Anti-Inflammatory Dysidazirine Carboxylic Acid from the Marine Cyanobacterium Caldora sp. Collected from the Reefs of Fort Lauderdale, Florida †
Abstract
:1. Introduction
2. Results and Discussion
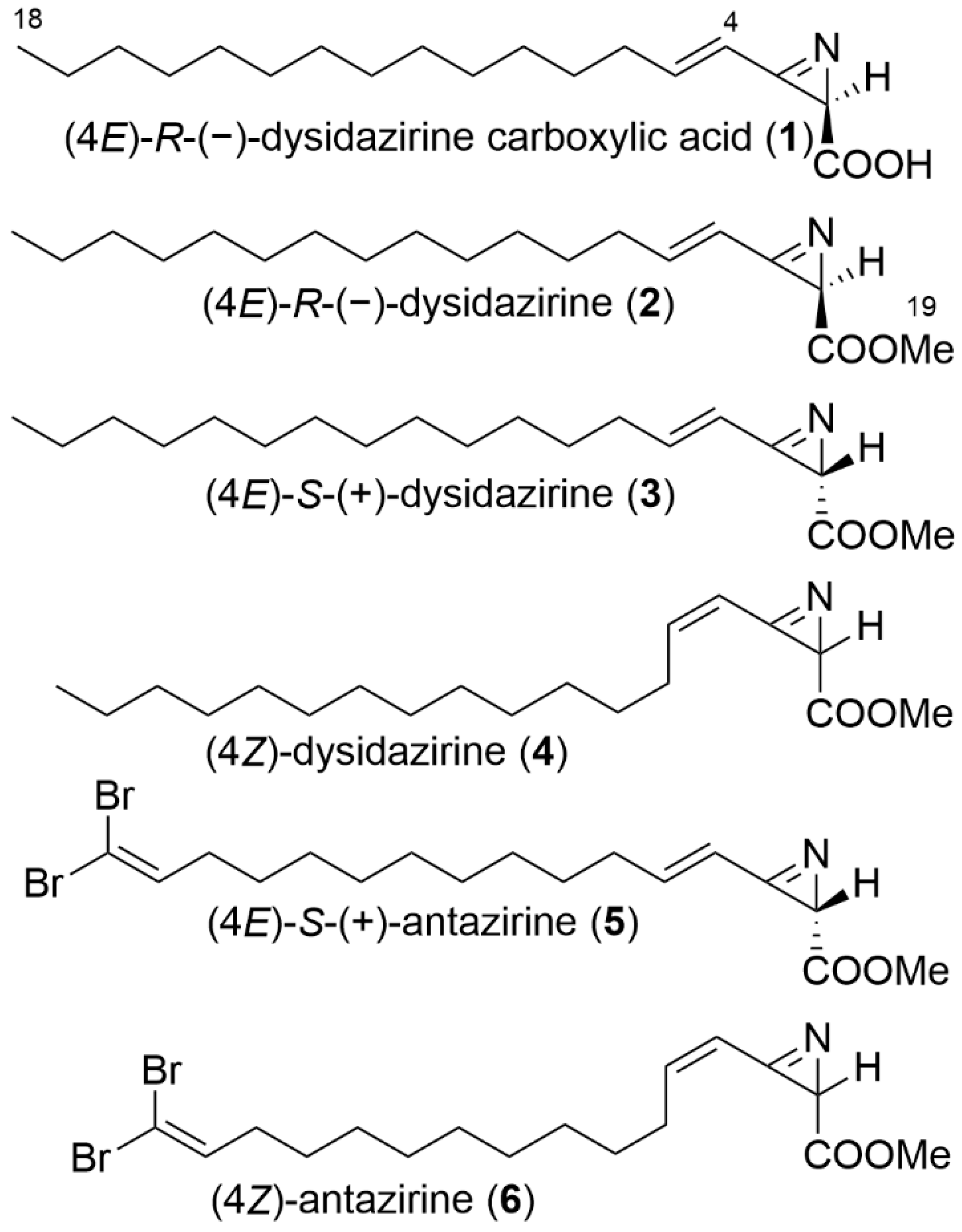
2.1. Isolation and Structure Elucidation
2.2. Identification of the Cyanobacterium
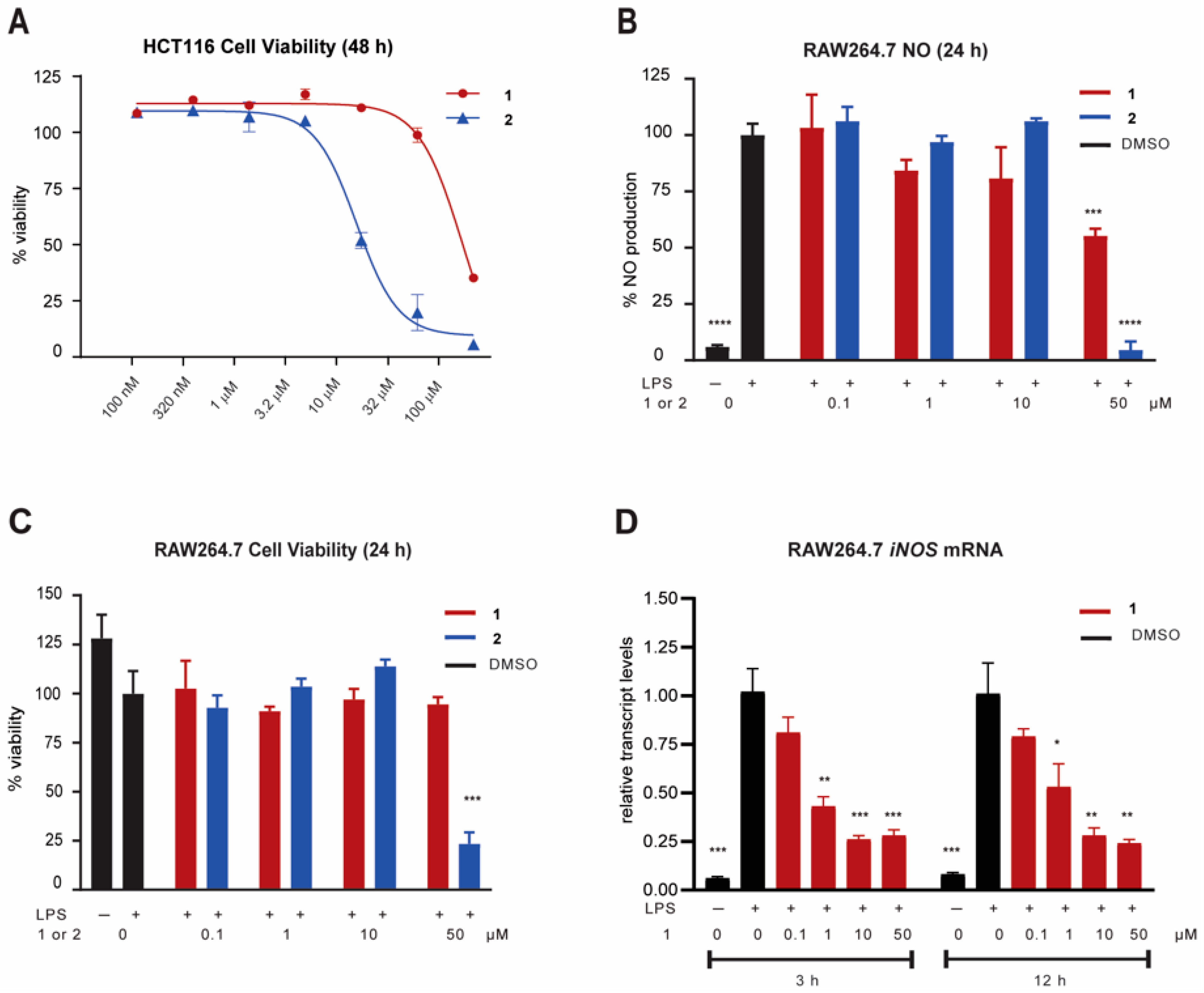
2.3. Biological Activity
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Collection, Extraction and Isolation
3.3. Molecular Identification
3.4. Cancer Cell Viability Assay (HCT116)
3.5. NO Assay and Cell Viability Assay (RAW264.7 Cells)
3.6. iNOS mRNA Measurement in RAW264.7 Cells
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Miller, T.W.; Tristram, E.W.; Wolf, F.J. Azirinomycin. II. Isolation and chemical characterization as 3-methyl-2(2H) azirinecarboxylic acid. J. Antibiot. 1971, 24, 48–50. [Google Scholar] [CrossRef] [Green Version]
- Stapley, E.O.; Hendlin, D.; Jackson, M.; Miller, A.; Hernandez, S.; Mata, J.M. Azirinomycin. I. Microbial production and biological characteristics. J. Antibiot. 1971, 24, 42–47. [Google Scholar] [CrossRef]
- Molinski, T.F.; Ireland, C.M. Dysidazirine, a cytotoxic azacyclopropene from the marine sponge Dysidea fragilis. J. Org. Chem. 1988, 53, 2103–2105. [Google Scholar] [CrossRef]
- Salomon, C.E.; Williams, D.H.; Faulkner, D.J. New azacyclopropene derivatives from Dysidea fragilis collected in Pohnpei. J. Nat. Prod. 1995, 58, 1463–1466. [Google Scholar] [CrossRef] [PubMed]
