Polysaccharides from Radix Peucedani: Extraction, Structural Characterization and Antioxidant Activity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of Ultrasound-Assisted Extraction of Polysaccharides Using DESs
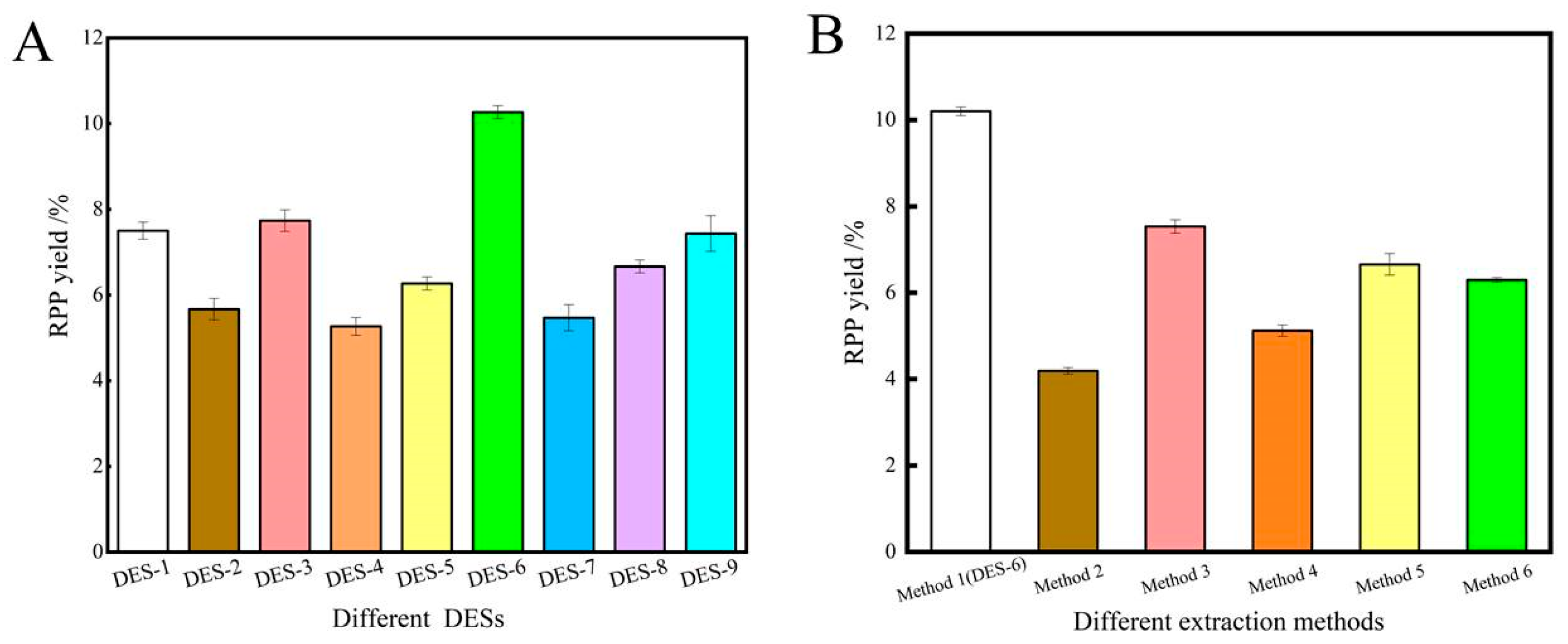
2.1.1. Influence of the DES System
2.1.2. Influence of DES Water Content on Extraction Yield
2.1.3. Influence of Other Factors on Extraction Yield
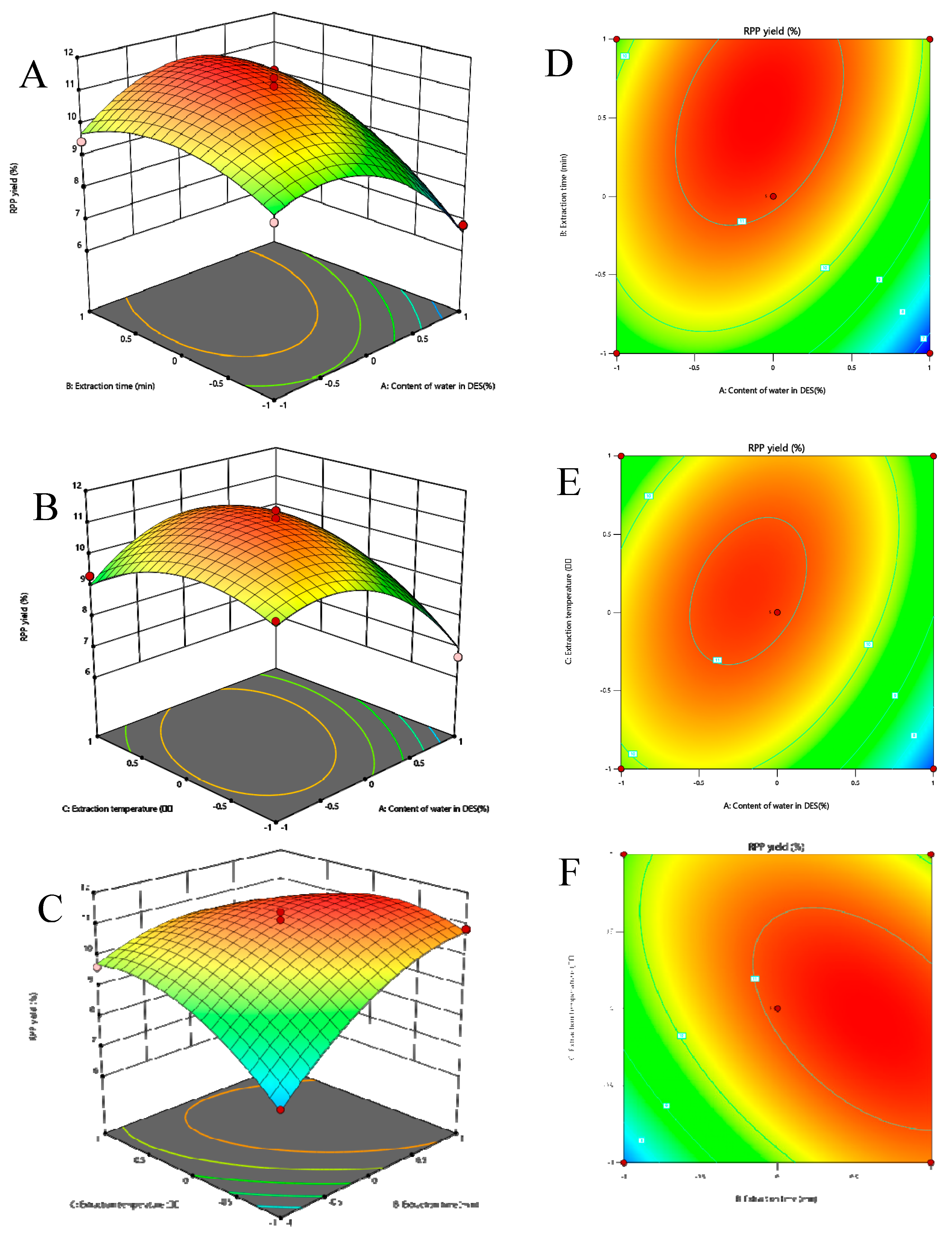
2.1.4. Optimization of Extraction Conditions by Box–Behnken Design
2.1.5. Optimization and Validation Experiments
2.1.6. Effect of Response Surface Interaction on Extraction Rate
2.2. Scanning Electron Microscopy (SEM) Image
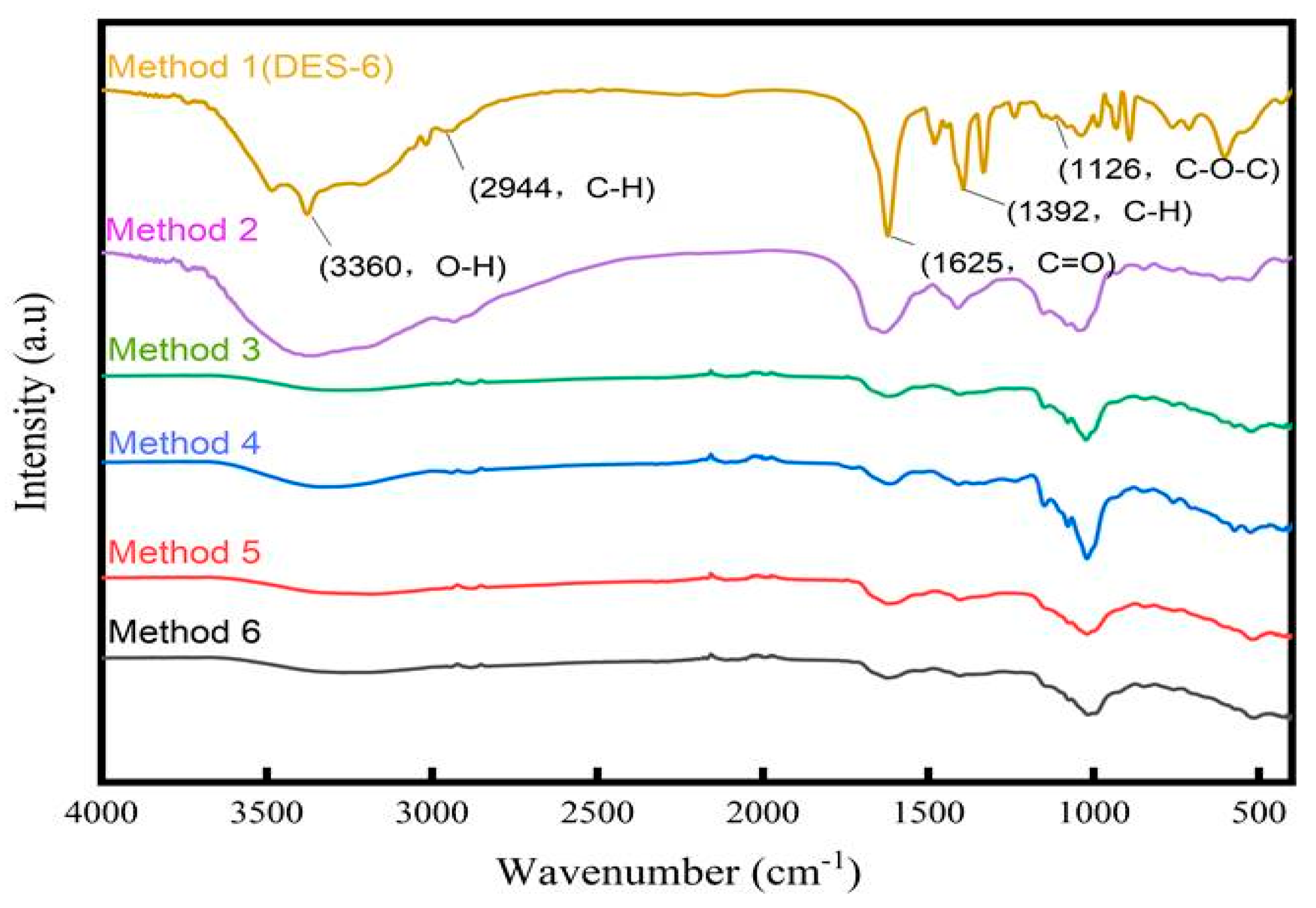
2.3. Fourier-Transform Infrared (FT-IR) Spectroscopy
2.4. Antioxidant Activity
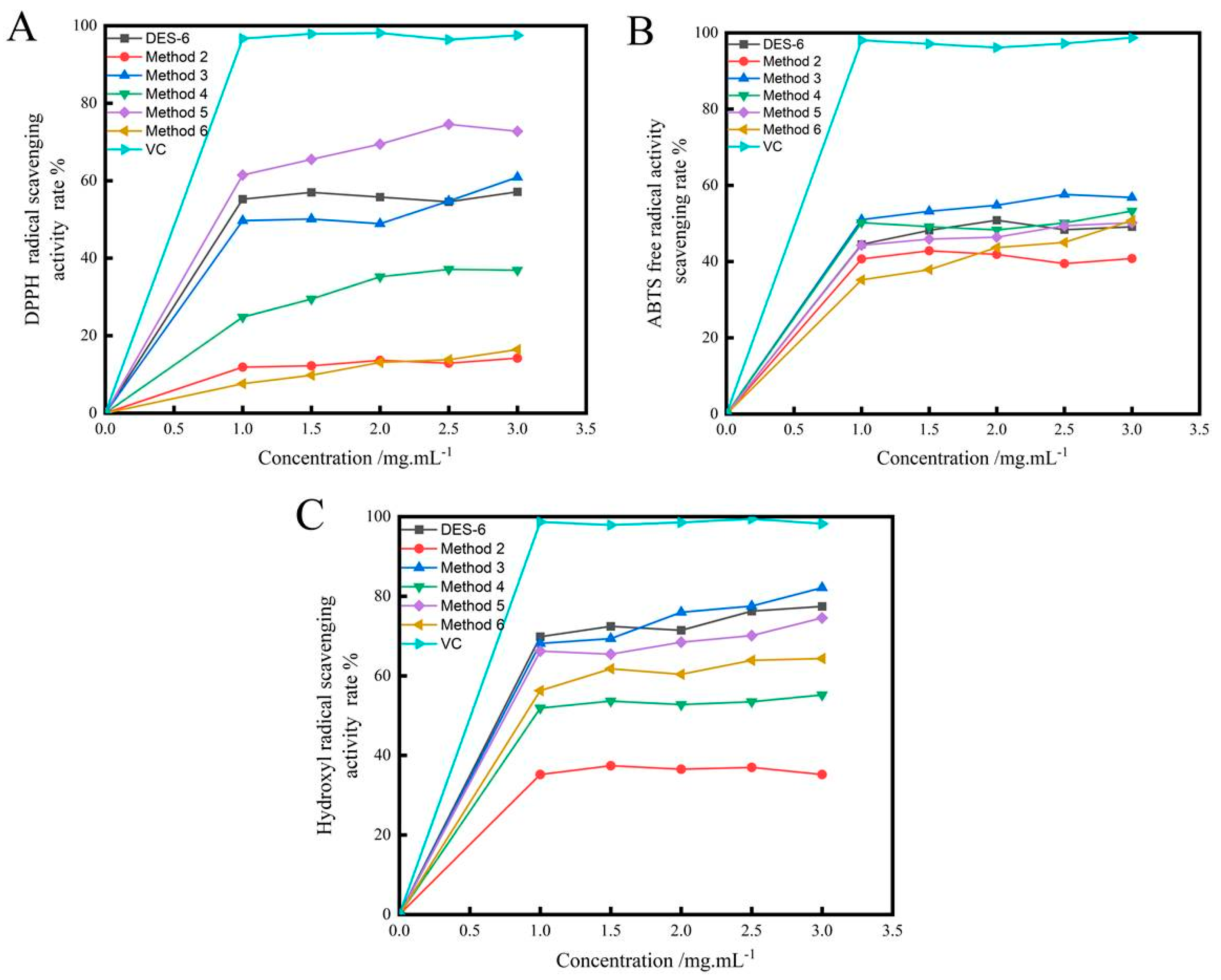
2.4.1. Scavenging of 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Radicals
2.4.2. Scavenging of 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS)radicals
2.4.3. Scavenging of Hydroxyl Radicals
2.5. Chemical Modification
2.5.1. Molecular Weight
2.5.2. Polysaccharide Molecular Conformation
2.6. Monosaccharaide Composition
2.7. Pharmacological Effects of Polysaccharides Antioxidant Networks
2.7.1. Venny Diagram Construction
2.7.2. Target Screening and Enrichment Analysis
2.7.3. Constructing the “Herb-Polysaccharide Fractions-Target-Pathway” Network
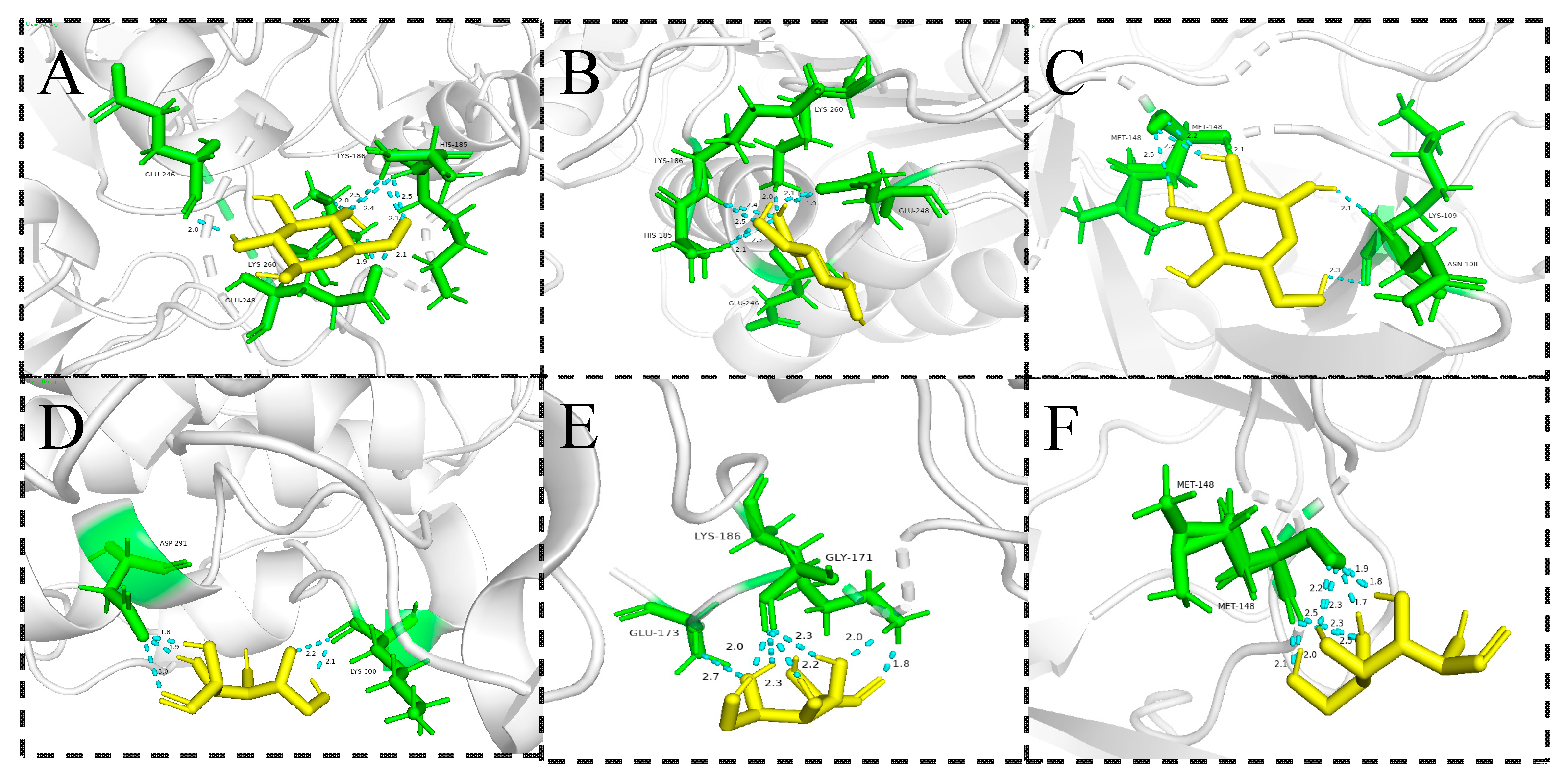
2.7.4. Component-Target Molecular Docking Analysis
3. Materials and Methods
3.1. Plant Materials and Chemicals
3.2. Experimental Methods
3.2.1. Preparation of DESs
3.2.2. Powder Pretreatment
3.2.3. Extraction of Radix Peucedani Polysaccharides
3.2.4. Plotting of Standard Curves for Polysaccharides Content
3.3. Process Optimization for Polysaccharides Extraction by Ultrasound-Assisted DESs
3.3.1. Single-Factor Test
3.3.2. Response Surface Optimization Test
3.4. Structural Analysis of Polysaccharides
3.4.1. Scanning Electron Microscopy (SEM) Analysis
3.4.2. Fourier Transform Infrared (FT-IR) Spectra
