1. Introduction
Newcastle disease (ND) is a highly contagious viral disease affecting many bird species. It is one of the most important and serious infections for the poultry industry, causing severe economic losses worldwide [
1,
2]. The causative agent of this serious infection is the Newcastle disease virus (NDV), a virulent form of Avian Paramyxovirus serotype1 (AMPV-1) belonging to the genus
Avulavirus, the family
Paramyxo viridae and the order
Mononega virales [
3]. It is an enveloped virus containing a single stranded, non-segmented, negative sense RNA genome, which encodes six structural proteins consisting of the nucleoprotein (NP), the phosphoprotein (P), the matrix protein (M), the fusion protein (F), the hemagglutinin-neuraminidase (HN) and the RNA polymerase (L) [
4].
The HN has three functions: (i) it recognizes sialic-acid-containing receptors on cell surfaces, (ii) promotes the fusion activity of the F protein, allowing the virus to penetrate the cell surface and (iii) acts as a neuraminidase (sialidase), removing sialic acids from progeny virus particles to prevent viral self-agglutination, thereby aiding viral spread [
3].
Vaccination is the current means for the control and prevention of NDV infection [
5]. However, despite the availability of various types of vaccines (live, inactivated and recombinant vaccines), NDV outbreaks are still occurring due to a lack of efficacious sanitary protocols and especially to mutations within viral strains, resulting in significant differences between current vaccine strains and field prevailing ones [
6,
7]. On the other hand, anti-NDV drugs are not available, due to their significant toxicity and high production cost [
8]. Therefore, the development of efficient antiviral molecules is crucial to fight NDV infections. Natural resources, such as animal venoms, have been shown to be endowed with anti-microbial effects [
9].
In particular, scorpion venom is a complex mixture containing neurotoxins, protease inhibitors, enzymes, antimicrobial peptides (AMPs) and some other compounds [
10,
11]. AMPs from scorpion venoms have demonstrated relevant activities against bacteria, fungi, and viruses [
11].
Scorpion venom AMPs are positively charged amphipathic peptides and can be conveniently divided into three structural categories: (1) cysteine containing peptides with disulfide bridges, called disulfide-bridged (DB) AMPs; (2) peptides with an amphipathic α-helix but lacking cysteine residues, called non-disulfide-bridged (NDB) AMPs and (3) peptides rich in certain amino acids, such as proline and glycine [
10].
More attention is being given to antiviral peptides, due to their unique biological activities that can make them potentially useful as antiviral drugs [
12]. Therefore, these studies highlight the diversity and the complexity of such arachnid venoms, which need to be explored for new antiviral candidates.
In this study, we screened Buthus occitanus tunetanus (Bot) scorpion venom fractions for their anti-NDV effect and identified a new peptide exhibiting in vitro and in ovo antiviral activities. It appears that this peptide belongs to a scorpion venom peptides family, not reported to exhibit a antimicrobial effect before now.
3. Materials and Methods
3.1. Chemicals and Reagents
The Bot scorpions were collected from Beni Khedach (South of Tunisia) by the veterinary services of the Institute Pasteur of Tunis. The venom was obtained through electric stimulation of scorpion post-abdomen and kept frozen at −20 °C, in its crude form, until used. Chemicals (reagent grade) were purchased from Sigma–Aldrich® (St. Louis, MO, USA) chemical company, except otherwise indicated. African green monkey kidney cells (Vero, ATCC CCL 85) were purchased from the American Type Culture Collection (ATCC, Boston, MA, USA) and the live virus vaccine ND (LaSota strain) (Cevac New L, Budapest, Hongrie) was used in all the assays. Cell culture supplements and reagents were purchased from GIBCO (Cergy-Pontoise, France); UltraPure™ Agarose was from Invitrogen (Waltham, MA, USA).
3.2. Purification
Crude venom (250 mg) from
Bot scorpion was dissolved in cold water (1:4
v/
v), and centrifuged at 15,000×
g for 15 min. The supernatant was loaded on Sephadex G-50 gel filtration chromatography column (K26/100), equilibrated at room temperature (25 °C) and eluted with 0.1 M acetic acid at a flow rate of 0.5 mL/min. The collection was carried out by an automatic fraction collector every 5 min with a volume collection of 2.5 mL per tube. The fractions pooling led to the obtaining of 4 fractions, as previously described [
23]. Only the fraction named BotG50, containing toxins with molecular mass ranging between 3000 Da and 7000 Da, was retained.
