The Ultratrace Determination of Fluoroquinolones in River Water Samples by an Online Solid-Phase Extraction Method Using a Molecularly Imprinted Polymer as a Selective Sorbent
Abstract
1. Introduction
2. Materials and Methods
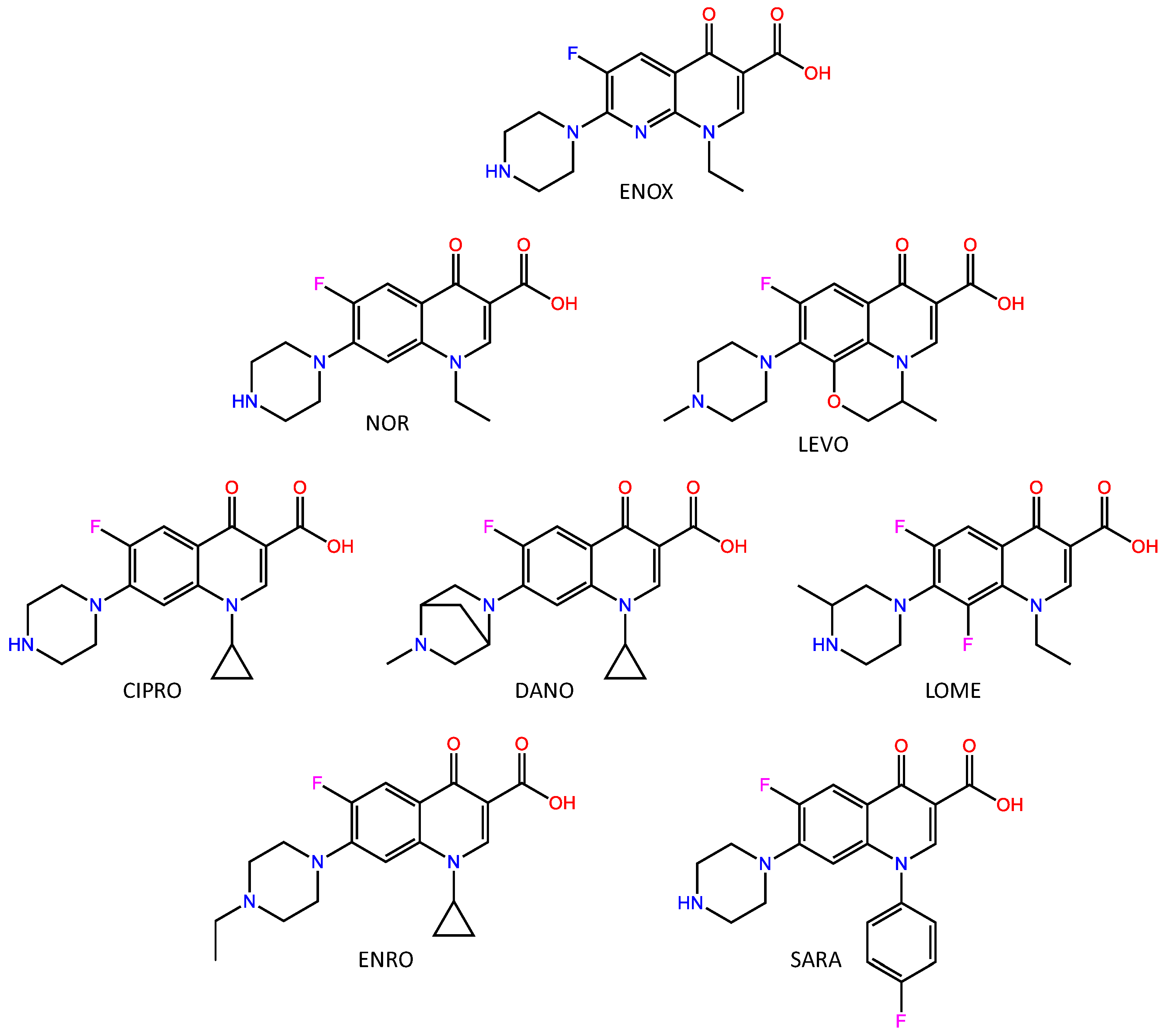
2.1. Chemicals
2.2. Polymer Synthesis
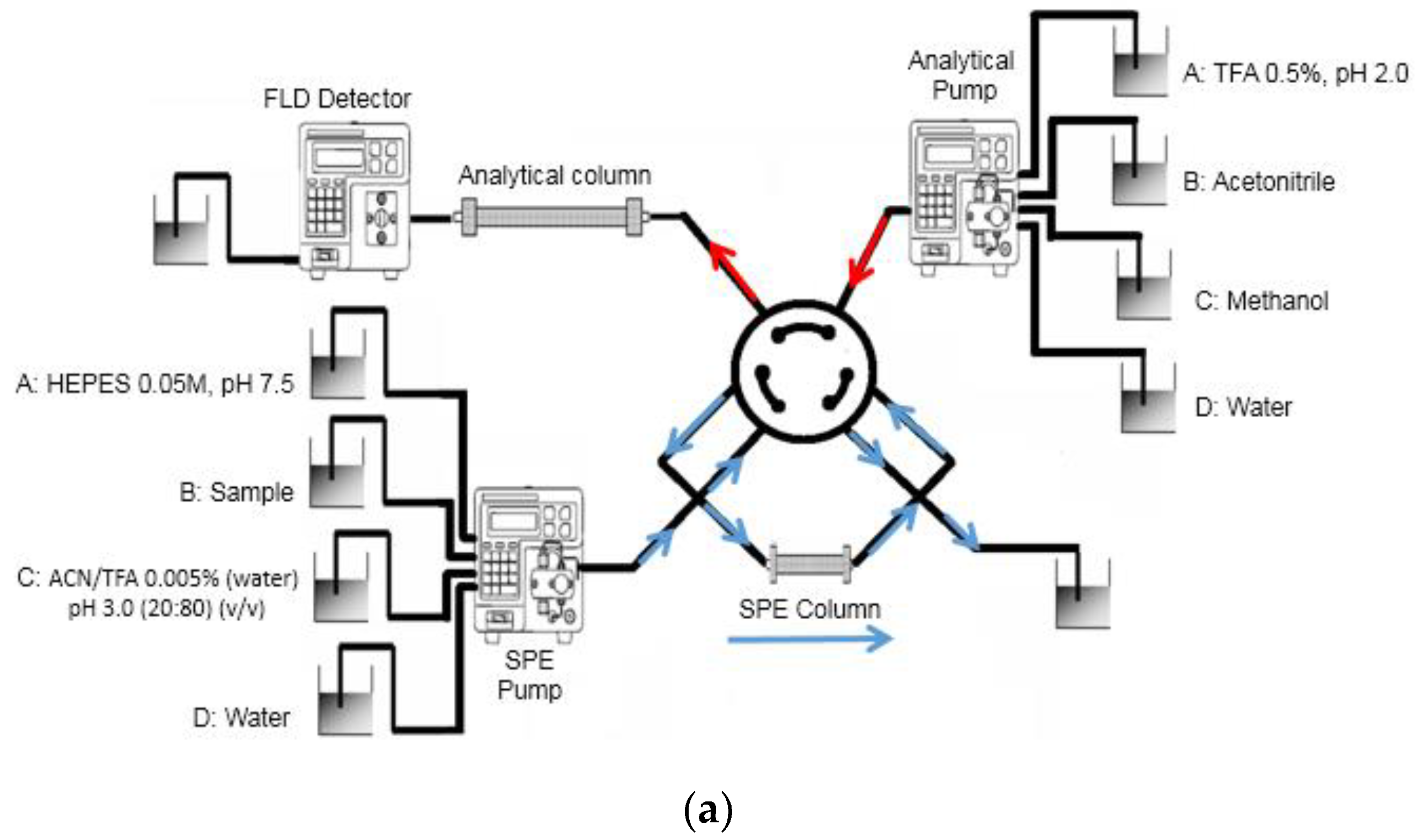
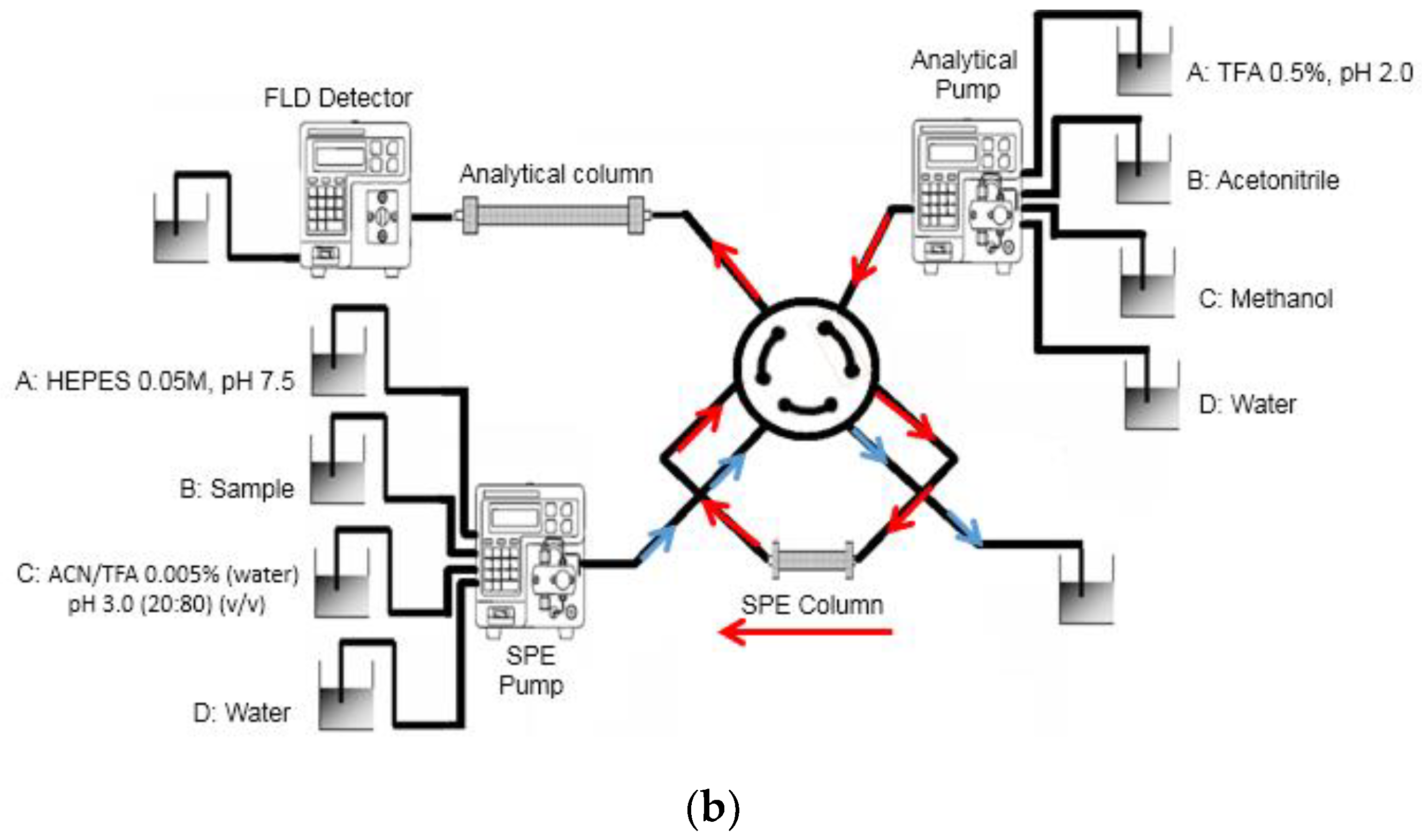
2.3. Apparatus and Analytical Conditions