3.4.3. Evaluation of Antioxidant Activity
3.4.4. Monosaccharide Analysis
3.5. Network Pharmacology Analysis
3.5.1. Prediction and Intersection of Targets
3.5.2. Network Creation and Enrichment Analysis
3.5.3. Molecular Docking
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- National Pharmacopoeia Committee (NPC). 2020 Pharmacopoeia of the People’s Republic of China (a); Medical Science and Technology Press: Beijing, China, 2020.
- Wu, W.K.; Yan, Q.R.; Song, W. Current situation, problems and countermeasures of the quality of Chinese medicine qian hu.J/OL. J. Yunnan Univ. Natl. (Nat. Sci. Ed.) 2023, 32, 421–427. [Google Scholar]
- Li, B.; Lu, F.; Wei, X.; Zhao, R. Fucoidan: Structure and Bioactivity. Molecules 2008, 13, 1671. [Google Scholar] [CrossRef] [PubMed]
- Usui, T.; Asari, K.; Mizuno, T. Isolation of Highly Purified “Fucoidan” from Eisenia bicyclis and Its Anticoagulant and Antitumor Activities. Agric. Biol. Chem. 1980, 44, 1965–1966. [Google Scholar] [CrossRef]
- Song, J.Q.; Xu, Y.T.; Zhang, H.K. Immunomodulation action of sulfate polysaccharide of Laminaria japonica on peritoneal macrophages of mice. Chin. J. Immunol. 2000, 16, 70. [Google Scholar]
- Yoshinobu, A.; Yoshitaka, M.; Tomonori, N. Fucoidan induces apoptosis of human HS-sultan cells accompanied by activation of caspase-3 and down-regulation of ERK pathways. Am. J. Hematol. 2005, 78, 7–14. [Google Scholar]
- Liu, F.; Chen, H.J.; Qin, L.; Akram, A.N.M.A.; Xu, J.; Zhou, W.Z.; Zhu, S.N.; Wang, Z.M. Effect and characterization of polysaccharides extracted from Chlorella sp. by hot-water and alkali extraction methods. Algal. Res. 2023, 70, 102970. [Google Scholar] [CrossRef]
- Emanuela, M.; Franca, C.; Matteo, D.; Valerio, D.; Alessandro, T.; Olga, R. Choline chloride-water mixtures as new generation of green solvents: A comprehensive physico-chemical study. J. Mol. Liq. 2023, 383, 122120. [Google Scholar]
