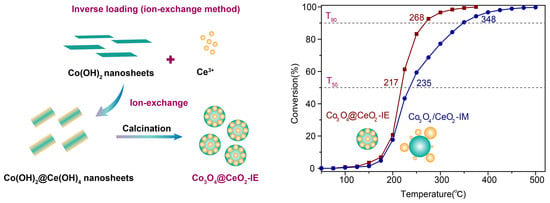
Enabling High Activity Catalyst Co3O4@CeO2 for Propane Catalytic Oxidation via Inverse Loading
Abstract
1. Introduction
2. Results
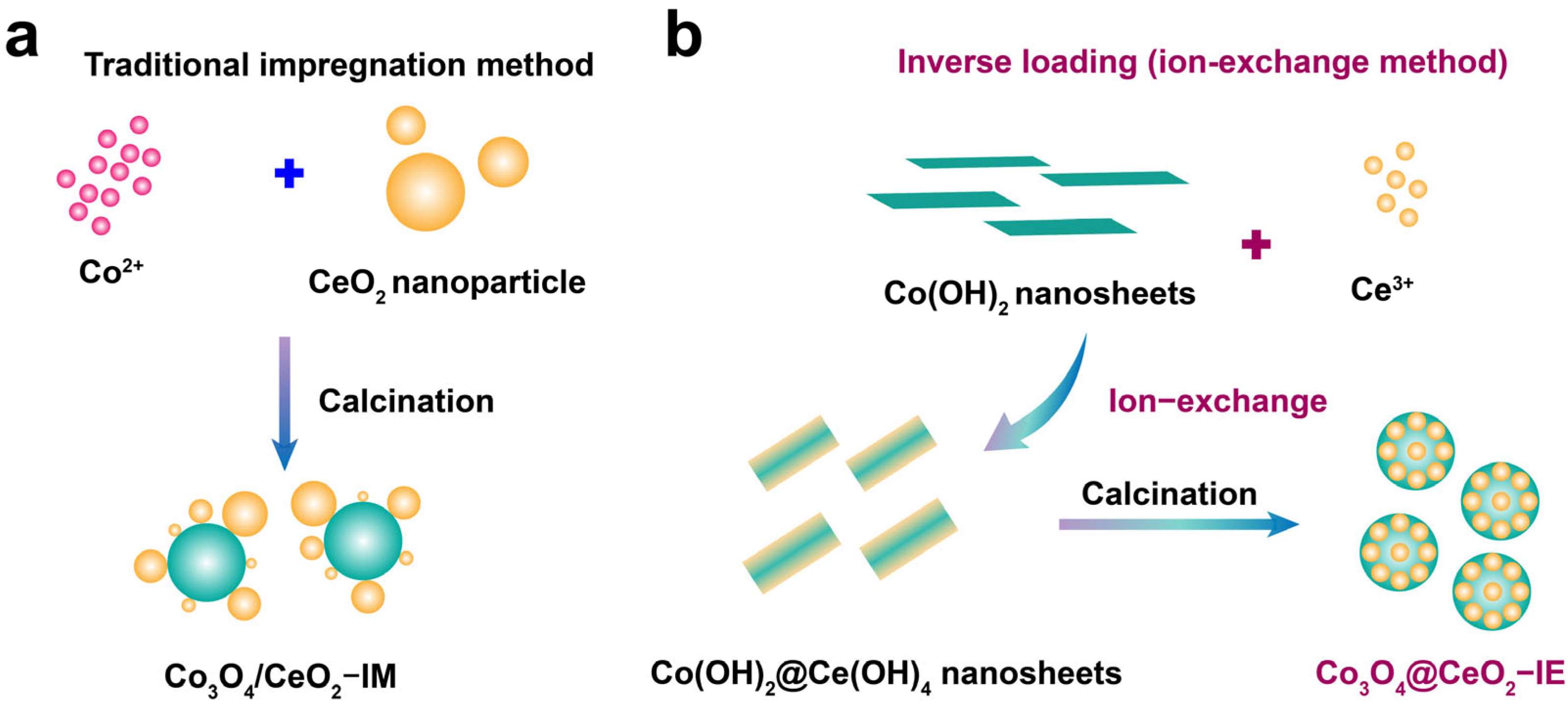
2.1. Structure and Morphology of the Obtained Samples
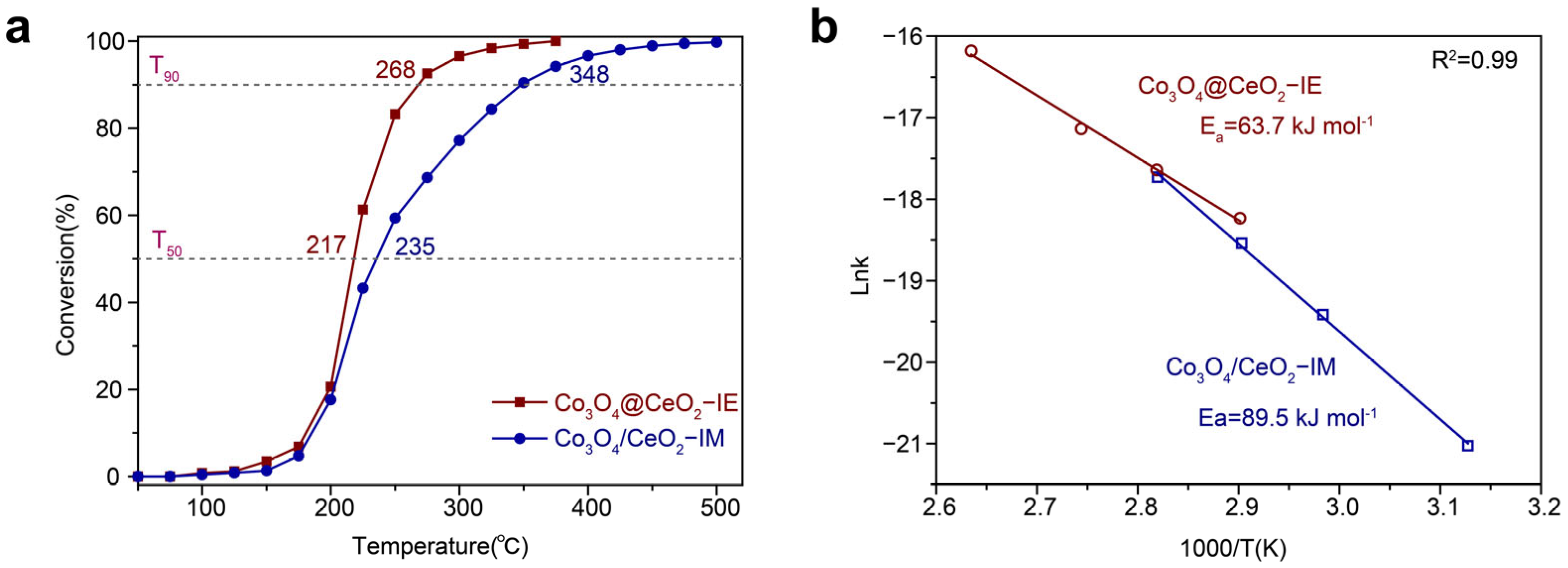
2.2. Catalytic Oxidation Performance
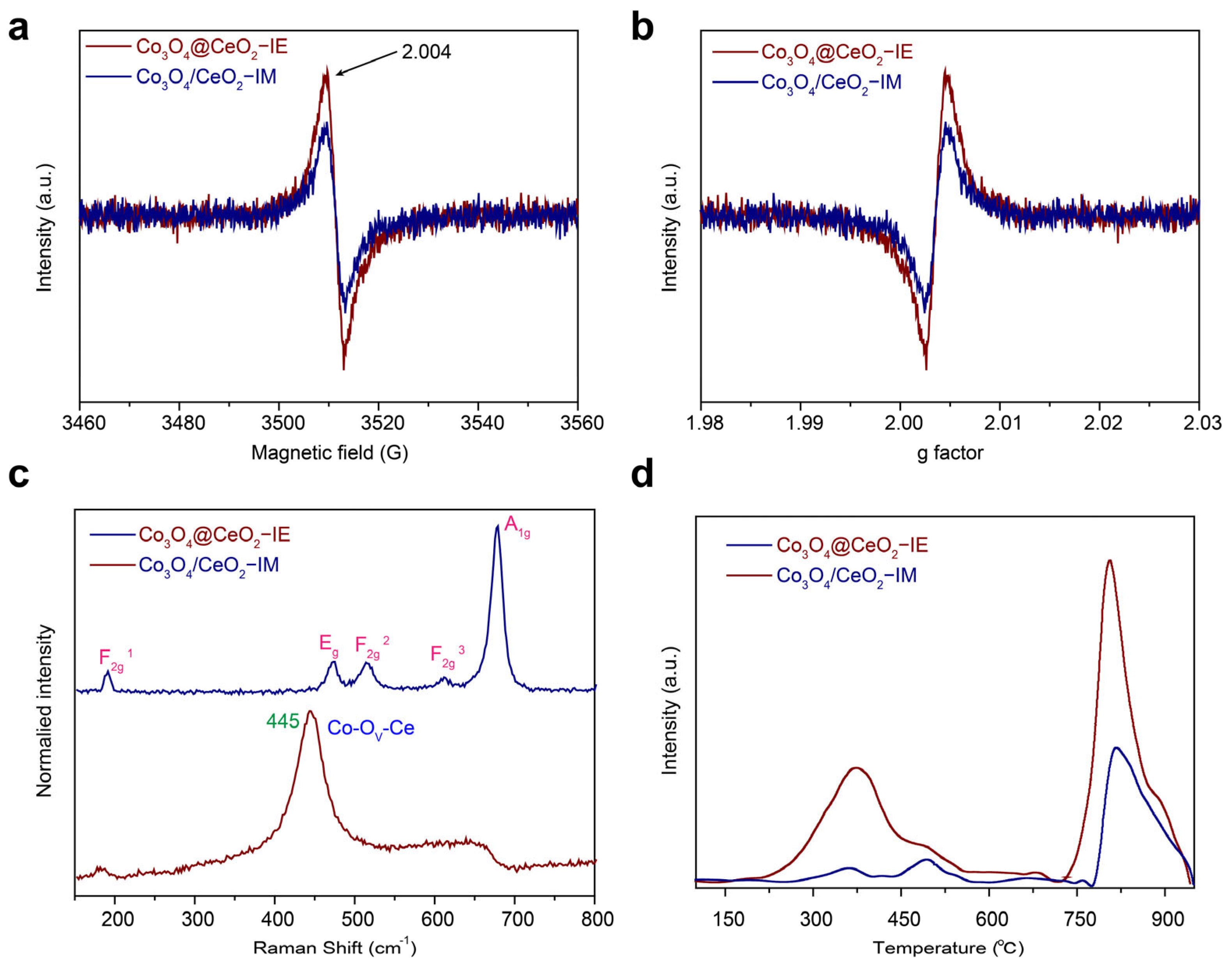
2.3. Surface Chemistry Analysis of the Catalysts
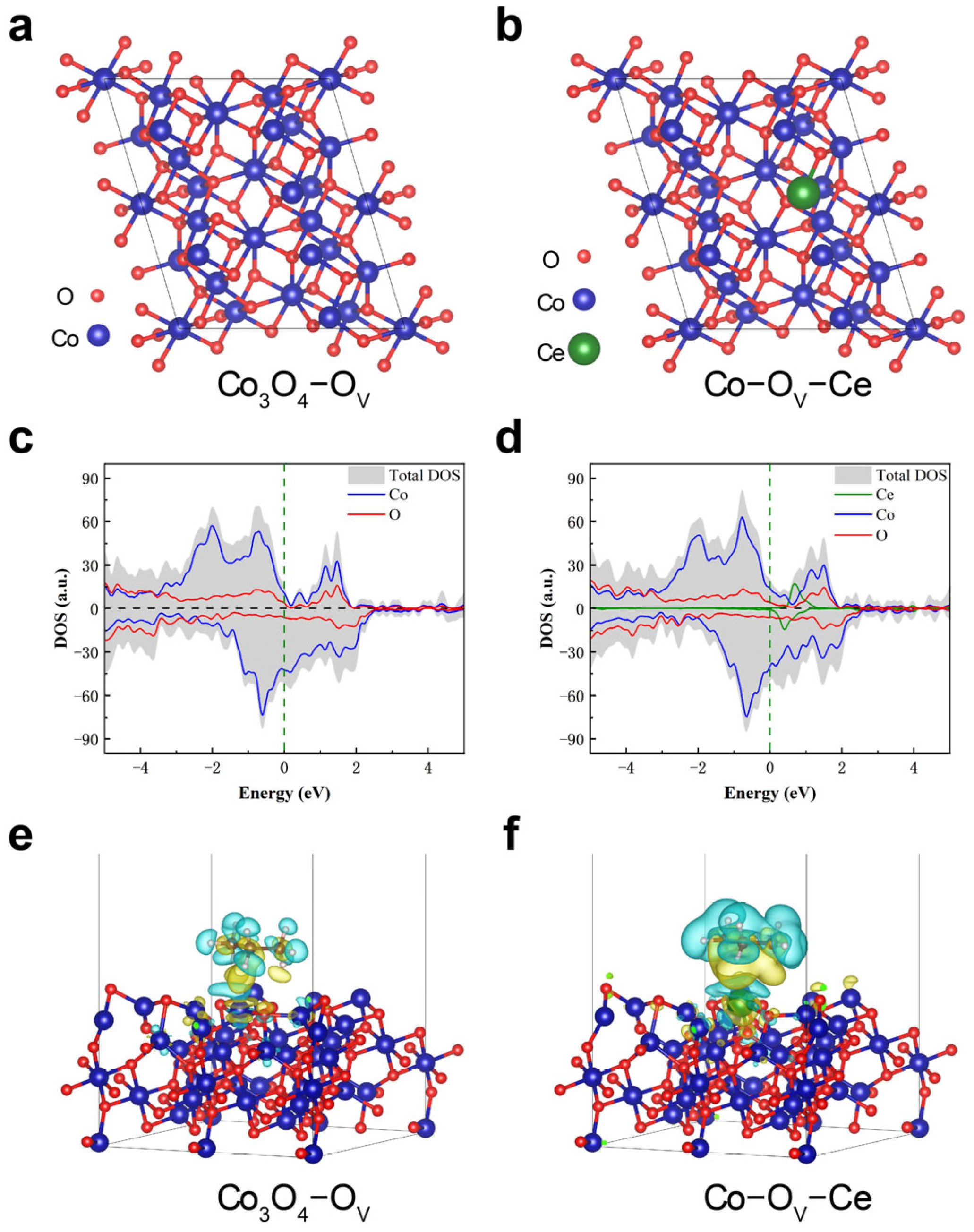
2.4. Catalytic Mechanism and Density−Functional Theory (DFT) Calculations
3. Discussion
4. Materials and Methods
4.1. Catalyst Preparation
4.1.1. Preparation of Co(OH)2
4.1.2. Preparation of Co3O4@CeO2–IE Catalyst
4.1.3. Preparation of Co3O4/CeO2–IM Catalyst
4.2. Characterizations
4.3. Catalyst Evaluation
4.4. Computational Details
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Mellouki, A.; Wallington, T.J.; Chen, J. Atmospheric chemistry of oxygenated volatile organic compounds: Impacts on air quality and climate. Chem. Rev. 2015, 115, 3984–4014. [Google Scholar] [CrossRef]
- Ravi Kumar, Y.; Deshmukh, K.; Kovářík, T.; Khadheer Pasha, S.K. A systematic review on 2D materials for volatile organic compound sensing. Coordin. Chem. Rev. 2022, 461, 214502. [Google Scholar] [CrossRef]
- He, C.; Cheng, J.; Zhang, X.; Douthwaite, M.; Pattisson, S.; Hao, Z. Recent Advances in the Catalytic Oxidation of Volatile Organic Compounds: A Review Based on Pollutant Sorts and Sources. Chem. Rev. 2019, 119, 4471–4568. [Google Scholar] [CrossRef]
- Li, P.; Kim, S.; Jin, J.; Do, H.C.; Park, J.H. Efficient photodegradation of volatile organic compounds by iron-based metal-organic frameworks with high adsorption capacity. Appl. Catal. B Environ. 2020, 263, 118284. [Google Scholar] [CrossRef]
