Multidimensional Chromatographic Fingerprinting Combined with Chemometrics for the Identification of Regulated Plants in Suspicious Plant Food Supplements
Abstract
1. Introduction
2. Results
2.1. Final Chromatographic Conditions
2.2. Correlation Analysis and Choice of Wavelengths
2.3. Chemometric Analysis
2.3.1. Partial Least Squares–Discriminant Analysis
Binary Models
Multiclass Models
2.3.2. Real Samples
2.3.3. Comparison of Results Obtained for 254 nm and for Selected Wavelengths
3. Materials and Methods
3.1. Samples and Reagents
3.2. Sample Preparation
3.2.1. Preparation of Reference Solutions
3.2.2. Preparation of Triturations
3.2.3. Preparation of Samples
3.3. Instrumentation and Conditions
3.3.1. Preparation of Reference Solutions
Method Development
3.4. Selection of Wavelengths
3.5. Data
3.5.1. Data Set Preparation and Multidimensional Fingerprints
3.5.2. Peak Alignment
3.5.3. Data Pretreatment
3.5.4. Test Set Selection
3.5.5. PLS-DA
3.5.6. Software
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Skalicka-Woźniak, K.; Georgiev, M.I.; Orhan, I.E. Adulteration of Herbal Sexual Enhancers and Slimmers: The Wish for Better Sexual Well-Being and Perfect Body Can Be Risky. Food Chem. Toxicol. 2017, 108, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.M.; Zhang, X. The World Medicines Situation 2011. Traditional Medicines: Global Situation, Issues and Challenges; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Werdemberg dos Santos, L.C.; Granja Arakaki, D.; Silva de Pádua Melo, E.; Nascimento, V.A. Health Hazard Assessment Due to Slimming Medicinal Plant Intake. Biol. Trace Elem. Res. 2022, 200, 1442–1454. [Google Scholar] [CrossRef] [PubMed]
- Pallarés, N.; Berrada, H.; Font, G.; Ferrer, E. Mycotoxins Occurrence in Medicinal Herbs Dietary Supplements and Exposure Assessment. J. Food Sci. Technol. 2022, 59, 2830–2841. [Google Scholar] [CrossRef] [PubMed]
- Jokar, N.K.; Noorhosseini, S.A.; Allahyari, M.S.; Damalas, C.A. Consumers’ Acceptance of Medicinal Herbs: An Application of the Technology Acceptance Model (TAM). J. Ethnopharmacol. 2017, 207, 203–210. [Google Scholar] [CrossRef]
- Rocha, T.; Amaral, J.S.; Oliveira, M.B.P.P. Adulteration of Dietary Supplements by the Illegal Addition of Synthetic Drugs: A Review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 43–62. [Google Scholar] [CrossRef]
- Corazza, O.; Martinotti, G.; Santacroce, R.; Chillemi, E.; Di Giannantonio, M.; Schifano, F.; Cellek, S. Sexual Enhancement Products for Sale Online: Raising Awareness of the Psychoactive Effects of Yohimbine, Maca, Horny Goat Weed, and Ginkgo biloba. BioMed Res. Int. 2014, 2014, 841798. [Google Scholar] [CrossRef]
- Esslinger, S.; Riedl, J.; Fauhl-Hassek, C. Potential and Limitations of Non-Targeted Fingerprinting for Authentication of Food in Official Control. Food Res. Int. 2014, 60, 189–204. [Google Scholar] [CrossRef]
- Sammut, J.; Gopi, K.; Saintilan, N.; Mazumder, D. Facing the Challenges of Food Fraud in the Global Food System. In Food Authentication and Traceability; Elsevier: Amsterdam, The Netherlands, 2021; pp. 35–63. ISBN 978-0-12-821104-5. [Google Scholar]
- Deconinck, E.; Djiogo, C.A.S.; Kamugisha, A.; Courselle, P. The Use of Stationary Phase Optimized Selectivity Liquid Chromatography for the Development of Herbal Fingerprints to Detect Targeted Plants in Plant Food Supplements. Talanta 2017, 170, 441–450. [Google Scholar] [CrossRef]
- Fibigr, J.; Šatínský, D.; Solich, P. Current Trends in the Analysis and Quality Control of Food Supplements Based on Plant Extracts. Anal. Chim. Acta 2018, 1036, 1–15. [Google Scholar] [CrossRef]
- Riswanto, F.D.O.; Windarsih, A.; Lukitaningsih, E.; Rafi, M.; Fadzilah, N.A.; Rohman, A. Metabolite Fingerprinting Based on 1H-NMR Spectroscopy and Liquid Chromatography for the Authentication of Herbal Products. Molecules 2022, 27, 1198. [Google Scholar] [CrossRef]
- Calixto, J.B. Efficacy, Safety, Quality Control, Marketing and Regulatory Guidelines for Herbal Medicines (Phytotherapeutic Agents). Braz. J. Med. Biol. Res. 2000, 33, 179–189. [Google Scholar] [CrossRef]
- KONINKLIJK BESLUIT van 29 AUGUSTUS 1997 Betreffende de Fabricage van En de Handel in Voedingsmiddelen Die Uit Planten of Uit Plantenbereidingen Samengesteld Zijn of Deze Bevatten (B.S. 21.XI.1997). Available online: https://www.health.belgium.be/nl/koninklijk-besluit-van-29-augustus-1997-planten (accessed on 18 January 2023).