- Skepper, C.K.; Molinski, T.F. Long-chain 2H-azirines with heterogeneous terminal halogenation from the marine sponge Dysidea fragilis. J. Org. Chem. 2008, 73, 2592–2597. [Google Scholar] [CrossRef] [PubMed]
- Keffer, J.L.; Plaza., A.; Bewley, C.A. Motualevic acids A-F, antimicrobial acids from the sponge Siliquariaspongia sp. Org. Lett. 2009, 11, 1087–1090. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skepper, C.K.; Dalisay, D.S.; Molinski, T.F. Synthesis and chain-dependent antifungal activity of long chain 2H-azairine-carboxylate esters related to dysidazirine. Bioorg. Med. Chem. Lett. 2010, 20, 2029–2032. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakharov, P.A.; Koronatov, A.N.; Khlebnikov, A.F.; Novikov, M.S.; Glukharev, A.G.; Rogacheva, E.V.; Kraeva, L.A.; Sharoyko, V.V.; Tennikova, T.B.; Rostovskii, N.V. Non-natural 2H-azirine-2-carboxylic acids: An expedient synthesis and antimicrobial activity. RSC Adv. 2019, 9, 37901–37905. [Google Scholar] [CrossRef] [Green Version]
- Brumley, D.; Spencer, K.A.; Gunasekera, S.P.; Sauvage, T.; Biggs, J.; Paul, V.J.; Luesch, H. Isolation and characterization of Anaephenes A–C, alkylphenols from a filamentous cyanobacterium (Hormoscilla sp., Oscillatoriales). J. Nat. Prod. 2018, 81, 2716–2721. [Google Scholar] [CrossRef] [PubMed]