- Yin, F.; Yue, W.; Li, Y.; Gao, S.; Zhang, C.; Kan, H.; Niu, H.; Wang, W.; Guo, Y. Carbon–based nanomaterials for the detection of volatile organic compounds: A review. Carbon 2021, 180, 274–297. [Google Scholar] [CrossRef]
- Khatib, M.; Haick, H. Sensors for Volatile Organic Compounds. ACS Nano 2022, 16, 7080–7115. [Google Scholar] [CrossRef]
- Nadjafi, M.; Abdala, P.M.; Verel, R.; Hosseini, D.; Safonova, O.V.; Fedorov, A.; Müller, C.R. Reducibility and Dispersion Influence the Activity in Silica-Supported Vanadium-Based Catalysts for the Oxidative Dehydrogenation of Propane: The Case of Sodium Decavanadate. ACS Catal. 2020, 10, 2314–2321. [Google Scholar] [CrossRef]
- Zhang, M.; Sui, X.; Zhang, X.; Niu, M.; Li, C.; Wan, H.; Qiao, Z.-A.; Xie, H.; Li, X. Multi-defects engineering of NiCo2O4 for catalytic propane oxidation. Appl. Surf. Sci. 2022, 600, 154040. [Google Scholar] [CrossRef]
- Liotta, L.F. Catalytic oxidation of volatile organic compounds on supported noble metals. Appl. Catal. B Environ. 2010, 100, 403–412. [Google Scholar] [CrossRef]
- Scirè, S.; Liotta, L.F. Supported gold catalysts for the total oxidation of volatile organic compounds. Appl. Catal. B Environ. 2012, 125, 222–246. [Google Scholar] [CrossRef]
- Wang, B.; Wu, X.; Ran, R.; Si, Z.; Weng, D. IR characterization of propane oxidation on Pt/CeO2–ZrO2: The reaction mechanism and the role of Pt. J. Mol. Catal. A 2012, 356, 100–105. [Google Scholar] [CrossRef]
- Chen, J.; Lv, X.; Xu, W.; Li, X.; Chen, J.; Jia, H. Utilizing Cl coordination to facilitate Ru–Ag self–assembling into alloy and recover thermally-inactivated catalyst for propane combustion. Appl. Catal. B Environ. 2021, 290, 119989. [Google Scholar] [CrossRef]
