Applications of Capillary Electrophoresis for the Determination of Cannabinoids in Different Matrices
Abstract
1. Introduction
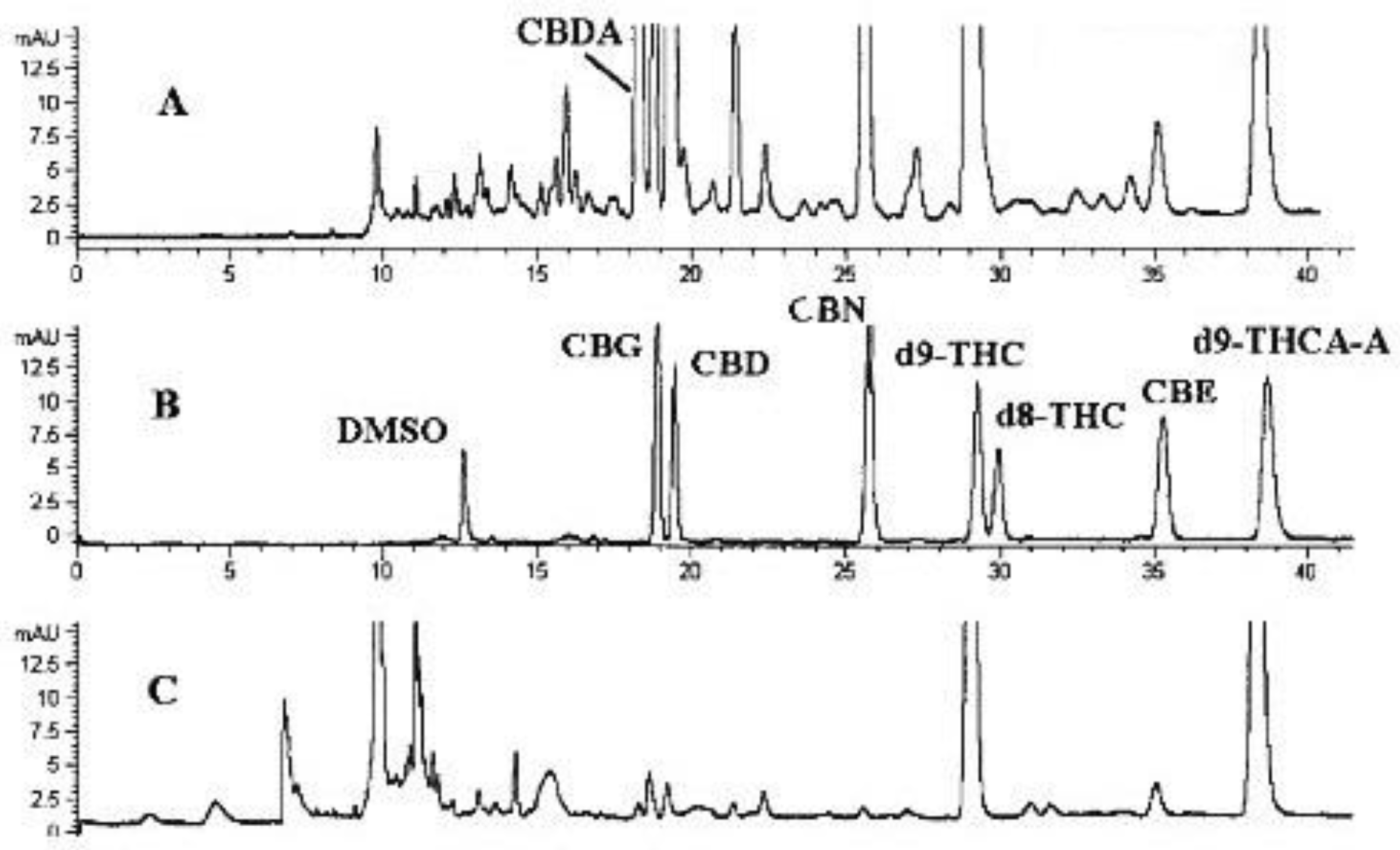
2. CE Methods for the Determination of CBD and Related Substances
2.1. Non-Aqueous Capillary Electrophoresis (NACE)
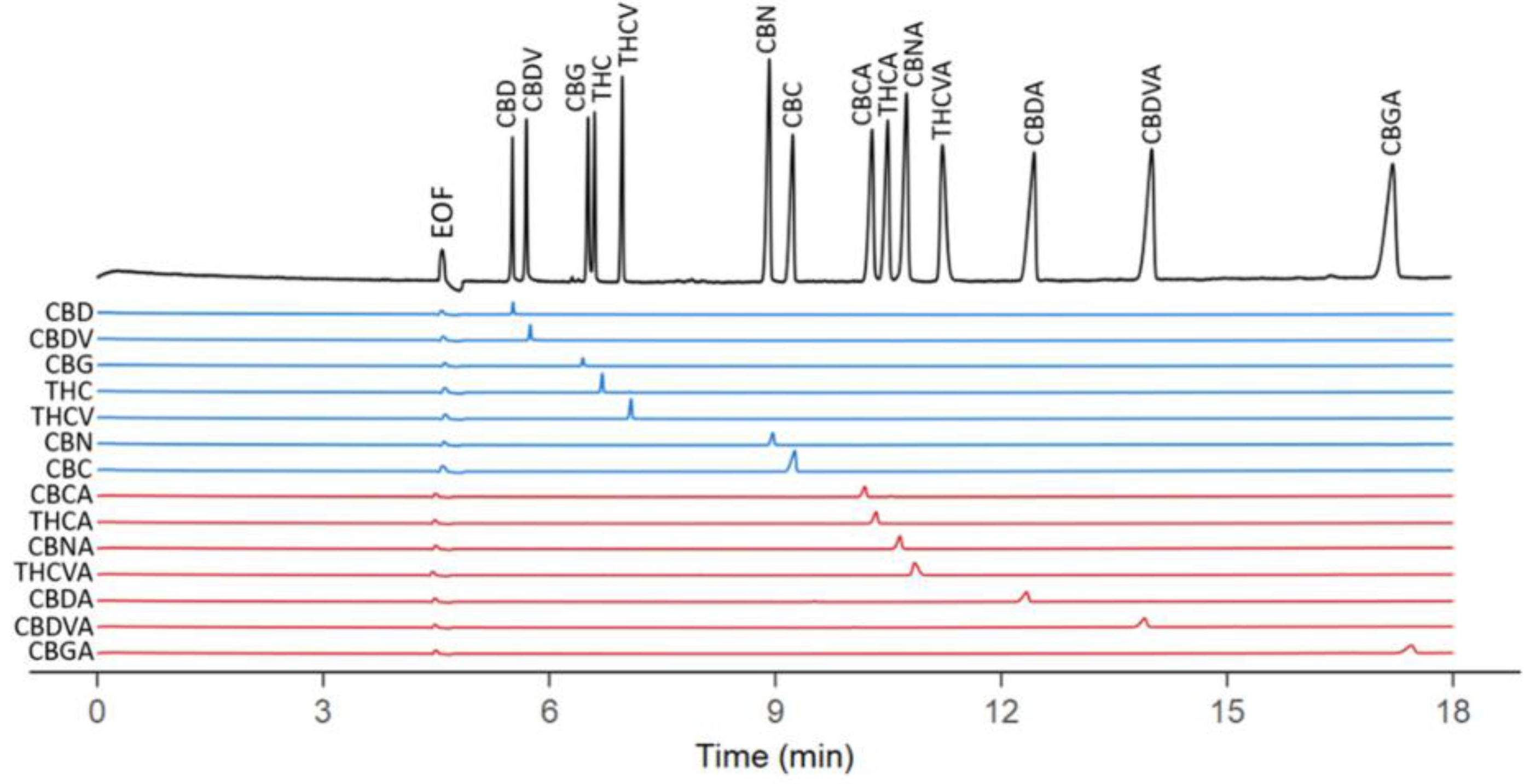
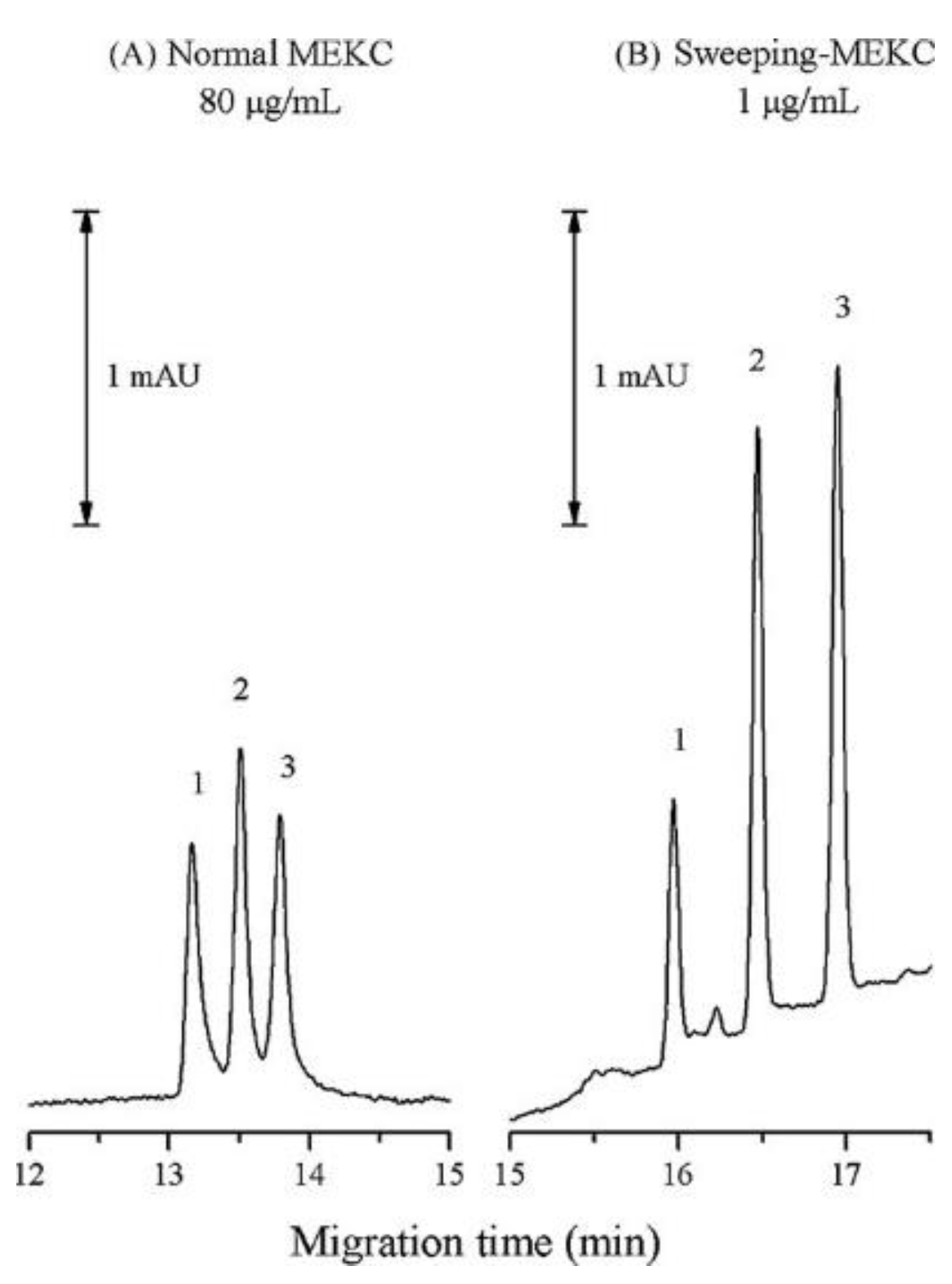
2.2. Micellar Electrokinetic Chromatography (MEKC)
2.3. Capillary Electrochromatography (CEC)
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
Abbreviations
References
- Klumpers, L.E.; Thacker, D.L. A brief background on cannabis: From plant to medical indications. J. AOAC Int. 2019, 102, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef] [PubMed]
- Hazekamp, A.; Fischedick, J.T. Cannabis-from cultivar to chemovar. Drug Test. Anal. 2012, 4, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Morales, P.; Reggio, P.H.; Jagerovic, N. An overview on medicinal chemistry of synthetic and natural derivatives of cannabidiol. Front. Pharmacol. 2017, 8, 422. [Google Scholar] [CrossRef] [PubMed]
- Hanuš, L.O.; Meyer, S.M.; Muñoz, E.; Taglialatela-Scafati, O.; Appendino, G. Phytocannabinoids: A unified critical inventory. Nat. Prod. Rep. 2016, 33, 1357–1392. [Google Scholar] [CrossRef]
- Reekie, T.A.; Scott, M.P.; Kassiou, M. The evolving science of phytocannabinoids. Nat. Rev. Chem. 2017, 2, 1–12. [Google Scholar] [CrossRef]
- Carliner, H.; Brown, Q.L.; Sarvet, A.L.; Hasin, D.S. Cannabis use, attitudes, and legal status in the US: A review. Prev. Med. 2017, 104, 13–23. [Google Scholar] [CrossRef]
- Vlad, R.A.; Hancu, G.; Ciurba, A.; Antonoaea, P.; Rédai, E.M.; Todoran, N.; Silasi, O.; Muntean, D.L. Cannabidiol-therapeutic and legal aspects. Die Pharmazie 2020, 75, 463–469. [Google Scholar]
