Nontoxic Levels of Se-Containing Compounds Increase Survival by Blocking Oxidative and Inflammatory Stresses via Signal Pathways Whereas High Levels of Se Induce Apoptosis
Abstract
1. Introduction
2. Selenium Compounds and Their Metabolism
3. Signaling Pathways and Molecular Targets Pertinent to Biological Selenium
4. Anti-Inflammatory Activity and Immune Functions of Selenium Addition
5. The Role of Selenium in Senescence and Ferroptosis
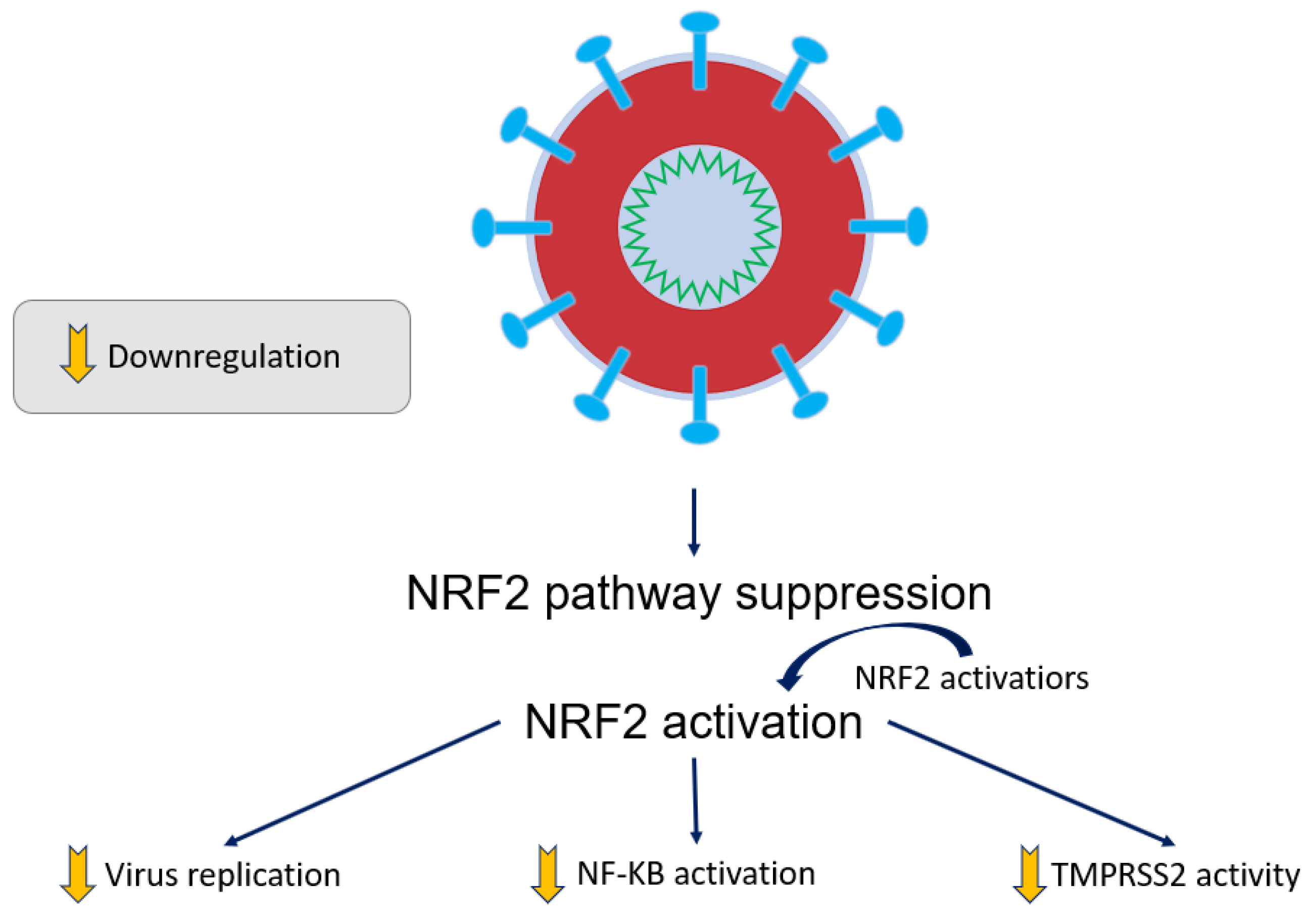
6. Benefits of COVID-19 Se Treatment and Related Sepsis Conditions
7. Clinical Applications of High Doses of Sodium Selenite (Cancer Studies)
8. Conclusions and Outlook
- Medicinal and Small molecule
- There are small molecule chemistry studies that can be tested as cocktails.
- How chemical cocktails of synthetic Se-containing compounds can be prepared (chemical synthesis) and formulations and their synthesis, which contain selenium, are of research interest.
- A host of combinations of both reduced and oxidized forms of selenium can be tested in combination in cocktail form.
- New small synthetic molecules of selenium can be used in biological testing as fluorescent probes in biological studies. The molecules could be natural products in which the selenium is taking the place of the O or S atom, which is an isosteric position.
- How other trace elements or main group elements can be involved in facilitating the treatment of cancer and aging—elements such as fluorine, boron, gallium, etc.—is of interest. Compounds that are fluorescent, phosphorescent, or luminescent can be tested for their efficacy and for their ability to serve as chemosensors with proper selectivity and sensitivity; moreover, their biological activity can be determined.
- A deeper understanding of tellurium-containing compounds in which the tellurium center is isosteric with that of selenium and sulfur is of importance because the tellurium stays away from sulfur metabolism because Te is comparatively more metallic. The understanding of tellurium compounds will impact further understanding of selenium-containing compounds and their capacity. As a warning, the Te–C bonds are weaker, and, therefore, the research is more challenging.
- Biophysical and Pharmacological
- The field shall require more biophysical chemistry studies, protein-related studies in the form of mutation studies with selenium-containing proteins, and novel unnatural selenium-containing AAs.
- As mentioned above, the assessment of the pharmacokinetics of new compounds will be essential and helps quantify the performance of the compounds.
- Biology and Biochemistry
- Further exploration of the (side) effects of Se and Se-containing small molecules on biology is required. One goal of these medicinal studies would be to check the relationship between the induction of Akt activity and the reduction of the ASK1 complex.
- The pro/antioxidant effect of selenite molecular action can be better defined and more clearly researched under a variety of biological conditions.
- How metabolic engineering can be used to help prepare further synthetic compounds of selenium is an important branch of this study.
- Clinical and Animal Testing
- It will be incredibly important to carry out further in vivo studies of MSeC.
- MSeC studies that can help assess the maximum tolerable dose and the half-life of the concentration of MSeC in human biology will be greatly welcomed by the community.
- Because MSeC is commercially available as a readily available over-the-counter supplement form, the application of efficacy of treating cancer in humans with this chemical would be an interesting investigation.
- As mentioned above, the NRF2 activators and Keap1 modification incurred by NF-κB activation would be a great focus of study. An exact understanding of selenium involvement in these processes would be important to investigate to ascertain whether selenium-containing compounds may be involved in altering NRF2 activity; this could be achieved by reducing NF-κB or modifying Keap1.
- GPX3 from the blood of cancer patients should be monitored, as well as the influence of selenium supplementation can be better defined in clinical studies.
- During the hospital stays (intensive care unit stay duration), the duration of ventilation by mechanical means and the overall survival rate were not altered by selenium intervention so far. Therefore, the possible positive clinical effectiveness can be observed upon completing, summarizing, and analyzing more clinical trials when they become available [105].
- High research standards will have to be involved in intervention studies that better scrutinize a proposed underlying feed-forward mechanism because of our observed association of Se deficiency with mortality risk. Safety and limited expenses are suitable for adjuvant treatments.
- Se supplementation with patients enduring ARDS can be sought, as well as other clinical applications which might be applicable and accessible. Hypothetical mechanisms can be proposed for Se supplements that would then prevent COVID-19 [123].
