Natural Deep Eutectic Solvents for the Extraction of Triterpene Saponins from Aralia elata var. mandshurica (Rupr. & Maxim.) J. Wen
Abstract
1. Introduction
2. Results
2.1. Identification of Triterpene Saponins in A. elata Roots
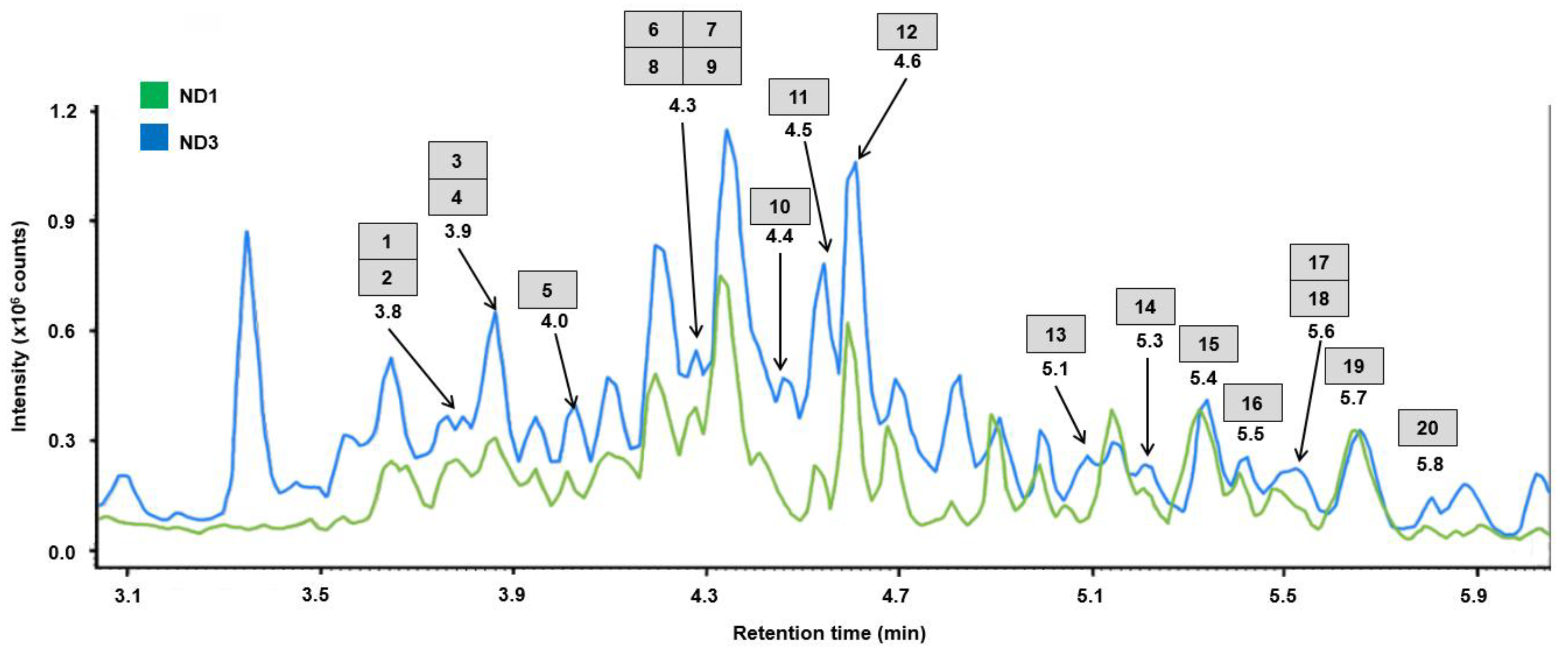
2.2. Extraction of Triterpene Saponins with NADES
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Plant Material and Extraction Procedures
4.3. Metabolite Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wen, J. New Taxa and Nomenclatural Changes in Aralia (Araliaceae). Novon 1994, 4, 400–403. [Google Scholar] [CrossRef]
- Xu, Y.; Liu, J.; Zeng, Y.; Jin, S.; Liu, W.; Li, Z.; Qin, X.; Bai, Y. Traditional Uses, Phytochemistry, Pharmacology, Toxicity and Quality Control of Medicinal Genus Aralia: A Review. J. Ethnopharmacol. 2022, 284, 114671. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Yoshizumi, S.; Ueno, T.; Matsuda, H.; Murakami, T.; Yamahara, J.; Murakami, N. Medicinal Foodstuffs. I. Hypoglycemic Constituents from a Garnish Foodstuff “Taranome,” the Young Shoot of Aralia Elata SEEM.: Elatosides G, H, I, J, and K. Chem. Pharm. Bull. 1995, 43, 1878–1882. [Google Scholar] [CrossRef]
- Shikov, A.N.; Pozharitskaya, O.N.; Makarov, V.G. Aralia elata var. mandshurica (Rupr. & Maxim.) J. Wen: An overview of pharmacological studies. Phytomedicine 2016, 23, 1409–1421. [Google Scholar] [CrossRef]
- Panossian, A.G.; Efferth, T.; Shikov, A.N.; Pozharitskaya, O.N.; Kuchta, K.; Mukherjee, P.K.; Banerjee, S.; Heinrich, M.; Wu, W.; Guo, D.; et al. Evolution of the adaptogenic concept from traditional use to medical systems: Pharmacology of stress- and aging-related diseases. Med. Res. Rev. 2021, 41, 630–703. [Google Scholar] [CrossRef]
- Chernyak, D.M.; Titova, M.S. Anti-Stress Effect of the Far Eastern Plants. Pacific Med. J. 2014, 2, 28–30. [Google Scholar]
- Jung, C.; Lee, U. Pharmacological Studies on Root Bark Extract of Aralia elata—Antigastritic and Antiulcerative Effects in Rats. Yakhak Hoeji 1993, 37, 581–590. [Google Scholar]
- Hernandez, D.E.; Hancke, J.L.; Wikman, G. Evaluation of the Anti-Ulcer and Antisecretory Activity of Extracts of Aralia elata Root and Schizandra chinensis Fruit in the Rat. J. Ethnopharmacol. 1988, 23, 109–114. [Google Scholar] [CrossRef]
- Luo, Y.; Dong, X.; Yu, Y.; Sun, G.; Sun, X. Total Aralosides of Aralia elata (Miq) Seem (TASAES) Ameliorate Nonalcoholic Steatohepatitis by Modulating IRE1α-Mediated JNK and NF-ΚB Pathways in ApoE–/– Mice. J. Ethnopharmacol. 2015, 163, 241–250. [Google Scholar] [CrossRef]
- Saito, S.; Ebashi, J.; Sumita, S.; Furumoto, T.; Nagamura, Y.; Nishida, K.; Isiguro, I. Comparison of Cytoprotective Effects of Saponins Isolated from Leaves of Aralia elata SEEM. (Araliaceae) with Synthesized Bisdesmosides of Oleanoic Acid and Hederagenin on Carbon Tetrachloride-Induced Hepatic Injury. Chem. Pharm. Bull. 1993, 41, 1395–1401. [Google Scholar] [CrossRef]
- Ahumada, F.; Trincado, M.A.; Arellano, J.A.; Hancke, J.; Wikman, G. Effect of Certain Adaptogenic Plant Extracts on Drug-Induced Narcosis in Female and Male Mice. Phytother. Res. 1991, 5, 29–31. [Google Scholar] [CrossRef]
- Wojcicki, J.; Samochowiec, L.; Kadlubowska, D.; Lutomski, J. Studies on the Saponin Fraction from the Root of Aralia mandshurica Rupr. et Maxim. Part IV. Influence of the Saponosides on the Content of Lipids in Blood Serum and Liver in Experimental Hyperlipemia. Herba Pol. 1977, 23, 285–289. [Google Scholar]