- Tistaert, C.; Dejaegher, B.; Heyden, Y.V. Chromatographic Separation Techniques and Data Handling Methods for Herbal Fingerprints: A Review. Anal. Chim. Acta 2011, 690, 148–161. [Google Scholar] [CrossRef]
- Goodarzi, M.; Russell, P.J.; Vander Heyden, Y. Similarity Analyses of Chromatographic Herbal Fingerprints: A Review. Anal. Chim. Acta 2013, 804, 16–28. [Google Scholar] [CrossRef]
- Nasim, N.; Sandeep, I.S.; Mohanty, S. Plant-Derived Natural Products for Drug Discovery: Current Approaches and Prospects. Nucleus 2022, 65, 399–411. [Google Scholar] [CrossRef]
- Custers, D.; Van Praag, N.; Courselle, P.; Apers, S.; Deconinck, E. Chromatographic Fingerprinting as a Strategy to Identify Regulated Plants in Illegal Herbal Supplements. Talanta 2017, 164, 490–502. [Google Scholar] [CrossRef]
- Xie, P.; Chen, S.; Liang, Y.; Wang, X.; Tian, R.; Upton, R. Chromatographic Fingerprint Analysis—A Rational Approach for Quality Assessment of Traditional Chinese Herbal Medicine. J. Chromatogr. A 2006, 1112, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Noviana, E.; Indrayanto, G.; Rohman, A. Advances in Fingerprint Analysis for Standardization and Quality Control of Herbal Medicines. Front. Pharmacol. 2022, 13, 853023. [Google Scholar] [CrossRef] [PubMed]
- Rohman, A.; Windarsih, A. The Application of Molecular Spectroscopy in Combination with Chemometrics for Halal Authentication Analysis: A Review. Int. J. Mol. Sci. 2020, 21, 5155. [Google Scholar] [CrossRef] [PubMed]
- Wahyuni, W.T.; Saharah, M.; Arif, Z.; Rafi, M. Thin Layer Chromatographic Fingerprint and Chemometrics Analysis for Identification of Phyllanthus Niruri from Its Related Species. J. Indones. Chem. Soc. 2020, 3, 47. [Google Scholar] [CrossRef]
- Han, J.; Xu, K.; Yan, Q.; Sui, W.; Zhang, H.; Wang, S.; Zhang, Z.; Wei, Z.; Han, F. Qualitative and Quantitative Evaluation of Flos Puerariae by Using Chemical Fingerprint in Combination with Chemometrics Method. J. Pharm. Anal. 2022, 12, 489–499. [Google Scholar] [CrossRef]
