Promising Strategies in Plant-Derived Treatments of Psoriasis-Update of In Vitro, In Vivo, and Clinical Trials Studies
Abstract
:1. Introduction
2. Epidemiology and Pathophysiology of Psoriasis
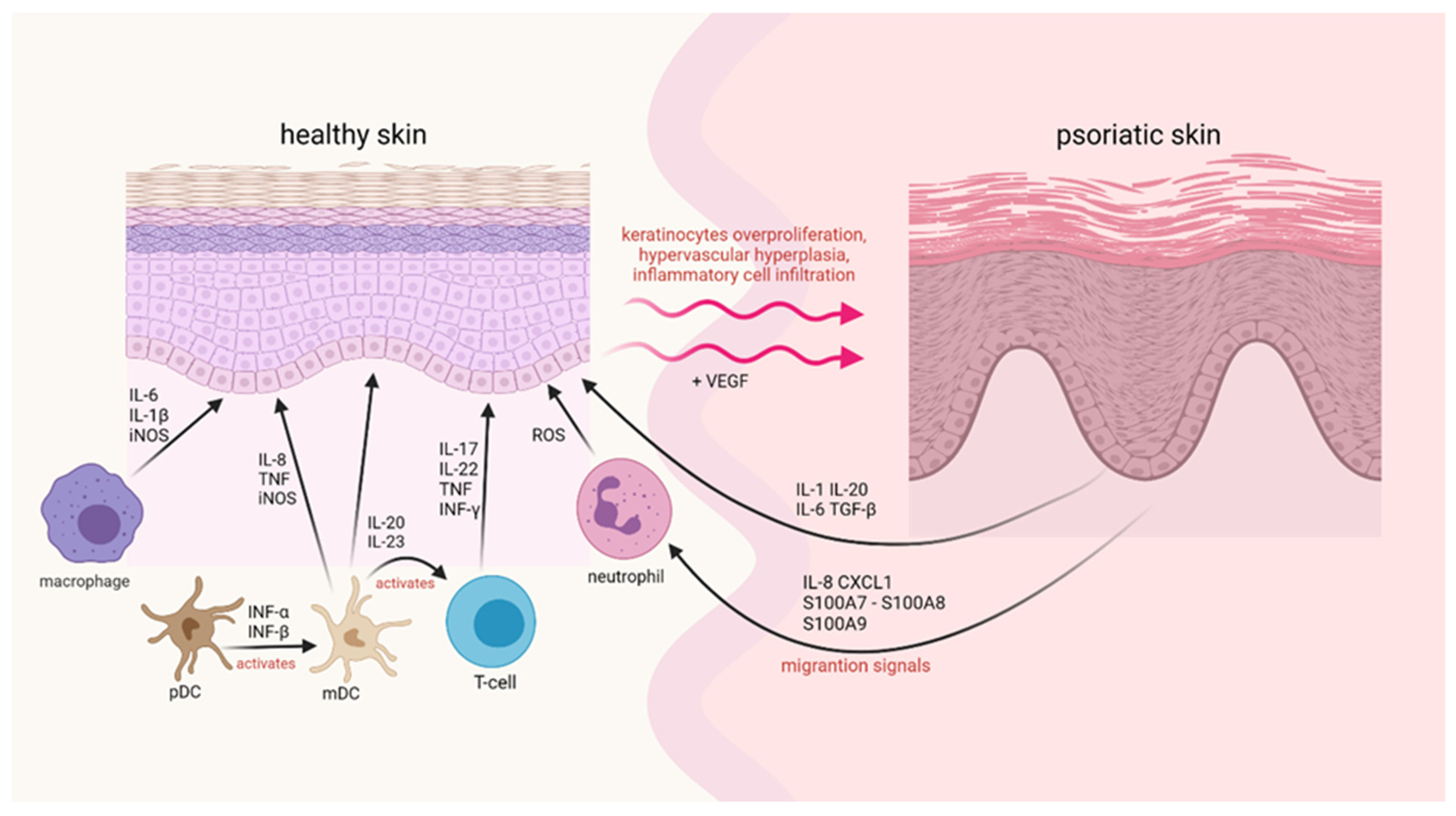
3. Immune System Response in Psoriasis
4. Oxidative Stress in the Course of Psoriasis
4.1. ROS
4.2. NOS
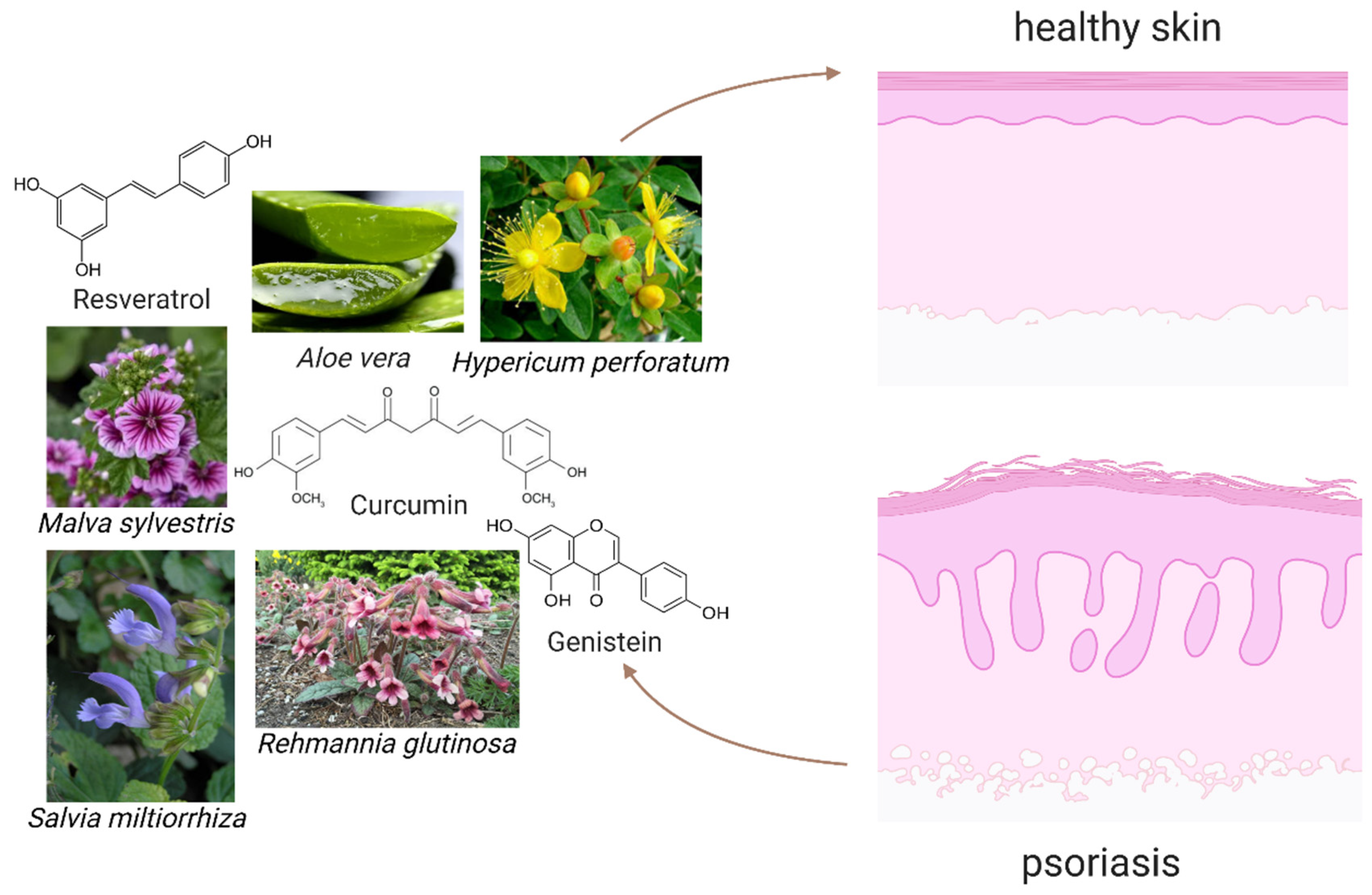
5. Substances of Natural Origin—Plants and Phytochemicals with Potential Therapeutic Significance in Reduction of ROS and iNOS
5.1. Vegetal Medicinal Species
5.1.1. Aloe Vera
5.1.2. Artemisia Capillaris
5.1.3. St. John’s Wort
5.1.4. Rehmannia Glutinosa
5.1.5. Salvia Miltiorrhiza
5.2. Alkaloid
Capsaicin
5.3. Polyphenol
5.3.1. Resveratrol
5.3.2. Curcumin
5.3.3. Rottlerin
5.4. Flavonoids
5.4.1. Quercetin
5.4.2. Apigenin
5.4.3. Kaempferol
5.4.4. Genistein
5.4.5. Rutin
5.4.6. Naringenin
5.4.7. Naringin
5.4.8. Epigallocatechin-3-gallate (EGCG)
5.4.9. Anthocyanidins
5.5. Carotenoid
Lycopene
5.6. Anthraquinone
Emodin
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Krueger, J.G. The Immunologic Basis for the Treatment of Psoriasis with New Biologic Agents. J. Am. Acad. Dermatol. 2002, 46, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, C.E.M.; Christophers, E.; Barker, J.N.W.N.; Chalmers, R.J.G.; Chimenti, S.; Krueger, G.G.; Leonardi, C.; Menter, A.; Ortonne, J.-P.; Fry, L. A Classification of Psoriasis Vulgaris According to Phenotype. Br. J. Dermatol. 2007, 156, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Di Meglio, P.; Villanova, F.; Nestle, F.O. Psoriasis. Cold Spring Harb. Perspect. Med. 2014, 4, a015354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benhadou, F.; Mintoff, D.; Schnebert, B.; Thio, H. Psoriasis and Microbiota: A Systematic Review. Diseases 2018, 6, 47. [Google Scholar] [CrossRef] [Green Version]
- Bowcock, A.M.; Krueger, J.G. Getting under the Skin: The Immunogenetics of Psoriasis. Nat. Rev. Immunol. 2005, 5, 699–711. [Google Scholar] [CrossRef]
- Schön, M.P.; Boehncke, W.-H. Psoriasis. N. Engl. J. Med. 2005, 352, 1899–1912. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A.V.E.D.; Romiti, R.; Souza, C.D.S.; Paschoal, R.S.; Milman, L.D.M.; Meneghello, L.P. Psoriasis comorbidities: Complications and benefits of immunobiological treatment. An. Bras. Dermatol. 2016, 91, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Aghmiuni, A.I.; Khiavi, A.A. Medicinal plants to calm and treat psoriasis disease. Aromat. Med. Plants–Back Nat. 2016, 1, 1–28. [Google Scholar] [CrossRef] [Green Version]
- Kaur, A.; Kumar, S. Plants and plant products with potential antipsoriatic activity—A review. Pharm. Biol. 2012, 50, 1573–1591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christophers, E. Psoriasis—Epidemiology and Clinical Spectrum. Clin. Exp. Dermatol. 2001, 26, 314–320. [Google Scholar] [CrossRef]
- Burch, P.R.J. Mode of Inheritance in Psoriasis. Arch. Dermatol. 1981, 117, 251. [Google Scholar] [CrossRef]
- Smith, A.E.; Kassab, J.Y.; Payne, R.; Beer, W.E. Bimodality in Age of Onset of Psoriasis, in Both Patients and Their Relatives. Dermatology 1993, 186, 181–186. [Google Scholar] [CrossRef]
- Ferrándiz, C.; Pujol, R.M.; Garc, P.V.; Bordas, X.; Smand, J.A. Psoriasis of Early and Late Onset: A Clinical and Epidemiologic Study from Spain. J. Am. Acad. Dermatol. 2002, 46, 867–873. [Google Scholar] [CrossRef]
- Fahlén, A.; Engstrand, L.; Baker, B.S.; Powles, A.; Fry, L. Comparison of bacterial microbiota in skin biopsies from normal and psoriatic skin. Arch. Dermatol. Res. 2012, 304, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Rendon, A.; Schäkel, K. Psoriasis Pathogenesis and Treatment. Int. J. Mol. Sci. 2019, 20, 1475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dopytalska, K.; Ciechanowicz, P.; Wiszniewski, K.; Szymańska, E.; Walecka, I. The role of epigenetic factors in psoriasis. Int. J. Mol. Sci. 2021, 22, 9294. [Google Scholar] [CrossRef]
- Shao, S.; Gudjonsson, J.E. Epigenetics of psoriasis. Epigenet. Allergy Autoimmun. 2020, 1253, 209–221. [Google Scholar] [CrossRef]
- Henseler, T.; Christophers, E. Psoriasis of Early and Late Onset: Characterization of Two Types of Psoriasis Vulgaris. J. Am. Acad. Dermatol. 1985, 13, 450–456. [Google Scholar] [CrossRef]
- Ayala, F. Clinical Presentation of Psoriasis. Reumatismo 2011, 59, 40–45. [Google Scholar] [CrossRef]
- Takeshita, J.; Grewal, S.; Langan, S.M.; Mehta, N.N.; Ogdie, A.; van Voorhees, A.S.; Gelfand, J.M. Psoriasis and Comorbid Diseases. J. Am. Acad. Dermatol. 2017, 76, 377–390. [Google Scholar] [CrossRef] [Green Version]
- Nestle, F.O.; Conrad, C.; Tun-Kyi, A.; Homey, B.; Gombert, M.; Boyman, O.; Burg, G.; Liu, Y.-J.; Gilliet, M. Plasmacytoid Predendritic Cells Initiate Psoriasis through Interferon-Production. J. Exp. Med. 2005, 202, 135–143. [Google Scholar] [CrossRef]