- Zhang, Y.L.; He, L.; Li, Q.; Cheng, J.W.; Wang, Y.B.; Zhao, J.C.; Yuan, S.F.; Chen, Y.J.; Shi, R. Optimization of ultrasonic-assisted deep eutectic solvent for the extraction of polysaccharides from Indocalamus tessellatus leaves and their biological studies. Sustain. Chem Pharm. 2022, 30, 100855. [Google Scholar] [CrossRef]
- Meng, Y.; Sui, X.Y.; Pan, X.; Zhang, X.Y.; Sui, H.M.; Xu, T.; Ge, P.L. Density-oriented deep eutectic solvent-based system for the selective separation of polysaccharides from Astragalus membranaceus var. Mongholicus under ultrasound-assisted conditions. Ultrason. Sonochemistry 2023, 98, 106522. [Google Scholar] [CrossRef]
- Bai, J.J.; Si, S.H.; Tao, L.; Zhang, N.; Shi, W.Y.; Zhu, Q.J. Synergy of a deep eutectic solvent and tea saponin on foam flooding EOR in a high salinity clay mineral rich heavy oil reservoir. J. Mol. Liq. 2023, 378, 121588. [Google Scholar]
- Zhao, H.; Baker, G.A. Ionic liquids and deep eutectic solvents for biodiesel synthesis: A review (Review). J. Chem. Technol. Biotechnol. 2013, 88, 3–12. [Google Scholar] [CrossRef]
- Wagle, D.V.; Zhao, H.; Baker, G.A.S. Deep Eutectic olvents: Sustainable Media for Nanoscale and Functional Materials. Acc. Chem. Res. 2014, 47, 2299–2308. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Xu, J.; Xie, J.X.; Zhu, S.Y.; Li, J.; Ying, G.D.; Chen, K.F. A low-energy and sustainable pulping technology for eucalyptus slabs using a deep eutectic solvent. Green Chem. 2023, 25, 3256–3266. [Google Scholar] [CrossRef]
- Wei, F.; Gao, G.Z.; Wang, X.F.; Dong, X.Y.; Li, P.P.; Hua, W.; Wang, X.; Wu, X.; Chen, H. Quantitative determination of oil content in small quantity of oilseed rape by ultrasound-assisted extraction combined with gas chromatography. Ultrason. Sonochemistry 2008, 15, 938–942. [Google Scholar] [CrossRef]
- Silva, D.T.D.; Pauletto, R.; Cavalheiro, S.D.S.; Emanuelli, T. Natural deep eutectic solvents as a biocompatible tool for the extraction of blueberry anthocyanins. J. Food Compos. Anal. 2020, 89, 103470. [Google Scholar] [CrossRef]
- Cui, H.; Li, Z.C.; Hong, J.D.; Wen, G.X.; Fu, M.X.; Yan, C. Extraction of Flavonoids from Scutellariae Radix using Ultrasound-Assisted Deep Eutectic Solvents and Evaluation of Their Anti-Inflammatory Activities. ACS Omega 2020, 5, 23140–23147. [Google Scholar]