- Wang, Y.; He, T.; Wang, J.; Wang, L.; Ren, X.; He, S.; Liu, X.; Dong, Y.; Ma, J.; Song, R.; et al. High Performance Liquid Chromatography Fingerprint and Headspace Gas Chromatography-Mass Spectrometry Combined with Chemometrics for the Species Authentication of Curcumae Rhizoma. J. Pharm. Biomed. Anal. 2021, 202, 114144. [Google Scholar] [CrossRef]
- Deconinck, E.; Vanhamme, M.; Bothy, J.L.; Courselle, P. A Strategy Based on Fingerprinting and Chemometrics for the Detection of Regulated Plants in Plant Food Supplements from the Belgian Market: Two Case Studies. J. Pharm. Biomed. Anal. 2019, 166, 189–196. [Google Scholar] [CrossRef]
- Koncz, D.; Tóth, B.; Roza, O.; Csupor, D. A Systematic Review of the European Rapid Alert System for Food and Feed: Tendencies in Illegal Food Supplements for Weight Loss. Front. Pharmacol. 2021, 11, 611361. [Google Scholar] [CrossRef]
- Bersani, F.S.; Coviello, M.; Imperatori, C.; Francesconi, M.; Hough, C.M.; Valeriani, G.; De Stefano, G.; Bolzan Mariotti Posocco, F.; Santacroce, R.; Minichino, A.; et al. Adverse Psychiatric Effects Associated with Herbal Weight-Loss Products. BioMed Res. Int. 2015, 2015, 120679. [Google Scholar] [CrossRef]
- Hawkins, N.; Richards, P.S.; Granley, H.M.; Stein, D.M. The Impact of Exposure to the Thin-Ideal Media Image on Women. Eat. Disord. 2004, 12, 35–50. [Google Scholar] [CrossRef]
- Farrington, R.; Musgrave, I.F.; Byard, R.W. Evidence for the Efficacy and Safety of Herbal Weight Loss Preparations. J. Integr. Med. 2019, 17, 87–92. [Google Scholar] [CrossRef]
- Custers, D.; Krakowska, B.; De Beer, J.O.; Courselle, P.; Daszykowski, M.; Apers, S.; Deconinck, E. Testing of Complementarity of PDA and MS Detectors Using Chromatographic Fingerprinting of Genuine and Counterfeit Samples Containing Sildenafil Citrate. Anal. Bioanal. Chem. 2016, 408, 1643–1656. [Google Scholar] [CrossRef]
- Pravdova, V.; Walczak, B.; Massart, D.L. A Comparison of Two Algorithms for Warping of Analytical Signals. Anal. Chim. Acta 2002, 456, 77–92. [Google Scholar] [CrossRef]
- van Nederkassel, A.M.; Daszykowski, M.; Massart, D.L.; Vander Heyden, Y. Prediction of Total Green Tea Antioxidant Capacity from Chromatograms by Multivariate Modeling. J. Chromatogr. A 2005, 1096, 177–186. [Google Scholar] [CrossRef]
- Snee, R.D. Validation of Regression Models: Methods and Examples. Technometrics. 1977, 19, 415–428. [Google Scholar] [CrossRef]
- Gromski, P.S.; Muhamadali, H.; Ellis, D.I.; Xu, Y.; Correa, E.; Turner, M.L.; Goodacre, R. A Tutorial Review: Metabolomics and Partial Least Squares-Discriminant Analysis—A Marriage of Convenience or a Shotgun Wedding. Anal. Chim. Acta 2015, 879, 10–23. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Perez, D.; Guan, H.; Madhivanan, P.; Mathee, K.; Narasimhan, G. So You Think You Can PLS-DA? BMC Bioinformatics. 2020, 21, 2. [Google Scholar] [CrossRef] [PubMed]
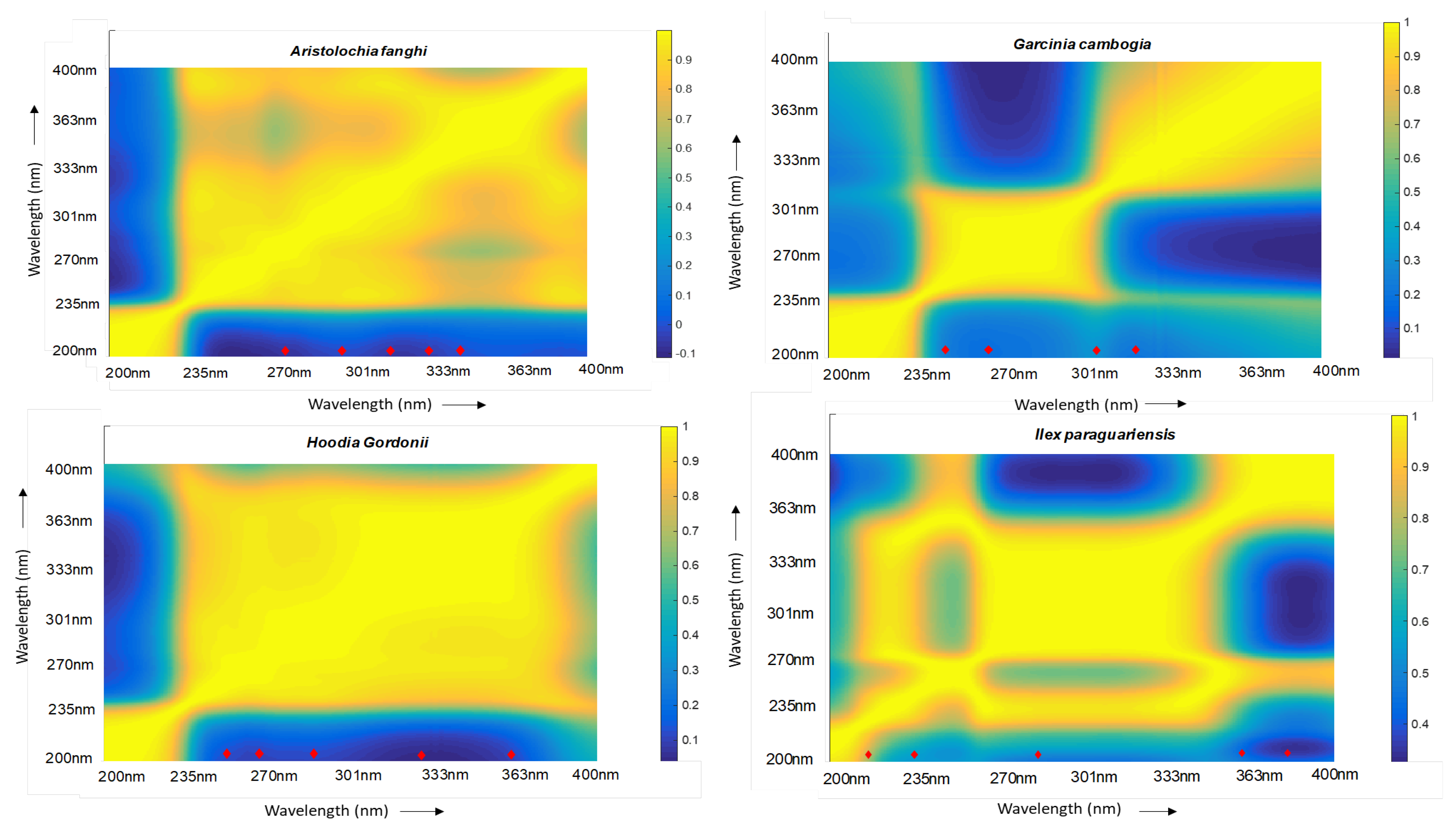
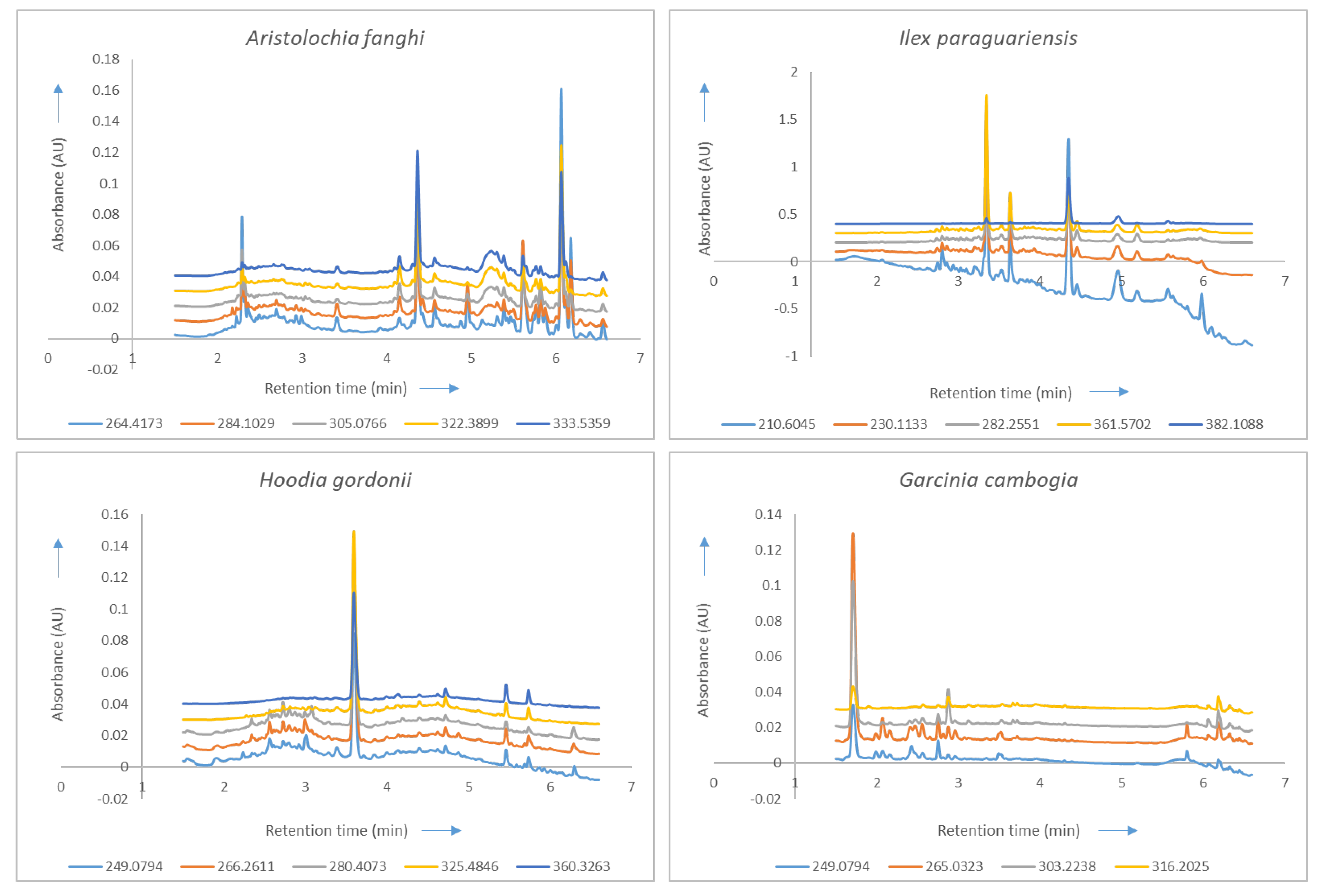
| Aristolochia fanghi | Ilex paraguariensis | Hoodia gordonii | Garcinia cambogia |
|---|---|---|---|
| 264 | 210 | 249 | 249 |
| 284 | 230 | 266 | 265 |
| 305 | 282 | 280 | 303 |
| 322 | 361 | 325 | 316 |
| 333 | 382 | 360 |
| Type of Data Set | Class Assigned | Total Samples in the Data Set | No. of Samples in Training Set | No. of Samples in Test Set |
|---|---|---|---|---|
| Binary data set | 1, 2 | 67 | 50 | 17 |
| Multiclass data set | 1, 2, 3, 4, 5 | 235 | 176 | 59 |
| Slimming Plant | PLS Factors | Cross Validation (ccr%) with Misclassified Samples between Brackets () | Modelling (ccr%) | External Test Set Prediction (ccr%) with Misclassified Samples between Brackets () |
|---|---|---|---|---|
| Aristolochia fanghi | 23 | 94% (3/50) | 96% | 94% (1/17) |
| Ilex paraguariensis | 14 | 96% (2/50) | 100% | 94% (1/17) |
| Hoodia gordonii | 20 | 88% (6/50) | 96% | 94% (1/17) |
| Garcinia cambogia | 22 | 96% (1/50) | 98% | 88% (2/17) |
| Slimming Plant | True Positives Training Set Test Set (cv) | False Positives Training Set Test Set (cv) | True Negatives Training Set Test Set (cv) | False Negatives Training Set Test Set (cv) | ||||
