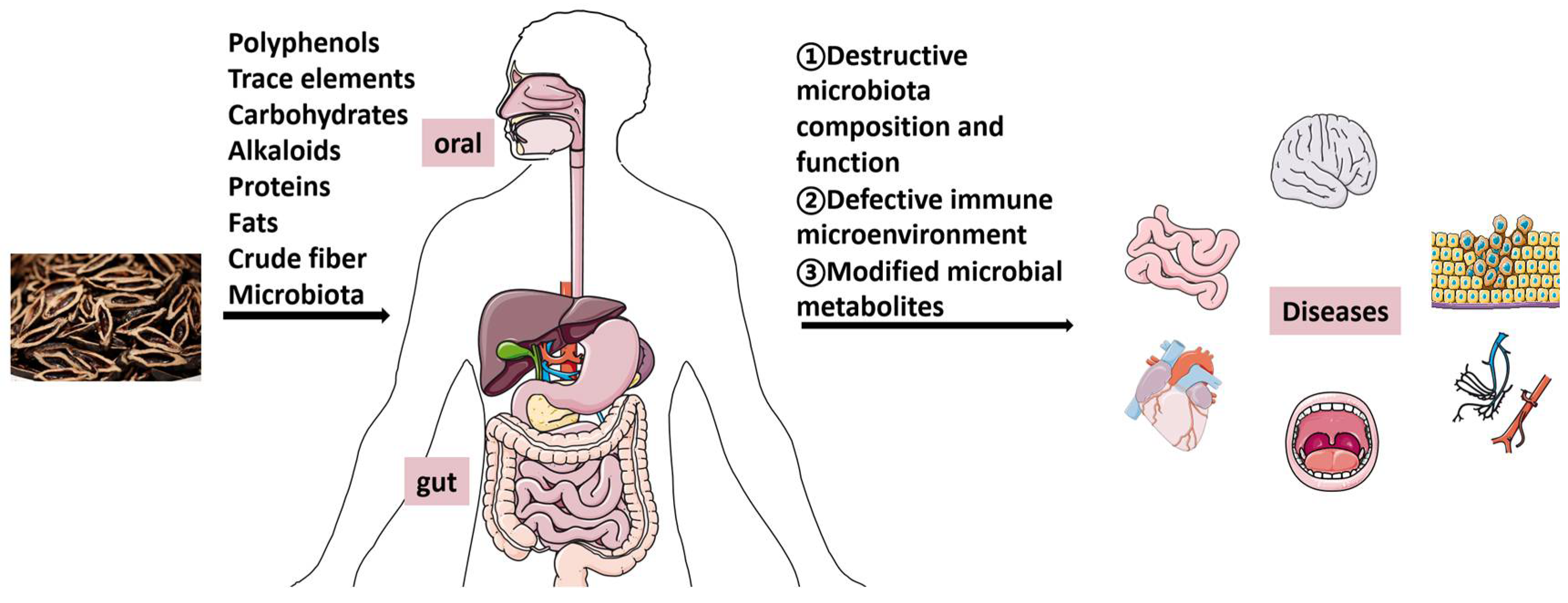
Potential Role of Host Microbiome in Areca Nut-Associated Carcinogenesis and Addiction
Abstract
1. Introduction
2. Microorganisms Contained in Areca Nut Alter Oral and Intestinal Microenvironment
3. Major Constituents in Areca Nut
4. Carcinogens in Areca Nut
4.1. Contradictory Role of Polyphenols in Cancers
4.2. Bidirectional Interaction between Polyphenols and Intestinal Microbes
5. Addiction in Areca Nut
5.1. Areca Alkaloids(Arecoline)
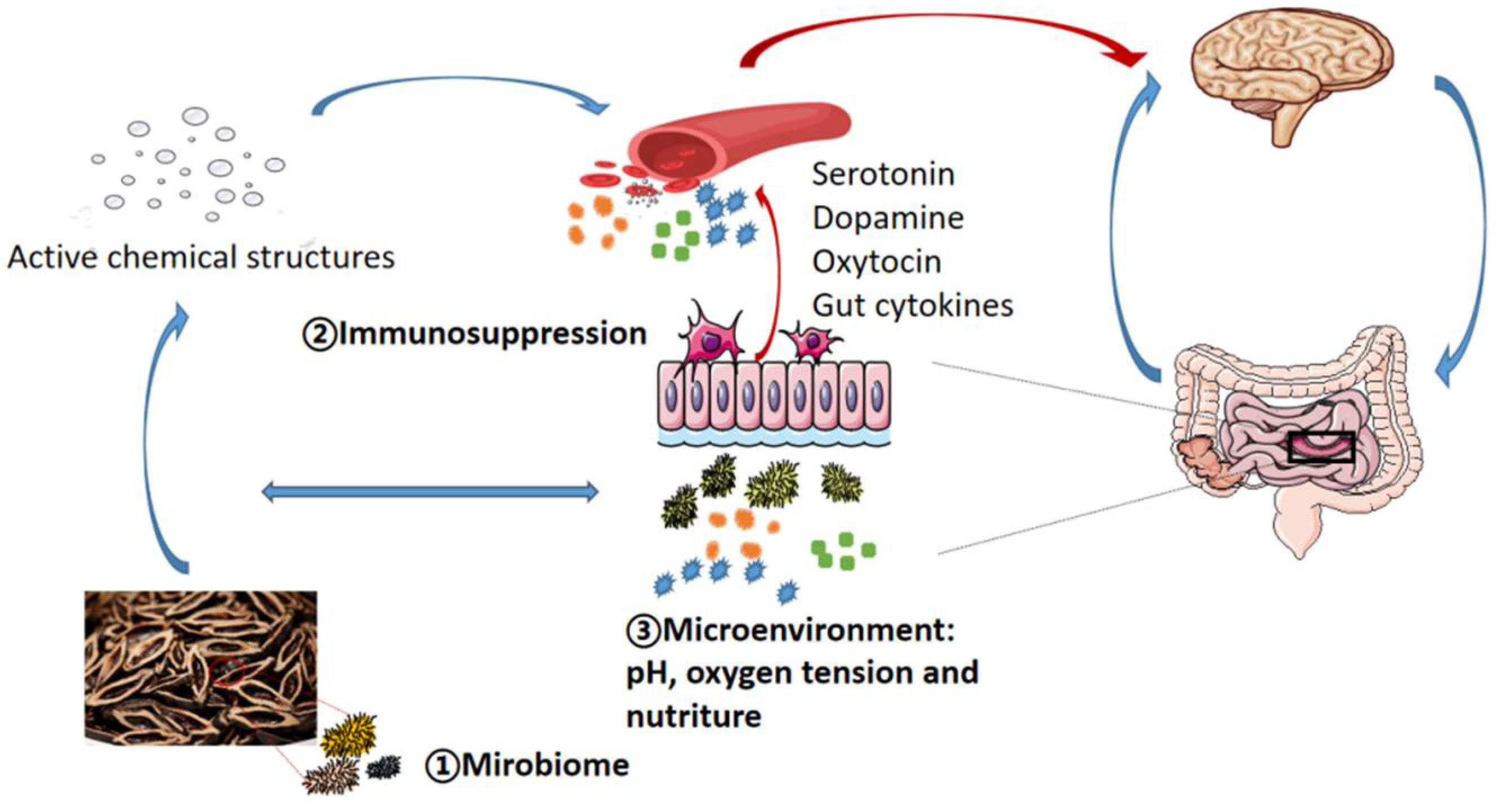
5.2. Microbiota-Gut-Brain Axis: A Potential Regulator of AN Addiction
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Nilesh, C.; Himanshu, A.G. A Qualitative Study of Perceptions and Practices Related to Areca Nut Use Among Adolescents in Mumbai, India. Nicotine Tob. Res. 2021, 23, 1793–1800. [Google Scholar]
- Hongmei, H.; Ting, W. Occurrence of areca alkaloids in wastewater of major Chinese cities. Sci. Total Environ. 2021, 783, 146961. [Google Scholar]
- Nidhi, S.; Rona, P. Perceptions and Practices of General Practitioners towards Oral Cancer and Emerging Risk Factors among Indian Immigrants in Australia: A Qualitative Study. Int. J. Environ. Res. Public Health 2021, 18, 11111. [Google Scholar]
- Strickland, S.S. Anthropological perspectives on use of the areca nut. Addict. Biol. 2002, 7, 85–97. [Google Scholar] [CrossRef]
- Liu, F.L.; Chen, C.L.; Lai, C.C.; Lee, C.C.; Chang, D.M. Arecoline suppresses RANKL-induced osteoclast differentiation in vitro and attenuates LPS-induced bone loss in vivo. Phytomedicine 2020, 69, 153195. [Google Scholar] [CrossRef] [PubMed]
- Benegal, V.; Rajkumar, R.P.; Muralidharan, K. Does areca nut use lead to dependence? Drug Alcohol Depend. 2008, 97, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Pandya, S.; Chaudhary, A.K.; Singh, M.; Singh, M.; Mehrotra, R. Correlation of histopathological diagnosis with habits and clinical findings in oral submucous fibrosis. Head Neck Oncol. 2009, 1, 10. [Google Scholar] [CrossRef] [PubMed]
- Mirza, S.S.; Shafique, K.; Vart, P.; Arain, M.I. Areca nut chewing and dependency syndrome: Is the dependence comparable to smoking? a cross sectional study. Subst. Abuse Treat. Prev. Policy 2011, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Nurmeen, A.; Hamad, A. Evaluation of cytotoxicity of areca nut and its commercial products on normal human gingival fibroblast and oral squamous cell carcinoma cell lines. J. Hazard. Mater. 2021, 403, 123872. [Google Scholar]
- Gupta, S.; Gupta, R.; Sinha, D.N.; Mehrotra, R. Relationship between type of smokeless tobacco & risk of cancer: A systematic review. Indian J. Med. Res. 2018, 148, 56–76. [Google Scholar] [PubMed]
- Shehryar, N.K.; Arif, J.; Raza, T.H. APrevalence of pain in oral cancer: A retrospective study. Oral Dis. 2021, 27, 1806–1812. [Google Scholar]
- Javed, F.; Al-Hezaimi, K.; Warnakulasuriya, S. Areca-nut chewing habit is a significant risk factor for metabolic syndrome: A systematic review. J. Nutr. Health Aging 2012, 16, 445–448. [Google Scholar] [CrossRef]
- Yen, A.M.; Chiu, Y.H.; Chen, L.S.; Wu, H.M.; Huang, C.C.; Boucher, B.J.; Chen, T.H. A population-based study of the association between betel-quid chewing and the metabolic syndrome in men. Am. J. Clin. Nutr. 2006, 83, 1153–1160. [Google Scholar] [CrossRef]
- Hsu, H.F.; Tsou, T.C.; Chao, H.R.; Shy, C.G.; Kuo, Y.T.; Tsai, F.Y.; Yeh, S.C.; Ko, Y.C. Effects of arecoline on adipogenesis, lipolysis, and glucose uptake of adipocytes-A possible role of betel-quid chewing in metabolic syndrome. Toxicol. Appl. Pharmacol. 2010, 245, 370–377. [Google Scholar] [CrossRef]
- Dasgupta, R.; Chatterji, U.; Nag, T.C.; Chaudhuri-Sengupta, S.; Nag, D.; Maiti, B.R. Ultrastructural and hormonal modulations of the thyroid gland following arecoline treatment in albino mice. Mol. Cell. Endocrinol. 2010, 319, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ogunkolade, W.B.; Boucher, B.J.; Bustin, S.A.; Burrin, J.M.; Noonan, K.; Mannan, N.; Hitman, G.A. Vitamin D metabolism in peripheral blood mononuclear cells is influenced by chewing “betel nut” (Areca catechu) and vitamin D status. J. Clin. Endocrinol. Metab. 2006, 91, 2612–2617. [Google Scholar] [CrossRef]