- Chen, K.; Li, W.; Zhu, C.; Yuan, L. Construction of Hollow Cobalt Tetroxide Nanocages through the Metal Salt Bifunctional Etching Strategy for Catalytic Oxidation of Propane at Ultrahigh Space Velocity. ACS Appl. Nano Mater. 2022, 5, 6575–6584. [Google Scholar] [CrossRef]
- Li, W.; Wang, J.; Gong, H. Catalytic Combustion of VOCs on Non-noble Metal Catalysts. Catal. Today 2009, 148, 81–87. [Google Scholar] [CrossRef]
- Feng, C.; Jiang, F.; Xiong, G.; Chen, C.; Wang, Z.; Pan, Y.; Fei, Z.; Lu, Y.; Li, X.; Zhang, R.; et al. Revelation of Mn4+–Osur–Mn3+ active site and combined Langmuir-Hinshelwood mechanism in propane total oxidation at low temperature over MnO2. Chem. Eng. J. 2023, 451, 138868. [Google Scholar] [CrossRef]
- Sun, L.; Liang, X.; Liu, H.; Cao, H.; Liu, X.; Jin, Y.; Li, X.; Chen, S.; Wu, X. Activation of Co–O bond in (110) facet exposed Co3O4 by Cu doping for the boost of propane catalytic oxidation. J. Hazard Mater. 2023, 452, 131319. [Google Scholar] [CrossRef]
- Chen, K.; Li, W.; Zhou, Z.; Huang, Q.; Liu, Y.; Duan, Q. Hydroxyl groups attached to Co2+ on the surface of Co3O4: A promising structure for propane catalytic oxidation. Catal. Sci. Technol. 2020, 10, 2573–2582. [Google Scholar] [CrossRef]
- Wu, S.; Liu, H.; Huang, Z.; Xu, H.; Shen, W. O-vacancy-rich porous MnO2 nanosheets as highly efficient catalysts for propane catalytic oxidation. Appl. Catal. B Environ. 2022, 312, 121387. [Google Scholar] [CrossRef]
- Xiao, Z.; Huang, Y.C.; Dong, C.L.; Xie, C.; Liu, Z.; Du, S.; Chen, W.; Yan, D.; Tao, L.; Shu, Z.; et al. Operando Identification of the Dynamic Behavior of Oxygen Vacancy-Rich Co3O4 for Oxygen Evolution Reaction. J. Am. Chem. Soc. 2020, 142, 12087–12095. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, Y.; Feng, Y.; Cheng, C.Q.; Qiu, W.K.; Dong, C.K.; Liu, H.; Du, X.W. Co3O4 Nanoparticles with Ultrasmall Size and Abundant Oxygen Vacancies for Boosting Oxygen Involved Reactions. Adv. Fun. Mater. 2019, 29, 1903444. [Google Scholar] [CrossRef]
