Synthesis of Fibrin-Type I Collagen Biomaterials via an Acidic Gel
Abstract
:1. Introduction
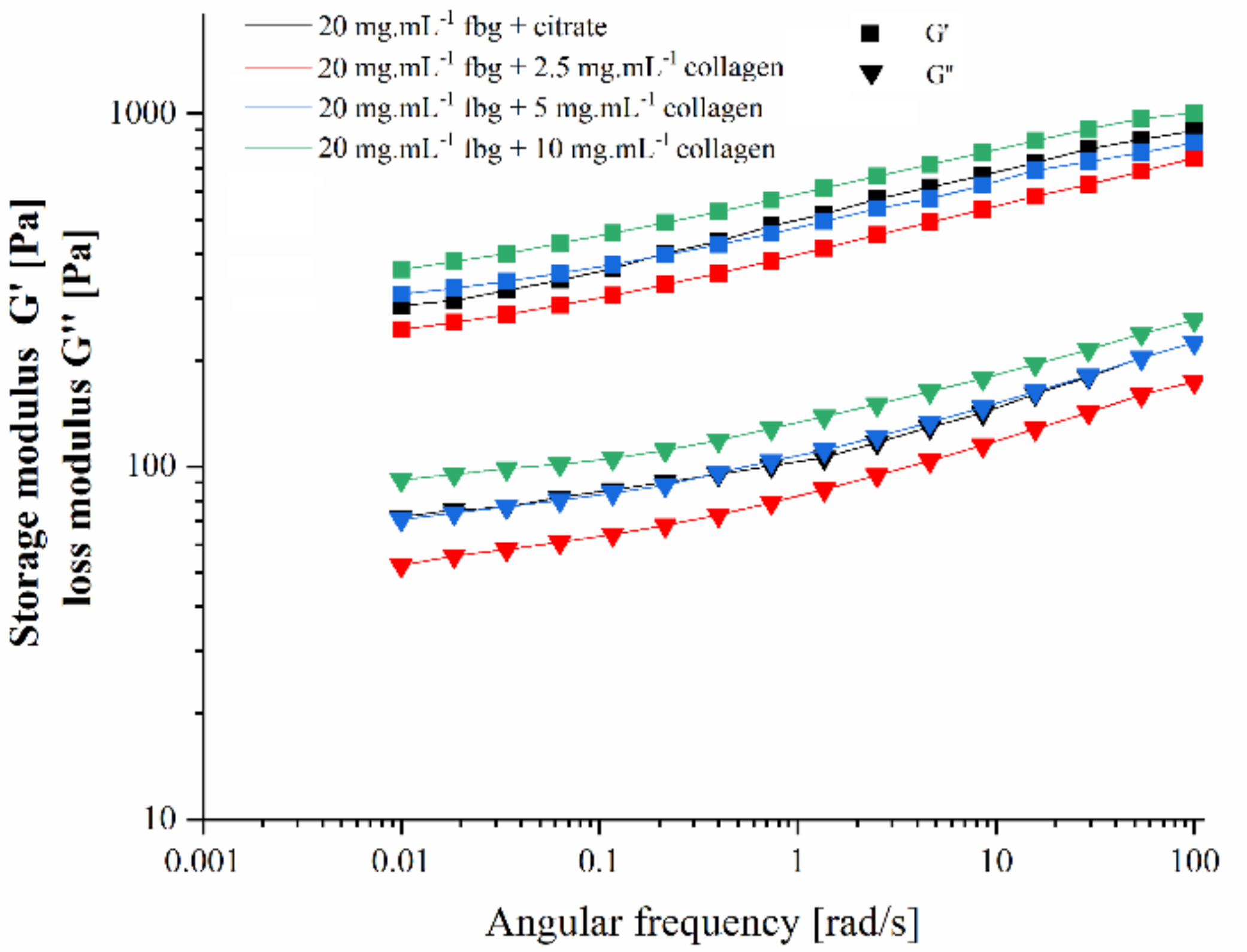
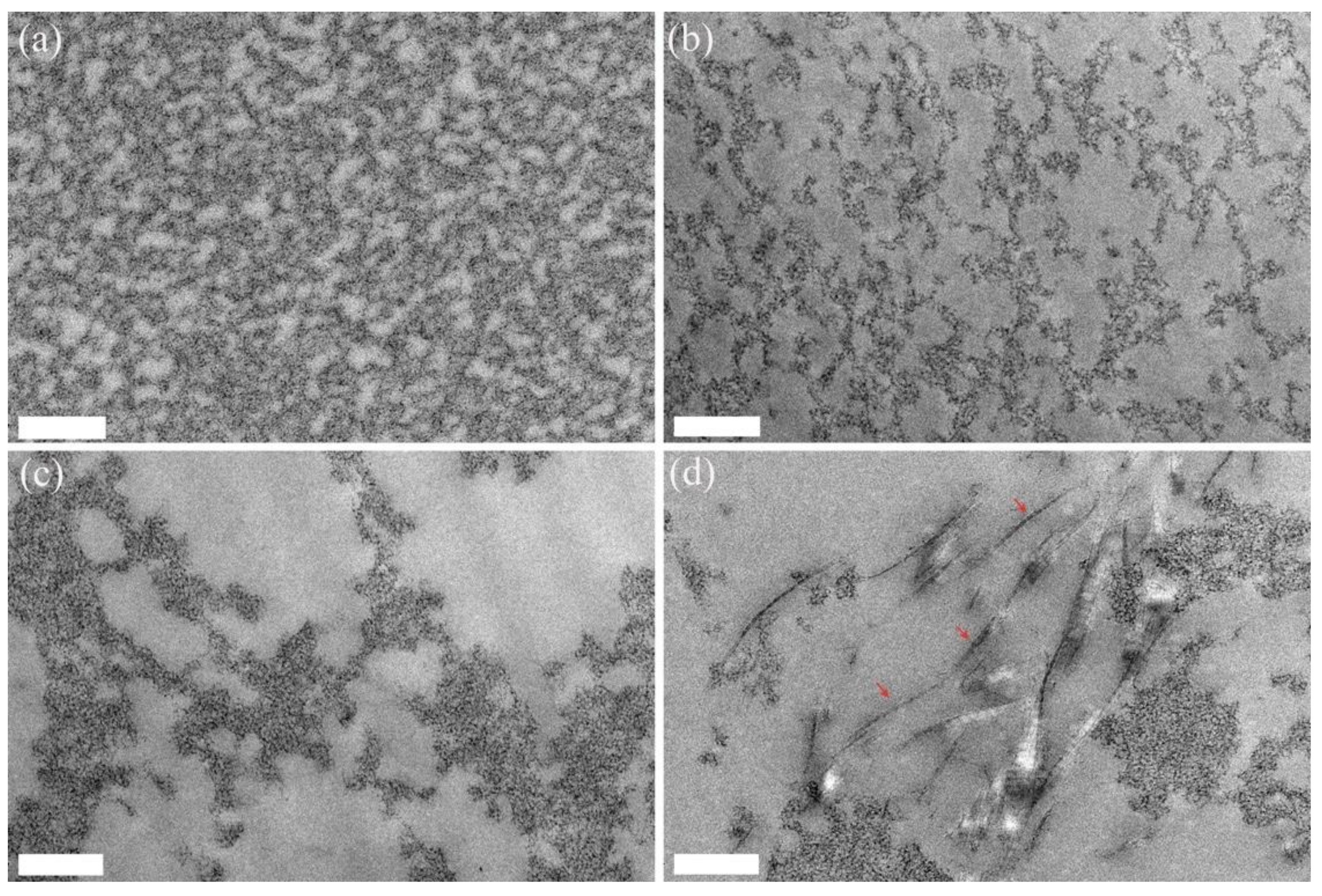
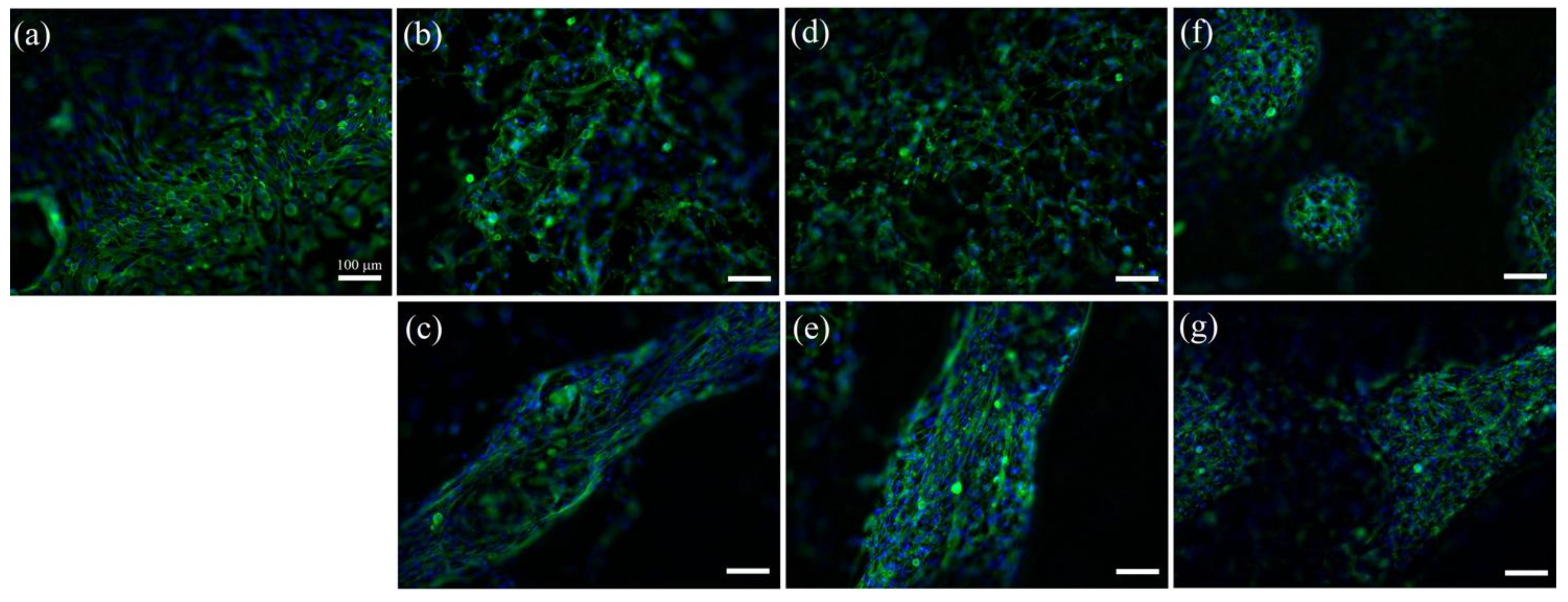
2. Results
3. Discussion
4. Materials and Methods
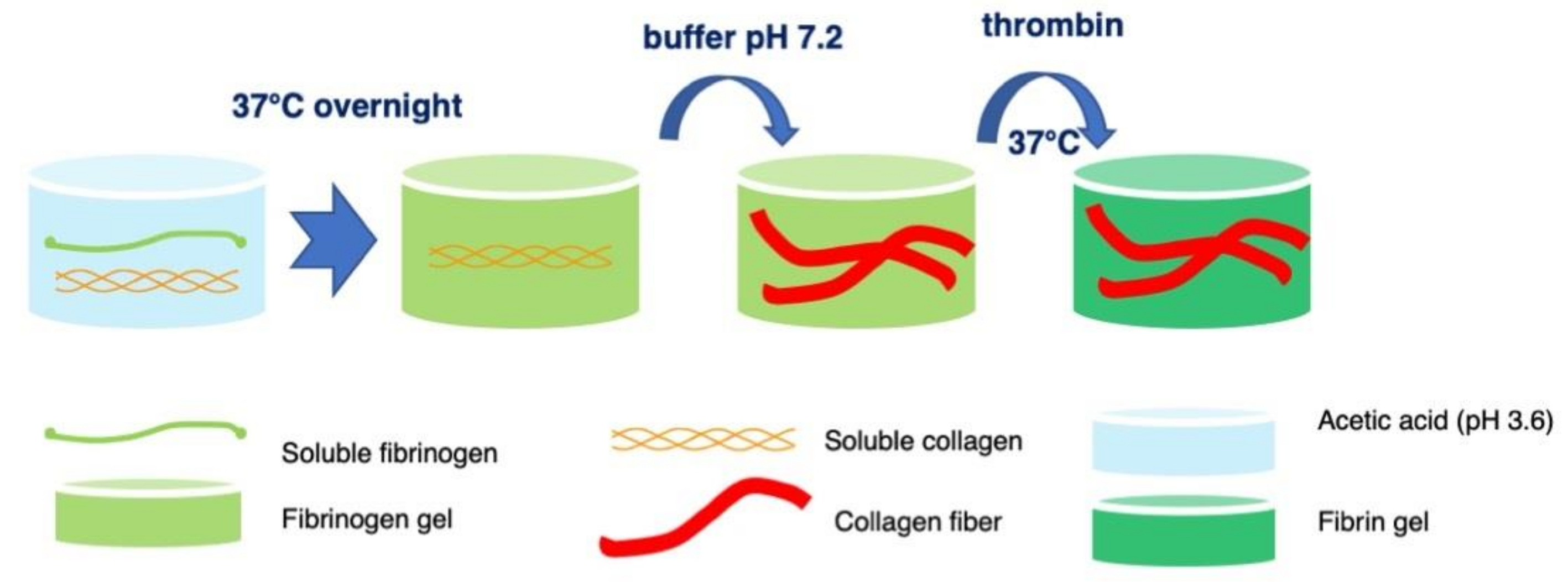
4.1. Formation of Fibrin-Type I Collagen Hydrogels
4.2. Rheological Studies
4.3. Scanning Electron Microscopy (SEM) Analysis
4.4. Transmission Electron Microscopy (TEM)
4.5. Cell Studies
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pieters, M.; Wolberg, A.S. Fibrinogen and fibrin: An illustrated review. Res. Pract. Thromb. Haemost. 2019, 3, 161–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weisel, J.W.; Litvinov, R.I. Fibrin formation, Structure and Properties. Subcell Biochem. 2017, 82, 405–456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferry, J.D.; Morrison, P.R. Preparation and Properties of Serum and Plasma Proteins. VIII. The Conversion of Human Fibrinogen to Fibrin under Various Conditions. J. Am. Chem. Soc. 1947, 69, 388–400. [Google Scholar] [CrossRef] [PubMed]
- Hethershaw, E.L.; Cilia La Corte, A.L.; Duval, C.; Ali, M.; Grant, P.J.; Ariëns, R.A.S.; Philippou, H. The effect of blood coagulation factor XIII on fibrin clot structure and fibrinolysis. J. Thromb. Haemost. 2014, 12, 197–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, H.; Ma, L.; Zhou, J.; Mao, Z.; Gao, C.; Shen, J. Fabrication and physical and biological properties of fibrin gel derived from human plasma. Biomed. Mater. 2008, 3, 015001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janmey, P.A.; Winer, J.P.; Weisel, J.W. Fibrin gels and their clinical and bioengineering applications. J. R. Soc. Interface 2009, 6, 1–10. [Google Scholar] [CrossRef]
- Rowe, S.L.; Lee, S.; Stegemann, J.P. Influence of thrombin concentration on the mechanical and morphological properties of cell-seeded fibrin hydrogels. Acta Biomater. 2007, 3, 59–67. [Google Scholar] [CrossRef] [Green Version]
