1. Introduction
Interactions at the interface are of fundamental importance in chemistry, physics, and biology. The thermodynamic parameters of these interactions can be modulated using surfactants. Owing to their amphiphilic structure, which comprises a hydrophilic and hydrophobic part, surfactants decrease the surface tension or interfacial tension between two liquids, a liquid and gas, or a liquid and solid. Hence, they can act as wetting, dispersing, and emulsifying agents. These properties enable the production of a large number of products required in household chemistry and cosmetic, pharmaceutical, agrochemical, petrochemical, textile, and paper industries.
The dynamic development of surfactant chemistry is a continuation of what was initiated by nature, which created the biosurfactants necessary for the functioning of living organisms. Biosurfactants usually refer to surfactants of microbial origin and comprise lecithin, rhamnolipids, sophorolipids, and emulsan [
1,
2]. Biosurfactants, in addition to their intrinsic role, have a wide range of technical applications. For instance, they can solubilise hydrocarbon contaminants and can be used in enhanced oil recovery [
3,
4]. Owing to their low toxicity and biodegradability, biosurfactants are extremely valuable products from the viewpoint of environmental protection. The global market size for biosurfactants reached a value of more than USD 2.33 billion in 2021 and is expected to grow at a compound annual growth rate (CAGR) of 5.8% between 2023 and 2028, reaching a projected value of USD 3.27 billion by 2027 [
5,
6]. Unfortunately, the number of available biosurfactants as well as the range of their applications does not meet the requirements expected from surfactants.
Currently, the widest groups of surfactants that meet the application requirements are nonionic, anionic, cationic, and amphoteric synthetic surfactants. The global demand for these surfactants, including soaps, exceeds 20 million tons per year [
6]. The global surfactant market stood at a value of approximately USD 41.84 billion in 2022. The market is further expected to grow in the forecast period 2023–2030 at a CAGR of 4.6% to reach a value of USD 59.95 billion by 2030 [