|---|---|---|---|---|---|---|---|---|
| Aristolochia fanghi | 45 | 12 | 2 | 1 | 3 | 4 | 0 | 0 |
| Ilex paraguariensis | 43 | 12 | 2 | 0 | 5 | 4 | 0 | 1 |
| Hoodia gordonii | 42 | 14 | 6 | 1 | 2 | 2 | 0 | 0 |
| Garcinia cambogia | 43 | 13 | 2 | 2 | 5 | 2 | 0 | 0 |
| Slimming Plant | PLS Factors | Cross Validation (ccr%) with Misclassified Samples between Brackets () | Modelling (ccr%) | External Test Set Validation (ccr%) with Misclassified Samples between Brackets () |
|---|---|---|---|---|
| Aristolochia fanghi | 22 | 91% (15/176) | 93% | 90% (6/59) |
| Ilex paraguariensis | 18 | 93% (12/176) | 97% | 88% (7/59) |
| Hoodia gordonii | 23 | 87% (23/176) | 94% | 90% (6/59) |
| Garcinia cambogia | 22 | 84% (28/176) | 93% | 90% (6/59) |
| Slimming Plant | True Positives Training Set Test Set (cv) | False Positives Training Set Test Set (cv) | ||
|---|---|---|---|---|
| A multiclass model with wavelengths of Aristolochia fanghi | ||||
| Aristolochia fanghi | 43 | 13 | 6 | 0 |
| Hoodia gordonii | 49 | 6 | 6 | 4 |
| Ilex paraguariensis | 29 | 20 | 2 | 0 |
| Garcinia cambogia | 39 | 14 | 1 | 2 |
| Blank | 0 | 0 | 0 | 0 |
| A multiclass model with wavelengths of Ilex paraguariensis | ||||
| Aristolochia fanghi | 46 | 10 | 1 | 4 |
| Hoodia gordonii | 44 | 9 | 5 | 2 |
| Ilex paraguariensis | 34 | 21 | 1 | 0 |
| Garcinia cambogia | 40 | 12 | 5 | 0 |
| Blank | 0 | 0 | 0 | 1 |
| A multiclass model with wavelengths of Hoodia gordonii | ||||
| Aristolochia fanghi | 43 | 11 | 8 | 0 |
| Hoodia gordonii | 46 | 10 | 10 | 6 |
| Ilex paraguariensis | 26 | 23 | 1 | 0 |
| Garcinia cambogia | 38 | 8 | 4 | 0 |
| Blank | 0 | 1 | 0 | 0 |
| A multiclass model with wavelengths of Garcinia cambogia | ||||
| Aristolochia fanghi | 40 | 12 | 10 | 2 |
| Hoodia gordonii | 35 | 10 | 2 | 2 |
| Ilex paraguariensis | 27 | 23 | 1 | 0 |
| Garcinia cambogia | 45 | 8 | 13 | 1 |
| Blank | 0 | 0 | 2 | 1 |
| Samples | Plants Classified According to Binary Modelling (Comparative) | Plants Classified with the Multiclass Model |