- Liu, X.-H.; Li, X.-M.; Han, C.-C.; Fang, X.-F.; Ma, L. Effects of Combined Therapy with Glipizide and Aralia Root Bark Extract on Glycemic Control and Lipid Profiles in Patients with Type 2 Diabetes Mellitus: Effects of Aralia on Type 2 Diabetes Mellitus. J. Sci. Food Agric. 2015, 95, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Xu, X.; Xu, H.; Wen, F.; Zhang, X.; Sun, H.; Yao, F.; Sun, G.; Sun, X. Effect of the total saponins of Aralia elata (Miq) seem on cardiac contractile function and intracellular calcium cycling regulation. J. Ethnopharmacol. 2014, 155, 240–247. [Google Scholar] [CrossRef] [PubMed]
- State Register of Pharmaceutical Products for Medical Purposes. Available online: https://grls.rosminzdrav.ru/default.aspx (accessed on 9 November 2022).
- Lee, J.E.; Sim, S.J.; Jeong, W.; Choi, C.W.; Kim, N.; Park, Y.; Kim, M.-J.; Lee, D.; Hong, S.S. Diterpenoids and phenolic analogues from the roots of Aralia continentalis. J. Asian Nat. Prod. Res. 2021, 23, 371–378. [Google Scholar] [CrossRef]
- Sun, Y.; Li, B.; Lin, X.; Xue, J.; Wang, Z.; Zhang, H.; Jiang, H.; Wang, Q.; Kuang, H. Simultaneous Determination of Four Triterpene Saponins in Aralia elata Leaves by HPLC-ELSD Combined with hierarchical clustering analysis: Simultaneous determination of four triterpene saponins. Phytochem. Anal. 2017, 28, 202–209. [Google Scholar] [CrossRef]
- Xing, X.; Yan, M.; Zhang, X.; Yang, L.; Jiang, H. Quantitative analysis of triterpenes in different parts of Aralia elata (Miq.) seem using HPLC–ELSD and their inhibition of human umbilical vein endothelial cell ox-ldl-induced apoptosis. J. Liq. Chromatogr. Relat. Technol. 2017, 40, 984–990. [Google Scholar] [CrossRef]
- Zhang, Y.; Peng, Y.; Li, L.; Zhao, L.; Hu, Y.; Hu, C.; Song, S. Studies on cytotoxic triterpene saponins from the leaves of Aralia elata. Food Chem. 2013, 138, 208–213. [Google Scholar] [CrossRef]
- Han, F.; Liang, J.; Yang, B.-Y.; Kuang, H.-X.; Xia, Y.-G. Identification and comparison of triterpene saponins in Aralia elata leaves and buds by the energy-resolved MSAll technique on a liquid chromatography/quadrupole time-of-flight mass spectrometry. J. Pharm. Biomed. Anal. 2021, 203, 114176. [Google Scholar] [CrossRef]
- Choi, Y.H.; van Spronsen, J.; Dai, Y.; Verberne, M.; Hollmann, F.; Arends, I.W.C.E.; Witkamp, G.-J.; Verpoorte, R. Are natural deep eutectic solvents the missing link in understanding cellular metabolism and physiology? Plant Physiol. 2011, 156, 1701–1705. [Google Scholar] [CrossRef] [PubMed]
- Paiva, A.; Craveiro, R.; Aroso, I.; Martins, M.; Reis, R.L.; Duarte, A.R.C. Natural Deep Eutectic Solvents—Solvents for the 21st century. ACS Sustain. Chem. Eng. 2014, 2, 1063–1071. [Google Scholar] [CrossRef]
- Dai, Y.; van Spronsen, J.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Natural Deep Eutectic Solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef]
- Fuad, F.M.; Nadzir, M.M.; Harun, A. Hydrophilic Natural Deep Eutectic Solvent: A review on physicochemical properties and extractability of bioactive compounds. J. Mol. Liq. 2021, 339, 116923. [Google Scholar] [CrossRef]
- Ivanović, M.; Alañón, M.E.; Arráez-Román, D.; Segura-Carretero, A. Enhanced and Green Extraction of Bioactive Compounds from Lippia citriodora by Tailor-Made Natural Deep Eutectic Solvents. Food Res. Int. 2018, 111, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.C.; Wu, P.; Li, Y.B.; Liu, T.C.; Zhang, L.; Zhou, Y.H. Natural Deep Eutectic Solvents as new green solvents to extract anthraquinones from Rheum palmatum L. RSC Adv. 2018, 8, 15069–15077. [Google Scholar] [CrossRef] [PubMed]
- Bosiljkov, T.; Dujmić, F.; Cvjetko Bubalo, M.; Hribar, J.; Vidrih, R.; Brnčić, M.; Zlatic, E.; Radojčić Redovniković, I.; Jokić, S. Natural Deep Eutectic Solvents and ultrasound-assisted extraction: Green approaches for extraction of wine lees anthocyanins. Food Bioprod. Process. 2017, 102, 195–203. [Google Scholar] [CrossRef]
- Alrugaibah, M.; Yagiz, Y.; Gu, L. Use Natural Deep Eutectic Solvents as efficient green reagents to extract procyanidins and anthocyanins from cranberry pomace and predictive modeling by RSM and artificial neural networking. Sep. Purif. Technol. 2021, 255, 117720. [Google Scholar] [CrossRef]
- He, X.; Yang, J.; Huang, Y.; Zhang, Y.; Wan, H.; Li, C. Green and efficient ultrasonic-assisted extraction of bioactive components from Salvia miltiorrhiza by Natural Deep Eutectic Solvents. Molecules 2019, 25, 140. [Google Scholar] [CrossRef]
- Torres-Vega, J.; Gómez-Alonso, S.; Pérez-Navarro, J.; Alarcón-Enos, J.; Pastene-Navarrete, E. Polyphenolic compounds extracted and purified from Buddleja globosa Hope (Buddlejaceae) leaves using Natural Deep Eutectic Solvents and centrifugal partition chromatography. Molecules 2021, 26, 2192. [Google Scholar] [CrossRef]
- Shang, X.; Dou, Y.; Zhang, Y.; Tan, J.-N.; Liu, X.; Zhang, Z. Tailor-made Natural Deep Eutectic Solvents for green extraction of isoflavones from Chickpea (Cicer arietinum L.) Sprouts. Ind. Crops Prod. 2019, 140, 111724. [Google Scholar] [CrossRef]
- Nadhira, A.; Febianli, D.; Fransisca, F.; Mun’Im, A.; Aryati, W.D. Natural Deep Eutectic Solvents ultrasound-assisted extraction (NADES-UAE) of trans-cinnamaldehyde and coumarin from cinnamon bark [Cinnamomum burmannii (Nees T. Nees) Blume]. JRP 2020, 24, 389–398. [Google Scholar] [CrossRef]
- Obluchinskaya, E.D.; Daurtseva, A.V.; Pozharitskaya, O.N.; Flisyuk, E.V.; Shikov, A.N. Natural Deep Eutectic Solvents as alternatives for extracting phlorotannins from brown algae. Pharm. Chem. J. 2019, 53, 243–247. [Google Scholar] [CrossRef]
- Obluchinskaya, E.D.; Pozharitskaya, O.N.; Zakharova, L.V.; Daurtseva, A.V.; Flisyuk, E.V.; Shikov, A.N. Efficacy of Natural Deep Eutectic Solvents for extraction of hydrophilic and lipophilic compounds from Fucus vesiculosus. Molecules 2021, 26, 4198. [Google Scholar] [CrossRef]
- Patil, S.S.; Pathak, A.; Rathod, V.K. Optimization and kinetic study of ultrasound Assisted Deep Eutectic Solvent based extraction: A greener route for extraction of curcuminoids from Curcuma longa. Ultrason. Sonochem. 2021, 70, 105267. [Google Scholar] [CrossRef] [PubMed]
- Wils, L.; Leman-Loubière, C.; Bellin, N.; Clément-Larosière, B.; Pinault, M.; Chevalier, S.; Enguehard-Gueiffier, C.; Bodet, C.; Boudesocque-Delaye, L. Natural Deep Eutectic Solvent formulations for spirulina: Preparation, intensification, and skin impact. Algal Res. 2021, 56, 102317. [Google Scholar] [CrossRef]
- Fang, X.; Li, Y.; Kua, Y.L.; Chew, Z.L.; Gan, S.; Tan, K.W.; Lee, T.Z.E.; Cheng, W.K.; Lau, H.L.N. Insights on the Potential of Natural Deep Eutectic Solvents (NADES) to Fine-Tune Durian Seed Gum for Use as Edible Food Coating. Food Hydrocoll. 2022, 132, 107861. [Google Scholar] [CrossRef]
- Li, Y.; Pan, Z.; Wang, B.; Yu, W.; Song, S.; Feng, H.; Zhao, W.; Zhang, J. Ultrasound-assisted extraction of bioactive alkaloids from Phellodendri amurensis cortex using Deep Eutectic Solvent aqueous solutions. New J. Chem. 2020, 44, 9172–9178. [Google Scholar] [CrossRef]
