Antioxidant Guided Fractionation of Blackberry Polyphenols Show Synergistic Role of Catechins and Ellagitannins
Abstract
1. Introduction
2. Results and Discussion
2.1. Antioxidant Indices of Phenolic-Rich Fractions of Blackberries
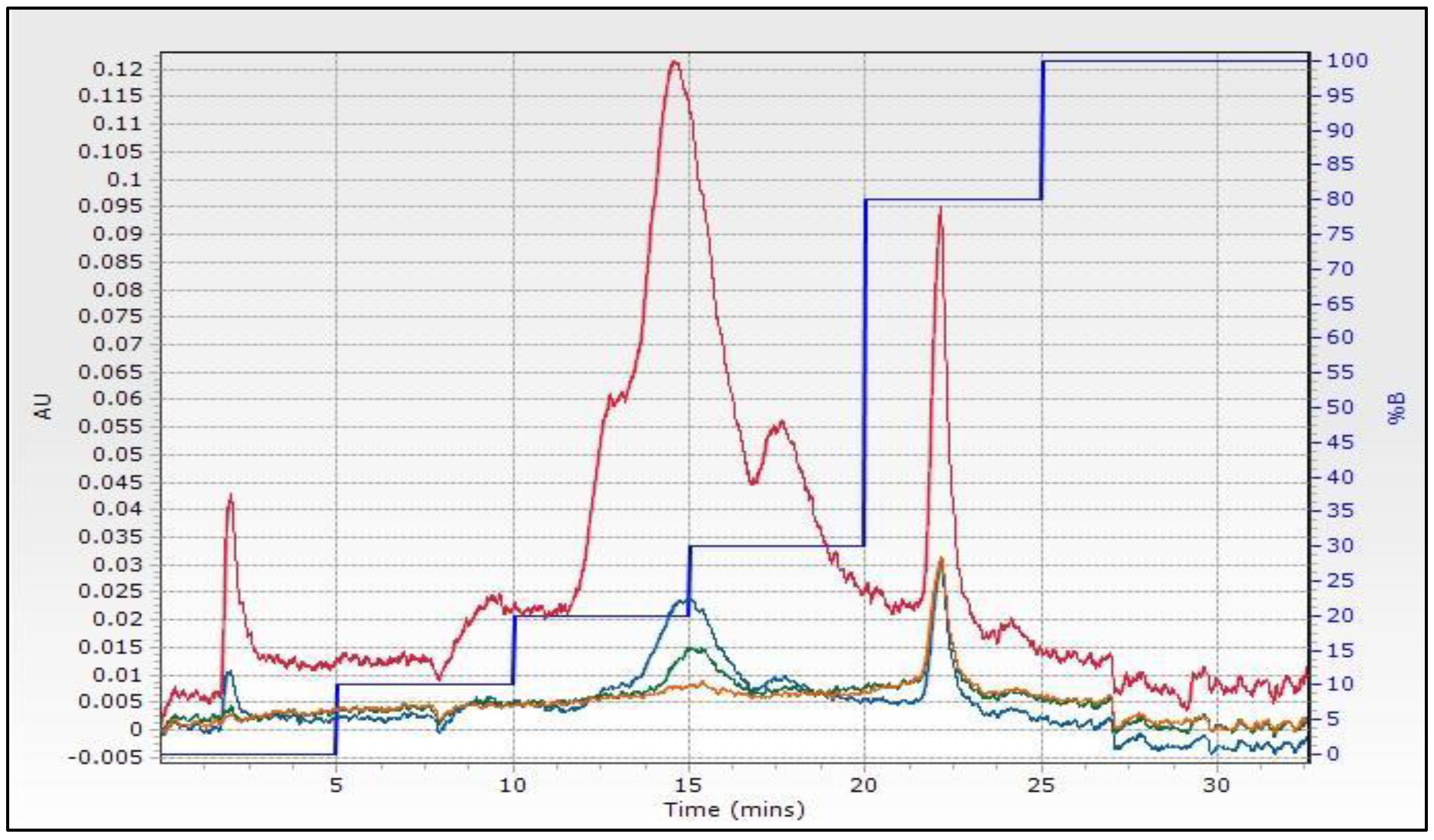
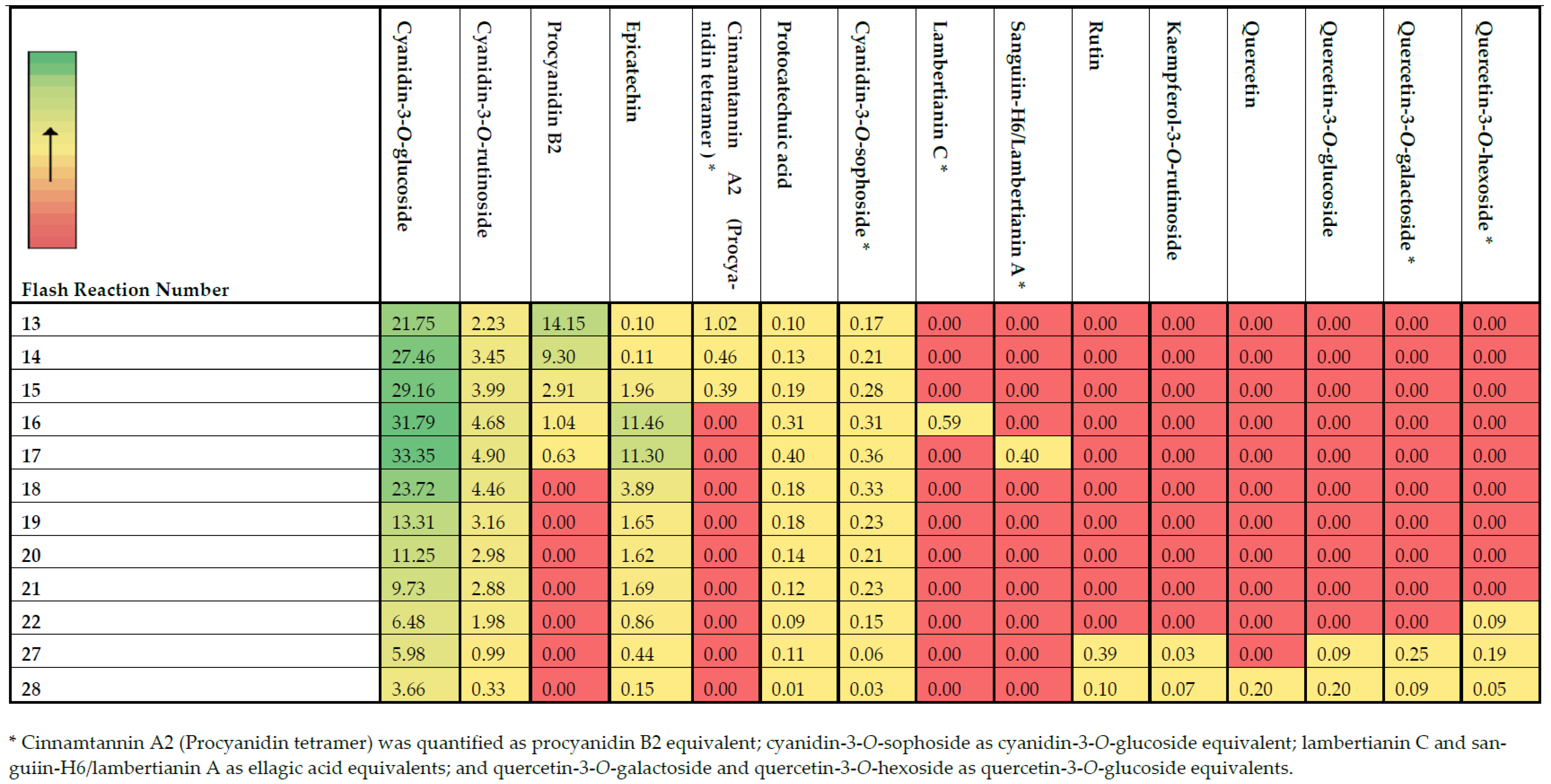
2.2. Characterization of Phenolic Compounds by LC-Q-TOF-MS/MS and UPLC-TQD-MS/MS Analyses
| Peak No. | Name | tR (min.) | Molecular Formula | Observed [M-H]− m/z | Calculated [M-H]− m/z | Mass Error (ppm) | MS/MS Ions m/z | References |
|---|---|---|---|---|---|---|---|---|
| 1 | Gallic acid | 0.93 | C7H6O5 | 169.0143 | 169.0137 | 3.5 | 125.03; 111.03; 96.98; 155.03 | [20,21] 1 |
| 2 | Neochlorogenic acid | 1.25 | C16H18O9 | 353.0866 | 353.0873 | −2.0 | 191.09; 155.05; 179.07 | [22] 1 |
| 3 | Catechin | 2.21 | C15H14O6 | 289.0723 | 289.0712 | 3.8 | 245.09; 203.08; 125.04 | [22] 1 |
| 4 | Protocatechuic acid | 2.40 | C7H6O4 | 153.0185 | 153.0188 | −2.0 | 109.03; 91.02 | [22] 1 |
| 5 | Dihydromyricetin rhamnoside | 2.48 | C21H22O12 | 465.1015 | 465.1033 | −3.9 | 125.03; 109.01; 241.06; 329.11 | [22] |
| 6 | Cyanidin-3-O-glucoside | 2.53 | [C21H21O11]+ | 447.0929 | 447.0927 | 0.4 | 285.05; 269.06; 125.03 | [20] 1 |
| 7 | Cyanidin-3-O-rutinoside | 2.66 | [C27H31O15]+ | 593.1597 | 593.1506 | −1.5 | 285.06; 284.05; 269.06; 287.07 | [23,24] 1 |
| 8 | Procyanidin trimer C type | 2.85 | C45H38O18 | 865.1987 | 865.1980 | 0.8 | 407.18; 289.14; 243.10 | [25,26] |
| 9 | 3-p-Coumaroylquinic acid | 2.95 | C16H18O8 | 337.0925 | 337.0923 | 0.6 | 173.05; 191.07 | [27,28] |
| 10 | Procyanthocyanidin type | 2.98 | C43H32O11 | 723.1888 | 723.1866 | 3.0 | 289.15; 407.19; 525.26 | [29] |
| 11 | Cyanidin-3-O-sophoroside | 3.11 | [C27H31O16]+ | 611.1586 | 611.1612 | −1.2 | 285.06; 475.16; 241.07 | [30] |
| 12 | Procyanidin B2 | 3.25 | C30H26O12 | 577.1339 | 577.1346 | −4.3 | 289.15; 407.18; 125.07; 408.19 | [31,32] 1 |
| 13 | Cinnamtannin A2 | 3.35 | C60H50O24 | 1155.2719 | 1155.2771 | 0.6 | 577.3; 425.2; 407.2; 287.1; 451.2 | [33] 2 |
| 14 | 3-Feruloylquinic acid | 3.44 | C17H20O9 | 367.1012 | 367.1029 | −4.6 | 166.58; 193.02 | [28,34] |