After freeze-drying, BotG50 was fractionated using FPLC on a cation exchange Resource S column (GE Healthcare, Uppsala, Sweden), pre-equilibrated by a solution buffer of 0.05 M ammonium acetate (pH 6.7). Molecules were eluted at a constant flow rate of 1 mL/min, using a linear gradient from 0.05 to 0.5 M ammonium acetate, for 58 min. Absorbance was monitored at 280 nm and fractions were collected manually and lyophilized, then dissolved in water. The obtained fractions were tested for their activities against NDV development. The fraction showing the best antiviral activity was purified on a C18 reversed phase HPLC column (250 × 10 mm, 5 μm; Beckman Fullerton, CA, USA), equilibrated in 0.1% trifluoro-acetic acid (TFA) in water. Peptides were eluted at a flow rate of 0.8 mL/min, using a linear gradient from 10 to 100% of solvent B (0.1% TFA in acetonitrile) in solvent A (0.1% TFA in water) in 45 min and collected according to their absorbance, monitored at 214 nm. Peptide concentration was determined by a BCA assay kit (Sigma-Aldrich, Burghausen, Germany), using Bovine Serum Albumin as a standard.
3.3. Mass Spectrometry and Amino Acid Sequence Determination
The H6 fraction was desalted by C18 ZipTip® (Millipore, Taufkirchen, Germany) and eluted directly into a 10 µL spray solution of acetonitrile: water: formic acid (75:25:0.1). An aliquot of H6 fraction was reduced with a 0.5M TCEP solution, then desalted with C18 ZipTip®.
A small amount (2–6 µL) was introduced into an Orbitrap Velos mass spectrometer, equipped with ETD module (Thermo Fisher Scientific, Bremen, Germany) using a TriVersaNanoMate® (Advion Biosciences, Ithaca, NY, USA) in positive ion mode. The spray voltage was set at 1.2–1.6 kV and the backpressure at 0.3–0.4 psi. A full set of automated positive ion calibrations was performed immediately prior to mass measurement. The transfer capillary temperature was set to 100 °C, the sheath and the auxiliary gasses switched off, and the source transfer parameters optimized using the auto tune feature. The FT automatic gain control (AGC) was set at 1 × 106 for MS experiments. The spectra were acquired in the FTMS in full profile mode with 10 micro-scans 1 min, with averaging on and set to max, and a resolution of 30,000 at m/z 400. For MS/MS experiments, the FT automatic gain control (AGC) was set at 2 × 105. Ions corresponding to the isotopic distribution of a single charge state were selected with the largest possible window, to avoid overlap with neighboring species but minimize signal loss. HCD (High Collision Dissociation) was performed at 30 eV and spectra were acquired in the FTMS in full profile mode at a resolution of 30,000 at m/z 400, with 10 microscans and with averaging on and set to the maximum value. The spectra were averaged using Qualbrowser in ThermoXcalibur 2.1 and deconvoluted using Xtract to produce zero charge mass spectra, and de novo sequencing was realized manually.
3.4. Cell Culture
African green monkey kidney cells (Vero, ATCC CCL 85) were grown as monolayers in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS) (Gibco, Cergy-Pontoise, France) and incubated at 37 °C with 5% CO2 in tissue culture flask, until total confluency.
3.5. Vero Cell Viability Assay
The effect of the venom peptide on cell viability was determined by the MTT (3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide) assay. Vero cells were grown at a cell density of 2 × 105 cells/well, in 96 well plates, and cultured at 37 °C for 24 h. The peptide was added to the wells at different concentrations (from 100 µg/mL to 5 µg/mL). After 72 h incubation, the medium was removed and 100 µL of MTT solution (5 mg/mL in PBS was added to each well, then re-incubated for 4 h at 37 °C. The supernatants were aspired out and 100 µL DMSO was added to solubilize the formazan crystals in each well. The absorbance was monitored in a reader plate (MULTISKAN, Labsystems, Vantaa, Finland) at 560 nm.
