Skin Pigmentation Types, Causes and Treatment—A Review
Abstract
1. Introduction
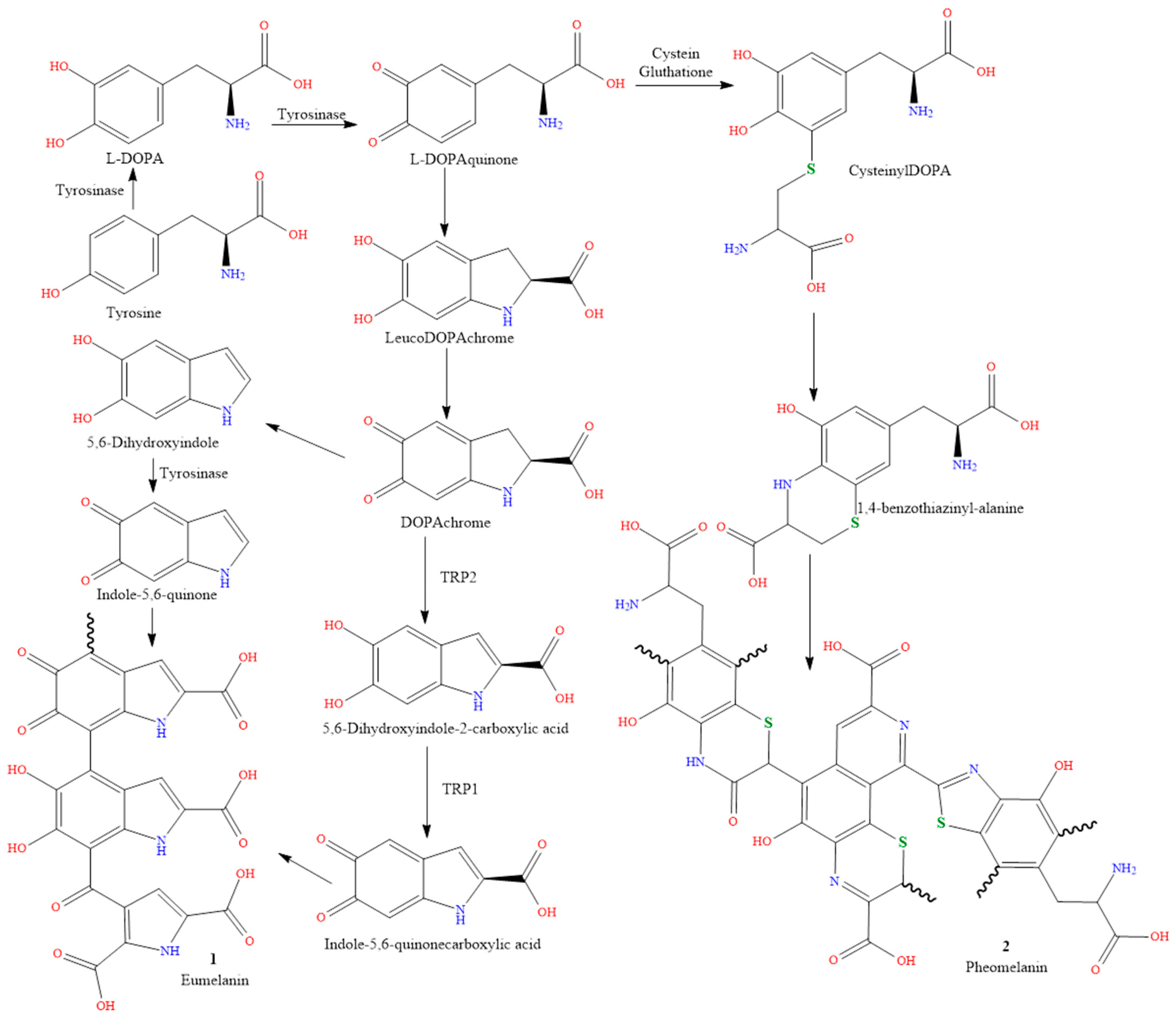
2. Causes of Skin Pigmentation
2.1. Genetics
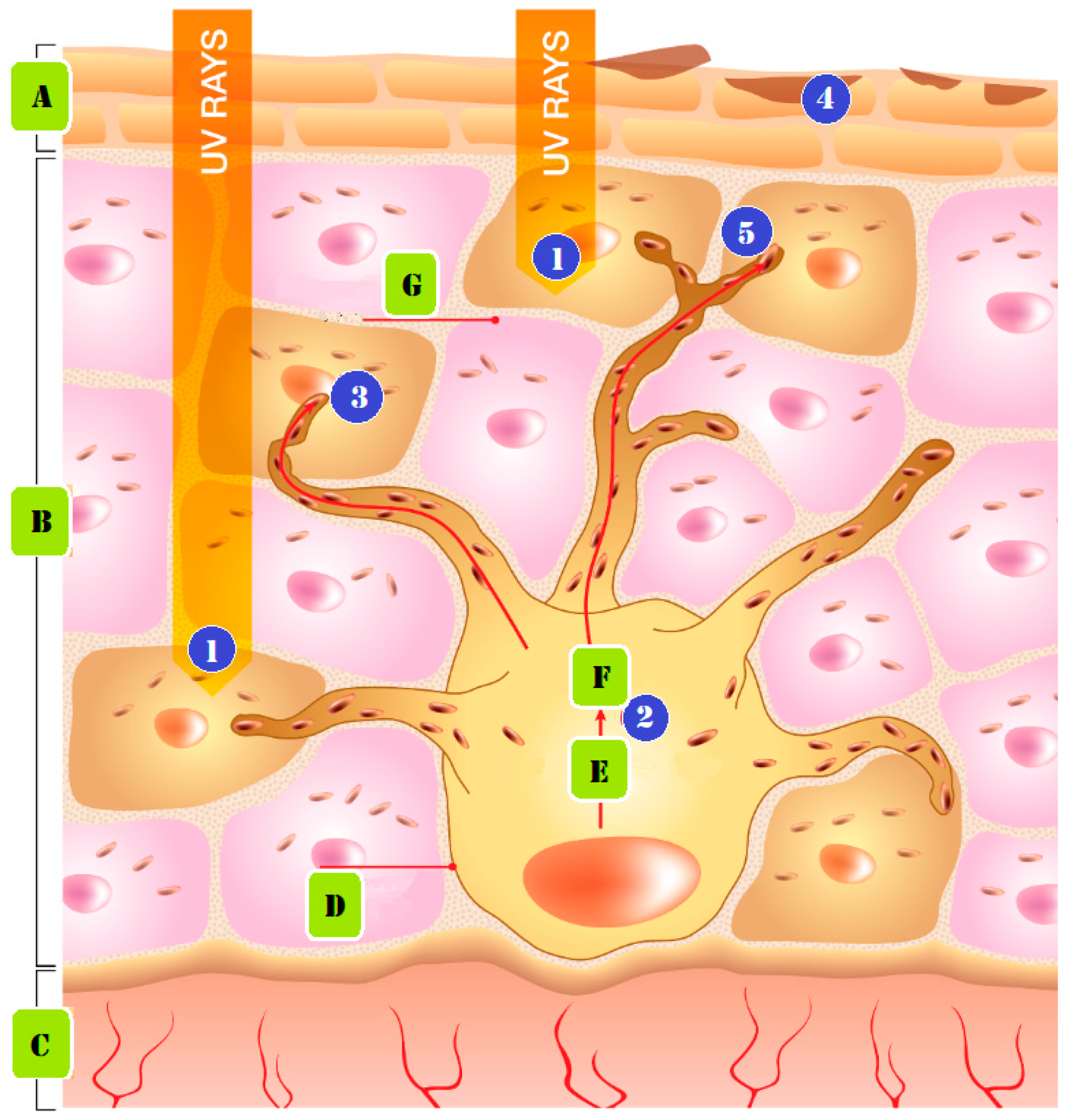
2.2. Sun Exposure
2.3. Medications
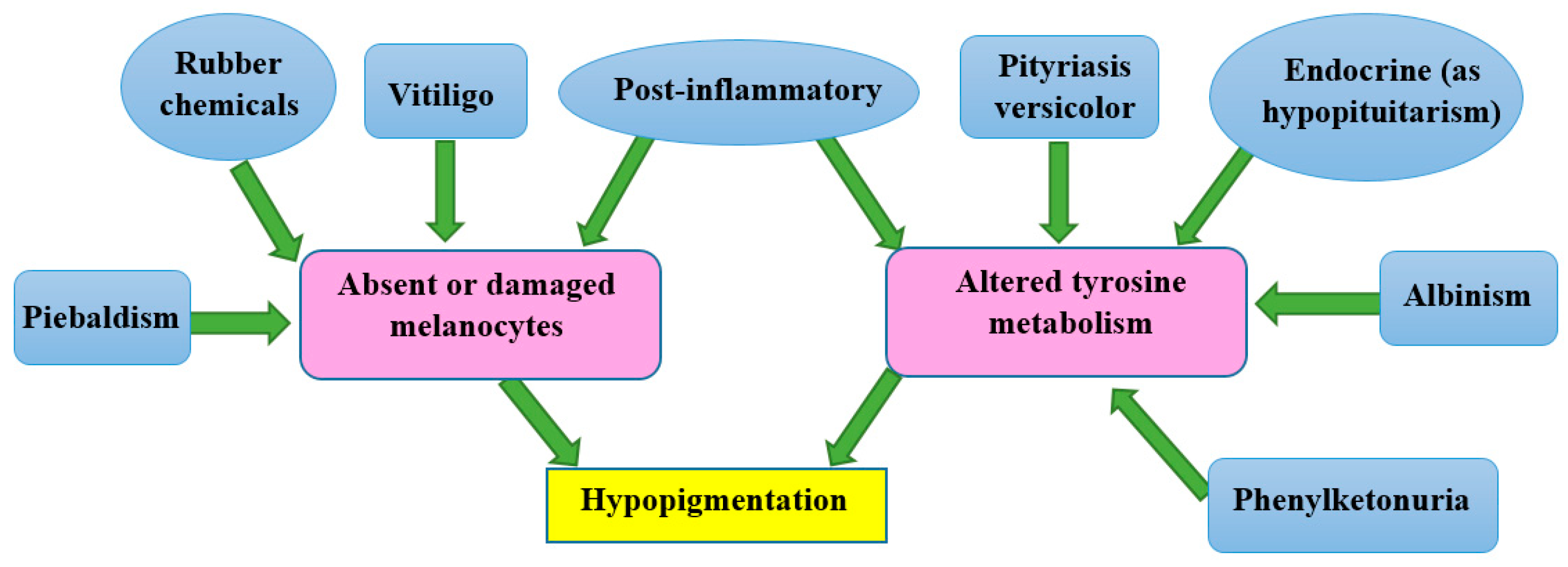
3. Types of Pigmentation Disorders
3.1. Causes of Hypopigmentation
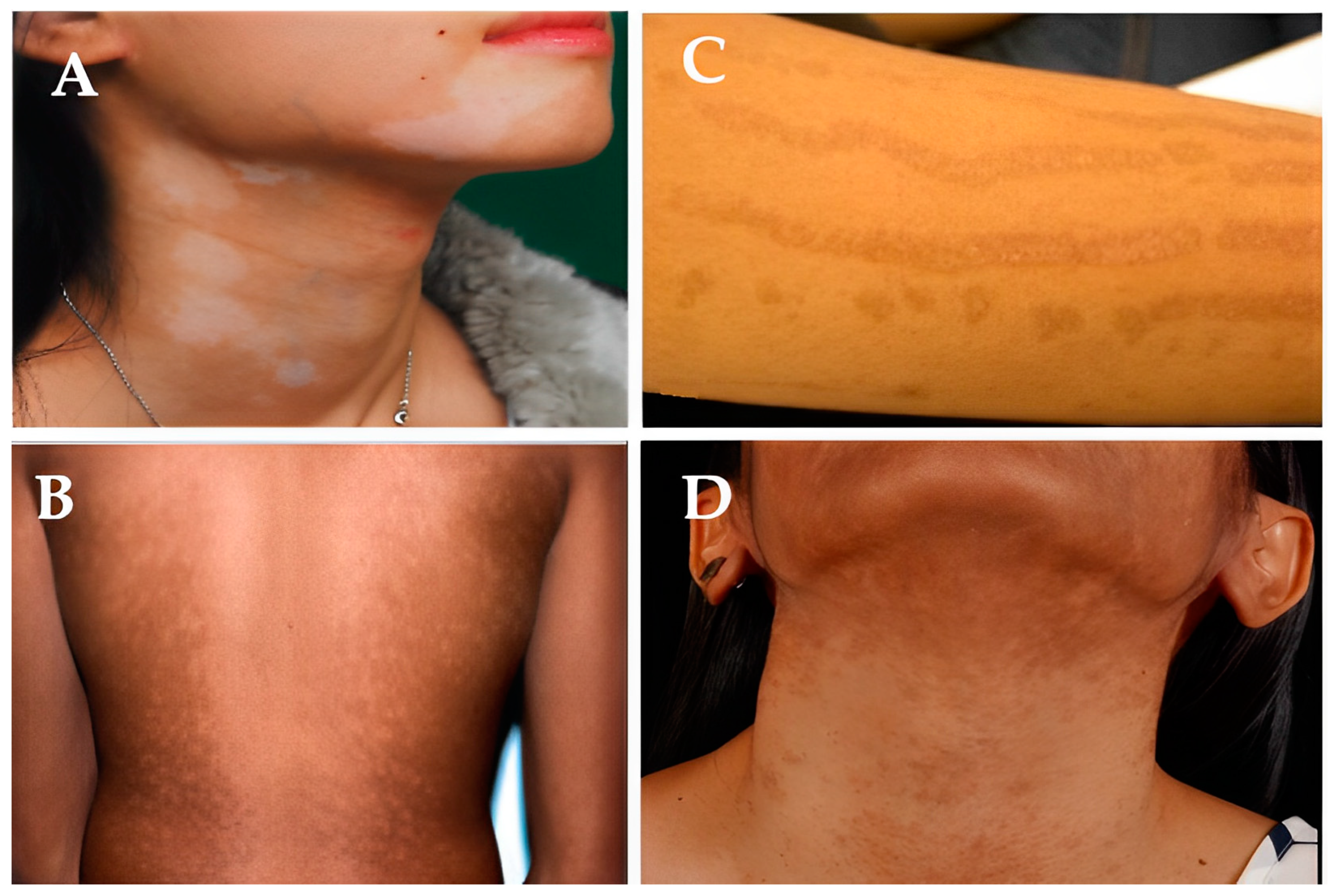
3.2. Causes of Hyperpigmentation
4. Drugs for Treatment of Skin Pigmentation
4.1. Oral Medications
4.2. Topical Creams
5. Natural Hyperpigmentation Treatment
| Name of Plant | Family | Growth Place | Active Compounds | Type of Pigmentation Targeted |
|---|---|---|---|---|
| Angelica sinensis [192] | Apiaceae | East Asia | 4-ethylresorcinol, 4-ethylphenol, 1-tetradecalnol | Hyperpigmentation agent combating skin-darkening. Study on Melan-A cells |
| Artocarpus [193] | Moraceae | Southeast Asia | Artocarpin, cudraflavone C, artocarpanone | TYR inhibitor. Hyperpigmentation–skin-whitening agents |
| Callicarpa longissima [194] | Lamiaceae | Southeast Asia | Carnosol | Antimelanogenesis in B16F10. Hyperpigmentation agents |
| Crataegus azarolus [195] | Rosaceae | European | Ursolic acid, hyperoside, virtexin-2″-O-rhamnoside | Antimelanogenesis in B16F10. Hyperpigmentation agents |
| Cyperus rotundus [196] | Cyperaceae | Africa, France, Austria, southern Asia | Valencene, camphene, carryophyllene oxide | Antimelanogenesis mechanism via the ion-channels in B16F10. Hyperpigmentation agents |
| Juniperus chinensis [197] | Cupressaceae | China, Myanmar, Russian, Korea | Widdrol | α-Melanocyte-stimulating hormone inhibition in B16F10 and TYR. Hyperpigmentation agents |
| Morus nigra [198] | Moraceae | Iberian Peninsula | Isoquercitrin, rutin, chlorogenic acid | Inhibit mushroom TYR. Hyperpigmentation agents |
| Oryza sativa [199] | Poaceae | China | p-Coumaric, ferulic | Antimelanogenesis in B16F10 melanoma by TYR. Hyperpigmentation agents |
| Passiflora edulis [200] | Passifloraceae | Brazil, Paraguay, Argentina | Piceatannol, resveratrol, quercetin | Antimelanogenesis in melanoma cells. Hyperpigmentation agents |
| Salvia officinalis [201] | Lamiaceae | Mediterranean region | 7a-methoxyrosmanol, isorosmanol | Antimelanogenesis in B16. Hyperpigmentation agents |
| Sesamum indicum [202] | Pedaliaceae | Africa, India | Sesamol | Antimelanogenesis in B16F10. Hyperpigmentation agents |
| Punica granatum [203] | Lythraceae | Mediterranean | Punicalgin | Antimelanogenesis in Melan-A. Hyperpigmentation agents |
| Litchi chinensis [204] | Sapindaceae | China, India, Bangladesh, Vietnam, Thailand, Malaysia, Indonesia, Pakistan, Cambodia, Bangladesh, Himalayas | Rosmarinc acid, gallic acid | Suppressed melanin production in B16F10 melanoma cells. Hyperpigmentation agents |
6. Modern Skin Pigmentation Treatments and Promising New Technologies
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Del Bino, S.; Duval, C.; Bernerd, F. Clinical and biological characterization of skin pigmentation diversity and its consequences on UV impact. Int. J. Mol. Sci. 2018, 19, 2668. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.R.; Lin, M.; Granka, J.M.; Myrick, J.W.; Liu, X.; Sockell, A.; Atkinson, E.G.; Werely, C.J.; Möller, M.; Sandhu, M.S.; et al. An unexpectedly complex architecture for skin pigmentation in Africans. Cell 2017, 171, 1340–1353. [Google Scholar] [CrossRef]
- Nasti, T.H.; Timares, L. MC 1R, Eumelanin and Pheomelanin: Their role in determining the susceptibility to skin cancer. Photochem. Photobiol. 2015, 91, 188–200. [Google Scholar] [CrossRef]
- Solano, F. Photoprotection and skin pigmentation: Melanin-related molecules and some other new agents obtained from natural sources. Molecules 2020, 25, 1537. [Google Scholar] [CrossRef]
- Polidori, C.; Jorge, A.; Ornosa, C. Eumelanin and pheomelanin are predominant pigments in bumblebee (Apidae: Bombus) pubescence. PeerJ 2017, 5, e3300–e3321. [Google Scholar] [CrossRef]
- Hu, S.; Zhai, P.; Chen, Y.; Zhao, B.; Yang, N.; Wang, M.; Xiao, Y.; Bao, G.; Wu, X. Morphological characterization and gene expression patterns for melanin pigmentation in Rex rabbit. Biochem. Genet. 2019, 57, 734–744. [Google Scholar] [CrossRef]
- Baek, S.H.; Lee, S.H. Sesamol decreases melanin biosynthesis in melanocyte cells and zebrafish: Possible involvement of MITF via the intracellular cAMP and p38/JNK signalling pathways. Exp. Dermatol. 2015, 24, 761–766. [Google Scholar] [CrossRef]
- Madelaine, R.; Ngo, K.J.; Skariah, G.; Mourrain, P. Genetic deciphering of the antagonistic activities of the melanin-concentrating hormone and melanocortin pathways in skin pigmentation. PLoS Genet. 2020, 16, e1009244-65. [Google Scholar] [CrossRef] [PubMed]
- Ozdeslik, R.N.; Olinski, L.E.; Trieu, M.M.; Oprian, D.D.; Oancea, E. Human nonvisual opsin 3 regulates pigmentation of epidermal melanocytes through functional interaction with melanocortin 1 receptor. Proc. Natl. Acad. Sci. USA 2019, 116, 11508–11517. [Google Scholar] [CrossRef] [PubMed]
- Wolf Horrell, E.M.; Boulanger, M.C.; D’Orazio, J.A. Melanocortin 1 receptor: Structure, function, and regulation. Front. Genet. 2016, 7, 95–111. [Google Scholar] [CrossRef]
- Suherlan, S.; Fakih, T.M.; Effendi, D.H. Uji In-Silico Aktivitas Melanogenesis Senyawa Ternatin Bunga Kembang Telang (Clitoria ternatea) terhadap Reseptor Tirosinase. Pros. Farm. 2021, 7, 849–856. [Google Scholar]
- Jablonski, N.G. The evolution of human skin pigmentation involved the interactions of genetic, environmental, and cultural variables. Pigment Cell Melanoma Res. 2021, 34, 707–729. [Google Scholar] [CrossRef] [PubMed]
- Ainger, S.A.; Jagirdar, K.; Lee, K.J.; Soyer, H.P.; Sturm, R.A. Skin pigmentation genetics for the clinic. Dermatology 2017, 233, 1–15. [Google Scholar] [CrossRef]
- Feng, Y.; McQuillan, M.A.; Tishkoff, S.A. Evolutionary genetics of skin pigmentation in African populations. Human Mol. Genet. 2021, 30, 88–97. [Google Scholar] [CrossRef]
- Kita, R.; Fraser, H.B. Local adaptation of sun-exposure-dependent gene expression regulation in human skin. PLoS Genet. 2016, 12, e1006382. [Google Scholar] [CrossRef]
- Armenta, A.M.; Henkel, E.D.; Ahmed, A.M. Pigmentation disorders in the elderly. Drugs Aging 2019, 36, 235–245. [Google Scholar] [CrossRef]
- Adigun, C.G. Adverse drug reactions of the lower extremities. Clin. Podiatr. Med. Surg. 2016, 33, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Nicolaidou, E.; Katsambas, A.D. Pigmentation disorders: Hyperpigmentation and hypopigmentation. Clin. Dermatol. 2014, 32, 66–72. [Google Scholar] [CrossRef]
- Böhm, M. Disorders of Melanin Pigmentation. In Braun-Falco’s Dermatology; Springer: Berlin/Heidelberg, Germany, 2021; pp. 1–35. [Google Scholar]
- Ma, E.Z.; Zhou, A.E.; Hoegler, K.M.; Khachemoune, A. Oculocutaneous albinism: Epidemiology, genetics, skin manifestation, and psychosocial issues. Arch. Dermatol. Res. 2023, 315, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Federico, J.R.; Krishnamurthy, K. Albinism. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Lee, D.Y.; Kim, C.R.; Lee, J.H. Trichrome vitiligo in segmental type. Photodermatol. Photoimmunol. Photomed. 2011, 27, 111–112. [Google Scholar] [CrossRef]