- Hu, J.; Zeng, X.; Wang, G.; Qian, B.; Liu, Y.; Hu, X.; He, B.; Zhang, L.; Zhang, X. Modulating mesoporous Co3O4 hollow nanospheres with oxygen vacancies for highly efficient peroxymonosulfate activation. Chem. Eng. J. 2020, 400, 125869. [Google Scholar] [CrossRef]
- Iablokov, V.; Barbosa, R.; Pollefeyt, G.; Van Driessche, I.; Chenakin, S.; Kruse, N. Catalytic CO Oxidation over Well-Defined Cobalt Oxide Nanoparticles: Size-Reactivity Correlation. ACS Catal. 2015, 5, 5714–5718. [Google Scholar] [CrossRef]
- Sun, H.; Ang, H.M.; Tadé, M.O.; Wang, S. Co3O4 nanocrystals with predominantly exposed facets: Synthesis, environmental and energy applications. J. Mater. Chem. A 2013, 1, 14427–14442. [Google Scholar] [CrossRef]
- Xie, X.; Li, Y.; Liu, Z.Q.; Haruta, M.; Shen, W.-J. Low-temperature oxidation of CO catalysed by Co3O4 nanorods. Nature 2009, 458, 746–749. [Google Scholar] [CrossRef] [PubMed]
- Tompkins, F.C. Superficial Chemistry and Solid Imperfections. Nature 1960, 186, 3–6. [Google Scholar] [CrossRef]
- Li, H.; Xiao, Z.; Liu, P.; Wang, H.; Geng, J.; Lei, H.; Zhuo, O. Interfaces and Oxygen Vacancies-Enriched Catalysts Derived from Cu-Mn-Al Hydrotalcite towards High-Efficient Water–Gas Shift Reaction. Molecules 2023, 28, 1522. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Loh, H.; Low, B.Q.L.; Zhu, H.; Low, J.; Heng, J.Z.X.; Tang, K.Y.; Li, Z.; Loh, X.J.; Ye, E.; et al. Role of oxygen vacancy in metal oxides for photocatalytic CO2 reduction. Appl. Catal. B Environ. 2023, 321, 122079. [Google Scholar] [CrossRef]
- Zhu, K.; Shi, F.; Zhu, X.; Yang, W. The roles of oxygen vacancies in electrocatalytic oxygen evolution reaction. Nano Energy 2020, 73, 104761. [Google Scholar] [CrossRef]
- Zhang, G.; Zhou, Y.; Yang, Y.; Kong, T.; Song, Y.; Zhang, S.; Zheng, H. Elucidating the Role of Surface Ce4+ and Oxygen Vacancies of CeO2 in the Direct Synthesis of Dimethyl Carbonate from CO2 and Methanol. Molecules 2023, 28, 3785. [Google Scholar] [CrossRef]
- Su, Z.; Yang, W.; Wang, C.; Xiong, S.; Cao, X.; Peng, Y.; Si, W.; Weng, Y.; Xue, M.; Li, J. Roles of Oxygen Vacancies in the Bulk and Surface of CeO2 for Toluene Catalytic Combustion. Environ. Sci. Technol. 2020, 54, 12684–12692. [Google Scholar] [CrossRef]
- Yang, M.; Shen, G.; Wang, Q.; Deng, K.; Liu, M.; Chen, Y.; Gong, Y.; Wang, Z. Roles of Oxygen Vacancies of CeO2 and Mn-Doped CeO2 with the Same Morphology in Benzene Catalytic Oxidation. Molecules 2021, 26, 6363. [Google Scholar] [CrossRef]
- Lou, B.; Shakoor, N.; Adeel, M.; Zhang, P.; Huang, L.; Zhao, Y.; Zhao, W.; Jiang, Y.; Rui, Y. Catalytic oxidation of volatile organic compounds by non-noble metal catalyst: Current advancement and future prospectives. J. Clean. Prod. 2022, 363, 132523. [Google Scholar] [CrossRef]
- Singha, R.K.; Shukla, A.; Yadav, A.; Sivakumar Konathala, L.N.; Bal, R. Effect of Metal-Support Interaction on Activity and Stability of Ni-CeO2 Catalyst for Partial Oxidation of Methane. Appl. Catal. B Environ. 2017, 202, 473–488. [Google Scholar] [CrossRef]
- Wei, X.; Wang, X.; Pu, Y.; Liu, A.; Chen, C.; Zou, W.; Zheng, Y.; Huang, J.; Zhang, Y.; Yang, Y.; et al. Facile Ball-Milling Synthesis of CeO2/g-C3N4 Z-Scheme Heterojunction for Synergistic Adsorption and Photodegradation of Methylene Blue: Characteristics, Kinetics, Models, and Mechanisms. Chem. Eng. J. 2021, 420, 127719. [Google Scholar] [CrossRef]