3.6. Virus Titration
The viral titer was assessed using a plaque reduction assay. Vero cell monolayers, seeded in 24 well plates, were infected with 10-fold dilutions of NDV suspension. After virus adsorption for 1h at 37 °C, the inoculum was removed. The cells were washed three times with PBS and overlaid with a medium containing 1.2% methylcellulose. After 72 h incubation at 37 °C, cells were fixed and stained with 1% crystal violet containing 20% methanol. The plaque viral titers were counted and expressed as plaque forming units (PFU), calculated according to the plaque numbers and dilution ratio [
24].
3.7. Viral Inactivation Assay
Screening of venom fraction on NDV inactivation was carried out by plaque reduction assay. Vero cell monolayers were cultivated in 24-well plates (105 cells/well) and incubated at 37 °C in 5% CO2. Each fraction was pre-incubated, at 10 µg/mL, with NDV (MOI = 0.001) for 2 h at 37 °C, then added to the Vero cell layers. After 2 h of incubation, cells were washed with PBS and overlaid with a medium containing 1.2% methylcellulose. After 72 h incubation at 37 °C, cells were fixed and stained with 1% crystal violet containing 20% methanol to count viral plaques. All antiviral assays were repeated three times. The concentration reducing 50% (EC50) of the plaque forming units was calculated by comparing treated and untreated cells (100% infectivity, as positive control), using GraphPad Prism software Version 4.0.
The most active peptide was tested at different concentrations (10 µg/mL; 5 µg/mL; 2.5 µg/mL; and 1.25 µg/mL). This peptide was then tested at the highest concentration (10 µg/mL), as described by Zhengyang et al. [
17], to study its effect on the different steps of the multiplication cycle of NDV.
3.7.1. Cell Protection Assay (Pretreatment Assay)
Vero cells were incubated in presence of the venom peptide for 2 h at 37 °C in 5% CO2. Subsequently, the cells were washed with PBS and infected with NDV at 0.001 MOI. After 2 h incubation, cells were washed with PBS then replenished with a cover layer as described above.
3.7.2. During Infection Assay
Vero cells were infected with the NDV (0.001 MOI) in the presence of the venom peptide (10 µg/mL), then incubated at 37 °C. After 2 h incubation, the cell layers were washed three times with PBS, and then replenished with a cover layer.
3.7.3. Post Treatment Assay
Vero cells were infected with NDV at MOI 0.001 for 2 h at 37 °C. Afterwards, cells were washed three times with PBS to remove unbound virus and then a cover layer containing the venom peptide was added.
3.8. Hemolysis Assays
The hemolytic activity of the venom peptide was assessed using chicken erythrocytes, according to the method of Maston [
25]. Blood was centrifuged for 5 min at 250×
g and washed three times with PBS, then resuspended to make a solution of 1% erythrocytes for the hemolytic assay. Serial dilutions from 100 µg/mL to 5 µg/mL of the scorpion venom peptide were mixed with the erythrocytes suspension and incubated for 45 min at 37 °C, then centrifuged at 250×
g for 5 min at 4 °C. The absorbance of the supernatants was measured at 545 nm. The negative control consisted of treatment with sterile PBS, while the positive control consisted of treatment with 0.1% Triton X-100.
3.9. In Ovo Toxicity Assay
The venom fraction (BotG50) as well as the active peptide, were evaluated at different concentrations for their effect on specific pathogen-free (SPF) embryos (ValoBio Media GmbH, Osterholz-Scharmbeck, Germany). The fraction/peptide were diluted in cell culture medium (DMEM) at 10 µg/mL, and then inoculated into 9-day-old embryonated SPF eggs via the allantoic route. Treated eggs were incubated at 37 °C and 80% humidity with a daily candling to examine the embryos’ viability. Dead embryos were immediately stored at 4 °C. The in ovo toxicity assay was repeated three times. After 5 days of incubation, the chicken embryos were photographed and examined.
3.10. In Ovo Antiviral Activity
The selected peptide was tested for its effect in reducing the infectivity of NDV in ovo. A mixture of 100 μL of the scorpion peptide (at 10 μg/mL) and 100 μL of NDV at 10
3 TCID 50/mL (Median Tissue Culture Infectious Dose/mL) was prepared and incubated at 37 °C for 2 h, then inoculated in the allantoic cavity of 9-day-old embryonated SPF eggs. Embryonated eggs inoculated with only the NDV were used as control. Treated SPF eggs were incubated at 37 °C and 80% humidity for 5 days with a daily candling to examine the embryos’ viability. Dead embryos were immediately stored at 4 °C. The allantoic fluids of live and dead embryos were aseptically harvested and stored at −80 °C until used for infectivity titration. To determine the embryo infectious dose (EID
50) titer, 200 µL of serial dilutions (10
−1 to 10
−10) of the harvested allantoic fluid of eggs treated with the scorpion peptide were inoculated to 9-day-old SPF embryonated eggs and incubated for 5 days at 37 °C and 80% humidity with daily candling. At the end of the incubation, the standard hemagglutination assay (HA) was carried out on harvested allantoic fluids to detect the presence of the viral antigens. The EID
50 was calculated according to the method of Reed and Muench [
26].
3.11. Hemagglutination Assay
The harvested allantoic fluid was added to the first well of a 96-well plate already containing 25 µL PBS per well. A two-fold dilution was prepared by mixing the suspension and transferring 25 µL to the next well. A volume of 25 µL of 5% (v/v) chicken blood suspension was added to each well and mixed gently before incubating the plate for 40 min at room temperature. Experiments were performed in triplicate using allantoic fluids from three eggs per treatment.
3.12. Sequence Analysis
In order to check the structural relationship between the chlorotoxin-like peptides and the reported scorpion venom AMPs, the sequences of all chlorotoxin-like peptides were first aligned only with antimicrobial scorpion venom peptides containing cysteines using the ClustalW program (
https://www.genome.jp/tools-bin/clustalw (accessed on 5 December 2022)) according to the default parameters (fast/rough alignments). The Clustalw starts with a pairwise sequence alignment, and then builds the tree of evolutionary relations between the sequences. A complementary phylogenetic analysis was then carried out by introducing antimicrobial peptide and antiviral sequences from scorpion venom with or without cysteines. The phylogenetic tree was generated by the Mega software package (version 3.1), according to the JTT (Jones–Taylor–Thornton) model. The algorithm of this template allows for sequence comparison based on approximate peptides, and defined sequences are clustered at the 85% level of identity. The closest sequence pairs are aligned, and the observed acid exchanges are taken into account in a matrix. All peptide sequences were taken from published reviews [
27,
28].
3.13. Protein–Protein Docking (In Silico Analyses)
In this study, docking analysis was established by a docking algorithm of HDOCK software (
http://hdock.phys.hust.edu.cn/ (accessed on 21 January 2023)) to predict structures of the complex scorpion toxin-HN of NDV. The crystal structure of the bind protein HN-NDV (PDB accession 3T1E) was taken from the Protein Data Bank (
https://www.rcsb.org/structure/3t1e (accessed on 21 January 2023)). The 3D structure model of AaCtx was obtained as reported in [
20]. Water molecules as well as other hetero-atoms were removed from the crystal structure of the HN-NDV structure before proceeding with the docking stage. The HDOCK server, for integrated protein–protein docking, automatically predicts their interaction through a hybrid algorithm of template-based and template-free docking. Both the cleaned bind protein and the ligand files were loaded on the web server and docked according to the default parameters. The top ten solutions, ranked according to the docking scores, were retained to select the best one, according to its binding site in HN-NDV, the binding affinity (ΔG) and the dissociation constants (Kd), calculated with the PRODIGY program [
29,
30]. The visualization of complexes as well as the electrostatic charge, were established with the PyMOL software (version 2.5).
3.14. Statistical Analysis
Statistical analysis was carried out with one-way ANOVA followed by Bonferroni test, using Graph-Pad Prism software (version 4.0). Numerical results were one representative of experiments repeated at least three times expressed as mean ± standard error of the mean (SEM). Significance was set at the 95% level. Statistical significance levels were defined as * p < 0.01, ** p < 0.001 and *** p < 0.0001. The EC50 and CC50 values were calculated with regression analysis using the software GraphPad Prism Version 4.0 (GraphPad Software, San Diego, CA, USA) by fitting a variable slope-sigmoid dose–effect curve. The selectivity index (SI) was calculated by dividing the CC50 by the EC50 value.
