Structural, Electronic, Elastic, and Optical Characteristics of AgZF3 (Z = Sb and Bi) Fluoro-Perovskites: Using a Computational Approach for Energy Generation
Abstract
1. Introduction
2. Results and Discussion
2.1. Structural Properties
2.2. Electronic Properties
2.3. Elastic Properties
3. Optical Properties
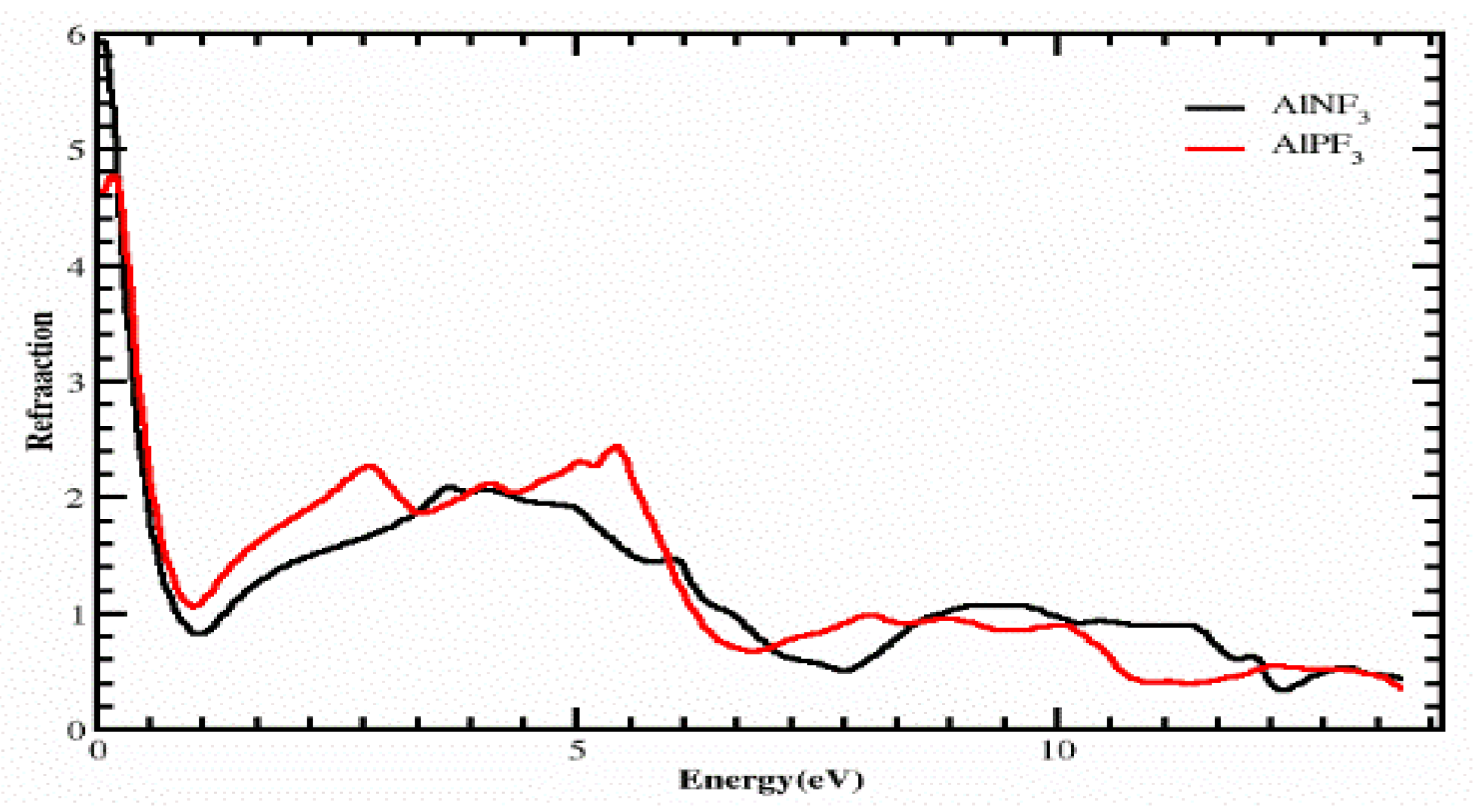
3.1. The Refractive Index
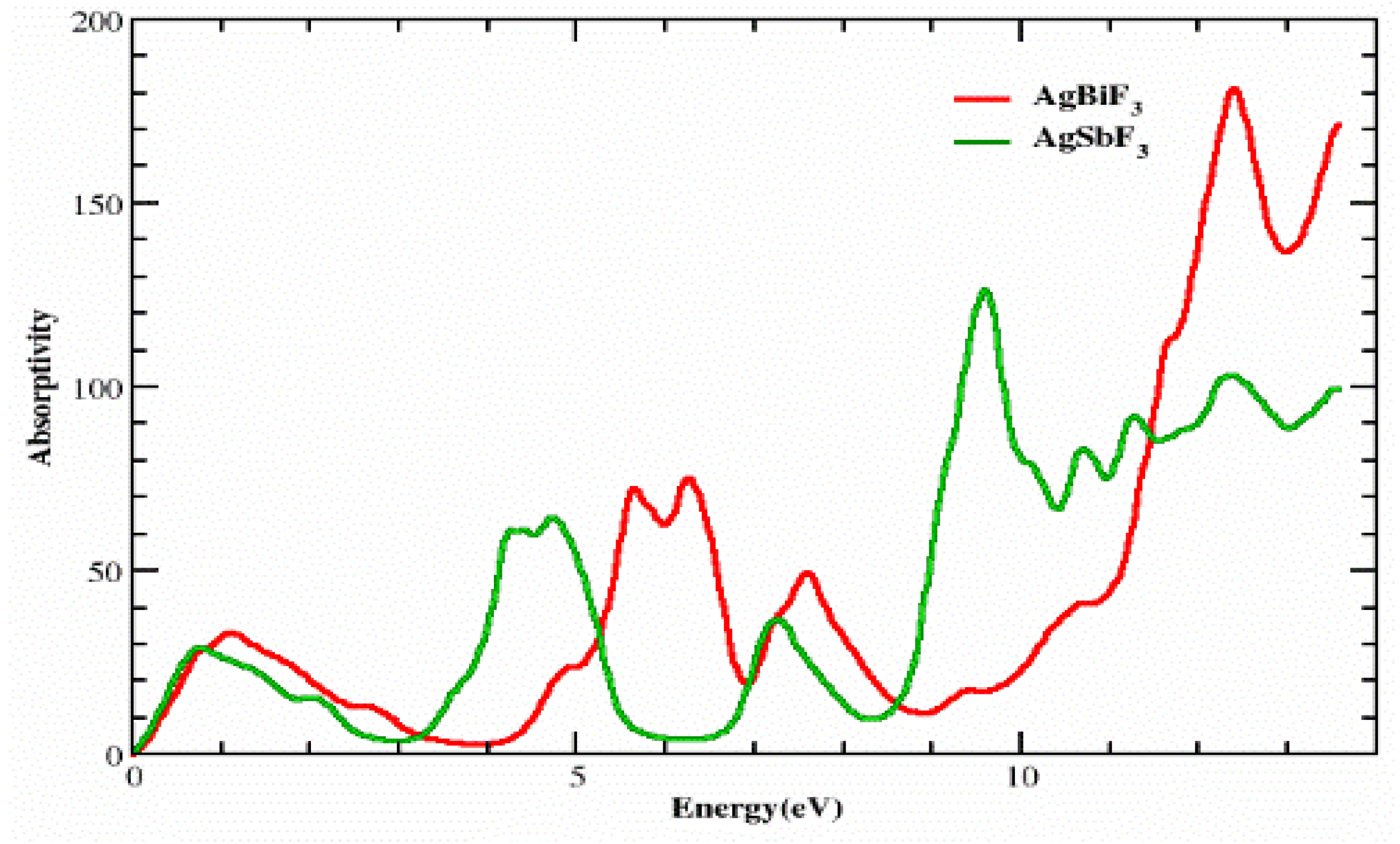
3.2. The Absorption Coefficient
3.3. The Reflectivity
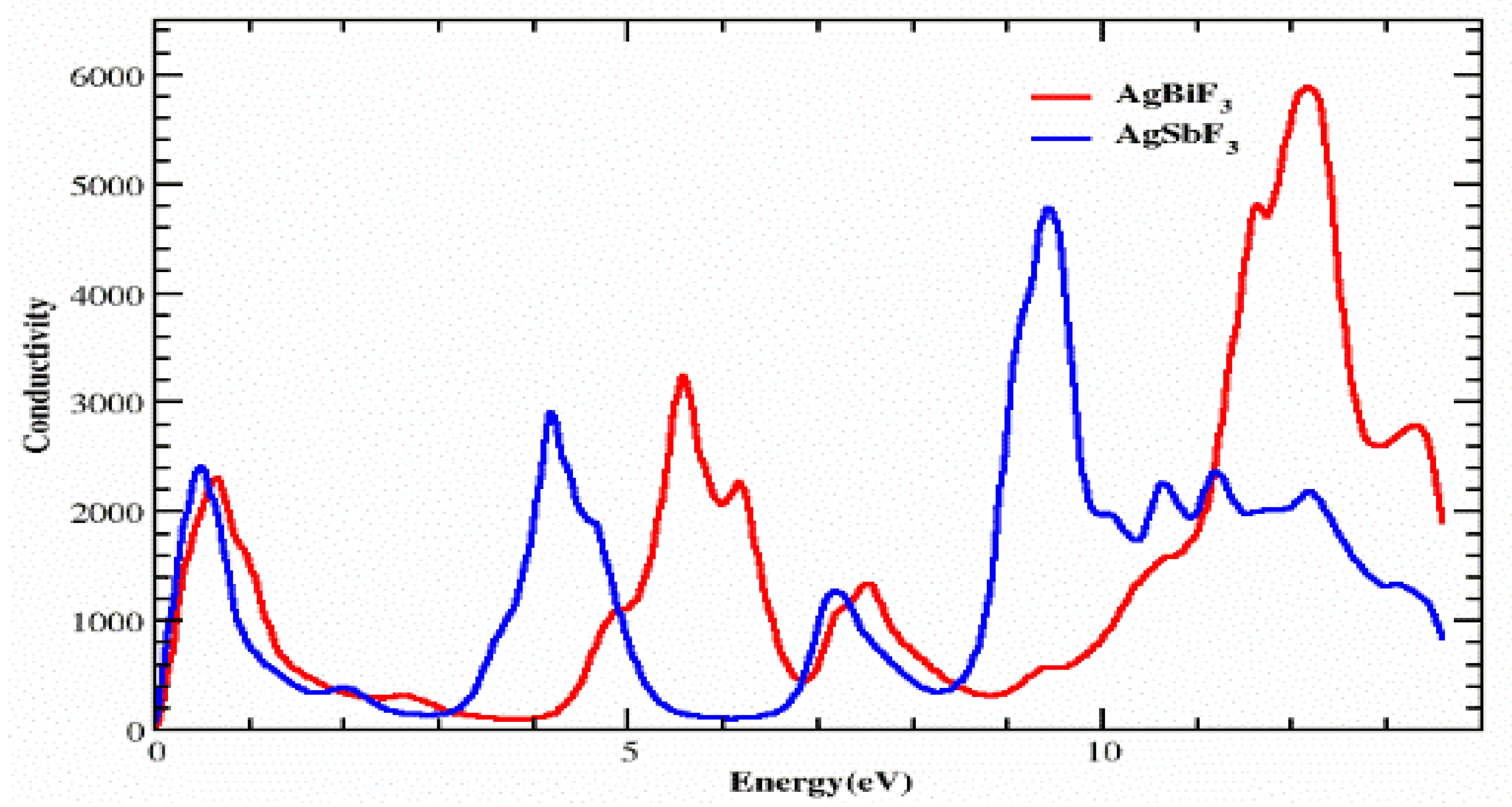
3.4. Optical Conductivity
4. Computational Methodology
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Nishimatsu, T.; Terakubo, N.; Mizuseki, H.; Kawazoe, Y.; Pawlak, D.A.; Shimamura, K.; Fukuda, T. Band Structures of Perovskite-Like Fluorides for Vacuum-Ultraviolet-Transparent Lens Materials. Jpn. J. Appl. Phys. 2002, 41, L365–L367. [Google Scholar] [CrossRef]
- Dotzler, C.; Williams, G.V.M.; Edgar, A. RbCdF3:Mn2+: A potential ultraviolet dosimeter material. Appl. Phys. Lett. 2007, 91, 181909. [Google Scholar] [CrossRef]
- Vaitheeswaran, G.; Kanchana, V.; Kumar, R.S.; Cornelius, A.; Nicol, M.; Svane, A.; Delin, A.; Johansson, B. High-pressure structural, elastic, and electronic properties of the scintillator host material KMgF3. Phys. Rev. B 2007, 76, 14107. [Google Scholar] [CrossRef]
- Hayatullah; Naeem, S.; Murtaza, G.; Khenata, R.; Khalid, M. First principle study of CsSrM3 (M = F, Cl). Phys. B Condens. Matter 2013, 414, 91–96. [Google Scholar] [CrossRef]