- Environmental
- The effect on human health (possibly positive) of the prevalence of selenium-containing material in common environmental and commercial forms such as CdSe can be explored. In particular, there are different forms (crystal forms, bulk, and nanocrystalline) of, e.g., CdSe, that could be studied for their biological activity. Their ability and mechanism to enter the biological system and the rate at which it can do it and also later be eliminated would be important biological data for the community.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAs | Amino acids |
| AIDS | Acquired immune deficiency syndrome |
| Akt | Ak strain transforming |
| ALL | Acute lymphoblastic leukemia |
| AML | Acute myeloid leukemia |
| ARDS | Acute respiratory distress syndrome |
| ARE | Antioxidant response element |
| ASK1 | Apoptosis signal-regulating kinase 1 |
| ATM | Ataxia telangiectasia mutated |
| ATP | Adenosine triphosphate |
| Bcl-2 | B-cell lymphoma 2 |
| CD4+T | Cluster of differentiation 4-positive T lymphocyte |
| CD8+ | Cluster of differentiation 8-positive |
| COPD | Chronic obstructive pulmonary disease |
| COVID-19 | Coronavirus disease 2019 |
| CRL-1790 | Line of normal colon fibroblasts |
| CT | Computed tomography |
| Cys | Cysteine |
| DMF | Dimethyl fumarate |
| DNA | Deoxyribonucleic acid |
| EXM | Extracellular matrix |
| FN | Fibronectin |
| FRAP | Ferric-reducing antioxidant power |
| GPX | Glutathione peroxidase |
| GPX1 | Glutathione peroxidase 1 |
| GPX3 | Glutathione peroxidase 3 |
| GPX4 | Glutathione peroxidase 4 |
| GSH | Glutathione (γ-Glutamylcysteinylglycine) |
| HCT-116 | Line of colon cancer cells |
| H2Se | Hydrogen selenide |
| HT1080 | HT-1080 human cells |
| IFN | Interferon |
| IgG | Immunoglobulin G |
| IκBα | Nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor |
| IL-2 | Interleukin-2 |
| JNK | Jun N-terminal kinase |
| Keap1 | Kelch-like ECH-associated protein 1 |
| MMP-2 | Matrix metalloproteinase-2 |
| MMP-9 | Matrix metalloproteinase-9 |
| MRC-5 | Line of normal lung fibroblasts |
| MSeA | Methylseleninic acid |
| MSeC | Methylselenocysteine |
| MSeCN | Methylselenocyanate |
| MSeH | Methylselenol |
| MT1-MMP | Membrane-type I matrix metalloproteinase |
| NAC | N-Acetylcysteine |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| NFAT | Nuclear factor of activated T cells |
| NF-κB | Nuclear factor kappa B |
| NOX2 | NADPH oxidase 2 |
| NRF2 | Nuclear factor erythroid 2-related factor 2 |
| 8-OHdG | 8-Hydroxy-2′-deoxyguanosine |
| PARP | Poly (ADP-ribose) polymerase |
| PI3K | Phosphoinositide 3-kinase |
| PC-3 | Line of prostate cancer cells |
| PDI | Protein disulfide isomerase |
| p65 | Transcription factor p65 |
| ROS | Reactive oxygen species |
| S/G2 Phase | S Phase/G2 Phase |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| SeM | Selenomethionine |
| SeM-METase | Methioninase |
| TCR | T-cell receptor |
| TM | Transaction Monitoring |
| TMPRSS2 | Transmembrane serine protease 2 |
| Trx | Thioredoxin |
| TrxR | Thioredoxin reductase |
| VN | Vitronectin |
References
- Marshall, J.R. Selenium in cancer prevention: Did we move too quickly? In Selenium in the Environment and Human Health; Banuelos, G.S., Lin, Z.-Q., Yin, X., Eds.; Taylor and Francis Group: London, UK, 2013; Volume 55, pp. 55–56. [Google Scholar]
- Kim, S.J.; Choi, M.C.; Park, J.M.; Chung, A.S. Antitumor Effects of Selenium. Int. J. Mol. Sci. 2021, 22, 11844. [Google Scholar] [CrossRef]
- Harvard, T.H. Chan School of Public Health. Available online: https://www.hsph.harvard.edu/nutritionsource/selenium/ (accessed on 30 June 2023).
- Mayo Foundation for Medical Education and Research (MFMER). Available online: https://www.mayoclinic.org/drugs-supplements/selenium-supplement-oral-route/description/drg-20063649 (accessed on 30 June 2023).
- Simon, H.U.; Haj-Yehia, A.; Levi-Schaffer, F. Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 2000, 5, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.-O.; Kim, M.-M.; Park, S.-J.; Kim, D.; Chung, J.; Chung, A.-S. Selenite suppresses hydrogen peroxide-induced cell apoptosis through inhibition of ASK1/JNK and activation of PI3-K/Akt pathways. FASEB J. 2002, 16, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Mthembu, S.N.; Sharma, A.; Albericio, F.; de la Torre, B.G. Breaking a couple: Disulfide reducing agents. ChemBioChem 2020, 21, 1947–1954. [Google Scholar] [CrossRef] [PubMed]
- Burk, R.F.; Hill, K.E. Regulation of selenium metabolism and transport. Annu. Rev. Nutr. 2015, 35, 109–134. [Google Scholar] [CrossRef] [PubMed]
- Spallholz, J.E. On the nature of selenium toxicity and carcinostatic activity. Free Radic. Biol. Med. 1994, 17, 45–64. [Google Scholar] [CrossRef]