4. Discussion
The development of new AMPs can expand the existing databases of antimicrobials and give hope for a possible solution for animal and human health, in the face of emerging pandemics and antimicrobial resistance. The glycoprotein hemagglutinin-neuraminidase (HN) on the surface of the NDV is responsible for cell attachment, the promotion of fusion and the release of progeny virions. This multifunctional nature of HN makes it an attractive target for the development of inhibitors of NDV.
Interestingly, many amino acid residues of the HN-NDV binding site are conserved with that of human parainfluenza HN, suggesting that it could be used as a model for the structure-based design of potential inhibitors as treatments for different diseases [
22].
Many research studies have shown that scorpion venom compounds possess antiviral activities in vitro, and are considered as promising tools for developing effective antiviral drugs [
31,
32]. Hp1090 was the first reported natural scorpion venom antiviral peptide. It is an NDB peptide discovered through cDNA technology from the gland of
Heterometrus petersii scorpion. This peptide inhibits Hepatitis C virus infection in vitro with an IC
50 value of 7.62 µg/mL [
33]. Afterward, several other scorpion venom antiviral peptides were reported. However, no study has been reported for scorpion venom peptides with anti-NDV activity. In Tunisia, the
Buthus occitanus tunetatus (
Bot) scorpion is the most represented species and its venom is considered to be the most toxic after that of
Androctonus australis species. In this study, we screened the venom of
Bot scorpion and isolated an active peptide BotCl, with an antiviral effect against NDV. It exhibited a significant dose-dependent infectivity inhibition when incubated with NDV for 2 h at 37 °C, then inoculated to Vero cells with an IC
50 of 2.5 µg/mL (0.69 µM) (
Figure 3B).
In particular, we have demonstrated that BotCl acted directly on NDV to inhibit its viral infectivity (
Figure 5). BotCl acts by disrupting the structure of the virus particles, resulting in their inactivation and inability to infect host cells. This mechanism of action is effective against NDV, as it directly targets the virus particles outside of the host cells, preventing their entry into the cells and their multiplication.
Interestingly, the MTT assay showed that BotCl has no cytotoxic effect on Vero cells up to a concentration of 100 µg/mL (
Figure 3A). Furthermore, at this concentration, no hemolytic activity was observed when added to chicken blood (
Supplementary Data: Figure S1), contrary to the majority of scorpion AMPs, whose therapeutic value is limited by their hemolytic activity [
31]. These interesting results prompted us to evaluate the antiviral activity of BotCl in ovo. We found that BotCl exerted a significant inhibition on NDV growth by decreasing its viral titer in SPF chicken embryos by 73% (
Figure 7). Thus, all these activities demonstrate that BotCl has an antiviral activity.
On the other hand, according to the purification scheme, we noted that the fraction containing BotCl was retained by the Resource S cation-exchange chromatography column, indicating that it carried a positive charge. Furthermore, BotCl was also eluted late in the gradient of acetonitrile from the RP-HPLC column, showing that this peptide is hydrophobic. These characteristics are similar to all the AMPs, especially those identified from scorpion venoms.