|---|---|---|
| Sample 1 | Ilex paraguariensis, Garcinia cambogia | Garcinia cambogia |
| Sample 2 | -- | Garcinia cambogia |
| Sample 3 | Aristolochia fanghi, Hoodia gordonii, Garcinia cambogia | Hoodia gordonii |
| Sample 4 | Hoodia gordonii, Ilex paraguariensis | Aristolochia fanghi |
| Sample 5 | Hoodia gordonii, | Aristolochia fanghi |
| Sample 6 | Garcinia cambogia, Ilex paraguariensis | Ilex paraguariensis |
| Sample 7 | Aristolochia fanghi, Hoodia gordonii, Garcinia cambogia | Hoodia gordonii |
| Sample 8 | -- | -- |
| Sample 9 | Aristolochia fanghi, Hoodia gordonii, Garcinia cambogia | Garcinia cambogia |
| Sample 10 | Garcinia cambogia, Ilex paraguariensis | Garcinia cambogia |
| Sample 11 | Aristolochia fanghi, Hoodia gordonii | Aristolochia fanghi |
| Sample 12 | Garcinia cambogia | Aristolochia fanghi |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ranjan, S.; Adams, E.; Deconinck, E. Multidimensional Chromatographic Fingerprinting Combined with Chemometrics for the Identification of Regulated Plants in Suspicious Plant Food Supplements. Molecules 2023, 28, 3632. https://doi.org/10.3390/molecules28083632
Ranjan S, Adams E, Deconinck E. Multidimensional Chromatographic Fingerprinting Combined with Chemometrics for the Identification of Regulated Plants in Suspicious Plant Food Supplements. Molecules. 2023; 28(8):3632. https://doi.org/10.3390/molecules28083632
Chicago/Turabian StyleRanjan, Surbhi, Erwin Adams, and Eric Deconinck. 2023. "Multidimensional Chromatographic Fingerprinting Combined with Chemometrics for the Identification of Regulated Plants in Suspicious Plant Food Supplements" Molecules 28, no. 8: 3632. https://doi.org/10.3390/molecules28083632
APA StyleRanjan, S., Adams, E., & Deconinck, E. (2023). Multidimensional Chromatographic Fingerprinting Combined with Chemometrics for the Identification of Regulated Plants in Suspicious Plant Food Supplements. Molecules, 28(8), 3632. https://doi.org/10.3390/molecules28083632