- Labunskyy, V.M.; Hatfield, D.L.; Gladyshev, V.N. Selenoproteins: Molecular pathways and physiological roles. Physiol. Rev. 2014, 94, 739–777. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; McIntosh, G.H.; Young, G.P. Selenium-rich foods: A promising approach to colorectal cancer prevention. Curr. Pharm. Biotechnol. 2012, 13, 165–172. [Google Scholar] [CrossRef]
- Rayman, M.P.; Infante, H.G.; Sargent, M. Food-chain selenium and human health: Spotlight on speciation. Br. J. Nutr. 2008, 100, 238–253. [Google Scholar] [CrossRef]
- Fernandes, A.P.; Wallenberg, M.; Gandin, V.; Misra, S.; Tisato, F.; Marzano, C.; Rigobello, M.P.; Kumar, S.; Björnstedt, M. Methylselenol Formed by Spontaneous Methylation of Selenide Is a Superior Selenium Substrate to the Thioredoxin and Glutaredoxin Systems. PLoS ONE 2012, 7, e50727. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Jung, U.; Cho, D.-Y.; Chung, A.-S. Se-Methylselenocysteine induces apoptosis through caspase activation in HL-60 cells. Carcinogenesis 2001, 22, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Jung, U.; Zheng, X.; Yoon, S.-O.; Chung, A.-S. Se-Methylselenocysteine induces apoptosis mediated by reactive oxygen species in HL-60 cells. Free Radic. Biol. Med. 2001, 31, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Jung, J.-Y.; Son, M.; Lee, S.-H.; Lim, J.-S.; Chung, A.-S. Long exposure of non-cytotoxic concentrations of methylselenol suppresses the invasive potential of B16F10 melanoma. Oncol. Rep. 2008, 20, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Lu, J. Apoptosis and angiogenesis in cancer prevention by selenium. Adv. Exp. Med. Biol. 2001, 492, 131–146. [Google Scholar] [PubMed]
- Lu, J.; Pei, H.; Ip, C.; Lisk, D.J.; Ganther, H.; Thompson, H.J. Effect of an aqueous extract of selenium-enriched garlic on in vitro markers and in vivo efficacy in cancer prevention. Carcinogenesis 1996, 17, 1903–1907. [Google Scholar] [CrossRef]
- Kaeck, M.; Lu, J.; Strange, R.; Ip, C.; Ganther, H.E.; Thompson, H.J. Differential induction of growth arrest inducible genes by selenium compounds. Biochem. Pharmacol. 1997, 53, 921–926. [Google Scholar] [CrossRef]
- Zhong, W.; Oberley, T.D. Redox-mediated Effects of Selenium on Apoptosis and Cell Cycle in the LNCaP Human Prostate Cancer Cell Line1. Cancer Res. 2001, 61, 7071–7078. [Google Scholar]
- Lu, J.; Kaeck, M.; Jiang, C.; Wilson, A.C.; Thompson, H.J. Selenite induction of DNA strand breaks and apoptosis in mouse leukemic L1210 cells. Biochem. Pharmacol. 1994, 47, 1531–1535. [Google Scholar] [CrossRef]
- Zhu, Z.; Kimura, M.; Itokawa, Y.; Aoki, T.; Takahashi, J.A.; Nakatsu, S.; Oda, Y.; Kikuchi, H. Apoptosis induced by selenium in human glioma cell lines. Biol. Trace Elem. Res. 1996, 54, 123–134. [Google Scholar] [CrossRef]
- Stewart, M.S.; Davis, R.L.; Walsh, L.P.; Pence, B.C. Induction of differentiation and apoptosis by sodium selenite in human colonic carcinoma cells (HT29). Cancer Lett. 1997, 117, 35–40. [Google Scholar] [CrossRef]
- Shen, H.-M.; Yang, C.-F.; Ong, C.-N. Sodium selenite-induced oxidative stress and apoptosis in human hepatoma HepG2 cells. Int. J. Cancer 1999, 81, 820–828. [Google Scholar] [CrossRef]
- Yoon, S.-O.; Kim, M.-M.; Chung, A.-S. Inhibitory Effect of Selenite on Invasion of HT1080 Tumor Cells*. J. Biol. Chem. 2001, 276, 20085–20092. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Wang, Z.; Ganther, H.; Lü, J. Distinct Effects of Methylseleninic Acid versus Selenite on Apoptosis, Cell Cycle, and Protein Kinase Pathways in DU145 Human Prostate Cancer Cells1. Mol. Cancer Ther. 2002, 1, 1059–1066. [Google Scholar]
- Wang, Z.; Jiang, C.; Ganther, H.; Lü, J. Antimitogenic and Proapoptotic Activities of Methylseleninic Acid in Vascular Endothelial Cells and Associated Effects on PI3K-AKT, ERK, JNK and p38 MAPK Signaling1. Cancer Res. 2001, 61, 7171–7178. [Google Scholar] [PubMed]
- Jiang, C.; Wang, Z.; Ganther, H.; Lu, J. Caspases as Key Executors of Methyl Selenium-induced Apoptosis (Anoikis) of DU-145 Prostate Cancer Cells1. Cancer Res. 2001, 61, 3062–3070. [Google Scholar]
- Ip, C.; Ganther, H.E. Comparison of selenium and sulfur analogs in cancer prevention. Carcinogenesis 1992, 13, 1167–1170. [Google Scholar] [CrossRef]
- Lü, J.; Jiang, C. Selenium and Cancer Chemoprevention: Hypotheses Integrating the Actions of Selenoproteins and Selenium Metabolites in Epithelial and Non-Epithelial Target Cells. Antioxid. Redox Signal. 2005, 7, 1715–1727. [Google Scholar] [CrossRef]
- Varlamova, E.G.; Turovsky, E.A. The main cytotoxic effects of methylseleninic acid on various cancer cells. Int. J. Mol. Sci. 2021, 22, 6614. [Google Scholar] [CrossRef]