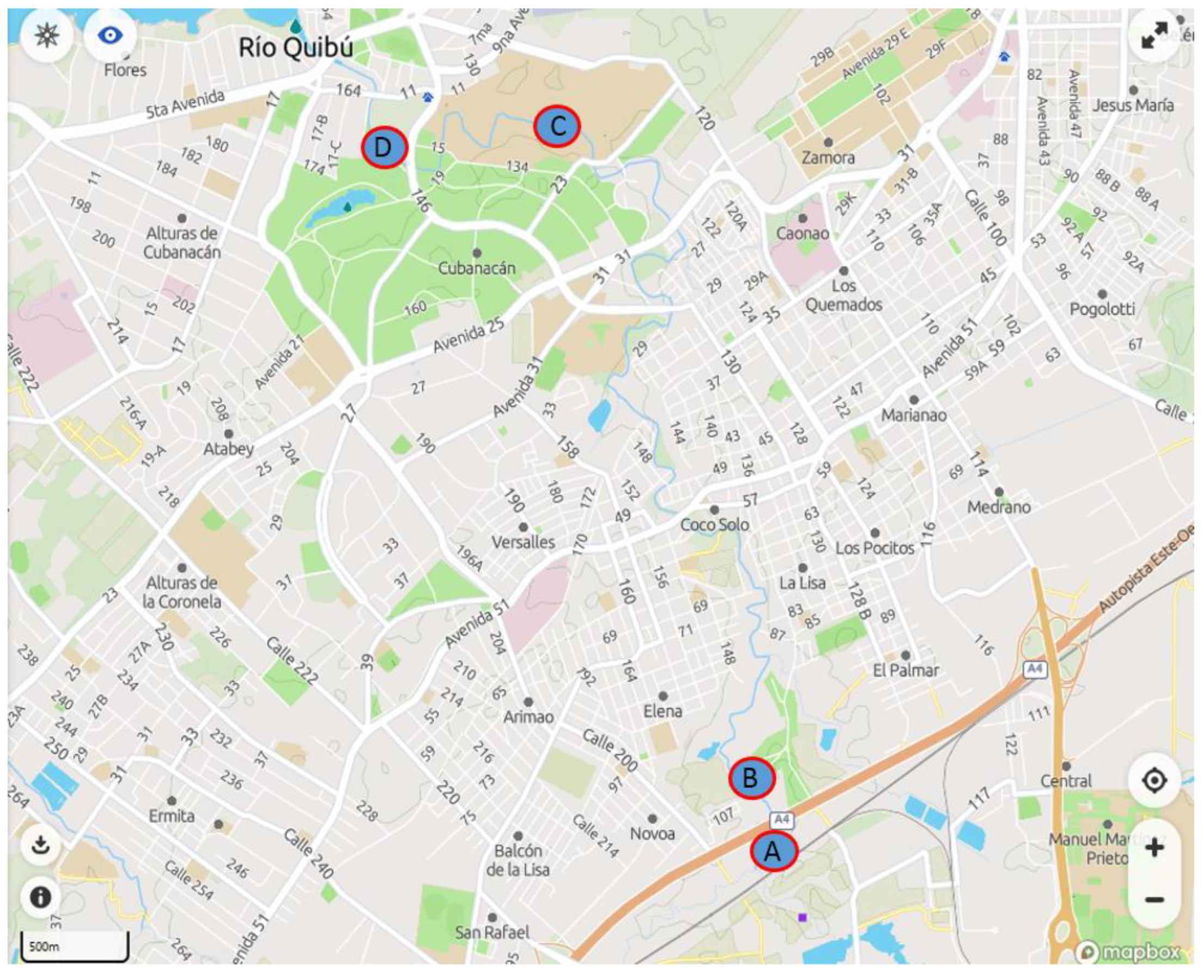
2.4. Sample Collection and Analysis
3. Results and Discussion
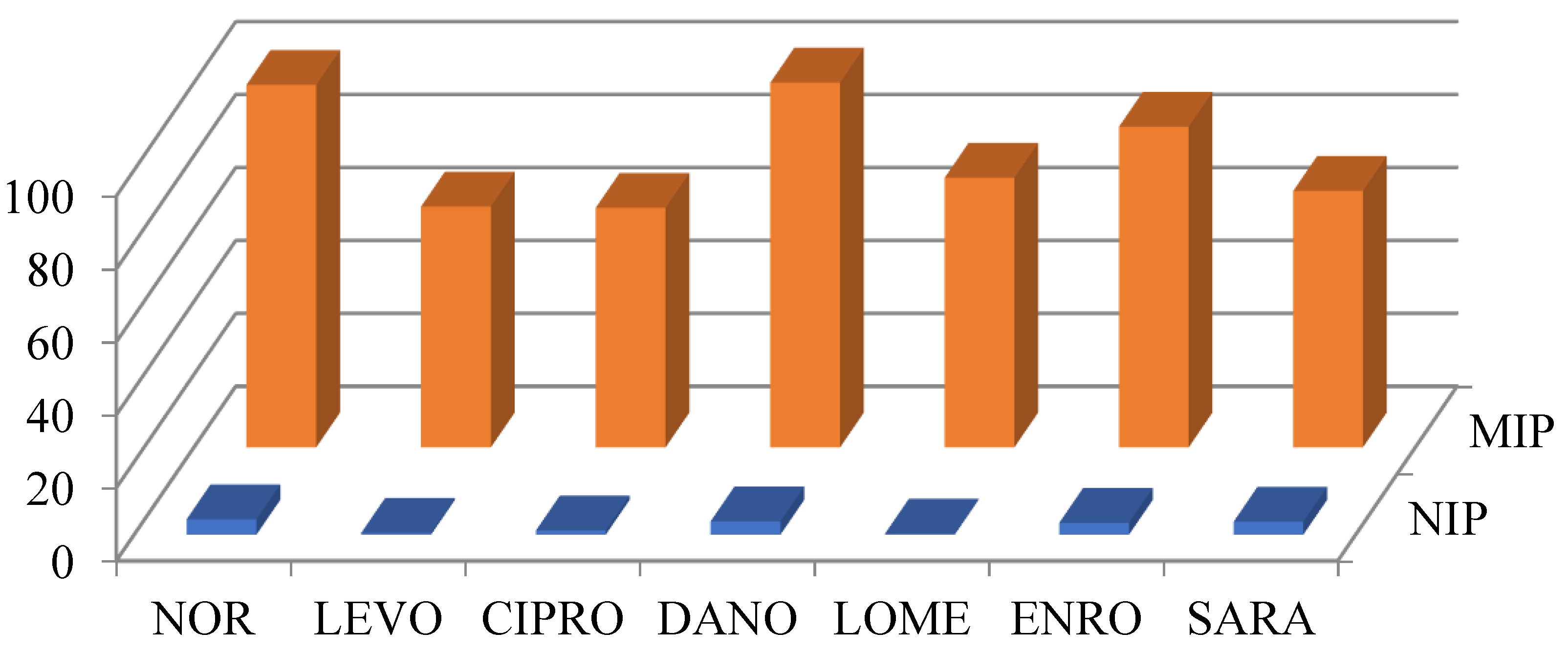
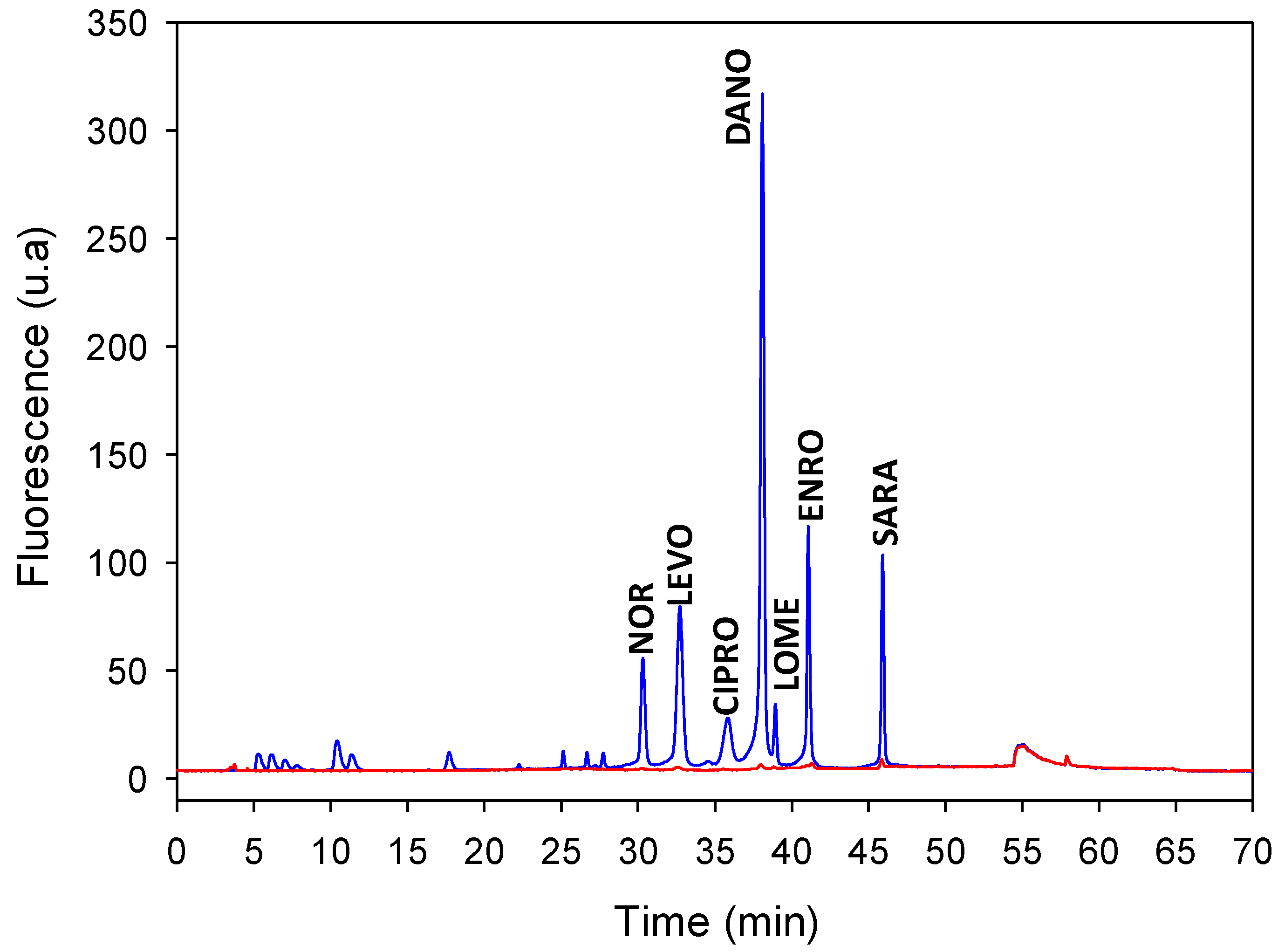
3.1. Online MISPE Optimization
3.2. Method Validation
3.3. Analysis of Quibú River Water
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- World Health Organization. Antibiotic Resistance. Available online: https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance (accessed on 10 October 2022).
- Alanis, A.J. Resistance to antibiotics: Are we in the post-antibiotic era? Med. Res. 2005, 36, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Boxall, A.B.A.; Kolpin, D.W.; Halling-Sørensen, B.; Tolls, J. Are veterinary medicines causing environmental risks? Environ. Sci. Technol. 2003, 37, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Golet, E.M.; Alder, A.C.; Hartmann, A.; