Chitosan and Its Carboxymethyl-Based Membranes Produced by Crosslinking with Magnesium Phytate
Abstract
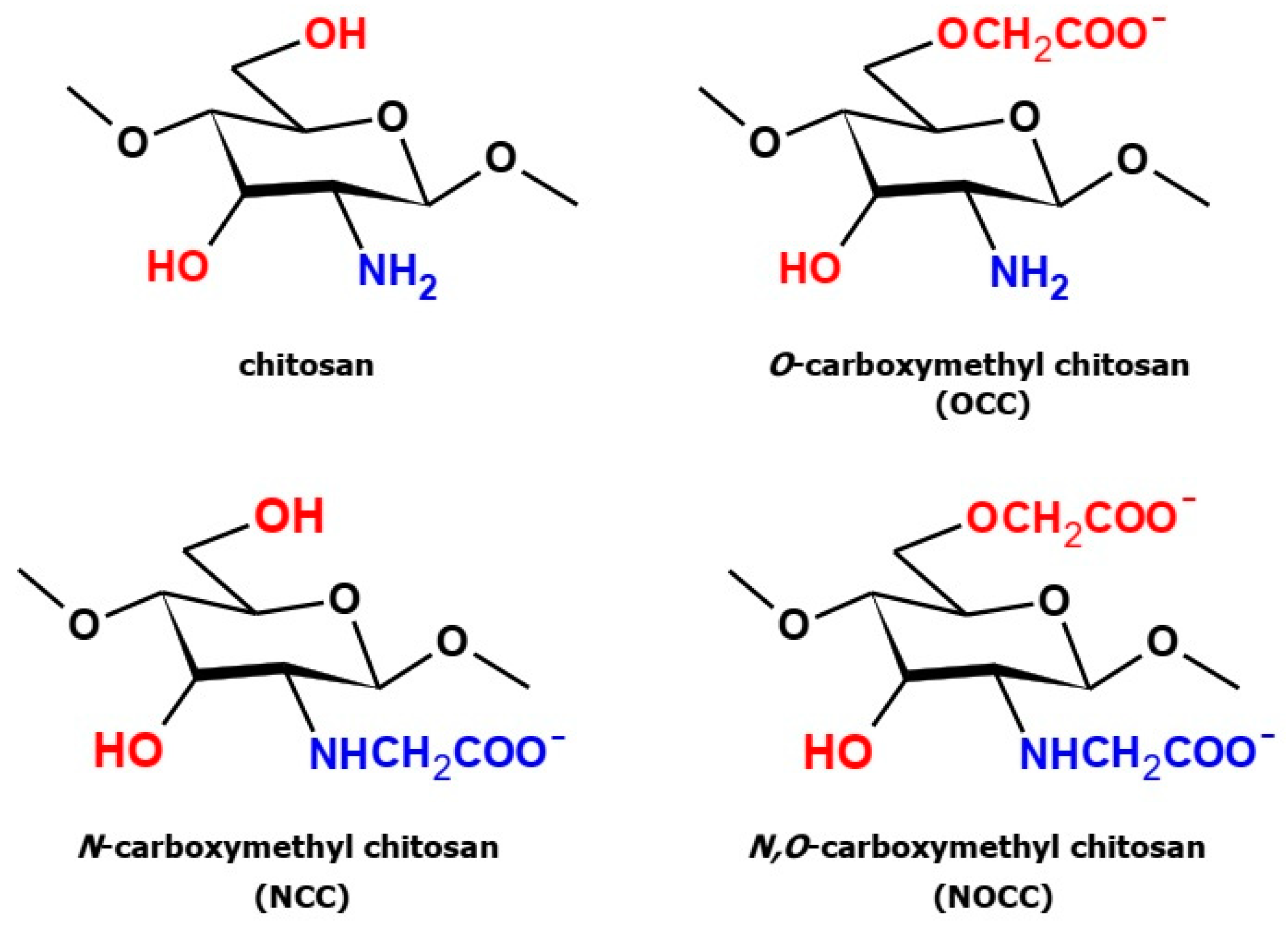
1. Introduction
2. Results and Discussion
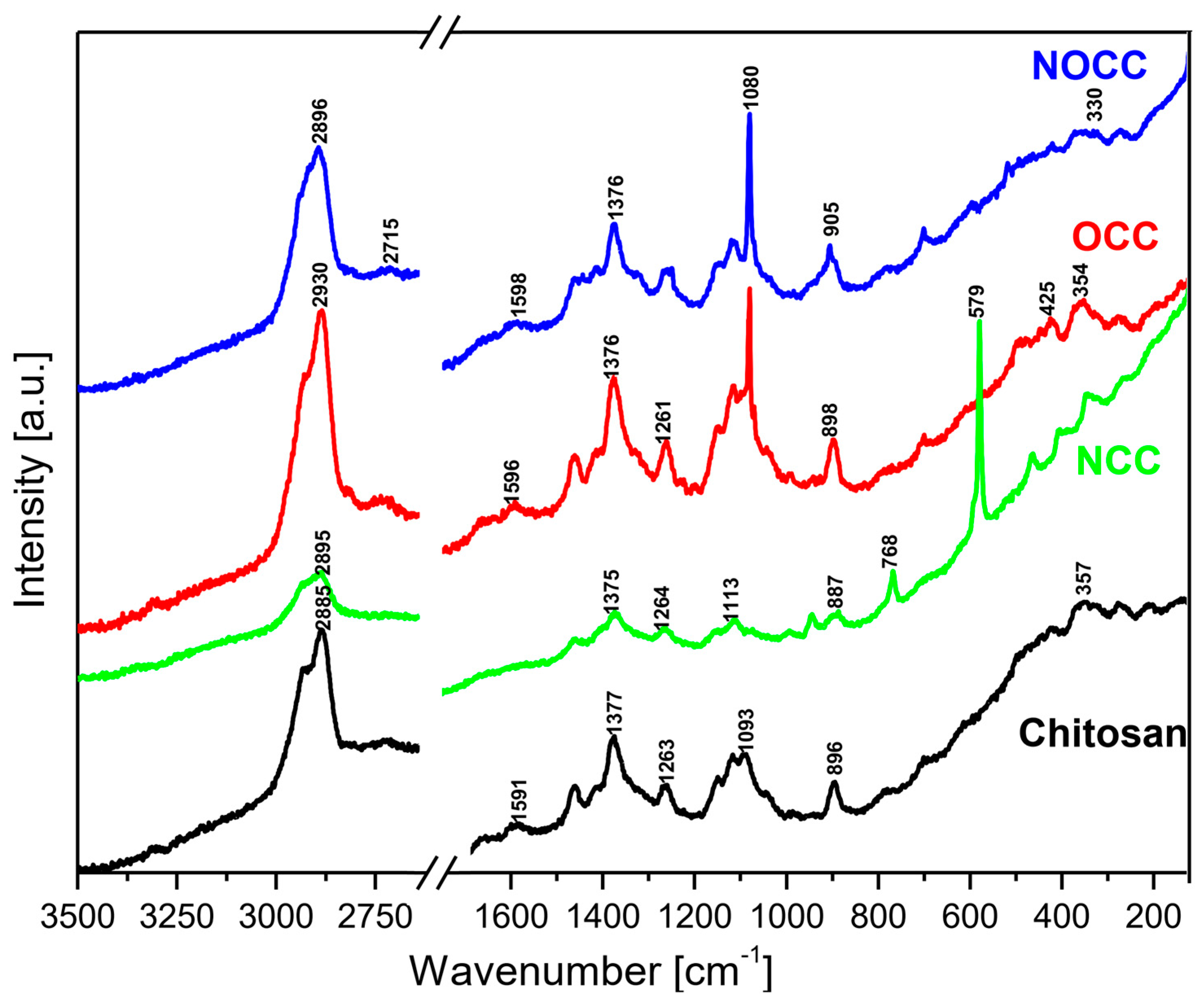
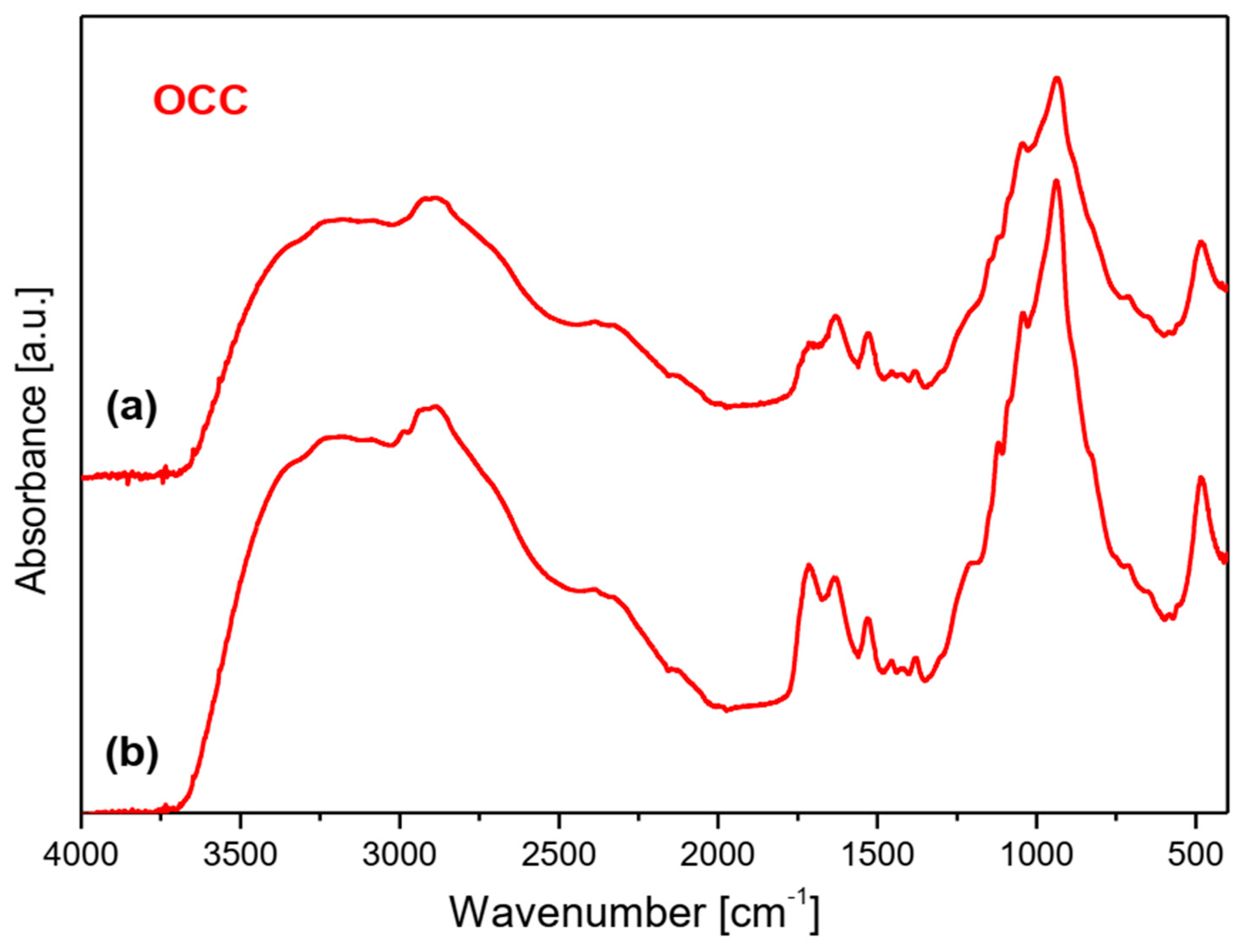
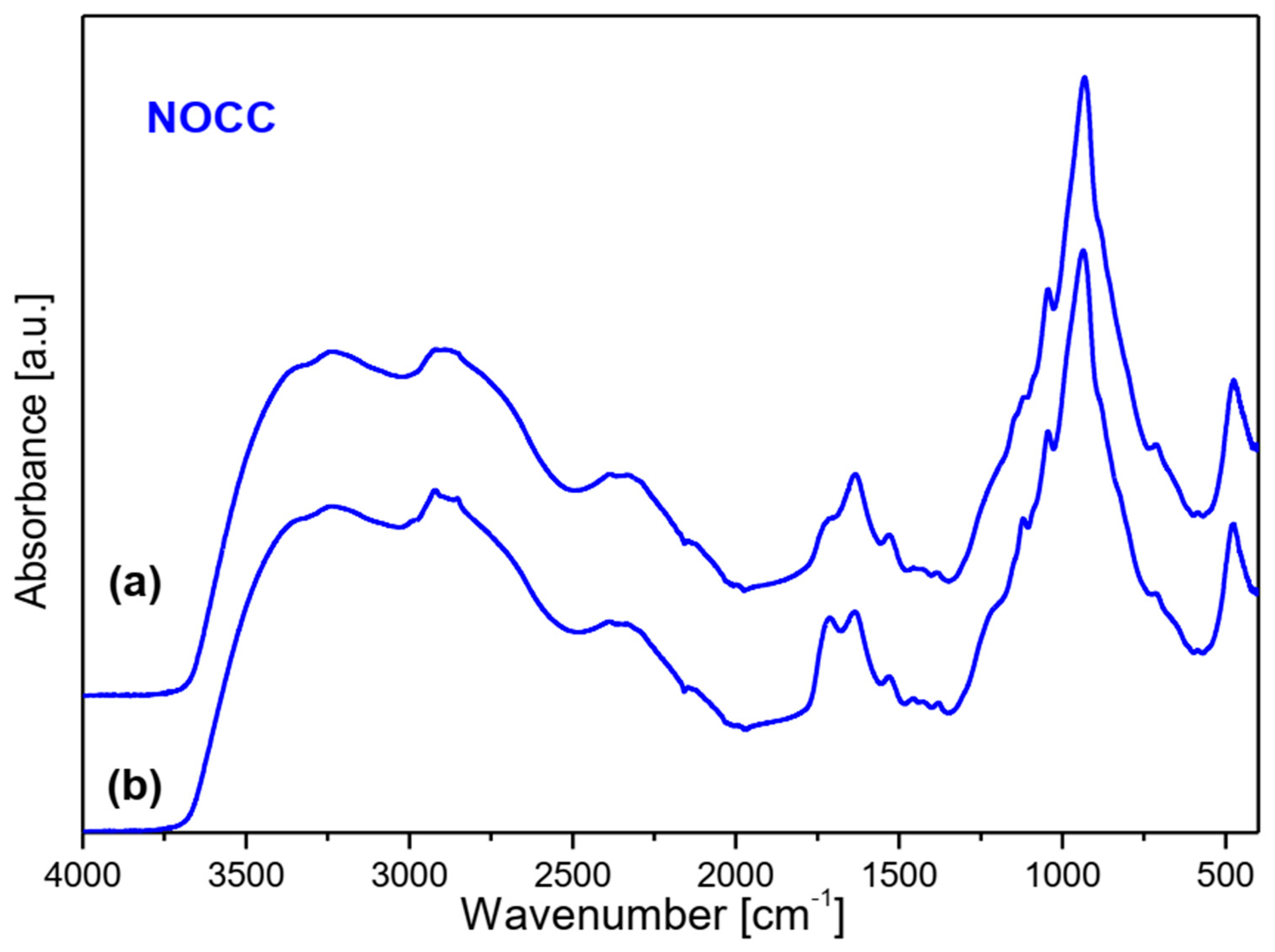
2.1. Vibrational Spectra
- (a)
- Very strong bands observed at 1080 cm−1 in the Raman spectra of NOCC and OCC derivatives correspond to the symmetric υs(C-O-C) vibration; they originate from the φ-CH2-OCH2COO fragment of these compounds.
- (b)
- The carboxymethylation of the pyranoid ring at the C-CH2-OH substituent also brings about changes in the IR spectra of OCC and NOCC derivatives—medium intensity bands are observed at 890 and 879 cm−1. They vibrations correspond to coupled υas(φ) + δ(CH) + ρ(CH2) vibrations of the pyranoid ring and methyl group of the φ-CH2-O- substituent.
- (c)
- Other changes are observed in the Raman spectra of the NCC and NOCC derivatives—new bands observed at 960 and 955 cm−1 correspond to the vibrations of the pyranoid ring coupled with the γ(NH) motion.
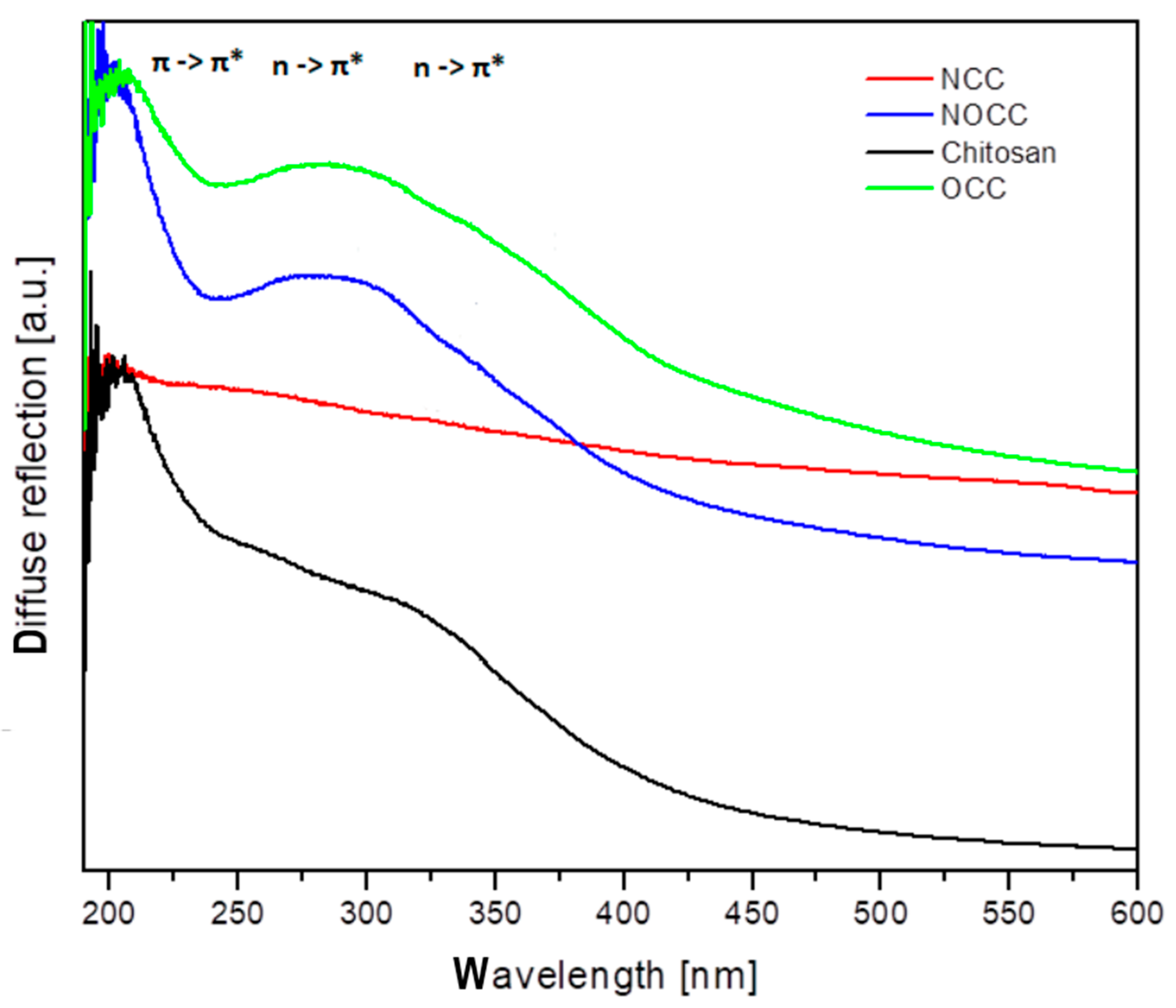
2.2. UV–Vis Spectra
2.3. Emission Spectra
2.4. Spectral and Microscopic Studies of the Crosslinked Chitosan
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Magnesium Phytate
3.3. Synthesis of Chitosan Derivatives