| 15 | Procyanidin trimer C type | 3.75 | C45H38O18 | 865.1976 | 865.1980 | −0.5 | 287.13; 125.06; 407.18; 243.10 | [25,26] |
| 16 | Epicatechin | 4.31 | C15H14O6 | 289.0726 | 289.0712 | 4.8 | 245.08; 203.06; 125.02; 179.04 | [24,35] 1 |
| 17 | Rutin | 6.50 | C27H30O16 | 609.1430 | 609.1456 | −4.3 | 301.05; 271.04; 255.05; 151.02 | [3] 1 |
| 18 | Epigallocatechin | 6.55 | C15H14O7 | 305.0663 | 305.0661 | 0.7 | 219.11; 146.99; 225.92; 196.97 | [36] |
| 19 | Lambertianin C isomer 3 | 6.87 | [C123H80O78]2- | 1401.1112 | 1401.1068 | 3.1 | 301.01; 633.09; 1250.64; 935.11 | [37] |
| 20 | Sanguiin H-6/Lambertianin A | 7.19 | C82H54O52 | 1869.1470 | 1869.1503 | −1.8 | 935.09; 1235.16; 1567.15; 633.08 | [37] |
| 21 | Ellagic acid pentoside | 8.09 | C19H14O12 | 433.0403 | 433.0407 | −0.9 | 300.98; 153.03; 78.98 | [3] |
| 22 | Ellagic acid | 8.14 | C14H6O8 | 300.9998 | 300.9984 | 4.7 | 301.01; 117.05; 284.02; 229.03 | [37] 1 |
| 23 | Quercetin-3-O-galactoside | 8.15 | C21H20O12 | 463.0867 | 463.0877 | −2.2 | 301.03; 271.02; 255.03 | [3] 1 |
| 24 | Quercetin-3-O-glucoside | 8.46 | C21H20O12 | 463.0866 | 463.0877 | −2.4 | 301.06; 151.01; 271.05; 255.04 | [3] 1 |
| 25 | Quercetin-3-O-hexoside | 8.68 | C21H20O12 | 463.0865 | 463.0877 | −2.0 | 301.05; 271.03; 151.02 | [3] |
| 26 | Quercetin-3-pentoside | 9.19 | C20H18O11 | 433.0783 | 433.0771 | 2.8 | 301.06; 271.09; 154.04 | [3] |
| 27 | Kaempferol-3-O-rutinoside | 9.32 | C27H30O15 | 593.1491 | 593.1506 | −2.5 | 269.10; 285.09; 257.11; 225.11; 125.05 | [3] 1 |
| 28 | Quercetin | 12.25 | C15H10O7 | 301.0357 | 301.0348 | 3.0 | 178.05; 125.05; 137.05; 147.00 | [37] 1 |
3. Materials and Methods
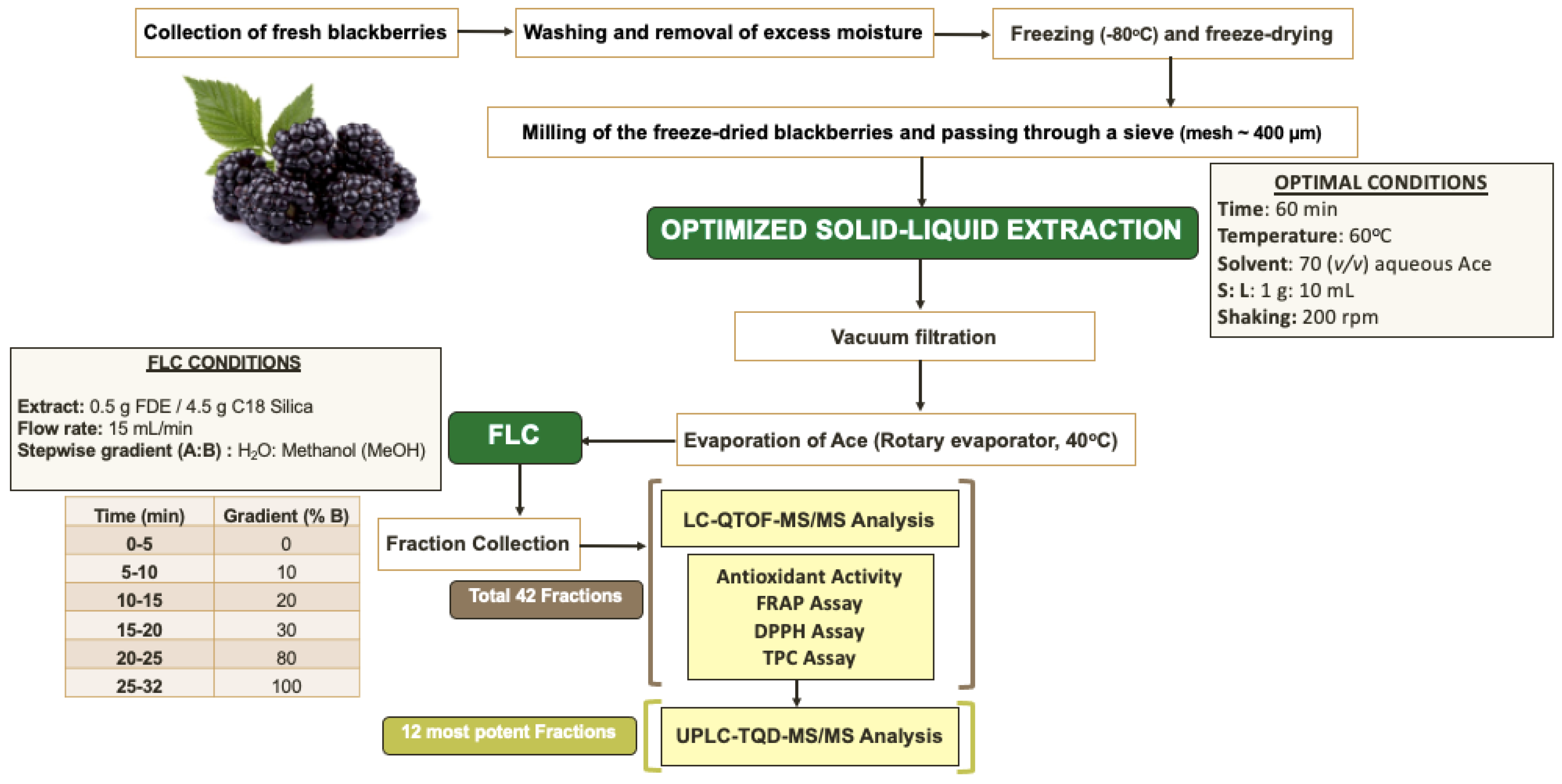
3.1. Samples and Reagents
3.2. Extraction of Phenolic Compounds from Blackberries
3.3. Flash Chromatography
3.4. Determination of TPC with the F-C Assay
3.5. Ferric Reducing Antioxidant Power (FRAP) Assay
3.6. 2,2′-Diphenyl-1-picrylhydrazyl Radical (DPPH•) Assay
3.7. Characterization of Phenolic Compounds by LC-QTOF-MS/MS Analysis
3.8. Quantification of Phenolic Compounds through UPLC-TQD-MS/MS Analysis