- Jiang, F.; Wang, S.; Liu, B.; Liu, J.; Wang, L.; Xiao, Y.; Xu, Y.; Liu, X. Insights into the Influence of CeO2 Crystal Facet on CO2 Hydrogenation to Methanol over Pd/CeO2 Catalysts. ACS Catal. 2020, 10, 11493–11509. [Google Scholar] [CrossRef]
- Chang, S.; Li, M.; Hua, Q.; Zhang, L.; Ma, Y.; Ye, B.; Huang, W. Shape-Dependent Interplay Between Oxygen Vacancies and Ag–CeO2 Interaction in Ag/CeO2 Catalysts and Their Influence on the Catalytic Activity. J. Catal. 2012, 293, 195–204. [Google Scholar] [CrossRef]
- Han, X.; Li, J.; Lu, J.; Luo, S.; Wan, J.; Li, B.; Hu, C.; Cheng, X. High mass-loading NiCoLDH nanosheet arrays grown on carbon cloth by electrodeposition for excellent electrochemical energy storage. Nano Energy 2021, 86, 106079. [Google Scholar] [CrossRef]
- Mo, S.; Zhang, Q.; Li, S.; Ren, Q.; Zhang, M.; Xue, Y.; Peng, R.; Xiao, H.; Chen, Y.; Ye, D. Integrated Cobalt Oxide Based Nanoarray Catalysts with Hierarchical Architectures: In Situ Raman Spectroscopy Investigation on the Carbon Monoxide Reaction Mechanism. ChemCatChem 2018, 10, 3012–3026. [Google Scholar] [CrossRef]
- Spanier, J.E.; Robinson, R.D.; Zhang, F.; Chan, S.-W.; Herman, I.P. Size-Dependent Properties of CeO2−y Nanoparticles as Studied by Raman Scattering. Phys. Rev. B 2001, 64, 245407–245414. [Google Scholar] [CrossRef]
- Lin, J.; Li, L.; Huang, Y.; Zhang, W.; Wang, X.; Wang, A.; Zhang, T. In Situ Calorimetric Study: Structural Effects on Adsorption and Catalytic Performances for CO Oxidation over Ir-in-CeO2 and Ir-on-CeO2 Catalysts. J. Phys. Chem. C 2011, 115, 16509–16517. [Google Scholar] [CrossRef]