- Zhou, L.L.; Luo, J.Q.; Xie, Q.T.; Huang, L.H.; Shen, D.; Li, G.Y. Dietary Fiber from Navel Orange Peel Prepared by Enzymatic and Ultrasound-Assisted Deep Eutectic Solvents: Physicochemical and Prebiotic Properties. Foods 2023, 12, 2007. [Google Scholar] [CrossRef]
- Zubera, N.; Muhammad, Z.; Muhammad, A.H.; Muhammad, S. Green extraction of ethnomedicinal compounds from Cymbopogon citratus Stapf using hydrogen-bonded supramolecular network. Sep. Sci. Technol. 2020, 56, 1520–1533. [Google Scholar]
- Xing, C.; Cui, W.Q.; Zhang, Y.; Zou, X.S.; Hao, J.Y.; Zheng, S.D.; Li, Y.H. Ultrasound-assisted deep eutectic solvents extraction of glabridin and isoliquiritigenin from Glycyrrhiza glabra: Optimization, extraction mechanism and in vitro bioactivities. Ultrason. Sonochemistry 2022, 83, 1350–4177. [Google Scholar] [CrossRef]
- Niu, M.; Zhang, S.Q.; Zhang, B. Methodological Guidelines for the Evaluation of Network Pharmacology. Chin. Herb. Med. 2021, 52, 4119–4129. [Google Scholar]
- Chen, J.; Chen, Q.L. Current status and reflection of network pharmacology in traditional Chinese medicine research. J. Shanghai Univ. Tradit. Chin. Med. 2021, 35, 1–6+13. [Google Scholar]
- Zhang, X.Y.; Zhang, Y.Y.; Liu, M.L. Application of virtual screening technology in traditional Chinese medicine research. Chin. J. New Drugs 2022, 31, 1676–1683. [Google Scholar]
- Yan, W.H.; Li, C.H.; Li, S.Y. A study to explore the potential mechanism of action of epileptin tablets in the treatment of epilepsy based on network pharmacology and molecular docking. Mod. Drugs Clin. 2023, 38, 1629–1637. [Google Scholar]
- Zhang, J.; Ye, Z.; Liu, G.; Liang, L.; Wen, C.; Liu, X.; Li, Y.; Ji, T.; Liu, D. Subcritical Water Enhanced with Deep Eutectic Solvent for Extracting Polysaccharides from Lentinus edodes and Their Antioxidant Activities. Molecules 2022, 27, 3612. [Google Scholar] [CrossRef]