- Lowes, M.A.; Chamian, F.; Abello, M.V.; Fuentes-Duculan, J.; Lin, S.-L.; Nussbaum, R.; Novitskaya, I.; Carbonaro, H.; Cardinale, I.; Kikuchi, T.; et al. Increase in TNF-~ and Inducible Nitric Oxide Synthase-Expressing Dendritic Cells in Psoriasis and Reduction with Efalizumab (Anti-CD11a). Proc. Natl. Acad. Sci. USA 2005, 102, 19057–19062. [Google Scholar] [CrossRef] [Green Version]
- Lowes, M.A.; Bowcock, A.M.; Krueger, J.G. Pathogenesis and Therapy of Psoriasis. Nature 2007, 445, 866–873. [Google Scholar] [CrossRef]
- Serbina, N.V.; Salazar-Mather, T.P.; Biron, C.A.; Kuziel, W.A.; Pamer, E.G. TNF/INOS-Producing Dendritic Cells Mediate Innate Immune Defense against Bacterial Infection. Immunity 2003, 19, 59–70. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Lee, E.; Lowes, M.A.; Haider, A.S.; Fuentes-Duculan, J.; Abello, M.V.; Chamian, F.; Cardinale, I.; Krueger, J.G. Prominent Production of IL-20 by CD68/CD11c Myeloid-Derived Cells in Psoriasis: Gene Regulation and Cellular Effects. J. Investig. Dermatol. 2006, 126, 1590–1599. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Krueger, J.G.; Kao, M.-C.J.; Lee, E.; Du, F.; Menter, A.; Wong, W.H.; Bowcock, A.M. Novel Mechanisms of T-Cell and Dendritic Cell Activation Revealed by Profiling of Psoriasis on the 63,100-Element Oligonucleotide Array. Physiol. Genom. 2003, 13, 69–78. [Google Scholar] [CrossRef] [Green Version]
- Lew, W.; Bowcock, A.M.; Krueger, J.G. Psoriasis Vulgaris: Cutaneous Lymphoid Tissue Supports T-Cell Activation and ‘Type 1’ Inflammatory Gene Expression. Trends Immunol. 2004, 25, 295–305. [Google Scholar] [CrossRef]
- McKenzie, B.S.; Kastelein, R.A.; Cua, D.J. Understanding the IL-23IL-17 Immune Pathway. Trends Immunol. 2006, 27, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Nickoloff, B.J.; Bonish, B.; Huang, B.B.; Porcelli, S.A. Characterization of a T Cell Line Bearing Natural Killer Receptors and Capable of Creating Psoriasis in a SCID Mouse Model System. J. Dermatol. Sci. 2000, 24, 212–225. [Google Scholar] [CrossRef]
- Prinz, J.C.; Groß, B.; Vollmer, S.; Trommler, P.; Strobel, I.; Meurer, M.; Plewig, G. T Cell Clones from Psoriasis Skin Lesions Can Promote Keratinocyte Proliferationin Vitro via Secreted Products. Eur. J. Immunol. 1994, 24, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, H.; Gyulai, R.; Toichi, E.; Garaczi, E.; Shimada, S.; Stevens, S.R.; McCormick, T.S.; Cooper, K.D. Dysfunctional Blood and Target Tissue CD4CD25high Regulatory T Cells in Psoriasis: Mechanism Underlying Unrestrained Pathogenic Effector T Cell Proliferation. J. Immunol. 2004, 174, 164–173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Green, S.; Dobrjansky, A.; Carswell, E.A.; Kassel, R.L.; Old, L.J.; Fiore, N.; Schwartz, M.K. Partial Purification of a Serum Factor That Causes Necrosis of Tumors. Proc. Natl. Acad. Sci. USA 1976, 73, 381–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finch, P.W.; Murphy, F.; Cardinale, I.; Krueger, J.G. Altered Expression of Keratinocyte Growth Factor and Its Receptor in Psoriasis. Am. J. Pathol. 1997, 151, 1619–1628. [Google Scholar]
- Lambert, A.J.; Brand, M.D. Reactive Oxygen Species Production by Mitochondria. In Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2009; pp. 165–181. [Google Scholar]
- Bayr, H. Reactive Oxygen Species. Crit. Care Med. 2005, 33, S498–S501. [Google Scholar] [CrossRef] [PubMed]
- Turrens, J.F. Mitochondrial Formation of Reactive Oxygen Species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef]
- Slimen, I.B.; Najar, T.; Ghram, A.; Dabbebi, H.; Ben Mrad, M.; Abdrabbah, M. Reactive Oxygen Species, Heat Stress and Oxidative-Induced Mitochondrial Damage. A Review. Int. J. Hyperth. 2014, 30, 513–523. [Google Scholar] [CrossRef]
- Brieger, K.; Schiavone, S.; Miller, F.J., Jr.; Krause, K.H. Reactive Oxygen Species: From Health to Disease. Swiss Med. Wkly. 2012, 142, w13659. [Google Scholar] [CrossRef] [PubMed]
- Richter, K.; Kietzmann, T. Reactive Oxygen Species and Fibrosis: Further Evidence of a Significant Liaison. Cell Tissue Res. 2016, 365, 591–605. [Google Scholar] [CrossRef] [Green Version]
- Alderton, W.K.; Cooper, C.E.; Knowles, R.G. Nitric Oxide Synthases: Structure, Function and Inhibition. Biochem. J. 2001, 357, 593–615. [Google Scholar] [CrossRef]
- Aktan, F. INOS-Mediated Nitric Oxide Production and Its Regulation. Life Sci. 2004, 75, 639–653. [Google Scholar] [CrossRef]
- Zhou, Q.; Mrowietz, U.; Rostami-Yazdi, M. Oxidative Stress in the Pathogenesis of Psoriasis. Free Radic. Biol. Med. 2009, 47, 891–905. [Google Scholar] [CrossRef] [PubMed]
- Kroencke, K.-D.; Fehsel, K.; Kolb-bachofen, V. Inducible Nitric Oxide Synthase and Its Product Nitric Oxide, a Small Molecule with Complex Biological Activities. Biol. Chem. Hoppe-Seyler. 1995, 376, 327–343. [Google Scholar] [CrossRef]
- Forstermann, U.; Sessa, W.C. Nitric Oxide Synthases: Regulation and Function. Eur. Heart J. 2011, 33, 829–837. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sies, H. Oxidative Stress: A Concept in Redox Biology and Medicine. Redox Biol. 2015, 4, 180–183. [Google Scholar] [CrossRef] [Green Version]
- Poljsak, B.; Šuput, D.; Milisav, I. Achieving the Balance between ROS and Antioxidants: When to Use the Synthetic Antioxidants. Oxidative Med. Cell. Longev. 2013, 2013, 956792. [Google Scholar] [CrossRef] [PubMed]
- Briganti, S.; Picardo, M. Antioxidant Activity, Lipid Peroxidation and Skin Diseases. Whats New. J. Eur. Acad. Dermatol. Venereol. 2003, 17, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T.; Holbrook, N.J. Oxidants, Oxidative Stress and the Biology of Ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef]
- Havens, C.G.; Ho, A.; Yoshioka, N.; Dowdy, S.F. Regulation of Late G/S Phase Transition and APC Cdh1 by Reactive Oxygen Species. Mol. Cell. Biol. 2006, 26, 4701–4711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baek, J.-O.; Byamba, D.; Wu, W.H.; Kim, T.-G.; Lee, M.-G. Assessment of an Imiquimod-Induced Psoriatic Mouse Model in Relation to Oxidative Stress. Arch. Dermatol. Res. 2012, 304, 699–706. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Huang, T. Oxidative Stress in Psoriasis and Potential Therapeutic Use of Antioxidants. Free Radic. Res. 2016, 50, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Shilov, V.N.; Sergienko, V.I. Oxidative Stress in Keratinocytes as an Etiopathogenetic Factor of Psoriasis. Bull. Exp. Biol. Med. 2000, 129, 309–313. [Google Scholar] [CrossRef]
- Shindo, Y.; Witt, E.; Han, D.; Epstein, W.; Packer, L. Enzymic and Non-Enzymic Antioxidants in Epidermis and Dermis of Human Skin. J. Investig. Dermatol. 1994, 102, 122–124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kadam, D.P.; Suryakar, A.N.; Ankush, R.D.; Kadam, C.Y.; Deshpande, K.H. Role of Oxidative Stress in Various Stages of Psoriasis. Indian J. Clin. Biochem. 2010, 25, 388–392. [Google Scholar] [CrossRef] [Green Version]
- Shah, A.A.; Sinha, A.A. Oxidative Stress and Autoimmune Skin Disease. Eur. J. Dermatol. 2013, 23, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Oszukowska, M.; Kozłowska, M.; Kaszuba, A. Paraoxonase-1 and Other Factors Related to Oxidative in Psoriasis. Adv. Dermatol. Allergol. 2020, 37, 92–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pleńkowska, J.; Gabig-Cimińska, M.; Mozolewski, P. Oxidative Stress as an Important Contributor to the Pathogenesis of Psoriasis. Int. J. Mol. Sci. 2020, 21, 6206. [Google Scholar] [CrossRef] [PubMed]
- Houshang, N.; Reza, K.; Masoud, S.; Ali, E.; Mansour, R.; Vaisi-Raygani, A. Antioxidant Status in Patients with Psoriasis. Cell Biochem. Funct. 2013, 32, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Gabr, S.A.; Al-Ghadir, A.H. Role of Cellular Oxidative Stress and Cytochrome c in the Pathogenesis of Psoriasis. Arch. Dermatol. Res. 2012, 304, 451–457. [Google Scholar] [CrossRef]
- Pujari, V.K.M. The Serum Levels of Malondialdehyde, Vitamin E and Erythrocyte Catalase Activity in Psoriasis Patients. J. Clin. Diagn. Res. 2014, 8, CC14–CC16. [Google Scholar] [CrossRef] [PubMed]
- Lisse, T.S.; King, B.L.; Rieger, S. Comparative Transcriptomic Profiling of Hydrogen Peroxide Signaling Networks in Zebrafish and Human Keratinocytes: Implications toward Conservation, Migration and Wound Healing. Sci. Rep. 2016, 6, 20328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barygina, V.; Becatti, M.; Prignano, F.; Lotti, T.; Taddei, N.; Fiorillo, C. Fibroblasts to Keratinocytes Redox Signaling: The Possible Role of ROS in Psoriatic Plaque Formation. Antioxidants 2019, 8, 566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Young, C.N.; Koepke, J.I.; Terlecky, L.J.; Borkin, M.S.; Boyd, S.L.; Terlecky, S.R. Reactive Oxygen Species in Tumor Necrosis Factor—Activated Primary Human Keratinocytes: Implications for Psoriasis and Inflammatory Skin Disease. J. Investig. Dermatol. 2008, 128, 2606–2614. [Google Scholar] [CrossRef] [Green Version]
- Aksoy, M.; Kirmit, A. Thiol/Disulphide Balance in Patients with Psoriasis. Adv. Dermatol. Allergol. 2020, 37, 52–55. [Google Scholar] [CrossRef] [Green Version]
- Baran, A.; Kiluk, P.; Myśliwiec, H.; Flisiak, I. The Role of Lipids in Psoriasis. Dermatol. Rev. 2017, 104, 619–635. [Google Scholar] [CrossRef] [Green Version]
- Magenta, A.; Dellambra, E.; Ciarapica, R.; Capogrossi, M.C. Oxidative Stress, MicroRNAs and Cytosolic Calcium Homeostasis. Cell Calcium 2016, 60, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Perera, G.K.; di Meglio, P.; Nestle, F.O. Psoriasis. Annu. Rev. Pathol. Mech. Dis. 2012, 7, 385–422. [Google Scholar] [CrossRef] [PubMed]
