Aloe arborescens: In Vitro Screening of Genotoxicity, Effective Inhibition of Enzyme Characteristics for Disease Etiology, and Microbiological Activity
Abstract
1. Introduction
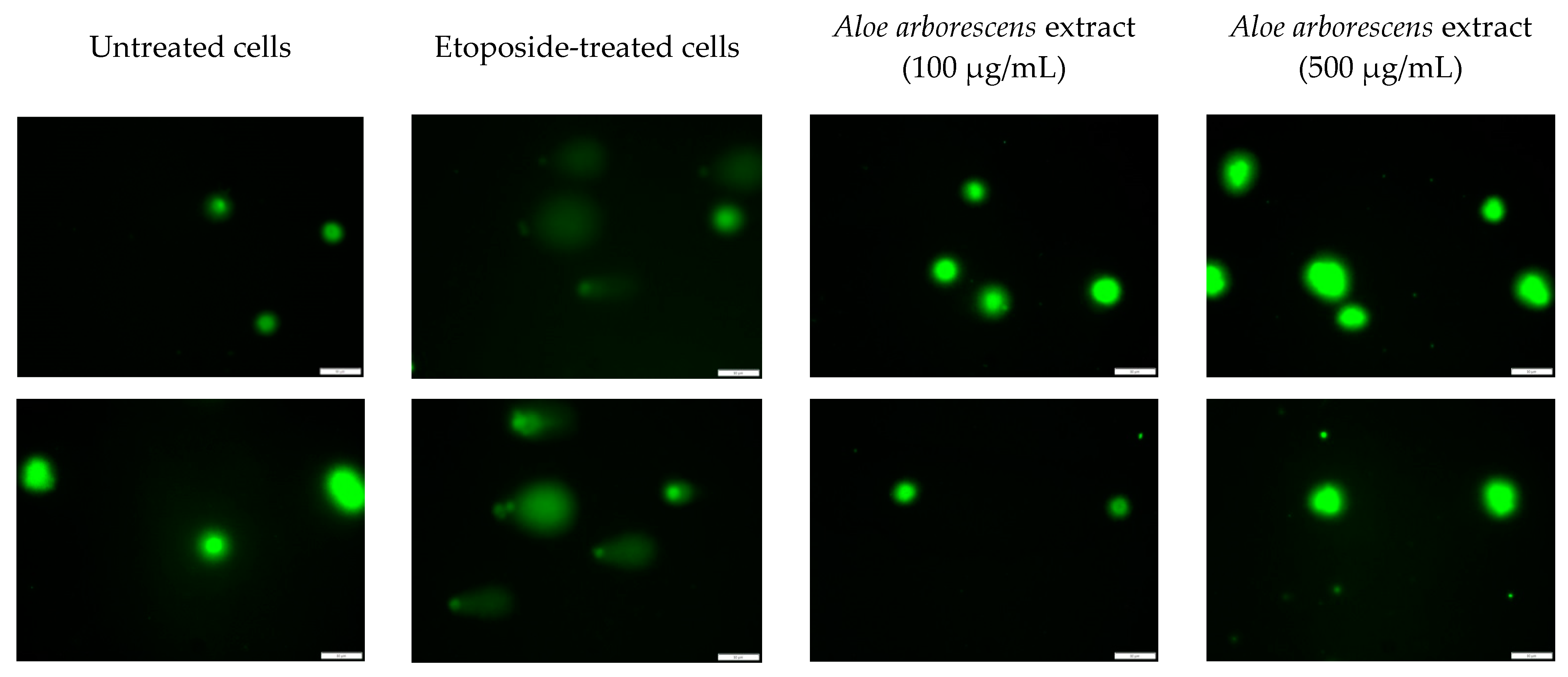
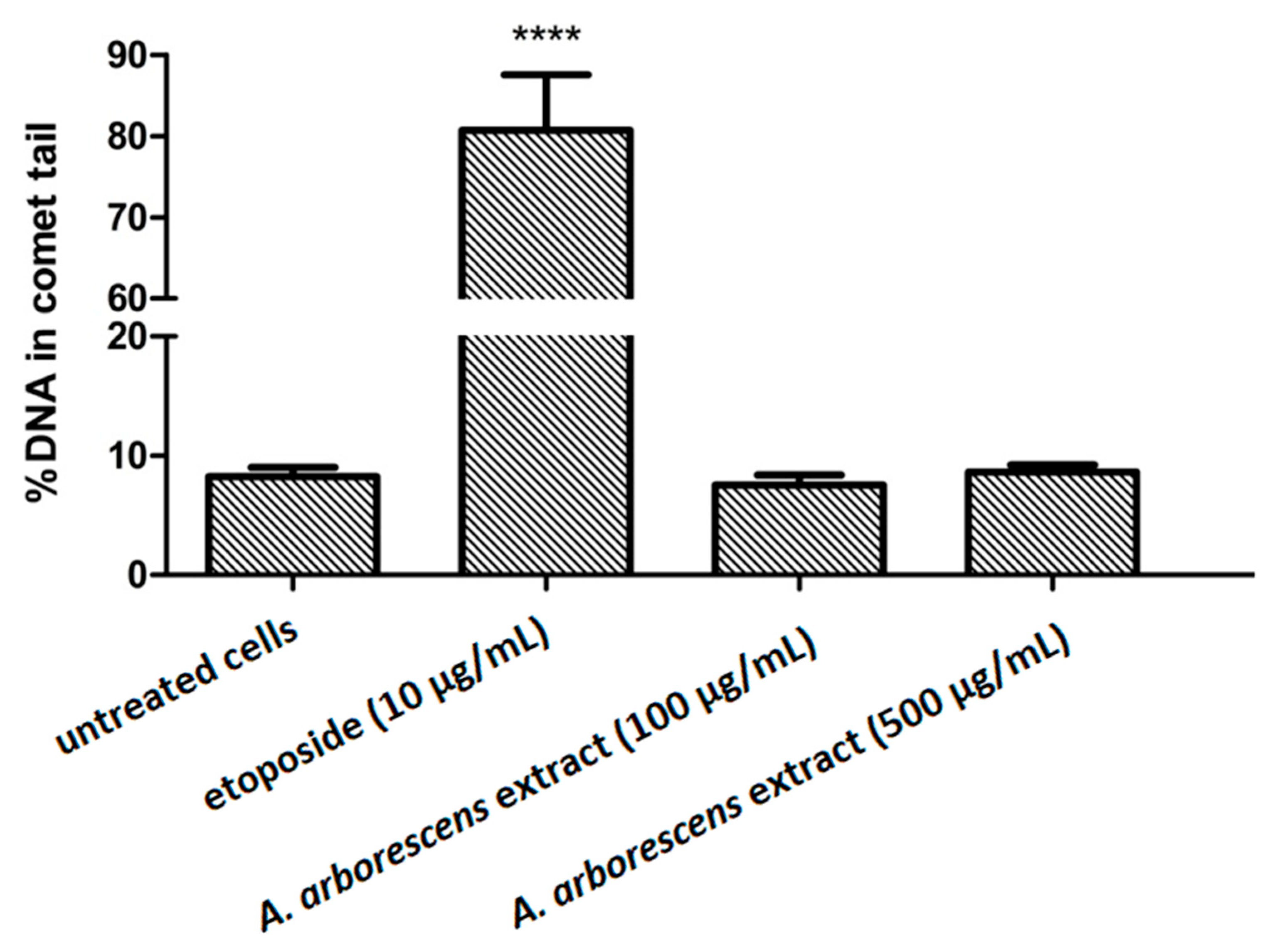
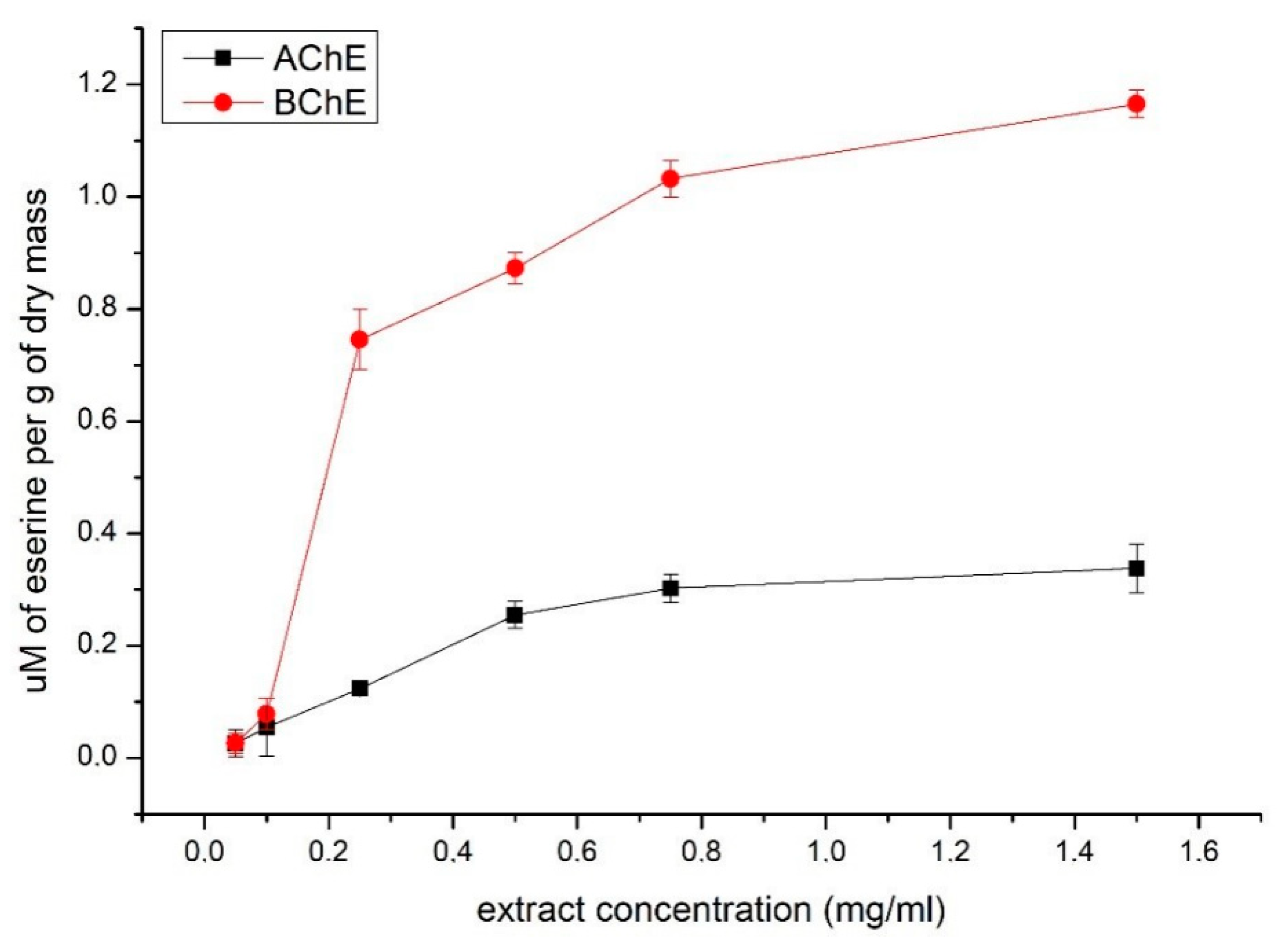
2. Results
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Lyophilisation of Aloe Gel
4.3. Extracts Preparation
4.4. HPLC-DAD Method
4.5. The Sum of Polyphenolic Compounds
4.6. The Sum of Phenolic Acids
4.7. Comet Assay
4.8. Enzyme Inhibition Assays
4.8.1. Hyaluronidase Inhibition Assay
4.8.2. α-Glucosidase Inhibition Assay
4.8.3. Acetylcholinesterase and Butyrylcholinesterase Inhibition Assay
4.9. Scratch Assay
4.10. Antimicrobial Studies
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Lundstrom, K.; Pham, H.T.; Dinh, L.D. Interaction of Plant Extracts with Central Nervous System Receptors. Medicines 2017, 4, 12. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Zhao, S.; Wu, Y.; Cao, H.; Xu, Y.; Liu, X.; Shui, W.; Cheng, J.; Zhao, S.; Shen, L.; et al. Identification of Natural Products as Novel Ligands for the Human 5-HT2C Receptor. Biophys. Rep. 2018, 4, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Lochner, M.; Thompson, A.J. The Muscarinic Antagonists Scopolamine and Atropine Are Competitive Antagonists at 5-HT3 Receptors. Neuropharmacology 2016, 108, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Oliver, M.; Díaz-Ríos, M. Using Caffeine and Other Adenosine Receptor Antagonists and Agonists as Therapeutic Tools against Neurodegenerative Diseases: A Review. Life Sci. 2014, 101, 1–9. [Google Scholar] [CrossRef]
- Umar, M.I.; Asmawi, M.Z.; Sadikun, A.; Abdul Majid, A.M.S.; Atangwho, I.J.; Khadeer Ahamed, M.B.; Altaf, R.; Ahmad, A. Multi-Constituent Synergism Is Responsible for Anti-Inflammatory Effect of Azadirachta Indica Leaf Extract. Pharm. Biol. 2014, 52, 1411–1422. [Google Scholar] [CrossRef]
- Maione, F.; Russo, R.; Khan, H.; Mascolo, N. Medicinal Plants with Anti-Inflammatory Activities. Nat. Prod. Res. 2016, 30, 1343–1352. [Google Scholar] [CrossRef]
- Zhang, L.; Virgous, C.; Si, H. Synergistic Anti-Inflammatory Effects and Mechanisms of Combined Phytochemicals. J. Nutr. Biochem. 2019, 69, 19–30. [Google Scholar] [CrossRef]