- Pan, X.; Xu, L.J.; Meng, J.L.; Chang, M.C.; Cheng, Y.F.; Geng, X.R.; Liu, R.Z. Ultrasound-Assisted Deep Eutectic Solvents Extraction of Polysaccharides from Morchella importuna: Optimization, Physicochemical Properties, and Bioactivities. Front Nutr. 2022, 9, 912014. [Google Scholar] [CrossRef] [PubMed]
- Purama, R.K.; Goswami, P.; Khan, A.T.; Goyal, A. Structural analysis and properties of dextran produced by Leuconostoc mesenteroides NRRL B-640. Carbohydr. Polym. 2008, 76, 30–35. [Google Scholar] [CrossRef]
- Ma, Y.H.; He, H.Q.; Wu, J.Z.; Huang, Q. Assessment of Polysaccharides from Mycelia of genus Ganoderma by Mid-Infrared and Near-Infrared Spectroscopy. Sci. Rep. 2018, 8, 10. [Google Scholar] [CrossRef]
- Han, C.F.; Liu, S.T.; Yan, R.R.; Li, J.; Chen, N.; Zhang, L.L.; Han, P.P. Salicylic Acid and Jasmonic Acid Increase the Polysaccharide Production of Nostoc flagelliforme via the Regulation of the Intracellular NO Level. Foods 2023, 12, 915. [Google Scholar] [CrossRef]
- Wang, Y.J.; Xiong, X.; Huang, G.L. Ultrasound-assisted extraction and analysis of maidenhairtree polysaccharides. Ultrason. Sonochemistry 2023, 95, 106395. [Google Scholar] [CrossRef]
- Gieroba, B.; Kalisz, G.; Krysa, M.; Khalavka, M.; Przekora, A. Application of Vibrational Spectroscopic Techniques in the Study of the Natural Polysaccharides and Their Cross-Linking Process. Int. J. Mol. Sci. 2023, 24, 2630. [Google Scholar] [CrossRef]
- Chen, Q.; Zhang, W.T.; Huang, G.L. Preparation and Antioxidant Activity of Acetylated Mung Bean Peel Polysaccharides. Chem. Biodivers. 2023, 20, e202300175. [Google Scholar] [CrossRef]
- Hao, J.W.; Chen, N.D.; Fu, X.C.; Zhang, J. Predicting the contents of polysaccharides and its monosugars in Dendrobium huoshanense by partial least squares regression model using attenuated total reflectance Fourier transform infrared spectroscopy. Spectrosc. Lett. 2019, 52, 297–305. [Google Scholar] [CrossRef]
- AlWraikat, M.; Liu, Y.; Wu, L.M.; Ali, Z.S.; Li, J.K. Structural Characterization of Degraded Lycium barbarum L. Leaves’ Polysaccharide Using Ascorbic Acid and Hydrogen Peroxide. Polymers 2022, 14, 1404. [Google Scholar] [CrossRef]
- Mao, G.H.; Zou, Y.; Feng, W.W.; Wang, W.; Zhao, T.; Ye, C.W.; Wu, X.Y. Extraction, preliminary characterization and antioxidant activity of Se-enriched Maitake polysaccharide. Carbohydr. Polym. 2014, 101, 213–219. [Google Scholar] [CrossRef]
- Wang, Z.C.; Zhou, X.Y.; Shu, Z.H.; Zheng, Y.; Hu, X.L.; Zhang, P.Y.; Li, N. Regulation strategy, bioactivity, and physical property of plant and microbial polysaccharides based on molecular weight. Int. J. Biol. Macromol. 2023, 244, 125360. [Google Scholar] [CrossRef]
- Feng, Z.; Xu, X.J.; Zhang, L. Natural polysaccharides with different conformations: Extraction, structure and anti-tumor activity. J. Mater. Chem. B 2020, 8, 9652–9667. [Google Scholar]
- Zeng, H.L.; Zhang, Y.; Liu, J. Molar Mass Distribution and Chain Conformation of Polysaccharides from Fortunella margarita (Lour.) Swingle. Struct. Chem. 2014, 33, 1245–1252. [Google Scholar]
- Zeng, H.L.; Miao, S.; Zhang, Y.; Zheng, B.D. Isolation, preliminary structural characterization and hypolipidemic effect of polysaccharide fractions from Fortunella margarita (Lour.) Swingle. Food Hydrocoll. 2016, 52, 126–136. [Google Scholar] [CrossRef]
- Jung, M.K.; In, S.B. Gene Expression Profiles and Antioxidant Effects of Houttuynia cordata Thunb Extract in Human Keratinocyte HaCaT Cells. J. Life Sci. 2018, 28, 1406–1415. [Google Scholar]
- Wang, B.; Yan, S.; Yi, Y.; Huang, Y.; Deng, Z.; Zhang, Y.; Zheng, Q.; Xie, H. Purified Vitexin Compound 1 Inhibits UVA-Induced Cellular Senescence in Human Dermal Fibroblasts by Binding Mitogen-Activated Protein Kinase 1. Cell Dev. Biol. 2020, 8, 691. [Google Scholar] [CrossRef]
- Naghdi, S.; Rezaei, M.; Tabarsa, M.; Abdollahi, M. Ultrasonic-assisted enzymatic extraction of sulfated polysaccharide from Skipjack tuna by-products. Ultrason. Sonochemistry 2023, 95, 106385. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.C.; Chu, D.P.; Zhang, J.X.; Zheng, Y.F.; Li, Y.Q. Microwave-assisted extraction, partial purification and biological activity in vitro of polysaccharides from bladder-wrack (Fucus vesiculosus) by using deep eutectic solvents. Sep. Purif. Technol. 2021, 259, 118169. [Google Scholar] [CrossRef]