- Yen, C.Y.; Chiang, W.F.; Liu, S.Y.; Cheng, P.C.; Lee, S.Y.; Hong, W.Z.; Lin, P.Y.; Liu, Y.C. Long-term stimulation of areca nut components results in increased chemoresistance through elevated autophagic activity. J. Oral Pathol. Med. 2014, 43, 91–96. [Google Scholar] [CrossRef]
- Hu, S.; Chen, W.C.; Hwang, G.S.; Chen, S.T.; Kuo, S.B.; Chen, Y.; Idova, G.; Wang, S.W. Changes in plasma steroids and cytokines levels in betel chewing patients in Taiwan. Steroids 2016, 111, 134–138. [Google Scholar] [CrossRef]
- Li, K.; Wang, C.; Zhao, Z.; Wu, Z.; Wu, Z.; Tian, X.; Xiao, Y.; Li, Z.; Wang, Y. A comparison for the effects of raw, smoked, and smoked and brined areca nut extracts on the immune and inflammatory responses in the Kunming mice. J. Food Biochem. 2020, 44, e13319. [Google Scholar] [CrossRef]
- Garg, A.; Chaturvedi, P.; Gupta, P.C. A review of the systemic adverse effects of areca nut or betel nut. Indian J. Med. Paediatr. Oncol. 2014, 35, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, M.D.; Chetia, P.; Choudhury, K.D.; Talukdar, A.D.; Datta-Choudhari, M. Atherogenic effect of Arecoline: A computational study. Bioinformation 2012, 8, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Bawany, F.I.; Ahmed, M.U.; Hussain, M.; Khan, A.; Lashari, M.N. Betel nut usage is a major risk factor for coronary artery disease. Glob. J. Health. Sci. 2013, 6, 189–195. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chiang, W.T.; Yang, C.C.; Deng, J.F.; Bullard, M. Cardiac arrhythmia and betel nut chewing--is there a causal effect? Vet. Hum. Toxicol. 1998, 40, 287–289. [Google Scholar] [PubMed]
- Owyang, C.; Wu, G.D. The gut microbiome in health and disease. Gastroenterology 2014, 146, 1433–1436. [Google Scholar] [CrossRef] [PubMed]
- The Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef]
- Hernandez, B.Y.; Zhu, X.; Goodman, M.T.; Gatewood, R.; Mendiola, P.; Quinata, K.; Paulino, Y.C. Betel nut chewing, oral premalignant lesions, and the oral microbiome. PLoS ONE 2017, 12, e0172196. [Google Scholar] [CrossRef]
- Zhong, X.; Lu, Q.; Zhang, Q.; He, Y.; Wei, W.; Wang, Y. Oral microbiota alteration associated with oral cancer and areca chewing. Oral Dis. 2021, 27, 226–239. [Google Scholar] [CrossRef]
- Mei, F.; Meng, K.; Gu, Z.; Yun, Y.; Zhang, W.; Zhang, C.; Zhong, Q.; Pan, F.; Shen, X.; Xia, G.; et al. Arecanut (Areca catechu L.) Seed Polyphenol-Ameliorated Osteoporosis by Altering Gut Microbiome via LYZ and the Immune System in Estrogen-Deficient Rats. J. Agric. Food Chem. 2021, 69, 246–258. [Google Scholar] [CrossRef] [PubMed]
- Chande, M.; Suba, K. Tackling the Use of Supari (Areca Nut) and Smokeless Tobacco Products in the South Asian Community in the United Kingdom. Dent. Update 2016, 43, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, A.; Zubairi, H.; Irfan, S.; Ghias, K. Microbiological safety of areca nut-containing, ready-to-eat chewing substances common among Pakistani paediatric population: A pilot study. J. Pak. Med. Assoc. 2019, 69, 450–454. [Google Scholar]
- Gupta, A.K.; Tulsyan, S.; Thakur, N.; Sharma, V.; Sinha, D.N.; Mehrotra, R. Chemistry, metabolism and pharmacology of carcinogenic alkaloids present in areca nut and factors affecting their concentration. Regul. Toxicol. Pharmacol. 2020, 110, 104548. [Google Scholar] [CrossRef]
- Faouzi, M.; Neupane, R.P.; Yang, J.; Williams, P.; Penner, R. Areca nut extracts mobilize calcium and release pro-inflammatory cytokines from various immune cells. Sci. Rep. 2018, 8, 1075. [Google Scholar] [CrossRef]
- Zhou, M.X.; Tian, X.; Wu, Z.Q.; Li, K.; Li, Z.J. Fuzhuan brick tea supplemented with areca nuts: Effects on serum and gut microbiota in mice. J. Food Biochem. 2021, 45, e13737. [Google Scholar] [CrossRef] [PubMed]
- Anand, R.; Dhingra, C.; Prasad, S.; Menon, I. Betel nut chewing and its deleterious effects on oral cavity. J. Cancer Res. Ther. 2014, 10, 499–505. [Google Scholar]
- Volgin, A.D.; Bashirzade, A.; Amstislavskaya, T.G.; Yakovlev, O.A.; Demin, K.A.; Ho, Y.J.; Wang, D.; Shevyrin, V.A.; Alpyshov, E.T.; Wappler-Guzzetta, E.A.; et al. DARK Classics in Chemical Neuroscience: Arecoline. ACS Chem. Neurosci. 2019, 10, 2176–2185. [Google Scholar] [CrossRef]
- Su, S.; Chien, M.; Lin, C.; Chen, M.; Yang, S. RAGE gene polymorphism and environmental factor in the risk of oral cancer. J. Dent. Res. 2015, 94, 403–411. [Google Scholar] [CrossRef]