- Mubarak, A. Ab initio study of the structural, electronic and optical properties of the fluoropervskite SrXF3 (X = Li, Na, K and Rb) compounds. Comput. Mater. Sci. 2014, 81, 478–482. [Google Scholar] [CrossRef]
- Mubarak, A.; Al-Omari, S. First-principles calculations of two cubic fluoropervskite compounds: RbFeF3 and RbNiF3. J. Magn. Magn. Mater. 2015, 382, 211–218. [Google Scholar] [CrossRef]
- Khan, R.; Althubeiti, K.; Algethami, M.; Rahman, N.; Sohail, M.; Mao, Q.; Zaman, Q.; Ullah, A.; Ilyas, N.; Afzal, A.M.; et al. Observation of Quan-tum Criticality in Antiferromagnetic based (Ce1−xYx)2Ir3Ge5 Kondo-Lattice System. J. Magn. Magn. Mater. 2022, 556, 169361. [Google Scholar] [CrossRef]
- Dimov, N.; Nishimura, A.; Chihara, K.; Kitajou, A.; Gocheva, I.D.; Okada, S. Transition metal NaMF3 compounds as model systems for studying the feasibility of ternary Li-M-F and Na-M-F single phases as cathodes for lithium–ion and sodium–ion batteries. Electrochim. Acta 2013, 110, 214–220. [Google Scholar] [CrossRef]
- Husain, M.; Sohail, M.; Rahman, N.; Khan, R.; Khan, A.A.; Elansary, H.O.; El-Abedin, T.K.Z.; Mahmoud, E.A.; Abdelmohsen, S.A.M.; Khan, A.Z. Exploring the exemplary structural, electronic, optical and elastic nature of inorganic ternary cubic fluoroperovskites XBaF3 (X = Al and Tl) employing the accurate TB-mBJ approach. Sci. Semicond. Sci. Technol. 2022, 37, 075004–075014. [Google Scholar] [CrossRef]