3.9. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Madrigal-Gamboa, V.; Jiménez-Arias, J.; Hidalgo, O.; Quesada, S.; Pérez, A.M.; Azofeifa, G. Membrane Processing Effect of Blackberry (Rubus Adenotrichos) on Cytotoxic and pro-Apoptotic Activities against Cancer Cell Lines. J. Food Process. Preserv. 2021, 45, e15575. [Google Scholar] [CrossRef]
- Ahmed, S.; Mia, S.; Shipon, T.P.; Kanan, K.A.; Ahmed, M. Physicochemical Properties, Bioactive Compounds and Total Antioxidant Activity of Blackberry (Syzygium Cumini L.) Juice Retained by Preservatives during Storage. J. Food Meas. Charact. 2021, 15, 3660–3669. [Google Scholar] [CrossRef]
- Oszmiański, J.; Nowicka, P.; Teleszko, M.; Wojdyło, A.; Cebulak, T.; Oklejewicz, K. Analysis of Phenolic Compounds and Antioxidant Activity in Wild Blackberry Fruits. Int. J. Mol. Sci. 2015, 16, 14540–14553. [Google Scholar] [CrossRef] [PubMed]
- Gong, E.S.; Li, B.; Li, B.; Podio, N.S.; Chen, H.; Li, T.; Sun, X.; Gao, N.; Wu, W.; Yang, T.; et al. Identification of Key Phenolic Compounds Responsible for Antioxidant Activities of Free and Bound Fractions of Blackberry Varieties’ Extracts by Boosted Regression Trees. J. Sci. Food Agric. 2022, 102, 984–994. [Google Scholar] [CrossRef] [PubMed]
- Sariburun, E.; Şahin, S.; Demir, C.; Türkben, C.; Uylaşer, V. Phenolic Content and Antioxidant Activity of Raspberry and Blackberry Cultivars. J. Food Sci. 2010, 75, C328–C335. [Google Scholar] [CrossRef] [PubMed]
- Acosta, O.; Vaillant, F.; Pérez, A.M.; Dornier, M. Potential of Ultrafiltration for Separation and Purification of Ellagitannins in Blackberry (Rubus Adenotrichus Schltdl.) Juice. Sep. Purif. Technol. 2014, 125, 120–125. [Google Scholar] [CrossRef]
- Ghosh, P.; Pradhan, R.C.; Mishra, S.; Rout, P.K. Quantification and Concentration of Anthocyanidin from Indian Blackberry (Jamun) by Combination of Ultra- and Nano-Filtrations. Food Bioprocess Technol. 2018, 11, 2194–2203. [Google Scholar] [CrossRef]
- Elisia, I.; Hu, C.; Popovich, D.G.; Kitts, D.D. Antioxidant Assessment of an Anthocyanin-Enriched Blackberry Extract. Food Chem. 2007, 101, 1052–1058. [Google Scholar] [CrossRef]