- Mehrtash, H.; Duncan, K.; Parascandola, M.; David, A.; Gritz, E.R.; Gupta, P.C.; Mehrotra, R.; Nordin, A.S.A.; Pearlman, P.C.; Warnakulasuriya, S.; et al. Defining a global research and policy agenda for betel quid and areca nut. Lancet Oncol. 2017, 18, e767–e775. [Google Scholar] [CrossRef]
- Tsai, J.F.; Jeng, J.E.; Chuang, L.Y.; Ho, M.S.; Ko, Y.C.; Lin, Z.Y.; Hsieh, M.Y.; Chen, S.C.; Chuang, W.L.; Wang, L.Y.; et al. Habitual betel quid chewing and risk for hepatocellular carcinoma complicating cirrhosis. Medicine 2004, 83, 176–187. [Google Scholar] [CrossRef]
- Cao, J.; Xu, R.; Zhao, X.; Zhong, Z.; Zhang, L.; Zhu, X.; Wu, S.W.; Ai, K. Areca Nut Chewing and an Impaired Estimated Glomerular Filtration Rate as Significant Risk Factors for Non-Muscle-Invasive Bladder Cancer Recurrence. Sci. Rep. 2016, 6, 29466. [Google Scholar] [CrossRef]
- Tseng, Y.H.; Chang, K.W.; Yang, C.C.; Liu, C.J.; Kao, S.Y.; Liu, T.Y.; Lin, S.C. Association between areca-stimulated vimentin expression and the progression of head and neck cancers. Head Neck 2012, 34, 245–253. [Google Scholar] [CrossRef]
- Jeng, J.H.; Hahn, L.J.; Lu, F.J.; Wang, Y.J.; Kuo, M.Y. Eugenol triggers different pathobiological effects on human oral mucosal fibroblasts. J. Dent. Res. 1994, 73, 1050–1055. [Google Scholar] [CrossRef]
- Kumpawat, K.; Deb, S.; Ray, S.; Chatterjee, A. Genotoxic effect of raw betel-nut extract in relation to endogenous glutathione levels and its mechanism of action in mammalian cells. Mutat. Res. 2003, 538, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Yadav, P.; Banerjee, A.; Boruah, N.; Singh, C.S.; Chatterjee, P.; Mukherjee, S.; Dakhar, H.; Nongrum, H.B.; Bhattacharjee, A.; Chatterjee, A. Glutathione S-transferasesP1 AA (105Ile) allele increases oral cancer risk, interacts strongly with c-Jun Kinase and weakly detoxifies areca-nut metabolites. Sci. Rep. 2020, 10, 6032. [Google Scholar] [CrossRef]
- Kiraly, D.D.; Walker, D.M.; Calipari, E.S.; Labonte, B.; Issler, O.; Pena, C.J.; Ribeiro, E.A.; Russo, S.J.; Nestler, E.J. Alterations of the Host Microbiome Affect Behavioral Responses to Cocaine. Sci. Rep. 2016, 6, 35455. [Google Scholar] [CrossRef]
- Nair, U.J.; Friesen, M.; Richard, I.; MacLennan, R.; Thomas, S.; Bartsch, H. Effect of lime composition on the formation of reactive oxygen species from areca nut extract in vitro. Carcinogenesis 1990, 11, 2145–2148. [Google Scholar] [CrossRef]
- Nagabhushan, M.; Bhide, S.V. Anti-mutagenicity of catechin against environmental mutagens. Mutagenesis 1988, 3, 293–296. [Google Scholar] [CrossRef]
- Azuine, M.A.; Bhide, S.V. Protective single/combined treatment with betel leaf and turmeric against methyl (acetoxymethyl) nitrosamine-induced hamster oral carcinogenesis. Int. J. Cancer 1992, 51, 412–415. [Google Scholar] [CrossRef]
- Jurga, B.; Dalia, M.K. The Role of Catechins in Cellular Responses to Oxidative Stress. Molecules 2018, 20, 965. [Google Scholar]
- Huang, P.L.; Chi, C.W.; Liu, T.Y. Areca nut procyanidins ameliorate streptozocin-induced hyperglycemia by regulating gluconeogenesis. Food Chem. Toxicol. 2013, 55, 137–143. [Google Scholar] [CrossRef]
- Daneel, F.; Jannie, P.; Desmond, S.; Larry, A.W. Circular dichroic properties of flavan-3,4-diols. J. Nat. Prod. 2004, 67, 174–178. [Google Scholar]
- Hu, L.; Wu, F.; He, J.; Zhong, L.; Song, Y.; Shao, H. Cytotoxicity of safrole in HepaRG cells: Studies on the role of CYP1A2-mediated ortho-quinone metabolic activation. Xenobiotica 2019, 49, 1504–1515. [Google Scholar] [CrossRef]
- Taleuzzaman, M.; Jain, P.; Verma, R.; Iqbal, Z.; Mirza, M.A. Eugenol as a Potential Drug Candidate: A Review. Curr. Top. Med. Chem. 2021, 21, 1804–1815. [Google Scholar] [CrossRef] [PubMed]
- Rauf, A.; Imran, M.; Butt, M.S.; Nadeem, M.; Peters, D.G.; Mubarak, M.S. Resveratrol as an anti-cancer agent: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 1428–1447. [Google Scholar] [CrossRef] [PubMed]
- Gan, R.Y.; Li, H.B.; Sui, Z.Q.; Corke, H. Absorption, metabolism, anti-cancer effect and molecular targets of epigallocatechin gallate (EGCG): An updated review. Crit. Rev. Food Sci. Nutr. 2018, 58, 924–941. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Arif, Y.; Bajguz, A.; Hayat, S. The role of quercetin in plants. Plant Physiol. Biochem. 2021, 166, 10–19. [Google Scholar] [CrossRef]
