Mg-LDH Nanoclays Intercalated Fennel and Green Tea Active Ingredient: Field and Laboratory Evaluation of Insecticidal Activities against Culex pipiens and Their Non-Target Organisms
Abstract
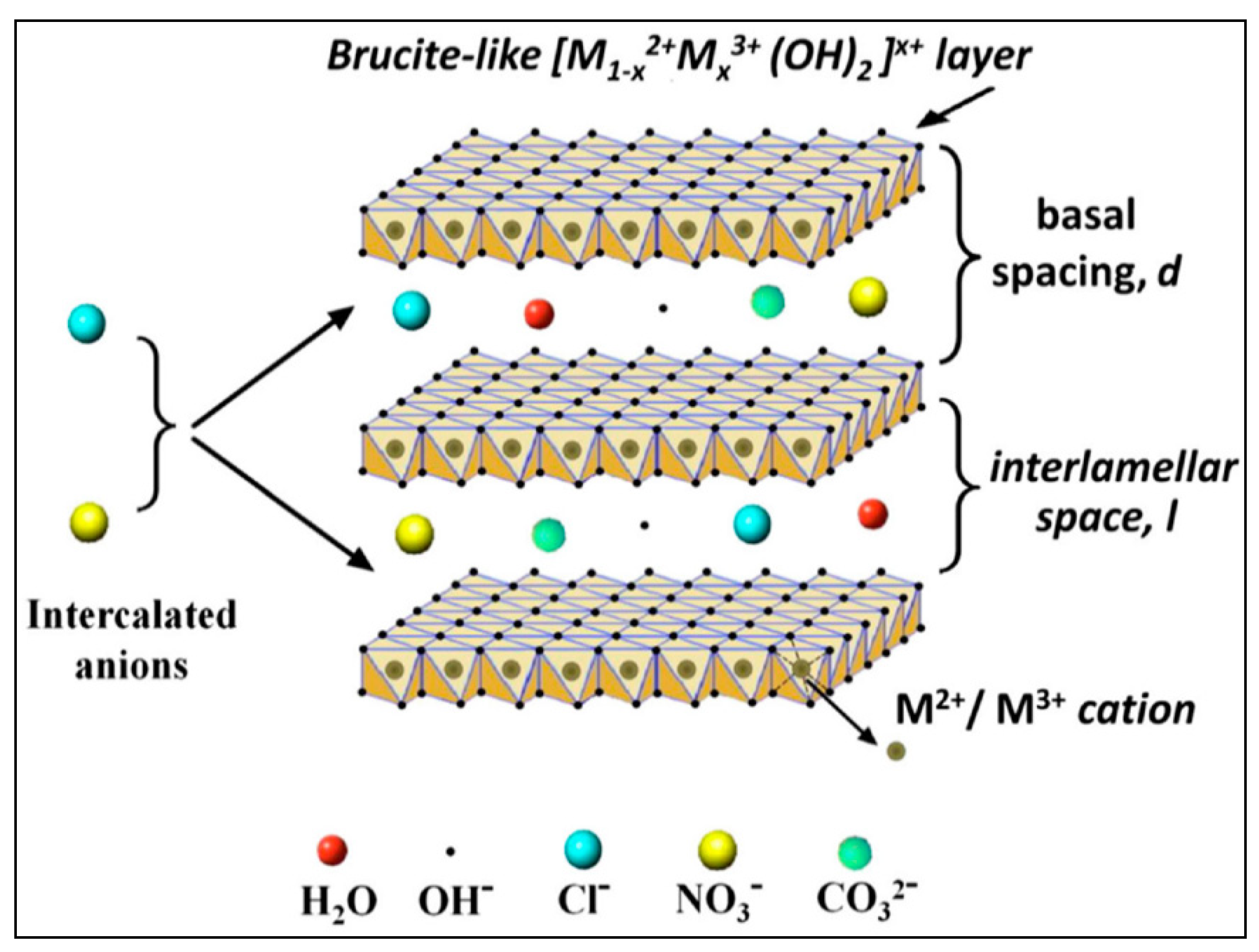
:1. Introduction
2. Materials and Methods
2.1. Chemistry
2.1.1. Materials
2.1.2. Synthesis of Mg and Ni Al-LDHs
2.1.3. Characterization of LDH
2.1.4. Adsorption of Active Ingredient Study
2.1.5. Adsorption Data Analysis and Drug Loading
2.2. Entomology Work
2.2.1. Culex pipiens
2.2.2. In Vitro Larvicidal Efficacy
2.2.3. Larvicidal Field Evaluation
2.2.4. In Vitro Adulticidal Efficacy
2.2.5. Adulticidal Field Evaluation
2.2.6. The Efficacy against Non-Target Predators
2.2.7. Data Analysis
3. Results
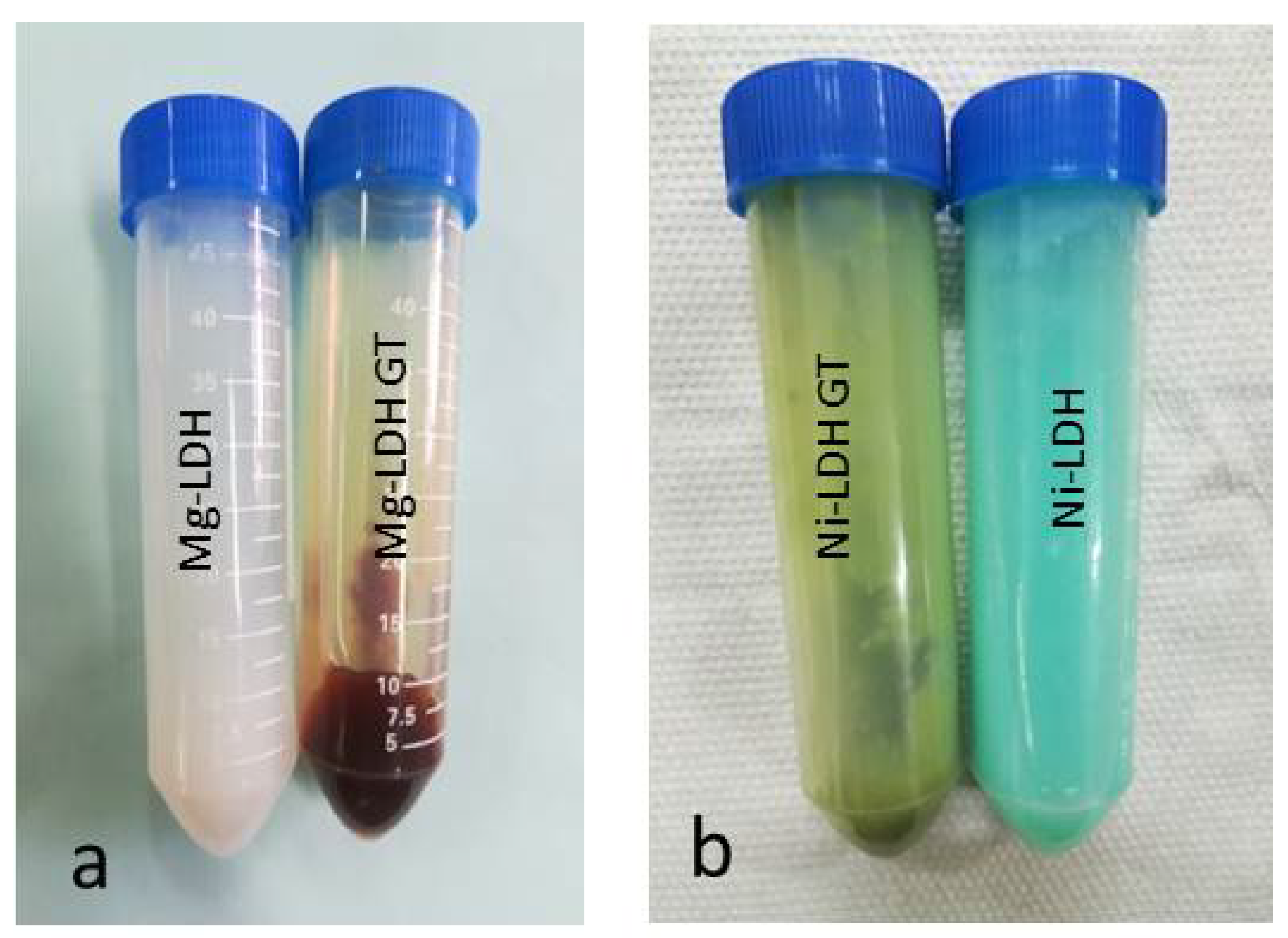
3.1. Synthesis of M(II)LDH
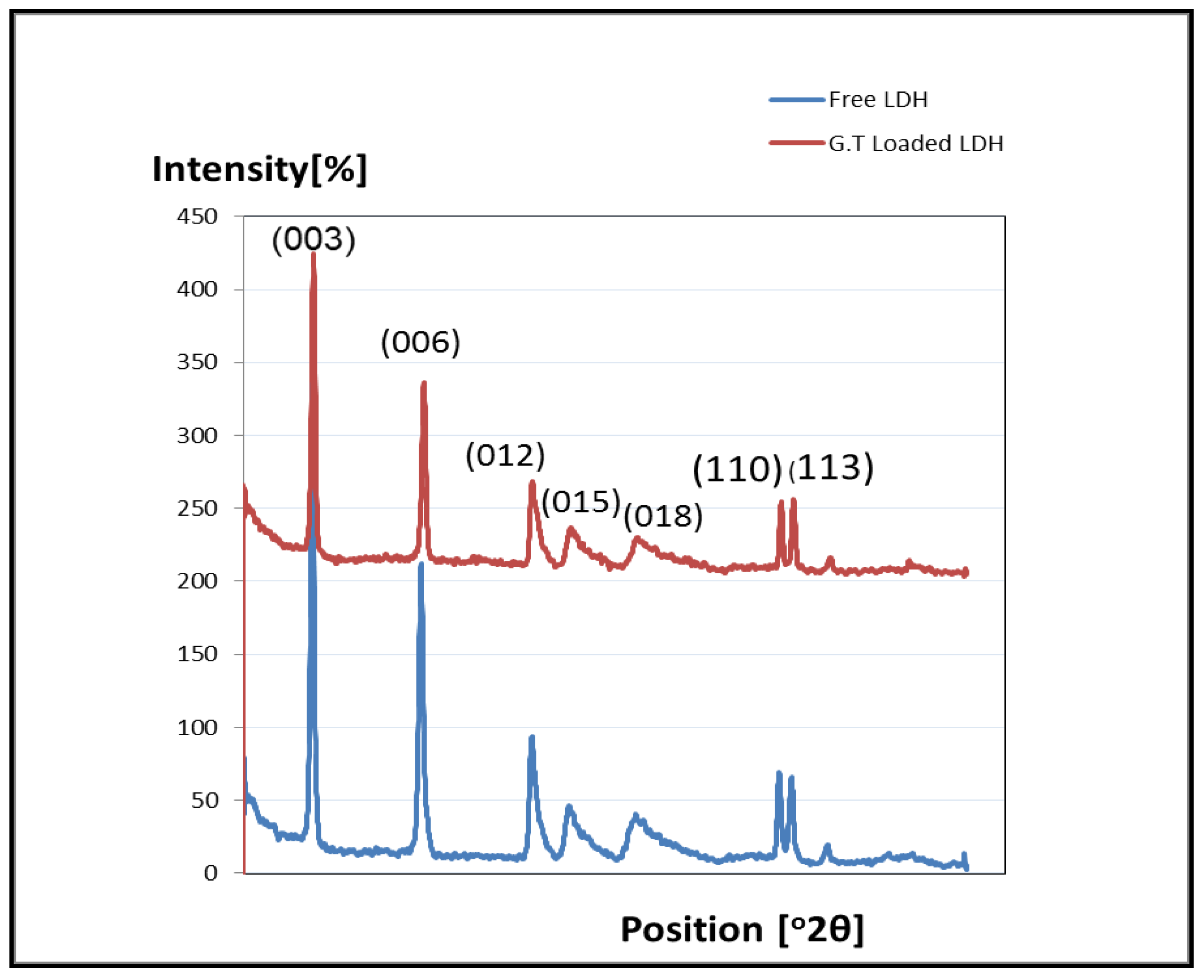
3.1.1. Characterization of M(II)LDH
3.1.2. Desorption of Active Ingredient and Drug Loading
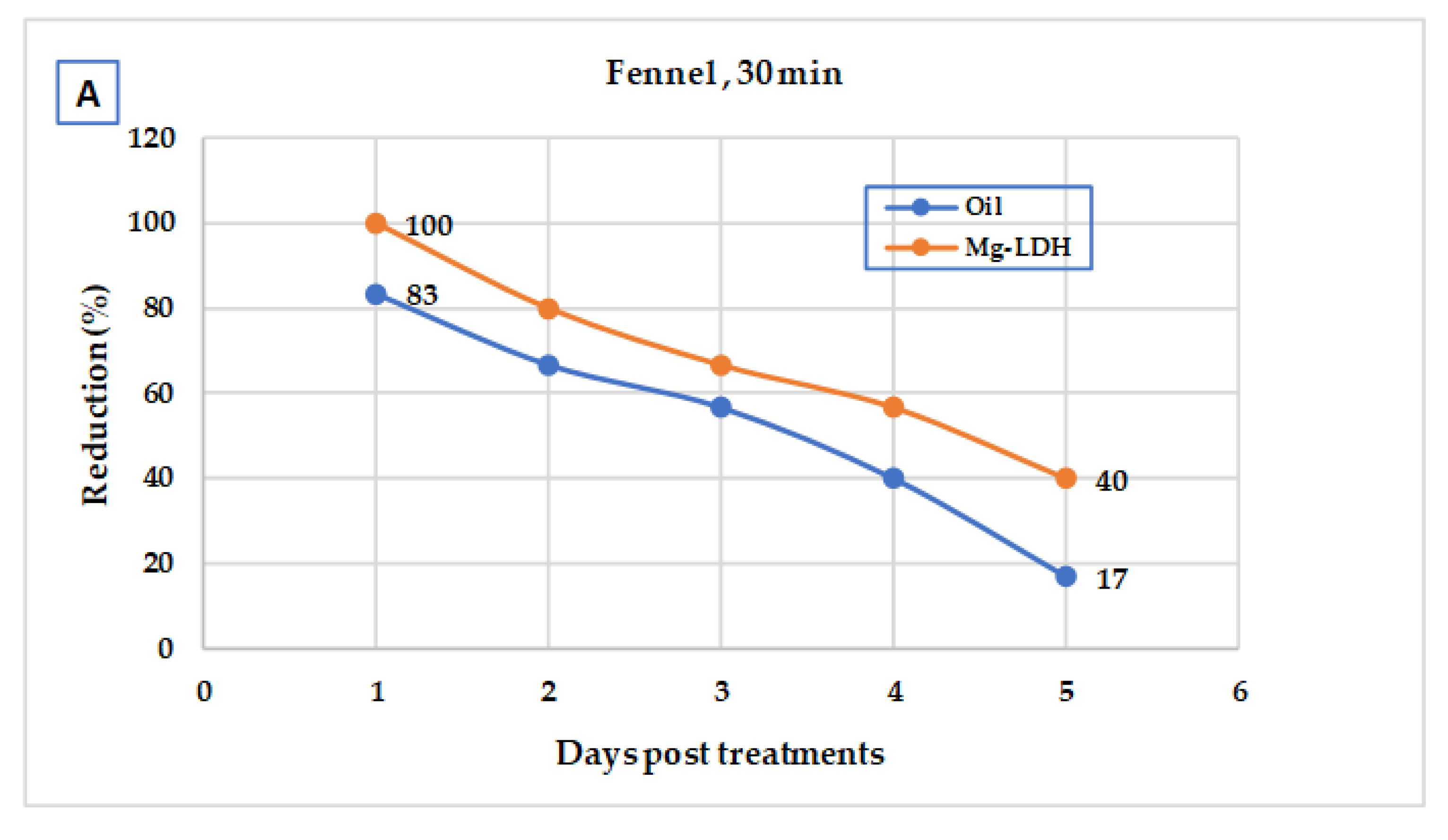
3.2. Larval Laboratory Experiments
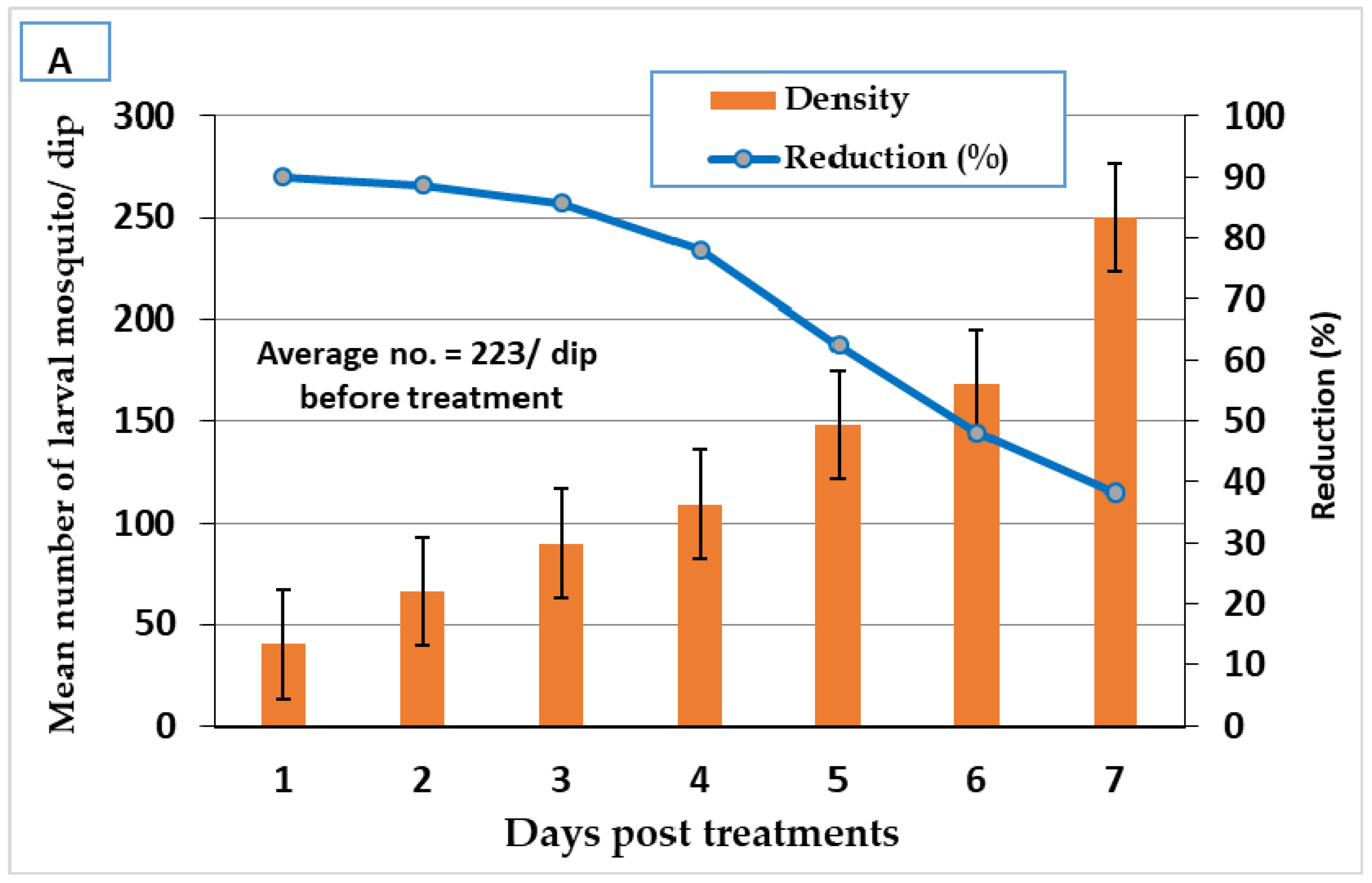
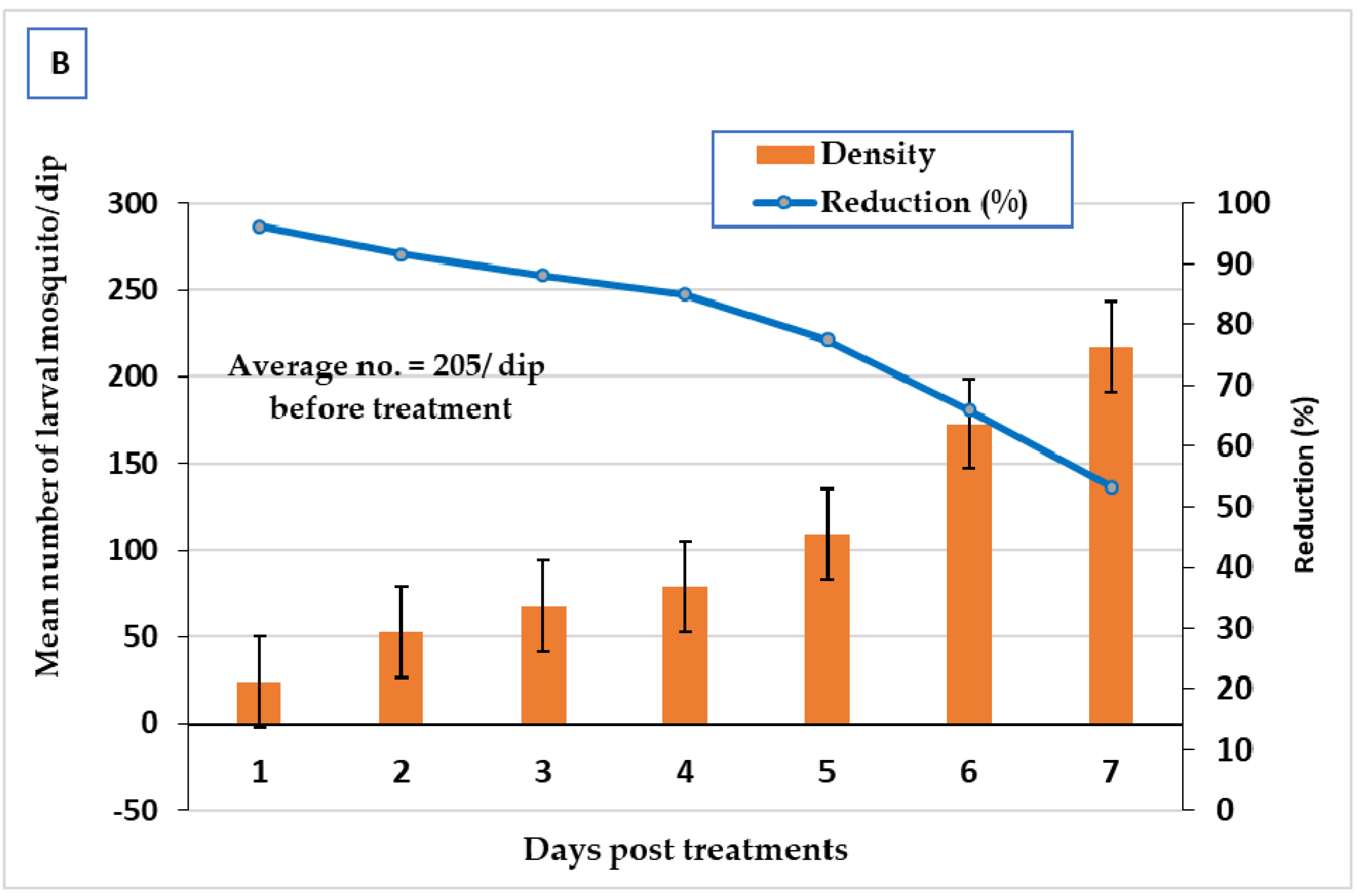
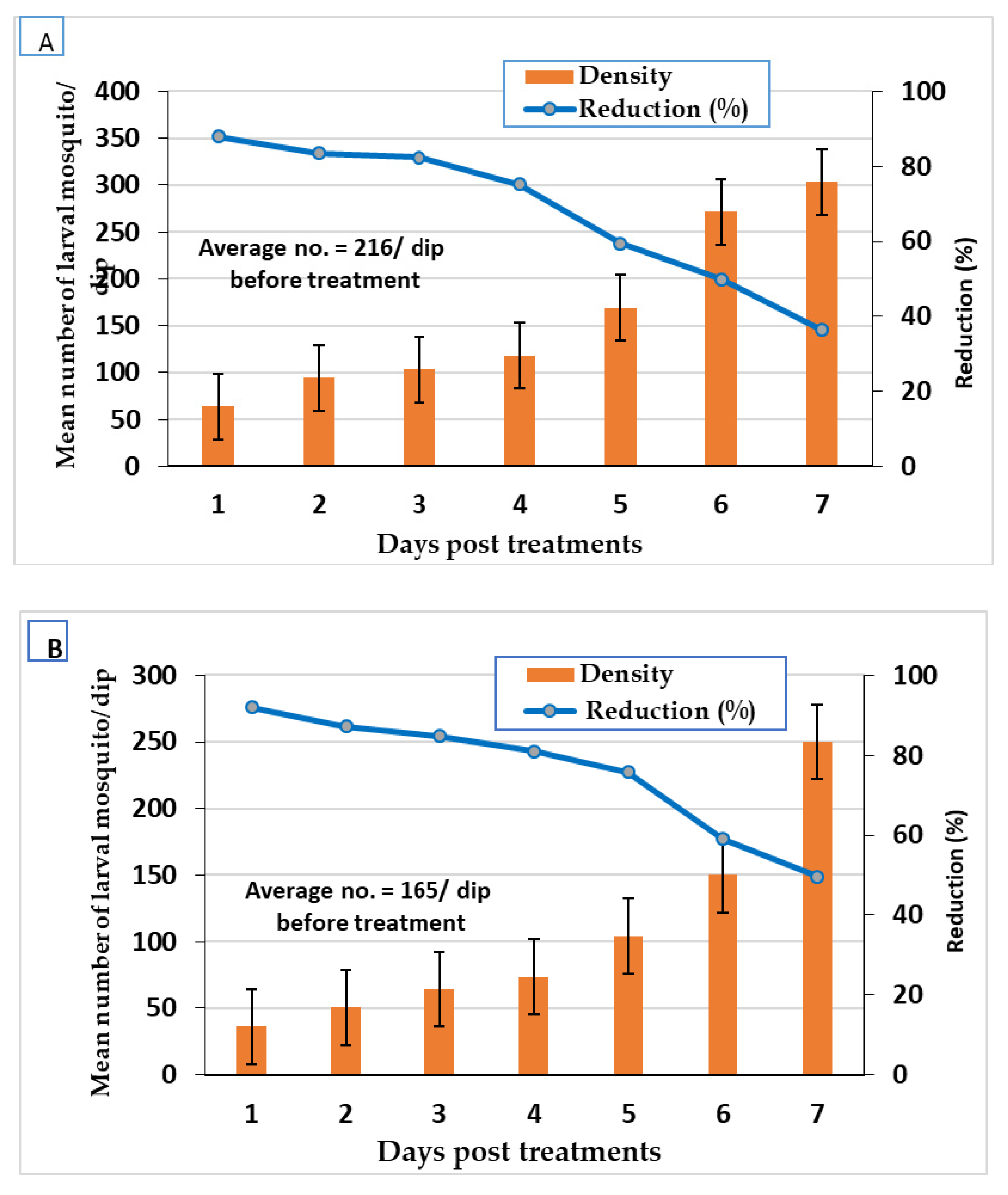
3.3. Larvicidal Field Evaluation
3.4. In Vitro Adulticidal Effect
3.5. Adult Field Experiments
3.6. The Efficacy against Non-Target Predators
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Li, F.; Duan, X. Applications of layered double hydroxides. ChemInform 2006, 37, 193–223. [Google Scholar]
- Evans, D.G.; Duan, X. Preparation of layered double hydroxides and their applications as additives in polymers, as precursors to magnetic materials and in biology and medicine. Chem. Commun. 2006, 5, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Mayadevi, S. Adsorption of fluoride ions by Zn–Al layered double hydroxides. Appl. Clay Sci. 2008, 40, 54–62. [Google Scholar] [CrossRef]
- McKenzie, A.L.; Fishel, C.T.; Davis, R.J. Investigation of the surface structure and basic properties of calcined hydrotalcites. J. Catal. 1992, 138, 547–561. [Google Scholar] [CrossRef]
- Schaper, H.; Berg-Slot, J.; Stork, W. Stabilized magnesia: A novel catalyst (support) material. Appl. Catal. 1989, 54, 79–90. [Google Scholar] [CrossRef]
- Ishikawa, T.; Matsumoto, K.; Kandori, K.; Nakayama, T. Anion-exchange and thermal change of layered zinc hydroxides formed in the presence of Al (III). Colloids Surf. A Physicochem. Eng. Asp. 2007, 293, 135–145. [Google Scholar] [CrossRef]
- Boclair, J.W.; Braterman, P.S. Layered double hydroxide stability: 1 Relative stabilities of layered double hydroxides and their simple counterparts. Chem. Mater. 1999, 11, 298–302. [Google Scholar] [CrossRef]
- Das, J.; Das, D.; Parida, K. Preparation and characterization of Mg–Al hydrotalcite-like compounds containing cerium. J. Colloid Interface Sci. 2006, 301, 569–574. [Google Scholar] [CrossRef]
- Prasanna, S.V.; Kamath, P.V. Chromate uptake characteristics of the pristine layered double hydroxides of Mg with Al. Solid State Sci. 2008, 10, 260–266. [Google Scholar] [CrossRef] [Green Version]
- Cantú, M.; López-Salinas, E.; Valente, J.S.; Montiel, R. SOx removal by calcined MgAlFe hydrotalcite-like materials: Effect of the chemical composition and the cerium incorporation method. Environ. Sci. Technol. 2005, 39, 9715–9720. [Google Scholar] [CrossRef]
- Soares, J.L.; Moreira, R.F.; José, H.J.; Grande, C.A.; Rodrigues, A.E. Hydrotalcite materials for carbon dioxide adsorption at high temperatures: Characterization and diffusivity measurements. Sep. Sci. Technol. 2005, 39, 1989–2010. [Google Scholar] [CrossRef]
- Yamamoto, T.; Kodama, T.; Hasegawa, N.; Tsuji, M.; Tamaura, Y. Synthesis of hydrotalcite with high layer charge for CO2 adsorbent. Energy Convers. Manag. 1995, 36, 637–640. [Google Scholar] [CrossRef]
