci. B 2010, 49, 1029–1043. [Google Scholar] [CrossRef]
- Jiang, G.; Song, S.; Zhai, Y.; Feng, C.; Zhang, Y. Improving the filler dispersion of polychloroprene/carboxylated multi-walled carbon nanotubes composites by non-covalent functionalization of carboxylated ionic liquid. Compos. Sci. Technol. 2016, 123, 171–178. [Google Scholar] [CrossRef]
- Lin, Q.; Lu, Y.B.; Ren, W.T.; Zhang, Y. The grafting reaction of epoxidized natural rubber with carboxyl ionic liquids and the ionic conductivity of solid electrolyte composites. RSC Adv. 2015, 5, 90031–90040. [Google Scholar] [CrossRef]
- Zheng, X.Y.; Li, D.K.; Feng, C.Y.; Chen, X.T. Thermal properties and non-isothermal curing kinetics of carbon nanotubes/ionic liquid/epoxy resin systems. Thermochim. Acta 2015, 618, 18–25. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Li, X.X.; Ge, X.; Deng, F.; Cho, U.R. Effect of coupling agents and ionic liquid on the properties of rice bran carbon/carboxylated styrene butadiene rubber composites. Macromol. Res. 2015, 23, 952–959. [Google Scholar] [CrossRef]
- Xu, P.; Wang, X.X.; Hu, Y.D.; Ding, Y.S. Piezoresistive properties of nanocomposites based on silicone rubber and ionic liquid-functionalized carbon black. Mater. Lett. 2016, 182, 218–222. [Google Scholar] [CrossRef]
- Kusmierek, M.; Szadkowski, B.; Marzec, A. The essential role of 1-butyl-3-methylimidazolium-based ionic liquids in the development of transparent silica-filled elastomer systems. Materials 2020, 13, 4337. [Google Scholar] [CrossRef] [PubMed]
- Marzec, A.; Laskowska, A.; Boiteux, G.; Zaborski, M.; Gain, O.; Serghei, A. Properties of carboxylated nitrile rubber/hydrotalcite composites containing imidazolium ionic liquids. Macromol. Symp. 2014, 341, 7–17. [Google Scholar] [CrossRef]
- Cho, M.; Seo, H.; Nam, J.; Choi, H.; Koo, J.; Lee, Y. High ionic conductivity and mechanical strength of solid polymer electrolytes based on NBR/ionic liquid and its application to an electrochemical actuator. Sens. Actuators B Chem. 2007, 128, 70–74. [Google Scholar] [CrossRef]
- Cho, M.S.; Nam, J.D.; Lee, Y.; Choi, H.R.; Koo, J.C. Dry type conducting polymer actuator based on polypyrrole–NBR/ionic liquid system. Mol. Cryst. Liq. Cryst. 2006, 444, 241–246. [Google Scholar] [CrossRef]
- Krainoi, A.; Kummerlowe, C.; Nakaramontri, Y.; Wisunthorn, S.; Vennemann, N.; Pichaiyut, S.; Kiatkamjornwong, S.; Nakason, C. Influence of carbon nanotube and ionic liquid on properties of natural rubber nanocomposites. Express Polym. Lett. 2019, 13, 327–348. [Google Scholar] [CrossRef]
- Subramaniam, K.; Das, A.; Simon, F.; Heinrich, G. Networking of ionic liquid modified CNTs in SSBR. Eur. Polym. J. 2013, 49, 345–352. [Google Scholar] [CrossRef]