- Zhou, Y.X.; Zhang, H.; Peng, C. Puerarin: A review of pharmacological effects. Phytother. Res. 2014, 28, 961–975. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Yoon, Y.; Yoon, H.; Park, H.M.; Song, S.; Yeum, K.J. Dietary Anthocyanins against Obesity and Inflammation. Nutrients 2017, 9, 1089. [Google Scholar] [CrossRef]
- Shim, G.; Ko, S.; Park, J.Y.; Suh, J.H.; Le, Q.V.; Kim, D.; Kim, Y.B.; Im, G.H.; Kim, H.N.; Choe, Y.S.; et al. Tannic acid-functionalized boron nitride nanosheets for theranostics. J. Control Release 2020, 327, 616–626. [Google Scholar] [CrossRef]
- Ozdal, T.; Sela, D.A.; Xiao, J.; Boyacioglu, D.; Chen, F.; Capanoglu, E. The Reciprocal Interactions between Polyphenols and Gut Microbiota and Effects on Bioaccessibility. Nutrients 2016, 8, 78. [Google Scholar] [CrossRef]
- Stevens, J.F.; Maier, C.S. The Chemistry of Gut Microbial Metabolism of Polyphenols. Phytochem. Rev. 2016, 15, 425–444. [Google Scholar] [CrossRef]
- Williamson, G.; Clifford, M.N. Role of the small intestine, colon and microbiota in determining the metabolic fate of polyphenols. Biochem. Pharmacol. 2017, 139, 24–39. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Shan, S.; Zhang, C.; Shi, J.; Li, H.; Li, Z. Inhibitory Effects of Bound Polyphenol from Foxtail Millet Bran on Colitis-Associated Carcinogenesis by the Restoration of Gut Microbiota in a Mice Model. J. Agric. Food Chem. 2020, 68, 3506–3517. [Google Scholar] [CrossRef] [PubMed]
- Jiao, X.; Wang, Y.; Lin, Y.; Lang, Y.; Li, E.; Zhang, X.; Zhang, Q.; Feng, Y.; Meng, X.; Li, B. Blueberry polyphenols extract as a potential prebiotic with anti-obesity effects on C57BL/6 J mice by modulating the gut microbiota. J. Nutr. Biochem. 2019, 64, 88–100. [Google Scholar] [CrossRef] [PubMed]
- Li, X.W.; Chen, H.P.; He, Y.Y.; Chen, W.L.; Chen, J.W.; Gao, L.; Hu, H.Y.; Wang, J. Effects of Rich-Polyphenols Extract of Dendrobium loddigesii on Anti-Diabetic, Anti-Inflammatory, Anti-Oxidant, and Gut Microbiota Modulation in db/db Mice. Molecules 2018, 23, 3245. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, Q.; Ma, W.; Tian, F.; Shen, H.; Zhou, M. A combination of quercetin and resveratrol reduces obesity in high-fat diet-fed rats by modulation of gut microbiota. Food Funct. 2017, 8, 4644–4656. [Google Scholar] [CrossRef]
- Zhao, H.; Cheng, N.; Zhou, W.; Chen, S.; Wang, Q.; Gao, H.; Xue, X.; Wu, L.; Cao, W. Honey Polyphenols Ameliorate DSS-Induced Ulcerative Colitis via Modulating Gut Microbiota in Rats. Mol. Nutr. Food Res. 2019, 63, e1900638. [Google Scholar] [CrossRef]
- Ho, L.; Zhao, D.; Ono, K.; Ruan, K.; Mogno, I.; Tsuji, M.; Carry, E.; Brathwaite, J.; Sims, S.; Frolinger, T.; et al. Heterogeneity in gut microbiota drive polyphenol metabolism that influences alpha-synuclein misfolding and toxicity. J. Nutr. Biochem. 2019, 64, 170–181. [Google Scholar] [CrossRef]
- Song, Z.; Revelo, X.; Shao, W.; Tian, L.; Zeng, K.; Lei, H.; Sun, H.S.; Woo, M.; Winer, D.; Jin, T. Dietary Curcumin Intervention Targets Mouse White Adipose Tissue Inflammation and Brown Adipose Tissue UCP1 Expression. Obesity 2018, 26, 547–558. [Google Scholar] [CrossRef]
- Wang, L.; Zeng, B.; Liu, Z.; Liao, Z.; Zhong, Q.; Gu, L.; Wei, H.; Fang, X. Green Tea Polyphenols Modulate Colonic Microbiota Diversity and Lipid Metabolism in High-Fat Diet Treated HFA Mice. J. Food Sci. 2018, 83, 864–873. [Google Scholar] [CrossRef]
- Wong, S.H.; Yu, J. Gut microbiota in colorectal cancer: Mechanisms of action and clinical applications. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 690–704. [Google Scholar] [CrossRef]
- Wu, X.; Zhang, T.; Chen, X.; Ji, G.; Zhang, F. Microbiota transplantation: Targeting cancer treatment. Cancer Lett. 2019, 452, 144–151. [Google Scholar] [CrossRef]
- Gori, S.; Inno, A.; Belluomini, L.; Bocus, P.; Bisoffi, Z.; Russo, A.; Arcaro, G. Gut microbiota and cancer: How gut microbiota modulates activity, efficacy and toxicity of antitumoral therapy. Crit. Rev. Oncol. Hematol. 2019, 143, 139–147. [Google Scholar] [CrossRef]
- Wei, M.Y.; Shi, S.; Liang, C.; Meng, Q.C.; Hua, J.; Zhang, Y.Y.; Liu, J.; Zhang, B.; Xu, J.; Yu, X.J. The microbiota and microbiome in pancreatic cancer: More influential than expected. Mol. Cancer 2019, 18, 97. [Google Scholar] [CrossRef]