- Stamboroski, S.; Joshi, A.; Noeske, P.-L.M.; Köppen, S.; Brüggemann, D. Principles of fibrinogen fiber assembly in vitro. Macromol. Biosci. 2021, 21, 2000412. [Google Scholar] [CrossRef]
- Gamboa-Martinez, T.C.; Luque-Guillen, V.; Gonzalez-Garcia, C.; Gomez Ribelles, J.L.; Gallego-Ferrer, G. Crosslinked fibrin gels for tissue engineering: Two approaches to improve their properties. J. Biomed. Mater. Res. 2014, 103, 614–621. [Google Scholar] [CrossRef]
- Wang, K.; Trichet, L.; Rieu, C.; Peccate, C.; Pembouong, G.; Bouteiller, L.; Coradin, T. Interactions of organosilanes with fibrinogen and their influence on muscle cell proliferation in 3D fibrin hydrogels. Biomacromolecules 2019, 20, 3684–3695. [Google Scholar] [CrossRef]
- Bujoli, B.; Scimeca, J.-C.; Verron, E. Fibrin as a multipurpose physiological platform for bone tissue engineering and targeted delivery of bioactive compounds. Pharmaceutics 2019, 11, 556. [Google Scholar] [CrossRef]
- Wang, K.; Albert, K.; Mosser, G.; Haye, B.; Percot, A.; Paris, C.; Peccate, C.; Trichet, L.; Coradin, T. Self-assembly/condensation interplay in nano-to-microfibrillar silicified fibrin hydrogels. Int. J. Biol. Macromol. 2020, 164, 1422–1431. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Mosser, G.; Haye, B.; Baccile, N.; Le Griel, P.; Pernot, P.; Cathala, B.; Trichet, L.; Coradin, T. Cellulose nanocrystal-fibrin nanocomposite hydrogels promoting myotube formation. Biomacromolecules 2021, 22, 2740–2753. [Google Scholar] [CrossRef] [PubMed]
- Ducret, M.; Montembault, A.; Josse, J.; Pasdeloup, M.; Cell, A.; Benchrih, R.; Mallein-Gerin, F.; Alliot-Licht, B.; David, L.; Farges, J.C. Design and characterization of a chitosan-enriched fibrin hydrogel for human dental pulp regeneration. Dental Mater. 2019, 35, 523–533. [Google Scholar] [CrossRef]