- Naseem, Z.; Zahid, M.; Hanif, M.; Shahid, M. Environmentally friendly extraction of bioactive compounds from Mentha arvensis using Deep Eutectic Solvent as green extraction media. Pol. J. Environ. Stud. 2020, 29, 3749–3757. [Google Scholar] [CrossRef]
- Yang, G.-Y.; Song, J.-N.; Chang, Y.-Q.; Wang, L.; Zheng, Y.-G.; Zhang, D.; Guo, L. Natural deep eutectic solvents for the extraction of bioactive steroidal saponins from Dioscoreae Nipponicae Rhizoma. Molecules 2021, 26, 2079. [Google Scholar] [CrossRef]
- Lanjekar, K.J.; Rathod, V.K. Green extraction of Glycyrrhizic acid from Glycyrrhiza glabra using choline chloride based Natural Deep Eutectic Solvents (NADESs). Process Biochem. 2021, 102, 22–32. [Google Scholar] [CrossRef]
- Xia, Y.-G.; Liang, J.; Li, G.-Y.; Yang, B.-Y.; Kuang, H.-X. Energy-Resolved Technique for Discovery and Identification of Malonyl-Triterpene Saponins in Caulophyllum robustum by UHPLC-Electrospray Fourier Transform Mass Spectrometry: Liquid Chromatography-Mass Spectrometry. J. Mass Spectrom. 2016, 51, 947–958. [Google Scholar] [CrossRef] [PubMed]
- Chernushevich, I.V.; Thomson, B.A. Collisional Cooling of Large Ions in Electrospray Mass Spectrometry. Anal. Chem. 2004, 76, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, V.U.; Perveen, S.; Bano, S. Guaiacin A and B from the Leaves of Guaiacum officinale. Planta Med. 1989, 55, 307–308. [Google Scholar] [CrossRef] [PubMed]
- Shao, C.-J.; Kasai, R.; Xu, J.-D.; Tanaka, O. Saponins from Roots of Kalopanax septemlobus (Thunb.) Koidz., Ciqiu: Structures of Kalopanax-Saponins C, D, E and F. Chem. Pharm. Bull. 1989, 37, 311–314. [Google Scholar] [CrossRef]
- Kasprzyk, Z.; Wojciechowski, Z. The Structure of Triterpenic Glycosides from the Flowers of Calendula officinalis L. Phytochemistry 1967, 6, 69–75. [Google Scholar] [CrossRef]
- Yen, P.H.; Cuc, N.T.; Huong, P.T.T.; Nhiem, N.X.; Hong Chuong, N.T.; Lien, G.T.K.; Huu Tai, B.; Tuyen, N.V.; Van Minh, C.; Van Kiem, P. Araliaarmoside: A New Triterpene Glycoside Isolated from the Leaves of Aralia armata. Nat. Prod. Commun. 2020, 15, 1–5. [Google Scholar] [CrossRef]
- Kochetkov, N.K.; Khorlin, A.J.; Vaskovsky, V.E. The Structures of Aralosides A and B. Tetrahedron Lett. 1962, 3, 713–716. [Google Scholar] [CrossRef]
- Song, S.; Nakamura, N.; Ma, C.; Hattori, M.; Xu, S. Four New Saponins from the Root Bark of Aralia elata. Chem. Pharm. Bull. 2000, 48, 838–842. [Google Scholar] [CrossRef]
- Ali, M.C.; Chen, J.; Zhang, H.; Li, Z.; Zhao, L.; Qiu, H. Effective Extraction of Flavonoids from Lycium barbarum L. Fruits by Deep Eutectic Solvents-Based Ultrasound-Assisted Extraction. Talanta 2019, 203, 16–22. [Google Scholar] [CrossRef]
- Maimulyanti, A.; Nurhidayati, I.; Mellisani, B.; Amelia Rachmawati Putri, F.; Puspita, F.; Restu Prihadi, A. Development of Natural Deep Eutectic Solvent (NADES) Based on Choline Chloride as a Green Solvent to Extract Phenolic Compound from Coffee Husk Waste. Arab. J. Chem. 2023, 16, 104634. [Google Scholar] [CrossRef]
- Lanjekar, K.J.; Rathod, V.K. Application of Ultrasound and Natural Deep Eutectic Solvent for the Extraction of Glycyrrhizic Acid from Glycyrrhiza glabra: Optimization and Kinetic Evaluation. Ind. Eng. Chem. Res. 2021, 60, 9532–9538. [Google Scholar] [CrossRef]
- Suresh, P.S.; Singh, P.P.; Anmol; Kapoor, S.; Padwad, Y.S.; Sharma, U. Lactic Acid-Based Deep Eutectic Solvent: An Efficient Green Media for the Selective Extraction of Steroidal Saponins from Trillium govanianum. Sep. Purif. Technol. 2022, 294, 121105. [Google Scholar] [CrossRef]