- Guo, S.; Chen, J.; Chen, F.; Zeng, Q.; Liu, W.L.; Zhang, G. Exosomes derived from Fusobacterium nucleatum-infected colorectal cancer cells facilitate tumour metastasis by selectively carrying miR-1246/92b-3p/27a-3p and CXCL. Gut 2020, 71, e1–e3. [Google Scholar] [CrossRef]
- Chen, S.; Su, T.; Zhang, Y.; Lee, A.; He, J.; Ge, Q.; Wang, L.; Si, J.; Zhuo, W.; Wang, L. Fusobacterium nucleatum promotes colorectal cancer metastasis by modulating KRT7-AS/KRT. Gut Microbes 2020, 11, 511–525. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, Y.; Weng, W.; Guo, B.; Cai, G.; Ma, Y.; Cai, S. Fusobacterium nucleatum promotes chemoresistance to 5-fluorouracil by upregulation of BIRC3 expression in colorectal cancer. J. Exp. Clin. Cancer Res. 2019, 38, 14. [Google Scholar] [CrossRef]
- Geller, L.T.; Barzily-Rokni, M.; Danino, T.; Jonas, O.H.; Shental, N.; Nejman, D.; Gavert, N.; Zwang, Y.; Cooper, Z.A.; Shee, K.; et al. Potential role of intratumor bacteria in mediating tumor resistance to the chemotherapeutic drug gemcitabine. Science 2017, 357, 1156–1160. [Google Scholar] [CrossRef]
- Viaud, S.; Saccheri, F.; Mignot, G.; Yamazaki, T.; Daillere, R.; Hannani, D.; Enot, D.P.; Pfirschke, C.; Engblom, C.; Pittet, M.J.; et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science 2013, 342, 971–976. [Google Scholar] [CrossRef]
- Daillere, R.; Vetizou, M.; Waldschmitt, N.; Yamazaki, T.; Isnard, C.; Poirier-Colame, V.; Duong, C.P.M.; Flament, C.; Lepage, P.; Roberti, M.P.; et al. Enterococcus hirae and Barnesiella intestinihominis Facilitate Cyclophosphamide-Induced Therapeutic Immunomodulatory Effects. Immunity 2016, 45, 931–943. [Google Scholar] [CrossRef]
- Wallace, B.D.; Wang, H.; Lane, K.T.; Scott, J.E.; Orans, J.; Koo, J.S.; Venkatesh, M.; Jobin, C.; Yeh, L.A.; Mani, S.; et al. Alleviating cancer drug toxicity by inhibiting a bacterial enzyme. Science 2010, 330, 831–835. [Google Scholar] [CrossRef]
- Bhatt, A.P.; Pellock, S.J.; Biernat, K.A.; Walton, W.G.; Wallace, B.D.; Creekmore, B.C.; Letertre, M.M.; Swann, J.R.; Wilson, I.D.; Roques, J.R.; et al. Targeted inhibition of gut bacterial beta-glucuronidase activity enhances anticancer drug efficacy. Proc. Natl. Acad. Sci. USA 2020, 117, 7374–7381. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, L.; Chen, S.; Guo, S.; Yue, T.; Hou, Q.; Feng, M.; Xu, H.; Liu, Y.; Wang, P.; et al. The administration of Escherichia coli Nissle 1917 ameliorates irinotecan-induced intestinal barrier dysfunction and gut microbial dysbiosis in mice. Life Sci. 2019, 231, 116529. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.H.; Ko, J.L.; Liao, J.M.; Huang, S.S.; Lin, M.Y.; Lee, L.H.; Chang, L.Y.; Ou, C.C. D-methionine alleviates cisplatin-induced mucositis by restoring the gut microbiota structure and improving intestinal inflammation. Ther. Adv. Med. Oncol. 2019, 11, 1758835918821021. [Google Scholar] [CrossRef]
- Perales-Puchalt, A.; Perez-Sanz, J.; Payne, K.K.; Svoronos, N.; Allegrezza, M.J.; Chaurio, R.A.; Anadon, C.; Calmette, J.; Biswas, S.; Mine, J.A.; et al. Frontline Science: Microbiota reconstitution restores intestinal integrity after cisplatin therapy. J. Leukoc. Biol. 2018, 103, 799–805. [Google Scholar] [CrossRef]
- Lee, T.H.; Park, D.; Kim, Y.J.; Lee, I.; Kim, S.; Oh, C.T.; Kim, J.K.; Jo, S.K. Lactobacillus salivarius BP121 prevents cisplatininduced acute kidney injury by inhibition of uremic toxins such as indoxyl sulfate and pcresol sulfate via alleviating dysbiosis. Int. J. Mol. Med. 2020, 45, 1130–1140. [Google Scholar]
- Gluschnaider, U.; Hidas, G.; Cojocaru, G.; Yutkin, V.; Ben-Neriah, Y.; Pikarsky, E. beta-TrCP inhibition reduces prostate cancer cell growth via upregulation of the aryl hydrocarbon receptor. PLoS ONE 2010, 5, e9060. [Google Scholar] [CrossRef]
- Portal-Nunez, S.; Shankavaram, U.T.; Rao, M.; Datrice, N.; Atay, S.; Aparicio, M.; Camphausen, K.A.; Fernández-Salguero, P.M.; Chang, H.; Lin, P.; et al. Aryl hydrocarbon receptor-induced adrenomedullin mediates cigarette smoke carcinogenicity in humans and mice. Cancer Res. 2012, 72, 5790–5800. [Google Scholar] [CrossRef]
- Zhang, S.; Lei, P.; Liu, X.; Li, X.; Walker, K.; Kotha, L.; Rowlands, C.; Safe, S. The aryl hydrocarbon receptor as a target for estrogen receptor-negative breast cancer chemotherapy. Endocr. Relat. Cancer 2009, 16, 835–844. [Google Scholar] [CrossRef]