- Sánchez-Velázquez, O.A.; Cuevas-Rodríguez, E.-O.; Reyes-Moreno, C.; Ríos-Iribe, Y.; Hernández-Álvarez, A.J.; León-López, L.; Milán-Carrillo, J. Profiling Modifications in Physicochemical, Chemical and Antioxidant Properties of Wild Blackberry (Rubus Sp.) during Fermentation with EC 1118 Yeast. J. Food Sci. Technol. 2021, 58, 4654–4665. [Google Scholar] [CrossRef]
- Cho, M.J.; Howard, L.R.; Prior, R.L.; Clark, J.R. Flavonoid Glycosides and Antioxidant Capacity of Various Blackberry, Blueberry and Red Grape Genotypes Determined by High-Performance Liquid Chromatography/Mass Spectrometry. J. Sci. Food Agric. 2004, 84, 1771–1782. [Google Scholar] [CrossRef]
- Roge, A.B.; Firke, S.N.; Kawade, R.M.; Sarje, S.K.; Vadvalkar, S.M. Brief Review on: Flash Chromatography. IJPSR 2011, 2, 1930. [Google Scholar]
- Gangopadhyay, N.; Rai, D.K.; Brunton, N.P.; Gallagher, E.; Hossain, M.B. Antioxidant-Guided Isolation and Mass Spectrometric Identification of the Major Polyphenols in Barley (Hordeum Vulgare) Grain. Food Chem. 2016, 210, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Barbosa-Pereira, L.; Pocheville, A.; Angulo, I.; Paseiro-Losada, P.; Cruz, J.M. Fractionation and Purification of Bioactive Compounds Obtained from a Brewery Waste Stream. BioMed Res. Int. 2013, 2013, 408491. [Google Scholar] [CrossRef] [PubMed]
- Covarrubias-Cárdenas, A.G.; Martínez-Castillo, J.I.; Medina-Torres, N.; Ayora-Talavera, T.; Espinosa-Andrews, H.; García-Cruz, N.U.; Pacheco, N. Antioxidant Capacity and UPLC-PDA ESI-MS Phenolic Profile of Stevia Rebaudiana Dry Powder Extracts Obtained by Ultrasound Assisted Extraction. Agronomy 2018, 8, 170. [Google Scholar] [CrossRef]
- Covarrubias-Cárdenas, A.; Patrón-Vázquez, J.; Espinosa-Andrews, H.; Ayora-Talavera, T.; García-Cruz, U.; Pacheco, N. Antioxidant Capacity and UPLC-PDA ESI-MS Polyphenolic Profile of Citrus Aurantium Extracts Obtained by Ultrasound Assisted Extraction. J. Food Sci. Technol. 2018, 55, 5106–5114. [Google Scholar] [CrossRef]
- Zhang, D.; Chu, L.; Liu, Y.; Wang, A.; Ji, B.; Wu, W.; Zhou, F.; Wei, Y.; Cheng, Q.; Cai, S.; et al. Analysis of the Antioxidant Capacities of Flavonoids under Different Spectrophotometric Assays Using Cyclic Voltammetry and Density Functional Theory. J. Agric. Food Chem. 2011, 59, 10277–10285. [Google Scholar] [CrossRef] [PubMed]