- Nelson, K.M.; Bisson, J.; Singh, G.; Graham, J.G.; Chen, S.N.; Friesen, J.B.; Dahlin, J.L.; Niemitz, M.; Walters, M.A.; Pauli, G.F. The essential medicinal chemistry of cannabidiol (CBD). J. Med. Chem. 2020, 63, 12137–12155. [Google Scholar] [CrossRef]
- Abu-Sawwa, R.; Scutt, B.; Park, Y. Emerging use of Epidiolex (cannabidiol) in epilepsy. J. Pediatr. Pharmacol. Ther. 2020, 25, 485–499. [Google Scholar] [CrossRef]
- Sastre-Garriga, J.; Vila, C.; Clissold, S.; Montalban, X. THC and CBD oromucosal spray (Sativex®) in the management of spasticity associated with multiple sclerosis. Expert Rev. Neurother. 2011, 11, 627–637. [Google Scholar] [CrossRef] [PubMed]
- White, C.M. A review of human studies assessing cannabidiol’s (CBD) therapeutic actions and potential. J. Clin. Pharmacol. 2019, 59, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Lucas, C.J.; Galettis, P.; Schneider, J. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br. J. Clin. Pharmacol. 2018, 84, 2477–2482. [Google Scholar] [CrossRef]
- Laprairie, R.B.; Bagher, A.M.; Kelly, M.E.M.; Denovan-Wright, E.M. Cannabidiol is a negative allosteric modulator of the cannabinoid CB 1 receptor. Br. J. Pharmacol. 2015, 172, 4790–4805. [Google Scholar] [CrossRef]
- Mills, B.; Yepes, A.; Nugent, K. Synthetic cannabinoids. Am. J. Med. Sci. 2015, 350, 59–62. [Google Scholar] [CrossRef]
- Fasinu, P.S.; Phillips, S.; ElSohly, M.A.; Walker, L.A. Current status and prospects for cannabidiol preparations as new therapeutic agents. Pharmacotherapy 2016, 36, 781–796. [Google Scholar] [CrossRef] [PubMed]
- Presley, B.C.; Jansen-Varnum, S.A.; Logan, B.K. Analysis of synthetic cannabinoids in botanical material: A review of analytical methods and findings. Forensic Sci. Rev. 2013, 25, 27–46. [Google Scholar] [PubMed]
- Raharjo, T.J.; Verpoorte, R. Methods for the analysis of cannabinoids in biological materials: A review. Phytochem. Anal. 2004, 15, 79–94. [Google Scholar] [CrossRef]
