1. Introduction
A growing number of researchers and industrialists now believe that perovskite solar cells (PSCs) are on the verge of commercialization as the power conversion efficiency (PCE) gears up to an excellent level, i.e., 26.1% [
1]. It is an unusual outlook at a time when other thin-films emerging photovoltaics are expensive or not comparatively efficient, and the PCE in conventional silicon solar cells is saturated at a specific value. Although the commercialization of PSCs seems interesting to lower the energy conversion cost, long-term stability, and high PCE via industrially available fabrication techniques, it is a sine qua non. In this virtue, depositing perovskite via an anti-solvent approach is the main barrier to scalable perovskite layers [
2]. Therefore, a two-step method, where two different precursors are prepared, i.e., for the BX
2 (generally PbI
2) and AX (organic salts) layers, are deposited sequentially to engineer the perovskite (ABX
3) [
3]. Nevertheless, the mechanism of perovskite formation in the two-step method is more complicated than the one-step method, owing to the interface formation between the PbI
2 and organic salts [
4]. More specifically, the physicochemical characteristics of both the lead and organic halides govern the perovskite formation [
5]. Organic salt’s evaporation and incomplete penetration in the PbI
2 bulk leave behind the unreacted PbI
2, which generally remains as a residue in the final perovskite layer [
6]. Although the moderate amount of PbI
2 within the grain boundaries is beneficial, owing to its passivation effect [
7,
8,
9], its excessive amount suppresses the carrier transportation near the perovskite interfaces, especially under illumination [
10,
11]. Therefore, controlling the amount of excess PbI
2, especially at the perovskite interfaces, can boost the PSCs’ performance through suppressing the nonradiative recombination [
12,
13].
Several additives have been applied to the PbI
2 layer, which enhanced the PSCs’ performances by passivating the Pb
2+ defects. For example, Boopathi et al. introduced alkali metal halides in the PbI
2 layer and improved the PbI
2 film’s compactness by passivating the Pb
2+ [
14]. In 2015, Zhang et al. incorporated an additive (4-tert-butylpyridine, TBP) to create a porous PbI
2 to enhance the organic salt penetration in the PbI
2 layer [
15]. Zhao et al. used carbonyl group-based monomers as an additive in the PbI
2 precursor, improving the interactions between the carbonyl groups and the Pb
2+ [
16]. Furthermore, Wang et al. partially substituted Pb
2+ with neodymium cations (Nd
3+) by doping PbI
2 with Nd and achieved a PCE over 21% [
17]. In more than 90% of cases, the additives are introduced in the PbI
2 precursor during the two-step perovskite method, and in each of these cases, the additives provide Lewis acid passivation to the Pb
2+ defects. Nonetheless, the reason behind the additive importation in the PbI
2 part, rather than salt precursors, was ill-defined until Liang et al. examined the Pb
0 defects in detail and concluded that the Pb
0 defects are induced by the PbI
2 layer under illumination. They further proved that the Pb
0 impurities are decomposition byproducts of residual PbI
2 [
18]. Very recently, Yang et al. also found that the Pb
2+ and Pb
0 defects are abundantly present at the residual PbI
2 sites [
19].
Herein, the Pb
2+ passivation effect at the interface of the SnO
2 and PbI
2 layer is studied, where the chances of PbI
2 residue are comparatively high [
20]. As on the amines’ deposition at PbI
2, the perovskite conversion stops right after the resultant wall formation in the form of the perovskite layer, further inhibiting the penetration of organic cations. Comparatively, this perovskite barrier formation is more critical in FA-riched organic cations where the PbI
2 with FAI reaction is fast [
21,
22,
23,
24]. Consequently, a large amount of PbI
2 residue is generated in the FA-based perovskite layers beneath the perovskite. This work uses FAI-rich amine precursors, and the SnO
2 substrate is decorated with a functional biological molecule, artemisinin (ART). On the ART interaction with the resultant perovskite, the interface defects, especially the Pb
2+ defects, are strongly hindered. Furthermore, the amount of PbI
2 residue is decreased in the final perovskite film. As a result, PCE is enhanced from 20.21% to 21.72%, mainly due to the improvement in the FF and V
OC of the device. Commercially available ART has a unique molecular structure composed of sesquiterpene lactones surrounded by alkyls [
25], which makes it hydrophobic, and its ketone group provides the Lewis base interaction and passivates the Pb–I antisite defects [
26]. Previously, it was used as an additive of perovskite a couple of times, where it successfully reduced the Pb
2+ defects and improved the PSCs’ performances [
27,
28]. Nevertheless, to the best of our knowledge, we are studying it as a passivation layer at the defective interface of the SnO
2/perovskite to control the PbI
2 residue and Pb
2+ defects for the first time.
2. Results and Discussions
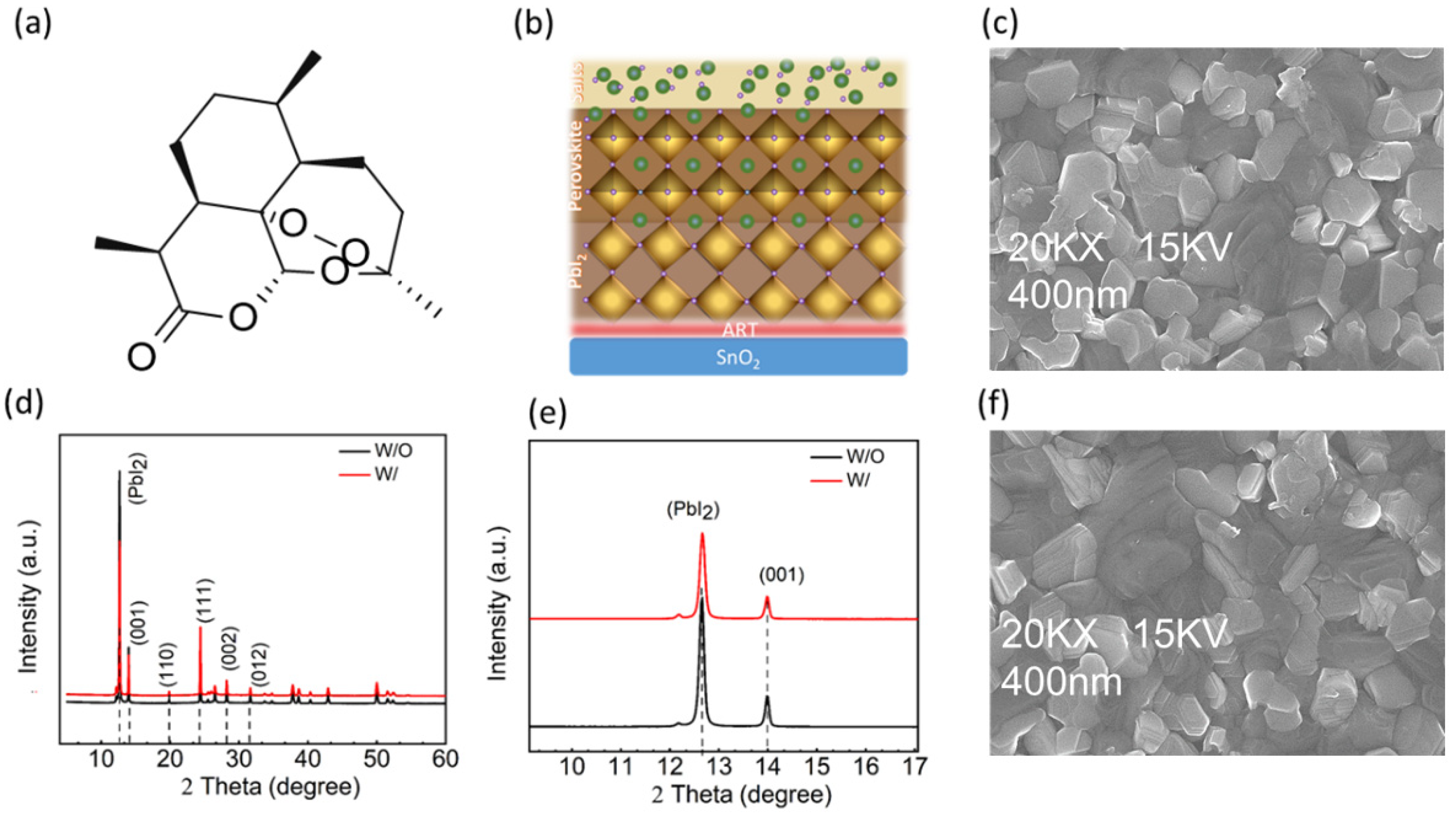
Molecular structures of small molecules or polymers have their specific effect on the performances of PSCs when used as additives or in the contacts of the perovskite layer [
29,
30,
31]. However, tedious experimental synthesis and reproducibility remained a question mark for these organic materials. Therefore, commercially available and cheaper organic materials can pave the commercialization. ART is a cost-effective organic biological molecule with a ketone group (C=O) (
Figure 1a) that enhances the Pb
2+ interaction and passivates the Pb-I antisite defects [
32]. ART is spin-casted on the SnO
2 layer, which sandwiches it as a SnO
2/ART/perovskite, as shown in
Figure 1b, where it has maximum interaction with the perovskite region and, moreover, this interface has a high concentration of PbI
2 residue. In the two-step fabricated perovskite layers, the Pb
2+ defects increase due to the PbI
2 residue [
18,
19]. Therefore, there are more chances that the unreacted PbI
2 is clustered at the SnO
2/perovskite interface as the perovskite conversion process starts from the top and then penetrates into the PbI
2 bulk under fast conversion conditions [
5,
33]. This quick process creates the perovskite barrier between the salts and the PbI
2, which is presented schematically in
Figure 1b. The unreacted salt can be managed via evaporation, leaving behind the unreacted PbI
2. Although, a moderate amount of PbI
2 residue is advantageous for the perovskite, if it lies within the grain boundaries. At the interfaces, PbI
2 has adverse effects in enhancing the nonradiative recombination and thus retards the performance of the devices.