- Katalinic, V.; Milos, M.; Kulisic, T.; Jukic, M. Screening of 70 Medicinal Plant Extracts for Antioxidant Capacity and Total Phenols. Food Chem. 2006, 94, 550–557. [Google Scholar] [CrossRef]
- Clarke, G.; Ting, K.; Wiart, C.; Fry, J. High Correlation of 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Radical Scavenging, Ferric Reducing Activity Potential and Total Phenolics Content Indicates Redundancy in Use of All Three Assays to Screen for Antioxidant Activity of Extracts of Plants from the Malaysian Rainforest. Antioxidants 2013, 2, 1–10. [Google Scholar] [CrossRef]
- Landete, J.M. Ellagitannins, Ellagic Acid and Their Derived Metabolites: A Review about Source, Metabolism, Functions and Health. Food Res. Int. 2011, 44, 1150–1160. [Google Scholar] [CrossRef]
- Chen, W.; Xu, Y.; Zhang, L.; Su, H.; Zheng, X. Blackberry Subjected to in Vitro Gastrointestinal Digestion Affords Protection against Ethyl Carbamate-Induced Cytotoxicity. Food Chem. 2016, 212, 620–627. [Google Scholar] [CrossRef]
- Singh, A.; Bajapai, V.; Kumar, S.; Sharma, K.R.; Kumara, B. Profiling of Gallic and Ellagic Acid Derivatives in Different Plant Parts of Terminalia Arjuna by HPLC-ESI-QTOF-MS/MS. Nat. Prod. Commun. 2016, 11, 239–244. [Google Scholar] [CrossRef]
- D’Angelo, R.W.O.; Gonçalves, M.M.; Fachi, M.M.; de Vilhena, R.O.; Pontarolo, R.; Maluf, D.F. UPLC–QToF-MS Characterization of Blackberry Extracts of Cultivars ‘Tupy’, ‘Guarani’, and ‘Xavante’: Development of Extract-Loaded Niosomes. Rev. Bras. Farmacogn. 2020, 30, 519–527. [Google Scholar] [CrossRef]
- Vagiri, M.; Ekholm, A.; Andersson, S.; Johansson, E.; Rumpunen, K. An Optimized Method for Analysis of Phenolic Compounds in Buds, Leaves, and Fruits of Black Currant (Ribes nigrum L.). J. Agric. Food Chem. 2012, 60, 10501–10510. [Google Scholar] [CrossRef]
- de Gomes, M.G.; Del Fabbro, L.; Rossito Goes, A.T.; Souza, L.C.; Donato, F.; Boeira, S.P.; Prigol, M.; Jesse, C.R. Blackberry Juice Anthocyanidins Limit Cisplatin-Induced Renal Pathophysiology in Mice. Pathophysiol. Off. J. Int. Soc. Pathophysiol. 2019, 26, 137–143. [Google Scholar] [CrossRef]