- Ramirez, C.L.; Fanovich, M.A.; Churio, M.S. Cannabinoids: Extraction methods, analysis, and physicochemical characterization. Stud. Nat. Prod. Chem. 2019, 61, 143–173. [Google Scholar]
- Pourseyed Lazarjani, M.; Torres, S.; Hooker, T.; Fowlie, C.; Young, O.; Seyfoddin, A. Methods for quantification of cannabinoids: A narrative review. J. Cannabis Res. 2020, 2, 1–10. [Google Scholar] [CrossRef]
- Micalizzi, G.; Vento, F.; Alibrando, F.; Donnarumma, D.; Dugo, P.; Mondello, L. Cannabis Sativa L.: A comprehensive review on the analytical methodologies for cannabinoids and terpenes characterization. J. Chromat. A 2021, 1637, 461864. [Google Scholar] [CrossRef] [PubMed]
- Citti, C.; Braghiroli, D.; Vandelli, M.A.; Cannazza, G. Pharmaceutical and biomedical analysis of cannabinoids: A critical review. J. Pharm. Biomed. Anal. 2018, 147, 565–579. [Google Scholar] [CrossRef] [PubMed]
- Brighenti, V.; Protti, M.; Anceschi, L.; Zanardi, C.; Mercolini, L.; Pellati, F. Emerging challenges in the extraction, analysis and bioanalysis of cannabidiol and related compounds. J. Pharm. Biomed. Anal. 2021, 192, 113633. [Google Scholar] [CrossRef] [PubMed]
- Řemínek, R.; Foret, F. Capillary electrophoretic methods for quality control analyses of pharmaceuticals: A review. Electrophoresis 2021, 42, 19–37. [Google Scholar] [CrossRef] [PubMed]
- Kenndler, E. A critical overview of non-aqueous capillary electrophoresis. Part I: Mobility and separation selectivity. J. Chromat. A 2014, 1335, 16–30. [Google Scholar] [CrossRef]
- Kenndler, E. A critical overview of non-aqueous capillary electrophoresis. Part II: Separation efficiency and analysis time. J. Chromat. A 2014, 1335, 31–41. [Google Scholar] [CrossRef]
- Backofen, U.; Matysik, F.M.; Lunte, C.E. Determination of cannabinoids in hair using high-pH non-aqueous electrolytes and electrochemical detection: Some aspects of sensitivity and selectivity. J. Chromat. A 2002, 942, 259–269. [Google Scholar] [CrossRef]