- Zhou, H.Z.; Xu, H.H.K. The fast release of stem cells from alginate-fibrin microbeads in injectable scaffolds for bone tissue engineering. Biomaterials 2011, 32, 7503–7513. [Google Scholar] [CrossRef] [Green Version]
- Hakam, M.S.; Imani, R.; Abolfathi, N.; Fakhrzadeh, H.; Sharifi, A.M. Evaluation of fibrin-gelatin hydrogel as biopaper for application in skin bioprinting: An in-vitro study. Biomed. Mater. Eng. 2016, 27, 669–682. [Google Scholar] [CrossRef]
- Parenteau-Bareil, R.; Gauvin, R.; Berthod, F. Collagen-Based Biomaterials for Tissue Engineering Applications. Materials 2010, 3, 1863–1887. [Google Scholar] [CrossRef] [Green Version]
- Antoine, E.E.; Vlachos, P.P.; Rylander, M.N. Review of Collagen I Hydrogels for Bioengineered Tissue Microenvironments: Characterization of Mechanics, Structure and Transport. Tissue Eng. Part B Rev. 2014, 20, 683–696. [Google Scholar] [CrossRef] [Green Version]
- Zhu, S.; Yuan, Q.; Yin, T.; You, J.; Gu, Z.; Xiong, S.; Hu, Y. Self-assembly of collagen-based biomaterials: Preparation, characterizations and biomedical applications. J. Mater. Chem. B 2018, 6, 2650–2676. [Google Scholar] [CrossRef]
- Lin, K.; Zhang, D.; Macedo, M.H.; Cui, W.; Sarmento, B.; Shen, G. Advanced Collagen-Based Biomaterials for Regenerative Biomedicine. Adv. Funct. Mater. 2019, 29, 1804943. [Google Scholar] [CrossRef]
- Walters, B.D.; Stegemann, J.P. Strategies for directing the structure and function of three-dimensional collagen biomaterials across length scales. Acta Biomater. 2014, 10, 1488–1501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hélary, C.; Abde, A.; Mosser, G.; Louedec, L.; Letourneur, D.; Coradin, T.; Giraud-Guille, M.M.; Meddahi-Pellé, A. Evaluation of dense collagen matrices as medicated wound dressing for the treatment of cutaneous chronic wounds. Biomater. Sci. 2015, 3, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Meyer, M. Processing of collagen-based biomaterials and the resulting materials properties. BioMed. Eng. OnLine 2019, 18, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Griffanti, G.; Nazhat, S.N. Dense fibrillar collagen-based hydrogels as functional osteoid-mimicking scaffolds. Int. Mater. Rev. 2020, 65, 502–521. [Google Scholar] [CrossRef]