- Ambolet-Camoit, A.; Bui, L.C.; Pierre, S.; Chevallier, A.; Marchand, A.; Coumoul, X.; Garlatti, M.; Andreau, K.; Barouki, R.; Aggerbeck, M. 2,3,7,8-tetrachlorodibenzo-p-dioxin counteracts the p53 response to a genotoxicant by upregulating expression of the metastasis marker agr2 in the hepatocarcinoma cell line HepG. Toxicol. Sci. 2010, 115, 501–512. [Google Scholar] [CrossRef]
- Flaveny, C.A.; Murray, I.A.; Chiaro, C.R.; Perdew, G.H. Ligand selectivity and gene regulation by the human aryl hydrocarbon receptor in transgenic mice. Mol. Pharmacol. 2009, 75, 1412–1420. [Google Scholar] [CrossRef]
- Zhang, S.; Qin, C.; Safe, S.H. Flavonoids as aryl hydrocarbon receptor agonists/antagonists: Effects of structure and cell context. Environ. Health Perspect. 2003, 111, 1877–1882. [Google Scholar] [CrossRef]
- Xue, Z.; Li, D.; Yu, W.; Zhang, Q.; Hou, X.; He, Y.; Kou, X. Mechanisms and therapeutic prospects of polyphenols as modulators of the aryl hydrocarbon receptor. Food Funct. 2017, 8, 1414–1437. [Google Scholar] [CrossRef]
- Shinde, R.; McGaha, T.L. The Aryl Hydrocarbon Receptor: Connecting Immunity to the Microenvironment. Trends Immunol. 2018, 39, 1005–1020. [Google Scholar] [CrossRef]
- Roman, A.C.; Carvajal-Gonzalez, J.M.; Merino, J.M.; Mulero-Navarro, S.; Fernandez-Salguero, P.M. The aryl hydrocarbon receptor in the crossroad of signalling networks with therapeutic value. Pharmacol. Ther. 2018, 185, 50–63. [Google Scholar] [CrossRef]
- Chen, A.Y.; Chen, Y.C. A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef]
- Polozhentsev, S.D.; Reiza, V.A.; Malinskii, D.M.; Lebedev, M.F. [Clinico-social aspects of arterial hypertension in the population of the Karelian ASSR]. Sov. Zdravookhr. 1989, 1, 50–53. [Google Scholar]
- Murota, K.; Nakamura, Y.; Uehara, M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018, 82, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Schanz, O.; Chijiiwa, R.; Cengiz, S.C.; Majlesain, Y.; Weighardt, H.; Takeyama, H.; Förster, I. Dietary AhR Ligands Regulate AhRR Expression in Intestinal Immune Cells and Intestinal Microbiota Composition. Int. J. Mol. Sci. 2020, 21, 3189. [Google Scholar] [CrossRef]
- Takamura, T.; Harama, D.; Fukumoto, S.; Nakamura, Y.; Shimokawa, N.; Ishimaru, K.; Ikegami, S.; Makino, S.; Kitamura, M.; Nakao, A. Lactobacillus bulgaricus OLL1181 activates the aryl hydrocarbon receptor pathway and inhibits colitis. Immunol. Cell Biol. 2011, 89, 817–822. [Google Scholar] [CrossRef]
- Cervantes-Barragan, L.; Chai, J.N.; Tianero, M.D.; Di Luccia, B.; Ahern, P.P.; Merriman, J.; Cortez, V.S.; Caparon, M.G.; Donia, M.S.; Gilfillan, S.; et al. Lactobacillus reuteri induces gut intraepithelial CD4(+)CD8alphaalpha(+) T cells. Science 2017, 357, 806–810. [Google Scholar] [CrossRef]
- Shah, G.; Chaturvedi, P.; Vaishampayan, S. Arecanut as an emerging etiology of oral cancers in India. Indian J. Med. Paediatr. Oncol. 2012, 33, 71–79. [Google Scholar] [CrossRef]
- Liu, Y.J.; Peng, W.; Hu, M.B.; Xu, M.; Wu, C.J. The pharmacology, toxicology and potential applications of arecoline: A review. Pharm. Biol. 2016, 54, 2753–2760. [Google Scholar] [CrossRef] [PubMed]
- Jain, V.; Garg, A.; Parascandola, M.; Chaturvedi, P.; Khariwala, S.S.; Stepanov, I. Analysis of Alkaloids in Areca Nut-Containing Products by Liquid Chromatography-Tandem Mass Spectrometry. J. Agric. Food Chem. 2017, 65, 1977–1983. [Google Scholar] [CrossRef]
- Chu, N.S. Neurological aspects of areca and betel chewing. Addict. Biol. 2002, 7, 111–114. [Google Scholar] [CrossRef]
- Papke, R.L.; Horenstein, N.A.; Stokes, C. Nicotinic Activity of Arecoline, the Psychoactive Element of “Betel Nuts”, Suggests a Basis for Habitual Use and Anti-Inflammatory Activity. PLoS ONE 2015, 10, e0140907. [Google Scholar] [CrossRef]
- Venkatesh, D.; Puranik, R.S.; Vanaki, S.S.; Puranik, S.R. Study of salivary arecoline in areca nut chewers. J. Oral Maxillofac. Pathol. 2018, 22, 446. [Google Scholar] [CrossRef] [PubMed]