- Iwamuro, Y.; Iio-Ishimaru, R.; Chinaka, S.; Takayama, N.; Hayakawa, K. Analysis of 11-nor-Δ9-tetrahydrocannabinol-9-carboxylic acid and its glucuronide in urine by capillary electrophoresis/mass spectrometry. Biomed. Chromat. 2012, 26, 1452–1456. [Google Scholar] [CrossRef]
- Mazina, J.; Spiljova, A.; Vaher, M.; Kaljurand, M.; Kulp, M. A rapid capillary electrophoresis method with LED-induced native fluorescence detection for the analysis of cannabinoids in oral fluid. Anal. Methods 2015, 7, 7741–7747. [Google Scholar] [CrossRef]
- Saar-Reismaa, P.; Erme, E.; Vaher, M.; Kulp, M.; Kaljurand, M.; Mazina-Sinkar, J. In situ determination of illegal drugs in oral fluid by portable capillary electrophoresis with deep UV excited fluorescence detection. Anal. Chem. 2018, 90, 6253–6258. [Google Scholar] [CrossRef]
- Zaripov, E.A.; Lee, T.; Dou, Y.; Harris, C.S.; Egorov, A.; Berezovski, M.V. Single-run separation and quantification of 14 cannabinoids using capillary electrophoresis. Separations 2021, 8, 30. [Google Scholar] [CrossRef]
- Hiroyuki, N.; Terabe, S. Micellar electrokinetic chromatography perspectives in drug analysis. J. Chromat. A 2006, 735, 3–27. [Google Scholar] [CrossRef]
- Silva, M. Micellar electrokinetic chromatography: A review of methodological and instrumental innovations focusing on practical aspects. Electrophoresis 2013, 34, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Weinberger, R.; Lurie, I.S. Micellar electrokinetic capillary chromatography of illicit drug substances. Anal. Chem. 1991, 63, 823–827. [Google Scholar] [CrossRef]
- Wernly, P.; Thormann, W. Confirmation testing of 11-nor-Δ9-tetrahydrocannabinol-9-carboxylic acid in urine with micellar electrokinetic capillary chromatography. J. Chromat. A 1992, 608, 251–256. [Google Scholar] [CrossRef]