3.4. Spectroscopic Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Rinaudo, M. Chitin and Chitosan: Properties and Applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Dash, M.; Chiellini, F.; Ottenbrite, R.M.; Chiellini, E. Chitosan—A Versatile Semi-Synthetic Polymer in Biomedical Applications. Prog. Polym. Sci. 2011, 36, 981–1014. [Google Scholar] [CrossRef]
- Bernkop-Schnürch, A.; Dünnhaupt, S. Chitosan-Based Drug Delivery Systems. Eur. J. Pharm. Biopharm. 2012, 81, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Yogeshkumar, N.G.; GuravAtul, S.; Adhikrao, V.Y. Chitosan and Its Applications: A Review of Literature. Int. J. Res. Pharm. Biomed. Sci. 2013, 4, 312–331. [Google Scholar]
- Vimal, S.; Abdul Majeed, S.; Taju, G.; Nambi, K.S.N.; Sundar Raj, N.; Madan, N.; Farook, M.A.; Rajkumar, T.; Gopinath, D.; Sahul Hameed, A.S. Chitosan Tripolyphosphate (CS/TPP) Nanoparticles: Preparation, Characterization and Application for Gene Delivery in Shrimp. Acta Trop. 2013, 128, 486–493. [Google Scholar] [CrossRef]
- Cheung, R.; Ng, T.; Wong, J.; Chan, W. Chitosan: An Update on Potential Biomedical and Pharmaceutical Applications. Mar. Drugs 2015, 13, 5156–5186. [Google Scholar] [CrossRef]
- Hamed, I.; Özogul, F.; Regenstein, J.M. Industrial Applications of Crustacean By-Products (Chitin, Chitosan, and Chitooligosaccharides): A Review. Trends Food Sci. Technol. 2016, 48, 40–50. [Google Scholar] [CrossRef]
- Oladzadabbasabadi, N.; Mohammadi Nafchi, A.; Ariffin, F.; Wijekoon, M.M.J.O.; Al-Hassan, A.A.; Dheyab, M.A.; Ghasemlou, M. Recent Advances in Extraction, Modification, and Application of Chitosan in Packaging Industry. Carbohydr. Polym. 2022, 277, 118876. [Google Scholar] [CrossRef]