X-ray diffraction (XRD) without (W/O) ART and with (W/) ART-deposited perovskite is conducted to study the effect of ART on the PbI2 residue and the perovskite defects’ passivation. Improvement in the crystallization has been noticed, and the peak of PbI
2 is substantially reduced (
Figure 1d). Specifically, the Bragg peak at the 2theta = 12.6° position, which represents the PbI
2, is suppressed under the effect of ART (
Figure 1e). At the X-axis, the diffraction peaks of the W/ and W/O perovskite samples are approximately the same, indicating that ART does not affect the perovskite’s crystal lattice (
Figure 1e,
Supporting Figure S1a,b). Furthermore, the perovskite crystallinity is enhanced in the perovskite deposited on ART, which can be seen from the sharpness of the W/ peaks (
Figure 1d). For obtaining information on the surface morphology of the perovskite film, a scanning electron microscope (SEM) and atomic force microscopy (AFM) characterization tests are performed (
Supporting Information, Figure S2a,b).
Figure 2c,f shows the top-view SEM images of the perovskite thin films, which show only a slight improvement in the film smoothness, coverage, and grain sizes. Modification of the perovskite layer via ART led to a significant reduction in the perovskite film’s roughness, as observed in the AFM. Root mean square (RMS) analysis revealed that the perovskite deposition on ART reduced the roughness of the perovskite film from 42.67 nm to 35.61 nm.
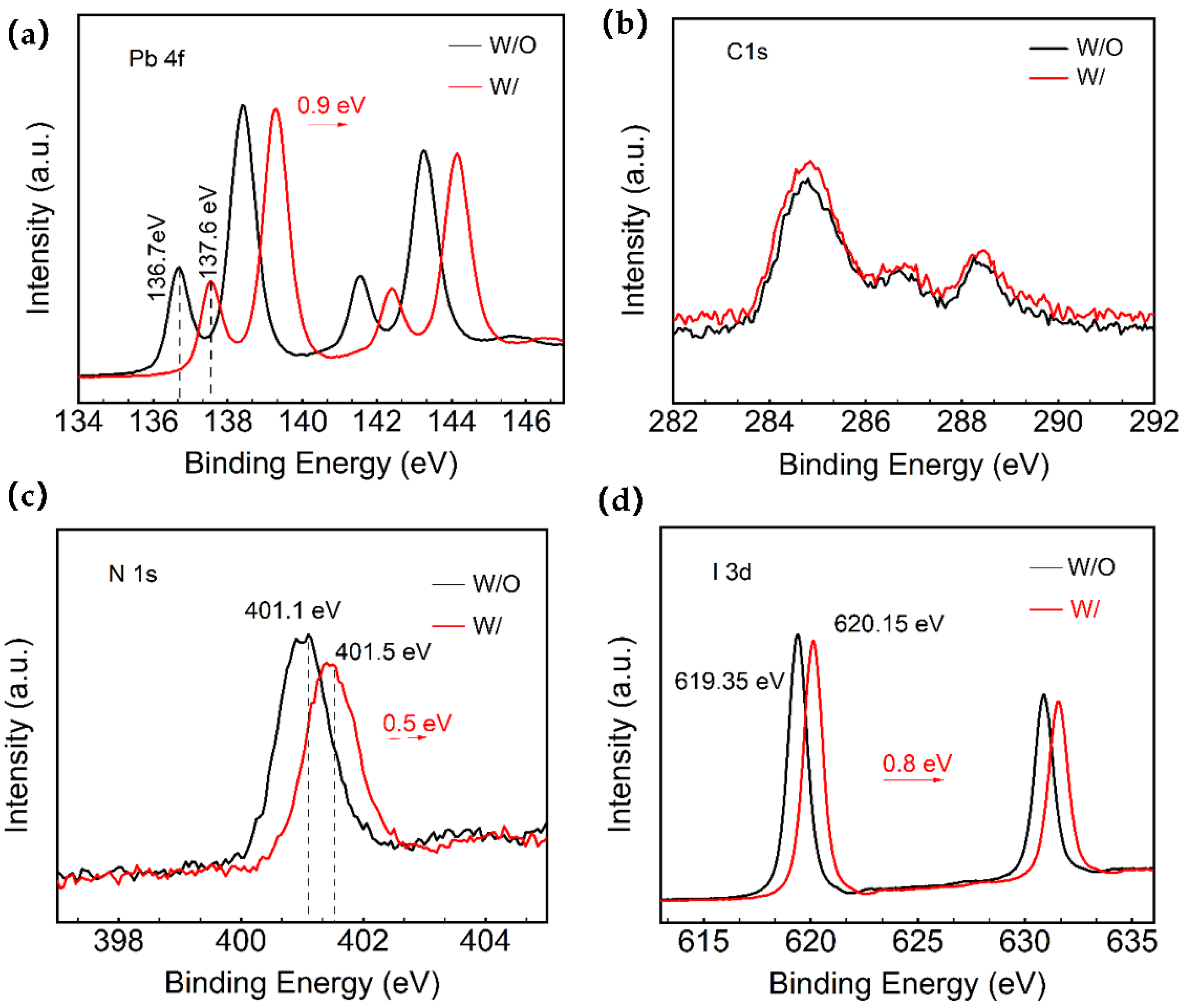
Moreover, the Pb
2+ defects in the W and W/O ART perovskite samples are analyzed via X-ray photoemission spectroscopy (XPS).
Figure S3 shows the full XPS spectrum. From the XPS spectra of Pb4f in
Figure 2a, it can be noticed that the two prominent peaks of Pb4f (4f7/2: 138.45 eV/139.15 eV and 4f5/2: 143.25 eV/144.15 eV) are seen in both the W and W/O ART perovskite samples. In particular, the small peaks of Pb
2+ corresponding to the saturated coordination and low binding energy (4f7/2: 136.7 eV/137.6 eV and 4f5/2: 141.6 eV/142.5 eV) were attributed to the uncoordinated metallic Pb (Pb
0). These findings suggest that the use of ART at the perovskite interface could impact the chemical composition of the perovskite films. The Pb 4f spectrum of both the control group and the ART/perovskite films showed the presence of metallic lead, indicating the reduction in Pb
2+ to Pb
0. The Pb 4f7/2 and Pb 4f5/2 binding energies of the experimental group were significantly shifted towards a higher binding energy by 0.9 eV compared to the control group. These results indicate the effective passivation of the perovskite defects around the unreacted PbI
2 sites, as the small peak of low binding energy decreased in intensity and shifted by 0.9 eV towards a higher binding energy. Additionally, the introduction of the ART layer effectively passivates the Pb-related surface defects and stabilizes the surface dangling bonds, as supported by the attenuation of the Pb
0 peak in the sulfur compounds [
34,
35].
Moreover, the obtained spectra exhibited characteristic perovskite crystal peaks such as C1s, N1s, O1s, Pb4f, and I3d (
Figure S3). Fine scan spectra of C1s, N1s, O1s, Pb4f, and I3d were obtained under different X-ray irradiation conditions before the measurements. The detailed Pb4f, C1s, N1s, and I3d spectra of the films are depicted in
Figure 2a–d. The results indicated a slight shift in the peak positions towards a higher binding energy in the films deposited on ART. The split-peak fits of I3d, Pb4f, and C1s for the control and the ART-based films can be found in
Supporting information Figure S4, with the corresponding elemental contents in
Table S1. The effects of this study on the Pb defects via XPS analysis is supported by the previous finding of Tsutomu Miyasaka’s team [
28]. They demonstrated that the strong interaction between the ART molecules and the uncoordinated Pb
2+ ions inhibits the formation of new Pb clusters with deep capture defects during aging, as indicated by the theoretical and experimental calculations. These findings highlight the potential of ART as a passivation layer for improving the stability and performance of devices.
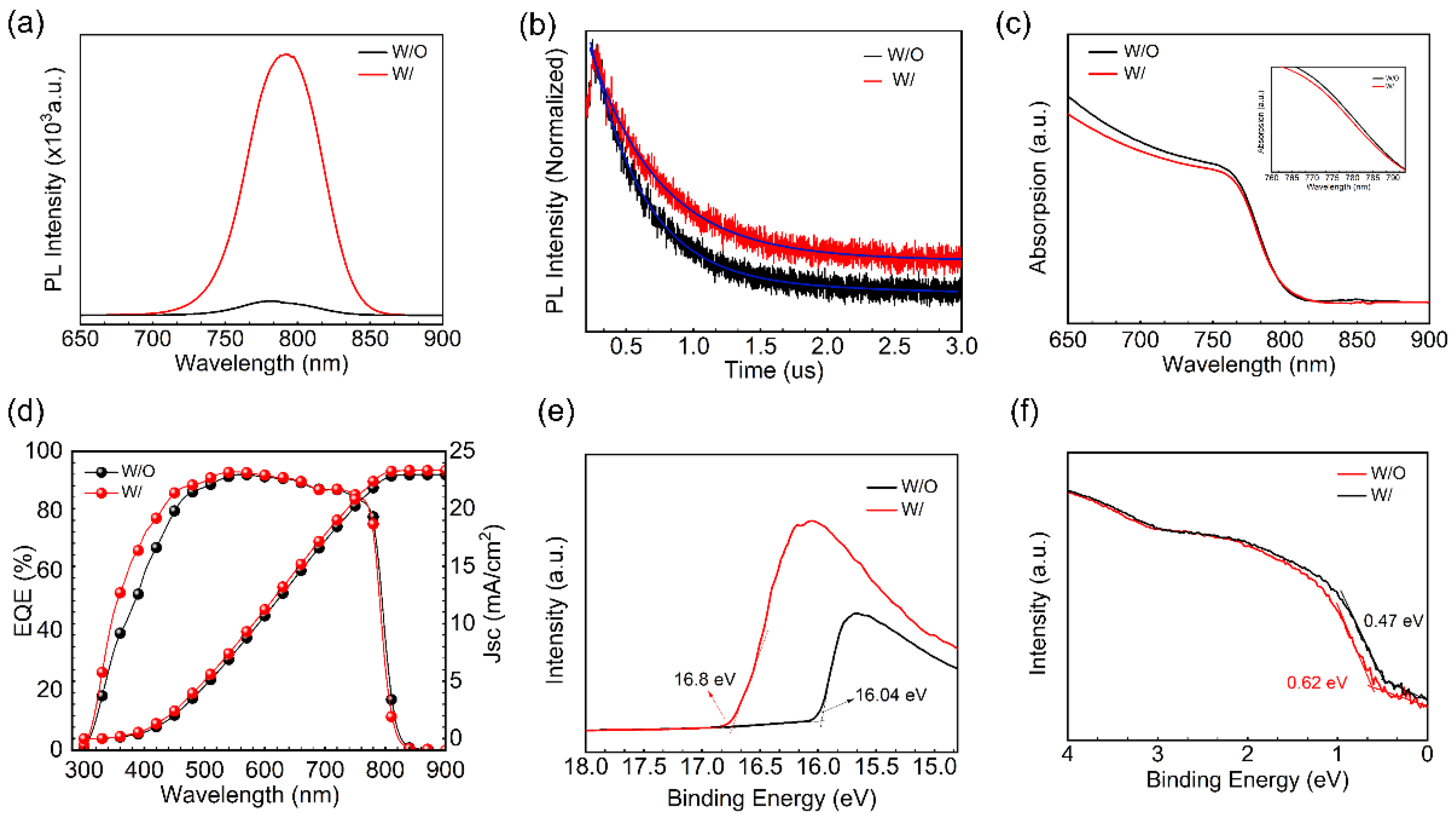
Time-resolved photoluminescence (TRPL) measurements are performed to investigate the optical and carrier properties of the perovskite films for both samples (W/ and W/O ART). The results indicate that the PL intensity in
Figure 3a, the ART/perovskite film, is significantly enhanced, and a red-shift phenomenon was noticed. The ART/perovskite films exhibited an increase in the carrier lifetime, as shown in
Figure 3b (fitting parameters are included in
Table S2), suggesting that the inclusion of the ART layer effectively improved the quality of the perovskite films by reducing the PbI
2 residue and the Pb