- Chen, L.; Liu, F.; Li, X.; Tao, Q.; Huang, Z.; Zuo, Q.; Chen, Y.; Li, T.; Fu, M.; Ye, D. Surface adsorbed and lattice oxygen activated by the CeO2/Co3O4 interface for enhancive catalytic soot combustion: Experimental and theoretical investigations. J. Colloid Interface Sci. 2023, 638, 109–122. [Google Scholar] [CrossRef]
- Wu, S.; Liu, H.; Huang, Z.; Xu, H.; Shen, W. Mn1ZrxOy mixed oxides with abundant oxygen vacancies for propane catalytic oxidation: Insights into the contribution of Zr doping. Chem. Eng. J. 2023, 452, 139341. [Google Scholar] [CrossRef]
- Zhu, W.; Chen, X.; Jin, J.; Di, X.; Liang, C.; Liu, Z. Insight into catalytic properties of Co3O4-CeO2 binary oxides for propane total oxidation. Chin. J. Catal. 2020, 41, 679–690. [Google Scholar] [CrossRef]
- Maintz, S.; Deringer, V.; Tchougreeff, A.; Dronskowski, R. LOBSTER: A Tool to Extract Chemical Bonding from Plane-Wave Based DFT. J. Comput. Chem. 2016, 37, 1030–1035. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Zhan, Q.; Liu, F.; Wang, C.; Li, H.; Wang, X.; Guo, X.; Cheng, Y.; Sun, W.; Wang, L.; et al. Overturned Loading of Inert CeO2 to Active Co3O4 for Unusually Improved Catalytic Activity in Fenton-Like Reactions. Angew. Chem. Int. Ed. 2022, 61, e202200406. [Google Scholar] [CrossRef]
- Wang, X.; Song, H.; Ma, S.; Li, M.; He, G.; Xie, M.; Guo, X. Template ion-exchange synthesis of Co-Ni composite hydroxides nanosheets for supercapacitor with unprecedented rate capability. Chem. Eng. J. 2021, 432, 134319. [Google Scholar] [CrossRef]
- Giannozzi, P.; Baroni, S.; Bonini, N.; Calandra, M.; Car, R.; Cavazzoni, C.; Ceresoli, D.; Chiarotti, G.L.; Cococcioni, M.; Dabo, I.; et al. QUANTUM ESPRESSO: A modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 2009, 21, 395502. [Google Scholar] [CrossRef] [PubMed]