- Hill, J.P.; Batchelor, J.M. An approach to hypopigmentation. BMJ 2017, 356, 356–362. [Google Scholar] [CrossRef]
- Silpa-Archa, N.; Kohli, I.; Chaowattanapanit, S.; Lim, H.W.; Hamzavi, I. Postinflammatory hyperpigmentation: A comprehensive overview: Epidemiology, pathogenesis, clinical presentation, and noninvasive assessment technique. J. Am. Acad. Dermatol. 2017, 77, 591–605. [Google Scholar] [CrossRef]
- Kallini, J.R.; Riaz, F.; Khachemoune, A. Tinea versicolor in dark-skinned individuals. Int. J. Dermatol. 2014, 53, 137–141. [Google Scholar] [CrossRef]
- Miazek, N.; Michalek, I.; Pawlowska-Kisiel, M.; Olszewska, M.; Rudnicka, L. Pityriasis Alba—Common Disease, Enigmatic Entity: Up-to-Date Review of the Literature. Pediatr. Dermatol. 2015, 32, 786–791. [Google Scholar] [CrossRef]
- Jing, J.; Man, X.Y. Vitiligo-like depigmentation in a patient treated with PD-1 antibody. BMJ 2021, 374, n1982. [Google Scholar] [CrossRef]
- McMichael, L. Skin camouflage. BMJ 2012, 344, d7921. [Google Scholar] [CrossRef]
- Sheth, P.B.; Shah, H.A.; Dave, J.N. Periorbital hyperpigmentation: A study of its prevalence, common causative factors and its association with personal habits and other disorders. Indian J. Dermatol. 2014, 59, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.; Yin, L.; Smuda, C.; Batzer, J.; Hearing, V.J.; Kolbe, L. Molecular and histological characterization of age spots. Exp. Dermatol. 2017, 26, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Baxter, L.L.; Pavan, W.J. The etiology and molecular genetics of human pigmentation disorders. Wiley Interdiscip. Rev. Dev. Biol. 2013, 2, 379–392. [Google Scholar] [CrossRef] [PubMed]
- Yadalla, H.K.K.; Aradhya, S. Post acne hyperpigmentation: A brief review. Our Dermatol. Online 2011, 2, 230–231. [Google Scholar]
- Plensdorf, S.; Livieratos, M.; Dada, N. Pigmentation disorders: Diagnosis and management. Am. Fam. Physician 2017, 96, 797–804. [Google Scholar] [PubMed]
- Woolery-Lloyd, H.; Kammer, J.N. Treatment of Hyperpigmentation. In Seminars in Cutaneous Medicine and Surgery; WB Saunders: Philadelphia, PA, USA, 2011; Volume 30, pp. 171–175. [Google Scholar]
- Bala, H.R.; Lee, S.; Wong, C.; Pandya, A.G.; Rodrigues, M. Oral tranexamic acid for the treatment of melasma: A review. Dermatol. Surg. 2018, 44, 814–825. [Google Scholar] [CrossRef]
- Ali, A.A.; Al-Obaidi, Z.M.J.; Raauf, A.M.; Mahmood, H.S. A Comparative, Randomized, Double-Blinded, and Vehicle-Controlled Study for the Reduction in Facial Pigmentation after Treatment with both Tranexamic Acid and Tranexamic Acid Ethyl Ester. Syst. Rev. Pharm. 2020, 11, 563–567. [Google Scholar]
- Kaur, A.; Bhalla, M.; Sarkar, R. Tranexamic acid in melasma: A review. Pigment Int. 2020, 7, 12–25. [Google Scholar]
- Maeda, K. Mechanism of Action of Topical Tranexamic Acid in the Treatment of Melasma and Sun-Induced Skin Hyperpigmentation. Cosmetics 2022, 9, 108. [Google Scholar] [CrossRef]
- McKesey, J.; Tovar-Garza, A.; Pandya, A.G. Melasma treatment: An evidence-based review. Am. J. Clin. Dermatol. 2020, 21, 173–225. [Google Scholar] [CrossRef]
- Grimes, P.E.; Ijaz, S.; Nashawati, R.; Kwak, D. New oral and topical approaches for the treatment of melasma. Int. J. Women’s Dermatol. 2019, 5, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Artzi, O.; Horovitz, T.; Bar-Ilan, E.; Shehadeh, W.; Koren, A.; Zusmanovitch, L.; Mehrabi, J.; Salameh, F.; Nelkenbaum, G.I.; Zur, E.; et al. The pathogenesis of melasma and implications for treatment. J. Cosmet. Dermatol. 2021, 20, 3432–3445. [Google Scholar] [CrossRef]
- Taraz, M.; Niknam, S.; Ehsani, A.H. Tranexamic acid in treatment of melasma: A comprehensive review of clinical studies. Dermatol. Ther. 2017, 30, e12465. [Google Scholar] [CrossRef]
- Tse, T.W.; Hui, E. Tranexamic acid: An important adjuvant in the treatment of melasma. J. Cosmet. Dermatol. 2013, 12, 57–66. [Google Scholar] [CrossRef]
- Sharma, R.; Mahajan, V.K.; Mehta, K.S.; Chauhan, P.S.; Rawat, R.; Shiny, T.N. Therapeutic efficacy and safety of oral tranexamic acid and that of tranexamic acid local infiltration with microinjections in patients with melasma: A comparative study. Clin. Exp. Dermatol. 2017, 42, 728–734. [Google Scholar] [CrossRef]
- Kim, S.J.; Park, J.Y.; Shibata, T.; Fujiwara, R.; Kang, H.Y. Efficacy and possible mechanisms of topical tranexamic acid in melasma. Clin. Exp. Dermatol. 2016, 41, 480–485. [Google Scholar] [CrossRef]
- Sofen, B.; Prado, G.; Emer, J. Melasma and post inflammatory hyperpigmentation: Management update and expert opinion. Skin. Ther. Lett. 2016, 21, 1–7. [Google Scholar]
- Demir, B.; Çiçek, D.; Bilik, L.; Aydoğdu, E.G.; Artaş, H.; Demirpolat, N.; Ergin, C. Oral isotretinoin induced pigmentation disorder: A case report. Firat Tip Derg 2017, 22, 143–145. [Google Scholar]
- Mysore, V.; Mahadevappa, O.H.; Barua, S.; Majid, I.; Viswanath, V.; Bhat, R.M.; Talwar, S.; Thurakkal, S.; Aurangabadkar, S.J.; Chatterjee, M.; et al. Standard guidelines of care: Performing procedures in patients on or recently administered with isotretinoin. J. Cutan. Aesthetic Surg. 2017, 10, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Bagatin, E.; Costa, C.S. The use of isotretinoin for acne–an update on optimal dosing, surveillance, and adverse effects. Expert Rev. Clin. Pharmacol. 2020, 13, 885–897. [Google Scholar] [CrossRef] [PubMed]
- Fallah, H.; Rademaker, M. Isotretinoin in the management of acne vulgaris: Practical prescribing. Int. J. Dermatol. 2021, 60, 451–460. [Google Scholar] [CrossRef]
- Villani, A.; Nastro, F.; Di Vico, F.; Fabbrocini, G.; Annunziata, M.C.; Genco, L. Oral isotretinoin for acne: A complete overview. Expert Opin. Drug Saf. 2022, 21, 1027–1037. [Google Scholar] [CrossRef]
- Spring, L.K.; Krakowski, A.C.; Alam, M.; Bhatia, A.; Brauer, J.; Cohen, J.; Rosso, J.Q.; Diaz, L.; Dover, J.; Eichenfield, L.F.; et al. Isotretinoin and timing of procedural interventions: A systematic review with consensus recommendations. JAMA Dermatol. 2017, 153, 802–809. [Google Scholar] [CrossRef]
- Chu, S.; Michelle, L.; Ekelem, C.; Sung, C.T.; Rojek, N.; Mesinkovska, N.A. Oral isotretinoin for the treatment of dermatologic conditions other than acne: A systematic review and discussion of future directions. Arch. Dermatol. Res. 2021, 313, 391–430. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.; Chen, Y.; Zhang, L.; Zhang, Y.; Ariyawati, A.; Chen, T.; Chen, J.; Liu, L.; Pu, Y.; Li, Y.; et al. Effect of 30% supramolecular salicylic acid peel on skin microbiota and inflammation in patients with moderate-to-severe acne vulgaris. Dermatol. Ther. 2023, 13, 155–168. [Google Scholar] [CrossRef] [PubMed]
- Dréno, B.; Araviiskaia, E.; Kerob, D.; Andriessen, A.; Anfilova, M.; Arenbergerova, M.; Barrios, L.O.; Mokos, Z.B.; Haedersdal, M.; Hofmann, M.A. Nonprescription acne vulgaris treatments: Their role in our treatment armamentarium—An international panel discussion. J. Cosmet. Dermatol. 2020, 19, 2201–2211. [Google Scholar] [CrossRef] [PubMed]
- Rachmin, I.; Ostrowski, S.M.; Weng, Q.Y.; Fisher, D.E. Topical treatment strategies to manipulate human skin pigmentation. Adv. Drug Deliv. Rev. 2020, 153, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.K.; Ortonne, J.P. Pigmentation: Dyschromia. In Textbook of Cosmetic Dermatology; Martin-Dunitz Ltd.: London, UK, 1998; pp. 391–415. [Google Scholar]