6]. However, the large volume of surfactants disposed into the environment, despite the wastewater treatment processes, is a serious burden and threat to the environment. Thus, owing to the increasing demand for surfactants, the development of new and more effective surfactants is extremely important.
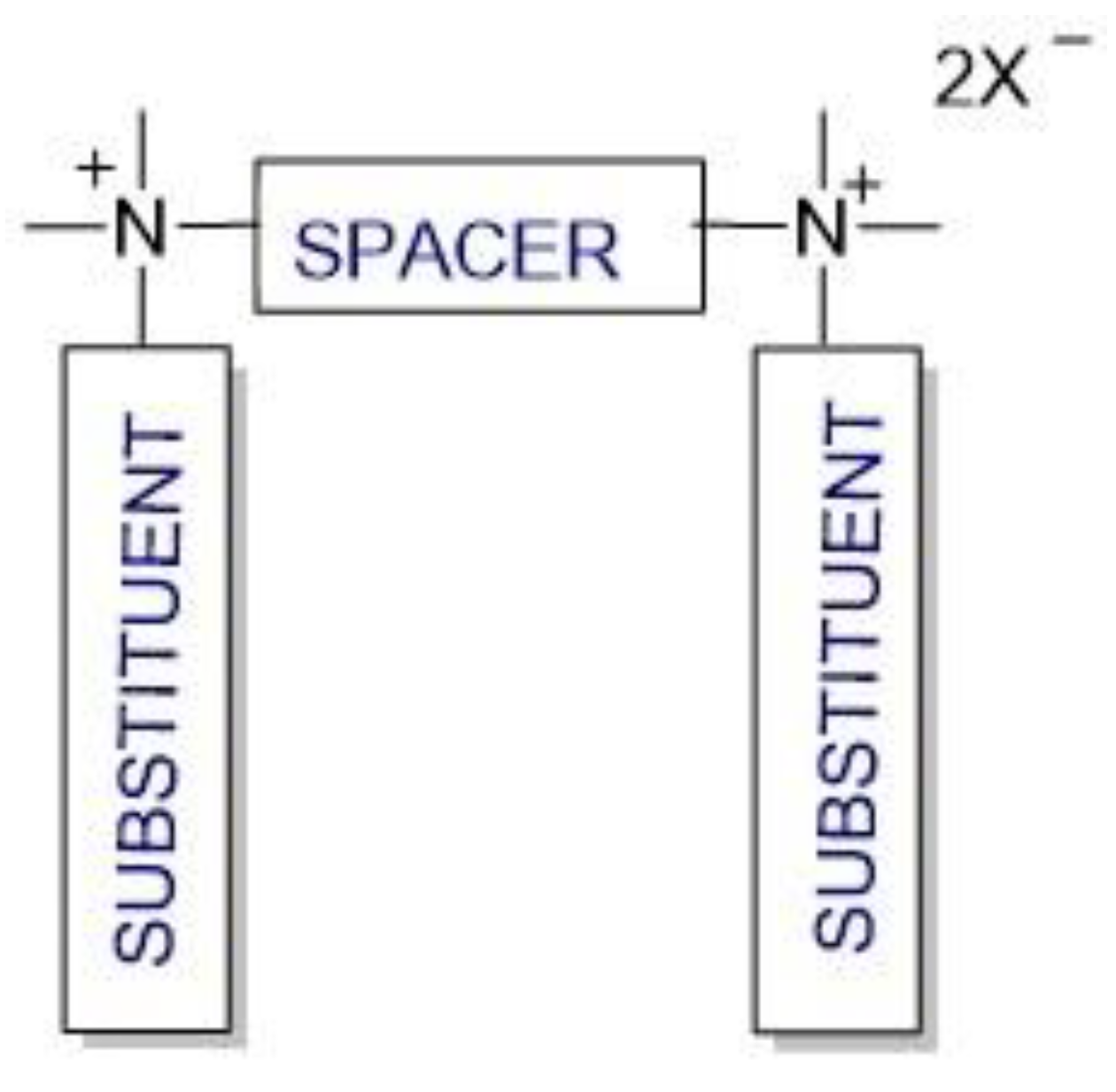
Cationic gemini surfactants have emerged as a result of the research conducted in this field over the last several years. Gemini surfactants are compounds that are composed of two hydrophilic head groups and two hydrophobic tails linked by a spacer at the head groups or close to them (
Figure 1). The spacers can be flexible (methylenes) or rigid (aromatic structures). The type of spacer (short or long, hydrophilic or hydrophobic) influences the shape of the micelles. The neutral charge of the molecule is retained in the presence of organic or inorganic counterions [
7,
8,
9].
The critical micelle concentration (CMC), surface tension (γ), and minimal inhibitory concentration (MIC) of gemini surfactants are a dozen times lower than those of monomeric surfactants. The unique properties of gemini surfactants with a wide range of hydrophilic–lipophilic balance (HLB) render them a very useful and innovative material in chemistry (e.g., for corrosion inhibition, micellar catalysis, nanoparticle synthesis, preparation of supramolecular solvents, nanoemulsion preparation, and synthesis of precisely defined polymers) [
10,
11,
12,
13,
14], medicine (as biocides, drug carriers, and capping agents for metal nanoparticles with biocidal properties or for preparing nonviral gene delivery systems and inducing protein conformational changes) [
15,
16,
17,
18,
19,
20], and optoelectronics (through a spatial network of well-dispersed molecules) [
7]. Gemini surfactants are a modern solution for all areas that need surfactants, including households, detergents, personal care, institutional and industrial cleaning, food processing, plastics, paints and coatings, oilfield chemicals, petrochemistry, agricultural chemicals, adhesives, and textiles [
20].
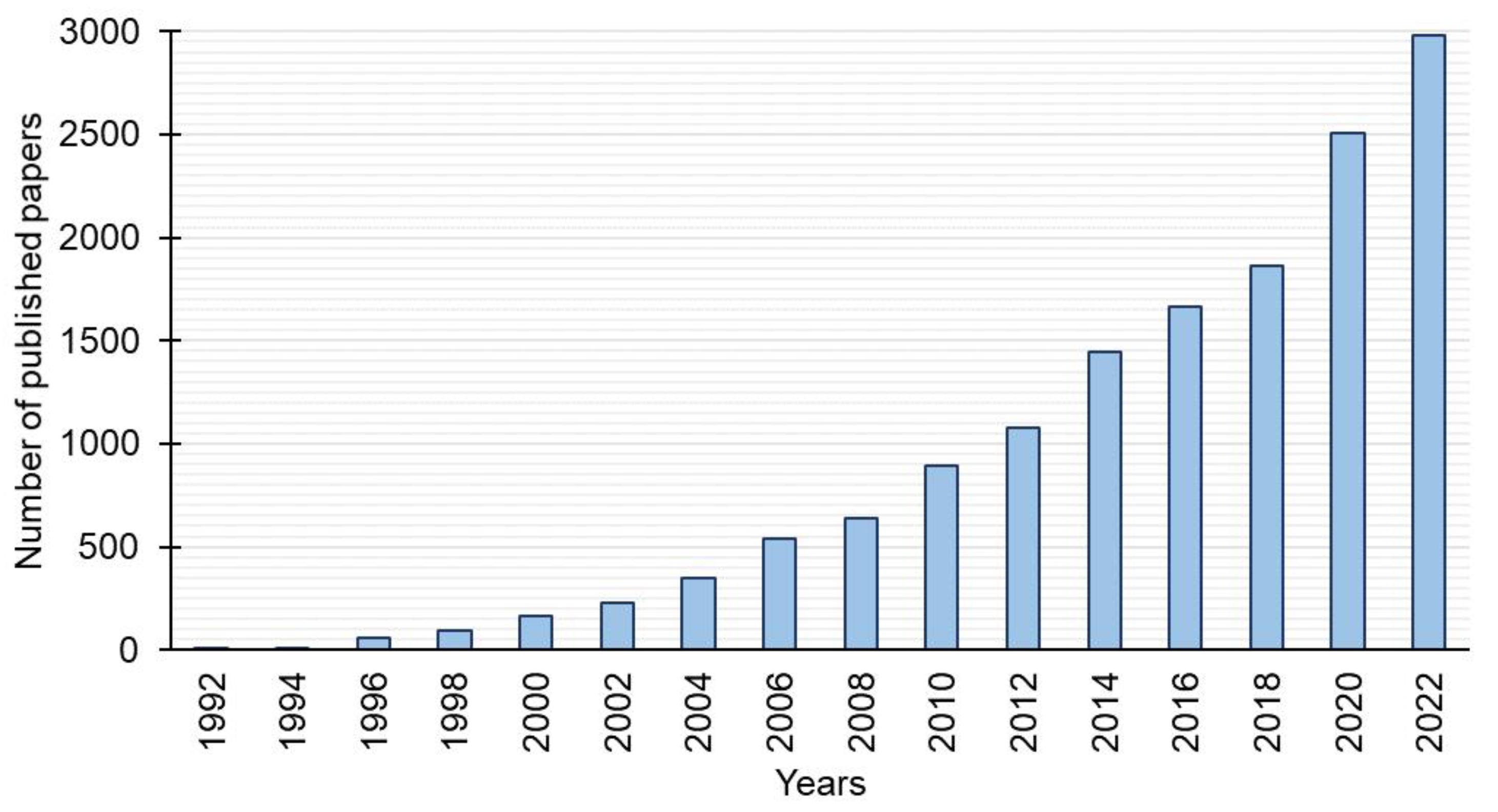
There is an increasing interest in gemini surfactants (
Figure 2). Among the large number of gemini surfactants reported in recent years, hexamethylene-1,6-bis(
N-dodecyl-
N,
N-dimethylammonium bromide) (12-6-12) deserves a special mention from the viewpoint of application. This review systematically presents the current studies devoted to the structure elucidation, synthesis, properties, and applications of 12-6-12, which can be a safer alternative to the surfactants used so far.
2. Structure and Synthesis
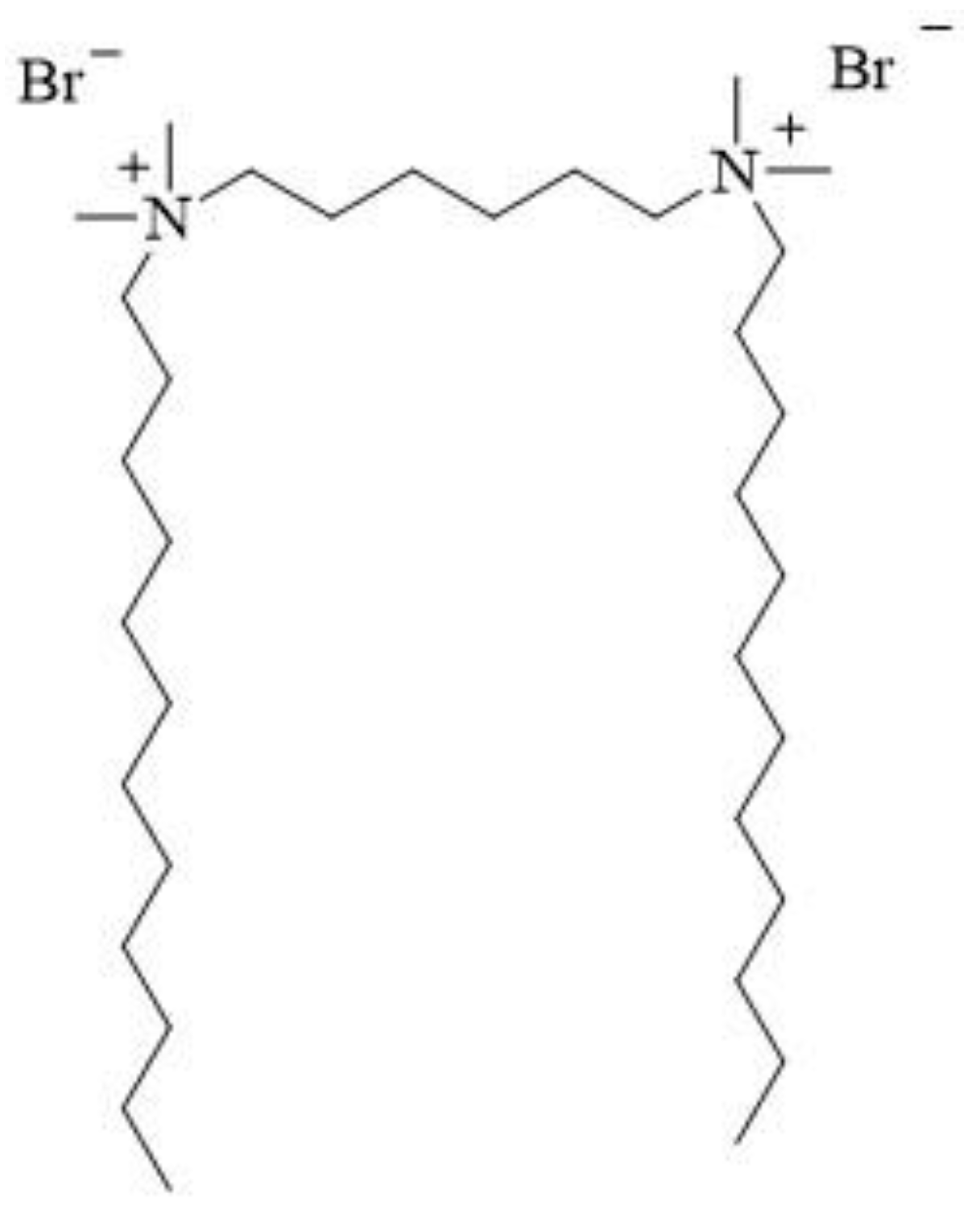
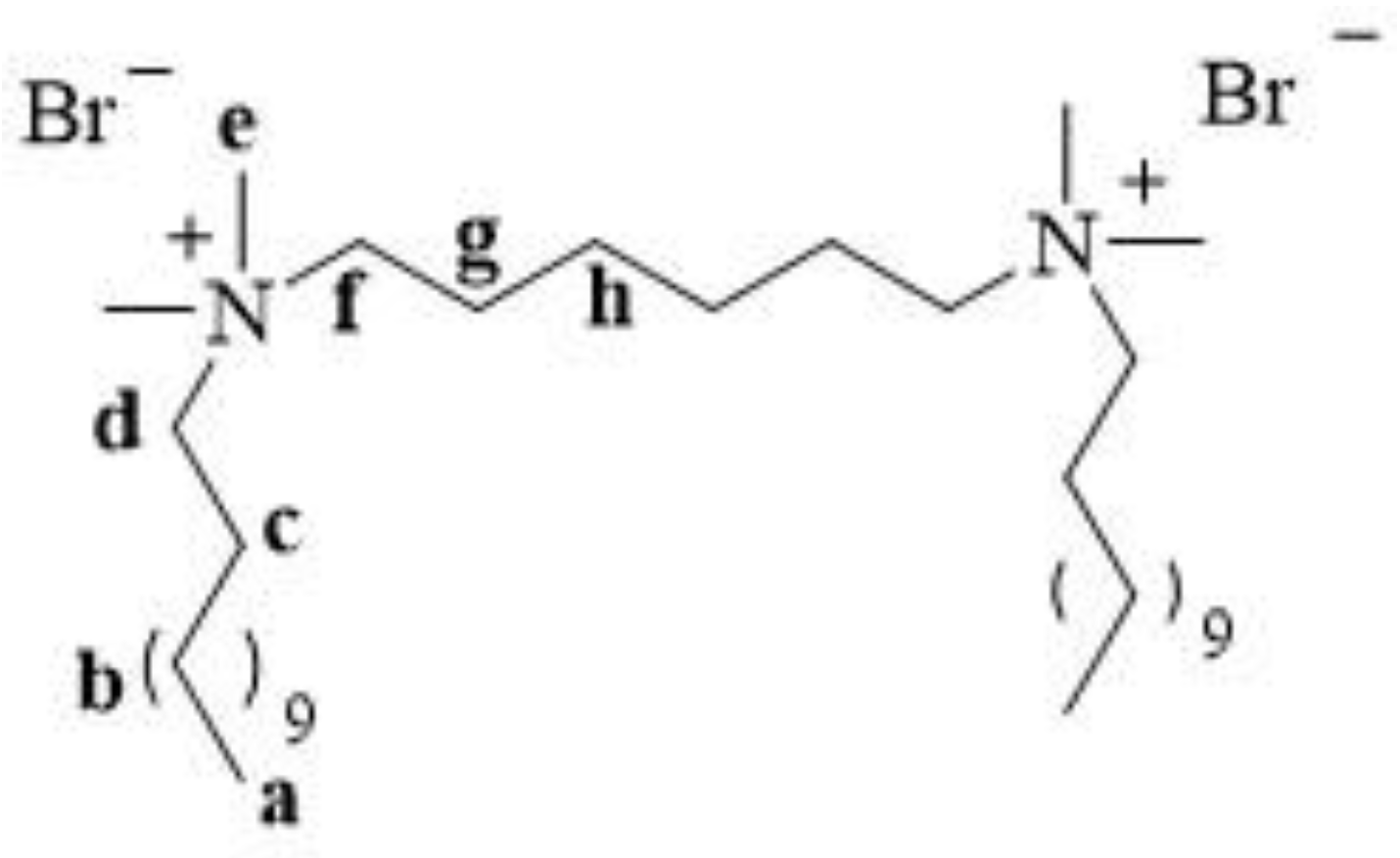
12-6-12 consists of two
N-dodecyl-
N,
N-dimethylammonium units connected with a chain of six methylene groups as a spacer. Bromine ions are present as counterions (
Figure 3). This compound has been classified under chemical abstracts service (CAS) number 18507-15-8 and is a dimeric analogue of
N-dodecyl-
N,
N,
N-trimethylammonium bromide (DTAB), which is commonly used as a microbiocide.
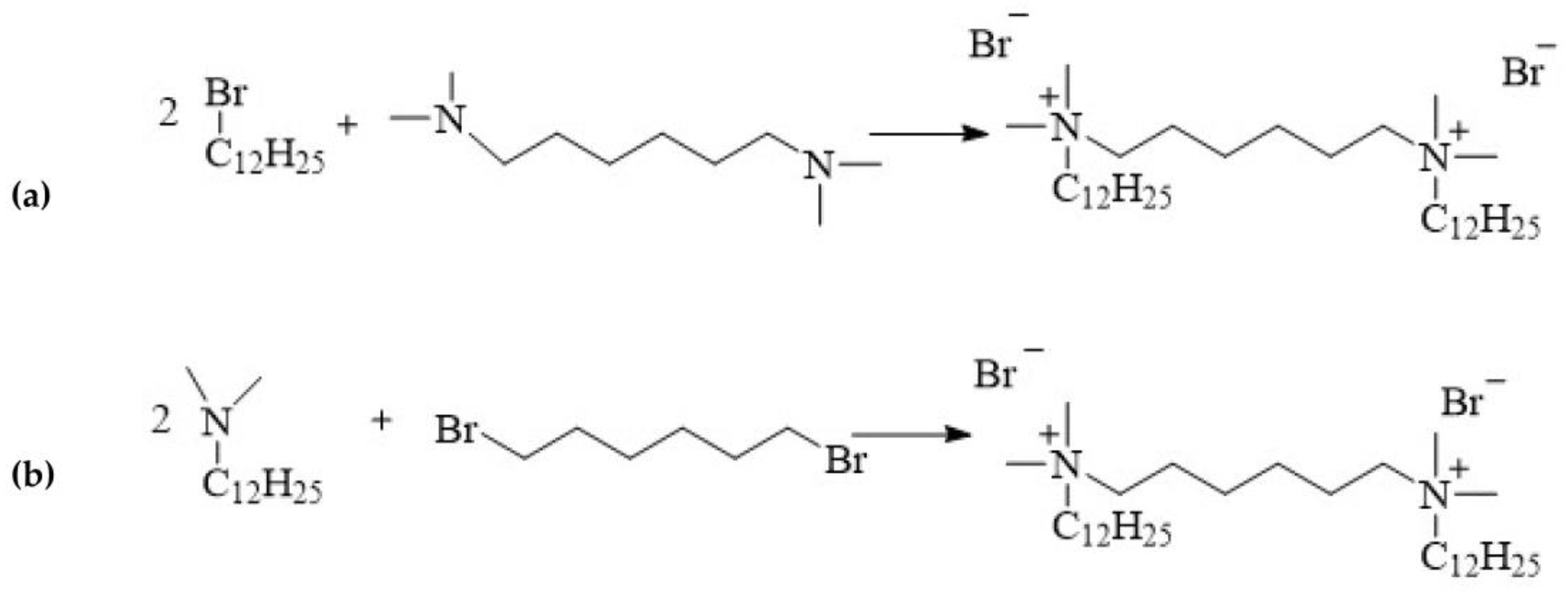
12-6-12 is one of the first compound to be classified as a double quaternary ammonium salt and defined as a gemini surfactant. This compound is synthesised via the quaternisation of amines, a process referred to as the Menschutkin reaction:
alkylation of hexamethylene-bis(
N,
N-dimethylamine) with 1-dodecylbromide (
Figure 4a);
linking of
N-dodecyl-
N,
N-dimethylamine with 1,6-dibromohexane (
Figure 4b).