- Shimamura, K.; Fujita, T.; Sato, H.; Bensalah, A.; Sarukura, N.; Fukuda, T. Growth and Characterization of KMgF3 Single Crystals by the Czochralski Technique under CF4 Atmosphere. Jpn. J. Appl. Phys. 2000, 39, 6807–6809. [Google Scholar] [CrossRef]
- Bensalah, A.; Shimamura, K.; Nakano, K.; Fujita, T.; Fukuda, T. Growth and characterization of LiSrGaF6 single crystal. J. Cryst. Growth 2001, 231, 143–147. [Google Scholar] [CrossRef]
- Husain, M.; Ahmad, M.S.; Rahman, N.; Sajjad, M.; Rauf, A.; Habib, A.; Mahmood, H.; Nisar, M.; Hussain, A.; Imran, M.; et al. First principle study of the structural, electronic, and Mechanical properties of cubic fluoroperovskites: (ZnXF3, X = Y, Bi). Fluoride 2020, 53, 657–667. [Google Scholar]
- Rahman, N.; Husain, M.; Yang, J.; Sajjad, M.; Ahmad, M.S.; Habib, A.; Rauf, A.; Haq, M.U.; Saddique, J.; Nisar, M.; et al. Theoretical investigation of the structural, electronic, and mechanical properties of the magnesium-based fluoroperovskite compounds XMgF3 (X=Ga, Al, In). Fluoride 2020, 53, 542–553. [Google Scholar]
- Rahman, N.; Husain, M.; Yang, J.; Sajjad, M.; Murtaza, G.; Haq, M.U.; Habib, A.; Zulfiqar; Rauf, A.; Karim, A.; et al. First principle study of structural, electronic, optical and mechanical properties of cubic fluoro-perovskites: (CdXF3, X = Y, Bi). Eur. Phys. J. Plus 2021, 136, 374. [Google Scholar] [CrossRef]