- Liu, Z.; Fan, B.; Zhao, J.; Yang, B.; Zheng, X. Benzothiazole Derivatives-Based Supramolecular Assemblies as Efficient Corrosion Inhibitors for Copper in Artificial Seawater: Formation, Interfacial Release and Protective Mechanisms. Corros. Sci. 2023, 212, 110957. [Google Scholar] [CrossRef]
- Higuchi, A.; Komiyama, J.; Iijima, T. The States of Water in Gel Cellophane Membranes. Polym. Bull. 1984, 11, 203–208. [Google Scholar] [CrossRef]
- Amaral, I.F.; Granja, P.L.; Melo, L.V.; Saramago, B.; Barbosa, M.A. Functionalization of Chitosan Membranes through Phosphorylation: Atomic Force Microscopy, Wettability, and Cytotoxicity Studies. J. Appl. Polym. Sci. 2006, 102, 276–284. [Google Scholar] [CrossRef]
- López-Pérez, P.M.; Marques, A.P.; da Silva, R.M.P.; Pashkuleva, I.; Reis, R.L. Effect of Chitosan Membrane Surface Modification via Plasma Induced Polymerization on the Adhesion of Osteoblast-like Cells. J. Mater. Chem. 2007, 17, 4064. [Google Scholar] [CrossRef]
- Xu, D.; Hein, S.; Wang, K. Chitosan Membrane in Separation Applications. Mater. Sci. Technol. 2008, 24, 1076–1087. [Google Scholar] [CrossRef]
- Lue, S.J.; Shieh, S.-J. Water States in Perfluorosulfonic Acid Membranes Using Differential Scanning Calorimetry. J. Macromol. Sci. Part B 2009, 48, 114–127. [Google Scholar] [CrossRef]