- Yasir, M.; Goyal, A.; Sonthalia, S. Corticosteroid adverse effects. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Yélamos, O.; Alejo, B.; Ertekin SSVilla-Crespo, L.; Zamora-Barquero, S.; Martinez, N.; Domínguez, M.; Iglesias, P.; Herrero, A.; Malvehy, J.; Puig, S. Non-invasive clinical and microscopic evaluation of the response to treatment with clobetasol cream vs. calcipotriol/betamethasone dipropionate foam in mild to moderate plaque psoriasis: An investigator-initiated, phase IV, unicentric, open, randomized clinical trial. J. Eur. Acad. Dermatol. Venereol. 2021, 35, 143–149. [Google Scholar] [PubMed]
- Cho, J.H.; Bhutani, S.; Kim, C.H.; Irwin, M.R. Anti-inflammatory effects of melatonin: A systematic review and meta-analysis of clinical trials. Brain Behav. Immun. 2021, 93, 245–253. [Google Scholar] [CrossRef]
- Karekar, S.R.; Marathe, P.A.; Nagarajan, V.B.; Khopkar, U.S.; Chikhalkar, S.B.; Desai, P.K.; Dongre, M.S. Use of topical steroids in dermatology: A questionnaire based study. Indian Dermatol. Online J. 2020, 11, 725–730. [Google Scholar]
- Chadderdon, C.; Gaston, R.G.; Loeffler, B.J.; Lewis, D. Betamethasone Versus Ketorolac Injection for the Treatment of De Quervain’s Tenosynovitis: A Double-Blind Randomized Clinical Trial: Level 1 Evidence. J. Hand Surg. 2017, 42, S45–S46. [Google Scholar] [CrossRef]
- Patel, H.K.; Barot, B.S.; Parejiya, P.B.; Shelat, P.K.; Shukla, A. Topical delivery of clobetasol propionate loaded microemulsion based gel for effective treatment of vitiligo: Ex vivo permeation and skin irritation studies. Colloids Surf. B Biointerfaces 2013, 102, 86–94. [Google Scholar] [CrossRef]
- Khaitan, B.K.; Sindhuja, T. Autoimmunity in vitiligo: Therapeutic implications and opportunities. Autoimmun. Rev. 2022, 21, 102932. [Google Scholar] [CrossRef]
- Eleftheriadou, V.; Atkar, R.; Batchelor, J.; McDonald, B.; Novakovic, L.; Patel, J.V.; Ravenscroft, J.; Rush, E.; Shah, D.; Shah, R.; et al. British Association of Dermatologists’ Clinical Standards Unit. British Association of Dermatologists guidelines for the management of people with vitiligo 2021. Br. J. Dermatol. 2022, 186, 18–29. [Google Scholar] [CrossRef]
- Habet, K.A.; Kolli, S.S.; Pona, A.; Feldman, S.R. A review of topical corticosteroid sprays for the treatment of inflammatory dermatoses. Dermatol. Online J. 2019, 25, 3–12. [Google Scholar] [CrossRef]
- Gajinov, Z. Corticosteroid topical therapy range: Fluocinolone-acetonide gel. Galen. Med. J. 2022, 1, 17–22. [Google Scholar] [CrossRef]
- Medici, S.; Peana, M.; Nurchi, V.M.; Zoroddu, M.A. Medical uses of silver: History, myths, and scientific evidence. J. Med. Chem. 2019, 62, 5923–5943. [Google Scholar] [CrossRef]
- Bandyopadhyay, D. Topical antibacterials in dermatology. Indian J. Dermatol. 2021, 66, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Adhya, A.; Bain, J.; Ray, O.; Hazra, A.; Adhikari, S.; Dutta, G.; Ray, S.; Majumdar, B.K. Healing of burn wounds by topical treatment: A randomized controlled comparison between silver sulfadiazine and nano-crystalline silver. J. Basic Clin. Pharm. 2014, 6, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Nethi, S.K.; Das, S.; Patra, C.R.; Mukherjee, S. Recent advances in inorganic nanomaterials for wound-healing applications. Biomater. Sci. 2019, 7, 2652–2674. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Patil, A.; Prakash, C.; Kumari, H. Comparison of intralesional triamcinolone acetonide, 5-fluorouracil, and their combination in treatment of keloids. World J. Plast. Surg. 2018, 7, 212–219. [Google Scholar] [CrossRef]
- Khan, H.A.; Sahibzada, M.N.; Paracha, M.M. Comparison of the efficacy of intralesional bleomycin versus intralesional triamcinolone acetonide in the treatment of keloids. Dermatol. Ther. 2019, 32, e13036-40. [Google Scholar] [CrossRef]
- Raman, P.; Pitty, R.; Krithika, C.L.; Anand, S.N.; Subramani, G.P. Topical curcumin and triamcinolone acetonide in recurrent minor aphthous ulcers: A pilot trial. J. Contemp. Dent. Pract. 2020, 21, 884–890. [Google Scholar]
- Kwiatkowska, D.; Wicka, M.; Bulska, E.; Kaliszewski, P. Investigation of the Excretion of Triamcinolone Acetonide and Its Metabolite. Separations 2023, 10, 164. [Google Scholar] [CrossRef]
- Mangion, S.E.; Mackenzie, L.; Roberts, M.S.; Holmes, A.M. Seborrheic dermatitis: Topical therapeutics and formulation design. Eur. J. Pharm. Biopharm. 2023, 185, 148–164. [Google Scholar] [CrossRef]
- Pinto, L.M.; Chiricozzi, A.; Calabrese, L.; Mannino, M.; Peris, K. Novel Therapeutic Strategies in the Topical Treatment of Atopic Dermatitis. Pharmaceutics 2022, 14, 2767. [Google Scholar] [CrossRef]
- Sandhu, S.; Klein, B.A.; Al-Hadlaq, M.; Chirravur, P.; Bajonaid, A.; Xu, Y.; Intini, R.; Hussein, M.; Vacharotayangul, P.; Sroussi, H.; et al. Oral lichen planus: Comparative efficacy and treatment costs—A systematic review. BMC Oral Health 2022, 22, 161. [Google Scholar] [CrossRef]
- Pakravan, F.; Ghalayani, P.; Emami, H.; Isfahani, M.N.; Noorshargh, P. A novel formulation for radiotherapy-induced oral mucositis: Triamcinolone acetonide mucoadhesive film. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2019, 24, 63. [Google Scholar]
- Mangold, A.R.; Pittelkow, M.R. Lichen planus. Clin. Basic Immunodermatology 2017, 551–576. [Google Scholar] [CrossRef]
- Ahmad Nasrollahi, S.; Sabet Nematzadeh, M.; Samadi, A.; Ayatollahi, A.; Yadangi, S.; Abels, C.; Firooz, A. Evaluation of the safety and efficacy of a triple combination cream (hydroquinone, tretinoin, and fluocinolone) for treatment of melasma in Middle Eastern skin. Clin. Cosmet. Investig. Dermatol. 2019, 12, 437–444. [Google Scholar] [CrossRef]
- Dreher, F.; Draelos, Z.D.; Gold, M.H.; Goldman, M.P.; Fabi, S.G.; Puissegur Lupo, M.L. Efficacy of hydroquinone-free skin-lightening cream for photoaging. J. Cosmet. Dermatol. 2013, 12, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Banihashemi, M.; Zabolinejad, N.; Jaafari, M.R.; Salehi, M.; Jabari, A. Comparison of therapeutic effects of liposomal tranexamic acid and conventional hydroquinone on melasma. J. Cosmet. Dermatol. 2015, 14, 174–177. [Google Scholar] [CrossRef]
- Pradhan, M.; Singh, D.; Murthy, S.N.; Singh, M.R. Design, characterization and skin permeating potential of Fluocinolone acetonide loaded nanostructured lipid carriers for topical treatment of psoriasis. Steroids 2015, 101, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Treesirichod, A.; Chaithirayanon, S.; Chaikul, T.; Chansakulporn, S. The randomized trials of 10% urea cream and 0.025% tretinoin cream in the treatment of acanthosis nigricans. J. Dermatol. Treat. 2021, 32, 837–842. [Google Scholar] [CrossRef]
- Bagatin, E.; Gonçalves, H.D.S.; Sato, M.; Almeida, L.M.C.; Miot, H.A. Comparable efficacy of adapalene 0.3% gel and tretinoin 0.05% cream as treatment for cutaneous photoaging. Eur. J. Dermatol. 2018, 28, 343–350. [Google Scholar] [CrossRef]
- Nautiyal, A.; Wairkar, S. Management of hyperpigmentation: Current treatments and emerging therapies. Pigment Cell Melanoma Res. 2021, 34, 1000–1014. [Google Scholar] [CrossRef]