- Harmel, M.; Khachai, H.; Haddou, A.; Khenata, R.; Murtaza, G.; Abbar, B.; Bin Omran, S.; Khalfa, M. Ab initio study of the mechanical, thermal and optoelectronic properties of the cubic CsBaF3. Acta Phys. Pol. 2015, 128, 34–42. [Google Scholar] [CrossRef]
- Daniel, D.J.; Madhusoodanan, U.; Nithya, R.; Ramasamy, P. Irradiation effect on luminescence properties of fluoroperovskite single crystal (LiBaF3:Eu2+). Radiat. Phys. Chem. 2013, 96, 135–139. [Google Scholar] [CrossRef]
- Charifi, Z.; Baaziz, H.; Hassan, F.E.H.; Bouarissa, N. High pressure study of structural and electronic properties of calcium chalcogenides. J. Phys. Condens. Matter 2005, 17, 4083–4092. [Google Scholar] [CrossRef]
- Ali, M.A.; Alam, N.; Meena; Ali, S.; Dar, S.A.; Khan, A.; Murtaza, G.; Laref, A. A theoretical study of the structural, thermoelectric, and spin-orbit coupling influenced optoelectronic properties of CsTmCl3 halide perovskite. Int. J. Quant. Chem. 2020, 120, 26141. [Google Scholar] [CrossRef]
- Mohamed, A.; El Houssine, A.; Nejmaa, F.; Ibrahim, B. Ab-initio study of electronic, optical and thermoelectric properties of TiO2 phases using mBJ approximation. In Proceedings of the 2020 IEEE 6th International Conference on Optimization and Applications, ICOA, Beni Mellal, Morocco, 20–21 April 2020; pp. 1–5. [Google Scholar]
- Mehl, M.J. Pressure dependence of the elastic moduli in aluminum-rich Al-Li compounds. Phys. Rev. B 1993, 47, 2493–2500. [Google Scholar] [CrossRef]