The synthesis of 12-6-12 was first reported in 1968 by Sindenko et al. [
21]. Stoichiometric amounts of dodecylbromide and hexamethylene-bis(
N,
N-dimethylamine) were reacted in ethanol for the synthesis (yield 70%) [
21]. Regardless of the synthetic pathway, the quaternisation reaction always follows the S
N2 nucleophilic substitution mechanism. The rate of the reaction depends on the concentrations of both reagents, although there are reports of the use of excess amine [
22,
23,
24,
25] or bromide [
26,
27,
28]. Typically, polar solvents such as alcohols (methanol [
27,
29], ethanol [
30,
31], isopropanol [
32]), acetone [
19,
23,
33], or acetonitrile [
22,
24,
34] are used in the synthesis of 12-6-12. These reactions occur at the boiling point of the solvent. The type of solvent used determines the reaction time because S
N2 reactions are the fastest in polar aprotic solvents such as acetonitrile. Replacing ethanol with acetonitrile reduces the reaction time from 24 to 5 h [
25,
35]. 12-6-12 can also be synthesised in solvent mixtures such as the acetonitrile/toluene mixture [
36]. The reaction time can be shortened using microwave radiation [
37]. However, the most economical and ecological approach is to synthesise 12-6-12 under stoichiometric conditions at room temperature without a solvent [
38]. In this case, good yields of over 90% can be achieved using small amounts of reagents over a reaction time of 0.5 h [
38]. Solvent-free synthesis can also be easily carried out on a large scale.
To obtain pure 12-6-12, the crude product can be crystallised from acetonitrile [
35] or from dichloromethane–diethyl ether [
39], acetone–methanol [
40,
41], ethanol–ethyl acetate [
23], ethanol–diethyl ether [
24], acetone–ethanol [
21], and acetone–ethyl acetate [
25] mixtures.
3. Analysis
12-6-12 is a white water-soluble solid [
26]. It melts with decomposition at 225–226 °C [
28,
30]. The structure of a compound is usually confirmed using proton and carbon nuclear magnetic resonance (
1H and
13C NMR). The structure and numbering of 12-6-12 are shown in
Figure 5.
In the
1H NMR spectra, signals from the protons of the terminal methyl groups of the long alkyl chains (a) were observed at the lowest ppm values. The protons of the methyl (e) and methylene groups (in spacer (f) and alkyl chains (d)) next to the quaternary nitrogen atom exhibited signals at the highest ppm values.
Table 1 shows the
1H NMR chemical shifts for 12-6-12.
In the
13C NMR spectra, signals from the carbons of the terminal methyl groups of the long alkyl chains were observed at the lowest ppm values, similar to that in the
1H NMR spectra. Signals from the methylene groups in the alkyl chains, spacers, and methyl groups adjacent to the quaternary ammonium nitrogen appeared at the highest ppm values.
Table 2 shows the
13C NMR chemical shifts of 12-6-12.
In the FTIR spectrum of 12-6-12, broad intense absorption bands corresponding to asymmetric stretching (ν
as) and symmetric stretching (ν
s) vibrations of the methyl and methylene groups were observed at 2980–2850 cm
−1, while typical bands corresponding to deformation vibrations (δ) of the methyl and methylene groups appeared at 1490–1370 cm
−1. At 720 cm
−1, there was a typical band corresponding to the rocking vibrations (ρ) of the methylene groups derived from the long alkyl hydrocarbon chains. No stretching vibration bands for the N-H and O-H bonds were observed, which confirmed the purity of the compound [
38].
Mass spectrometry is another analytical method for confirming the structure and purity of synthesised compounds. Currently, soft ionisation techniques, such as electrospray ionisation (ESI), are used for diagnostic purposes. Although methods leading to many decays and fragmentation ions for 12-6-12 have been published [
43], they are currently of little diagnostic importance. Buse et al. published the electrospray ionisation quadrupole time-of-flight hybrid tandem mass spectrometry of a homologous series of gemini surfactants [
44]. The fragments and their corresponding
m/
z values for 12-6-12 are listed in
Table 3.
5. Toxicity and Environmental Impact
Generally, amphiphiles are known to influence the organisation of lipid membranes, and surfactants have been extensively studied in systems involving interactions with lipid membranes [
87]. Above a certain concentration, these compounds may exhibit undesirable properties, including toxicity. Therefore, it is very important to study the effects of substances on cells and living organisms before introducing them for use. Toxicity and CMC have been linked to gemini surfactants. Above a certain concentration, the toxicity of these compounds increases. It has also been shown that gemini surfactants are less toxic than their single-chain analogues [
88].