- Steinhauser, D.; Subramaniam, K.; Das, A.; Heinrich, G.; Klupel, M. Influence of ionic liquids on the dielectric relaxation behavior of CNT based elastomer nanocomposites. Express Polym. Lett. 2012, 6, 927–936. [Google Scholar] [CrossRef]
- Le, H.H.; Hoang, X.T.; Das, A.; Gohs, U.; Stoeckelhuber, K.W.; Boldt, R.; Heinrich, G.; Adhikari, R.; Radusch, H.J. Kinetics of filler wetting and dispersion in carbon nanotube/rubber composites. Carbon 2012, 50, 4543–4556. [Google Scholar] [CrossRef]
- Yin, Q.; Wen, Y.W.; Jia, H.B.; Hong, L.; Ji, Q.M.; Xu, Z.D. Enhanced mechanical, dielectric, electrical and thermal conductive properties of HXNBR/HNBR blends filled with ionic liquid-modified multiwalled carbon nanotubes. J. Mater. Sci. 2017, 52, 10814–10828. [Google Scholar] [CrossRef]
- Subramaniam, K.; Das, A.; Haussler, L.; Harnisch, C.; Stockelhuber, K.W.; Heinrich, G. Enhanced thermal stability of polychloroprene rubber composites with ionic liquid modified MWCNTs. Polym. Degrad. Stab. 2012, 97, 776–785. [Google Scholar] [CrossRef]
- Subramaniam, K.; Das, A.; Steinhauser, D.; Kluppel, M.; Heinrich, G. Effect of ionic liquid on dielectric, mechanical and dynamic mechanical properties of multi-walled carbon nanotubes/polychloroprene rubber composites. Eur. Polym. J. 2011, 47, 2234–2243. [Google Scholar] [CrossRef]
- Subramaniam, K.; Das, A.; Heinrich, G. Development of conducting polychloroprene rubber using imidazolium based ionic liquid modified multi-walled carbon nanotubes. Compos. Sci. Technol. 2011, 71, 1441–1449. [Google Scholar] [CrossRef]
- Das, M.; Aswathy, T.R.; Pal, S.; Naskar, K. Effect of ionic liquid modified graphene oxide on mechanical and self-healing application of an ionic elastomer. Eur. Polym. J. 2021, 158, 110691–110702. [Google Scholar] [CrossRef]
- Liu, C.; Ruan, G.; Wang, P.; Zhou, Y.Y.; Xu, P.; Ding, Y.S. Synergistic effect of ILs modified MWCNTs on enhanced dielectric properties of silicone rubber/POE blends. Mater. Lett. 2019, 239, 203–206. [Google Scholar] [CrossRef]
- Hussain, M.; Yasin, S.; Ali, A.; Li, Z.Y.; Fan, X.P.; Song, Y.H.; Zheng, Q.; Wang, W.J. Synergistic impact of ionic liquid on interfacial interaction and viscoelastic behaviors of silica filled nitrile butadiene rubber nanocomposites. Compos. Part A-Appl. Sci. Manuf. 2022, 163, 107202–107211. [Google Scholar] [CrossRef]
- Yasin, S.; Hussain, M.; Zheng, Q.; Song, Y. Effects of ionic liquid on cellulosic nanofiller filled natural rubber bionanocomposites. J. Colloid Interface Sci. 2021, 591, 409–417. [Google Scholar] [CrossRef]
- Hussain, M.; Yasin, S.; Memon, H.; Li, Z.; Fan, X.; Akram, M.A.; Wang, W.; Song, Y.; Zheng, Q. Rheological and mechanical properties of silica/nitrile butadiene rubber vulcanizates with eco-friendly ionic liquid. Polymers 2020, 12, 2763. [Google Scholar] [CrossRef]