- Loftsson, T. Introduction. In Essential Pharmacokinetics; Elsevier: Amsterdam, The Netherlands, 2015; pp. 1–8. ISBN 9780128014110. [Google Scholar]
- Grace, O.M. Current Perspectives on the Economic Botany of the Genus Aloe L. (Xanthorrhoeaceae). S. Afr. J. Bot. 2011, 77, 980–987. [Google Scholar] [CrossRef]
- Eshun, K.; He, Q. Aloe Vera: A Valuable Ingredient for the Food, Pharmaceutical and Cosmetic Industries--a Review. Crit. Rev. Food Sci. Nutr. 2004, 44, 91–96. [Google Scholar] [CrossRef]
- Boudreau, M.D.; Beland, F.A. An Evaluation of the Biological and Toxicological Properties of Aloe Barbadensis (Miller), Aloe Vera. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2006, 24, 103–154. [Google Scholar] [CrossRef]
- Muñiz-Ramirez, A.; Perez, R.M.; Garcia, E.; Garcia, F.E. Antidiabetic Activity of Aloe Vera Leaves. Evid. Based Complement. Altern. Med. 2020, 2020, e6371201. [Google Scholar] [CrossRef] [PubMed]
- Kahramanoğlu, İ.; Chen, C.; Chen, J.; Wan, C. Chemical Constituents, Antimicrobial Activity, and Food Preservative Characteristics of Aloe Vera Gel. Agronomy 2019, 9, 831. [Google Scholar] [CrossRef]
- Shakib, Z.; Shahraki, N.; Razavi, B.M.; Hosseinzadeh, H. Aloe Vera as an Herbal Medicine in the Treatment of Metabolic Syndrome: A Review. Phytother. Res. 2019, 33, 2649–2660. [Google Scholar] [CrossRef] [PubMed]
- Fehrmann-Cartes, K.; Coronado, M.; Hernández, A.J.; Allende, M.L.; Feijoo, C.G. Anti-Inflammatory Effects of Aloe Vera on Soy Meal-Induced Intestinal Inflammation in Zebrafish. Fish Shellfish. Immunol. 2019, 95, 564–573. [Google Scholar] [CrossRef]
- Mangaiyarkarasi, S.P.; Manigandan, T.; Elumalai, M.; Cholan, P.K.; Kaur, R.P. Benefits of Aloe Vera in Dentistry. J. Pharm. Bioallied. Sci. 2015, 7, S255–S259. [Google Scholar] [CrossRef]
- Nazeam, J.A.; Gad, H.A.; Esmat, A.; El-Hefnawy, H.M.; Singab, A.-N.B. Aloe Arborescens Polysaccharides: In Vitro Immunomodulation and Potential Cytotoxic Activity. J. Med. Food 2017, 20, 491–501. [Google Scholar] [CrossRef]
- Glatthaar-Saalmüller, B.; Fal, A.M.; Schönknecht, K.; Conrad, F.; Sievers, H.; Saalmüller, A. Antiviral Activity of an Aqueous Extract Derived from Aloe Arborescens Mill. against a Broad Panel of Viruses Causing Infections of the Upper Respiratory Tract. Phytomedicine 2015, 22, 911–920. [Google Scholar] [CrossRef]
- Luccia, B.D.; Manzo, N.; Vivo, M.; Galano, E.; Amoresano, A.; Crescenzi, E.; Pollice, A.; Tudisco, R.; Infascelli, F.; Calabrò, V. A Biochemical and Cellular Approach to Explore the Antiproliferative and Prodifferentiative Activity of Aloe Arborescens Leaf Extract. Phytother. Res. 2013, 27, 1819–1828. [Google Scholar] [CrossRef]
- Singab, A.-N.B.; El-Hefnawy, H.M.; Esmat, A.; Gad, H.A.; Nazeam, J.A. A Systemic Review on Aloe Arborescens Pharmacological Profile: Biological Activities and Pilot Clinical Trials. Phytother. Res. 2015, 29, 1858–1867. [Google Scholar] [CrossRef]
- Leitgeb, M.; Kupnik, K.; Knez, Ž.; Primožič, M. Enzymatic and Antimicrobial Activity of Biologically Active Samples from Aloe Arborescens and Aloe Barbadensis. Biology 2021, 10, 765. [Google Scholar] [CrossRef]
- Kupnik, K.; Primožič, M.; Knez, Ž.; Leitgeb, M. Antimicrobial Efficiency of Aloe Arborescens and Aloe Barbadensis Natural and Commercial Products. Plants 2021, 10, 92. [Google Scholar] [CrossRef] [PubMed]
- Pawłowicz, K.; Ludowicz, D.; Karaźniewicz-Łada, M.; Wdowiak, K.; Cielecka-Piontek, J. Analysis of the Composition of Lyophilisates Obtained from Aloe Arborescens Gel of Leaves of Different Ages from Controlled Crops. Molecules 2021, 26, 3204. [Google Scholar] [CrossRef]
- Abatangelo, G.; Vindigni, V.; Avruscio, G.; Pandis, L.; Brun, P. Hyaluronic Acid: Redefining Its Role. Cells 2020, 9, 1743. [Google Scholar] [CrossRef] [PubMed]
- Petrey, A.C.; de la Motte, C.A. Hyaluronan, a Crucial Regulator of Inflammation. Front. Immunol. 2014, 5, 101. [Google Scholar] [CrossRef] [PubMed]
- Oryan, A.; Mohammadalipour, A.; Moshiri, A.; Tabandeh, M.R. Topical Application of Aloe Vera Accelerated Wound Healing, Modeling, and Remodeling: An Experimental Study. Ann. Plast. Surg. 2016, 77, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Chandu, A.N.; Kumar, C.S.; Bhattacharjee, C.; Debnath, S.; Kannan, K.K. Studies on Immunomodulatory Activity of Aloe Vera (Linn). Int. J. Appl. Biol. Pharm. Technol. 2011. [Google Scholar]
- Salawu, K.M.; Ajaiyeoba, E.O.; Ogbole, O.O.; Adeniji, J.A.; Faleye, T.C.; Agunu, A. Antioxidant, Brine Shrimp Lethality, and Antiproliferative Properties of Gel and Leaf Extracts of Aloe Schweinfurthii and Aloe Vera. J. Herbs Spices Med. Plants 2017, 23, 263–271. [Google Scholar] [CrossRef]