- Ostrowska-Czubenko, J.; Gierszewska-Drużyńska, M. Effect of Ionic Crosslinking on the Water State in Hydrogel Chitosan Membranes. Carbohydr. Polym. 2009, 77, 590–598. [Google Scholar] [CrossRef]
- Mengatto, L.; Luna, J.A.; Cabrera, M.I. Influence of Cross-Linking Density on Swelling and Estradiol Permeation of Chitosan Membranes. J. Mater. Sci. 2010, 45, 1046–1051. [Google Scholar] [CrossRef]
- Chakrabarty, T.; Kumar, M.; Shahi, V.K. Chitosan Based Membranes for Separation, Pervaporation and Fuel Cell Applications: Recent Developments. In Biopolymers; Sciyo: Rijeka, Croatia, 2010. [Google Scholar]
- Wu, X.; He, G.; Gu, S.; Hu, Z.; Yan, X. The State of Water in the Series of Sulfonated Poly (Phthalazinone Ether Sulfone Ketone) (SPPESK) Proton Exchange Membranes. Chem. Eng. J. 2010, 156, 578–581. [Google Scholar] [CrossRef]
- Ostrowska-Czubenko, J.; Pieróg, M.; Gierszewska-Drużyńska, M. Water State in Chemically and Physically Crosslinked Chitosan Membranes. J. Appl. Polym. Sci. 2013, 130, 1707–1715. [Google Scholar] [CrossRef]
- Lima, H.A.; Lia, F.M.V.; Ramdayal, S. Preparation and Characterization of Chitosan-Insulin-Tripolyphosphate Membrane for Controlled Drug Release: Effect of Cross Linking Agent. J. Biomater. Nanobiotechnol. 2014, 05, 211–219. [Google Scholar] [CrossRef]
- Gierszewska, M.; Ostrowska-Czubenko, J. Chitosan-Based Membranes with Different Ionic Crosslinking Density for Pharmaceutical and Industrial Applications. Carbohydr. Polym. 2016, 153, 501–511. [Google Scholar] [CrossRef]