- Verhasselt, V.; Goldman, M.; Willems, F. Oxidative Stress Up-Regulates IL-8 and TNF- Synthesis by Human Dendritic Cells. Eur. J. Immunol. 1998, 28, 3886–3890. [Google Scholar] [CrossRef]
- Rutault, K.; Alderman, C.; Chain, B.M.; Katz, D.R. Reactive Oxygen Species Activate Human Peripheral Blood Dendritic Cells. Free Radic. Biol. Med. 1999, 26, 232–238. [Google Scholar] [CrossRef]
- Jain, S.; Kaur, I.R.; Das, S.; Bhattacharya, S.N.; Singh, A. T Helper 1 to T Helper 2 Shift in Cytokine Expression: An Autoregulatory Process in Superantigen-Associated Psoriasis Progression? J. Med. Microbiol. 2009, 58, 180–184. [Google Scholar] [CrossRef]
- Gostner, J.M.; Becker, K.; Fuchs, D.; Sucher, R. Redox Regulation of the Immune Response. Redox Rep. 2013, 18, 88–94. [Google Scholar] [CrossRef]
- Campanati, A.; Goteri, G.; Simonetti, O.; Ganzetti, G.; Giuliodori, K.; Giuliano, A.; Sabato, S.; Stramazzotti, D.; Gulini, E.; Dusi, D.; et al. Angiogenesis in Psoriatic Skin and Its Modifications after Administration of Etanercept: Videocapillaroscopic, Histological and Immunohistochemical Evaluation. Int. J. Immunopathol. Pharmacol. 2009, 22, 371–377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nofal, A.; Al-Makhzangy, I.; Attwa, E.; Nassar, A.; Abdalmoati, A. Vascular Endothelial Growth Factor in Psoriasis: An Indicator of Disease Severity and Control. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.W.; Voyles, S.V.; Armstrong, E.J.; Fuller, E.N.; Rutledge, J.C. Angiogenesis and Oxidative Stress: Common Mechanisms Linking Psoriasis with Atherosclerosis. J. Dermatol. Sci. 2011, 63, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Detmar, M.; Brown, L.F.; Schön, M.P.; Elicker, B.M.; Velasco, P.; Richard, L.; Fukumura, D.; Monsky, W.; Claffey, K.P.; Jain, R.K. Increased Microvascular Density and Enhanced Leukocyte Rolling and Adhesion in the Skin of VEGF Transgenic Mice. J. Investig. Dermatol. 1998, 111, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takahashi, H.; Ibe, M.; Nakamura, S.; Ishida-Yamamoto, A.; Hashimoto, Y.; Iizuka, H. Extracellular Regulated Kinase and C-Jun N-Terminal Kinase Are Activated in Psoriatic Involved Epidermis. J. Dermatol. Sci. 2002, 30, 94–99. [Google Scholar] [CrossRef]
- Johansen, C.; Kragballe, K.; Westergaard, M.; Henningsen, J.; Kristiansen, K.; Iversen, L. The Mitogen-Activated Protein Kinases P38 and ERK1/2 Are Increased in Lesional Psoriatic Skin. Br. J. Dermatol. 2005, 152, 37–42. [Google Scholar] [CrossRef]
- Yu, X.-J.; Li, C.-Y.; Dai, H.-Y.; Cai, D.-X.; Wang, K.-Y.; Xu, Y.-H.; Chen, L.-M.; Zhou, C.-L. Expression and Localization of the Activated Mitogen-Activated Protein Kinase in Lesional Psoriatic Skin. Exp. Mol. Pathol. 2007, 83, 413–418. [Google Scholar] [CrossRef]
- Efimova, T.; Broome, A.M.; Eckert, R.L. A regulatory role for p38 delta MAPK in keratinocyte differentiation. Evidence for p38 delta-ERK1/2 complex formation. J. Biol. Chem. 2003, 278, 34277–34285. [Google Scholar] [CrossRef] [Green Version]
- Morgan, M.J.; Liu, Z. Crosstalk of Reactive Oxygen Species and NF-B Signaling. Cell Res. 2010, 21, 103–115. [Google Scholar] [CrossRef] [Green Version]
- Carballo, M.; Conde, M.; el Bekay, R.; Martín-Nieto, J.; Camacho, M.J.; Monteseirín, J.; Conde, J.; Bedoya, F.J.; Sobrino, F. Oxidative Stress Triggers STAT3 Tyrosine Phosphorylation and Nuclear Translocation in Human Lymphocytes. J. Biol. Chem. 1999, 274, 17580–17586. [Google Scholar] [CrossRef] [Green Version]
- Svendsen, M.T.; Jeyabalan, J.; Andersen, K.E.; Andersen, F.; Johannessen, H. Worldwide Utilization of Topical Remedies in Treatment of Psoriasis: A Systematic Review. J. Dermatol. Treat. 2016, 28, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Farahnik, B.; Sharma, D.; Alban, J.; Sivamani, R.K. Topical Botanical Agents for the Treatment of Psoriasis: A Systematic Review. Am. J. Clin. Dermatol. 2017, 18, 451–468. [Google Scholar] [CrossRef] [PubMed]
- May, B.H.; Deng, S.; Zhang, A.L.; Lu, C.; Xue, C.C.L. In Silico Database Screening of Potential Targets and Pathways of Compounds Contained in Plants Used for Psoriasis Vulgaris. Arch. Dermatol. Res. 2015, 307, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Radha, M.H.; Laxmipriya, N.P. Evaluation of Biological Properties and Clinical Effectiveness of Aloe vera: A systematic Review. J. Tradit. Complement. Med. 2015, 5, 21–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leng, H.; Pu, L.; Xu, L.; Shi, X.; Ji, J.; Chen, K. Effects of Aloe Polysaccharide, a Polysaccharide Extracted from Aloe-vera, on TNF—induced HaCaT Cell Proliferation and the Underlying Mechanism in Psoriasis. Mol. Med. Rep. 2018, 18, 3537–3543. [Google Scholar] [CrossRef] [Green Version]