- Al-Oqail, M.M.; El-Shaibany, A.; Al-Jassas, E.; Al-Sheddi, E.S.; Al-Massarani, S.M.; Farshori, N.N. In Vitro Anti-Proliferative Activities of Aloe Perryi Flowers Extract on Human Liver, Colon, Breast, Lung, Prostate and Epithelial Cancer Cell Lines. Pak. J. Pharm. Sci. 2016, 8, 723–729. [Google Scholar]
- Mehrabi, Z.; Firouzbakhsh, F.; Rahimi-Mianji, G.; Paknejad, H. Immunostimulatory Effect of Aloe Vera (Aloe Barbadensis) on Non-Specific Immune Response, Immune Gene Expression, and Experimental Challenge with Saprolegnia Parasitica in Rainbow Trout (Oncorhynchus Mykiss). Aquaculture 2019, 503, 330–338. [Google Scholar] [CrossRef]
- Celestino, V.R.L.; Maranhão, H.M.L.; Vasconcelos, C.F.B.; Lima, C.R.; Medeiros, G.C.R.; Araújo, A.V.; Wanderley, A.G. Acute Toxicity and Laxative Activity of Aloe Ferox Resin. Rev. Bras. Farmacogn. 2013, 23, 279–283. [Google Scholar] [CrossRef]
- Galli, C.L.; Cinelli, S.; Ciliutti, P.; Melzi, G.; Marinovich, M. Aloe-Emodin, a Hydroxyanthracene Derivative, Is Not Genotoxic in an In Vivo Comet Test. Regul. Toxicol. Pharmacol. 2021, 124, 104967. [Google Scholar] [CrossRef] [PubMed]
- Banni, M. Evidence of the Genotoxic Effect of Aloin on Earth Worms Eisenia Andrei. Focus Med. Sci. J. 2017, 3, 1–6. [Google Scholar]
- Nesslany, F.; Simar-Meintières, S.; Ficheux, H.; Marzin, D. Aloe-Emodin-Induced DNA Fragmentation in the Mouse In Vivo Comet Assay. Mutat. Res. 2009, 678, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Younes, M.; Aggett, P.; Aguilar, F.; Crebelli, R.; Filipič, M.; Frutos, M.J.; Galtier, P.; Gott, D.; Gundert-Remy, U.; Kuhnle, G.G.; et al. Safety of Hydroxyanthracene Derivatives for Use in Food. EFSA J. 2018, 16, e05090. [Google Scholar] [CrossRef]
- Boudreau, M.D.; Beland, F.A.; Nichols, J.A.; Pogribna, M. Toxicology and Carcinogenesis Studies of a Nondecolorized [Corrected] Whole Leaf Extract of Aloe Barbadensis Miller (Aloe Vera) in F344/N Rats and B6C3F1 Mice (Drinking Water Study). Natl. Toxicol. Program Tech. Rep. Ser. 2013, Aug, 1–266, Erratum in: Natl. Toxicol. Program Tech. Rep. Ser. 2013, Aug, 1. PMID: 24042237. [Google Scholar] [PubMed]
- Sehgal, I.; Winters, W.D.; Scott, M.; David, A.; Gillis, G.; Stoufflet, T.; Nair, A.; Kousoulas, K. Toxicologic Assessment of a Commercial Decolorized Whole Leaf Aloe Vera Juice, Lily of the Desert Filtered Whole Leaf Juice with Aloesorb. J. Toxicol. 2013, 2013, 802453. [Google Scholar] [CrossRef]