- Monteiro, O.A.; Airoldi, C. Some Studies of Crosslinking Chitosan–Glutaraldehyde Interaction in a Homogeneous System. Int. J. Biol. Macromol. 1999, 26, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Crescenzi, V.; Francescangeli, A.; Taglienti, A.; Capitani, D.; Mannina, L. Synthesis and Partial Characterization of Hydrogels Obtained via Glutaraldehyde Crosslinking of Acetylated Chitosan and of Hyaluronan Derivatives. Biomacromolecules 2003, 4, 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- Harish Prashanth, K.V.; Tharanathan, R.N. Crosslinked Chitosan—Preparation and Characterization. Carbohydr. Res. 2006, 341, 169–173. [Google Scholar] [CrossRef]
- Li, H.; Gao, X.; Wang, Y.; Zhang, X.; Tong, Z. Comparison of Chitosan/Starch Composite Film Properties before and after Cross-Linking. Int. J. Biol. Macromol. 2013, 52, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Nagireddi, S.; Katiyar, V.; Uppaluri, R. Pd(II) Adsorption Characteristics of Glutaraldehyde Cross-Linked Chitosan Copolymer Resin. Int. J. Biol. Macromol. 2017, 94, 72–84. [Google Scholar] [CrossRef]
- Guerrero, P.; Muxika, A.; Zarandona, I.; de la Caba, K. Crosslinking of Chitosan Films Processed by Compression Molding. Carbohydr. Polym. 2019, 206, 820–826. [Google Scholar] [CrossRef]
- Yeamsuksawat, T.; Liang, J. Characterization and Release Kinetic of Crosslinked Chitosan Film Incorporated with α-Tocopherol. Food Packag. Shelf Life 2019, 22, 100415. [Google Scholar] [CrossRef]