- Wang, J.; Yip, S.; Phillpot, S.R.; Wolf, D. Crystal instabilities at finite strain. Phys. Rev. Lett. 1993, 71, 4182–4185. [Google Scholar] [CrossRef]
- Hill, R. The Elastic Behaviour of a Crystalline Aggregate. Proc. Phys. Soc. Sect. A 1952, 65, 349. [Google Scholar] [CrossRef]
- Voigt, W. Lehrbuch der Kristallphysik (Textbook of Crystal Physics); BG Teubner: Leipzig/Berlin, Germany, 1928. [Google Scholar]
- Russ, A. Computation of the yield point of mixed crystals due to hiring for single crystals. Mater. Phys. 1929, 9, 49–58. [Google Scholar]
- Pettifor, D. Theoretical predictions of structure and related properties of intermetallics. Mater. Sci. Technol. 1992, 8, 345–349. [Google Scholar] [CrossRef]
- Pugh, S.F. XCII. Relations between the elastic moduli and the plastic properties of polycrystalline pure metals. Lond. Edinb. Dublin Philos. Mag. J. Sci. 1954, 45, 823–843. [Google Scholar] [CrossRef]
- Frantsevich, I. Elastic Constants and Elastic Moduli of Metals and Insulators; Reference Book; Naukova Dumka: Kiev, Ukraine, 1982. [Google Scholar]
- Penn, D.R. Electron mean-free-path calculations using a model dielectric function. Phys. Rev. B 1987, 35, 482–486. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16 Revision C.01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Tao, P.; Liu, S.; Wong, W. Phosphorescent Manganese(II) Complexes and Their Emerging Applications. Adv. Opt. Mater. 2020, 8, 2000985. [Google Scholar] [CrossRef]