- Ferraris, C.; Rimicci, C.; Garelli, S.; Ugazio, E.; Battaglia, L. Nanosystems in cosmetic products: A brief overview of functional, market, regulatory and safety concerns. Pharmaceutics 2021, 13, 1408. [Google Scholar] [CrossRef] [PubMed]
- Usatine, R.; Tinitigan, M. Diagnosis and treatment of lichen planus. Am. Fam. Physician 2011, 84, 53–60. [Google Scholar] [PubMed]
- Korabiusz, K.; Wawryków, A.; Fabian-Danielewska, A.; Stecko, M.; Wilczyńska, A.; Janik-Fuks, I.; Martyna, M.; Harasimowicz, J. Laser removal of tattoo-A case report. J. Educ. Health Sport 2019, 9, 415–419. [Google Scholar]
- Doucette, K.; Forster, S.; Marcus, A. Study to Assess Visual Elimination of a Novel Otic Gel (Florfenicol, Terbinafine, Betamethasone Acetate) in Comparison to an Otic Solution (Florfenicol, Terbinafine, Mometasone Furoate) and an Otic Suspension (Gentamicin Sulfate, Clotrimazole, Mometasone Furoate Monohydrate) in Dogs Immediately after Application to the Ear Canal. In BSAVA Congress Proceedings 2018; BSAVA Library: Gloucester, UK, 2018; p. 489. [Google Scholar]
- Grammatikova, N.É. Comparative study of the antimicrobial activity of combined topical medicinal formulations of betamethasone, gentamicin, and clotrimazole in vitro. Pharm. Chem. J. 2020, 53, 971–975. [Google Scholar] [CrossRef]
- Cole, L.K.; Rajala-Schultz, P.J.; Lorch, G. Conductive hearing loss in four dogs associated with the use of ointment-based otic medications. Vet. Dermatol. 2018, 29, 341-e120. [Google Scholar] [CrossRef] [PubMed]
- Hikmatovich, I.N. Evaluation of the Efficacy of External Therapy in Sick Children with Alergodermatosis. Web Semant. Univers. J. Innov. Educ. 2023, 2, 50–54. [Google Scholar]
- Harris, J.E.; Rashighi, M.; Nguyen, N.; Jabbari, A.; Ulerio, G.; Clynes, R.; Christiano, A.M.; Mackay-Wiggan, J. Rapid skin repigmentation on oral ruxolitinib in a patient with coexistent vitiligo and alopecia areata (AA). J. Am. Acad. Dermatol. 2016, 74, 370–371. [Google Scholar] [CrossRef]
- Rosmarin, D.; Pandya, A.G.; Lebwohl, M.; Grimes, P.; Hamzavi, I.; Gottlieb, A.B.; Butler, K.; Kuo, F.; Sun, K.; Ji, T.; et al. Ruxolitinib cream for treatment of vitiligo: A randomised, controlled, phase 2 trial. Lancet 2020, 396, 110–120. [Google Scholar] [CrossRef]
- Rothstein, B.; Joshipura, D.; Saraiya, A.; Abdat, R.; Ashkar, H.; Turkowski, Y.; Vaneeta Sheth, V.; Huang, V.; Chung, S.; Kachuk, C.; et al. Treatment of vitiligo with the topical Janus kinase inhibitor ruxolitinib. J. Am. Acad. Dermatol. 2017, 76, 1054–1060. [Google Scholar] [CrossRef]
- Sheikh, A.; Rafique, W.; Owais, R.; Malik, F.; Ali, E. FDA approves Ruxolitinib (Opzelura) for Vitiligo Therapy: A breakthrough in the field of dermatology. Ann. Med. Surg. 2022, 81, 104499–104506. [Google Scholar] [CrossRef]
- Shreberk-Hassidim, R.; Ramot, Y.; Zlotogorski, A. Janus kinase inhibitors in dermatology: A systematic review. J. Am. Acad. Dermatol. 2017, 76, 745–753. [Google Scholar] [CrossRef]
- Chapman, S.; Kwa, M.; Gold, L.S.; Lim, H.W. Janus kinase inhibitors in dermatology: Part I. A comprehensive review. J. Am. Acad. Dermatol. 2022, 86, 406–413. [Google Scholar] [CrossRef]
- Rosmarin, D.; Passeron, T.; Pandya, A.G.; Grimes, P.; Harris, J.E.; Desai, S.R.; Lebwohl, M.; Mulard, M.; Seneschal, J.; Wolkerstorfer, A.; et al. Two phase 3, randomized, controlled trials of ruxolitinib cream for vitiligo. N. Engl. J. Med. 2022, 387, 1445–1455. [Google Scholar] [CrossRef]
- Seneschal, J.; Wolkerstorfer, A.; Desai, S.R.; Grimes, P.; Ezzedine, K.; Kornacki, D.; Butler, K.; Kuo, F.I.; Sun, K.; Grimes, P.; et al. Efficacy and safety of ruxolitinib cream for the treatment of vitiligo by patient demographics and baseline clinical characteristics: Week 52 pooled subgroup analysis from two randomized phase 3 studies. Br. J. Dermatol. 2023, 188 (Suppl. S1), ljac106-006. [Google Scholar] [CrossRef]
- Sripathi, S.K.; Lalitha, P. Keratolytic Molecule Aided Inhibition of DNA Damage and Tyrosinase Activity of a Herbal Formulation. Int. J. BioSci. Technol. 2016, 9, 7–14. [Google Scholar]
- Arif, T. Salicylic acid as a peeling agent: A comprehensive review. Clin. Cosmet. Investig. Dermatol. 2015, 8, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Jiang, R.; Zhou, J.; Xu, X.; Sun, Z.; Li, J.; Chen, X.; Li, Z.; Yan, X.; Zhao, D.; et al. Salicylic acid in ginseng root alleviates skin hyperpigmentation disorders by inhibiting melanogenesis and melanosome transport. Eur. J. Pharmacol. 2021, 910, 174458. [Google Scholar] [CrossRef]
- Karnes, J.B.; Usatine, R.P. Management of external genital warts. Am. Fam. Physician 2014, 90, 312–318. [Google Scholar]
- Lu, J.; Cong, T.; Wen, X.; Li, X.; Du, D.; He, G.; Jiang, X. Salicylic acid treats acne vulgaris by suppressing AMPK/SREBP 1 pathway in sebocytes. Exp. Dermatol. 2019, 28, 786–794. [Google Scholar] [CrossRef]
- Yeoh, S.C.; Goh, C.F. Topical delivery of salicylates. Drug Deliv. Transl. Res. 2021, 12, 981–1001. [Google Scholar] [CrossRef]
- Ogbechie-Godec, O.A.; Elbuluk, N. Melasma: An up-to-date comprehensive review. Dermatol. Ther. 2017, 7, 305–318. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, B.P.; Aman, T.; Alexis, A.F. Postinflammatory hyperpigmentation: Epidemiology, clinical presentation, pathogenesis and treatment. Am. J. Clin. Dermatol. 2018, 19, 489–503. [Google Scholar] [CrossRef]
- Al-Niaimi, F.; Chiang, N.Y.Z. Topical vitamin C and the skin: Mechanisms of action and clinical applications. J. Clin. Aesthetic Dermatol. 2017, 10, 14–17. [Google Scholar]
- Ravetti, S.; Clemente, C.; Brignone, S.; Hergert, L.; Allemandi, D.; Palma, S. Ascorbic acid in skin health. Cosmetics 2019, 6, 58. [Google Scholar] [CrossRef]
- Goik, U.; Goik, T.; Załęska, I. The properties and application of argan oil in cosmetology. Eur. J. Lipid Sci. Technol. 2019, 121, 1800313–1800342. [Google Scholar] [CrossRef]
- Phong, C.; Lee, V.; Yale, K.; Sung, C.; Mesinkovska, N. Coconut, Castor, and Argan Oil for Hair in Skin of Color Patients: A Systematic Review. J. Drugs Dermatol. JDD 2022, 21, 751–757. [Google Scholar]
- Charrouf, Z.; Guillaume, D. The argan oil project: Going from utopia to reality in 20 years. OCL 2018, 25, D209–D214. [Google Scholar] [CrossRef]
- Kanlayavattanakul, M.; Lourith, N. Plants and natural products for the treatment of skin hyperpigmentation–A review. Planta Med. 2018, 84, 988–1006. [Google Scholar] [CrossRef]
- Hollinger, J.C.; Angra, K.; Halder, R.M. Are natural ingredients effective in the management of hyperpigmentation? A systematic review. J. Clin. Aesthetic Dermatol. 2018, 11, 28–37. [Google Scholar]
- Panzella, L.; Napolitano, A. Natural and bioinspired phenolic compounds as tyrosinase inhibitors for the treatment of skin hyperpigmentation: Recent advances. Cosmetics 2019, 6, 57. [Google Scholar] [CrossRef]
- Grimes, P.; Bhawan, J.; Howell, M.; Desai, S.; Coryell, E.; Einziger, M.; Simpson, A.; Yaroshinsky, A.; McCraw, T. Histopathological Changes Induced by Malassezin: A Novel Natural Microbiome Indole for Treatment of Facial Hyperpigmentation. J. Drugs Dermatol. JDD 2022, 21, 141–145. [Google Scholar] [CrossRef]
- Karadas, F.; Erdoğan, S.; Kor, D.; Oto, G.; Uluman, M. The effects of different types of antioxidants (Se, vitamin E and carotenoids) in broiler diets on the growth performance, skin pigmentation and liver and plasma antioxidant concentrations. Braz. J. Poult. Sci. 2016, 18, 101–116. [Google Scholar] [CrossRef]