- Hussain, M.; Yasin, S.; Akram, M.A.; Xu, H.L.; Song, Y.H.; Zheng, Q. Influence of ionic liquids on structure and rheological behaviors of silica-filled butadiene rubber. Ind. Eng. Chem. Res. 2019, 58, 18205–18212. [Google Scholar] [CrossRef]
- Sarath, P.S.; Mahesh, T.Y.; Pandey, M.K.; Haponiuk, J.T.; Thomas, S.; George, S.C. Tribological performance of ionic liquid modified graphene oxide/silicone rubber composite and the correlation of properties using machine learning methods. Polym. Eng. Sci. 2022, 62, 1473–1484. [Google Scholar]
- Ghosh Dasgupta, M.; Dev, S.A.; Muneera Parveen, A.B.; Sarath, P.; Sreekumar, V.B. Draft genome of Korthalsia laciniosa (Griff.) Mart., a climbing rattan elucidates its phylogenetic position. Genomics 2021, 113, 2010–2022. [Google Scholar] [CrossRef]
- Lei, Y.D.; Tang, Z.H.; Zhu, L.X.; Guo, B.C.; Jia, D.M. Functional thiol ionic liquids as novel interfacial modifiers in SBR/HNTs composites. Polymer 2011, 52, 1337–1344. [Google Scholar] [CrossRef]
- Gaca, M.; Ilcikova, M.; Mrlik, M.; Cvek, M.; Vaulot, C.; Urbanek, P.; Pietrasik, R.; Krupa, I.; Pietrasik, J. Impact of ionic liquids on the processing and photo-actuation behavior of SBR composites containing graphene nanoplatelets. Sens. Actuators B-Chem. 2021, 329, 129195–129203. [Google Scholar] [CrossRef]
- Gaca, M.; Vaulot, C.; Maciejewska, M.; Lipinska, M. Preparation and properties of SBR composites containing graphene nanoplatelets modified with pyridinium derivative. Materials 2020, 13, 5407. [Google Scholar] [CrossRef] [PubMed]
- Cappillino, P.J.; Pratt, H.D., III; Hudak, N.S.; Tomson, N.C.; Anderson, T.M.; Anstey, M.R. Application of redox non-innocent ligands to non-aqueous flow battery electrolytes. Adv. Energy Mater. 2014, 4, 1300566. [Google Scholar] [CrossRef]
- Chang, F.; Hu, C.; Liu, X.; Liu, L.; Zhang, J. Coulter dispersant as positive electrolyte additive for the vanadium redox flow battery. Electrochim. Acta 2012, 60, 334–338. [Google Scholar] [CrossRef]
- Shafik, E.S.; Rozik, N.N.; Younan, A.F.; Abd El-Messieh, S.L. Novel plasticizer for acrylonitrile butadiene rubber (NBR) and its effect on physico-mechanical and electrical properties of the vulcanizates. Bull. Mater. Sci. 2020, 43, 1–8. [Google Scholar] [CrossRef]
- Sun, J.; Shi, X.; Wu, Y.; Du, Y.; Jiang, X. The synthesis of a new ionic liquid and its use as a multifunctional additive in different rubber composites. Mater. Res. Express 2018, 5, 075703–0753712. [Google Scholar] [CrossRef]
- Lei, Y.D.; Tang, Z.H.; Guo, B.C.; Zhu, L.X.; Jia, D.M. Synthesis of novel functional liquid and its application as a modifier in SBR/silica composites. Express Polym. Lett. 2010, 4, 692–703. [Google Scholar] [CrossRef]
- Maciejewska, M.; Zaborski, M. Ionic liquids as coagents for sulfur vulcanization of butadiene–styrene elastomer filled with carbon black. Polym. Bull. 2018, 75, 4499–4514. [Google Scholar] [CrossRef]