- Cummings, C.L.; Gawlitta, D.; Nerem, R.M.; Stegemann, J.P. Properties of engineered vascular constructs made from collagen, fibrin, and collagen–fibrin mixtures. Biomaterials 2004, 25, 3699–3706. [Google Scholar] [CrossRef]
- Rowe, S.L.; Stegemann, J.P. Interpenetrating collagen-fibrin composite matrices with varying protein contents and ratios. Biomacromolecules 2006, 7, 2942–2948. [Google Scholar] [CrossRef] [Green Version]
- Lai, V.K.; Lake, S.P.; Frey, C.R.; Tranquillo, R.T.; Barocas, V.H. Mechanical behavior of collagen-fibrin co-gels reflects transition from series to parallel interactions with increasing collagen content. J. Biomech. Eng. 2012, 134, 011004. [Google Scholar] [CrossRef]
- Hong, H.; Stegemann, J.P. 2D and 3D collagen and fibrin biopolymers promote specific ECM and integrin gene expression by vascular smooth muscle cells. J. Biomater. Sci. Polym. Ed. 2008, 19, 1279–1293. [Google Scholar] [CrossRef] [Green Version]
- Kaiser, N.J.; Kant, R.J.; Minor, A.J.; Coulombe, K.L.K. Optimizing blended collagen-fibrin hydrogels for cardiac tissue engineering with human ipsc-derived cardiomyocytes. ACS Biomater. Sci. Eng. 2018, 5, 887–899. [Google Scholar] [CrossRef] [Green Version]
- Coradin, T.; Wang, K.; Law, T.; Trichet, L. Type I collagen-fibrin mixed hydrogels:preparation, properties and biomedical applications. Gels 2020, 6, 36. [Google Scholar] [CrossRef]
- Nedrelow, D.S.; Bankwala, D.; Hyypio, J.D.; Lai, V.K.; Barocas, V.H. Mechanics of a two-fiber model with one nested fiber network, as applied to the collagen-fibrin system. Acta Biomater. 2018, 72, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Giraud-Guille, M.M.; Hélary, C.; Vigier, S.; Nassif, N. Dense fibrillar collagen matrices for tissue repair. Soft Matter 2010, 6, 4963–4967. [Google Scholar] [CrossRef] [PubMed]