- Su, H.L.; Feng, L.I.; Jen, H.P.; Hsieh, Y.Z. Using sweeping micellar electrokinetic chromatography to analyze Δ9-tetrahydrocannabinol and its major metabolites. J. Chromat. A 2009, 1216, 3512–3517. [Google Scholar] [CrossRef] [PubMed]
- Gottardo, R.; Bertaso, A.; Pascali, J.; Sorio, D.; Musile, G.; Trapani, E.; Seri, C.; Sepelloni, G.; Tagliaro, F. Micellar electrokinetic chromatography: A new simple tool for the analysis of synthetic cannabinoids in herbal blends and for the rapid estimation of their log P values. J. Chromat. A 2012, 1267, 198–205. [Google Scholar] [CrossRef]
- Akamatsu, S.; Mitsuhashi, T. MEKC–MS/MS method using a volatile surfactant for the simultaneous determination of 12 synthetic cannabinoids. J. Sep. Sci. 2014, 37, 304–307. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.L.; Tsai, Y.H.; Hsu, W.L.; Lin, Y.H. An on-line stacking capillary electrophoresis method for the analysis of Δ9-tetrahydrocannabinol and its metabolites. J. Chromat. A 2015, 1426, 226–232. [Google Scholar] [CrossRef]
- Švidrnoch, M.; Přibylka, A.; Maier, V. Determination of selected synthetic cannabinoids and their metabolites by micellar electrokinetic chromatography–mass spectrometry employing perfluoroheptanoic acid-based micellar phase. Talanta 2016, 150, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Bartle, K.D.; Myers, P. Theory of capillary electrochromatography. J. Chromat. A 2001, 916, 3–23. [Google Scholar] [CrossRef]
- Lurie, I.S.; Meyers, R.P.; Conver, T.S. Capillary electrochromatography of cannabinoids. Anal. Chem. 1998, 70, 3255–3260. [Google Scholar] [CrossRef]
- Toraño, J.S.; Ramautar, R.; de Jong, G. Advances in capillary electrophoresis for the life sciences. J. Chromat. B 2019, 1118, 116–136. [Google Scholar] [CrossRef] [PubMed]
- Kristoff, C.J.; Bwanali, L.; Veltri, L.M.; Gautam, G.P.; Rutto, P.K.; Newton, E.O.; Holland, L.A. Challenging bioanalyses with capillary electrophoresis. Anal. Chem. 2019, 92, 49–66. [Google Scholar] [CrossRef] [PubMed]