- Madsen, G.K.; Blaha, P.; Schwarz, K.; Sjostedt, E. Efficient linearization of the augmented plane-wave method. Phys. Rev. B 2001, 64, 195134. [Google Scholar] [CrossRef]
- Maqbool, M.; Ahmad, I.; Richardson, H.; Kordesch, M. Direct ultraviolet excitation of an amorphous AlN: Praseodymium phosphor by codoped Gd3+ cathodoluminescence. Appl. Phys. Lett. 2007, 91, 193511. [Google Scholar] [CrossRef]
- Murtaza, G.; Ahmad, I. Shift of indirect to direct bandgap and optical response of LaAlO3 under pressure. J. Appl. Phys. 2012, 111, 123116. [Google Scholar] [CrossRef]
- Murtaza, G.; Ahmad, I.; Amin, B.; Maqssod, J.; Afaq, A.; Maqbool, M.; Khan, I.; Zahid, M. Investigation of structural and optoelectronic properties of BaThO3. Opt. Mater. 2011, 33, 553–557. [Google Scholar] [CrossRef]
- Saddique, J.; Husain, M.; Rahman, N.; Khan, R.; Zulfiqar; Iqbal, A.; Sohail, M.; Khattak, S.A.; Khan, S.N.; Khan, A.A.; et al. Modeling structural, elastic, electronic and optical properties of ternary cubic barium based fluoroperovskites MBaF3 (M = Ga and In) compounds based on DFT. Mater. Sci. Semicond. Process. 2022, 139, 106345. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Tran, F.; Blaha, P. Accurate Band Gaps of Semiconductors and Insulators with a Semilocal Exchange-Correlation Potential. Phys. Rev. Lett. 2009, 102, 226401. [Google Scholar] [CrossRef]