- Rieu, C.; Mosser, G.; Haye, B.; Sanson, N.; Coradin, T.; Trichet, L. Thrombin-free polymerization leads to pure fibrin(ogen) materials with extended processing capacity. bioRxiv 2020. [Google Scholar] [CrossRef]
- Mahmood, A.; Patel, D.; Hickson, B.; DesRochers, J.; Hu, X. Recent progress in biopolymer-based hydrogel materials for biomedical applications. Int. J. Mol. Sci. 2022, 23, 1415. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.; Poverenov, E. Natural biopolymer-based hydrogels for use in food and agriculture. J. Sci. Food Agric. 2020, 100, 2337–2347. [Google Scholar] [CrossRef] [PubMed]
- Tabani, H.; Alexovic, M.; Sabo, J.; Payan, M.R. An overview on the recent applications of agarose as a green biopolymer in micro-extraction-based sample preparation techniques. Talanta 2021, 224, 121892. [Google Scholar] [CrossRef] [PubMed]
- Light, N.; Champion, A.E. Characterization of muscle epimysium, perimysium and endomysium collagens. Biochem. J. 1984, 219, 1017–1026. [Google Scholar] [CrossRef] [Green Version]
- Kitakaze, T.; Sakamoto, T.; Kitano, T.; Inoue, N.; Sugihara, F.; Harada, N.; Yamaji, R. The collagen derived dipeptide hydroxyprolyl-glycine promotes C2C12 myoblast differentiation and myotube hypertrophy. Biochem. Biophys. Res. Commun. 2016, 478, 1292–1297. [Google Scholar] [CrossRef]
- Picaut, L.; Trichet, L.; Hélary, C.; Ducourthial, G.; Bonnin, M.-A.; Haye, B.; Ronsin, O.; Schanne-Klein, M.-C.; Duprez, D.; Baumberger, T. Core-shell pure collagen threads extruded from highly concentrated solutions promote colonization and differentiation of C3H10T1/2 cells. ACS Biomater. Sci. Eng. 2021, 7, 626–635. [Google Scholar] [CrossRef]
- O’Brien, F.J.; Harley, B.A.; Yannas, I.V.; Gibson, L.J. The effect of pore size on cell adhesion in collagen-GAG scaffolds. Biomaterials 2005, 26, 433–441. [Google Scholar] [CrossRef]