- Zhong, B.C.; Zeng, X.Q.; Chen, W.J.; Luo, Q.W.; Hu, D.C.; Jia, Z.X.; Jia, D.M. Nonsolvent-assisted surface modification of silica by silane and antioxidant for rubber reinforcement. Polym. Test. 2019, 78, 105949–105957. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, T.; Kim, K.; Kim, B.; Kwag, G.; Kim, J.Y.; Ji, S.; Kim, W.; Paik, H.J. Poly(styrene-r-butadiene)-b-poly(poly(ethylene glycol) methyl ether methacrylate) as a silica dispersant in rubber compounds. Polym. Int. 2014, 63, 908–914. [Google Scholar] [CrossRef]
- Shanmugharaj, A.M.; Bhowmick, A.K. Dynamic mechanical properties of styrene-butadiene rubber vulcanizate filled with electron beam modified surface-treated dual-phase filler. J. Appl. Polym. Sci. 2003, 88, 2992–3004. [Google Scholar] [CrossRef]
- Xiao, Y.K.; Zou, H.; Zhang, L.Q.; Ye, X.; Han, D.L. Surface modification of silica nanoparticles by a polyoxyethylene sorbitan and silane coupling agent to prepare high-performance rubber composites. Polym. Test. 2020, 81, 106195–106204. [Google Scholar] [CrossRef]
- Zhang, X.P.; Cai, L.; He, A.H.; Ma, H.W.; Li, Y.; Hu, Y.M.; Zhang, X.Q.; Liu, L. Facile strategies for green tire tread with enhanced filler-matrix interfacial interactions and dynamic mechanical properties. Compos. Sci. Technol. 2021, 203, 108601–108628. [Google Scholar] [CrossRef]
- Maciejewska, M. Ionic liquids and calcium oxide grafted with allylmalonic acid applied to support the peroxide crosslinking of an Ethylene-Propylene copolymer. Materials 2020, 13, 3260. [Google Scholar] [CrossRef]
- Tang, Z.H.; Huang, J.; Wu, X.H.; Guo, B.C.; Zhang, L.Q.; Liu, F. Interface engineering toward promoting silanization by ionic liquid for high-performance rubber/silica composites. Ind. Eng. Chem. Res. 2015, 54, 10747–10756. [Google Scholar] [CrossRef]
- Sattar, M.A.; Patnaik, A. Role of interface structure and chain dynamics on the diverging glass transition behavior of SSBR-SiO2-PIL elastomers. ACS Omega 2020, 5, 21191–21202. [Google Scholar] [CrossRef]
- Zhou, H.; Bai, S.; Zhang, Y.; Xu, D.; Wang, M. Recent advances in ionic liquids and ionic liquid-functionalized graphene: Catalytic application and environmental remediation. Int. J. Environ. Res. Public Health 2022, 19, 75–84. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, H.; Zhang, J.H.; Zhao, Y.F. Improving tribological performance of fluoroether rubber composites by ionic liquid modified graphene. Compos. Sci. Technol. 2019, 170, 109–115. [Google Scholar] [CrossRef]
- Sanchez-Rodriguez, C.; Aviles, M.D.; Pamies, R.; Carrion-Vilches, F.J.; Sanes, J.; Bermudez, M.D. Extruded PLA nanocomposites modified by graphene oxide and ionic liquid. Polymers 2021, 13, 655. [Google Scholar] [CrossRef]
- Fukushima, T.; Aida, T. Ionic liquids for soft functional materials with carbon nanotubes. Chemistry 2007, 13, 5048–5058. [Google Scholar] [CrossRef] [PubMed]
- Peng, R.G.; Wang, Y.Z.; Tang, W.; Yang, Y.K.; Xie, X.L. Progress in imidazolium ionic liquids assisted fabrication of carbon nanotube and graphene polymer composites. Polymers 2013, 5, 847–872. [Google Scholar] [CrossRef]
- Tunckol, M.; Durand, J.; Serp, P. Carbon nanomaterial–ionic liquid hybrids. Carbon 2012, 50, 4303–4334. [Google Scholar] [CrossRef]
- Zhou, X.; Wu, T.; Ding, K.; Hu, B.; Hou, M.; Han, B. Dispersion of graphene sheets in ionic liquid [bmim][PF6] stabilized by an ionic liquid polymer. Chem. Commun. 2010, 46, 386–388. [Google Scholar] [CrossRef]
- Abraham, J.; Arif, P.M.; Kailas, L.; Kalarikkal, N.; George, S.C.; Thomas, S. Developing highly conducting and mechanically durable styrene butadiene rubber composites with tailored microstructural properties by a green approach using ionic liquid modified MWCNTs. RSC Adv. 2016, 6, 32493–32504. [Google Scholar] [CrossRef]
- Demétrio da Silva, V.; Ferrari, M.D.; Brandalise, R.N.; Benvenutti, E.V.; Schrekker, H.S.; Amico, S.C. EPDM with biochar, carbon black, aramid pulp and ionic liquid-compatibilized aramid pulp. Fibers Polym. 2021, 22, 1180–1188. [Google Scholar] [CrossRef]
- Sowińska, A.; Maciejewska, M. Thermal analysis applied to studying the influence of ionic liquids on the vulcanization, thermal stability and damping properties of ethylene-propylene-diene rubber. J. Therm. Anal. Calorim. 2019, 138, 2669–2681. [Google Scholar] [CrossRef]
- Demétrio da Silva, V.; Jacobi, M.M.; Schrekker, H.S.; Amico, S.C. Imidazolium ionic liquid compatibilizers in melt-blended styrene-butadiene rubber/aramid pulp composites. Polym. Bull. 2018, 76, 3451–3462. [Google Scholar] [CrossRef]
- Demétrio da Silva, V.; Jacobi, M.M.; Schrekker, H.S.; Amico, S.C. Aramid pulp with physisorbed imidazolium ionic liquids for solvent-casted enhanced styrene-butadiene rubber composites. J. Appl. Polym. Sci. 2018, 135, 46693–46699. [Google Scholar] [CrossRef]
- Demétrio da Silva, V.; Barros, Í.R.D.; Conceição, D.K.S.D.; Almeida, K.N.D.; Schrekker, H.S.; Amico, S.C.; Jacobi, M.M. Aramid pulp reinforced hydrogenated nitrile butadiene rubber composites with ionic liquid compatibilizers. J. Appl. Polym. Sci. 2019, 137, 48702–48708. [Google Scholar] [CrossRef]
- Maciejewska, M.; Zaborski, M. Thermal analysis and mechanical methods applied to studying properties of SBR compounds containing ionic liquids. Polym. Test. 2017, 61, 349–363. [Google Scholar] [CrossRef]
- Pernak, J.; Walkiewicz, F.; Maciejewska, M.; Zaborski, M. Ionic liquids as vulcanization accelerators. Ind. Eng. Chem. Res. 2010, 49, 5012–5017. [Google Scholar] [CrossRef]
- Maciejewska, M.; Zaborski, M. Effect of ionic liquids on the dispersion of zinc oxide and silica nanoparticles, vulcanisation behaviour and properties of NBR composites. Express Polym. Lett. 2014, 8, 932–940. [Google Scholar] [CrossRef]
- Przybyszewska, M.; Zaborski, M. Effect of ionic liquids and surfactants on zinc oxide nanoparticle activity in crosslinking of acrylonitrile butadiene elastomer. J. Appl. Polym. Sci. 2010, 116, 155–164. [Google Scholar] [CrossRef]
- Maciejewska, M.; Walkiewicz, F.; Zaborski, M. Novel ionic liquids as accelerators for the sulfur vulcanization of butadiene–styrene elastomer composites. Ind. Eng. Chem. Res. 2013, 52, 8410–8415. [Google Scholar] [CrossRef]
- Sowinska, A.; Maciejewska, M.; Guo, L.; Delebecq, E. Task-specific ionic liquids with lactate anion applied to improve ZnO dispersibility in the Ethylene-Propylene-Diene elastomer. Polymers 2021, 13, 774. [Google Scholar] [CrossRef]
- Pingot, M.; Szadkowski, B.; Zaborski, M. Experimental investigation on activity of cumene hydroperoxide and selected ionic liquids in butadiene rubber vulcanization. Adv. Polym. Technol. 2018, 37, 3432–3437. [Google Scholar] [CrossRef]
- Belesov, A.V.; Shkaeva, N.V.; Popov, M.S.; Skrebets, T.E.; Faleva, A.V.; Ul’yanovskii, N.V.; Kosyakov, D.S. New insights into the thermal stability of 1-butyl-3-methylimidazolium-based ionic liquids. Int. J. Mol. Sci. 2022, 23, 10966. [Google Scholar] [CrossRef]
- Yin, B.; Zhang, X.M.; Zhang, X.; Wang, J.Y.; Wen, Y.W.; Jia, H.B.; Ji, Q.M.; Ding, L.F. Ionic liquid functionalized graphene oxide for enhancement of styrene-butadiene rubber nanocomposites. Polym. Adv. Technol. 2017, 28, 293–302. [Google Scholar] [CrossRef]
- De Vries, H.; Jeong, S.; Passerini, S. Ternary polymer electrolytes incorporating pyrrolidinium-imide ionic liquids. RSC Adv. 2015, 5, 13598–13606. [Google Scholar] [CrossRef]
- Joost, M.; Kim, G.T.; Winter, M.; Passerini, S. Phase stability of Li-ion conductive, ternary solid polymer electrolytes. Electrochim. Acta 2013, 113, 181–185. [Google Scholar] [CrossRef]
- Yan, X.; Anguille, S.; Bendahan, M.; Moulin, P. Ionic liquids combined with membrane separation processes: A review. Sep. Purif. Technol. 2019, 222, 230–253. [Google Scholar] [CrossRef]
- Abraham, J.; Jose, T.; Moni, G.; George, S.C.; Kalarikkal, N.; Thomas, S. Ionic liquid modified multiwalled carbon nanotube embedded styrene butadiene rubber membranes for the selective removal of toluene from toluene/methanol mixture via pervaporation. J. Taiwan Inst. Chem. Eng. 2019, 95, 594–601. [Google Scholar] [CrossRef]
- Friess, K.; Izak, P.; Karaszova, M.; Pasichnyk, M.; Lanc, M.; Nikolaeva, D.; Luis, P.; Jansen, J.C. A review on ionic liquid gas separation membranes. Membranes 2021, 11, 97. [Google Scholar] [CrossRef]
- Nguyen, P.T.; Voss, B.A.; Wiesenauer, E.F.; Gin, D.L.; Noble, R.D. Physically gelled room-temperature ionic liquid-based composite membranes for CO2/N2 separation: Effect of composition and thickness on membrane properties and performance. Ind. Eng. Chem. Res. 2013, 52, 8812–8821. [Google Scholar] [CrossRef]
- Gu, Y.; Cussler, E.L.; Lodge, T.P. ABA-triblock copolymer ion gels for CO2 separation applications. J. Membr. Sci. 2012, 423, 20–26. [Google Scholar] [CrossRef]
- Tomé, L.C.; Marrucho, I.M. Ionic liquid-based materials: A platform to design engineered CO2 separation membranes. Chem. Soc. Rev. 2016, 45, 2785–2824. [Google Scholar] [CrossRef]
- Sharma, M.; Sharma, S.; Abraham, J.; Thomas, S.; Madras, G.; Bose, S. Flexible EMI shielding materials derived by melt blending PVDF and ionic liquid modified MWNTs. Mater. Res. Express 2014, 1, 035003–0350019. [Google Scholar] [CrossRef]
- Zhang, D.; Liu, Q.; Shi, X.; Li, Y. Tetrabutylammonium hexafluorophosphate and 1-ethyl-3-methyl imidazolium hexafluorophosphate ionic liquids as supporting electrolytes for non-aqueous vanadium redox flow batteries. J. Power Sources 2012, 203, 201–205. [Google Scholar] [CrossRef]
- Li, M.; Xie, X.; Yang, C.; Wang, J.; Wang, S.; Shang, Y. Characteristics of BMIMBF4 as anode electrolyte additive for vanadium redox flow battery (In Chinese). CIESC J. 2011, 62 (S2), 135–139. [Google Scholar]
- Wen, Y.; Zhang, H.; Qian, P.; Zhou, H.; Zhao, P.; Yi, B.; Yang, Y. A study of the Fe (III)/Fe (II)–triethanolamine complex redox couple for redox flow battery application. Electrochim. Acta 2006, 51, 3769–3775. [Google Scholar] [CrossRef]

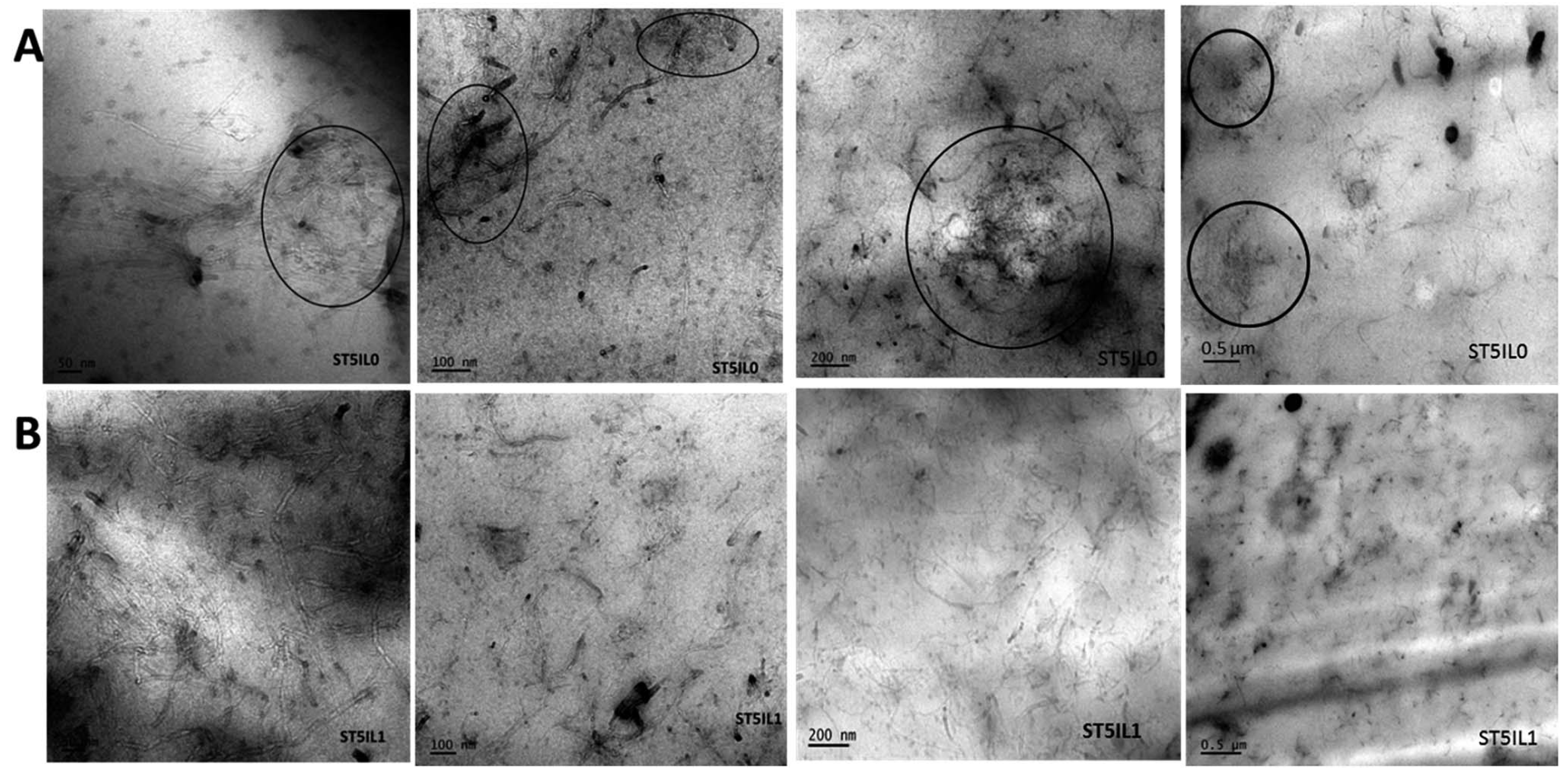


| ILs | Abbreviation | Application Field | References |
|---|---|---|---|
| 1-allyl-3-methylimidazolium chloride | [AMIM] [Cl] | dispersant, solvent | [37,38,39,40,41,42,43,44] |
| 1-decyl-3-methylimidazolium chloride | [DMIM] [Cl] | dispersant, sensor | [12,45,46,47,48] |
| 1-methylimidazole chloride | [MIM] [Cl] | dispersant, modifier | [30,49] |
| 1-butyl-3-methylimidazolium chloride | [BMIM] [Cl] | membrane separation, solvent, plasticizer, surfactant, conductive, battery | [20,50,51,52] |
| 1-butyl-3-methylimidazolium bromide | [BMIM] [Br] | dispersant, solvent, conductive | [53,54,55] |
| 1-ethyl-3-methylimidazolium chloride | [EMIM] [Cl] | [56,57,58,59] | |
| 1-ethyl-3-methylimidazolium bromide | [EMIM] [Br] | [60,61] | |
| 1-benzyl-3-methylimidazolium chloride | [BZMIM] [Cl] | membrane separation | [62,63] |
| 1-benzyl-3-ethylimidazolium chloride | [BZEIM] [Cl] | [13,64,65] | |
| 1-butyl-3-methylimidazolium tetrafluoroborate | [BMIM] [BF4] | dispersant, plasticizer, conductive, membrane separation, battery | [22,66,67,68,69,70] |
| 1-butyl-3-methylimidazolium hexafluorophosphate | [BMIM] [PF6] | [19,61,66,71,72,73,74,75] | |