- Gierszewska, M.; Jakubowska, E.; Olewnik-Kruszkowska, E. Effect of Chemical Crosslinking on Properties of Chitosan-Montmorillonite Composites. Polym. Test. 2019, 77, 105872. [Google Scholar] [CrossRef]
- Hunger, M.; Domalik-Pyzik, P.; Reczyńska, K.; Chłopek, J. Double Crosslinking of Chitosan/Vanillin Hydrogels as a Basis for Mechanically Strong Gradient Scaffolds for Tissue Engineering. Eng. Biomater. 2020, 23, 2–11. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, G.; Murray, P.; Zhang, H. Porous Chitosan by Crosslinking with Tricarboxylic Acid and Tuneable Release. SN Appl. Sci. 2020, 2, 435. [Google Scholar] [CrossRef]
- Berger, J.; Reist, M.; Mayer, J.M.; Felt, O.; Peppas, N.A.; Gurny, R. Structure and Interactions in Covalently and Ionically Crosslinked Chitosan Hydrogels for Biomedical Applications. Eur. J. Pharm. Biopharm. 2004, 57, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Mi, F.-L.; Sung, H.-W.; Shyu, S.-S.; Su, C.-C.; Peng, C.-K. Synthesis and Characterization of Biodegradable TPP/Genipin Co-Crosslinked Chitosan Gel Beads. Polymer 2003, 44, 6521–6530. [Google Scholar] [CrossRef]
- Peppas, N.A.; Mikos, A.G. Preparation Methods and Structure of Hydrogels. In Hydrogels in Medicine and Pharmacy; Peppas, N.A., Ed.; CRC Press: Boca Raton, FL, USA, 1986; pp. 1–26. ISBN 9780429285097. [Google Scholar]