- Hariharan, S.; Dharmaraj, S. Selenium and selenoproteins: It’s role in regulation of inflammation. Inflammopharmacology 2020, 28, 667–695. [Google Scholar] [CrossRef]
- Álvarez-Pérez, M.; Ali, W.; Marć, M.A.; Handzlik, J.; Domínguez-Álvarez, E. Selenides and Diselenides: A Review of Their Anticancer and Chemopreventive Activity. Molecules 2018, 23, 628. [Google Scholar] [CrossRef]
- Lennicke, C.; Rahn, J.; Bukur, J.; Hochgräfe, F.; Wessjohann, L.A.; Lichtenfels, R.; Seliger, B. Modulation of MHC class I surface expression in B16F10 melanoma cells by methylseleninic acid. OncoImmunology 2017, 6, e1259049. [Google Scholar] [CrossRef] [PubMed]
- Wiesner-Reinhold, M.; Schreiner, M.; Baldermann, S.; Schwarz, D.; Hanschen, F.S.; Kipp, A.P.; Rowan, D.D.; Bentley-Hewitt, K.L.; McKenzie, M.J. Mechanisms of Selenium Enrichment and Measurement in Brassicaceous Vegetables, and Their Application to Human Health. Front. Plant Sci. 2017, 8, 1365. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Jiang, C.; Kaeck, M.; Ganther, H.; Vadhanavikit, S.; Clement, I.; Thompson, H. Dissociation of the genotoxic and growth inhibitory effects of selenium. Biochem. Pharmacol. 1995, 50, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Clement, I.; Vadhanavikit, S.; Ganther, H. Cancer chemoprevention by aliphatic selenocyanates: Effect of chain length on inhibition of mammary tumors and DMBA adducts. Carcinogenesis 1995, 16, 35–38. [Google Scholar] [CrossRef]
- Ip, C.; Zhu, Z.; Thompson, H.J.; Lisk, D.; Ganther, H.E. Chemoprevention of mammary cancer with Se-allylselenocysteine and other selenoamino acids in the rat. Anticancer Res. 1999, 19, 2875–2880. [Google Scholar]
- Wang, Z.; Jiang, C.; Lü, J. Induction of caspase-mediated apoptosis and cell-cycle G1 arrest by selenium metabolite methylselenol. Mol. Carcinog. 2002, 34, 113–120. [Google Scholar] [CrossRef]
- Drake, E.N. Cancer chemoprevention: Selenium as a prooxidant, not an antioxidant. Med. Hypotheses 2006, 67, 318–322. [Google Scholar] [CrossRef]
- Wallenberg, M.; Misra, S.; Björnstedt, M. Selenium Cytotoxicity in Cancer. Basic Clin. Pharmacol. Toxicol. 2014, 114, 377–386. [Google Scholar] [CrossRef]
- Francesconi, K.A.; Pannier, F. Selenium Metabolites in Urine: A Critical Overview of Past Work and Current Status. Clin. Chem. 2004, 50, 2240–2253. [Google Scholar] [CrossRef]
- Ogra, Y.; Ishiwata, K.; Takayama, H.; Aimi, N.; Suzuki, K.T. Identification of a novel selenium metabolite, Se-methyl-N-acetylselenohexosamine, in rat urine by high-performance liquid chromatography–inductively coupled plasma mass spectrometry and –electrospray ionization tandem mass spectrometry. J. Chromatogr. B 2002, 767, 301–312. [Google Scholar] [CrossRef]
- Barbazán, J.; Alonso-Alconada, L.; Elkhatib, N.; Geraldo, S.; Gurchenkov, V.; Glentis, A.; van Niel, G.; Palmulli, R.; Fernández, B.; Viaño, P.; et al. Liver Metastasis Is Facilitated by the Adherence of Circulating Tumor Cells to Vascular Fibronectin Deposits. Cancer Res. 2017, 77, 3431–3441. [Google Scholar] [CrossRef]
- Kim, A.; Oh, J.-H.; Park, J.-M.; Chung, A.-S. Methylselenol generated from selenomethionine by methioninase downregulates integrin expression and induces caspase-mediated apoptosis of B16F10 melanoma cells. J. Cell. Physiol. 2007, 212, 386–400. [Google Scholar] [CrossRef]
- Park, J.-M.; Kim, A.; Oh, J.-H.; Chung, A.-S. Methylseleninic acid inhibits PMA-stimulated pro-MMP-2 activation mediated by MT1-MMP expression and further tumor invasion through suppression of NF-κB activation. Carcinogenesis 2007, 28, 837–847. [Google Scholar] [CrossRef]
- Johnson, L.L.; Dyer, R.; Hupe, D.J. Matrix metalloproteinases. Curr. Opin. Chem. Biol. 1998, 2, 466–471. [Google Scholar] [CrossRef]
- Johnsen, M.; Lund, L.R.; Rømer, J.; Almholt, K.; Danø, K. Cancer invasion and tissue remodeling: Common themes in proteolytic matrix degradation. Curr. Opin. Cell Biol. 1998, 10, 667–671. [Google Scholar] [CrossRef]
- Liabakk, N.-B.; Talbot, I.; Smith, R.A.; Wilkinson, K.; Balkwill, F. Matrix Metalloprotease 2 (MMP-2) and Matrix Metalloprotease 9 (MMP-9) Type IV Collagenases in Colorectal Cancer. Cancer Res. 1996, 56, 190–196. [Google Scholar] [PubMed]
- Egeblad, M.; Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nat. Rev. Cancer 2002, 2, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Visse, R.; Nagase, H. Matrix Metalloproteinases and Tissue Inhibitors of Metalloproteinases. Circ. Res. 2003, 92, 827–839. [Google Scholar] [CrossRef]