- Rockenbach, I.I.; Jungfer, E.; Ritter, C.; Santiago-Schübel, B.; Thiele, B.; Fett, R.; Galensa, R. Characterization of Flavan-3-Ols in Seeds of Grape Pomace by CE, HPLC-DAD-MSn and LC-ESI-FTICR-MS. Food Res. Int. 2012, 48, 848–855. [Google Scholar] [CrossRef]
- Czyżowska, A.; Wilkowska, A.; Staszczak Mianowska, A.; Nowak, A. Characterization of Phytochemicals in Berry Fruit Wines Analyzed by Liquid Chromatography Coupled to Photodiode-Array Detection and Electrospray Ionization/Ion Trap Mass Spectrometry (LC-DAD-ESI-MSn) and Their Antioxidant and Antimicrobial Activity. Foods 2020, 9, 1783. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, C.; Wang, D.; Zhang, J.; Zhang, T. Emerging Small-Molecule Inhibitors of the Bruton’s Tyrosine Kinase (BTK): Current Development. Eur. J. Med. Chem. 2021, 217, 113329. [Google Scholar] [CrossRef] [PubMed]
- Schuster, B.; Herrmann, K. Hydroxybenzoic and Hydroxycinnamic Acid Derivatives in Soft Fruits. Phytochemistry 1985, 24, 2761–2764. [Google Scholar] [CrossRef]
- Liao, X.; Greenspan, P.; Srivastava, A.; Pegg, R.B. Characterizing the Phenolic Constituents of U.S. Southeastern Blackberry Cultivars. J. Berry Res. 2020, 10, 311–327. [Google Scholar] [CrossRef]
- Hillebrand, S.; Schwarz, M.; Winterhalter, P. Characterization of Anthocyanins and Pyranoanthocyanins from Blood Orange [Citrus sinensis (L.) Osbeck] Juice. J. Agric. Food Chem. 2004, 52, 7331–7338. [Google Scholar] [CrossRef]
- de Pascual-Teresa, S.; Santos-Buelga, C.; Rivas-Gonzalo, J.C. Quantitative Analysis of Flavan-3-Ols in Spanish Foodstuffs and Beverages. J. Agric. Food Chem. 2000, 48, 5331–5337. [Google Scholar] [CrossRef] [PubMed]
- Bystrom, L.M.; Lewis, B.A.; Brown, D.L.; Rodriguez, E.; Obendorf, R.L. Characterization of Phenolics by LC-UV/Vis, LC-MS/MS and Sugars by GC in Melicoccus Bijugatus Jacq. ‘Montgomery’ Fruits. Food Chem. 2008, 111, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Enomoto, H.; Takahashi, S.; Takeda, S.; Hatta, H. Distribution of Flavan-3-Ol Species in Ripe Strawberry Fruit Revealed by Matrix-Assisted Laser Desorption/Ionization-Mass Spectrometry Imaging. Molecules 2019, 25, 103. [Google Scholar] [CrossRef] [PubMed]