- Shi, X.; Yang, Y.; Ren, H.; Sun, S.; Mu, L.T.; Chen, X.; Wang, Y.; Zhang, Y.; Wang, L.H.; Sun, C. Identification of Multiple Components in Deep Eutectic Solvent Extract of Acanthopanax senticosus Root by Ultra-High-Performance Liquid Chromatography with Quadrupole Orbitrap Mass Spectrometry. Phytochem. Lett. 2020, 35, 175–185. [Google Scholar] [CrossRef]
- Liu, G.; Feng, S.; Sui, M.; Chen, B.; Sun, P. Extraction and Identification of Steroidal Saponins from Polygonatum cyrtonema Hua Using Natural Deep Eutectic Solvent-synergistic Quartz Sand Assisted Extraction Method. J. Sep. Sci. 2023, 2200823. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Tailoring properties of Natural Deep Eutectic Solvents with water to facilitate their applications. Food Chem. 2015, 187, 14–19. [Google Scholar] [CrossRef]
- Liu, X.; Ahlgren, S.; Korthout, H.A.A.J.; Salomé-Abarca, L.F.; Bayona, L.M.; Verpoorte, R.; Choi, Y.H. Broad Range Chemical Profiling of Natural Deep Eutectic Solvent Extracts Using a High Performance Thin Layer Chromatography–Based Method. J. Chromatogr. A 2018, 1532, 198–207. [Google Scholar] [CrossRef]
- Phaisan, S.; Makkliang, F.; Putalun, W.; Sakamoto, S.; Yusakul, G. Development of a Colorless Centella asiatica (L.) Urb. Extract Using a Natural Deep Eutectic Solvent (NADES) and Microwave-Assisted Extraction (MAE) Optimized by Response Surface Methodology. RSC Adv. 2021, 11, 8741–8750. [Google Scholar] [CrossRef]
- Dai, Y.; Verpoorte, R.; Choi, Y.H. Natural Deep Eutectic Solvents Providing Enhanced Stability of Natural Colorants from Safflower (Carthamus tinctorius). Food Chem. 2014, 159, 116–121. [Google Scholar] [CrossRef]
- Wang, Q.-H.; Zhang, J.; Ma, X.; Ye, X.-Y.; Yang, B.-Y.; Xia, Y.-G.; Kuang, H.-X. A New Triterpenoid Saponin from the Leaves of Aralia Elata. Chin. J. Nat. Med. 2011, 9, 17–21. [Google Scholar] [CrossRef]
- Jiang, Y.T.; Xu, S.X.; Gu, X.H.; Ren, L.; Chen, Y.J.; Yao, X.S.; Miao, Z.C. Studies on the chemical constituents from Aralia elata. Yao Xue Xue Bao 1992, 27, 528–532. [Google Scholar]
- Lin, G.; Yang, J.-s. Studies on the Chemical Constituents of Aralia decaisneane II. Chin. Pharm. J. Beijing 2004, 39, 575–578. [Google Scholar]
- Miyase, T.; Shiokawa, K.-I.; Zhang, D.M.; Ueno, A. Araliasaponins I–XI, Triterpene Saponins from the Roots of Aralia decaisneana. Phytochemistry 1996, 41, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Miyase, T.; Sutoh, N.; Zhang, D.M.; Ueno, A. Araliasaponins XII–XVIII, Triterpene Saponins from the Roots of Aralia chinensis. Phytochemistry 1996, 42, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Gao, R.; Liao, M.; Huang, X.; Chen, Y.; Yang, G.; Li, J. Six New Triterpene Derivatives from Aralia chinensis var. dasyphylloides. Molecules 2016, 21, 1700. [Google Scholar] [CrossRef]
- Sun, W.J.; Zhang, D.K.; Sha, Z.F.; Zhang, H.L.; Zhang, X.L. Studies on the Saponins from the Root Bark of Aralia chinensis L. Acta Pharm. Sin. 1991, 26, 197–202. [Google Scholar]
- Yu, S.-S.; Yu, D.-Q.; Liang, X.-T. Triterpenoid Saponins from the Roots of Aralia spinifolia. J. Nat. Prod. 1994, 57, 978–982. [Google Scholar] [CrossRef]
- Yen, P.H.; Chuong, N.T.H.; Lien, G.T.K.; Cuc, N.T.; Nhiem, N.X.; Thanh, N.T.V.; Tai, B.H.; Seo, Y.; Namkung, W.; Park, S. Oleanane-Type Triterpene Saponins from Aralia armata Leaves and Their Cytotoxic Activity. Nat. Prod. Res. 2021, 36, 142–149. [Google Scholar] [CrossRef]
- Zaki, A.A.; Qiu, L. Machaerinic Acid 3-O-β-D-Glucuronopyranoside from Calendula officinalis. Nat. Prod. Res. 2020, 34, 2938–2944. [Google Scholar] [CrossRef]