- Hu, J.; Lloyd, M.; Hobbs, C.; Cox, P.; Burke, K.; Pearce, G.; Streicker, M.A.; Gao, Q.; Frankos, V. Absence of Genotoxicity of Purified Aloe Vera Whole Leaf Dry Juice as Assessed by an In Vitro Mouse Lymphoma Tk Assay and an In Vivo Comet Assay in Male F344 Rats. Toxicol. Rep. 2021, 8, 511–519. [Google Scholar] [CrossRef]
- Matsuda, Y.; Yokohira, M.; Suzuki, S.; Hosokawa, K.; Yamakawa, K.; Zeng, Y.; Ninomiya, F.; Saoo, K.; Kuno, T.; Imaida, K. One-Year Chronic Toxicity Study of Aloe Arborescens Miller Var. Natalensis Berger in Wistar Hannover Rats. A Pilot Study. Food Chem. Toxicol. 2008, 46, 733–739. [Google Scholar] [CrossRef]
- Guo, X.; Mei, N. Aloe Vera: A Review of Toxicity and Adverse Clinical Effects. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2016, 34, 77–96. [Google Scholar] [CrossRef]
- Langmead, L.; Makins, R.J.; Rampton, D.S. Anti-Inflammatory Effects of Aloe Vera Gel in Human Colorectal Mucosa In Vitro. Aliment. Pharm. 2004, 19, 521–527. [Google Scholar] [CrossRef]
- Ajmera, N.; Chatterjee, A.; Goyal, V. Aloe Vera: It’s Effect on Gingivitis. J. Indian Soc. Periodontol. 2013, 17, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Klaikeaw, N.; Wongphoom, J.; Werawatganon, D.; Chayanupatkul, M.; Siriviriyakul, P. Anti-Inflammatory and Anti-Oxidant Effects of Aloe Vera in Rats with Non-Alcoholic Steatohepatitis. World J. Hepatol. 2020, 12, 363–377. [Google Scholar] [CrossRef] [PubMed]
- Gebrelibanos, M.; Gebremedhin, G.; Karim, A.; Sintayehu, B.; Periasamy, G. In-vitro hyaluronidase inhibition properties of aloe camperi, aloe percrassa and senna singueana. Int. J. Pharmacogn. 2014, 1, 701–704. [Google Scholar]
- Abo-Youssef, A.M.H.; Messiha, B.A.S. Beneficial Effects of Aloe Vera in Treatment of Diabetes: Comparative In Vivo and In Vitro Studies. Bull. Fac. Pharm. Cairo Univ. 2013, 51, 7–11. [Google Scholar] [CrossRef][Green Version]
- Pothuraju, R.; Sharma, R.K.; Onteru, S.K.; Singh, S.; Hussain, S.A. Hypoglycemic and Hypolipidemic Effects of Aloe Vera Extract Preparations: A Review. Phytother. Res. 2016, 30, 200–207. [Google Scholar] [CrossRef]
- Amare, G.G.; Meharie, B.G.; Belayneh, Y.M. Evaluation of Antidiabetic Activity of the Leaf Latex of Aloe Pulcherrima Gilbert and Sebsebe (Aloaceae). Evid. Based Complement. Altern. Med. 2020, 2020, e8899743. [Google Scholar] [CrossRef]
- Prasannaraja, C.; Kamalanathan, A.S.; Vijayalakshmi, M.A.; Venkataraman, K. A Dipyrrole Derivative from Aloe Vera Inhibits an Anti-Diabetic Drug Target Dipeptidyl Peptidase (DPP)-IV In Vitro. Prep. Biochem. Biotechnol. 2020, 50, 511–520. [Google Scholar] [CrossRef]
- Elsayed, A.; Ezzat, S.; Mohammed, N.; Seham, S. Chemical Composition and Evaluation of Possible Alpha Glucosidase Inhibitory Activity of Eight Aloe Species. J. Med. Plants Res. 2016, 10, 167–178. [Google Scholar] [CrossRef]
- Abbaoui, A.; Hiba, O.E.; Gamrani, H. Neuroprotective Potential of Aloe Arborescens against Copper Induced Neurobehavioral Features of Parkinson’s Disease in Rat. Acta Histochem. 2017, 119, 592–601. [Google Scholar] [CrossRef]
- Guven, M.; Gölge, U.H.; Aslan, E.; Sehitoglu, M.H.; Aras, A.B.; Akman, T.; Cosar, M. The Effect of Aloe Vera on Ischemia—Reperfusion Injury of Sciatic Nerve in Rats. Biomed. Pharmacother. 2016, 79, 201–207. [Google Scholar] [CrossRef]
- Rathor, N.; Arora, T.; Manocha, S.; Patil, A.N.; Mediratta, P.K.; Sharma, K.K. Anticonvulsant Activity of Aloe Vera Leaf Extract in Acute and Chronic Models of Epilepsy in Mice. J. Pharm. Pharmacol. 2014, 66, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Majdi, A.; Sadigh-Eteghad, S.; Aghsan, S.R.; Farajdokht, F.; Vatandoust, S.M.; Namvaran, A.; Mahmoudi, J. Amyloid-β, Tau, and the Cholinergic System in Alzheimer’s Disease: Seeking Direction in a Tangle of Clues. Rev. Neurosci. 2020, 31, 391–413. [Google Scholar] [CrossRef] [PubMed]
- Clementi, M.; Tringali, G.; Triggiani, D.; Giardina, B. Aloe Arborescens Extract Protects IMR-32 Cells against Alzheimer Amyloid Beta Peptide via Inhibition of Radical Peroxide Production. Nat. Prod. Commun. 2015, 10, 1993–1995. [Google Scholar] [CrossRef] [PubMed]
- Comparative Efficacy of Aloe Vera and Benzydamine Mouthwashes on Radiation-Induced Oral Mucositis: A Triple-Blind, Randomised, Controlled Clinical Trial. Oral Health Prev. Dent. 2015, 13, 309–315. [CrossRef]
- Liang, J.; Cui, L.; Li, J.; Guan, S.; Zhang, K.; Li, J. Aloe Vera: A Medicinal Plant Used in Skin Wound Healing. Tissue Eng. Part B Rev. 2020, 27, 455–474. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, M.N.; Ahmed, N. Effectiveness of Aloe Vera Gel Compared with 1% Silver Sulphadiazine Cream as Burn Wound Dressing in Second Degree Burns. J. Pak. Med. Assoc. 2013, 63, 225–230. [Google Scholar] [PubMed]
- Sari, Y.; Purnawan, I.; Kurniawan, D.; Sutrisna, E. A Comparative Study of the Effects of Nigella Sativa Oil Gel and Aloe Vera Gel on Wound Healing in Diabetic Rats. J. Evid. Based Integr. Med. 2018, 23, 2515690X1877280. [Google Scholar] [CrossRef]
- Hanieh Alamolhoda, S.; Mirabi, P.; Mojab, F. Effects of Both Aloe Vera Gel and Breast Milk on the Improvement of Nipple Soreness in Lactating Women—A Randomized Controlled Trial. J. Herb. Med. 2020, 21, 100327. [Google Scholar] [CrossRef]
- Fox, L.T.; Mazumder, A.; Dwivedi, A.; Gerber, M.; du Plessis, J.; Hamman, J.H. In Vitro Wound Healing and Cytotoxic Activity of the Gel and Whole-Leaf Materials from Selected Aloe Species. J. Ethnopharmacol. 2017, 200, 1–7. [Google Scholar] [CrossRef]
- Rodríguez-González, V.M.; Femenia, A.; González-Laredo, R.F.; Rocha-Guzmán, N.E.; Gallegos-Infante, J.A.; Candelas-Cadillo, M.G.; Ramírez-Baca, P.; Simal, S.; Rosselló, C. Effects of Pasteurization on Bioactive Polysaccharide Acemannan and Cell Wall Polymers from Aloe Barbadensis Miller. Carbohydr. Polym. 2011, 86, 1675–1683. [Google Scholar] [CrossRef]
- Minjares-Fuentes, R.; Rodríguez-González, V.M.; González-Laredo, R.F.; Eim, V.; González-Centeno, M.R.; Femenia, A. Effect of Different Drying Procedures on the Bioactive Polysaccharide Acemannan from Aloe Vera (Aloe Barbadensis Miller). Carbohydr. Polym. 2017, 168, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Grabowska, K.; Podolak, I.; Galanty, A.; Załuski, D.; Makowska-Wąs, J.; Sobolewska, D.; Janeczko, Z.; Żmudzki, P. In Vitro Anti-Denaturation and Anti-Hyaluronidase Activities of Extracts and Galactolipids from Leaves of Impatiens Parviflora DC. Nat. Prod. Res. 2016, 30, 1219–1223. [Google Scholar] [CrossRef] [PubMed]
- Chipiti, T.; Ibrahim, M.A.; Singh, M.; Islam, M.S. In Vitro α-Amylase and α-Glucosidase Inhibitory and Cytotoxic Activities of Extracts from Cissus Cornifolia Planch Parts. Pharm. Mag. 2017, 13, S329–S333. [Google Scholar] [CrossRef]
- Kobus-Cisowska, J.; Szymanowska, D.; Maciejewska, P.; Kmiecik, D.; Gramza-Michałowska, A.; Kulczyński, B.; Cielecka-Piontek, J. In Vitro Screening for Acetylcholinesterase and Butyrylcholinesterase Inhibition and Antimicrobial Activity of Chia Seeds (Salvia Hispanica). Electron. J. Biotechnol. 2019, 37, 1–10. [Google Scholar] [CrossRef]

| Compound | Content in a Freeze-Dried Leaf (mg/g d.m.) |
|---|---|
| Aloin A | 1.44 ± 0.24 |
| Aloenin A | 3.98 ± 0.89 |
| Sum of polyphenols | 352.24 ± 1.75 |
| Sum of phenolic acids | 187.38 ± 12.88 |
| Microorganism | Diameter of Inhibition Zone (mm)—Aloe arborescens Extract | Diameter of Inhibition Zone (mm)—5.25% Sodium Hypochlorite |
|---|---|---|
| Clostridium difficile ATCC 9689 | 0.8 | 15.3 |
| Clostridium butyricum ATTC 860 | 1.3 | 12.1 |
| Listeria monocytogenes ATCC 7644 | 4.0 | 18.4 |
| Bacillus subtilis ATCC 238557 | 8.2 | 16.8 |
| Enterococcus faecalis ATTC 29212 | 2.2 | 23.1 |
| Staphylococcus aureus ATCC 25923 | 9.1 | 18.9 |
| Staphylococcus pyogenes ATCC 19615 | 8.9 | 21.3 |
| Escherichia coli ATCC 25922 | 9.2 | 18.9 |
| Klebsiell apneumoniae ATCC 31488 | 11.3 | 24.5 |
| Proteus mirabilis ATCC 12453 | 4.3 | 19.8 |
| Salmonella typhimurium ATCC 14028 | 3.8 | 17.7 |
| Pseudomonas aereginosa ATCC 27853 | 9.3 | 24.8 |
| Enterobacter aerogenes ATCC 13048 | 1.2 | 19.3 |
| Candida krusei ATCC 14243 | 9.2 | 18.3 |
| Candida albicans ATTC 10231 | 10.7 | 20.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pawłowicz, K.; Sip, S.; Plech, T.; Kaproń, B.; Kobus-Cisowska, J.; Cielecka-Piontek, J. Aloe arborescens: In Vitro Screening of Genotoxicity, Effective Inhibition of Enzyme Characteristics for Disease Etiology, and Microbiological Activity. Molecules 2022, 27, 2323. https://doi.org/10.3390/molecules27072323
Pawłowicz K, Sip S, Plech T, Kaproń B, Kobus-Cisowska J, Cielecka-Piontek J. Aloe arborescens: In Vitro Screening of Genotoxicity, Effective Inhibition of Enzyme Characteristics for Disease Etiology, and Microbiological Activity. Molecules. 2022; 27(7):2323. https://doi.org/10.3390/molecules27072323
Chicago/Turabian StylePawłowicz, Kamil, Szymon Sip, Tomasz Plech, Barbara Kaproń, Joanna Kobus-Cisowska, and Judyta Cielecka-Piontek. 2022. "Aloe arborescens: In Vitro Screening of Genotoxicity, Effective Inhibition of Enzyme Characteristics for Disease Etiology, and Microbiological Activity" Molecules 27, no. 7: 2323. https://doi.org/10.3390/molecules27072323
APA StylePawłowicz, K., Sip, S., Plech, T., Kaproń, B., Kobus-Cisowska, J., & Cielecka-Piontek, J. (2022). Aloe arborescens: In Vitro Screening of Genotoxicity, Effective Inhibition of Enzyme Characteristics for Disease Etiology, and Microbiological Activity. Molecules, 27(7), 2323. https://doi.org/10.3390/molecules27072323