- Sato, H.; Takino, T.; Okada, Y.; Cao, J.; Shinagawa, A.; Yamamoto, E.; Seiki, M. A matrix metalloproteinase expressed on the surface of invasive tumour cells. Nature 1994, 370, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Sternlicht. M., D.; Werb, Z. How Matrix Metalloproteinases Regulate Cell Behavior. Annu. Rev. Cell Dev. Biol. 2001, 17, 463–516. [Google Scholar] [CrossRef]
- Zeng, H.; Briske-Anderson, M.; Idso, J.P.; Hunt, C.D. The Selenium Metabolite Methylselenol Inhibits the Migration and Invasion Potential of HT1080 Tumor Cells. J. Nutr. 2006, 136, 1528–1532. [Google Scholar] [CrossRef] [PubMed]
- Fratta Pasini, A.M.; Stranieri, C.; Cominacini, L.; Mozzini, C. Potential Role of Antioxidant and Anti-Inflammatory Therapies to Prevent Severe SARS-Cov-2 Complications. Antioxidants 2021, 10, 272. [Google Scholar] [CrossRef]
- Sadowska, A.M. N-Acetylcysteine mucolysis in the management of chronic obstructive pulmonary disease. Ther. Adv. Respir. Dis. 2012, 6, 127–135. [Google Scholar] [CrossRef]
- Kensler, T.W.; Wakabayashi, N.; Biswal, S. Cell Survival Responses to Environmental Stresses Via the Keap1-Nrf2-ARE Pathway. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 89–116. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Roche, L.; Mesta, F. Oxidative Stress as Key Player in Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) Infection. Arch. Med. Res. 2020, 51, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Khomich, O.A.; Kochetkov, S.N.; Bartosch, B.; Ivanov, A.V. Redox Biology of Respiratory Viral Infections. Viruses 2018, 10, 392. [Google Scholar] [CrossRef]
- Checconi, P.; De Angelis, M.; Marcocci, M.E.; Fraternale, A.; Magnani, M.; Palamara, A.T.; Nencioni, L. Redox-Modulating Agents in the Treatment of Viral Infections. Int. J. Mol. Sci. 2020, 21, 4084. [Google Scholar] [CrossRef]
- Cecchini, R.; Cecchini, A.L. SARS-CoV-2 infection pathogenesis is related to oxidative stress as a response to aggression. Med. Hypotheses 2020, 143, 110102. [Google Scholar] [CrossRef] [PubMed]
- Komaravelli, N.; Casola, A. Respiratory viral infections and subversion of cellular antioxidant defenses. J. Pharm. Pharm. 2014, 5, 1000141. [Google Scholar]
- Wyler, E.; Franke, V.; Menegatti, J.; Kocks, C.; Boltengagen, A.; Praktiknjo, S.; Walch-Ruckheim, B.; Bosse, J.; Rajewsky, N.; Grasser, F.; et al. Single-cell RNA-sequencing of herpes simplex virus 1-infected 482 cells connects NRF2 activation to an antiviral program. Nat. Commun. 2019, 10, 4878. [Google Scholar] [CrossRef]
- Halder, U.C.; Bagchi, P.; Chattopadhyay, S.; Dutta, D.; Chawla-Sarkar, M. Cell death regulation during influenza A virus infection by matrix (M1) protein: A model of viral control over the cellular survival pathway. Cell Death Dis. 2011, 2, e197. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.S.C.; Lee, N. Adjunctive therapies and immunomodulating agents for severe influenza. Influenza Other Respir. Viruses 2013, 7, 52–59. [Google Scholar] [CrossRef]
- Cuadrado, A.; Martín-Moldes, Z.; Ye, J.; Lastres-Becker, I. Transcription Factors NRF2 and NF-κB Are Coordinated Effectors of the Rho Family, GTP-binding Protein RAC1 during Inflammation*. J. Biol. Chem. 2014, 289, 15244–15258. [Google Scholar] [CrossRef] [PubMed]
- Lastres-Becker, I.; Innamorato, N.G.; Jaworski, T.; Rabano, A.; Kügler, S.; Van Leuven, F.; Cuadrado, A. Fractalkine activates NRF2/NFE2L2 and heme oxygenase 1 to restrain tauopathy-induced microgliosis. Brain 2014, 137, 78–91. [Google Scholar] [CrossRef] [PubMed]
- Boutten, A.; Goven, D.; Artaud-Macari, E.; Boczkowski, J.; Bonay, M. NRF2 targeting: A promising therapeutic strategy in chronic obstructive pulmonary disease. Trends Mol. Med. 2011, 17, 363–371. [Google Scholar] [CrossRef] [PubMed]
- DeDiego, M.L.; Nieto-Torres, J.L.; Regla-Nava, J.A.; Jimenez-Guardeño, J.M.; Fernandez-Delgado, R.; Fett, C.; Castaño-Rodriguez, C.; Perlman, S.; Enjuanes, L. Inhibition of NF-κB-Mediated Inflammation in Severe Acute Respiratory Syndrome Coronavirus-Infected Mice Increases Survival. J. Virol. 2014, 88, 913–924. [Google Scholar] [CrossRef]
- Dosch, S.F.; Mahajan, S.D.; Collins, A.R. SARS coronavirus spike protein-induced innate immune response occurs via activation of the NF-κB pathway in human monocyte macrophages in vitro. Virus Res. 2009, 142, 19–27. [Google Scholar] [CrossRef]
- Cuadrado, A.; Pajares, M.; Benito, C.; Jiménez-Villegas, J.; Escoll, M.; Fernández-Ginés, R.; Garcia Yagüe, A.J.; Lastra, D.; Manda, G.; Rojo, A.I.; et al. Can Activation of NRF2 Be a Strategy against COVID-19? Trends Pharmacol. Sci. 2020, 41, 598–610. [Google Scholar] [CrossRef]
- Broome, C.S.; McArdle, F.; Kyle, J.A.; Andrews, F.; Lowe, N.M.; Hart, C.A.; Arthur, J.R.; Jackson, M.J. An increase in selenium intake improves immune function and poliovirus handling in adults with marginal selenium status. Am. J. Clin. Nutr. 2004, 80, 154–162. [Google Scholar] [CrossRef]
- Bak, D.W.; Gao, J.; Wang, C.; Weerapana, E. A quantitative chemoproteomic platform to monitor selenocysteine reactivity within a complex proteome. Cell Chem. Biol. 2018, 25, 1157–1167.e4. [Google Scholar] [CrossRef]
- Roy, M.; Kiremidjian-Schumacher, L.; Wishe, H.I.; Cohen, M.W.; Stotzky, G. Selenium Supplementation Enhances the Expression of Interleukin 2 Receptor Subunits and Internalization of Interleukin 2. Proc. Soc. Exp. Biol. Med. 1993, 202, 295–301. [Google Scholar] [CrossRef]
- Rusu, P.M.; Chan, A.Y.; Heikenwalder, M.; Müller, O.J.; Rose, A.J. Dietary Essential Amino Acid Restriction Promotes Hyperdipsia via Hepatic FGF21. Nutrients 2021, 13, 1469. [Google Scholar] [CrossRef] [PubMed]
- Spallholz, J.; Boylan, M.; Larsen, H. Micronutrition and immune function. Ann. N. Y. Acad. Sci. 1990, 587, 123–139. [Google Scholar] [CrossRef]
- Peretz, A.; Neve, J.; Duchateau, J.; Famaey, J.P. Adjuvant Treatment of Recent Onset Rheumatoid Arthritis by Selenium Supplementation: Preliminary Observations. Rheumatology 1992, 31, 281–282. [Google Scholar] [CrossRef] [PubMed]
- McCloy, R. Chronic Pancreatitis at Manchester, UK. Digestion 1998, 59 (Suppl. S4), 36–48. [Google Scholar] [CrossRef]
- Shaheen, S.O.; Sterne, J.A.; Thompson, R.L.; Songhurst, C.E.; Margetts, B.M.; Burney, P.G. Dietary antioxidants and asthma in adults: Population-based case–control study. Am. J. Respir. Crit. Care Med. 2001, 164, 1823–1828. [Google Scholar] [CrossRef] [PubMed]
- Holland, S.M. Chronic Granulomatous Disease. Clin. Rev. Allergy Immunol. 2010, 38, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Schäppi, M.G.; Jaquet, V.; Belli, D.C.; Krause, K.-H. Hyperinflammation in chronic granulomatous disease and anti-inflammatory role of the phagocyte NADPH oxidase. Semin. Immunopathol. 2008, 30, 255–271. [Google Scholar] [CrossRef] [PubMed]
- Baker, S.S.; Cohen, H.J. Altered oxidative metabolism in selenium-deficient rat granulocytes. J. Immunol. 1983, 130, 2856–2860. [Google Scholar] [CrossRef]
- Verma, S.; Hoffmann, F.W.; Kumar, M.; Huang, Z.; Roe, K.; Nguyen-Wu, E.; Hashimoto, A.S.; Hoffmann, P.R. Selenoprotein K Knockout Mice Exhibit Deficient Calcium Flux in Immune Cells and Impaired Immune Responses. J. Immunol. 2011, 186, 2127–2137. [Google Scholar] [CrossRef]
- Fomenko, D.E.; Koc, A.; Agisheva, N.; Jacobsen, M.; Kaya, A.; Malinouski, M.; Rutherford, J.C.; Siu, K.-L.; Jin, D.-Y.; Winge, D.R.; et al. Thiol peroxidases mediate specific genome-wide regulation of gene expression in response to hydrogen peroxide. Proc. Natl. Acad. Sci. USA 2011, 108, 2729–2734. [Google Scholar] [CrossRef] [PubMed]
- Veal, E.A.; Day, A.M.; Morgan, B.A. Hydrogen Peroxide Sensing and Signaling. Mol. Cell 2007, 26, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Delaunay, A.; Pflieger, D.; Barrault, M.-B.; Vinh, J.; Toledano, M.B. A Thiol Peroxidase Is an H2O2 Receptor and Redox-Transducer in Gene Activation. Cell 2002, 111, 471–481. [Google Scholar] [CrossRef]
- Hoffmann, F.W.; Hashimoto, A.C.; Shafer, L.A.; Dow, S.; Berry, M.J.; Hoffmann, P.R. Dietary Selenium Modulates Activation and Differentiation of CD4+ T Cells in Mice through a Mechanism Involving Cellular Free Thiols. J. Nutr. 2010, 140, 1155–1161. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, P.R.; Jourdan-Le Saux, C.; Hoffmann, F.W.; Chang, P.S.; Bollt, O.; He, Q.; Tam, E.K.; Berry, M.J. A Role for Dietary Selenium and Selenoproteins in Allergic Airway Inflammation1. J. Immunol. 2007, 179, 3258–3267. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Rose, A.H.; Hoffmann, P.R. The Role of Selenium in Inflammation and Immunity: From Molecular Mechanisms to Therapeutic Opportunities. Antioxid. Redox Signal. 2011, 16, 705–743. [Google Scholar] [CrossRef] [PubMed]
- Avery, J.C.; Hoffmann, P.R. Selenium, selenoproteins, and immunity. Nutrients 2018, 10, 1203. [Google Scholar] [CrossRef]
- Razaghi, A.; Poorebrahim, M.; Sarhan, D.; Björnstedt, M. Selenium stimulates the antitumour immunity: Insights to future research. Eur. J. Cancer 2021, 155, 256–267. [Google Scholar] [CrossRef]
- Kiremidjian-Schumacher, L.; Roy, M.; Wishe, H.; Cohen, M.; Stotzky, G. Selenium and immune cell functions. I. Effect on lymphocyte proliferation and production of interleukin 1 and interleukin 2. Proc. Soc. Exp. Biol. Med. 1990, 193, 136–142. [Google Scholar] [CrossRef]
- Wu, M.; Kang, M.M.; Schoene, N.W.; Cheng, W.-H. Selenium Compounds Activate Early Barriers of Tumorigenesis*. J. Biol. Chem. 2010, 285, 12055–12062. [Google Scholar] [CrossRef]