- Lucock, M.D. The evolution of human skin pigmentation: A changing medley of vitamins, genetic variability, and UV radiation during human expansion. Am. J. Biol. Anthropol. 2023, 180, 252–271. [Google Scholar] [CrossRef] [PubMed]
- Rembe, J.D.; Fromm-Dornieden, C.; Stuermer, E.K. Effects of vitamin B complex and vitamin C on human skin cells: Is the perceived effect measurable? Adv. Skin Wound Care 2018, 31, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Farzanfar, S.; Kouzekonan, G.S.; Mirjani, R.; Shekarchi, B. Vitamin B12-loaded polycaprolacton/gelatin nanofibrous scaffold as potential wound care material. Biomed. Eng. Lett. 2020, 10, 547–554. [Google Scholar] [CrossRef]
- Vivcharenko, V.; Wojcik, M.; Przekora, A. Cellular response to vitamin C-enriched chitosan/agarose film with potential application as artificial skin substitute for chronic wound treatment. Cells 2020, 9, 1185. [Google Scholar] [CrossRef]
- Barrios-Garay, K.; Toledano-Serrabona, J.; Gay-Escoda, C.; Sánchez-Garcés, M.Á. Clinical effect of vitamin C supplementation on bone healing: A systematic review. Med. Oral Patol. Oral Y Cirugía Bucal 2022, 27, e205–e215. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K.Y.; Song, H.J.; Kim, D.C. Effect of alpha-tocopheryl acetate, retinyl palmitate, and phytantriol on hair protection. J. Appl. Biol. Chem. 2022, 65, 307–312. [Google Scholar] [CrossRef]
- Santos, J.S.; Tavares, G.D.; Barradas, T.N. Vitamin E and derivatives in skin health promotion. In Vitamin E in Health and Disease-Interactions, Diseases and Health Aspects; IntechOpen: London, UK, 2021. [Google Scholar]
- Putranti, A.R.; Primaharinastiti, R.; Hendradi, E. Effectivity and physicochemical stability of nanostructured lipid carrier coenzyme Q10 in different ratio of lipid cetyl palmitate and alpha tocopheryl acetate as carrier. Asian J. Pharm. Clin. Res. 2017, 10, 146–152. [Google Scholar] [CrossRef]
- Panichakul, T.; Rodboon, T.; Suwannalert, P.; Tripetch, C.; Rungruang, R.; Boohuad, N.; Youdee, P. Additive effect of a combination of Artocarpus lakoocha and Glycyrrhiza glabra extracts on tyrosinase inhibition in melanoma B16 cells. Pharmaceuticals 2020, 13, 310. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Pathak, U.; Medhi, M.; Mastinu, A.; Sikarwar, M.S.; Mishra, P. Botanical, chemical and pharmacological properties of artocarpus lakoocha (monkey fruit): A review. Agric. Rev. 2020, 41, 305–316. [Google Scholar] [CrossRef]
- Ullah, S.; Shoaib, R.; Khan, S.; Masood, A. Phytochemicals; Targeted-Based Therapeutic Approaches for Pigmentation Disorders. Open Access Indones. J. Med. Rev. 2023, 3, 368–381. [Google Scholar] [CrossRef]
- Vaughn, A.R.; Clark, A.K.; Sivamani, R.K.; Shi, V.Y. Natural oils for skin-barrier repair: Ancient compounds now backed by modern science. Am. J. Clin. Dermatol. 2018, 19, 103–117. [Google Scholar] [CrossRef] [PubMed]
- Sarkic, A.; Stappen, I. Essential oils and their single compounds in cosmetics—A critical review. Cosmetics 2018, 5, 11. [Google Scholar] [CrossRef]
- Ali, S.A.; Galgut, J.M.; Choudhary, R.K. On the novel action of melanolysis by a leaf extract of Aloe vera and its active ingredient aloin, potent skin depigmenting agents. Planta Med. 2012, 78, 767–771. [Google Scholar] [CrossRef]
- Gad, H.A.; Roberts, A.; Hamzi, S.H.; Gad, H.A.; Touiss, I.; Altyar, A.E.; Kensara, A.O.; Ashour, M.L. Jojoba Oil: An updated comprehensive review on chemistry, pharmaceutical uses, and toxicity. Polymers 2021, 13, 1711. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Ma, S.; Tominaga, T.; Yokoyama, K.; Kitatani, K.; Horikawa, K.; Suzuki, K. Acute effects of transdermal administration of jojoba oil on lipid metabolism in mice. Medicina 2019, 55, 594. [Google Scholar] [CrossRef]
- Blaak, J.; Staib, P. An updated review on efficacy and benefits of sweet almond, evening primrose and jojoba oils in skin care applications. Int. J. Cosmet. Sci. 2022, 44, 1–9. [Google Scholar] [CrossRef]
- Sturtevant, D.; Lu, S.; Zhou, Z.W.; Shen, Y.; Wang, S.; Song, J.M.; Zhong, J.; Burks, D.J.; Yang, Z.Q.; Yang, Q.Y.; et al. The genome of jojoba (Simmondsia chinensis): A taxonomically isolated species that directs wax ester accumulation in its seeds. Sci. Adv. 2020, 6, e3240–e3253. [Google Scholar] [CrossRef]
- Nasr, M.; Abdel-Hamid, S.; Moftah, N.H.; Fadel, M.; Alyoussef, A.A. Jojoba oil soft colloidal nanocarrier of a synthetic retinoid: Preparation, characterization and clinical efficacy in psoriatic patients. Curr. Drug Deliv. 2017, 14, 426–432. [Google Scholar] [CrossRef]
- Sánchez, M.; Avhad, M.R.; Marchetti, J.M.; Martínez, M.; Aracil, J. Jojoba oil: A state of the art review and future prospects. Energy Convers. Manag. 2016, 129, 293–304. [Google Scholar] [CrossRef]
- Manoharan, S.; Vishnupriya, V.; Gayathri, R. Phytochemical analysis and in vitro antioxidant activity of jojoba oil. J. Pharm. Sci. Res. 2016, 8, 512–516. [Google Scholar]
- Leite, C.D.S.; Bonafé, G.A.; Carvalho Santos, J.; Martinez, C.A.R.; Ortega, M.M.; Ribeiro, M.L. The anti-inflammatory properties of licorice (Glycyrrhiza glabra)-derived compounds in intestinal disorders. Int. J. Mol. Sci. 2022, 23, 4121. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.J.; Son, D.H.; Chung, T.H.; Lee, Y.J. A review of the pharmacological efficacy and safety of licorice root from corroborative clinical trial findings. J. Med. Food 2020, 23, 12–20. [Google Scholar] [CrossRef]
- Mohiuddin, A.K. Skin lightening & management of hyperpigmentation. Pharma Sci. Anal. Res. J. 2019, 2, 180020–180068. [Google Scholar]
- Kimyon, R.S.; Liou, Y.L.; Schlarbaum, J.P.; Warshaw, E.M. Allergic contact dermatitis to licorice root extract. Dermatitis® 2019, 30, 227–228. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J.M.; Accioly, J.; Kitchen, N. Double Blind, Placebo Controlled Evaluation of a Novel Skin Lightening Agent. J. Drugs Dermatol. JDD 2018, 17, 113–115. [Google Scholar]
- Dhariwala, M.Y.; Ravikumar, P. An overview of herbal alternatives in androgenetic alopecia. J. Cosmet. Dermatol. 2019, 18, 966–975. [Google Scholar] [CrossRef]
- Boscariol, R.; Junior, J.M.O.; Baldo, D.A.; Balcão, V.M.; Vila, M.M. Transdermal permeation of curcumin promoted by choline geranate ionic liquid: Potential for the treatment of skin diseases. Saudi Pharm. J. 2022, 30, 382–397. [Google Scholar] [CrossRef]
- Vo, T.S.; Vo, T.T.B.C.; Vo, T.T.T.N.; Lai, T.N.H. Turmeric (Curcuma longa L.): Chemical components and their effective clinical applications. J. Turk. Chem. Soc. Sect. A Chem. 2021, 8, 883–898. [Google Scholar] [CrossRef]
- Firmansyah, D.; Sumiwi, S.A.; Saptarini, N.M.; Levita, J. Curcuma longa extract inhibits the activity of mushroom tyrosinase and the growth of murine skin cancer B16F10 cells. J. Herbmed Pharmacol. 2023, 12, 153–158. [Google Scholar] [CrossRef]
- Farooqui, R.K.; Kaurav, M.; Kumar, M.; Sudheesh, M.S.; Pandey, R.S. Permeation enhancer nanovesicles mediated topical delivery of curcumin for the treatment of hyperpigmentation. J. Liposome Res. 2022, 32, 332–339. [Google Scholar] [CrossRef]
- Akter, J.; Islam, M.Z.; Hossain, M.A.; Takara, K. Anti-tyrosinase properties of different species of turmeric and isolation of active compounds from Curcuma amada. Med. Chem. Res. 2021, 30, 1669–1676. [Google Scholar] [CrossRef]