- Ha, H.; Lee, H.; Seo, C.S.; Lim, H.-S.; Lee, J.K.; Lee, M.-Y.; Shin, H. Artemisia Capillaris Inhibits Atopic Dermatitis-like Skin Lesions in Dermatophagoides Farinae-Sensitized Nc/Nga Mice. BMC Complement. Altern. Med. 2014, 14, 100. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.Y.; Nam, S.; Hong, I.K.; Kim, H.; Yang, H.; Cho, H.-J. Antiproliferation of Keratinocytes and Alleviation of Psoriasis by the Ethanol Extract of Artemisia Capillaris. Phytother. Res. 2018, 32, 923–932. [Google Scholar] [CrossRef]
- Lee, S.Y.; Nam, S.; Kim, S.; Koo, J.S.; Hong, I.K.; Kim, H.; Han, S.; Kang, M.; Yang, H.; Cho, H.-J. Therapeutic Efficacies of Artemisia Capillaris Extract Cream Formulation in Imiquimod-Induced Psoriasis Models. Evid.-Based Complement. Altern. Med. 2018, 2018, 3610494. [Google Scholar] [CrossRef] [Green Version]
- Najafizadeh, P.; Hashemian, F.; Mansouri, P.; Farshi, S.; Surmaghi, M.S.; Chalangari, R. The Evaluation of the Clinical Effect of Topical St Johns Wort (Hypericum perforatum L.) in Plaque Type Psoriasis Vulgaris: A Pilot Study. Australas. J. Dermatol. 2012, 53, 131–135. [Google Scholar] [CrossRef]
- Wölfle, U.; Seelinger, G.; Schempp, C. Topical Application of St. John’s Wort (Hypericum perforatum). Planta Med. 2013, 80, 109–120. [Google Scholar] [CrossRef] [Green Version]
- Mansouri, P.; Mirafzal, S.; Najafizadeh, P.; Safaei-Naraghi, Z.; Salehi-Surmaghi, M.; Hashemian, F. The Impact of Topical Saint John’s Wort (Hypericum perforatum) Treatment on Tissue Tumor Necrosis Factor-Alpha Levels in Plaque-Type Psoriasis: A Pilot Study. J. Postgrad. Med. 2017, 63, 215. [Google Scholar] [CrossRef]
- Yan, X.; Zhang, H.; Dang, M.; Chen, X. Rehmannia Radix Extract Ameliorates Imiquimod-Induced Psoriasis-like Skin Inflammation in a Mouse Model via the Janus-Kinase Signal Transducer and Activator of Transcription Pathway. Pharmacogn. Mag. 2020, 16, 613. [Google Scholar] [CrossRef]
- Liu, C.-L.; Cheng, L.; Ko, C.-H.; Wong, C.-W.; Cheng, W.-H.; Cheung, D.W.-S.; Leung, P.-C.; Fung, K.-P.; Lau, C.B.-S. Bioassay-Guided Isolation of Anti-Inflammatory Components from the Root of Rehmannia Glutinosa and Its Underlying Mechanism via Inhibition of INOS Pathway. J. Ethnopharmacol. 2012, 143, 867–875. [Google Scholar] [CrossRef] [PubMed]
- Baek, G.-H.; Jang, Y.-S.; Jeong, S.-I.; Cha, J.; Joo, M.; Shin, S.-W.; Ha, K.-T.; Jeong, H.-S. Rehmannia Glutinosa Suppresses Inflammatory Responses Elicited by Advanced Glycation End Products. Inflammation 2012, 35, 1232–1241. [Google Scholar] [CrossRef]
- Jia, J.; Mo, X.; Liu, J.; Yan, F.; Wang, N.; Lin, Y.; Li, H.; Zheng, Y.; Chen, D. Mechanism of Danshensu-Induced Inhibition of Abnormal Epidermal Proliferation in Psoriasis. Eur. J. Pharmacol. 2020, 868, 172881. [Google Scholar] [CrossRef]
- Tang, L.; He, S.; Wang, X.; Liu, H.; Zhu, Y.; Feng, B.; Su, Z.; Zhu, W.; Liu, B.; Xu, F.; et al. Cryptotanshinonereduces Psoriatic Epidermal Hyperplasia via Inhibiting the Activation of STAT3. Exp. Dermatol. 2018, 27, 268–275. [Google Scholar] [CrossRef]
- Maleš, Ž.; Drvar, D.L.; Duka, I.; Žužul, K. Application of Medicinal Plants in Several Dermatovenerological Entities. Acta Pharm. 2019, 69, 525–531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agrawal, U.; Gupta, M.; Vyas, S.P. Capsaicin Delivery into the Skin with Lipidic Nanoparticles for the Treatment of Psoriasis. Artif. Cells Nanomed. Biotechnol. 2013, 43, 33–39. [Google Scholar] [CrossRef]
- Khatoon, K.; Ali, A.; Ahmad, F.J.; Hafeez, Z.; Rizvi, M.M.A.; Akhter, S.; Beg, S. Novel Nanoemulsion Gel Containing Triple Natural Bio-Actives Combination of Curcumin, Thymoquinone, and Resveratrol Improves Psoriasis Therapy: In Vitro and in Vivo Studies. Drug Deliv. Transl. Res. 2020, 11, 1245–1260. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.-Y.; Su, Y.-W.; Lin, K.-I.; Hsu, L.-C.; Chuang, T.-H. Natural Modulators of Endosomal Toll-Like Receptor-Mediated Psoriatic Skin Inflammation. J. Immunol. Res. 2017, 2017, 7807313. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, A.; Monteiro, V.; Navegantes-Lima, K.; Reis, J.; Gomes, R.; Rodrigues, D.; Gaspar, S.; Monteiro, M. Resveratrol Role in Autoimmune Disease—A Mini-Review. Nutrients 2017, 9, 1306. [Google Scholar] [CrossRef] [Green Version]
- Shehzad, A.; Wahid, F.; Lee, Y.S. Curcumin in Cancer Chemoprevention: Molecular Targets, Pharmacokinetics, Bioavailability, and Clinical Trials. Arch. Pharm. 2010, 343, 489–499. [Google Scholar] [CrossRef]
- Kanai, M.; Yoshimura, K.; Asada, M.; Imaizumi, A.; Suzuki, C.; Matsumoto, S.; Nishimura, T.; Mori, Y.; Masui, T.; Kawaguchi, Y.; et al. A Phase I/II Study of Gemcitabine-Based Chemotherapy plus Curcumin for Patients with Gemcitabine-Resistant Pancreatic Cancer. Cancer Chemother. Pharmacol. 2010, 68, 157–164. [Google Scholar] [CrossRef] [Green Version]