2+ defects, which alternatively decreased the non-radiative recombination. The extension of the relaxation time resulting from the ART/perovskites indicates that the duration of the band-to-band recombination emission was prolonged, leading to the conclusion that the ART passivation layer decreased the density of the shallow traps. We utilized TRPL spectrum fitting, which revealed a remarkable improvement in the lifespan of the ART/perovskite film. Specifically, the ART/perovskite film exhibited a lifespan of 509.93 ns, which is significantly longer than that of the control sample (394.92 ns). These promising results can be attributed to the reduction in PbI
2 at the perovskite interface. Additionally, reductions in the non-radiative recombination further resulted in improved device stability. Notably, the modified perovskite film exhibited an enhanced long-term stability of the conversion efficiency. Overall, these findings suggest that the reduction in the PbI
2 residue at the interfaces has the potential to improve the performance of PSCs. The results of this study provide valuable insights into the mechanisms underlying the enhanced performance of perovskite solar cells with the ART passivation and PbI
2 reduction. We compared the absorption changes of the samples with and without an ART layer using UV-vis measurements. We found that the absorption edge of the perovskite film shifted slightly blue after the treatment with the artemisinin passivation layer. The same phenomenon was observed in the EQE measurements. This indicates that the perovskite defects were passivated and the PbI
2 residue is suppressed, thereby reducing the non-radiative recombination. In
Figure 4d, the integrated current densities of 22.96 mA/cm
2 and 23.33 mA/cm
2 for the external quantum efficiency (EQE) spectra of the W/O ART layer and ART passivation layer devices, respectively, are in good agreement with the J-V measurements.
The ART layer greatly affects the optical and electronic properties of the perovskite films. Ultraviolet photoelectron spectroscopy (UPS) and the photoluminescence (PL) absorption peaks were used to analyze these properties before and after the ART application (
Figure 3e,f). It is evident that the defect passivation of perovskite promotes a charge extraction and alters the energy levels accordingly [
36]. In this virtue, the ART layer’s influence on the energy scape of perovskite is studied via UPS. The results indicate that the ART layer slightly decreases the secondary electron cutoff region (SECO) (
Figure 3e,f), and there is a change in the valence band maximum (VBM) of the perovskite films (
Figure S5). Specifically, the work function (WF) values after modification were 4.42 eV for the control and 5.18 eV for the ART/perovskite. Moreover, the VBM values for the ART modified and control perovskite films were found to be 0.62 eV and 0.47 eV below the Fermi level (E
F), respectively. These changes in the energy levels of the perovskite films indicate a preferred arrangement of energy levels that may benefit the charge transfer and non-radiative composite suppression. Therefore, compared with the perovskite treated without the ART layer, the energy level of the perovskite treated with the ART passivation layer is more matched, which favors carrier extraction and increases the efficiency of the device.