- Rodríguez-Cid, L.; Qian, W.; Iribarra-Araya, J.; Etcheverry-Berríos, Á.; Martínez-Olmos, E.; Choquesillo-Lazarte, D.; Sañudo, E.C.; Roubeau, O.; Periago, A.M.L.; González-Camp, A.; et al. Broadening the scope of high structural dimensionality nanomaterials using pyridine-based curcuminoids. Dalton Trans. 2021, 50, 7056–7064. [Google Scholar] [CrossRef]
- Colantonio, S.; Rivers, J.K. Botanicals with dermatologic properties derived from first nations healing: Part 2—Plants and algae. J. Cutan. Med. Surg. 2017, 21, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, A.R.; Clark, A.K.; Notay, M.; Sivamani, R.K. Randomized controlled pilot study of dietary supplementation with turmeric or herbal combination tablets on skin barrier function in healthy subjects. J. Med. Food 2018, 21, 1260–1265. [Google Scholar] [CrossRef] [PubMed]
- Kallis, P.J.; Price, A.; Dosal, J.R.; Nichols, A.J.; Keri, J. A Biologically Based Approach to Acne and Rosacea. J. Drugs Dermatol. JDD 2018, 17, 611–617. [Google Scholar] [PubMed]
- Song, X.C.; Canellas, E.; Wrona, M.; Becerril, R.; Nerin, C. Comparison of two antioxidant packaging based on rosemary oleoresin and green tea extract coated on polyethylene terephthalate for extending the shelf life of minced pork meat. Food Packag. Shelf Life 2020, 26, 100588–100597. [Google Scholar] [CrossRef]
- Bagheri, R.; Rashidlamir, A.; Ashtary-Larky, D.; Wong, A.; Alipour, M.; Motevalli, M.S.; Chebbi, A.; Laher, I.; Zouhal, H. Does green tea extract enhance the anti-inflammatory effects of exercise on fat loss? Br. J. Clin. Pharmacol. 2020, 86, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Gaweł-Bęben, K.; Kukula-Koch, W.; Hoian, U.; Czop, M.; Strzępek-Gomółka, M.; Antosiewicz, B. Characterization of Cistus× incanus L. and Cistus ladanifer L. extracts as potential multifunctional antioxidant ingredients for skin protecting cosmetics. Antioxidants 2020, 9, 202. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.; Xu, Y.; Zhang, C.; Wang, N.; Li, H.; Feng, Y. Green tea and epigallocatechin gallate (EGCG) for the management of nonalcoholic fatty liver diseases (NAFLD): Insights into the role of oxidative stress and antioxidant mechanism. Antioxidants 2021, 10, 1076. [Google Scholar] [CrossRef]
- Hodges, J.K.; Sasaki, G.Y.; Bruno, R.S. Anti-inflammatory activities of green tea catechins along the gut–liver axis in nonalcoholic fatty liver disease: Lessons learned from preclinical and human studies. J. Nutr. Biochem. 2020, 85, 108478–108524. [Google Scholar] [CrossRef] [PubMed]
- Chaikul, P.; Sripisut, T.; Chanpirom, S.; Ditthawutthikul, N. Anti-skin aging activities of green tea (Camelliasinensis (L) Kuntze) in B16F10 melanoma cells and human skin fibroblasts. Eur. J. Integr. Med. 2020, 40, 101212–101240. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Sherje, A.P. Development of resveratrol and green tea sunscreen formulation for combined photoprotective and antioxidant properties. J. Drug Deliv. Sci. Technol. 2020, 60, 102000–102023. [Google Scholar] [CrossRef]
- Winkelman, W.J. Aromatherapy, botanicals, and essential oils in acne. Clin. Dermatol. 2018, 36, 299–305. [Google Scholar] [CrossRef]
- Patidar, K. Unmet Need and Challenges of Skin Aging by Herbal Anti-aging Cosmeceuticals: An Overview. Asian J. Pharm. (AJP) 2018, 12, 410–418. [Google Scholar]
- Phasha, V.; Senabe, J.; Ndzotoyi, P.; Okole, B.; Fouche, G.; Chuturgoon, A. Review on the use of kojic acid—A skin-lightening ingredient. Cosmetics 2022, 9, 64. [Google Scholar] [CrossRef]
- Saeedi, M.; Eslamifar, M.; Khezri, K. Kojic acid applications in cosmetic and pharmaceutical preparations. Biomed. Pharmacother. 2019, 110, 582–593. [Google Scholar] [CrossRef]
- Khezri, K.; Saeedi, M.; Morteza-Semnani, K.; Akbari, J.; Hedayatizadeh-Omran, A. A promising and effective platform for delivering hydrophilic depigmenting agents in the treatment of cutaneous hyperpigmentation: Kojic acid nanostructured lipid carrier. Artif. Cells Nanomed. Biotechnol. 2021, 49, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Park, T.J.; Ikram, M.; Ahmad, S.; Ahmad, R.; Jo, M.G.; Kim, M.O. Antioxidative and Anti-inflammatory Effects of Kojic Acid in Aβ-Induced Mouse Model of Alzheimer’s Disease. Mol. Neurobiol. 2021, 58, 5127–5140. [Google Scholar] [CrossRef] [PubMed]
- Zilles, J.C.; Dos Santos, F.L.; Kulkamp-Guerreiro, I.C.; Contri, R.V. Biological activities and safety data of kojic acid and its derivatives: A review. Exp. Dermatol. 2022, 31, 1500–1521. [Google Scholar] [CrossRef] [PubMed]
- Bakhouche, I.; Aliat, T.; Boubellouta, T.; Gali, L.; Şen, A.; Bellik, Y. Phenolic contents and in vitro antioxidant, anti-tyrosinase, and anti-inflammatory effects of leaves and roots extracts of the halophyte Limonium delicatulum. S. Afr. J. Bot. 2021, 139, 42–49. [Google Scholar] [CrossRef]
- Li, T.X.; Liang, J.X.; Liu, L.L.; Shi, F.C.; Jia, X.W.; Li, M.H.; Xu, C.P. Novel kojic acid derivatives with anti-inflammatory effects from Aspergillus versicolor. Fitoterapia 2021, 154, 105027–105033. [Google Scholar] [CrossRef]
- Gust, P.J.; Luke, J.D. Kojic acid. J. Dermatol. Nurses’ Assoc. 2016, 8, 338–340. [Google Scholar] [CrossRef]
- Searle, T.; Al-Niaimi, F.; Ali, F.R. The top 10 cosmeceuticals for facial hyperpigmentation. Dermatol. Ther. 2020, 33, e14095–e14139. [Google Scholar] [CrossRef]
- Ohno, Y.; Kondo, S.; Tajima, K.; Shibata, T.; Itoh, T. Effect of phlorotannins isolated from Eisenia bicyclis on melanogenesis in mouse B16 melanoma cells. Nat. Prod. Commun. 2021, 16, 1934578X211019264. [Google Scholar] [CrossRef]
- Phang, S.J.; Teh, H.X.; Looi, M.L.; Arumugam, B.; Fauzi, M.B.; Kuppusamy, U.R. Phlorotannins from brown algae: A review on their antioxidant mechanisms and applications in oxidative stress-mediated diseases. J. Appl. Phycol. 2023, 35, 867–892. [Google Scholar] [CrossRef]
- Jesumani, V.; Du, H.; Aslam, M.; Pei, P.; Huang, N. Potential use of seaweed bioactive compounds in skincare—A review. Mar. Drugs 2019, 17, 688. [Google Scholar] [CrossRef]
- Zhao, W.; Yang, A.; Wang, J.; Huang, D.; Deng, Y.; Zhang, X.; Qu, Q.; Ma, W.; Xiong, R.; Zhu, M.; et al. Potential application of natural bioactive compounds as skin-whitening agents: A review. J. Cosmet. Dermatol. 2022, 21, 6669–6687. [Google Scholar] [CrossRef]
- Kim, K.N.; Yang, H.M.; Kang, S.M.; Ahn, G.; Roh, S.W.; Lee, W.; Kim, D.K.; Jeon, Y.J. Whitening effect of octaphlorethol A isolated from Ishige foliacea in an in vivo zebrafish model. J. Microbiol. Biotechnol. 2015, 25, 448–451. [Google Scholar] [CrossRef]
- Lee, S.H.; Kang, N.; Kim, E.A.; Heo, S.J.; Moon, S.H.; Jeon, B.T.; Jeon, Y.J. Antidiabetogenic and antioxidative effects of octaphlorethol a isolated from the brown algae Ishige foliacea in streptozotocin-induced diabetic mice. Food Sci. Biotechnol. 2014, 23, 1261–1266. [Google Scholar] [CrossRef]
- Thawabteh, A.M.; Swaileh, Z.; Ammar, M.; Jaghama, W.; Yousef, M.; Karaman, R.; Bufo, S.A.; Scrano, L. Antifungal and Antibacterial Activities of Isolated Marine Compounds. Toxins 2023, 15, 93. [Google Scholar] [CrossRef]