- Qi, Y.; Schoene, N.W.; Lartey, F.M.; Cheng, W.-H. Selenium Compounds Activate ATM-dependent DNA Damage Response via the Mismatch Repair Protein hMLH1 in Colorectal Cancer Cells*. J. Biol. Chem. 2010, 285, 33010–33017. [Google Scholar] [CrossRef]
- Liu, H.; Forouhar, F.; Seibt, T.; Saneto, R.; Wigby, K.; Friedman, J.; Xia, X.; Shchepinov, M.S.; Ramesh, S.K.; Conrad, M.; et al. Characterization of a patient-derived variant of GPX4 for precision therapy. Nat. Chem. Biol. 2022, 18, 91–100. [Google Scholar] [CrossRef]
- Ingold, I.; Conrad, M. Selenium and iron, two elemental rivals in the ferroptotic death process. Oncotarget 2018, 9, 22241. [Google Scholar] [CrossRef]
- Ingold, I.; Berndt, C.; Schmitt, S.; Doll, S.; Poschmann, G.; Buday, K.; Roveri, A.; Peng, X.; Freitas, F.P.; Seibt, T. Selenium utilization by GPX4 is required to prevent hydroperoxide-induced ferroptosis. Cell 2018, 172, 409–422. [Google Scholar] [CrossRef] [PubMed]
- Cooke, M.S.; Evans, M.D.; Dizdaroglu, M.; Lunec, J. Oxidative DNA damage: Mechanisms, mutation, and disease. FASEB J. 2003, 17, 1195–1214. [Google Scholar] [CrossRef] [PubMed]
- Bjørklund, G.; Zou, L.; Wang, J.; Chasapis, C.T.; Peana, M. Thioredoxin reductase as a pharmacological target. Pharmacol. Res. 2021, 174, 105854. [Google Scholar] [CrossRef]
- Kamal, A.M.; Hamdy, N.M.; Hegab, H.M.; El-Mesallamy, H.O. Expression of thioredoxin-1 (TXN) and its relation with oxidative DNA damage and treatment outcome in adult AML and ALL: A comparative study. Hematology 2016, 21, 567–575. [Google Scholar] [CrossRef]
- Pan, S.; Yang, J.; Ji, S.; Li, T.; Gao, S.; Sun, C.; Xu, H. Cancer therapy by targeting thioredoxin reductase based on selenium-containing dynamic covalent bond. CCS Chem. 2020, 2, 225–235. [Google Scholar] [CrossRef]
- Rossato, M.; Russo, L.; Mazzocut, S.; Di Vincenzo, A.; Fioretto, P.; Vettor, R. Current smoking is not associated with COVID-19. Eur. Respir. J. 2020, 55, 2001290. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Li, R.; Lu, Z.; Huang, Y. Does comorbidity increase the risk of patients with COVID-19: Evidence from meta-analysis. Aging 2020, 12, 6049. [Google Scholar] [CrossRef]
- Song, P.; Li, W.; Xie, J.; Hou, Y.; You, C. Cytokine storm induced by SARS-CoV-2. Clin. Chim. Acta 2020, 509, 280–287. [Google Scholar] [CrossRef] [PubMed]
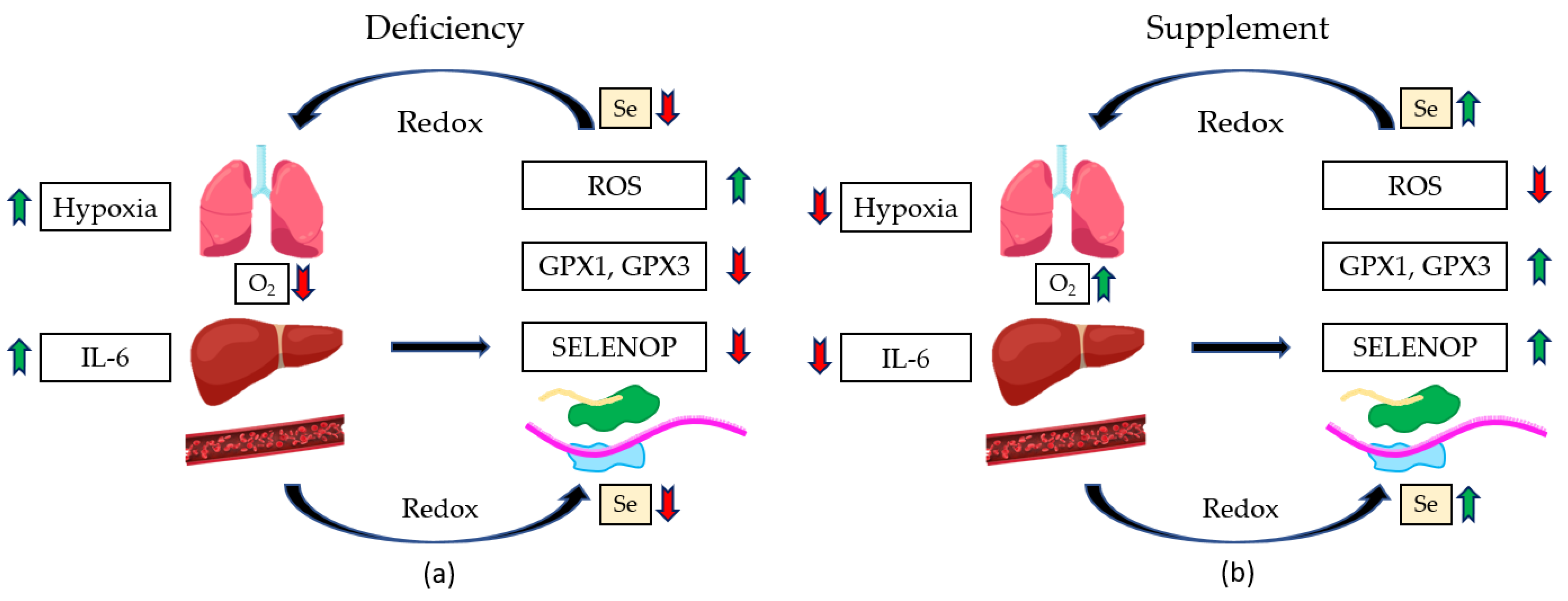
- Moghaddam, A.; Heller, R.A.; Sun, Q.; Seelig, J.; Cherkezov, A.; Seibert, L.; Hackler, J.; Seemann, P.; Diegmann, J.; Pilz, M.; et al. Selenium Deficiency Is Associated with Mortality Risk from COVID-19. Nutrients 2020, 12, 2098. [Google Scholar] [CrossRef] [PubMed]
- Mahase, E. COVID-19: Demand for dexamethasone surges as RECOVERY trial publishes preprint. BMJ 2020, 369, m2512. [Google Scholar] [CrossRef] [PubMed]
- Notz, Q.; Herrmann, J.; Schlesinger, T.; Helmer, P.; Sudowe, S.; Sun, Q.; Hackler, J.; Roeder, D.; Lotz, C.; Meybohm, P. Clinical significance of micronutrient supplementation in critically ill COVID-19 patients with severe ARDS. Nutrients 2021, 13, 2113. [Google Scholar] [CrossRef] [PubMed]
- Mahmoodpoor, A.; Hamishehkar, H.; Shadvar, K.; Ostadi, Z.; Sanaie, S.; Saghaleini, S.H.; Nader, N.D. The effect of intravenous selenium on oxidative stress in critically ill patients with acute respiratory distress syndrome. Immunol. Investig. 2019, 48, 147–159. [Google Scholar] [CrossRef]
- Budak, Ü.; Guo, Y.; Tanyildizi, E.; Şengür, A. Cascaded deep convolutional encoder-decoder neural networks for efficient liver tumor segmentation. Med. Hypotheses 2020, 134, 109431. [Google Scholar] [CrossRef]
- Diwaker, D.; Mishra, K.P.; Ganju, L. Potential roles of protein disulphide isomerase in viral infections. Acta Virol. 2013, 57, 293–304. [Google Scholar]
- Ware, L.B.; Matthay, M.A. The acute respiratory distress syndrome. N. Engl. J. Med. 2000, 342, 1334–1349. [Google Scholar] [CrossRef]
- Baldwin, S.; Grum, C.; Boxer, L.; Simon, R.; Ketai, L.; Devall, L. Oxidant activity in expired breath of patients with adult respiratory distress syndrome. Lancet 1986, 327, 11–14. [Google Scholar] [CrossRef]
- Rocksén, D.; Ekstrand-Hammarström, B.; Johansson, L.; Bucht, A. Vitamin E Reduces Transendothelial Migration of Neutrophils and Prevents Lung Injury in Endotoxin-Induced Airway Inflammation. Am. J. Respir. Cell Mol. Biol. 2003, 28, 199–207. [Google Scholar] [CrossRef]
- Nader, N.D.; McQuiller, P.S.; Raghavendran, K.; Spengler, R.N.; Knight, P.R. The role of alveolar macrophages in the pathogenesis of aspiration pneumonitis. Immunol. Investig. 2007, 36, 457–471. [Google Scholar] [CrossRef]