Almeida et al. studied the cytotoxicity of gemini surfactants and suggested that the toxicity increases with increasing spacer length and that surfactants with longer tails are less toxic than those with shorter tails [
87]. They chose the NCTC 2544 cell line, a human skin keratinocyte cell line, as a model of skin irritation. After 24 h of exposure to low concentrations of 12-6-12 (up to 10 mM), no significant cytotoxicity was observed in the cell line. However, at a 12-6-12 concentration of 50 mM, strong toxicity was observed [
87]. The cytotoxicity and skin irritation profiles of 12-6-12 were also studied by Silva et al. [
89]. These studies were performed in cultured human epidermal keratinocytes and human dermal fibroblasts. The skin integrity evaluation studies did not indicate relevant changes in the skin structure after the use of 12-6-12, while cytotoxicity studies established a relative cytotoxicity [
89].
Koziróg and Brycki tested the haemolytic activity of 12-6-12 and DTAB in terms of the MIC against the morphotic elements of sheep blood [
65]. 12-6-12 did not exhibit haemolytic activity at the highest MIC (0.029 μM/mL). A two-fold increase in the concentration lysed the erythrocytes slightly. Considerable disintegration of the erythrocyte membranes was observed at a 12-6-12 concentration of 0.58 μM/mL. DTAB, at the highest MIC of 1.01 μM/mL, caused slight haemolysis. In samples with 2.02 μM/mL of DTAB, a high degree (60%) of haemolysis was observed [
65]. Thus, 12-6-12 shows lower haemotoxicity at the used concentration than its monomeric analogue. However, considering the reduced cytotoxicity of 12-6-12 compared to DTAB and that its required concentrations are many times lower than those of DTAB in order to obtain the same effect, its environmental impact will be much less than that of monomeric cationic surfactants.
Zhang et al. studied the cytotoxicity of gemini surfactants against a rat glioma cell line (C6) and human kidney cell line (HEK293) [
32]. They presented the cytotoxic effect in terms of the IC
50 values, which were 5.1 and 3.3 µM for C6 and HEK293, respectively [
32]. Thus, 12-6-12 has potential applications in the medical field.
Research on the ecotoxicity of 12-6-12 and other gemini surfactants is sparse. Generally, these compounds are not readily biodegradable [
90]. However, their degree of biodegradation can be increased using bacteria immobilised on alginate [
91]. The degree of biodegradation of 12-6-12 determined by the CO
2 headspace test was 0% [
88]. Garcia et al. studied the aquatoxicity of 12-6-12 and DTAB against
D. magna, and the IC
50 values were 0.65 and 0.38 mg/L, respectively [
88]. This confirms the previous conclusion that gemini surfactants are less toxic to aquatic organisms than their monomeric analogues.
6. Applications
Surfactants are ubiquitous, being key components in a diverse range of complex industrial processes and utilitarian products such as dispersants, solubilisers, emulsifiers, demulsifiers, foaming agents, wetting agents, disinfectants, corrosion inhibitors, antistatic agents, and viscosity modifiers. In the last few decades, significant efforts have been made for the synthesis of new gemini surfactants, fuelled by their remarkably improved physicochemical properties that can be achieved by the modification of structural factors [
92]. Cationic gemini surfactants have wide applications due to their excellent surface activity [
7,
20]:
in foaming, agrichemical spreading aids, and cleaning;
from industrial to personal care applications;
phase transfer catalyst;
bleach activator;
as hair conditioners and fabric softeners.