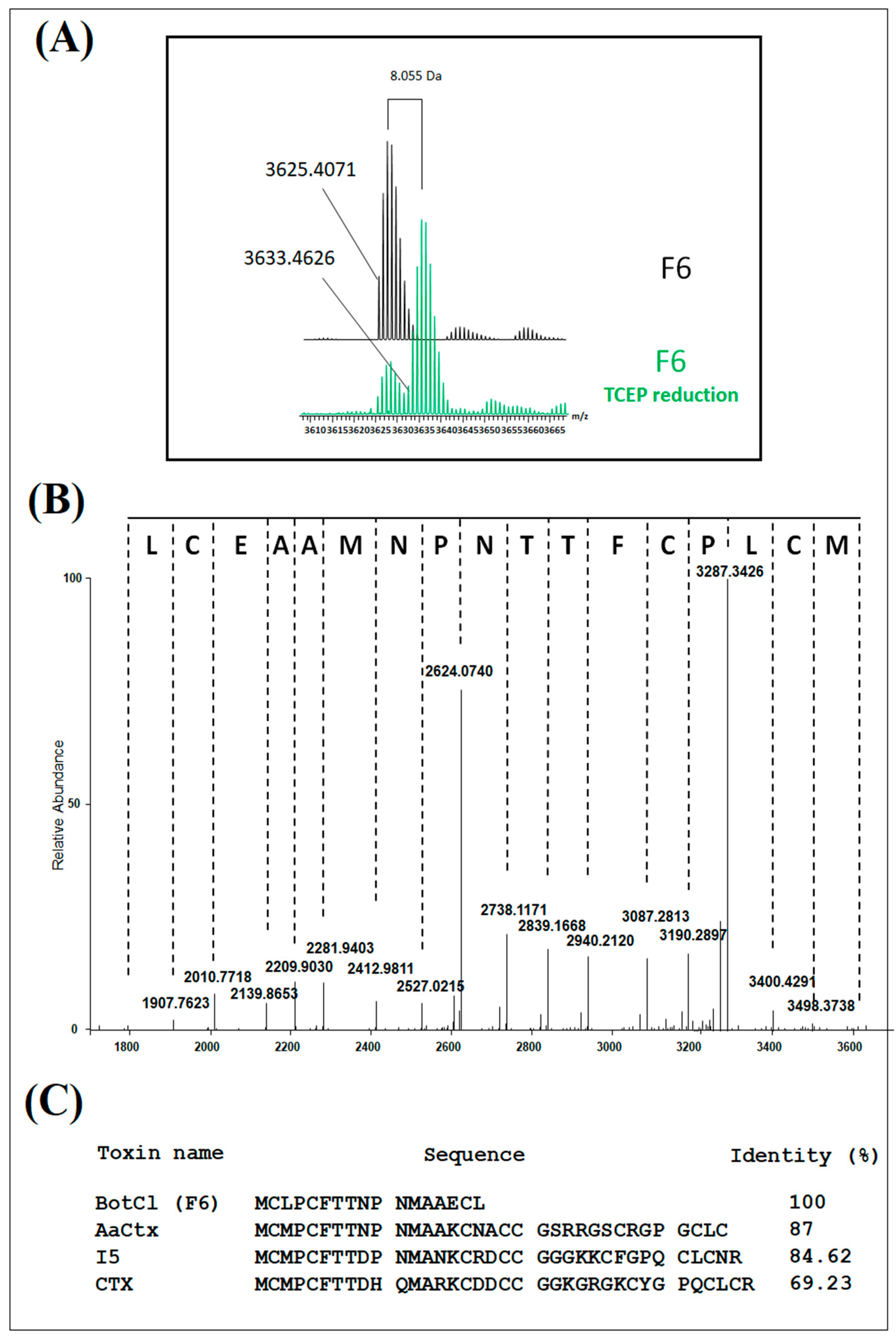
Interestingly, LC/MS analysis revealed the presence of four disulfide bonds, thus classifying BotCl in the family of DB-AMPs.
Interestingly, the molecular mass and the four disulphide bridges highlighted by mass spectrometry, as well as the N-terminal sequence of BotCl, showed that it belongs to the Chlorotoxin-like peptides family (
Figure 2).
Disulfide bonds often occur within well-defined structural motifs (such as the CSα/β or ICK motif), and typically have a role in stabilizing peptides by reducing their conformational flexibility. The disulfide bond connectivity in chlorotoxin was reported to be more like a CSα/β motif than an ICK motif [
34].
Chlorotxin-like peptides have been reported to be an interesting template for the development of diagnostic and therapeutic agents for cancer, but as far as we know no antimicrobial effect was reported for any of these peptides. Thus, BotCl is the first chlorotoxin-like peptide shown to exhibit antiviral activity.
Since we have already isolated AaCtx, another chlorotoxin-like peptide from scorpion Aa venom, we tested it at a concentration of 10 µg/mL and found that it had a three-fold lower effect than BotCl at the same concentration. This result suggests that the other chlorotoxin-like peptides may have an anti-NVD effect, with different affinities.