- Liang, X.F.; Zhao, Y.Y.; Liu, X.Z.; Yang, X.J.; Fan, Y.; Guo, D.Y.; Song, X.M.; Song, B. Isolation and Identification of Chemical Constituents from Aralia Taibaiensis Cortex. Chin. J. Exp. Tradit. Med. Formulae 2018, 20, 56–61. [Google Scholar]
- Yoshikawa, M.; Harada, E.; Matsuda, H.; Murakami, T.; Yamahara, J.; Murakami, N. Elatosides A and B, Potent Inhibitors of Ethanol Absorption in Rats from the Bark of Aralia elata SEEM.: The Structure-Activity Relationships of Oleanolic Acid Oligoglycosides. Chem. Pharm. Bull. 1993, 41, 2069–2071. [Google Scholar] [CrossRef]
- Song, S.-J.; Nakamura, N.; Ma, C.-M.; Hattori, M.; Xu, S.-X. Five Saponins from the Root Bark of Aralia elata. Phytochemistry 2001, 56, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Zhang, L.; Liu, Z. Analysis of Saponins from Leaves of Aralia elata by Liquid Chromatography and Multi-Stage Tandem Mass Spectrometry. Anal. Sci. 2009, 25, 753–755. [Google Scholar] [CrossRef]
- Shuyu, C.; Xianggao, L.; Chongxi, Z. Chemical components of Aralia elata. J. Jilin Agric. Univ. 1992, 14, 29–32. [Google Scholar]
- Ma, Z.; Song, S.; Xu, S. Two new saponins from Aralia elata (Miq.) see. Chin. J. Med. Chem. 2004, 14, 47–48. [Google Scholar]
- Kuang, H.-X.; Sun, H.; Zhang, N.; Okada, Y.; Okuyama, T. Two New Saponins, Congmuyenosides A and B, from the Leaves of Aralia elata Collected in Heilongjiang, China. Chem. Pharm. Bull. 1996, 44, 2183–2185. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Shim, S.H.; Chae, S.; Han, S.J.; Kang, S.S.; Son, K.H.; Chang, H.W.; Kim, H.P.; Bae, K. Saponins and Other Constituents from the Leaves of Aralia elata. Chem. Pharm. Bull. 2005, 53, 696–700. [Google Scholar] [CrossRef]
- Xi, F.-M.; Li, C.-T.; Han, J.; Yu, S.-S.; Wu, Z.-J.; Chen, W.-S. Thiophenes, Polyacetylenes and Terpenes from the Aerial Parts of Eclipata prostrata. Bioorg. Med. Chem. 2014, 22, 6515–6522. [Google Scholar] [CrossRef]
- Matsuo, Y.; Watanabe, K.; Mimaki, Y. Triterpene Glycosides from the Underground Parts of Caulophyllum thalictroides. J. Nat. Prod. 2009, 72, 1155–1160. [Google Scholar] [CrossRef]
- Nhiem, N.X.; Lim, H.Y.; Kiem, P.V.; Minh, C.V.; Thu, V.K.; Tai, B.H.; Quang, T.H.; Song, S.B.; Kim, Y.H. Oleanane-Type Triterpene Saponins from the Bark of Aralia elata and Their NF-ΚB Inhibition and PPAR Activation Signal Pathway. Bioorg. Med. Chem. Lett. 2011, 21, 6143–6147. [Google Scholar] [CrossRef]
- Tian, Y.; Zhang, X.; Liu, H.; Gong, D.; Li, X. Comparison of the Nutritional and Phytochemical Composition and Antioxidant Activities of Aralia elata (Miq.) Seem Fruits in Northeast China. Arab. J. Chem. 2021, 14, 103448. [Google Scholar] [CrossRef]
- Sakai, S.; Katsumata(Nee Ohtsuka), M.; Satoh, Y.; Nagasao, M.; Miyakoshi, M.; Ida, Y.; Shoji, J. Oleanolic Acid Saponins from Root Bark of Aralia elata. Phytochemistry 1994, 35, 1319–1324. [Google Scholar] [CrossRef] [PubMed]
- Kuljanabhagavad, T.; Thongphasuk, P.; Chamulitrat, W.; Wink, M. Triterpene Saponins from Chenopodium quinoa Willd. Phytochemistry 2008, 69, 1919–1926. [Google Scholar] [CrossRef] [PubMed]
- Waffo-Téguo, P.; Voutquenne, L.; Thoison, O.; Dumontet, V.; Nguyen, V.H.; Lavaud, C. Acetylated Glucuronide Triterpene Bidesmosidic Saponins from Symplocos glomerata. Phytochemistry 2004, 65, 741–750. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Murakami, T.; Harada, E.; Murakami, N.; Yamahara, J.; Matsuda, H. Bioactive Saponins and Glycosides. VI. Elatosides A and B, Potent Inhibitors of Ethanol Absorption, from the Bark of Aralia elata SEEM. (Araliaceae): The Structure-Requirement in Oleanolic Acid Glucuronide-Saponins for the Inhibitory Activity. Chem. Pharm. Bull. 1996, 44, 1915–1922. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.F.; Yi, Y.H.; Wang, Z.Z.; Jiang, Y.P.; Li, Y.Q. Oleanolic acid saponins from the root bark of Aralia taibaiensis. Yao Xue Xue Bao 1997, 32, 685–690. [Google Scholar] [PubMed]