- Yu, J.J.; Jiang, Z.; Zhu, L.; Hao, Z.P.; Xu, Z.P. Adsorption/desorption studies of NOx on well-mixed oxides derived from Co–Mg/Al hydrotalcite-like compounds. J. Phys. Chem. B 2006, 110, 4291–4300. [Google Scholar] [CrossRef] [Green Version]
- Das, N.; Konar, J.; Mohanta, M.; Srivastava, S. Adsorption of Cr (VI) and Se (IV) from their aqueous solutions onto Zr4+-substituted ZnAl/MgAl-layered double hydroxides: Effect of Zr4+ substitution in the layer. J. Colloid Interface Sci. 2004, 270, 1–8. [Google Scholar] [CrossRef]
- Dousova, B.; Machovic, V.; Kolousek, D.; Kovanda, F.; Dornicak, V. Sorption of As (V) species from aqueous systems. Water Air Soil Pollut. 2003, 149, 251. [Google Scholar] [CrossRef]
- Wang, H.; Chen, J.; Cai, Y.; Ji, J.; Liu, L.; Teng, H.H. Defluoridation of drinking water by Mg/Al hydrotalcite-like compounds and their calcined products. Appl. Clay Sci. 2007, 35, 59–66. [Google Scholar] [CrossRef]
- Choudary, B.; Kantam, M.L.; Reddy, C.V.; Rao, K.K.; Figueras, F. Henry reactions catalysed by modified Mg–Al hydrotalcite: An efficient reusable solid base for selective synthesis of β-nitroalkanols. Green Chem. 1999, 1, 187–189. [Google Scholar] [CrossRef]
- Cwik, A.; Fuchs, A.; Hell, Z.; Clacens, J.-M. An efficient and environmental-friendly synthesis of 4-hydroxy-arylpiperidines using hydrotalcite catalyst. J. Mol. Catal. A Chem. 2004, 219, 377–381. [Google Scholar] [CrossRef]
- Sels, B.F.; De Vos, D.E.; Jacobs, P.A. Hydrotalcite-like anionic clays in catalytic organic reactions. Catal. Rev. 2001, 43, 443–488. [Google Scholar] [CrossRef]
- Arrabito, G.; Bonasera, A.; Prestopino, G.; Orsini, A.; Mattoccia, A.; Martinelli, E.; Pignataro, B.; Medaglia, P.G. Layered double hydroxides: A toolbox for chemistry and biology. Crystals 2019, 9, 361. [Google Scholar] [CrossRef] [Green Version]
- Jones, R.T.; Ant, T.H.; Cameron, M.M.; Logan, J.G. Novel control strategies for mosquito-borne diseases. Philos. Trans. R. Soc. B Biol. Sci. 2021, 376, 20190802. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Shafi, I.R.; Shoeib, E.Y.; Attia, S.S.; Rubio, J.M.; Edmardash, Y.; El-Badry, A.A. Mosquito identification and molecular xenomonitoring of lymphatic filariasis in selected endemic areas in Giza and Qualioubiya Governorates, Egypt. J. Egypt. Soc. Parasitol. 2016, 46, 93–100. [Google Scholar] [PubMed] [Green Version]
- Cano, J.; Rebollo, M.P.; Golding, N.; Pullan, R.L.; Crellen, T.; Soler, A.; Kelly-Hope, L.A.; Lindsay, S.W.; Hay, S.I.; Bockarie, M.J. The global distribution and transmission limits of lymphatic filariasis: Past and present. Parasites Vectors 2014, 7, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Selim, A.; Radwan, A.; Arnaout, F.; Khater, H. The Recent Update of the Situation of West Nile Fever among Equids in Egypt after Three Decades of Missing Information. Pak. Vet. J. 2020, 40, 390–393. [Google Scholar]
- Govindarajan, M.; Rajeswary, M.; Hoti, S.; Bhattacharyya, A.; Benelli, G. Eugenol, α-pinene and β-caryophyllene from Plectranthus barbatus essential oil as eco-friendly larvicides against malaria, dengue and Japanese encephalitis mosquito vectors. Parasitol. Res. 2016, 115, 807–815. [Google Scholar] [CrossRef]
- Khater, H.F. Prospects of botanical biopesticides in insect pest management. Pharmacologia 2012, 3, 641–656. [Google Scholar]
- Khater, H.F. Bioactivity of essential oils as green biopesticides: Recent global scenario. Recent Prog. Med. Plants 2013, 37, 151–218. [Google Scholar]
- Noutcha, M.A.; Edwin-Wosu, N.I.; Ogali, R.E.; Okiwelu, S.N. The role of plant essential oils in mosquito (Diptera: Culicidae) control. Annu. Res. Rev. Biol. 2016, 10, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, N.; Alam, M.; Saeed, M.; Ullah, H.; Iqbal, T.; Al-Mutairi, K.A.; Shahjeer, K.; Ullah, R.; Ahmed, S.; Ahmed, N.A.A.H.; et al. Botanical Insecticides Are a Non-Toxic Alternative to Conventional Pesticides in the Control of Insects and Pests. In Global Decline of Insects; IntechOpen: London, UK, 2021. [Google Scholar]