- Wua, S.T.; Suna, J.C.; Leeb, K.; Sunc, Y.M. Docking Prediction for Tumor Necrosis Factor-α and Five Herbal Inhibitors. Int. J. Eng. Sci. Technol. 2010, 2, 4263–4270. [Google Scholar]
- Kurd, S.K.; Smith, N.; Van Voorhees, A.; Troxel, A.B.; Badmaev, V.; Seykora, J.T.; Gelfand, J.M. Oral Curcumin in the Treatment of Moderate to Severe Psoriasis Vulgaris: A Prospective Clinical Trial. J. Am. Acad. Dermatol. 2008, 58, 625–631. [Google Scholar] [CrossRef] [Green Version]
- Tu, C.; Han, B.; Yao, Q.; Zhang, Y.; Liu, H.; Zhang, S. Curcumin Attenuates Concanavalin A-Induced Liver Injury in Mice by Inhibition of Toll-like Receptor (TLR) 2, TLR4 and TLR9 Expression. Int. Immunopharmacol. 2012, 12, 151–157. [Google Scholar] [CrossRef]
- Cho, J.-W.; Lee, K.-S.; Kim, C.-W. Curcumin Attenuates the Expression of IL-1, IL-6, and TNF- as Well as Cyclin E in TNF--Treated HaCaT Cells NF-B and MAPKs as Potential Upstream Targets. Int. J. Mol. Med. 2007, 19, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Han, J.; Zhao, Y.; Zhu, Q.; Hu, J. Curcumin Induces Apoptosis in Tumor Necrosis Factor-Alpha-Treated HaCaT Cells. Int. Immunopharmacol. 2012, 13, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Heng, M.C.Y.; Song, M.K.; Harker, J.; Heng, M.K. Drug-Induced Suppression of Phosphorylase Kinase Activity Correlates with Resolution of Psoriasis as Assessed by Clinical, Histological and Immunohistochemical Parameters. Br. J. Dermatol. 2000, 143, 937–949. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Patchva, S.; Aggarwal, B.B. Therapeutic Roles of Curcumin: Lessons Learned from Clinical Trials. AAPS J. 2012, 15, 195–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torricelli, C.; Fortino, V.; Capurro, E.; Valacchi, G.; Pacini, A.; Muscettola, M.; Soucek, K.; Maioli, E. Rottlerin Inhibits the Nuclear Factor B/Cyclin-D1 Cascade in MCF-7 Breast Cancer Cells. Life Sci. 2008, 82, 638–643. [Google Scholar] [CrossRef]
- Putic, A.; Stecher, L.; Prinz, H.; Müller, K. Structureactivity Relationship Studies of Acridones as Potential Antipsoriatic Agents. 2. Synthesis and Antiproliferative Activity of 10-Substituted Hydroxy-10H-Acridin-9-Ones against Human Keratinocyte Growth. Eur. J. Med. Chem. 2010, 45, 5345–5352. [Google Scholar] [CrossRef] [PubMed]
- Min, M.; Yan, B.-X.; Wang, P.; Landeck, L.; Chen, J.-Q.; Li, W.; Cai, S.-Q.; Zheng, M.; Man, X.-Y. Rottlerin as a Therapeutic Approach0.6emin Psoriasis: Evidence from in Vitro and in Vivo Studies. PLoS ONE 2017, 12, e0190051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.-C.; Mani, A.; Cai, Y.; Thomson, J.; Ma, J.; Peudru, F.; Chen, S.; Luo, M.; Zhang, J.; Chapman, R.G.; et al. An Effective Identification and Quantification Method for Ginkgo Biloba Flavonol Glycosides with Targeted Evaluation of Adulterated Products. Phytomedicine 2016, 23, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Koyu, H.; Haznedaroglu, M.Z. Investigation of Impact of Storage Conditions on Hypericum perforatum L. Dried Total Extract. J. Food Drug Anal. 2015, 23, 545–551. [Google Scholar] [CrossRef] [Green Version]
- Xiong, H.; Xu, Y.; Tan, G.; Han, Y.; Tang, Z.; Xu, W.; Zeng, F.; Guo, Q. Glycyrrhizin Ameliorates Imiquimod-Induced Psoriasis-like Skin Lesions in BALB/c Mice and Inhibits TNF-a-Induced ICAM-1 Expression via NF-B/MAPK in HaCaT Cells. Cell. Physiol. Biochem. 2015, 35, 1335–1346. [Google Scholar] [CrossRef]
- Kiekow, C.J.; Figueiró, F.; Dietrich, F.; Vechia, L.D.; Pires, E.N.S.; Jandrey, E.H.F.; Gnoatto, S.C.B.; Salbego, C.G.; Battastini, A.M.O.; Gosmann, G. Quercetin Derivative Induces Cell Death in Glioma Cells by Modulating NF-B Nuclear Translocation and Caspase-3 Activation. Eur. J. Pharm. Sci. 2016, 84, 116–122. [Google Scholar] [CrossRef]
- Chen, H.; Lu, C.; Liu, H.; Wang, M.; Zhao, H.; Yan, Y.; Han, L. Quercetin Ameliorates Imiquimod-Induced Psoriasis-like Skin Inflammation in Mice via the NF-B Pathway. Int. Immunopharmacol. 2017, 48, 110–117. [Google Scholar] [CrossRef]
- Ross, J.A.; Kasum, C.M. Dietary Flavonoids: Bioavailability, Metabolic Effects, and Safety. Annu. Rev. Nutr. 2002, 22, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.-K.; Ecklund, D.; Liu, M.; Datta, S.K. Apigenin, a Non-Mutagenic Dietary Flavonoid, Suppresses Lupus by Inhibiting Autoantigen Presentation for Expansion of Autoreactive Th1 and Th17 Cells. Arthritis Res. Ther. 2009, 11, R59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirzoeva, S.; Tong, X.; Bridgeman, B.B.; Plebanek, M.P.; Volpert, O.V. Apigenin Inhibits UVB-Induced Skin Carcinogenesis: The Role of Thrombospondin-1 as an Anti-Inflammatory Factor. Neoplasia 2018, 20, 930–942. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.Y.; Chen, Y.C. A Review of the Dietary Flavonoid, Kaempferol on Human Health and Cancer Chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Liu, H.; Lu, C.; Deng, J.; Yan, Y.; Chen, H.; Wang, Y.; Liang, C.-L.; Wei, J.; Han, L.; et al. Kaempferol Attenuates Imiquimod-Induced Psoriatic Skin Inflammation in a Mouse Model. Clin. Exp. Immunol. 2019, 198, 403–415. [Google Scholar] [CrossRef]