- Leff, J.A.; Day, C.; McCord, J.; Repine, J.; Parsons, P.; Moore, F.; Moore, E.; Taniguchi, N.; Jochum, M.; Fritz, H. Serum antioxidants as predictors of adult respiratory distress syndrome in patients with sepsis. Lancet 1993, 341, 777–780. [Google Scholar] [CrossRef] [PubMed]
- Nader, N.D.; Davidson, B.A.; Tait, A.R.; Holm, B.A.; Knight, P.R. Serine antiproteinase administration preserves innate superoxide dismutase levels after acid aspiration and hyperoxia but does not decrease lung injury. Anesth. Analg. 2005, 101, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Angstwurm, M.W.A.; Engelmann, L.; Zimmermann, T.; Lehmann, C.; Spes, C.H.; Abel, P.; Strauß, R.; Meier-Hellmann, A.; Insel, R.; Radke, J.; et al. Selenium in Intensive Care (SIC): Results of a prospective randomized, placebo-controlled, multiple-center study in patients with severe systemic inflammatory response syndrome, sepsis, and septic shock. Crit. Care Med. 2007, 35, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, J.; Akbaraly, N.T.; Hininger, I.; Roussel, A.-M.; Berr, C. Factors associated with longitudinal plasma selenium decline in the elderly: The EVA Study. J. Nutr. Biochem. 2007, 18, 482–487. [Google Scholar] [CrossRef]
- Ames, B.N. Low micronutrient intake may accelerate the degenerative diseases of aging through allocation of scarce micronutrients by triage. Proc. Natl. Acad. Sci. USA 2006, 103, 17589–17594. [Google Scholar] [CrossRef]
- Knox, S.J.; Jayachandran, P.; Keeling, C.A.; Stevens, K.J.; Sandhu, N.; Stamps-DeAnda, S.L.; Savic, R.; Shura, L.; Buyyounouski, M.K.; Grimes, K. Results from a phase 1 study of sodium selenite in combination with palliative radiation therapy in patients with metastatic cancer. Transl. Oncol. 2019, 12, 1525–1531. [Google Scholar] [CrossRef]
- Brodin, O.; Eksborg, S.; Wallenberg, M.; Asker-Hagelberg, C.; Larsen, E.H.; Mohlkert, D.; Lenneby-Helleday, C.; Jacobsson, H.; Linder, S.; Misra, S.; et al. Pharmacokinetics and Toxicity of Sodium Selenite in the Treatment of Patients with Carcinoma in a Phase I Clinical Trial: The SECAR Study. Nutrients 2015, 7, 4978–4994. [Google Scholar] [CrossRef]
- Olm, E.; Fernandes, A.P.; Hebert, C.; Rundlöf, A.-K.; Larsen, E.H.; Danielsson, O.; Björnstedt, M. Extracellular thiol-assisted selenium uptake dependent on the xc− cystine transporter explains the cancer-specific cytotoxicity of selenite. Proc. Natl. Acad. Sci. USA 2009, 106, 11400–11405. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, Y. Potential interventions for novel coronavirus in China: A systematic review. J. Med. Virol. 2020, 92, 479–490. [Google Scholar] [CrossRef]
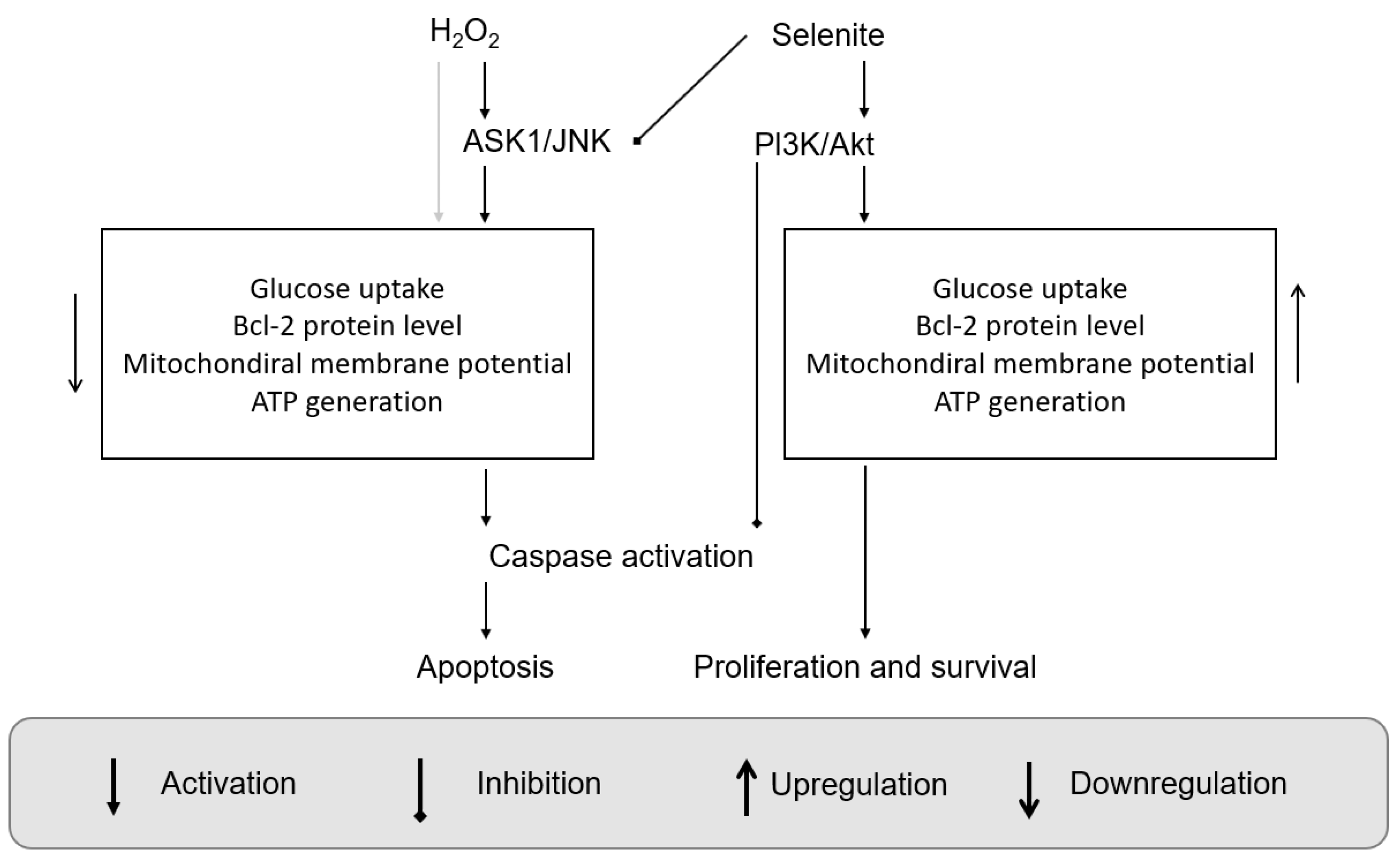
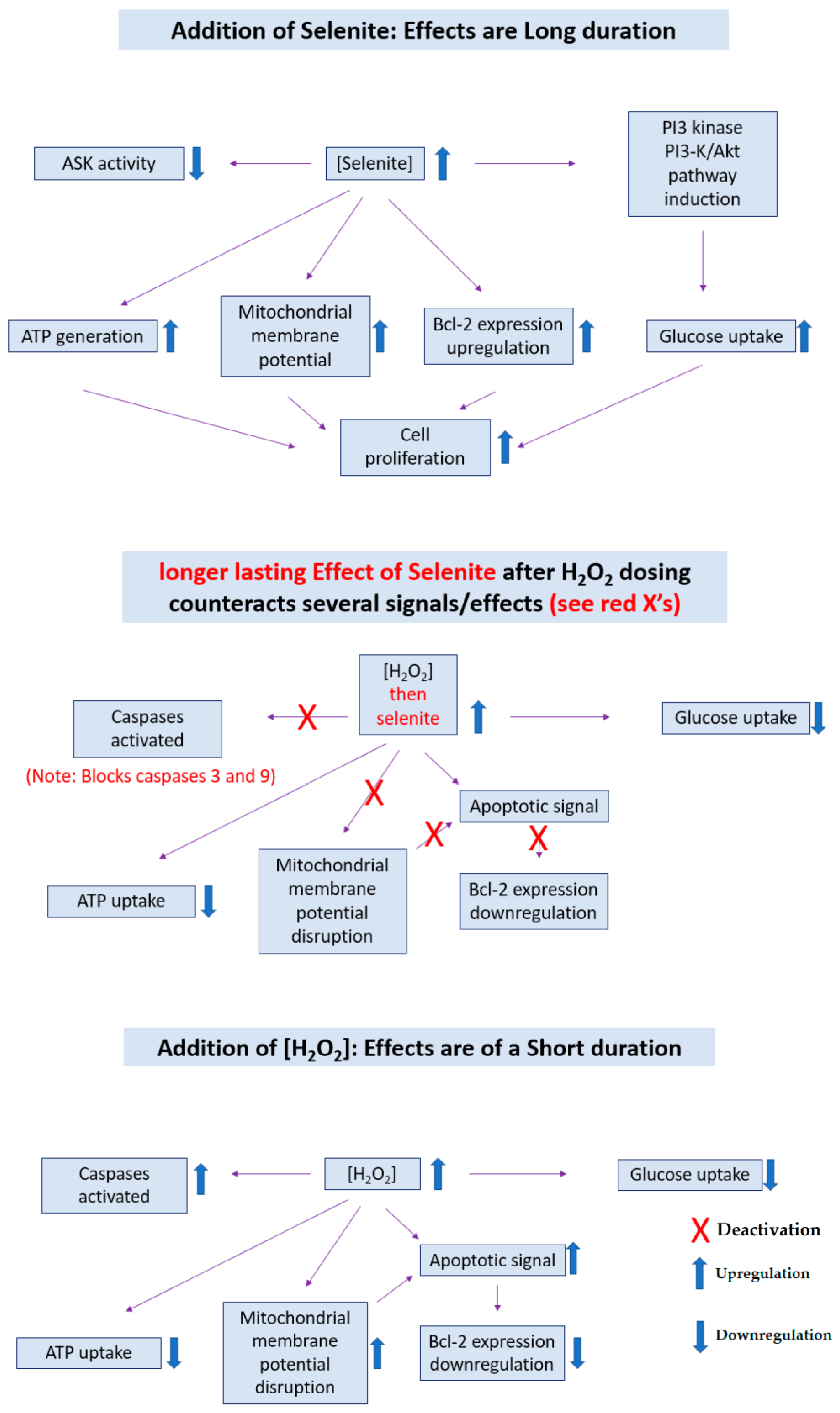
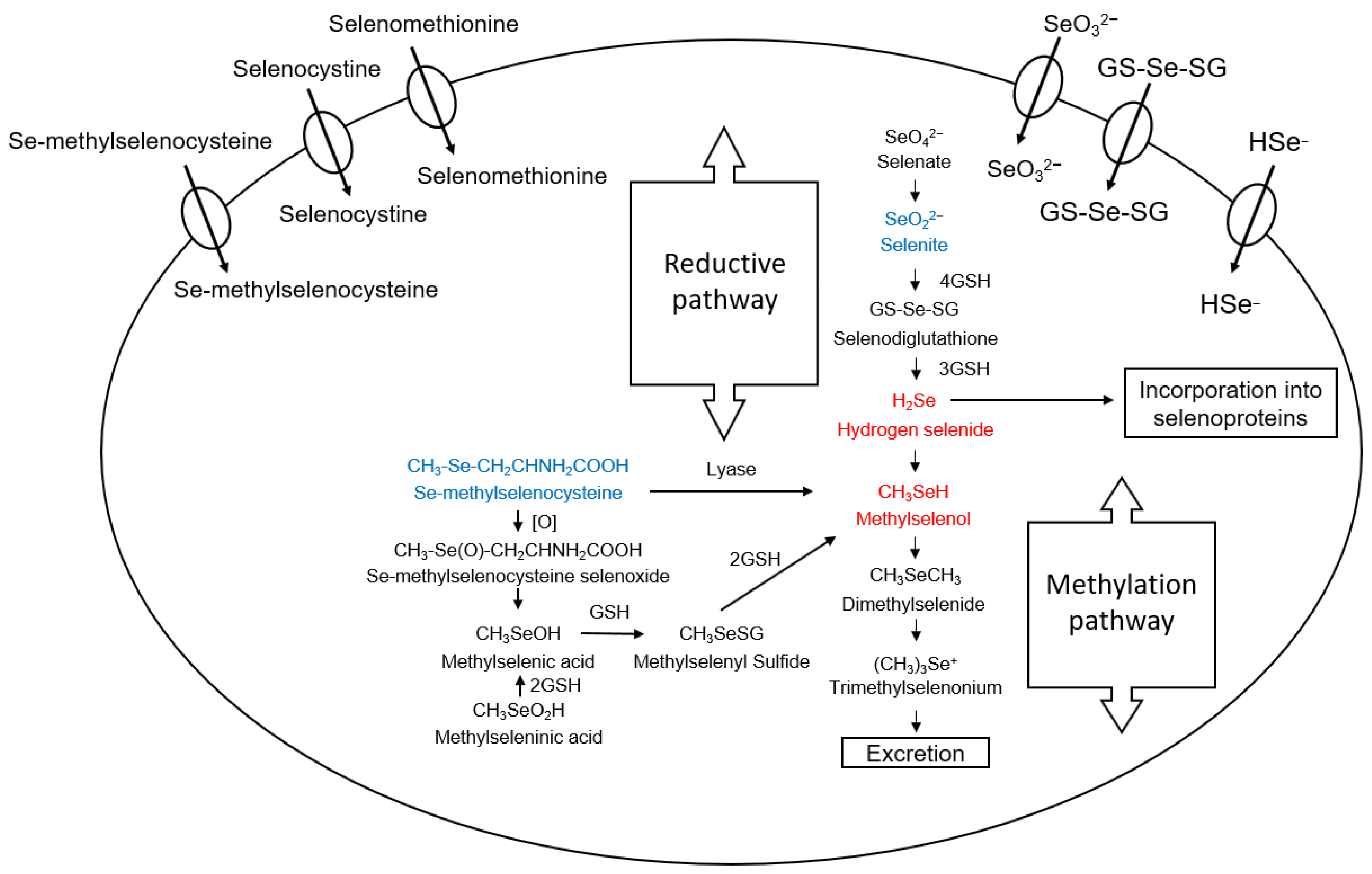
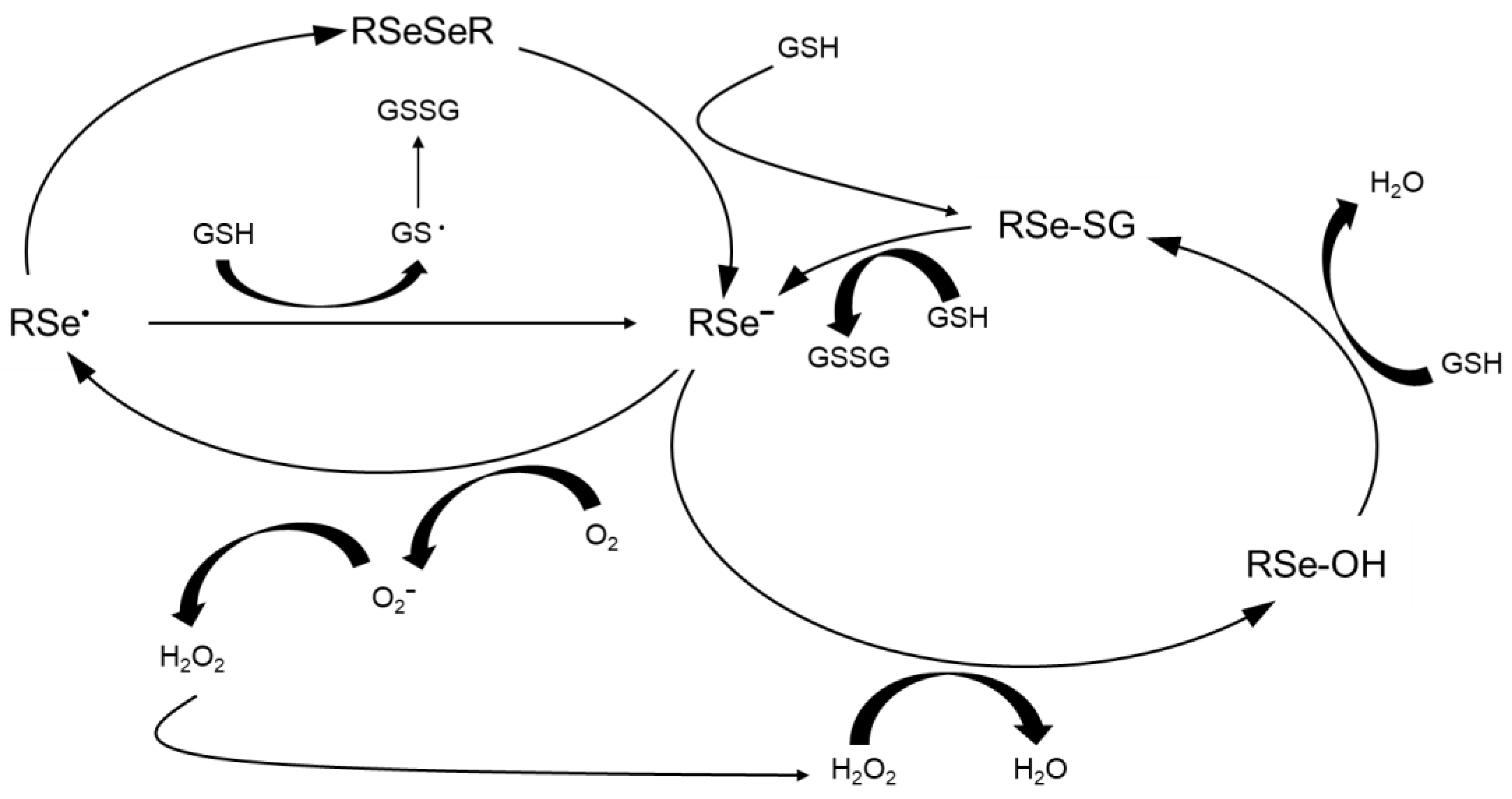



| Criteria | Values | Notes | References |
|---|---|---|---|
| Normal range of blood selenium | 120–160 μg/L | “The Tolerable Upper Intake Level (UL) for selenium for all adults 19+ years of age and pregnant and lactating women is 400 micrograms daily; a UL is the maximum daily intake unlikely to cause harmful effects on health.” | [3] |
| Tolerable intake, upper level(s) | 400 μg per day | [3] | |
| Daily required intake for human | See notes | 0–3 years of age: 10–20 micrograms (mcg) per day. 4–6 years of age: 20 mcg per day. 7–10 years of age: 30 mcg per day. Adolescent or adult males: 40–70 mcg per day. Adolescent or adult females: 45–55 mcg per day. | [4] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
An, J.-K.; Chung, A.-S.; Churchill, D.G. Nontoxic Levels of Se-Containing Compounds Increase Survival by Blocking Oxidative and Inflammatory Stresses via Signal Pathways Whereas High Levels of Se Induce Apoptosis. Molecules 2023, 28, 5234. https://doi.org/10.3390/molecules28135234
An J-K, Chung A-S, Churchill DG. Nontoxic Levels of Se-Containing Compounds Increase Survival by Blocking Oxidative and Inflammatory Stresses via Signal Pathways Whereas High Levels of Se Induce Apoptosis. Molecules. 2023; 28(13):5234. https://doi.org/10.3390/molecules28135234
Chicago/Turabian StyleAn, Jong-Keol, An-Sik Chung, and David G. Churchill. 2023. "Nontoxic Levels of Se-Containing Compounds Increase Survival by Blocking Oxidative and Inflammatory Stresses via Signal Pathways Whereas High Levels of Se Induce Apoptosis" Molecules 28, no. 13: 5234. https://doi.org/10.3390/molecules28135234
APA StyleAn, J.-K., Chung, A.-S., & Churchill, D. G. (2023). Nontoxic Levels of Se-Containing Compounds Increase Survival by Blocking Oxidative and Inflammatory Stresses via Signal Pathways Whereas High Levels of Se Induce Apoptosis. Molecules, 28(13), 5234. https://doi.org/10.3390/molecules28135234