Given the lack of anti-NDV scorpion peptides, the activity of BotCl could be compared to the few identified disulfide-bridged antiviral peptides such as Smp76, isolated from
Scorpiomaurus palmatus, which inhibits the ability of Hepatitis C virus (HCV) to infect the host cells with an IC
50 of 0.01 µg/mL [
28]. EV37 is a scorpine-like peptide of 78 amino acid residues, including the CSα/β motif. It inhibited DENV-2 infection and suppressed HCV and ZIKV infections with IC
50 of 10 µM [
35]. BmKDfsin3 is composed of 38 amino acid residues, with 3 disulfide bonds. It inhibits HCV replication and affects the attachment and post-entry stages of the viral infection cycle with an IC
50 of 3.35 µM [
27]. The BmKDfsin4 inhibits the activity of HBV in HepG2.2.15 cells with IC
50 values of 1.26–3.95 µM against the production of extracellular HBsAg and HBeAg. Thus, with its IC
50 of 0.69 µM, BotCl could be considered as a potent antiviral scorpion toxin, compared to these peptides.
However, there remains the question regarding the functionality of BotCl in
Bot scorpion venom, knowing that scorpions are not subject to NDV infections. Scorpion venomous gland contains a wide range of biological active molecules, including AMPs [
3,
4], that are believed to be an integral component of an innate immune system that serves to protect the scorpion and its gland against a variety of pathogens [
36,
37] and facilitate the action of other neurotoxins [
11]. Consequently, and given the conservation of haemagglutinin neuraminidase glycoproteins on the surface of different viruses, BotCl could probably block other viral infections.
As both BotCl and AaCtx from the chlorotoxin-like peptides family showed an antiviral effect, and assuming that structural homology leads, generally, to the exhibition of homologous functional properties, we can presume that the other chlorotoxin-like peptides could also have antiviral effects. Thus, we studied the potential structural relationship of the Chlorotoxin-like peptides with the reported scorpion venom DB-AMPs. The sequence alignment showed that the primary structures of Chlorotoxin-like peptides could be definitely aligned with those of the scorpion venom DB-AMPs. Particularly, we found that five of the eight cysteine residues of the chlorotoxin-like peptides were conserved in all the sequences. Another cysteine residue (Cys5 of ClTx)was also conserved, except with HgebKTx. Thus, according to the sequence alignment reported in
Figure 8A, it is obvious that the chlorotoxin-like peptides are structurally related to the reported scorpion venom DB-AMPs. The reinforcement of the arguments in favor of the structural relationship between Chlorotoxin-like peptides and AMPs was taken advantage of by a phylogenetic tree (
Figure 8B). Indeed, it is obvious that the evolutionary relationship between the sequences of chlorotoxin-like peptides and the sequences of scorpion venom DB- or NDB-AMPs shown in
Figure 8B is one of the effective ways to prove the existence of a new family of AMPs represented by the ClTx.
Since AaCtx has shown an inhibiting effect on NDV infectivity, and in order to identify and characterize the putative mode of interaction between the chlorotoxin-like peptide and the NDV, a blind docking was carried out with the crystal structure of the catalytically active head region (residues 124 to 569) of HN from the Kansas strain of NDV [
7]. HN has a uniquely large cavity around the O4 position of its substrate, which is lined with residues that are largely conserved across all HNs and play a functional role of fusion upon binding receptors [
38,
39].
The most reliable solution of the docking showed that AaCtx binds the cavity of the HN-NDV binding site, mainly with the loop connecting the α-helix to the β-sheet (
Figure 9A). The most important interaction was that of G21 of AaCtx with R416 (
Figure 9B). Interestingly, this later belonged to the tri-arginyl cluster (R174, R416 and R498), identified as a crucial triad for the activity of HN-NDV, and their mutations severely reduced HN activity [
22]. Interestingly, R498 is also implied in an important interaction with A13. Other interactions are important in the complex, such as that of R23 and R24 with E401/S237 and E258 of HN-NDV, respectively. In fact, the E258 and Y262 lie on a helix that is stabilized by the calcium ion, and it is interesting to note that the removal of Ca
2+ completely abolishes enzyme activity [
22].
Interestingly, the AaCtx interaction with HN-NDV could be compared to that of Neu5Ac2en, a HN inhibitor [
22]. This inhibitor binds the tri-arginyl residues, as well as I175, S237, E401, E547, E258 and Y526 of the HN active site [
22,
40]. These interactions were found in AaCtx, mainly by its R23 (with I175, S237 and E401), S22 (with R416, E401 and Y526 Y526) and R24 (with E258) residues. Thus, the interaction model of AaCtx with NDV was in accordance with its antiviral effect.