- Iqbal, T.; Ahmed, N.; Shahjeer, K.; Ahmed, S.; Al-Mutairi, K.A.; Khater, H.F.; Ali, R.F. Botanical Insecticides and their Potential as Anti-Insect/Pests: Are they Successful against Insects and Pests? In Global Decline of Insects; IntechOpen: London, UK, 2021. [Google Scholar]
- Yang, C.S.; Landau, J.M. Effects of tea consumption on nutrition and health. J. Nutr. 2000, 130, 2409–2412. [Google Scholar] [CrossRef]
- Chen, L.; Mo, H.; Zhao, L.; Gao, W.; Wang, S.; Cromie, M.M.; Lu, C.; Wang, J.-S.; Shen, C.-L. Therapeutic properties of green tea against environmental insults. J. Nutr. Biochem. 2017, 40, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Muema, J.M.; Bargul, J.L.; Nyanjom, S.G.; Mutunga, J.M.; Njeru, S.N. Potential of Camellia sinensis proanthocyanidins-rich fraction for controlling malaria mosquito populations through disruption of larval development. Parasites Vectors 2016, 9, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barbehenn, R.V.; Constabel, C.P. Tannins in plant–herbivore interactions. Phytochemistry 2011, 72, 1551–1565. [Google Scholar] [CrossRef] [PubMed]
- Rocha, D.K.; Matos, O.; Novo, M.T.; Figueiredo, A.C.; Delgado, M.; Moiteiro, C. Larvicidal activity against Aedes aegypti of Foeniculum vulgare essential oils from Portugal and Cape Verde. Nat. Prod. Commun. 2015, 10, 1934578X1501000438. [Google Scholar] [CrossRef] [Green Version]
- Modise, S.A.; Ashafa, A.O.T. Larvicidal, pupicidal and insecticidal activities of Cosmos bipinnatus, Foeniculum vulgare and Tagetes minuta against Culex quinquefasciatus mosquitoes. Trop. J. Pharm. Res. 2016, 15, 965–972. [Google Scholar] [CrossRef] [Green Version]
- WHO. Larval Source Management: A Supplementary Malaria Vector Control Measure: An Operational Manual; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- WHO. Instructions for Determining the Susceptibility or Resistance of Mosquito Larvae to Insecticides; World Health Organization: Geneva, Switzerland, 1981. [Google Scholar]
- WHO. Guidelines for Laboratory and Field Testing of Mosquito Larvicides; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Vatandoost, H.; Abai, M.R.; Akbari, M.; Raeisi, A.; Yousefi, H.; Sheikhi, S.; Bagheri, A. Comparison of CDC bottle bioassay with WHO standard method for assessment susceptibility level of malaria vector, Anopheles stephensi to three imagicides. J. Arthropod-Borne Dis. 2019, 13, 17. [Google Scholar] [CrossRef]
- WHO. Test Procedures for Insecticide Resistance Monitoring in Malaria Vector Mosquitoes; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Mulla, M.S.; Norland, L.R.; Fanara, D.M.; Darwazeh, H.A.; McKean, D.W. Control of chironomid midges in recreational lakes. J. Econ. Entomol. 1971, 64, 300–307. [Google Scholar] [CrossRef]
- Baz, M. Strategies for Mosquito Control. Ph.D. Thesis, Benha University, Faculty of Science, Benha, Egypt, 2013. [Google Scholar]
- Aydın, C.; Mammadov, R. Phenolic composition, antioxidant, antibacterial, larvacidal against Culex pipiens, and cytotoxic activities of Hyacinthella lineata steudel extracts. Int. J. Food Prop. 2017, 20, 2276–2285. [Google Scholar] [CrossRef] [Green Version]
- Poonsri, W.; Pengsook, A.; Pluempanupat, W.; Yooboon, T.; Bullangpoti, V. Evaluation of Alpinia galanga (Zingiberaceae) extracts and isolated trans-cinnamic acid on some mosquitoes larvae. Chem. Biol. Technol. Agric. 2019, 6, 1–7. [Google Scholar] [CrossRef]
- Baz, M.M.; Selim, A.; Radwan, I.T.; Alkhaibari, A.M.; Khater, H.F. Larvicidal and adulticidal effects of some Egyptian oils against Culex pipiens. Sci. Rep. 2022, 12, 1–18. [Google Scholar] [CrossRef]