- Simpson Jr, S.L.; Quirino, J.P.; Terabe, S. On-line sample preconcentration in capillary electrophoresis: Fundamentals and applications. J. Chromat. A 2008, 1184, 504–541. [Google Scholar] [CrossRef] [PubMed]
- Riekkola, M.L.; Jussila, M.; Porras, S.P.; Valkó, I.E. Non-aqueous capillary electrophoresis. J. Chromat. A 2000, 892, 155–170. [Google Scholar] [CrossRef]
- Quirino, J.P.; Terabe, S. Stacking of neutral analytes in micellar electrokinetic chromatography. J. Capill. Electrophor. 1997, 4, 233–245. [Google Scholar]
- Yang, L.; Lee, C.S. Micellar electrokinetic chromatography-mass spectrometry. J. Chromat. A 1997, 780, 207–218. [Google Scholar] [CrossRef]
| CE Technique | Analyte | Matrices | Analytical Conditions | References |
|---|---|---|---|---|
| CEC-UV | CBC, CBD, CBG, CBN, THC, Δ8-THC, THCA | plant extract | 25 mM phosphate in acetonitrile, pH 2.57, 20 °C, 25 kV, electrokinetic injection 5 kV × 3 s Hypersil C18 3-µm CEC capillary | [42] |
| NACE-ED | CBD, CBN, THC, THCA | hair | 5 mM sodium hydroxide dissolved in acetonitrile-methanol (1:1), 20 kV, 220 nm | [27] |
| MEKC-UV | THC, THC-OH, THC-COOH | urine | 25 mM phosphate, 75 mM SDS, 40% methanol (v/v), pH 2.6, 25 °C, −20 kV, UV 210 nm | [36] |
| MEKC-UV | 9 synthetic cannabinoids | herbal blends | 25 mM borax, 30 mM SDS, 20% n-propanol (v/v), pH 8.0, 25 °C, 30 kV, UV 220 nm | [37] |
| NACE-MS | THC, THC-COOH, THC-glucuronide | urine | 40 mM ammonium formate, pH 6.4, 25 °C, 30 kV MS: gas temperature, 300 °C; gas flow (N2) 8 L/min; nebulizer, N2 10 psi at electrophoresis, 0 psi at sample injection. capillary 4000 V, sheath liquid 5mM ammonium formate:methanol (50/50, v/v) | [28] |
| MEKC-MS | 12 synthetic cannabinoids | herbal blends | 50 mM perfluooctanoic acid, 20% acetonitrile/water (v/v), 25 °C, 30 kV MS: nebulizing and drying gas 69 kPa N2 and 10 L/min N2 at 30 °C, sheath liquid 5 mM ammonium formate in 50% v/v methanol/water | [38] |