- Shu, X.; Zhu, K. The Influence of Multivalent Phosphate Structure on the Properties of Ionically Cross-Linked Chitosan Films for Controlled Drug Release. Eur. J. Pharm. Biopharm. 2002, 54, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Zając, A.; Hanuza, J.; Wandas, M.; Dymińska, L. Determination of N-Acetylation Degree in Chitosan Using Raman Spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 134, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Zając, A.; Dymińska, L.; Lorenc, J.; Kaczmarek, S.M.; Leniec, G.; Ptak, M.; Hanuza, J. Spectroscopic Properties and Molecular Structure of Copper Phytate Complexes: IR, Raman, UV–Vis, EPR Studies and DFT Calculations. JBIC J. Biol. Inorg. Chem. 2019, 24, 11–20. [Google Scholar] [CrossRef]
- Zając, A.; Solarz, P.; Ptak, M.; Lorenc, J.; Kaczmarek, S.M.; Leniec, G.; Hermanowicz, K.; Hanuza, J. Synthesis, Optical and Magnetic Studies of Cerium and Europium Phytate Complexes—New Microporous Materials. J. Mol. Struct. 2021, 1233, 130114. [Google Scholar] [CrossRef]
- Novak, A. Hydrogen Bonding in Solids Correlation of Spectroscopic and Crystallographic Data. In Large Molecules; Springer: Berlin/Heidelberg, Germany, 1974; pp. 177–216. ISBN 978-3-540-37932-4. [Google Scholar]
- Lei, M.; Huang, W.; Jin, Z.; Sun, J.; Zhang, M.; Zhao, S. Effect of Molecular Structure and Ionization State on Aggregation of Carboxymethyl Chitosan: A Molecular Dynamics Study. Carbohydr. Polym. 2022, 297, 119993. [Google Scholar] [CrossRef]

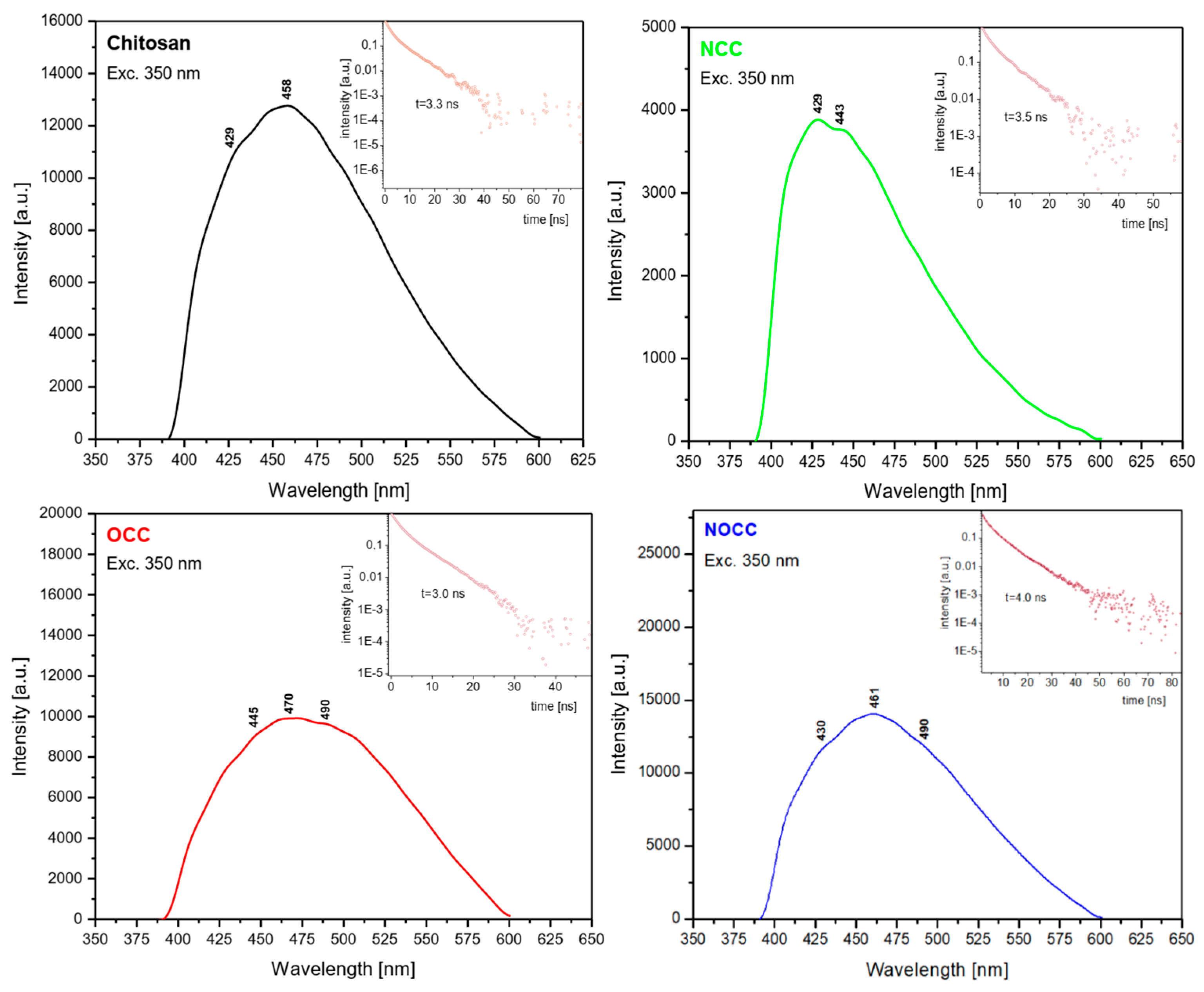


| T1 | T2 | T3 | |