- Kilian, J.; Millard, M.; Ozenil, M.; Krause, D.; Ghaderi, K.; Holzer, W.; Urban, E.; Spreitzer, H.; Wadsak, W.; Hacker, M.; et al. Synthesis, Biological Evaluation, and Docking Studies of Antagonistic Hydroxylated Arecaidine Esters Targeting mAChRs. Molecules 2022, 27, 3173. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, A.; Ray, J.G. Molecular Pathology of Malignant Transformation of Oral Submucous Fibrosis. J. Environ. Pathol. Toxicol. Oncol. 2016, 35, 193–205. [Google Scholar] [CrossRef]
- Ezquer, F.; Quintanilla, M.E.; Moya-Flores, F.; Morales, P.; Munita, J.M.; Olivares, B.; Landskron, G.; Hermoso, M.A.; Ezquer, M.; Herrera-Marschitz, M.; et al. Innate gut microbiota predisposes to high alcohol consumption. Addict. Biol. 2021, 26, e13018. [Google Scholar] [CrossRef]
- Cussotto, S.; Sandhu, K.V.; Dinan, T.G.; Cryan, J.F. The Neuroendocrinology of the Microbiota-Gut-Brain Axis: A Behavioural Perspective. Front. Neuroendocrinol. 2018, 51, 80–101. [Google Scholar] [CrossRef]
- Xu, Y.; Xie, Z.; Wang, H.; Shen, Z.; Guo, Y.; Gao, Y.; Chen, X.; Wu, Q.; Li, X.; Wang, K. Bacterial Diversity of Intestinal Microbiota in Patients with Substance Use Disorders Revealed by 16S rRNA Gene Deep Sequencing. Sci. Rep. 2017, 7, 3628. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, K.S.; Peterson, V.L.; Halfon, O.; Ahern, G.; Fouhy, F.; Stanton, C.; Dinan, T.G.; Cryan, J.F.; Boutrel, B. Gut microbiome correlates with altered striatal dopamine receptor expression in a model of compulsive alcohol seeking. Neuropharmacology 2018, 141, 249–259. [Google Scholar] [CrossRef]
- Zuo, Z.; Fan, H.; Tang, X.D.; Chen, Y.M.; Xun, L.T.; Li, Y.; Song, Z.J.; Zhai, H.Q. Effect of different treatments and alcohol addiction on gut microbiota in minimal hepatic encephalopathy patients. Exp. Ther. Med. 2017, 14, 4887–4895. [Google Scholar] [CrossRef] [PubMed]
- Peterson, V.L.; Jury, N.J.; Cabrera-Rubio, R.; Draper, L.A.; Crispie, F.; Cotter, P.D.; Dinan, T.G.; Holmes, A.; Cryan, J.F. Drunk bugs: Chronic vapour alcohol exposure induces marked changes in the gut microbiome in mice. Behav. Brain Res. 2017, 323, 172–176. [Google Scholar] [CrossRef]
- Volpe, G.E.; Ward, H.; Mwamburi, M.; Dinh, D.; Bhalchandra, S.; Wanke, C.; alKane, A.V. Associations of cocaine use and HIV infection with the intestinal microbiota, microbial translocation, and inflammation. J. Stud. Alcohol Drugs 2014, 75, 347–357. [Google Scholar] [CrossRef]
- Mutlu, E.A.; Gillevet, P.M.; Rangwala, H.; Sikaroodi, M.; Naqvi, A.; Engen, P.A.; Kwasny, M.; Lau, C.K.; Keshavarzian, A. Colonic microbiome is altered in alcoholism. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 302, G966–G978. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Liu, Q.; Guo, L.; Zeng, H.; Ding, C.; Zhang, W. Gut Microbiota and Relevant Metabolites Analysis in Alcohol Dependent Mice. Front. Microbiol. 2018, 9, 1874. [Google Scholar] [CrossRef] [PubMed]
- Scorza, C.; Piccini, C.; Martinez Busi, M.; Abin Carriquiry, J.A.; Zunino, P. Alterations in the Gut Microbiota of Rats Chronically Exposed to Volatilized Cocaine and Its Active Adulterants Caffeine and Phenacetin. Neurotox. Res. 2019, 35, 111–121. [Google Scholar] [CrossRef]
- de Timary, P.; Leclercq, S.; Starkel, P.; Delzenne, N. A dysbiotic subpopulation of alcohol-dependent subjects. Gut. Microbes 2015, 6, 388–391. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, C.; Dong, X.; Hu, T.; Wang, L.; Zhao, W.; Zhu, S.; Li, G.; Hu, Y.; Gao, Q.; et al. Chronic alcohol exposure induced gut microbiota dysbiosis and its correlations with neuropsychic behaviors and brain BDNF/Gabra1 changes in mice. Biofactors 2019, 45, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.W.; Ge, C.; Feng, G.X.; Li, Y.; Luo, D.; Dong, J.L.; Li, H.; Wang, H.; Cui, M.; Fan, S.J. Gut microbiota modulates alcohol withdrawal-induced anxiety in mice. Toxicol. Lett. 2018, 287, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Leclercq, S.; Le Roy, T.; Furgiuele, S.; Coste, V.; Bindels, L.B.; Leyrolle, Q.; Neyrinck, A.M.; Quoilin, C.; Amadieu, C.; Petit, G.; et al. Gut Microbiota-Induced Changes in beta-Hydroxybutyrate Metabolism Are Linked to Altered Sociability and Depression in Alcohol Use Disorder. Cell Rep. 2020, 33, 108238. [Google Scholar] [CrossRef] [PubMed]