- Wu, D.T.; Feng, K.L.; Huang, L.G.; Ren, Y.H.; Yi, C.; Zou, L. Deep Eutectic Solvent-Assisted Extraction, Partially Structural Characterization, and Bioactivities of Acidic Polysaccharides from Lotus Leaves. Foods 2021, 10, 2330. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.Q.; Huang, R.M.; Wen, P.; Song, Y.; He, B.L.; Tan, J.L.; Wang, H. Structural characterization and immunological activity of pectin polysaccharide from kiwano (Cucumis metuliferus) peels. Carbohydr. Polym. 2021, 254, 117371. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z. Preparation of Manno-Oligosaccharides by Enzymatic Hydrolysis and Its Function; Huazhong Agricultural University: Wuhan, China, 2016. [Google Scholar]
- Li, G.F.; Chen, H.F.; Lang, Y.F. Extraction process optimization and in vitro biological activities of total flavonoids from Terminalia chebula. Chin. Tradit. Pat. Med. 2021, 43, 2945–2951. [Google Scholar]
- Wang, N.; Li, Q. Study on extraction and antioxidant activity of polysaccharidesfrom Radix Bupleuri by natural deep eutectic solvents combined with ultrasound-assisted enzymolysis. Sustain. Chem. Pharm. 2022, 30, 100877. [Google Scholar] [CrossRef]
- Corderi, S.; Renders, T.; Servaes, K.; Vanbroekhoven, K.; De Roo, T.; Elst, K. Strategies for the Removal of Polysaccharides from Biorefinery Lignins: Process Optimization and Techno Economic Evaluation. Molecules 2021, 26, 3324. [Google Scholar] [CrossRef] [PubMed]
- Periaswamy, S.S.; Yong, N.C.; Hee, C.W.; Byung, S.C. Green and efficient extraction of polysaccharides from brown seaweed by adding deep eutectic solvent in subcritical water hydrolysis. J. Clean Prod. 2018, 198, 1474–1484. [Google Scholar]