- Matsuda, S.; Minami, A.; Ono, Y.; Kitagishi, Y. Neuroprotection of Genistein in Alzheimer’s Disease. In Diet and Nutrition in Dementia and Cognitive Decline; Elsevier: Amsterdam, The Netherlands, 2015; pp. 1003–1010. [Google Scholar]
- Polkowski, K.; Mazurek, A.P. Biological Properties of Genistein. A Review of in Vitro and in Vivo Data. Acta Pol. Pharm. 2000, 57, 135–155. [Google Scholar]
- Wang, A.; Wei, J.; Lu, C.; Chen, H.; Zhong, X.; Lu, Y.; Li, L.; Huang, H.; Dai, Z.; Han, L. Genistein Suppresses Psoriasis-Related Inflammation through a STAT3NF-B-Dependent Mechanism in Keratinocytes. Int. Immunopharmacol. 2019, 69, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Smolińska, E.; Moskot, M.; Jakóbkiewicz-Banecka, J.; Wegrzyn, G.; Banecki, B.; Szczerkowska-Dobosz, A.; Purzycka-Bohdan, D.; Gabig-Cimińska, M. Molecular Action of Isoflavone Genistein in the Human Epithelial Cell Line HaCaT. PLoS ONE 2018, 13, e0192297. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, H.; Nassiri-Asl, M. Review of the Protective Effects of Rutin on the Metabolic Function as an Important Dietary Flavonoid. J. Endocrinol. Investig. 2014, 37, 783–788. [Google Scholar] [CrossRef]
- Ganeshpurkar, A.; Saluja, A.K. The Pharmacological Potential of Rutin. Saudi Pharm. J. 2017, 25, 149–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhanabal, S.P.; Muruganantham, N.; Basavaraj, K.H.; Wadhwani, A.; Shamasundar, N.M. Antipsoriatic Activity of Extracts and Fractions Obtained from Memecylon Malabaricum Leaves. J. Pharm. Pharmacol. 2012, 64, 1501–1509. [Google Scholar] [CrossRef]
- Lee, C.-H.; Jeong, T.-S.; Choi, Y.-K.; Hyun, B.-H.; Oh, G.-T.; Kim, E.-H.; Kim, J.-R.; Han, J.-I.; Bok, S.-H. Anti-Atherogenic Effect of Citrus Flavonoids, Naringin and Naringenin, Associated with Hepatic ACAT and Aortic VCAM-1 and MCP-1 in High Cholesterol-Fed Rabbits. Biochem. Biophys. Res. Commun. 2001, 284, 681–688. [Google Scholar] [CrossRef]
- Salehi, B.; Fokou, P.; Sharifi-Rad, M.; Zucca, P.; Pezzani, R.; Martins, N.; Sharifi-Rad, J. The Therapeutic Potential of Naringenin: A Review of Clinical Trials. Pharmaceuticals 2019, 12, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verri, W.A.; Vicentini, F.T.M.C.; Baracat, M.M.; Georgetti, S.R.; Cardoso, R.D.R.; Cunha, T.M.; Ferreira, S.H.; Cunha, F.Q.; Fonseca, M.J.V.; Casagrande, R. Flavonoids as Anti-Inflammatory and Analgesic Drugs: Mechanisms of Action and Perspectives in the Development of Pharmaceutical Forms. In Bioactive Natural Products; Elsevier: Amsterdam, The Netherlands, 2012; pp. 297–330. [Google Scholar]
- Igual, M.; García-Martínez, E.; Camacho, M.M.; Martínez-Navarrete, N. Jam Processing and Storage Effects on -Carotene and Flavonoids Content in Grapefruit. J. Funct. Foods 2013, 5, 736–744. [Google Scholar] [CrossRef]
- Bharti, S.; Rani, N.; Krishnamurthy, B.; Arya, D. Preclinical Evidence for the Pharmacological Actions of Naringin: A Review. Planta Med. 2014, 80, 437–451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Su, W.-W.; Wang, S.; Li, P.-B. Naringin Inhibits Chemokine Production in an LPS-Induced RAW 264.7 Macrophage Cell Line. Mol. Med. Rep. 2012, 6, 1343–1350. [Google Scholar] [CrossRef] [Green Version]
- Deenonpoe, R.; Prayong, P.; Thippamom, N.; Meephansan, J.; Na-Bangchang, K. Anti-Inflammatory Effect of Naringin and Sericin Combination on Human Peripheral Blood Mononuclear Cells (HPBMCs) from Patient with Psoriasis. BMC Complement. Altern. Med. 2019, 19, 168. [Google Scholar] [CrossRef]
- Winiarska-Mieczan, A.; Mieczan, T.; Wójcik, G. Importance of Redox Equilibrium in the Pathogenesis of PsoriasisImpact of Antioxidant-Rich Diet. Nutrients 2020, 12, 1841. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, X.; Mei, L.; Wang, H.; Fang, F. Epigallocatechin-3-Gallate (EGCG) Inhibits Imiquimod-Induced Psoriasis-like Inflammation of BALB/c Mice. BMC Complement. Altern. Med. 2016, 16, 334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and Anthocyanins: Colored Pigments as Food, Pharmaceutical Ingredients, and the Potential Health Benefits. Food Nutr. Res. 2017, 61, 1361779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kausar, H.; Jeyabalan, J.; Aqil, F.; Chabba, D.; Sidana, J.; Singh, I.P.; Gupta, R.C. Berry Anthocyanidins Synergistically Suppress Growth and Invasive Potential of Human Non-Small-Cell Lung Cancer Cells. Cancer Lett. 2012, 325, 54–62. [Google Scholar] [CrossRef]