- Roni, M.; Murugan, K.; Panneerselvam, C.; Subramaniam, J.; Nicoletti, M.; Madhiyazhagan, P.; Dinesh, D.; Suresh, U.; Khater, H.F.; Wei, H. Characterization and biotoxicity of Hypnea musciformis-synthesized silver nanoparticles as potential eco-friendly control tool against Aedes aegypti and Plutella xylostella. Ecotoxicol. Environ. Saf. 2015, 121, 31–38. [Google Scholar] [CrossRef]
- Abd El Meguid, A.D.; Mahmoud, S.H.; Baz, M.M. Toxicological activity of four plant oils against Aedes caspius and Culex pipiens (Diptera: Culicidae). Int. J. Mosq. Res. 2019, 6, 86–94. [Google Scholar]
- Baz, M.M.; Hegazy, M.M.; Khater, H.F.; El-Sayed, Y.A. Comparative Evaluation of Five Oil-Resin Plant Extracts against The Mosquito Larvae, Culex pipiens Say (Diptera: Culicidae). Pak. Vet. J. 2021, 41, 191–196. [Google Scholar] [CrossRef]
- Pavela, R.; Žabka, M.; Bednář, J.; Tříska, J.; Vrchotová, N. New knowledge for yield, composition and insecticidal activity of essential oils obtained from the aerial parts or seeds of fennel (Foeniculum vulgare Mill.). Ind. Crops Prod. 2016, 83, 275–282. [Google Scholar] [CrossRef]
- Sousa, R.M.O.; Rosa, J.S.; Silva, C.A.; Almeida, M.T.M.; Novo, M.T.; Cunha, A.C.; Fernandes-Ferreira, M. Larvicidal, molluscicidal and nematicidal activities of essential oils and compounds from Foeniculum vulgare. J. Pest Sci. 2015, 88, 413–426. [Google Scholar] [CrossRef]
- Zoubiri, S.; Baaliouamer, A.; Seba, N.; Chamouni, N. Chemical composition and larvicidal activity of Algerian Foeniculum vulgare seed essential oil. Arab. J. Chem. 2014, 7, 480–485. [Google Scholar] [CrossRef] [Green Version]
- Hassan, M.I.; Atwa, W.A.; Moselhy, W.A.; Mahmoud, D.A. Efficacy of the green tea, Camellia sinensis leaves extract on some biological activities of Culex pipiens and the detection of its phytochemical constituents. Egypt. Acad. J. Biol. Sci. F Toxicol. Pest Control 2020, 12, 59–70. [Google Scholar] [CrossRef]
- Žabar, A.; Cvetković, V.; Rajković, J.; Jović, J.; Vasiljević, P.; Mitrović, T. Larvicidal activity and in vitro effects of green tea (Camellia sinensis L.) water infusion. Biol. Nyssana 2013, 4, 75–79. [Google Scholar]
- Khater, H.F. Biocontrol of Some Insects. Ph.D. Thesis, Zagazig University, Benha Branch, Benha, Egypt, 2003. [Google Scholar]
- Khater, H.F.; Shalaby, A.A.-S. Potential of biologically active plant oils to control mosquito larvae (Culex pipiens, Diptera: Culicidae) from an Egyptian locality. Rev. Inst. Med. Trop. Sao Paulo 2008, 50, 107–112. [Google Scholar] [CrossRef]
- Chaiyasit, D.; Choochote, W.; Rattanachanpichai, E.; Chaithong, U.; Chaiwong, P.; Jitpakdi, A.; Tippawangkosol, P.; Riyong, D.; Pitasawat, B. Essential oils as potential adulticides against two populations of Aedes aegypti, the laboratory and natural field strains, in Chiang Mai province, northern Thailand. Parasitol. Res. 2006, 99, 715–721. [Google Scholar] [CrossRef]
- Kamsuk, K.; Choochote, W.; Chaithong, U.; Jitpakdi, A.; Tippawangkosol, P.; Riyong, D.; Pitasawat, B. Effectiveness of Zanthoxylum piperitum-derived essential oil as an alternative repellent under laboratory and field applications. Parasitol. Res. 2007, 100, 339–345. [Google Scholar] [CrossRef]
- Benelli, G.; Caselli, A.; Canale, A. Nanoparticles for mosquito control: Challenges and constraints. J. King Saud Univ. Sci. 2017, 29, 424–435. [Google Scholar] [CrossRef]
- Murugan, K.; Priyanka, V.; Dinesh, D.; Madhiyazhagan, P.; Panneerselvam, C.; Subramaniam, J.; Suresh, U.; Chandramohan, B.; Roni, M.; Nicoletti, M. Predation by Asian bullfrog tadpoles, Hoplobatrachus tigerinus, against the dengue vector, Aedes aegypti, in an aquatic environment treated with mosquitocidal nanoparticles. Parasitol. Res. 2015, 114, 3601–3610. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, M.; Khater, H.F.; Panneerselvam, C.; Benelli, G. One-pot fabrication of silver nanocrystals using Nicandra physalodes: A novel route for mosquito vector control with moderate toxicity on non-target water bugs. Res. Vet. Sci. 2016, 107, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, M.; Rajeswary, M.; Muthukumaran, U.; Hoti, S.; Khater, H.F.; Benelli, G. Single-step biosynthesis and characterization of silver nanoparticles using Zornia diphylla leaves: A potent eco-friendly tool against malaria and arbovirus vectors. J. Photochem. Photobiol. B Biol. 2016, 161, 482–489. [Google Scholar] [CrossRef]
- Sultana, N.; Raul, P.K.; Goswami, D.; Das, D.; Islam, S.; Tyagi, V.; Das, B.; Gogoi, H.K.; Chattopadhyay, P.; Raju, P.S. Bio-nanoparticle assembly: A potent on-site biolarvicidal agent against mosquito vectors. RSC Adv. 2020, 10, 9356–9368. [Google Scholar] [CrossRef]
- Jabbar, A.; Tariq, M.; Gulzar, A.; Mukhtar, T.; Zainab, T. Lethal and Sub Lethal Effects of Plant Extracts and Green Silver Nanoparticles against Culex pipiens. Pak. J. Zool. 2021, 54, 1259–1267. [Google Scholar] [CrossRef]
- Govindarajan, M.; AlQahtani, F.S.; AlShebly, M.M.; Benelli, G. One-pot and eco-friendly synthesis of silver nanocrystals using Adiantum raddianum: Toxicity against mosquito vectors of medical and veterinary importance. J. Appl. Biomed. 2017, 15, 87–95. [Google Scholar] [CrossRef]
- Rajasekharreddy, P.; Rani, P.U. Biofabrication of Ag nanoparticles using Sterculia foetida L. seed extract and their toxic potential against mosquito vectors and HeLa cancer cells. Mater. Sci. Eng. C 2014, 39, 203–212. [Google Scholar] [CrossRef]
- Hikal, W.M.; Baeshen, R.S.; Said-Al Ahl, H.A. Botanical insecticide as simple extractives for pest control. Cogent Biol. 2017, 3, 1404274. [Google Scholar] [CrossRef]
- Khater, H.; Geden, C. Potential of essential oils to prevent fly strike by Lucilia sericata, and effects of oils on longevity of adult flies. J. Vector Ecol. 2018, 43, 261–270. [Google Scholar] [CrossRef] [Green Version]
- Patil, C.D.; Borase, H.P.; Patil, S.V.; Salunkhe, R.B.; Salunke, B.K. Larvicidal activity of silver nanoparticles synthesized using Pergularia daemia plant latex against Aedes aegypti and Anopheles stephensi and nontarget fish Poecillia reticulata. Parasitol. Res. 2012, 111, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Sutthanont, N.; Choochote, W.; Tuetun, B.; Junkum, A.; Jitpakdi, A.; Chaithong, U.; Riyong, D.; Pitasawat, B. Chemical composition and larvicidal activity of edible plant-derived essential oils against the pyrethroid-susceptible and-resistant strains of Aedes aegypti (Diptera: Culicidae). J. Vector Ecol. 2010, 35, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.L.; Cho, K., II; Li, Q.X. Insecticidal activity of basil oil, trans-anethole, estragole, and linalool to adult fruit flies of Ceratitis capitata, Bactrocera dorsalis, and Bactrocera cucurbitae. J. Econ. Entomol. 2009, 102, 203–209. [Google Scholar] [CrossRef] [PubMed]



| Compounds | Retention Time | Std Conc. (μg/mL) | Samples (Conc. μg/g) | Structure |
|---|---|---|---|---|
| Chlorogenic acid | 7.30 | 0.05 | 0.23 | 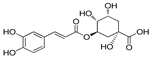 |
| Daidzein | 12.81 | 0.05 | 0.67 | 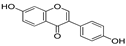 |
| Gallic acid | Not Detected | 0.05 | Not Detected | - |
| Caffeic acid | Not Detected | 0.05 | Not Detected | - |
| Rutin | Not Detected | 0.05 | Not Detected | - |
| Coumaric acid | Not Detected | 0.05 | Not Detected | - |
| Vanillin | 9.47 | 0.05 | 0.76 |  |
| Naringenin | 14.89 | 0.05 | 0.52 | 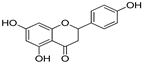 |
| Querectin | Not Detected | 0.05 | Not Detected | - |