- Giannozzi, P.; Andreussi, O.; Brumme, T.; Bunau, O.; Buongiorno Nardelli, M.; Calandra, M.; Car, R.; Cavazzoni, C.; Ceresoli, D.; Cococcioni, M.; et al. Advanced capabilities for materials modelling with Quantum ESPRESSO. J. Phys. Condens. Matter 2017, 29, 465901. [Google Scholar] [CrossRef] [PubMed]
- Van Setten, M.J.; Giantomassi, M.; Bousquet, E.; Verstraete, M.J.; Hamann, D.R.; Gonze, X.; Rignanese, G.M. The PseudoDojo: Training and grading a 85 element optimized norm-conserving pseudopotential table. Comput. Phys. Commun. 2018, 226, 39–54. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Liang, W.; Lin, C.; Zhang, T.; Zhang, J.; Sheng, N.; Song, Z.; Jiang, J.; Sun, B.; Xu, W. Enabling High Activity Catalyst Co3O4@CeO2 for Propane Catalytic Oxidation via Inverse Loading. Molecules 2023, 28, 5930. https://doi.org/10.3390/molecules28155930
Wang X, Liang W, Lin C, Zhang T, Zhang J, Sheng N, Song Z, Jiang J, Sun B, Xu W. Enabling High Activity Catalyst Co3O4@CeO2 for Propane Catalytic Oxidation via Inverse Loading. Molecules. 2023; 28(15):5930. https://doi.org/10.3390/molecules28155930
Chicago/Turabian StyleWang, Xuan, Wei Liang, Changqing Lin, Tie Zhang, Jing Zhang, Nan Sheng, Zhaoning Song, Jie Jiang, Bing Sun, and Wei Xu. 2023. "Enabling High Activity Catalyst Co3O4@CeO2 for Propane Catalytic Oxidation via Inverse Loading" Molecules 28, no. 15: 5930. https://doi.org/10.3390/molecules28155930
APA StyleWang, X., Liang, W., Lin, C., Zhang, T., Zhang, J., Sheng, N., Song, Z., Jiang, J., Sun, B., & Xu, W. (2023). Enabling High Activity Catalyst Co3O4@CeO2 for Propane Catalytic Oxidation via Inverse Loading. Molecules, 28(15), 5930. https://doi.org/10.3390/molecules28155930