- Ramabulana, A.-T.; Steenkamp, P.; Madala, N.; Dubery, I.A. Profiling of Chlorogenic Acids from Bidens Pilosa and Differentiation of Closely Related Positional Isomers with the Aid of UHPLC-QTOF-MS/MS-Based In-Source Collision-Induced Dissociation. Metabolites 2020, 10, 178. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Veberic, R.; Hudina, M.; Zorenc, Z.; Koron, D.; Senica, M. Fruit Quality Characteristics and Biochemical Composition of Fully Ripe Blackberries Harvested at Different Times. Foods 2021, 10, 1581. [Google Scholar] [CrossRef]
- Wang, W.; Ye, Z.; Gao, H.; Ouyang, D. Computational Pharmaceutics—A New Paradigm of Drug Delivery. J. Control. Release 2021, 338, 119–136. [Google Scholar] [CrossRef]
- Hager, T.J.; Howard, L.R.; Liyanage, R.; Lay, J.O.; Prior, R.L. Ellagitannin Composition of Blackberry as Determined by HPLC-ESI-MS and MALDI-TOF-MS. J. Agric. Food Chem. 2008, 56, 661–669. [Google Scholar] [CrossRef]
- Kolniak-Ostek, J.; Kucharska, A.Z.; Sokół-Łętowska, A.; Fecka, I. Characterization of Phenolic Compounds of Thorny and Thornless Blackberries. J. Agric. Food Chem. 2015, 63, 3012–3021. [Google Scholar] [CrossRef]
- Becker, C.; Klaering, H.-P.; Kroh, L.W.; Krumbein, A. Cool-Cultivated Red Leaf Lettuce Accumulates Cyanidin-3-O-(6″-O-Malonyl)-Glucoside and Caffeoylmalic Acid. Food Chem. 2014, 146, 404–411. [Google Scholar] [CrossRef]
- Amico, V.; Chillemi, R.; Mangiafico, S.; Spatafora, C.; Tringali, C. Polyphenol-Enriched Fractions from Sicilian Grape Pomace: HPLC–DAD Analysis and Antioxidant Activity. Bioresour. Technol. 2008, 99, 5960–5966. [Google Scholar] [CrossRef]
- Erkan, N.; Ayranci, G.; Ayranci, E. Antioxidant Activities of Rosemary (Rosmarinus officinalis L.) Extract, Blackseed (Nigella Sativa L.) Essential Oil, Carnosic Acid, Rosmarinic Acid and Sesamol. Food Chem. 2008, 110, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Boeing, J.S.; Barizão, E.O.; E Silva, B.C.; Montanher, P.F.; de Cinque Almeida, V.; Visentainer, J.V. Evaluation of Solvent Effect on