- Engene, N.; Tronholm, A.; Salvador-Reyes, L.; Luesch, H.; Paul, V.J. Caldora penicillata gen. nov., comb. nov. (Cyanobacteria), a pantropical marine species with biomedical relevance. J. Phycol. 2015, 51, 670–681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matthew, S.; Chen, Q.-Y.; Ratnayake, R.; Fermaintt, C.S.; Lucena-Agell, D.; Bonato, F.; Prota, A.E.; Lim, S.T.; Wang, X.; Diaz, J.F.; et al. Gatorbulin-1, a distinct cyclodepsipeptide chemotype, targets a seventh tubulin pharmacological site. Proc. Natl. Acad. Sci. USA 2021, 118, e2021847118. [Google Scholar] [CrossRef] [PubMed]


| C No | δC Mult. | δH (J in Hz) | COSY | HMBC a | δC Mult. | δH (J in Hz) |
|---|---|---|---|---|---|---|
| 1 b | 2 c,d | |||||
| 1 | 176.2, C | 2 | 172.2, C | |||
| 2 | 29.4, CH | 2.49, s | 4 | 28.3, CH | 2.45, s | |
| 3 | 157.1, C | 2, 4, 5 | 156.6, C | |||
| 4 | 114.1, CH | 6.61, d (15.1) | 5, 6 | 2, 5 | 112.9, CH | 6.47, d (15.8) |
| 5 | 158.4, CH | 6.75, dt (15.1, 6.9) | 4, 6 | 6, 7 | 155.8, CH | 6.64, dt (15.8, 6.8) |
| 6 | 34.1, CH2 | 2.38, dt (6.9, 7.4) | 5, 7 | 4, 5, 7 | 33.2, CH2 | 2.28, dt (6.8, 7.5) |
| 7 | 29.0, CH2 | 1.52, m | 6, 8 | 5, 6 | 29.1, CH2 | 1.44, m |
| 8–15 | 30.3–30.8, CH2 | 1.35–1.24, m | 29.2–29.7, CH2 | 1.1–1.3, m | ||
| 16 | 33.0, CH2 | 1.32, m | 18 | 33.2, CH2 | 1.17, m | |
| 17 | 23.7, CH2 | 1.24, m | 18 | 18 | 22.6, CH2 | 1.17, m |
| 18 | 14.4, CH3 | 0.89, t (6.8) | 17 | 16, 17 | 14.1, CH3 | 0.79, t (6.8) |