| Ellagic acid | 9.86 | 0.05 | 0.59 | 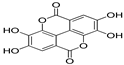 |
| 3,4-Dihydroxybenzoic acid | Not Detected | 0.05 | Not Detected | - |
| Hesperetin | Not Detected | 0.05 | Not Detected | - |
| Myricetin | 11.62 | 0.05 | 0.44 |  |
| Cinnamic acid | Not Detected | 0.05 | Not Detected | - |
| Methyl gallate | Not Detected | 0.05 | Not Detected | - |
| Kaempferol | Not Detected | 0.05 | Not Detected | - |
| Ferulic acid | Not Detected | 0.05 | Not Detected | - |
| Syringic acid | 8.35 | 0.05 | 0.42 | 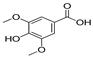 |
| Apigenin | Not Detected | 0.05 | Not Detected | - |
| Catechin | 7.30 | 0.05 | 0.73 | 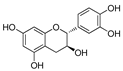 |
| Luteolin | Not Detected | 0.05 | Not Detected | - |
| Oil Name | Nanocomposite | Time (Hour) | Mortality % (Mean ± SE) | ||||
|---|---|---|---|---|---|---|---|
| 125 * | 250 * | 500 * | 1000 * | 2000 * | |||
| Fennel (Foeniculum vulgare) | Oil | 0.5 | 0.00 ± 0.00 c | 0.00 ± 0.00 c | 3.33 ± 0.66 d | 8.33 ± 1.66 e | 13.33 ± 0.33 e |
| 2 | 1.67 ± 0.33 c | 3.33 ± 0.33d c | 6.67 ± 0.33 d | 21.67 ± 0.33 d | 30.0 ± 0.57 d | ||
| 8 | 3.33 ± 0.33 c | 8.33 ± 0.33 c | 18.33 ± 0.33 c | 45.0 ± 1.15 c | 61.66 ± 1.20 c | ||
| 24 | 8.33 ± 0.33 b | 20.0 ± 0.57 b | 60.0 ± 0.57 b | 60.0 ± 0.57 b | 90.0 ± 0.00 a | ||
| 48 | 13.33 ± 0.33 a | 33.33 ± 0.33 a | 61.33 ± 0.66 a | 86.67 ± 0.66 a | 100.0 ± 0.00 a | ||
| Mg-LDH-F | 0.5 | 3.33 ± 0.33 c | 10.0 ± 0.00 c | 13.33 ± 0.33 d | 25.0 ± 0.57 d | 31.67 ± 0.33 d | |
| 2 | 6.67 ± 0.33 bc | 13.33 ± 0.33 c | 20.0 ± 0.58 d | 40.0 ± 0.57 c | 50.0 ± 0.57 c | ||
| 8 | 10.0 ± 0.57 b | 26.67 ± 0.88 b | 51.67 ± 0.67 c | 70.0 ± 0.57 b | 83.33 ± 0.33 b | ||
| 24 | 16.67 ± 0.33 a | 35.0 ± 0.57 ab | 65.0 ± 0.57 b | 90.0 ± 0.57 a | 100.0 ± 0.0 a | ||
| 48 | 18.33 ± 0.33 a | 40.0 ± 0.57 a | 75.0 ± 0.57 a | 96.67 ± 0.33 a | 100.0 ± 0.00 a | ||
| Ni-LDH-F | 0.5 | 1.67 ± 0.33 b | 5.0 ± 0.00 c | 11.67 ± 0.33 c | 16.67 ± 0.33 d | 20.0 ± 0.57 d | |
| 2 | 5.0 ± 0.00 b | 8.33 ± 0.33 c | 15.0 ± 0.58 c | 33.33 ± 0.88 c | 40.0 ± 1.0 c | ||
| 8 | 8.33 ± 0.67 ab | 21.67 ± 0.33 b | 46.67 ± 0.88 b | 65.0 ± 0.57 b | 75.0 ± 0.57 a | ||
| 24 | 13.33 ± 0.33 a | 30.0 ± 0.57 a | 60.0 ± 0.57 a | 83.33 ± 0.33 a | 98.33 ± 0.33 a | ||
| 48 | 15.0 ± 0.57 a | 35.0 ± 0.57 a | 66.67 ± 0.88 a | 90.0 ± 0.57 a | 100.0 ± 0.00 a | ||
| Mg-Free | 0.5 | 0.00 ± 0.00 c | 0.00 ± 0.00 c | 1.67 ± 0.33 d | 3.33 ± 0.33 e | 5.0 ± 0.00 f | |
| 2 | 0.00 ± 0.00 c | 1.67 ± 0.33 c | 1.67 ± 0.33 d | 3.33 ± 0.33 e | 8.33 ± 0.33 f | ||
| 8 | 0.00 ± 0.00 c | 1.67 ± 0.33 c | 3.33 ± 0.33 d | 5.0 ± 0.00 e | 8.33 ± 0.33 f | ||
| 24 | 0.00 ± 0.00 c | 1.67 ± 0.33 c | 3.33 ± 0.33 d | 5.0 ± 0.00 e | 8.33 ± 0.33 f | ||
| 48 | 0.00 ± 0.00 c | 1.67 ± 0.33 c | 3.33 ± 0.33 d | 5.0 ± 0.00 e | 10.0 ± 0.00 e | ||
| Oil Name | Nanocomposite | Time (Hour) | Mortality % (Mean ± SE) | ||||
|---|---|---|---|---|---|---|---|
| 125 * | 250 * | 500 * | 1000 * | 2000 * | |||
| Green tea (Camellia sinensis) | Oil | 0.5 | 0.00 ± 0.00 c | 1.67 ± 0.33 c | 3.33 ± 0.33 d | 6.67 ± 0.33 e | 11.66 ± 0.33 e |
| 2 | 1.67 ± 0.33 bc | 3.33 ± 0.33 c | 5.0 ± 0.00 d | 18.33 ± 0.33 d | 28.33 ± 0.66 d | ||
| 8 | 3.33 ± 0.33 bc | 8.33 ± 0.33 c | 15.0 ± 0.00 c | 41.67 ± 0.88 c | 53.33 ± 0.88 c | ||
| 24 | 5.0 ± 0.00 b | 16.67 ± 0.33 b | 28.33 ± 0.33 b | 56.67 ± 0.88 b | 85.0 ± 0.57 b | ||
| 48 | 13.33 ± 0.33 a | 31.67 ± 0.66 a | 53.33 ± 0.33 a | 86.6 ± 0.88 a | 95.00 ± 0.57a | ||
| Mg-LDH-GT | 0.5 | 3.33 ± 0.33 d | 8.33 ± 0.33 c | 11.67 ± 0.33 c | 21.67 ± 0.33 d | 28.33 ± 0.88 d | |
| 2 | 6.67 ± 0.33 bc | 10.0 ± 0.57 c | 16.67 ± 0.33 c | 36.67 ± 0.57 c | 46.67 ± 0.33 c | ||
| 8 | 11.67 ± 0.33 bc | 26.67 ± 0.33 b | 45.0 ± 0.57 b | 68.33 ± 0.88 b | 81.67 ± 0.88 b | ||