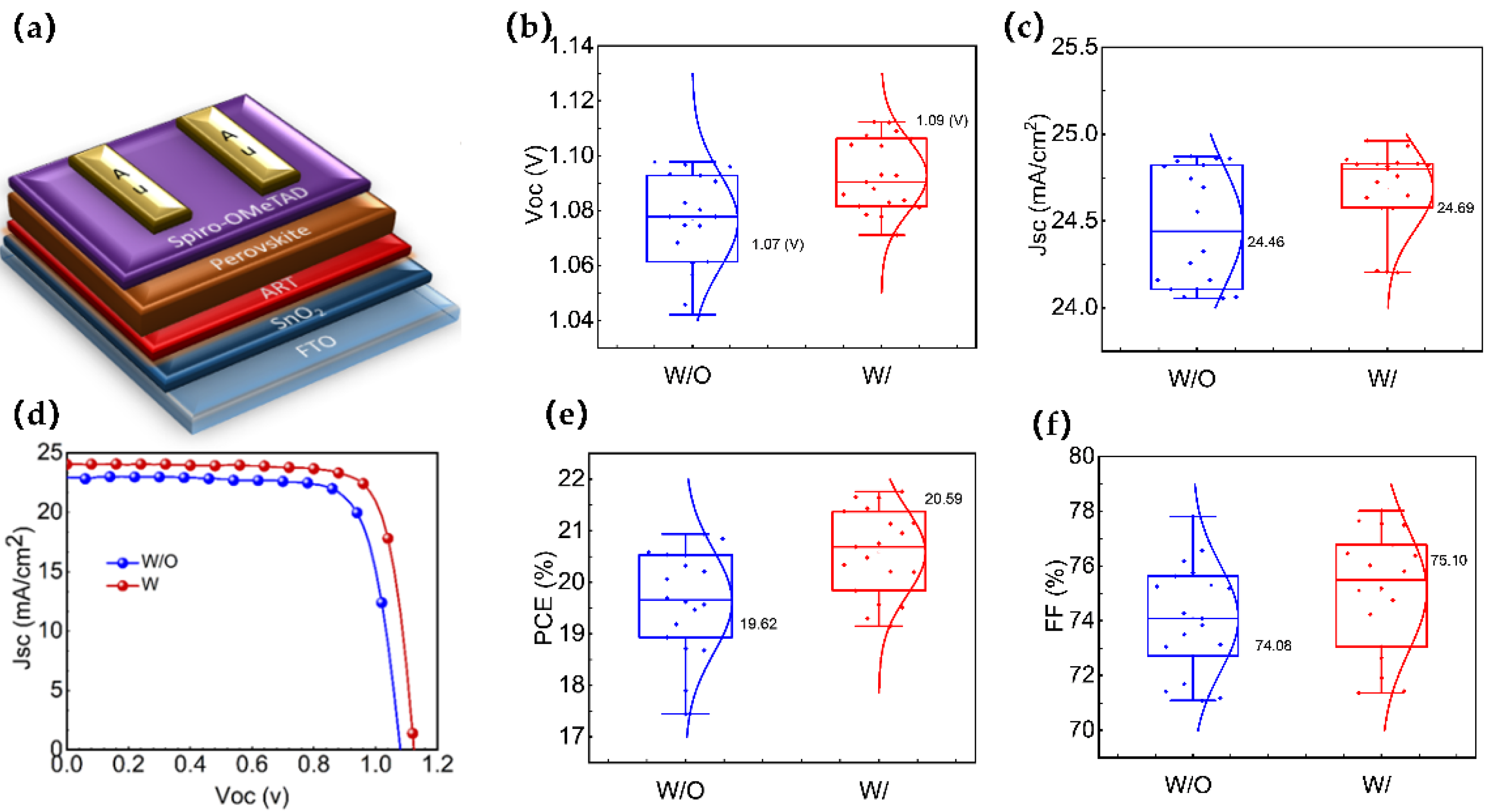
The final device structure is schematically presented in
Figure 4a, and the JV curves of the best devices for both W/ and W/O are shown in
Figure 4b. The maximum efficiency of the perovskite solar cells increased from 20.21% for the devices without the ART layer, with a Jsc of 24.86 mA/cm
2, Voc of 1.07 V, and an FF of 0.76, to 21.72% for the devices with the ART layer, with a Jsc of 24.69 mA/cm
2, Voc of 1.11 V, and an FF of 79.52 (Champion devices’ photovoltaic parameters for both types are included in
Table S3). We investigated the performance reproducibility of the ART-based PSCs by assessing the photovoltaic parameters of 30 devices for each type (W and W/O ART). The photovoltaic parameters (Jsc, Voc, FF, and PCE) are evaluated, as shown in
Figure 4b,c,e,f. Remarkably, all of the photovoltaic parameters improved after the inclusion of the ART layer. The calculated average Voc, Jsc, FF, and PCE values in the W/O ART devices were 1.07 V, 24.46 mA/cm
2, 74.08, and 19.62%, respectively. On the contrary, in the W/ ART devices, the average values of Voc, Jsc, FF, and PCE were 1.09 V, 24.69 mA/cm
2, 75.10, and 20.51%, respectively. In addition, the hysteresis index (HI) was determined to quantify the efficiency mismatch using the equation: HI = (PCE
reverse − PCE
forward)/PCE
reverse. The HI decreased from 0.64% for the devices without the ART layer to 0.41% for the devices with the ART layer interface modification (
Table S3), indicating a reduction in the defects and hysteresis effect. These results demonstrate that the ART passivation layer significantly improves the performance and stability of perovskite solar cells.