| 19 | 52.2, CH3 | 3.34, s | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gunasekera, S.P.; Kokkaliari, S.; Ratnayake, R.; Sauvage, T.; dos Santos, L.A.H.; Luesch, H.; Paul, V.J. Anti-Inflammatory Dysidazirine Carboxylic Acid from the Marine Cyanobacterium Caldora sp. Collected from the Reefs of Fort Lauderdale, Florida. Molecules 2022, 27, 1717. https://doi.org/10.3390/molecules27051717
Gunasekera SP, Kokkaliari S, Ratnayake R, Sauvage T, dos Santos LAH, Luesch H, Paul VJ. Anti-Inflammatory Dysidazirine Carboxylic Acid from the Marine Cyanobacterium Caldora sp. Collected from the Reefs of Fort Lauderdale, Florida. Molecules. 2022; 27(5):1717. https://doi.org/10.3390/molecules27051717
Chicago/Turabian StyleGunasekera, Sarath P., Sofia Kokkaliari, Ranjala Ratnayake, Thomas Sauvage, Larissa A. H. dos Santos, Hendrik Luesch, and Valerie J. Paul. 2022. "Anti-Inflammatory Dysidazirine Carboxylic Acid from the Marine Cyanobacterium Caldora sp. Collected from the Reefs of Fort Lauderdale, Florida" Molecules 27, no. 5: 1717. https://doi.org/10.3390/molecules27051717
APA StyleGunasekera, S. P., Kokkaliari, S., Ratnayake, R., Sauvage, T., dos Santos, L. A. H., Luesch, H., & Paul, V. J. (2022). Anti-Inflammatory Dysidazirine Carboxylic Acid from the Marine Cyanobacterium Caldora sp. Collected from the Reefs of Fort Lauderdale, Florida. Molecules, 27(5), 1717. https://doi.org/10.3390/molecules27051717