- Ross, J.J.; Tranquillo, R.T. ECM gene expression correlates with in vitro tissue growth and development in fibrin gel remodeled by neonatal smooth muscle cells. Matrix Biol. 2003, 22, 477–490. [Google Scholar] [CrossRef] [PubMed]
- Delaporte, C.; Dautreaux, B.; Fardeau, M. Human myotube differentiation in vitro in different culture conditions. Biol. Cell 1986, 57, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Van Wachem, P.B.; Brouwer, L.A.; van Luyn, M.J. Absence of muscle regeneration after implantation of a collagen matrix seeded with myoblasts. Biomaterials 1999, 20, 416–419. [Google Scholar] [CrossRef] [PubMed]
- Hung, F.H.; Sillitti, D.; Shrirao, A.B.; Shrieber, D.I.; Firestein, B.L. Collagen nanofiber anisotropy induces myotube differentiation and acetylcholine receptor clustering. J. Tissue Eng. Regen. Med. 2018, 12, e2010–e2019. [Google Scholar] [CrossRef]
- Hu, L.-Y.; Mileti, C.J.; Loomis, T.; Brashear, S.E.; Ahmad, S.; Chellakudam, R.R.; Wohlgemuth, R.P.; Gionet-Gonzales, M.A.; Leach, K.; Smith, L.R. Skeletal muscle progenitors are sensitive to collagen architectural features of fibril size and cross-linking. Am. J. Physiol. Cell Physiol. 2021, 321, C330–C342. [Google Scholar] [CrossRef]
- Rieu, C.; Rose, N.; Taleb, A.; Mosser, G.; Haye, B.; Coradin, T.; Le Grand, F.; Trichet, L. Differential myoblast and tenoblast affinity to collagen, fibrin and mixed threads in the prospect of muscle-tendon junction modelisation. bioRxiv 2020. [Google Scholar] [CrossRef]
- Rieu, C.; Parisi, C.; Mosser, G.; Haye, B.; Coradin, T.; Fernandes, F.M.; Trichet, L. Topotactic Fibrillogenesis of Freeze-Cast Microridged Collagen Scaffolds for 3D Cell Culture. ACS Appl. Mater. Interfaces 2019, 11, 14672–14683. [Google Scholar] [CrossRef]
- De Sa Peixoto, P.; Deniset-Besseau, A.; Schanne-Klein, M.-C.; Mosser, G. Achievement of cornea-like organizations in dense collagen I solutions: Clues to the physico-chemistry of cornea morphogenesis. Soft Matter. 2011, 7, 11203–11210. [Google Scholar] [CrossRef]
- Shrinivas, P.; Kasapis, S.; Tongdang, T. Morphology and mechanical properties of bicontinuous gels of agarose and gelatin and the effect of added lipid phase. Langmuir 2009, 25, 8763–8773. [Google Scholar] [CrossRef]
- Chicatun, F.; Pedraza, C.E.; Ghezzi, C.E.; Marelli, B.; Kaartinen, M.T.; McKee, M.D.; Nazhat, S.N. Osteoid-mimicking dense collagen/chitosan hybrid gels. Biomacromolecules 2011, 12, 2946–2956. [Google Scholar] [CrossRef]
- Bergman, I.; Loxley, R. Two improved ad simplified methods for the spectrophotometric determination of hydroxyproline. Anal. Chem. 1963, 35, 1961–1965. [Google Scholar] [CrossRef]
- Gobeaux, F.; Mosser, G.; Anglo, A.; Panine, P.; Davidson, P.; Giraud-Guille, M.-M.; Belamie, E. Fibrillogenesis in dense collagen solutions: A physicochemical study. J. Mol. Biol. 2008, 376, 1509–1522. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, K.; Camman, M.; Mosser, G.; Haye, B.; Trichet, L.; Coradin, T. Synthesis of Fibrin-Type I Collagen Biomaterials via an Acidic Gel. Molecules 2022, 27, 2099. https://doi.org/10.3390/molecules27072099
Wang K, Camman M, Mosser G, Haye B, Trichet L, Coradin T. Synthesis of Fibrin-Type I Collagen Biomaterials via an Acidic Gel. Molecules. 2022; 27(7):2099. https://doi.org/10.3390/molecules27072099
Chicago/Turabian StyleWang, Kun, Marie Camman, Gervaise Mosser, Bernard Haye, Léa Trichet, and Thibaud Coradin. 2022. "Synthesis of Fibrin-Type I Collagen Biomaterials via an Acidic Gel" Molecules 27, no. 7: 2099. https://doi.org/10.3390/molecules27072099
APA StyleWang, K., Camman, M., Mosser, G., Haye, B., Trichet, L., & Coradin, T. (2022). Synthesis of Fibrin-Type I Collagen Biomaterials via an Acidic Gel. Molecules, 27(7), 2099. https://doi.org/10.3390/molecules27072099