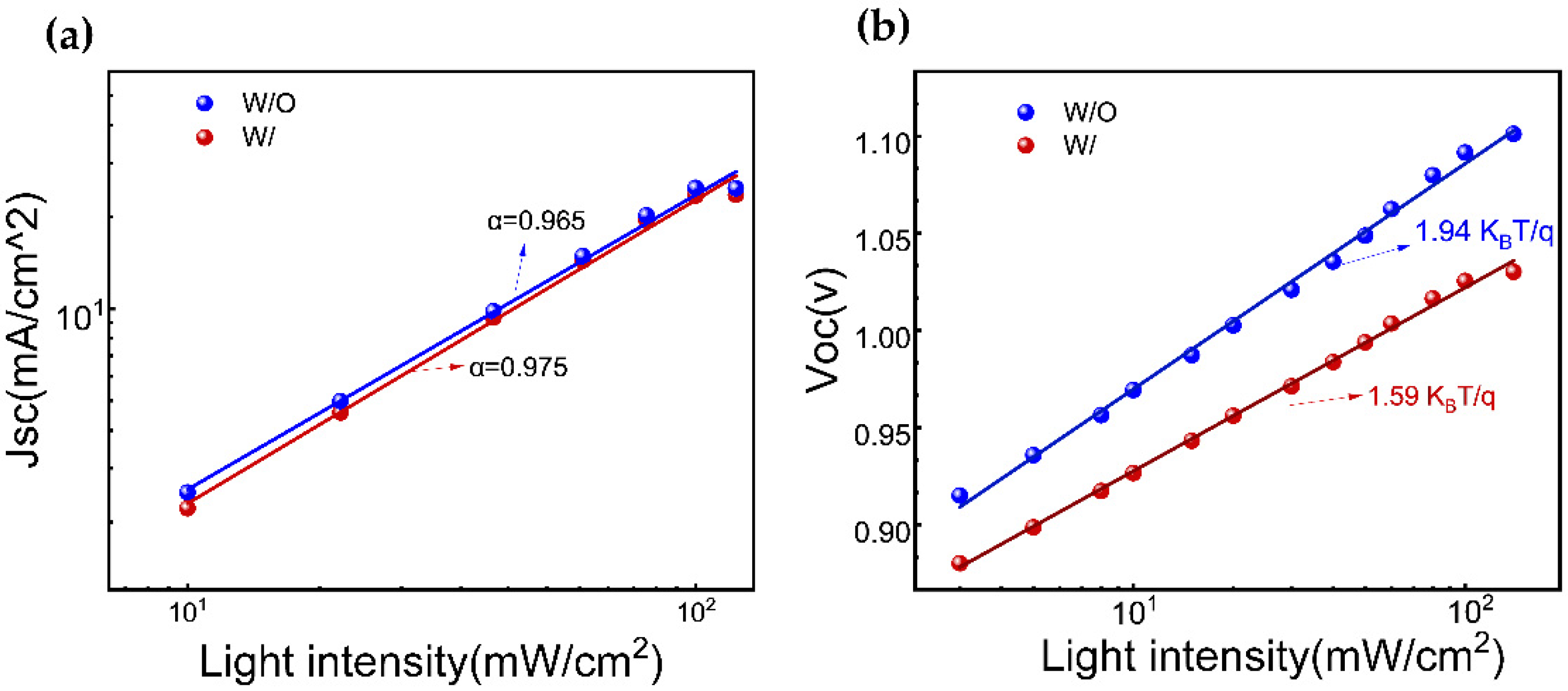
Furthermore, the charge recombination dynamics in a device are investigated by analyzing the functional relationships of short-circuit current density (Jsc) (
Figure 5a) andthe open-circuit voltage (Voc) (
Figure 5b)with incident light intensities. The ideal factor (α) of the device, calculated as (α ≤ 1), is found to be 0.965 and 0.975 for W/O and W/ ART, respectively, indicating that the bimolecular recombination approaches the minimum value, especially in ART-based devices. On the other hand, the ideal factor (ε) under the open-circuit conditions, with a range of 1 < ε < 2, reflects the extent of the monomolecular recombination. A value of ε closer to 1 indicates lesser monomolecular recombination. In contrast, a value more relative to 2 suggests more traps in the device, leading to increased monomolecular recombination, which is unfavorable for the charge transmission and collection. The experimental results reveal that the ε values of the devices are all greater than 1, indicating the existence of monomolecular recombination. As expected, on the ART incorporation, ε value is reduced from 1.94 K
BT/q to 1.59 K
BT/q. These findings are consistent with the results obtained from the TRPL and UV-visible absorption spectroscopy measurements. Consequently, these results are the response of the passivation treatment, and especially, the PbI
2 residue reduction via ART.
3. Materials and Methods
3.1. Materials
The SnO2 colloidal dispersions, featuring a particle size of 3–5 nm and diluted with 15% water, were obtained from Alfa Aesar. Meanwhile, flourine–doped tin oxide (FTO) with a Rsheet of 15 Ω/sq, thickness of 600 nm, and a light transmittance of >85% was procured from Beijing Huamin New Material Technology Co., Ltd., Beijing, China. Lead iodide (PbI2), MAI, FAI, MACl, MABr, and Spiro-OMeTAD were sourced from Xi’an Shuoyuan Optoelectronic Technology Co., Ltd., Xian, China, and supporting reagents such as K2SO4, triethylsilane, N, N-dimethylformamide (DMF), dimethyl sulfoxide (DMSO), chlorobenzene (CB), 4-tert-butylpyridine (TBP), anhydrous isopropanol (IPA), and lithium bisfluoromethane sulfonamide (Li-TFSI) were purchased from Sigma Aldrich Co., Ltd., St. Louis, MO, USA. Artemisinin was also procured from Sigma.