the Extraction of Phenolic Compounds and Antioxidant Capacities from the Berries: Application of Principal Component Analysis. Chem. Cent. J. 2014, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. In Methods in Enzymology; Oxidants and Antioxidants Part A; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Stratil, P.; Klejdus, B.; Kubán, V. Determination of Total Content of Phenolic Compounds and Their Antioxidant Activity in Vegetables--Evaluation of Spectrophotometric Methods. J. Agric. Food Chem. 2006, 54, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Goupy, P.; Hugues, M.; Boivin, P.; Amiot, M.J. Antioxidant Composition and Activity of Barley (Hordeum Vulgare) and Malt Extracts and of Isolated Phenolic Compounds. J. Sci. Food Agric. 1999, 79, 1625–1634. [Google Scholar] [CrossRef]
- Xu, L.; Liu, Y.; Wu, H.; Wu, H.; Liu, X.; Zhou, A. Rapid Identification of Chemical Profile in Gandou Decoction by UPLC-Q-TOF-MSE Coupled with Novel Informatics UNIFI Platform. J. Pharm. Anal. 2020, 10, 35–48. [Google Scholar] [CrossRef]

| DPPH | FRAP | TPC | |
|---|---|---|---|
| DPPH | 1.000 | ||
| FRAP | 0.986 | 1.000 | |
| TPC | 0.987 | 0.987 | 1.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tzima, K.; Putsakum, G.; Rai, D.K. Antioxidant Guided Fractionation of Blackberry Polyphenols Show Synergistic Role of Catechins and Ellagitannins. Molecules 2023, 28, 1933. https://doi.org/10.3390/molecules28041933
Tzima K, Putsakum G, Rai DK. Antioxidant Guided Fractionation of Blackberry Polyphenols Show Synergistic Role of Catechins and Ellagitannins. Molecules. 2023; 28(4):1933. https://doi.org/10.3390/molecules28041933
Chicago/Turabian StyleTzima, Katerina, Gontorn Putsakum, and Dilip K. Rai. 2023. "Antioxidant Guided Fractionation of Blackberry Polyphenols Show Synergistic Role of Catechins and Ellagitannins" Molecules 28, no. 4: 1933. https://doi.org/10.3390/molecules28041933
APA StyleTzima, K., Putsakum, G., & Rai, D. K. (2023). Antioxidant Guided Fractionation of Blackberry Polyphenols Show Synergistic Role of Catechins and Ellagitannins. Molecules, 28(4), 1933. https://doi.org/10.3390/molecules28041933