- Shanura Fernando, I.P.; Asanka Sanjeewa, K.K.; Samarakoon, K.W.; Kim, H.S.; Gunasekara, U.K.D.S.S.; Park, Y.J.; Abeytunga, D.T.U.; Lee, W.W.; Jeon, Y.J. The potential of fucoidans from Chnoospora minima and Sargassum polycystum in cosmetics: Antioxidant, anti-inflammatory, skin-whitening, and antiwrinkle activities. J. Appl. Phycol. 2018, 30, 3223–3232. [Google Scholar] [CrossRef]
- Kok, J.M.L.; Jee, J.M.; Chew, L.Y.; Wong, C.L. The potential of the brown seaweed Sargassum polycystum against acne vulgaris. J. Appl. Phycol. 2016, 28, 3127–3133. [Google Scholar] [CrossRef]
- Lourenço-Lopes, C.; Fraga-Corral, M.; Soria-Lopez, A.; Nuñes-Estevez, B.; Barral-Martinez, M.; Silva, A.; Li, N.; Liu, C.; Gandara, J.S.; Prieto, M.A. Fucoxanthin’s optimization from Undaria pinnatifida using conventional heat extraction, bioactivity assays and in silico studies. Antioxidants 2022, 11, 1296. [Google Scholar] [CrossRef]
- Park, E.J.; Choi, J.I. Melanogenesis inhibitory effect of low molecular weight fucoidan from Undaria pinnatifida. J. Appl. Phycol. 2017, 29, 2213–2217. [Google Scholar] [CrossRef]
- Lee, S.H.; Kang, S.M.; Ko, S.C.; Lee, D.H.; Jeon, Y.J. Octaphlorethol A, a novel phenolic compound isolated from a brown alga, Ishige foliacea, increases glucose transporter 4-mediated glucose uptake in skeletal muscle cells. Biochem. Biophys. Res. Commun. 2012, 420, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Lajis, A.F.B.; Ariff, A.B. Discovery of new depigmenting compounds and their efficacy to treat hyperpigmentation: Evidence from in vitro study. J. Cosmet. Dermatol. 2019, 18, 703–727. [Google Scholar] [CrossRef]
- Haggag, Y.A.; Abd Elrahman, A.A.; Ulber, R.; Zayed, A. Fucoidan in pharmaceutical formulations: A comprehensive review for smart drug delivery systems. Mar. Drugs 2023, 21, 112. [Google Scholar] [CrossRef] [PubMed]
- Guan, B.; Chen, K.; Tong, Z.; Chen, L.; Chen, Q.; Su, J. Advances in fucoxanthin research for the prevention and treatment of inflammation-related diseases. Nutrients 2022, 14, 4768. [Google Scholar] [CrossRef] [PubMed]
- Natsume, C.; Aoki, N.; Aoyama, T.; Senda, K.; Matsui, M.; Ikegami, A.; Tanaka, K.; Azuma, Y.T.; Fujita, T. Fucoxanthin ameliorates atopic dermatitis symptoms by regulating keratinocytes and regulatory innate lymphoid cells. Int. J. Mol. Sci. 2020, 21, 2180. [Google Scholar] [CrossRef]
- Garcia-Jimenez, A.; Teruel-Puche, J.A.; Berna, J.; Rodriguez-Lopez, J.N.; Tudela, J.; Garcia-Ruiz, P.A.; Garcia-Canovas, F. Characterization of the action of tyrosinase on resorcinols. Bioorg. Med. Chem. 2016, 24, 4434–4443. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.J.; Lin, C.C.; Lu, T.M.; Li, J.H.; Chen, I.S.; Kuo, Y.H.; Ko, H.H. Chemical constituents derived from Artocarpus xanthocarpus as inhibitors of melanin biosynthesis. Phytochemistry 2015, 117, 424–435. [Google Scholar] [CrossRef]
- Yamahara, M.; Sugimura, K.; Kumagai, A.; Fuchino, H.; Kuroi, A.; Kagawa, M.; Itoh, Y.; Kawahara, H.; Nagaoka, Y.; Iida, O.; et al. Callicarpa longissima extract, carnosol-rich, potently inhibits melanogenesis in B16F10 melanoma cells. J. Nat. Med. 2016, 70, 28–35. [Google Scholar] [CrossRef]
- Mustapha, N.; Bzéouich, I.M.; Ghedira, K.; Hennebelle, T.; Chekir-Ghedira, L. Compounds isolated from the aerial part of Crataegus azarolus inhibit growth of B16F10 melanoma cells and exert a potent inhibition of the melanin synthesis. Biomed. Pharmacother. 2015, 69, 139–144. [Google Scholar] [CrossRef]
- Nam, J.H.; Nam, D.Y.; Lee, D.U. Valencene from the rhizomes of Cyperus rotundus inhibits skin photoaging-related ion channels and UV-induced melanogenesis in B16F10 melanoma cells. J. Nat. Prod. 2016, 79, 1091–1096. [Google Scholar] [CrossRef]
- Jin, K.S.; Lee, J.Y.; Hyun, S.K.; Kim, B.W.; Kwon, H.J. Juniperus chinensis and the functional compounds, cedrol and widdrol, ameliorate α-melanocyte stimulating hormone-induced melanin formation in B16F10 cells. Food Sci. Biotechnol. 2015, 24, 611–618. [Google Scholar] [CrossRef]
- de Freitas, M.M.; Fontes, P.R.; Souza, P.M.; William Fagg, C.; Neves Silva Guerra, E.; de Medeiros Nobrega, Y.K.; Silveira, D.; Bazzo, Y.F.; Simeoni, L.A.; Homem-de-Mello, M.; et al. Extracts of Morus nigra L. leaves standardized in chlorogenic acid, rutin and isoquercitrin: Tyrosinase inhibition and cytotoxicity. PLoS ONE 2016, 11, e0163130–e0163154. [Google Scholar] [CrossRef]
- Kanlayavattanakul, M.; Lourith, N.; Chaikul, P. Jasmine rice panicle: A safe and efficient natural ingredient for skin aging treatments. J. Ethnopharmacol. 2016, 193, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Lourith, N.; Kanlayavattanakul, M.; Chingunpitak, J. Development of sunscreen products containing passion fruit seed extract. Braz. J. Pharm. Sci. 2017, 53, 1–8. [Google Scholar] [CrossRef]
- Sallam, A.; Mira, A.; Ashour, A.; Shimizu, K. Acetylcholine esterase inhibitors and melanin synthesis inhibitors from Salvia officinalis. Phytomedicine 2016, 23, 1005–1011. [Google Scholar] [CrossRef]
- Srisayam, M.; Weerapreeyakul, N.; Barusrux, S.; Kanokmedhakul, K. Antioxidant, antimelanogenic, and skin-protective effect of sesamol. J. Cosmet Sci. 2014, 65, 69–79. [Google Scholar] [PubMed]
- Diwakar, G.; Rana, J.; Saito, L.; Vredeveld, D.; Zemaitis, D.; Scholten, J. Inhibitory effect of a novel combination of Salvia hispanica (chia) seed and Punica granatum (pomegranate) fruit extracts on melanin production. Fitoterapia 2014, 97, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Lourith, N.; Kanlayavattanakul, M.; Chaikul, P.; Chansriniyom, C.; Bunwatcharaphansakun, P. In vitro and cellular activities of the selected fruits residues for skin aging treatment. An. Acad. Bras. Ciências 2017, 89, 577–589. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.D.; Mohan, G.C.; Bhattacharya, T.; Patel, R.A.; Tsoukas, M. Pediatric laser therapy in pigmented conditions. Am. J. Clin. Dermatol. 2019, 20, 647–655. [Google Scholar] [CrossRef]
- Xie, X.Y.; Fu, G.L.; Yang, Q.; Zeng, Y.; Ke, H.; Lu, J.J.; Yi, H. Clinical Efficacy of 755 nm Laser Treatment of Lip Mucosal Pigmentation in Children with Peutz–Jeghers Syndrome. Dermatol. Ther. 2023, 2023, 8020443–8020447. [Google Scholar] [CrossRef]
- Nunez, J.H.; Strong, A.L.; Comish, P.; Hespe, G.E.; Harvey, J.; Sorkin, M.; Levi, B. A Review of Laser Therapies for the Treatment of Scarring and Vascular Anomalies. Adv. Wound Care 2023, 12, 68–84. [Google Scholar] [CrossRef]
- Purohit, S.J.; Tharmavaram, M.; Rawtani, D.; Prajapati, P.; Pandya, H.; Dey, A. Niosomes as cutting edge nanocarrier for controlled and targeted delivery of essential oils and biomolecules. J. Drug Deliv. Sci. Technol. 2022, 73, 103438. [Google Scholar] [CrossRef]
- Sharma, S.; Vashist, S.; Lamba, A.K.; Arora, S. Novel Strategies in the Treatment of Acne: A Review. Int. J. Pharm. Investig. 2022, 12, 123–128. [Google Scholar] [CrossRef]
- Yonekawa, Y.; Thomas, B.J.; Thanos, A.; Todorich, B.; Drenser, K.A.; Trese, M.T.; Capone Jr, A. The cutting edge of retinopathy of prematurity care: Expanding the boundaries of diagnosis and treatment. Retina 2017, 37, 2208–2225. [Google Scholar] [CrossRef]
- Al Qarqaz, F.; Al-Yousef, A. Skin microneedling for acne scars associated with pigmentation in patients with dark skin. J. Cosmet. Dermatol. 2018, 17, 390–395. [Google Scholar] [CrossRef]