| 1-ethyl-3-methylimidazolium tetrafluoroborate | [EMIM] [BF4] | dispersant, conductive, electrolyte | [16] |
| 1-carboxyethyl-3-methylimidazolium bis(trifluoromethysulfonyl)imide | [CEMIM] [NTf2] | dispersant, conductive, grafting agent, battery | [76,77] |
| 1-hexyl-3-methylimidazolium hexafluorophosphate | [HMIM] [PF6] | dispersant, plasticizer, conductive, coupling agent | [71,72,78,79] |
| 1-hexadecyl-3- methylimidazolium bromide | [HDMIM] [Br] | dispersant, conductive | [47,80] |
| tributylmethylammoniu bis(trifluoromethylsulfonyl)imide | [TBMA] [NTf2] | [70] | |
| 1-butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide | [BMIM] [NTf2] | conductive, electromagnetic interference, vulcanization | [81,82,83,84,85,86,87,88,89,90,91,92] |
| 1-propyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide | [PMIM] [NTf2] | vulcanization | [15] |
| 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide | [EMIM] [NTf2] | dispersant, electromagnetic interference | [93] |
| 1-vinyl-3-ethylimidazolium bromide | [VEIM] [Br] | [34,35,94] | |
| 1-butyl-3-methylimidazolium tetrachloroaluminate | [BMIM] [AlCl4] | dispersant | [81,82] |
| 1-ethyl-3-methylimidazolium acetate | [EMIM] [Ac] | [95,96,97,98] | |
| 1-ethyl-3-methylimidazolium dicyanamide | [EMIM] [Dca] | catalyst | [99,100] |
| 1-methylimidazolium mercaptopropinate | [MIM] [MP] | dispersant, catalyst, modifier, vulcanization | [101] |
| bis(1-methylimidazolium) mercaptosuccinate | [BMIM] [MS] | [101] | |
| 1-butylpridinium bromide | [BBP] [Br] | dispersant, modifier | [102] |
| 4-methyl-1-butylpyridinium bromide | [BMBP] [Br] | [102] | |
| 1-butyl-4-methylpyridinium tetrafluoroborate | [BMP] [BF4] | [103] | |
| tetrabutylammonium hexafluorophosphate | [TBA] [PF6] | electrolyte | [104] |
| tetraethylammonium hexafluorophosphate | [TEA] [PF6] | [105] | |
| 1-ethyl-3-methyl imidazolium hexafluorophosphate | [EMIM] [PF6] | [105] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, L.; Wang, L.; Cui, Z.; Liu, Y.; Du, A. Multifunctional Applications of Ionic Liquids in Polymer Materials: A Brief Review. Molecules 2023, 28, 3836. https://doi.org/10.3390/molecules28093836
Wei L, Wang L, Cui Z, Liu Y, Du A. Multifunctional Applications of Ionic Liquids in Polymer Materials: A Brief Review. Molecules. 2023; 28(9):3836. https://doi.org/10.3390/molecules28093836
Chicago/Turabian StyleWei, Liping, Lin Wang, Ziwen Cui, Yingjun Liu, and Aihua Du. 2023. "Multifunctional Applications of Ionic Liquids in Polymer Materials: A Brief Review" Molecules 28, no. 9: 3836. https://doi.org/10.3390/molecules28093836
APA StyleWei, L., Wang, L., Cui, Z., Liu, Y., & Du, A. (2023). Multifunctional Applications of Ionic Liquids in Polymer Materials: A Brief Review. Molecules, 28(9), 3836. https://doi.org/10.3390/molecules28093836