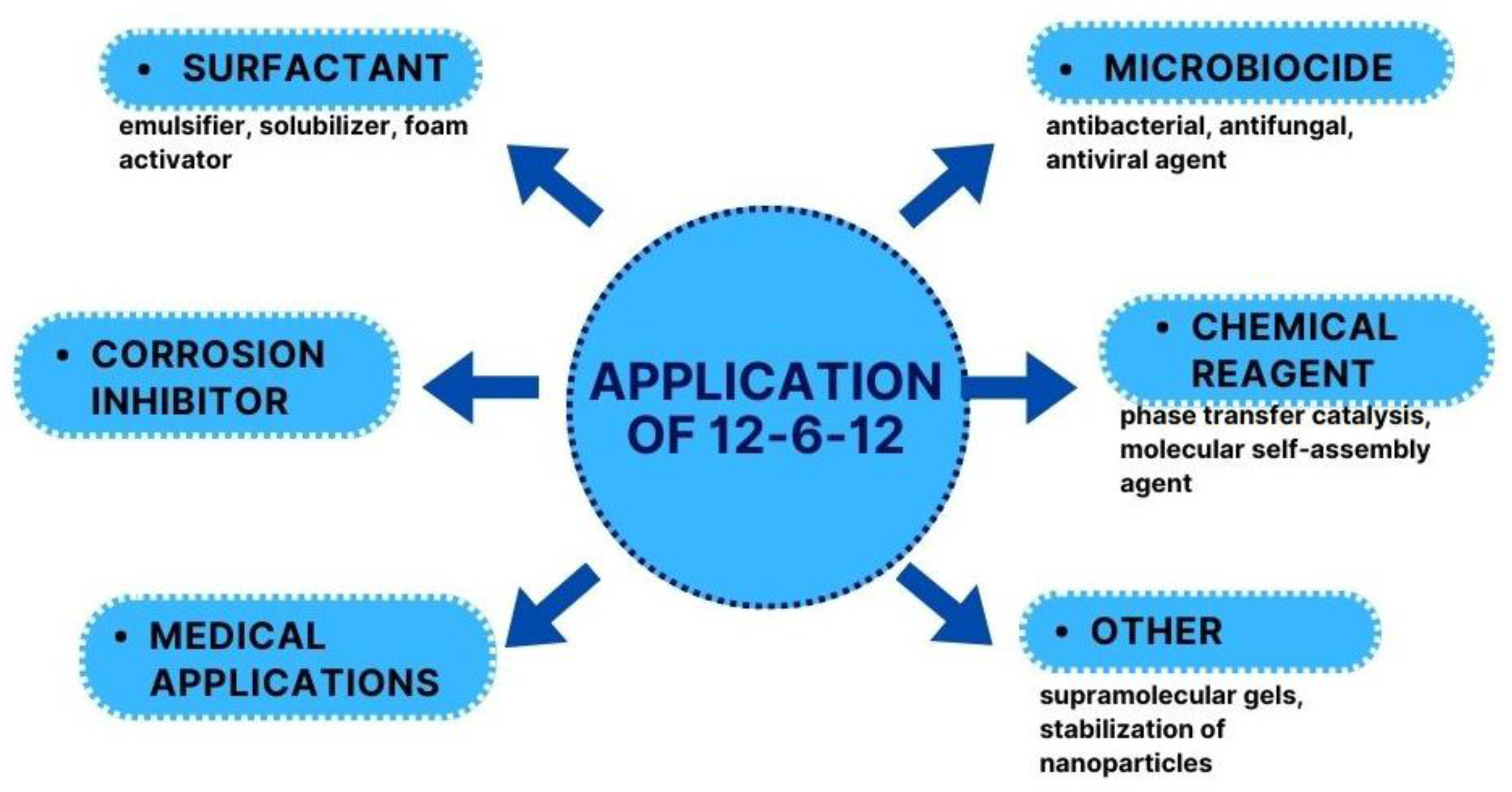
12-6-12 was first described 55 years ago, but the increased application interest of this compound can be defined in the 21st century. 12-6-12 can be used in several practical applications (
Figure 8).
12-6-12 is characterised by excellent surface, antimicrobial, and anticorrosion properties. As a compound that can be obtained economically and ecologically, it is an ideal product that has many industrial applications, including bioapplications (
Table 9).
Moreover, 12-6-12 can be used in chemical synthesis for the preparation of supramolecular gels [
101] and as a nanoparticle stabiliser [
28,
41,
102], an interfacial transfer catalyst [
30,
31,
103,
104,
105], and a molecular self-assembly agent [
106].
In conclusion, quaternary ammonium gemini surfactants are known for their multifunctional utility properties such as antimicrobial, surfactant, and anticorrosion properties. 12-6-12 has all the above mentioned properties of gemini surfactants and can be obtained in a one-step reaction, which is an indisputable and economically justified advantage over other gemini surfactants. Compared to its monochain counterpart, it shows better surface, antimicrobial, and anticorrosion activity and is characterised by a lower toxicity. The use of this compound in concentrations much lower than DTAB while providing the same utility effect makes 12-6-12 a candidate in many applications. Initially, this compound was used in applications as typical quaternary ammonium salts as an interfacial transfer catalyst. Currently, this compound finds potential applications in many different areas of life. Particularly noteworthy is the testing of this compound in biomedical applications. Due to its anticorrosive, biocidal, dispersing, and detergent properties, 12-6-12 can be used as an additive to car fuels and as an anticorrosion and antimicrobial agent in oil pipelines, smart anticorrosion coatings, etc. Hence, the possibility of producing it in large quantities in a one-step, waste-free synthesis is a huge advantage over other gemini surfactants.