- Ziaeifar, E.; Ziaeifar, F.; Mozafarpoor, S.; Goodarzi, A. Applications of microneedling for various dermatologic indications with a special focus on pigmentary disorders: A comprehensive review study. Dermatol. Ther. 2021, 34, e151–e159. [Google Scholar] [CrossRef] [PubMed]
- Lima, E.V.A.; Lima, M.M.D.A.; Miot, H.A. Induction of pigmentation through microneedling in stable localized vitiligo patients. Dermatol. Surg. 2020, 46, 434–435. [Google Scholar] [CrossRef]
- Ahmed, N.A.; Mohammed, S.S.; Fatani, M.I. Treatment of periorbital dark circles: Comparative study of carboxy therapy vs chemical peeling vs mesotherapy. J. Cosmet. Dermatol. 2019, 18, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, R.; Arsiwala, S.; Dubey, N.; Sonthalia, S.; Das, A.; Arya, L.; Gokhale, N.; Torsekar, R.G.; Somani, V.K.; Majid, I.; et al. Chemical peels in melasma: A review with consensus recommendations by Indian pigmentary expert group. Indian J. Dermatol. 2017, 62, 578–584. [Google Scholar] [CrossRef]
- Kadry, M.; Tawfik, A.; Abdallah, N.; Badawi, A.; Shokeir, H. Platelet-rich plasma versus combined fractional carbon dioxide laser with platelet-rich plasma in the treatment of vitiligo: A comparative study. Clin. Cosmet. Investig. Dermatol. 2018, 11, 551–559. [Google Scholar] [CrossRef]
- Mohamad, N.E.; ELgameel, R.M.; Mohamed, M.H. Comparative study between the effectiveness of plasma skin regeneration versus micro-needling in the treatment of striae distensae. J. Cosmet. Dermatol. 2022, 21, 4545–4553. [Google Scholar] [CrossRef] [PubMed]
- Wulandari, P.; Jusuf, N.K.; Nasution, K. Microneedling and platelet-rich plasma (PRP) treatment for mixed melasma. J. Gen. Proced. Dermatol. Venereol. Indones. 2022, 6, 44. [Google Scholar] [CrossRef]
- Rezapour, A.; Arabloo, J.; Moradi, N.; Ehsanzadeh, S.J.; Hourzad, M.; Alipour, V. Safety and effectiveness of endodermal radiofrequency for skin rejuvenation: A systematic review. Aesthetic Plast. Surg. 2023, 47, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Goel, A.; Gatne, V. Use of nanofractional radiofrequency for the treatment of acne scars in Indian skin. J. Cosmet. Dermatol. 2017, 16, 186–192. [Google Scholar] [CrossRef] [PubMed]
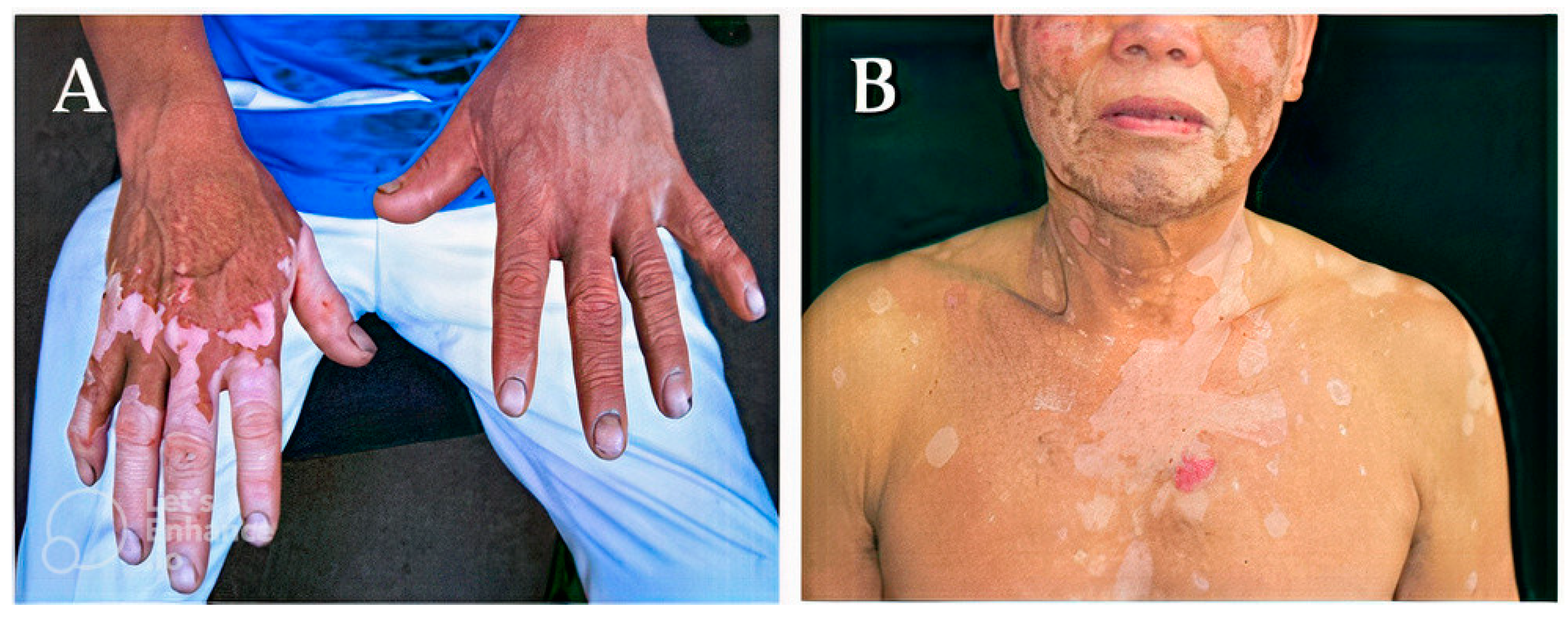

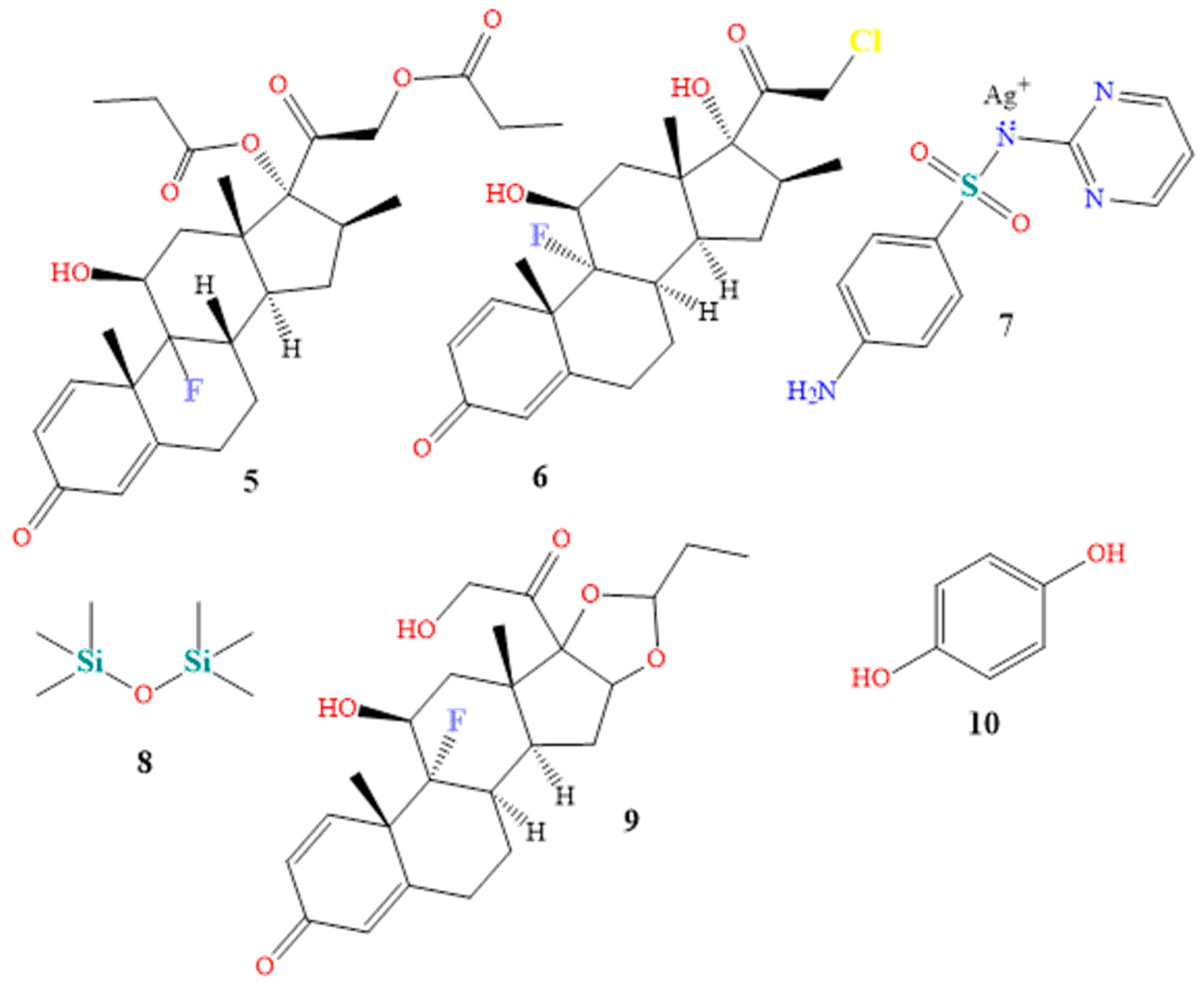

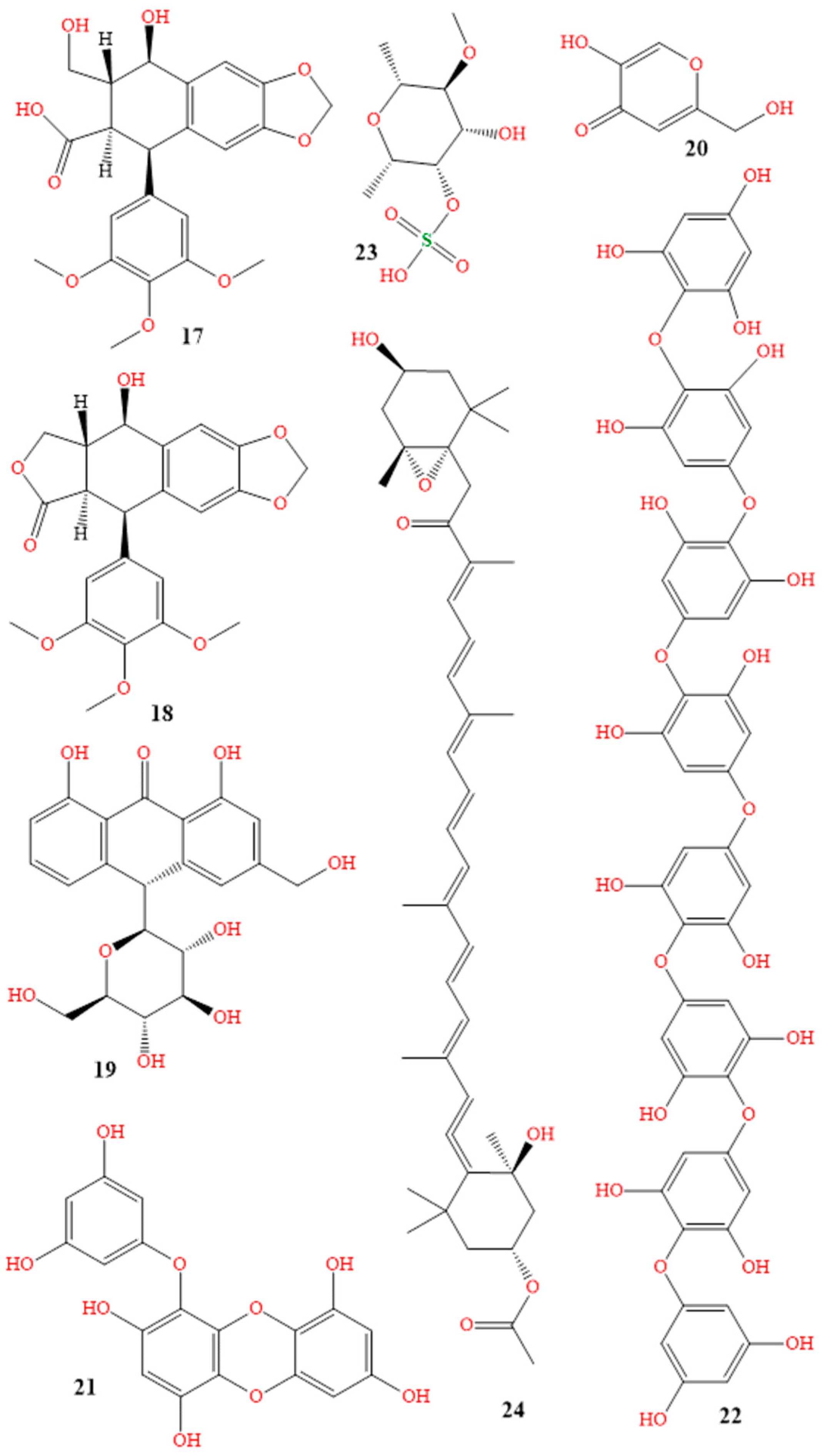
| Class | Generic Name | Brand Names® | Dosage Form |
|---|---|---|---|
| Topical steroids | Betamethasone | Etnovate, Diprolene, Luxiq, Beta-Val, Diprolene AF | Cream, gel, ointment, lotion |
| Clobetasol | Dermovate, Clobex, Olux, Olux-E, Temovate, Clobevate, Clodan, Cormax, Cormax Scalp, Embeline, Embeline E, Impeklo, Tovet | Solution, spray, ointment, gel, foam, lotion, cream, shampoo | |
| Triamcinolone acetonide | DermasilkRx SDS Pak, Dermasorb TA, DermaWerx SDS Pak, Kenalog, Oralone, Trianex, Triderm | Cream, ointment | |
| Topical anti-infectives | Silver topical | SilvaSorb, Aceso Ag, Solox | Cream, gel, foam |
| Topical steroids with anti-infectives | Dimethicone and triamcinolone topical | Yaliira Pak, Ellzia Pak, TriaDime-80, TriHeal-80 | Creams, shampoos, powders, gels |
| Topical depigmenting agents | Fluocinolone, hydroquinone and tretinoin topical | Tri-Luma, Triderma | Cream |
| Hydroquinone topical | Melquin HP, Alera, EpiQuin Micro, Esoterica, Hydro-Q, Melamin, Melpaque HP, Nuquin HP, AMBI Fade, Blanche, Esoterica Nighttime, Glytone, Lustra-Ultra, Melamin-C, NeoStrata HQ Skin Lightening, Olivia Quido, Fade cream, Remergent HQ | Cream | |
| Topical keratolytics | Salicylic acid topical | Bensal HP, KeralytGel, Salex, Acnex, Aliclen, DHS Salicylic Acid 3%, Durasal, Keralyt Shampoo, Stri-Dex, Akurza, DermalZone, Dr Scholl’s, Fostex, Freezeone, Rayasal, Salvax, Stridex, | Liquid, soap, cream, lotion, foam |
| Podophyllum resin topical | Podocon-25, Podofin, Pododerm | Topical solution | |
| Podofilox topical | Condylox | Topical gel, topical solution |
| Drug | Method of Evaluation | Duration | Performed Organism | Activity | Skin Disorder | Ref. |
|---|---|---|---|---|---|---|
| Tranexamic acid orally | Clinical and photographic | 6 months | Human | 10.8% excellent, 54% good, and 31.1% fair | Melasma | [41,42] |
| Topical tranexamic acid | MASI and chromameter | 12 weeks | Human | Improvement in the fourth and eighth weeks | Melasma | [45,46,47] |
| Isotretinoin orally | MASI | 16 weeks | Human | Reduction of roughly 73.4% | Acne | [56,57] |
| Betamethasone cream | Clinical | 3 month | Human | 40.0% no reaction, 46.7% moderate and 13.3% severe response | Vitiligo | [66,67] |
| Clobetasol propionate spray | Clinical | 4 weeks | Human | 80.0% of the patients in the therapy group had a decrease in severity from the beginning | Plaque psoriasis | [68,69] |
| Silver sulfadiazine cream | Clinical | 4 weeks | Human | 30% mild recovery, 48% quick recovery, 22% poor healing | 2° burn injuries | [75] |
| Triamcinolone mouthwash | Visual analog scale | 2 months | Human | Significant improvement in all patients | Oral lichen planus | [80,81,82] |
| Topical tretinoin, hydroquinone, and fluocinolone | Clinical | 8 weeks | Human | Improvement rate of 73% at weeks 4, 6, and 8 | Melasma | [90] |
| Tretinoin topically | Clinical | 12 week | Human | 77% of patients showed significant improvement | Melasma | [91] |
| Topically betamethasone, clotrimazole, and gentamicin | Clinical | 28 days | Human | At 7th day, a reduction in inflammation; at 14th day, scientific recovery in 42% of patients; at 28th day, the patients had fully recovered medically | Itchy dermatoses, including atopic dermatitis, contact dermatitis, and true eczema | [96] |
| Ruxolitinib | Clinical randomized controlled trials | 52 week | Human | Clinically excellent repigmentation after 24 weeks, with continued improvement through week 52 | Vitiligo | [103,104] |
| Salicylic acid | Mexameter and MASI | 14 weeks | Human | 83% believed that the peeled side had advanced more than the unpeeled side; 6% thought the unpeeled aspect was more advanced; 11% believed there was no difference | Bilateral melasma | [107,108] |
| L-ascorbic acid | Mexameter and MASI | 16 weeks | Human | Decrease from baseline | Enlarged pores, fine lines, and dullness | [111,112] |
| Argan oil | Clinical | 28 days | Human | Patients observed a minor decrease in melanin content in the vicinity of the bandage | Reducing the amount of pigmentation | [113,114,115] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thawabteh, A.M.; Jibreen, A.; Karaman, D.; Thawabteh, A.; Karaman, R. Skin Pigmentation Types, Causes and Treatment—A Review. Molecules 2023, 28, 4839. https://doi.org/10.3390/molecules28124839
Thawabteh AM, Jibreen A, Karaman D, Thawabteh A, Karaman R. Skin Pigmentation Types, Causes and Treatment—A Review. Molecules. 2023; 28(12):4839. https://doi.org/10.3390/molecules28124839
Chicago/Turabian StyleThawabteh, Amin Mahmood, Alaa Jibreen, Donia Karaman, Alà Thawabteh, and Rafik Karaman. 2023. "Skin Pigmentation Types, Causes and Treatment—A Review" Molecules 28, no. 12: 4839. https://doi.org/10.3390/molecules28124839
APA StyleThawabteh, A. M., Jibreen, A., Karaman, D., Thawabteh, A., & Karaman, R. (2023). Skin Pigmentation Types, Causes and Treatment—A Review. Molecules, 28(12), 4839. https://doi.org/10.3390/molecules28124839