|---|---|---|---|
| Chitosan | 19,607 | 21,834 | 23,310 cm−1 |
| NCC | 20,000 | 22,573 | 23,310 cm−1 |
| OCC | 20,408 | 21,276 | 22,471 cm−1 |
| NOCC | 20,408 | 21,691 | 23,255 cm−1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zając, A.; Sąsiadek, W.; Dymińska, L.; Ropuszyńska-Robak, P.; Hanuza, J.; Ptak, M.; Smółka, S.; Lisiecki, R.; Skrzypczak, K. Chitosan and Its Carboxymethyl-Based Membranes Produced by Crosslinking with Magnesium Phytate. Molecules 2023, 28, 5987. https://doi.org/10.3390/molecules28165987
Zając A, Sąsiadek W, Dymińska L, Ropuszyńska-Robak P, Hanuza J, Ptak M, Smółka S, Lisiecki R, Skrzypczak K. Chitosan and Its Carboxymethyl-Based Membranes Produced by Crosslinking with Magnesium Phytate. Molecules. 2023; 28(16):5987. https://doi.org/10.3390/molecules28165987
Chicago/Turabian StyleZając, Adam, Wojciech Sąsiadek, Lucyna Dymińska, Paulina Ropuszyńska-Robak, Jerzy Hanuza, Maciej Ptak, Szymon Smółka, Radosław Lisiecki, and Katarzyna Skrzypczak. 2023. "Chitosan and Its Carboxymethyl-Based Membranes Produced by Crosslinking with Magnesium Phytate" Molecules 28, no. 16: 5987. https://doi.org/10.3390/molecules28165987
APA StyleZając, A., Sąsiadek, W., Dymińska, L., Ropuszyńska-Robak, P., Hanuza, J., Ptak, M., Smółka, S., Lisiecki, R., & Skrzypczak, K. (2023). Chitosan and Its Carboxymethyl-Based Membranes Produced by Crosslinking with Magnesium Phytate. Molecules, 28(16), 5987. https://doi.org/10.3390/molecules28165987