| 24 | 18.33 ± 0.67 ab | 36.67 ± 0.33 a | 58.33 ± 0.88 a | 83.33 ± 0.67 a | 98.33 ± 0.33 a | ||
| 48 | 20.0 ± 0.57 a | 41.67 ± 0.33 a | 66.67 ± 0.33 a | 88.33 ± 0.33 a | 100.0 ± 0.00 a | ||
| Ni-LDH-GT | 0.5 | 1.67 ± 0.33 c | 3.33 ± 0.33 d | 8.33 ± 0.33 c | 11.67 ± 0.33 d | 16.67 ± 0.67 d | |
| 2 | 3.33 ± 0.33 bc | 8.33 ± 0.33 cd | 10.0 ± 0.58 c | 25.0 ± 0.58 c | 35.0 ± 0.57 c | ||
| 8 | 6.67 ± 0.33 ab | 13.33 ± 0.33 bc | 28.33 ± 0.33 b | 48.33 ± 0.33 b | 65.0 ± 0.57 b | ||
| 24 | 8.33 ± 0.33 a | 18.33 ± 0.33 b | 40.0 ± 0.57 a | 63.33 ± 0.57 a | 93.33 ± 0.33 a | ||
| 48 | 10.0 ± 0.00 a | 21.67 ± 0.57 a | 43.33 ± 0.33 a | 75.00 ± 0.00 a | 96.67 ± 0.33 a | ||
| Ni-Free | 0.5 | 0.00 ± 0.00 c | 0.00 ± 0.00 c | 1.67 ± 0.33 d | 3.33 ± 0.33 e | 5.0 ± 0.00 f | |
| 2 | 0.00 ± 0.00 c | 00.00 ± 0.00 c | 1.67 ± 0.33 d | 3.33 ± 0.33 e | 5.0 ± 0.00 f | ||
| 8 | 0.00 ± 0.00 c | 1.67 ± 0.33 c | 3.33 ± 0.33 d | 3.33 ± 0.33 e | 8.33 ± 0.33 f | ||
| 24 | 0.00 ± 0.00 c | 1.67 ± 0.33 c | 3.33 ± 0.33 d | 5.0 ± 0.00 e | 8.33 ± 0.33 f | ||
| 48 | 0.00 ± 0.00 c | 1.67 ± 0.33 c | 3.33 ± 0.33 d | 5.0 ± 0.00 e | 8.33 ± 0.33 f | ||
| Oil Name | Nanocomposite | LC50 (Low.–Up.) * | LC90 (Low.–Up.) | LC95 (Low.–Up.) | Chi (Sig) | Equation |
|---|---|---|---|---|---|---|
| Fennel (Foeniculum vulgare) | Oil | 843.88 (502.16–1458.19) | 1736.30 (1230.01–3494.77) | 1989.29 (1402.44–4106.03) | 19.74 (0.001 a) | Y = −1.04 + 1.24 × 10−3X |
| Mg-LDH-F | 451.95 (304.81–663.28) | 880.32 (667.46–1439.86) | 1001.75 (755.14–1675.14) | 12.76 (0.003 a) | Y = −1.2 + 1.70 × 10−3X | |
| Ni-LDH-F | 550.12 (187.53–3382.85) | 1116.31 (687.92–13,917) | 1276.82 (786.49–16,976.02) | 34.96 (0.000 a) | Y = −1.12 + 1.68 × 10−3X | |
| Green tea (Camellia sinensis) | Oil | 938.93 (634.38–1453.09) | 1944.13 (1365.39–3174.64) | 2100.75 (1547.09–3688.21) | 15.15 (0.004 a) | Y = −1.2 + 1.76 × 10−3X |
| Mg-LDH-GT | 530.46 (173.93–1416.95) | 1130.82 (729.36–4632.54) | 1301.02 (836.27–5594.66) | 31.95 (0.000 a) | Y = −1.2 + 1.76 × 10−3X | |
| Ni-LDH-GT | 769.94 (432.83–1362.39) | 1633.88 (1145.95–3407.41) | 1878.79 (1311.81–4023.44) | 20.20 (0.000 a) | Y = −1.2 + 1.76 × 10−3X |
| Oil Name | Nanocomposite | Conc. (%) | Mortality% (Mean ± SE) | LC50 (Low.–Up.) | LC90 (Low.–Up.) | LC95 (Low.–Up.) | Chi (Sig) | Equation |
|---|---|---|---|---|---|---|---|---|
| Fennel (Foeniculum vulgare) | Oil | 0 | 0.00 ± 0.00 c | 3.25 (2.00–4.35) | 34.72 (21.80–80.40) | 72.40 (38.76–230.68) | 26.52 (0.000 a) | Y = 0.64 + 0.6 × 10−3X |
| 2.0 | 10.00 ± 11.55 d | |||||||
| 5.0 | 53.33 ± 3.33 d | |||||||
| 10.0 | 66.67 ± 8.82 c | |||||||
| 15.0 | 76.67 ± 3.33 b | |||||||
| 20.0 | 86.67 ± 3.33 a | |||||||
| Mg-LDH-F | 0.0 | 0.00 ± 0.00 c | 1.75 (0.55–2.15) | 8.52 (5.80–20.4) | 14.16 (8.40–28.92) | 18.904 (0.001 a) | Y = −1.04 + 1.24 × 10−3X | |
| 0.5 | 66.67 ± 6.67 d | |||||||
| 1.0 | 70.00 ± 5.77 c | |||||||
| 2.0 | 83.33 ± 8.82 b | |||||||
| 3.0 | 96.67 ± 3.33 a | |||||||
| 4.0 | 100.00 ± 0.00 a | |||||||
| Ni-LDH-F | 0.0 | 0.00 ± 0.00 c | 2.30 (1.35–3.145) | 17.52 (12.72–29.52) | 33.16 (21.36–70.68) | 26.52 (0.000 a) | Y = 0.64 + 0.6 × 10−3X | |
| 0.5 | 56.67 ± 3.33 e | |||||||
| 1.0 | 63.33 ± 3.33 d | |||||||
| 2.0 | 73.33 ± 6.67 c | |||||||
| 3.0 | 86.67 ± 3.33 b | |||||||
| 4.0 | 93.33 ± 3.33 a |
| Oil Name | Nanocomposite | Conc. (%) | Mortality% (Mean ± SE) | LC50 (Low.–Up.) | LC90 (Low.–Up.) | LC95 (Low.–Up.) | Chi Sig) | Equation |
|---|---|---|---|---|---|---|---|---|
| Green tea (Camellia sinensis) | Oil | 0.0 | 0.00 ± 0.00 e | 5.45 (3.95–6.95) | 25.90 (18.83–47.45) | 35.39 (22.04–202.28) | 26.52 (0.000 a) | Y = 1.10 + 0.17 × 10−3X |
| 2.0 | 40.00 ± 5.77 d | |||||||
| 5.0 | 43.33 ± 3.33 d | |||||||
| 10.0 | 56.67 ± 3.33 c | |||||||
| 15.0 | 70.00 ± 5.77 b | |||||||
| 20.0 | 76.67 ± 3.33 a | |||||||
| Mg-LDH-GT | 0.0 | 0.00 ± 0.00 f | 0.42 (3.27–6.83) | 4.51 (2.30–7.99) | 8.84 (5.51–20.57) | 18.904 (0.001 a) | Y = −0.25 + 1.2 × 10−3X | |
| 0.5 | 56.67 ± 3.33 e | |||||||
| 1.0 | 66.67 ± 6.67 d | |||||||
| 2.0 | 76.67 ± 3.33 c | |||||||
| 3.0 | 83.33 ± 3.33 b | |||||||
| 4.0 | 93.33 ± 3.33 a | |||||||
| Ni-LDH-GT | 0.0 | 0.00 ± 0.00 f | 0.45 (031–0.79) | 12.32 (6.76–41.91) | 29.14 (12.90–159.03) | 16.35 (0.001 a) | Y = −0.97 + 0.17 × 10−3X | |
| 0.5 | 50.00 ± 0.00 e | |||||||
| 1.0 | 56.67 ± 3.33 d | |||||||
| 2.0 | 66.67 ± 3.33 c | |||||||
| 3.0 | 73.33 ± 3.33 b | |||||||
| 4.0 | 83.33 ± 3.33 a |
| Oil Name | Oil Formulated | Sites | Avg. Adult/Site | Mean Number ± SE | % Reduction | % Mean Reduction |
|---|---|---|---|---|---|---|
| Control | home 1 | 13.7 | 22.36 ± 5.92 a | 0 | 0 | |
| home 2 | 19.7 | 0 | ||||
| home 3 | 33.7 | 0 | ||||
|
Fennel (Foeniculum vulgare) | Oil | home 1 | 2.67 | 6.66 ± 1.38 b | 84.6 | 83.1 |
| home 2 | 5.00 | 81.2 | ||||
| home 3 | 7.33 | 83.6 | ||||
| Mg-LDH | home 1 | 0.00 | 0.22 ± 0.22 b | 100.0 | 100.0 | |
| home 2 | 0.00 | 100.0 | ||||
| home 3 | 0.00 | 100.0 | ||||
|
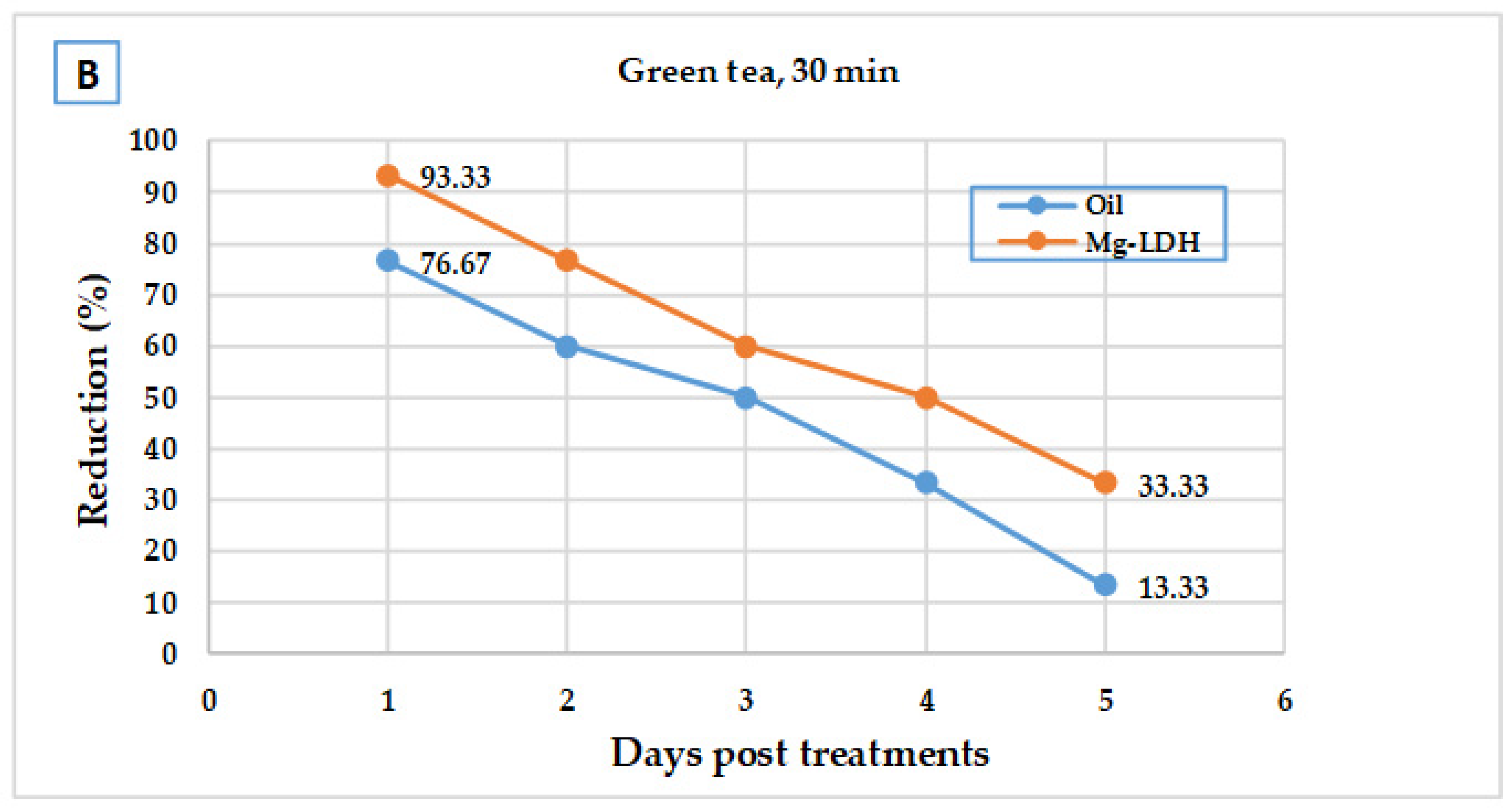
Green tea
(Camellia sinensis) | Oil | home 1 | 4.00 | 5.0 ± 1.34 b | 77.3 | 77.0 |
| home 2 | 7.33 | 72.8 | ||||
| home 3 | 8.67 | 80.8 | ||||
| Mg-LDH | home 1 | 0.00 | 0.00 ± 0.00 b | 98.3 | 99.0 | |
| home 2 | 0.00 | 100.0 | ||||
| home 3 | 0.67 | 98.6 |
| Oil Name | Treatment | Mosquito Predator Types | Control *** | Mean of Predation | ||
|---|---|---|---|---|---|---|
| G. affinis | C. tripunctatus | S. urinator | ||||
| Foeniculum vulgare | Free- Mg-LDH * | 80.00 ± 1.15 aB | 64.33 ± 4.18 bC | 25.00 ± 0.58 bC | 99.67 ± 0.33 aA | 67.25 ± 8.32 a |
| Oil | 74.67 ± 2.03 bB | 52.33 ± 2.03 cC | 22.33 ± 1.76 cC | 99.33 ± 0.67 aA | 62.17 ± 8.59 b | |
| Mg-LDH-F | 75.00 ± 1.15 bB | 69.00 ± 3.21 aC | 27.33 ± 2.19 aC | 99.00 ± 0.58 aA | 67.58 ± 7.83 a | |
| Camellia sinensis | Free Mg-LDH ** | 82.33 ± 1.15 bB | 65.00 ± 0.00 bC | 25.00 ± 0.58 bC | 97.67 ± 0.33 aA | 67.50 ± 6.32 a |
| Oil | 75.67 ± 1.76 cB | 55.67 ± 1.20 cC | 23.33 ± 2.03 bC | 99.33 ± 0.67 aA | 63.50 ± 8.43 b | |
| Mg-LDH-GT | 77.00 ± 3.21 cB | 71.00 ± 1.73 aC | 28.00 ± 1.53 aC | 99.00 ± 0.58 aA | 68.75 ± 7.81 a | |
| Mean of sample | 77.05 ± 0.94 B | 62.77 ± 1.72 C | 25.15 ± 0.65 C | 99.33 ± 0.19 A | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radwan, I.T.; Baz, M.M.; Khater, H.; Alkhaibari, A.M.; Selim, A.M. Mg-LDH Nanoclays Intercalated Fennel and Green Tea Active Ingredient: Field and Laboratory Evaluation of Insecticidal Activities against Culex pipiens and Their Non-Target Organisms. Molecules 2022, 27, 2424. https://doi.org/10.3390/molecules27082424
Radwan IT, Baz MM, Khater H, Alkhaibari AM, Selim AM. Mg-LDH Nanoclays Intercalated Fennel and Green Tea Active Ingredient: Field and Laboratory Evaluation of Insecticidal Activities against Culex pipiens and Their Non-Target Organisms. Molecules. 2022; 27(8):2424. https://doi.org/10.3390/molecules27082424
Chicago/Turabian StyleRadwan, Ibrahim Taha, Mohamed M. Baz, Hanem Khater, Abeer Mousa Alkhaibari, and Abdelfattah M. Selim. 2022. "Mg-LDH Nanoclays Intercalated Fennel and Green Tea Active Ingredient: Field and Laboratory Evaluation of Insecticidal Activities against Culex pipiens and Their Non-Target Organisms" Molecules 27, no. 8: 2424. https://doi.org/10.3390/molecules27082424
APA StyleRadwan, I. T., Baz, M. M., Khater, H., Alkhaibari, A. M., & Selim, A. M. (2022). Mg-LDH Nanoclays Intercalated Fennel and Green Tea Active Ingredient: Field and Laboratory Evaluation of Insecticidal Activities against Culex pipiens and Their Non-Target Organisms. Molecules, 27(8), 2424. https://doi.org/10.3390/molecules27082424