3.2. Device Fabrication
Substrate Preparation: The FTO conductive glass substrate was sequentially cleaned with a glass cleaner, ultrapure water, isopropanol, and ethanol for 15 min each, and then dried with a nitrogen gun for use. A two-step process was used to prepare the n-i-p perovskite solar cells with the FTO/SnO2&18C6&K/artemisinin passivation/perovskite/Spiro-OMeTAD/Au structure. The prepared FTO substrate was pretreated with UV-ozone for 15 min to ensure surface infiltration and establish good contact with SnO2.
In the context of the electron layer preparation, a precursor solution composed of SnO2 with a colloidal volume ratio of 15% and ultrapure water containing 0.2 mg of 18C6 and 2 mg of K2SO4 was combined at a ratio of 1:5. Following a 15 min sonication period, 100 uL of the electron layer precursor solution was subjected to a spin-coating process at 4000 rpm for 30 s, depositing a layer on the previously ozone-treated FTO substrate. The resultant film was annealed at 150 °C for 15 min, resulting in a dense SnO2 electron layer that was allowed to cool to room temperature before being exposed to an additional 15 min ozone pretreatment. The film was rapidly transferred to a glove box for subsequent spin-coating. This process was regarded as a vital component of the electron layer preparation for the intended perovskite solar cell structure.
The passivation layer was prepared by dissolving 1 mg of artemisinin in 1 mL of DMSO and subsequently deposited on the previously UV-treated SnO
2 electron layer film using a spin-coating process at 5000 rpm for 30 s. The concentration of ART in DMSO is optimized after trying different ratios in DMSO (
Supporting Information, Table S4). This preparation process is deemed critical for the successful fabrication of the intended perovskite solar cell structure.
A two-step method was utilized for the preparation of the perovskite layer films. The precursor solution (1)containing 1.5 M of PbI2, was dissolved in a mixture of DMF and DMSO (with a volume ratio of 9:1) and stirred at 60 °C for 4 h. The ammonium salt precursor solution (2) was composed of FAI, MABr, and MACl at a ratio of 90 mg:9 mg:9 mg and dissolved in 1 mL of anhydrous isopropanol. To deposit the perovskite layer, 60 μL of the prepared precursor solution (1) was spin-coated at 1500 rpm for 30 s, followed by annealing at 70 °C for 1 min, cooling to room temperature, and the subsequent deposition of 120 μL of the precursor solution (2) at 2000 rpm for 30 s with a quick drop at 5 s. The perovskite film was transferred to a drying air oven at 20–30% humidity for 15 min, annealed at 150 °C, and then swiftly moved to a nitrogen glove box. It was allowed to cool to room temperature and was then ready for the next step.
The hole precursor solution was formulated by dissolving 72.3 mg of Spiro-OMeTAD doped with 17.5 μL Li-(TFSI) in a 1 mL acetonitrile solution at a concentration of 520 mg, and 28.8 μL of 4-tert-butyl pyrimidine was added to the solution. The resulting 60 μL hole precursor solution was spin-coated and deposited onto the perovskite films at a rate of 4000 rpm for 30 s. These findings provide useful insights into the design and optimization of perovskite solar cells.
3.3. Photovoltaic Measurement
The manuscript reports the comprehensive characterization of a perovskite solar cell device. The current density–voltage (I-V) curve of the device was measured using a Keithley B2901A digital source (Keithley Instruments, Inc., Cleveland, OH, USA) meter under AM1.5G solar simulator irradiation, and the incident light intensity was calibrated with a quasi-silicon cell. The I-V curve was recorded with a scanning step of 0.02 V, a delay of 10 ms, and an effective area of 0.06 cm2. Time-resolved photoluminescence (Edinburgh Instruments, Bain Square Kirkton Campus Livingston EH54 7DQ, Britain, UK) spectra were acquired using a transient steady-state fluorescence lifetime test (FluoTime 300, Picoquant, Berlin, Germany). The surface roughness of the perovskite films was determined via atomic force microscopy (AFM) (Oxford Instruments Asylum Research, Inc., Santa Barbara, CA, USA). The morphology of the film surface and sections was analyzed using field emission scanning electron microscopy (SEM) (GeminiSEM 300 Carl Zeiss Microscopy Ltd., Britain, UK). Fourier transform infrared spectra were obtained using a Nicolet iS50, Thermo Fisher Scientific, Co. Ltd., Brno-Černovice, Czech Republic. X-ray diffraction (XRD) was performed using a SmartLab XRD, Rigaku Corporation of Japan, Tokyo, Japan. XPS Escalab Xi+, Thermo Fisher Scientific Co. Ltd., Brno-Černovice, Czech Republic. The TR-PL spectra had a transient lifetime of 30 ms to 20 ps and a wavelength range of 230 nm to 1700 nm. These results provide valuable insights into the structural and optical properties of the perovskite solar cell, which could help in the development of high-performance devices.