- Miyase, T.; Kohsaka, H.; Ueno, A. Tragopogonosides A-I, Oleanane Saponins from Tragopogon pratensis. Phytochemistry 1992, 31, 2087–2091. [Google Scholar] [CrossRef]

| No. | tR (min) | m/z [M−H]− Observed | m/z [M−H]− Calculated | Elemental Composition [M−H]− | MS2 Fragmentation Patterns-Product Ions, m/z (Rel. Intensity) | Δm (ppm) | Assignment | Plant Part | Solvent | Ref. | Suppl. Spectra |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 3.8 | 911.4993 | 911.5010 | C47H75O17− | 455.3507 (20), 617.4041 (15), 749.4472 (15), 911.4993 (100) | 1.9 | Guaiacin B isomer 1 | X,Y,Z 1 | W,E,ND1,2,3,4,6,7 2 | [44] | Figure S1 |
| 2 | 3.8 | 1235.6107 | 1235.6066 | C59H95O27− | 455.3522 (50), 617.4059 (15), 749.4492 (100), 911.5035 (10), 1235.6107 (10) | −3.3 | Oleanolic acid-3-O- (triglucopyranosyl-1-3- arabinopyranosyl)-28-1- glucopyranosyl | X | W,E,ND1,2,3,4 | Figure S2 | |
| Y | W,E,ND1,2,3,4,6,7 | ||||||||||
| Z | W,E,ND1,3,4,6,7 | ||||||||||
| 3 | 3.9 | 1087.5317 | 1087.5331 | C53H83O23− | 455.3509 (10), 701.4265 (5), 925.4814 (15), 1087.5317 (100) | 1.3 | Kalopanax-Saponin F isomer 1 | X | W,E, ND1,2,3,4,6,7 | [45] | Figure S3 |
| Y | All | ||||||||||
| Z | W,E,ND1,3,4,6,7 | ||||||||||
| 4 | 3.9 | 1117.5390 | 1117.5436 | C54H85O24− | 455.3499 (3), 731.4347 (5), 955.4898 (10), 1117.5390 (100) | 4.1 | Calendulaglycoside A | X,Z | W,E,ND1,2,3,4,6,7 | [46] | Figure S4 |
| Y | All | ||||||||||
| 5 | 4.0 | 1249.5869 | 1249.5859 | C59H93O28− | 455.3487 (5), 701.4254 (15), 925.4743 (7), 1057.5219 (80), 1087.5337 (50), 1153.5538 (100), 1249.5869 (70) | −0.8 | Araliaarmoside | X,Y,Z | W,E,ND1,2,3,4,6,7 | [47] | Figure S5 |
| 6 | 4.3 | 1087.5336 | 1087.5331 | C53H83O23− | 455.3509 (10), 701.4265 (5), 925.4814 (15), 1087.55336 (100) | −0.5 | Kalopanax-Saponin F isomer 2 | X,Z | W,E,ND1,2,3,4,6,7 | [45] | Figure S3 |
| Y | All | ||||||||||
| 7 | 4.3 | 955.4859 | 955.4908 | C48H75O19− | 455.3511 (5), 569.379 (5), 793.4317 (15), 955.4859 (100) | 5.1 | Calendulaglycoside C isomer 1 | X,Y,Z | W,E,ND1,2,3,4,6,7 | [46] | Figure S6 |
| 8 | 4.3 | 1073.5569 | 1073.5538 | C53H85O22− | 455.3518 (50), 617.4052 (20), 749.4485 (30), 911.5022 (100), 1073.5569 (15) | −2.9 | Oleanolic acid-3-O- (diglucopyranosyl-1-3- arabinopyranosyl)-28-1- glucopyranosyl ester | X | E,ND1,2,3,4,6,7 | Figure S7 | |
| Y | E,ND1,2,3,6,7 | ||||||||||
| Z | ND1,3,6,7 | ||||||||||
| 9 | 4.3 | 1119.5606 | 1119.5616 | C54H87O24− | 455.3521 (5), 617.4057 (5), 749.4490 (7), 911.5019 (100), 1073.5551 (15), 1119.5606 (13) | 0.9 | Oleanolic acid-3-O- (methyldioxy-trihexopyranosyl-1-3- pentopyranosyl)-28-1- hexopyranosyl ester | X,Y | W,E,ND1,2,3,4,6,7 | Figure S8 | |
| Z | W,E,ND1,3,4,6,7 | ||||||||||
| 10 | 4.4 | 1057.5249 | 1057.5225 | C52H81O22− | 455.3517 (5), 701.4286 (7), 763.4309 (5), 895.4698 (7), 1057.5249 (100) | −2.3 | Araloside B | X,Y,Z | W,E,ND1,2,3,4,6,7 | [48] | Figure S9 |
| 11 | 4.5 | 1089.5493 | 1089.5487 | C53H85O23− | 455.3501 (5), 719.4360 (10), 881.4901 (100), 1043.5437 (12), 1089.5493 (12) | −0.6 | Araliasaponin III | X,Z | W,E,ND1,2,3,4,6,7 | [49] | Figure S10 |
| Y | W,E,ND1,3,4,6,7 | ||||||||||
| 12 | 4.6 | 793.4312 | 793.4380 | C42H65O14− | 455.3471 (10), 631.3785 (15), 793.4312 (100) | 8.6 | Oleanolic acid-3-O- (hexosyl)-28-1-hexouronide ester isomer 1 | X,Z | W,E,ND1,2,3,4,6,7 | Figure S11 | |