- Peterson, V.L.; Richards, J.B.; Meyer, P.J.; Cabrera-Rubio, R.; Tripi, J.A.; King, C.P.; Polesskaya, O.; Baud, A.; Chitre, A.S.; Bastiaanssen, T.F.; et al. Sex-dependent associations between addiction-related behaviors and the microbiome in outbred rats. EBioMedicine 2020, 55, 102769. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Vuong, H.E.; Nusbaum, D.J.; Hsiao, E.Y.; Evans, C.J.; Taylor, A.M.W. The gut microbiota mediates reward and sensory responses associated with regimen-selective morphine dependence. Neuropsychopharmacology 2018, 43, 2606–2614. [Google Scholar] [CrossRef] [PubMed]
- Hoban, A.E.; Moloney, R.D.; Golubeva, A.V.; McVey Neufeld, K.A.; O’Sullivan, O.; Patterson, E.; Stanton, C.; Dinan, T.G.; Clarke, G.; Cryan, J.F. Behavioural and neurochemical consequences of chronic gut microbiota depletion during adulthood in the rat. Neuroscience 2016, 339, 463–477. [Google Scholar] [CrossRef]
- Burokas, A.; Arboleya, S.; Moloney, R.D.; Peterson, V.L.; Murphy, K.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Targeting the Microbiota-Gut-Brain Axis: Prebiotics Have Anxiolytic and Antidepressant-like Effects and Reverse the Impact of Chronic Stress in Mice. Biol. Psychiatry 2017, 82, 472–487. [Google Scholar] [CrossRef]
- Warda, A.K.; Rea, K.; Fitzgerald, P.; Hueston, C.; Gonzalez-Tortuero, E.; Dinan, T.G.; Hill, C. Heat-killed lactobacilli alter both microbiota composition and behaviour. Behav. Brain. Res. 2019, 362, 213–223. [Google Scholar] [CrossRef]
| Formulas | Polyphenols | Activity | Gut Microbiota-Relevant | Reference |
|---|---|---|---|---|
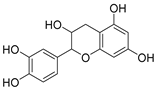 | Catechin | Antimicrobial, antioxidant, anti-cancer and carcinogenic activity | Yes | [48,49] |
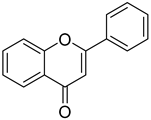 | Flavonoids | Anti-inflammatory, antioxidant and anti-cancer effects, anti-bacteria and anti-virus | Yes | [9,50] |
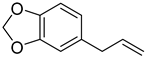 | Safrole | Anti-cancer and antioxidant effects | Yes | [51] |
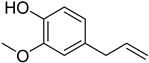 | Eugenol | Anti-bacteria and antihypertensive effect | Yes | [52] |
| Polyphenols | Representative Plant | Activity | Gut Microbiota-Dependent | Reference |
|---|---|---|---|---|
| Resveratrol | Reynoutria japonica Houtt. and Vitis | Antimicrobial, antioxidant, and anti-inflammatory activity | Yes | [53] |
| Quercetin | Fagopyrum esculentum Moench. | Anti-virus, anti-bacterial, anti-cancer, and cardiovascular-protective effect | Yes | [54] |
| Catechin | Green tea | Anti-cancer, anti-virus, anti-fungi, anti-bacterial, and cardiovascular-protective effect | Yes | [55] |
| Puerarin | Pueraria lobata | Anti-oxidant, anti-inflammatory, antihypertensive, and neuroprotective activity | Yes | [56] |
| Anthocyanidin | Elderberries | Anti-oxidant, anti-mutagenic, and anti-proliferative properties | Yes | [57] |
| Tannic acid | Fruit | Anti-oxidant and anti-bacterial effect | Yes | [58] |
| Formulas | Alkaloids | Activity | Reference |
|---|---|---|---|
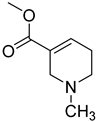 | Arecoline | Effects on nervous, cardiovascular, endocrine and digestive system; anti-parasitic effects; carcinogenic; and genotoxic | [90] |
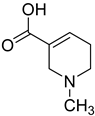 | Arecaidine | Effects on nervous and endocrine system | [107] |
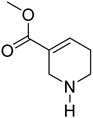 | Guvacoline | Effects on nervous system, anti-inflammatory, and anti-cancer activity | [91] |
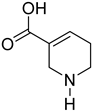 | Guvacine | Effects on nervous system and anti-cancer activity | [108] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, L.; Yuan, F.; Chen, S.; Li, X.; Kong, L.; Zhang, W. Potential Role of Host Microbiome in Areca Nut-Associated Carcinogenesis and Addiction. Molecules 2022, 27, 8171. https://doi.org/10.3390/molecules27238171
Chen L, Yuan F, Chen S, Li X, Kong L, Zhang W. Potential Role of Host Microbiome in Areca Nut-Associated Carcinogenesis and Addiction. Molecules. 2022; 27(23):8171. https://doi.org/10.3390/molecules27238171
Chicago/Turabian StyleChen, Lihui, Fulai Yuan, Sifang Chen, Xiong Li, Lingyu Kong, and Wei Zhang. 2022. "Potential Role of Host Microbiome in Areca Nut-Associated Carcinogenesis and Addiction" Molecules 27, no. 23: 8171. https://doi.org/10.3390/molecules27238171
APA StyleChen, L., Yuan, F., Chen, S., Li, X., Kong, L., & Zhang, W. (2022). Potential Role of Host Microbiome in Areca Nut-Associated Carcinogenesis and Addiction. Molecules, 27(23), 8171. https://doi.org/10.3390/molecules27238171