- Murnaghan, F.D. The Compressibility of Media under Extreme Pressures. Proc. Natl. Acad. Sci. USA 1944, 30, 244–247. [Google Scholar] [CrossRef]
- Jamal, M.; Bilal, M.; Ahmad, I.; Jalali-Asadabadi, S. IRelast package. J. Alloys Compd. 2018, 735, 569–579. [Google Scholar] [CrossRef]
- Bechhoefer, J. Kramers–kronig, bode, and the meaning of zero. Am. J. Phys. 2011, 79, 1053–1059. [Google Scholar] [CrossRef]
- Kim, C.C.; Garland, J.; Raccah, P. Modeling the optical dielectric function of the alloy system AlxGa1−xAs. Phys. Rev. B 1993, 47, 1876. [Google Scholar] [CrossRef]
- Dufek, P.; Blaha, P.; Schwarz, K. Applications of Engel and Vosko’s generalized gradient approximation in solids. Phys. Rev. B 1994, 50, 7279–7283. [Google Scholar] [CrossRef]
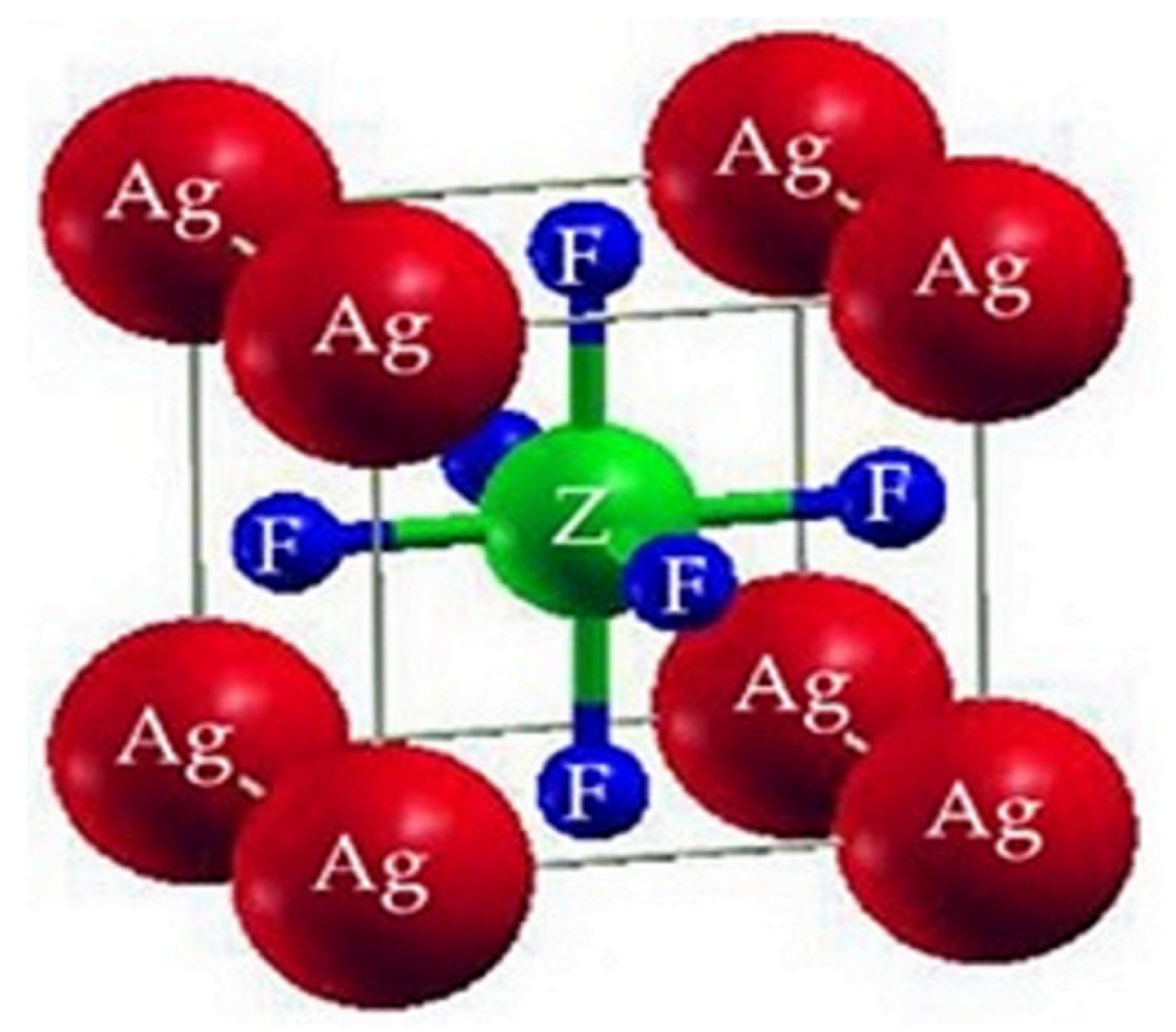
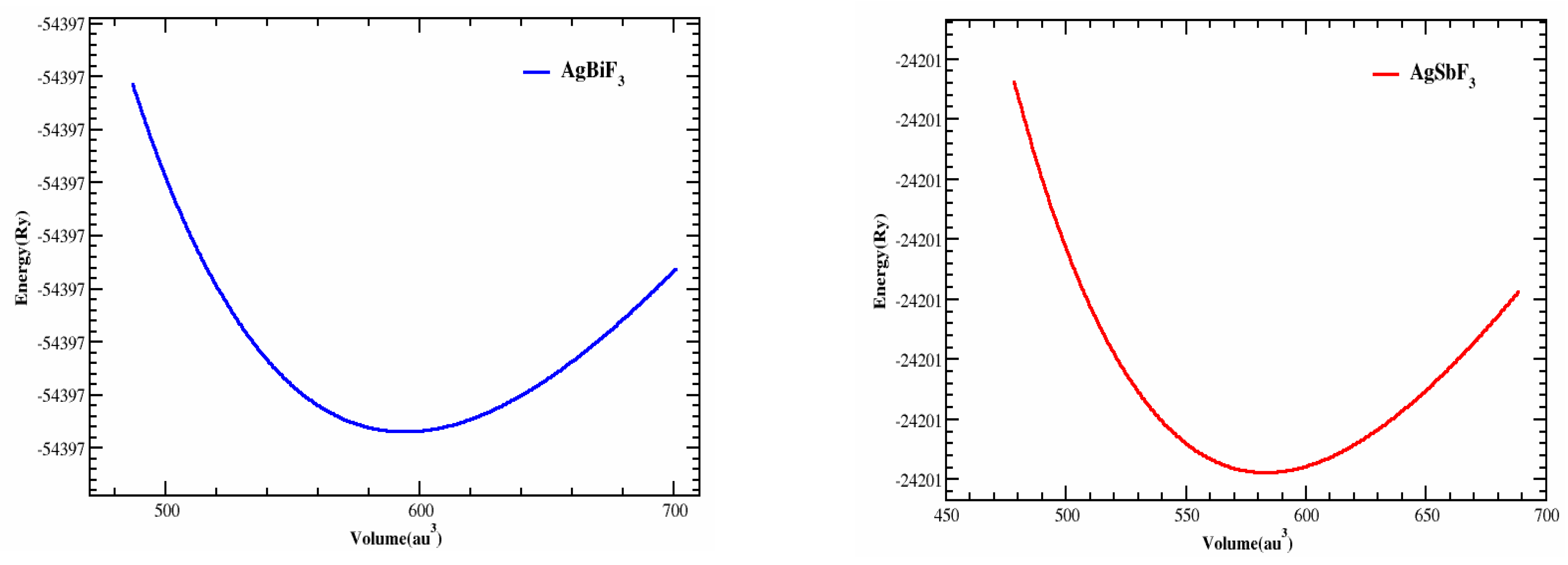
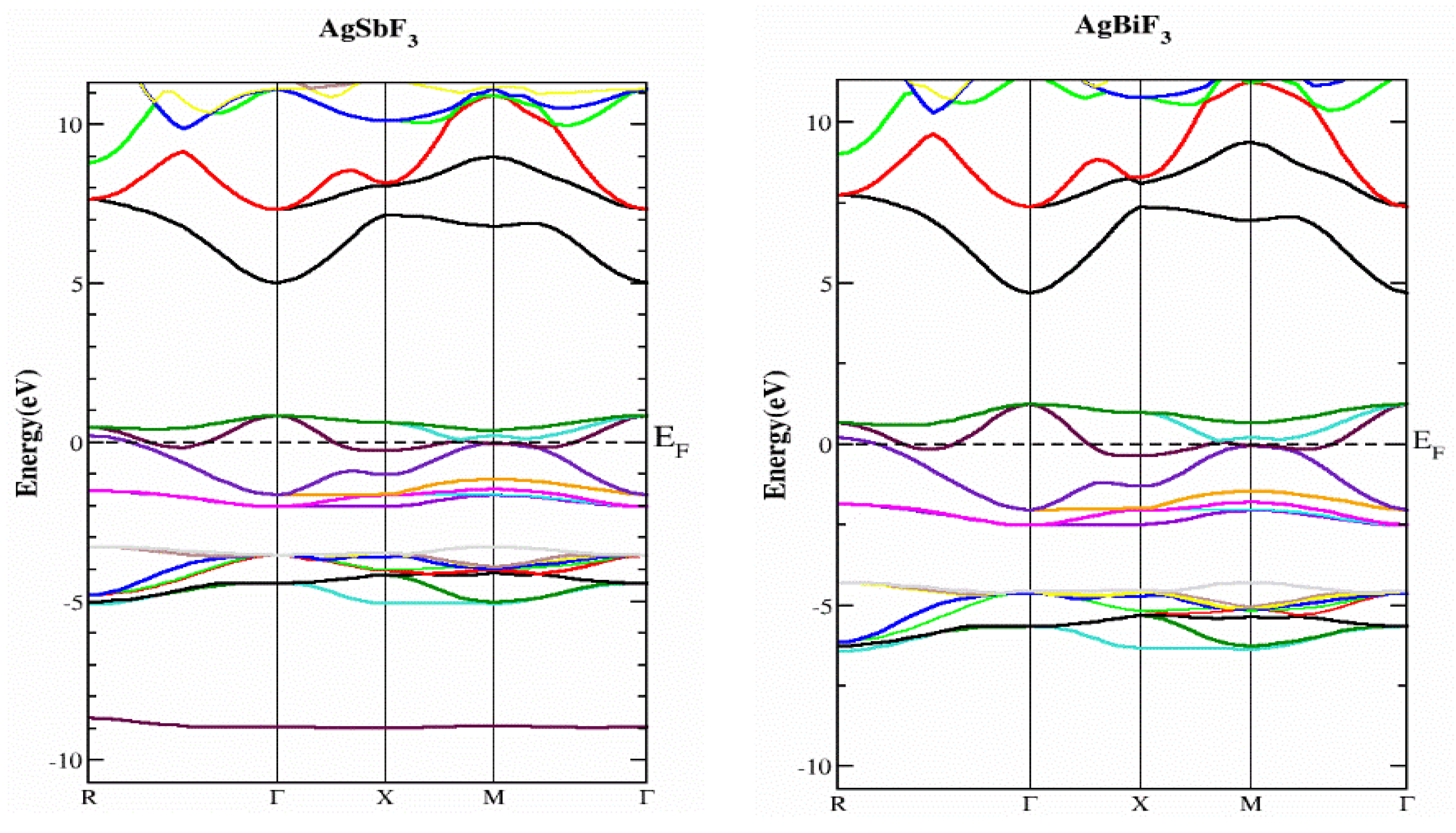
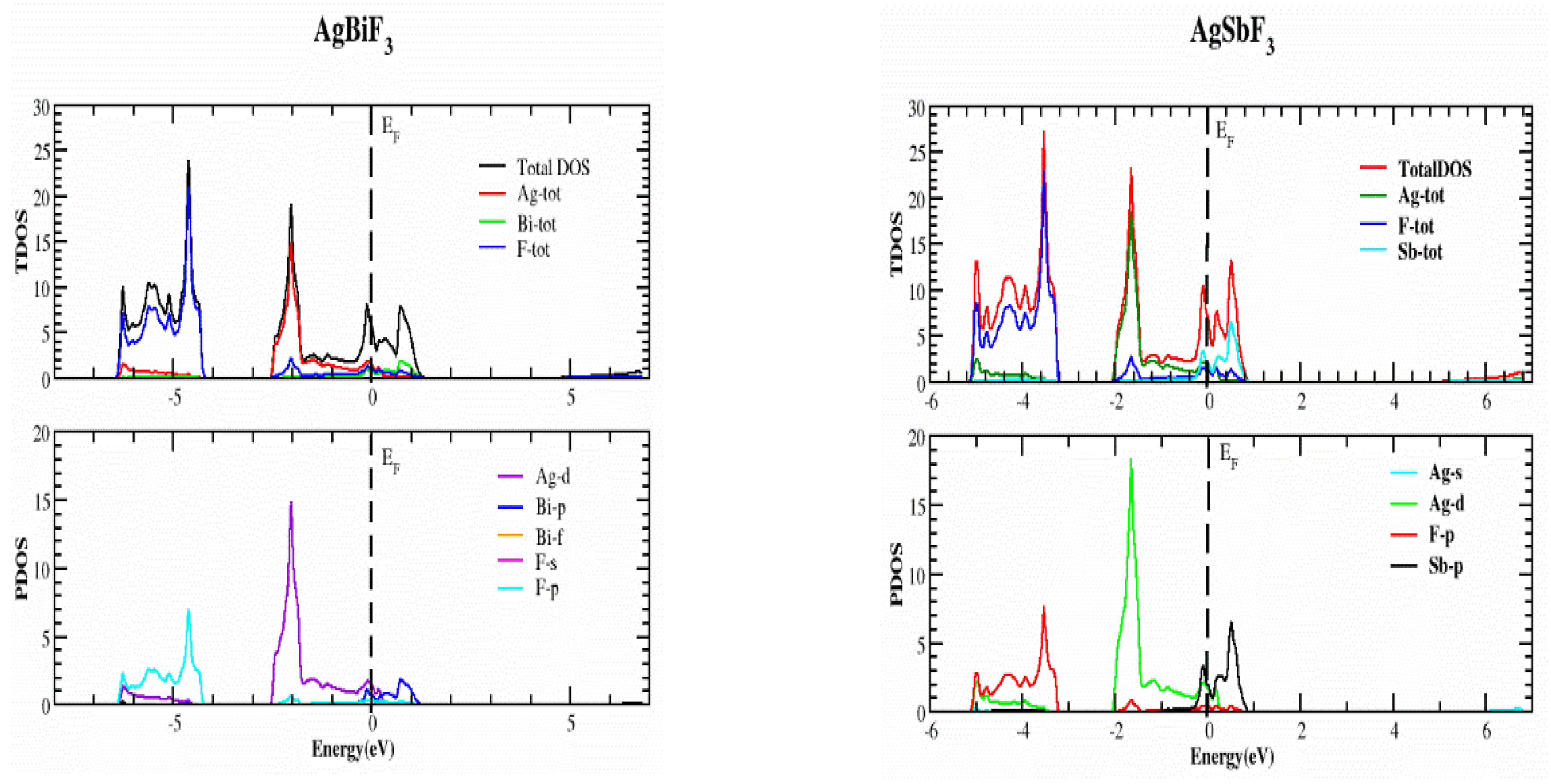

| Compounds | ao (Å) | B (GPa) | B’ | V0 (a.u3) | E0 (Ry) |
|---|---|---|---|---|---|
| AgSbF3 | 4.42 | 66.38 | 5.42 | 583.14 | −24,201.03 |
| AgBiF3 | 4.45 | 65.34 | 5.08 | 594.10 | −54,396.93 |
| Compounds | AgSbF3 | AgBiF3 |
|---|---|---|
| C11 | 14.61 | 69.74 |
| C12 | 46.02 | 67.09 |
| C44 | 7.64 | 11.12 |
| G | 6.60 | 5.01 |
| A | −0.37 | 8.39 |
| v | 0.528 | 0.45 |