| Y | All | ||||||||||
| 13 | 5.1 | 955.4899 | 955.4908 | C48H75O19− | 455.3511 (5), 569.379 (5), 793.4317 (15), 955.4899 (100) | 0.9 | Calendulaglycoside C isomer 2 | X,Y,Z | W,E,ND1,2,3,4,6,7 | [46] | Figure S6 |
| 14 | 5.3 | 925.4805 | 925.4802 | C47H73O18− | 455.3508 (2), 569.3831 (5), 731.4366 (20), 925.4803 (100) | 0.3 | Araloside A isomer 1 | X,Z | W,E,ND1,2,3,4,6,7 | [48] | Figure S12 |
| Y | All | ||||||||||
| 15 | 5.4 | 911.5016 | 911.5010 | C47H75O17− | 455.3507 (20), 617.4041 (15), 749.4472 (15), 911.5016 (100) | −0.7 | Guaiacin B isomer 2 | X,Z | W,E,ND1,2,3,4,6,7 | [44] | Figure S1 |
| Y | All | ||||||||||
| 16 | 5.5 | 925.4807 | 925.4802 | C47H73O18− | 455.3508 (2), 569.3831 (5), 731.4366 (20), 925.4803 (100) | 0.5 | Araloside A isomer 2 | X,Z | W,E,ND1,2,3,4,6,7 | [48] | Figure S12 |
| Y | All | ||||||||||
| 17 | 5.6 | 895.4696 | - | - | 455.3508 (5), 551.3731 (5), 895.4696 (100) | Oleanolic acid unknown derivatives | X,Y,Z | W,E,ND1,2,3,4,6,7 | Figure S13 | ||
| 18 | 5.6 | 793.4360 | 793.4380 | C42H65O14− | 455.3471 (10), 631.3785 (15), 793.4312 (100) | 2.5 | Oleanolic acid-3-O- (hexosyl)-28-1-hexouronide ester isomer 2 | X,Z | W,E,ND1,2,3,4,6,7 | Figure S11 | |
| Y | All | ||||||||||
| 19 | 5.7 | 763.4260 | 763.4274 | C41H63O13− | 455.3502 (3), 631.3822 (5), 763.4260 (100) | 1.8 | Oleanolic acid 3-O- hexuronide-(1-3- pentafuranoside) | X,Z | W,E,ND1,2,3,4,6,7 | Figure S14 | |
| Y | All | ||||||||||
| 20 | 5.8 | 911.4949 | 911.5010 | C47H75O17− | 455.3507 (20), 617.4041 (15), 749.4472 (15), 911.4949 (100) | 6.7 | Guaiacin B isomer 3 | X,Y,Z | W,E,ND1,2,3,4,6,7 | [44] | Figure S1 |
| Code | Component 1 | Component 2 | Molar Ratio | Amount of Water (% (v/v)) |
|---|---|---|---|---|
| ND1 | Choline chloride | Malic acid | 1:1 | - |
| ND2 | Choline chloride | Malic acid | 1:2 | - |
| ND3 | Choline chloride | Lactic acid | 1:3 | - |
| ND4 | Choline chloride | Lactic acid | 1:3 | 30 |
| ND5 | Choline chloride | Oxalic acid | 1:1 | 15 |
| ND6 | Sorbitol | Malic acid | 1:1 | 10 |
| ND7 | Sorbitol | Malic acid | 1:2 | 20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrochenko, A.A.; Orlova, A.; Frolova, N.; Serebryakov, E.B.; Soboleva, A.; Flisyuk, E.V.; Frolov, A.; Shikov, A.N. Natural Deep Eutectic Solvents for the Extraction of Triterpene Saponins from Aralia elata var. mandshurica (Rupr. & Maxim.) J. Wen. Molecules 2023, 28, 3614. https://doi.org/10.3390/molecules28083614
Petrochenko AA, Orlova A, Frolova N, Serebryakov EB, Soboleva A, Flisyuk EV, Frolov A, Shikov AN. Natural Deep Eutectic Solvents for the Extraction of Triterpene Saponins from Aralia elata var. mandshurica (Rupr. & Maxim.) J. Wen. Molecules. 2023; 28(8):3614. https://doi.org/10.3390/molecules28083614
Chicago/Turabian StylePetrochenko, Alyona A., Anastasia Orlova, Nadezhda Frolova, Evgeny B. Serebryakov, Alena Soboleva, Elena V. Flisyuk, Andrej Frolov, and Alexander N. Shikov. 2023. "Natural Deep Eutectic Solvents for the Extraction of Triterpene Saponins from Aralia elata var. mandshurica (Rupr. & Maxim.) J. Wen" Molecules 28, no. 8: 3614. https://doi.org/10.3390/molecules28083614
APA StylePetrochenko, A. A., Orlova, A., Frolova, N., Serebryakov, E. B., Soboleva, A., Flisyuk, E. V., Frolov, A., & Shikov, A. N. (2023). Natural Deep Eutectic Solvents for the Extraction of Triterpene Saponins from Aralia elata var. mandshurica (Rupr. & Maxim.) J. Wen. Molecules, 28(8), 3614. https://doi.org/10.3390/molecules28083614