- Tian, Q.; Hu, P.; Wang, M.H.; Hu, Y.; Xie, L.K.; Li, Z.X.; Xu, Z. Study on the mechanism of Gao-Liang-Huo decoction against dextran sulfate sodium-induced ulcerative colitis based on metabolomics and network pharmacology. Pharmacol. Res. 2023, 9, 100304. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, H.; Li, N.; Chen, J.M.; Xu, H.; Wang, Y.J.; Liang, Q.Q. Network pharmacology, a promising approach to reveal the pharmacology mechanism of Chinese medicine formula. J. Ethnopharmacol. 2023, 309, 116306. [Google Scholar] [CrossRef]





| Source | Sum of Squares | df | Mean Square | F Value | p-Value | Significant |
|---|---|---|---|---|---|---|
| Model | 34.45 | 9 | 3.83 | 43.31 | <0.0001 | significant |
| A | 3.25 | 1 | 3.25 | 36.81 | 0.0005 | ** |
| B | 6.6 | 1 | 6.6 | 74.67 | <0.0001 | ** |
| C | 1.2 | 1 | 1.2 | 13.62 | 0.0078 | * |
| AB | 2.14 | 1 | 2.14 | 24.19 | 0.0017 | * |
| AC | 2.38 | 1 | 2.38 | 26.97 | 0.0013 | * |
| BC | 2.78 | 1 | 2.78 | 31.43 | 0.0008 | ** |
| A2 | 8.53 | 1 | 8.53 | 96.54 | <0.0001 | ** |
| B2 | 2.65 | 1 | 2.65 | 30.01 | 0.0009 | ** |
| C2 | 3.36 | 1 | 3.36 | 38.02 | 0.0005 | ** |
| Residual | 0.6187 | 7 | 0.0884 | |||
| Lack of Fit | 0.4426 | 3 | 0.1475 | 3.35 | 0.1365 | not significant |
| Pure Error | 0.176 | 4 | 0.044 | |||
| Cor Total | 35.07 | 16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Wang, C.; Li, Q.; Liang, W. Polysaccharides from Radix Peucedani: Extraction, Structural Characterization and Antioxidant Activity. Molecules 2023, 28, 7845. https://doi.org/10.3390/molecules28237845
Zhang J, Wang C, Li Q, Liang W. Polysaccharides from Radix Peucedani: Extraction, Structural Characterization and Antioxidant Activity. Molecules. 2023; 28(23):7845. https://doi.org/10.3390/molecules28237845
Chicago/Turabian StyleZhang, Jie, Chenyue Wang, Qian Li, and Wei Liang. 2023. "Polysaccharides from Radix Peucedani: Extraction, Structural Characterization and Antioxidant Activity" Molecules 28, no. 23: 7845. https://doi.org/10.3390/molecules28237845
APA StyleZhang, J., Wang, C., Li, Q., & Liang, W. (2023). Polysaccharides from Radix Peucedani: Extraction, Structural Characterization and Antioxidant Activity. Molecules, 28(23), 7845. https://doi.org/10.3390/molecules28237845