| NACE-LEDIF | CBD, THC | saliva | 2.5 mM sodium hydroxide in methanol-acetonitrile (1:1), 17 kV, λex/em = 280/305 nm | [29] |
| MEKC-UV | THC, THC-OH, THC-COOH | urine | 30 mM phosphate, 100 mM SDS, 40% methanol (v/v), pH 2.5, 25 °C, 30 kV, UV 214 nm | [39] |
| MEKC-MS | synthetic cannabinoids | spiked urine spiked plasma | 150 mM ammonium hydroxide, 75 mM perfluoroheptanoic acid, 10% isopropanol (v/v), pH 9.20, 25 °C MS: ESI voltage +3.5 kV; drying gas (N2) flow rate 10 L/min, temperature 200 °C, nebulizing gas pressure 5 psi, sheath liquid methanol: water: formic acid (50:49.5:0.5, v/v/v) | [40] |
| NACE-UV | 14 phytocannabinoids (including CBD, THC) | plant extract | 6 mM sodium hydroxide dissolved in acetonitrile:water (60:40), 25 µM β-CD, 450V/cm, UV 230 nm | [31] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blebea, N.M.; Hancu, G.; Vlad, R.A.; Pricopie, A. Applications of Capillary Electrophoresis for the Determination of Cannabinoids in Different Matrices. Molecules 2023, 28, 638. https://doi.org/10.3390/molecules28020638
Blebea NM, Hancu G, Vlad RA, Pricopie A. Applications of Capillary Electrophoresis for the Determination of Cannabinoids in Different Matrices. Molecules. 2023; 28(2):638. https://doi.org/10.3390/molecules28020638
Chicago/Turabian StyleBlebea, Nicoleta Mirela, Gabriel Hancu, Robert Alexandru Vlad, and Andreea Pricopie. 2023. "Applications of Capillary Electrophoresis for the Determination of Cannabinoids in Different Matrices" Molecules 28, no. 2: 638. https://doi.org/10.3390/molecules28020638
APA StyleBlebea, N. M., Hancu, G., Vlad, R. A., & Pricopie, A. (2023). Applications of Capillary Electrophoresis for the Determination of Cannabinoids in Different Matrices. Molecules, 28(2), 638. https://doi.org/10.3390/molecules28020638