| B/G | 10.07 | 13.06 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hedhili, F.; Khan, H.; Sohail, M.; Rahman, N.; Khan, R.; Alahmad, W.; Albaqawi, H.S.; Al-Shomar, S.M.; Alsalmi, O. Structural, Electronic, Elastic, and Optical Characteristics of AgZF3 (Z = Sb and Bi) Fluoro-Perovskites: Using a Computational Approach for Energy Generation. Molecules 2023, 28, 4418. https://doi.org/10.3390/molecules28114418
Hedhili F, Khan H, Sohail M, Rahman N, Khan R, Alahmad W, Albaqawi HS, Al-Shomar SM, Alsalmi O. Structural, Electronic, Elastic, and Optical Characteristics of AgZF3 (Z = Sb and Bi) Fluoro-Perovskites: Using a Computational Approach for Energy Generation. Molecules. 2023; 28(11):4418. https://doi.org/10.3390/molecules28114418
Chicago/Turabian StyleHedhili, Fekhra, Hukam Khan, Mohammad Sohail, Nasir Rahman, Rajwali Khan, Waed Alahmad, Hissah Saedoon Albaqawi, Shereen Mohammed Al-Shomar, and Omar Alsalmi. 2023. "Structural, Electronic, Elastic, and Optical Characteristics of AgZF3 (Z = Sb and Bi) Fluoro-Perovskites: Using a Computational Approach for Energy Generation" Molecules 28, no. 11: 4418. https://doi.org/10.3390/molecules28114418
APA StyleHedhili, F., Khan, H., Sohail, M., Rahman, N., Khan, R., Alahmad, W., Albaqawi, H. S., Al-Shomar, S. M., & Alsalmi, O. (2023). Structural, Electronic, Elastic, and Optical Characteristics of AgZF3 (Z = Sb and Bi) Fluoro-Perovskites: Using a Computational Approach for Energy Generation. Molecules, 28(11), 4418. https://doi.org/10.3390/molecules28114418