- Chamcheu, J.C.; Pal, H.C.; Siddiqui, I.A.; Adhami, V.M.; Ayehunie, S.; Boylan, B.T.; Noubissi, F.K.; Khan, N.; Syed, D.N.; Elmets, C.A.; et al. Prodifferentiation, Anti-Inflammatory and Antiproliferative Effects of Delphinidin, a Dietary Anthocyanidin, in a Full-Thickness Three-Dimensional Reconstituted Human Skin Model of Psoriasis. Ski. Pharmacol. Physiol. 2015, 28, 177–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clinton, S.K. Lycopene: Chemistry, Biology, and Implications for Human Health and Disease. Nutr. Rev. 2009, 56, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Mangels, A.R.; Holden, J.M.; Beecher, G.R.; Forman, M.R.; Lanza, E. Carotenoid Content of Fruits and Vegetables: An Evaluation of Analytic Data. J. Am. Diet. Assoc. 1993, 93, 284–296. [Google Scholar] [CrossRef]
- Trejo-Solís, C.; Pedraza-Chaverrí, J.; Torres-Ramos, M.; Jiménez-Farfán, D.; Salgado, A.C.; Serrano-García, N.; Osorio-Rico, L.; Sotelo, J. Multiple Molecular and Cellular Mechanisms of Action of Lycopene in Cancer Inhibition. Evid.-Based Complement. Altern. Med. 2013, 2013, 705121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shih, C.-M.; Hsieh, C.-K.; Huang, C.-Y.; Huang, C.-Y.; Wang, K.-H.; Fong, T.-H.; Trang, N.T.T.; Liu, K.-T.; Lee, A.-W. Lycopene Inhibit IMQ-Induced Psoriasis-Like Inflammation by Inhibiting ICAM-1 Production in Mice. Polymers 2020, 12, 1521. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Fu, J.; Yin, X.; Cao, S.; Li, X.; Lin, L.; Ni, J. Emodin: A Review of Its Pharmacology, Toxicity and Pharmacokinetics. Phytother. Res. 2016, 30, 1207–1218. [Google Scholar] [CrossRef]
- Nguyen, U.T.; Nguyen, L.T.H.; Kim, B.-A.; Choi, M.-J.; Yang, I.-J.; Shin, H.-M. Natural Compound Mixture, Containing Emodin, Genipin, Chlorogenic Acid, Cimigenoside, and Ginsenoside Rb1, Ameliorates Psoriasis-Like Skin Lesions by Suppressing Inflammation and Proliferation in Keratinocytes. Evid.-Based Complement. Altern. Med. 2020, 2020, 9416962. [Google Scholar] [CrossRef] [PubMed]
| Phytochemicals | Classification | Natural Occurrence | Application with References |
|---|---|---|---|
| Aloe vera | Vegetal medicinal species | - | [85,86] |
| Artemisia capillaris | Vegetal medicinal species | - | [88,89] |
| St. John’s wort Hypericum perforatum | Vegetal medicinal species | - | [92] |
| Rehmannia glutinosa | Vegetal medicinal species | - | [93,94,95] |
| Salvia miltiorrhiza | Vegetal medicinal species | - | [96,97] |
| Capsaicin | Alkaloid | chili peppers | [98,99] |
| Resveratrol | Polyphenol | red grapes, peanuts, berries | [101,102] |
| Curcumin | Polyphenol | - | [105,106,107,108,109,110,111] |
| Rottlerin | Polyphenol | - | [113,114] |
| Quercetin | Polyphenol, flavonoids | onion, apple, broccoli, citrus fruits, cherries, green tea, coffee, red wine, capers | [118,119] |
| Apigenin | Polyphenol, flavonoids | sweet pepper, parsley, thyme, celery, onions, tea | [121,122] |
| Kaempferol | Polyphenol, flavonoids | tea, broccoli, apples, strawberries, beans | [124] |
| Genistein | Polyphenol, flavonoids | soy beans, fava beans | [127,128] |
| Rutin | Polyphenol, flavonoids | citrus, apples, Betula leaves, buckwheat, black tea, green tea | [131] |
| Naringenin | Polyphenol, flavonoids | grapefruit, lemon, tangerine, orange | [134] |
| Naringin | Polyphenol, flavonoids | citrus, cooked tomato paste, cherries, beans, and oregano | [138] |
| Epigallocatechin-3-gallate (EGCG) | Polyphenol, flavonoids | green tea, black tea | [140] |
| Anthocyanin | Polyphenol, flavonoids | berries, strawberries, currants, grapes, tropical fruits, aubergine skin, red cabbage | [143] |
| Lycopene | Carotenoid | tomatoes, rosehips, watermelons, red grapefruits, papayas, apricots, and pink guavas | [147] |
| Emodin | Anthraquinone | rhubarb, water pepper | [149] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nowak-Perlak, M.; Szpadel, K.; Jabłońska, I.; Pizon, M.; Woźniak, M. Promising Strategies in Plant-Derived Treatments of Psoriasis-Update of In Vitro, In Vivo, and Clinical Trials Studies. Molecules 2022, 27, 591. https://doi.org/10.3390/molecules27030591
Nowak-Perlak M, Szpadel K, Jabłońska I, Pizon M, Woźniak M. Promising Strategies in Plant-Derived Treatments of Psoriasis-Update of In Vitro, In Vivo, and Clinical Trials Studies. Molecules. 2022; 27(3):591. https://doi.org/10.3390/molecules27030591
Chicago/Turabian StyleNowak-Perlak, Martyna, Krzysztof Szpadel, Izabella Jabłońska, Monika Pizon, and Marta Woźniak. 2022. "Promising Strategies in Plant-Derived Treatments of Psoriasis-Update of In Vitro, In Vivo, and Clinical Trials Studies" Molecules 27, no. 3: 591. https://doi.org/10.3390/molecules27030591
APA StyleNowak-Perlak, M., Szpadel, K., Jabłońska, I., Pizon, M., & Woźniak, M. (2022). Promising Strategies in Plant-Derived Treatments of Psoriasis-Update of In Vitro, In Vivo, and Clinical Trials Studies. Molecules, 27(3), 591. https://doi.org/10.3390/molecules27030591







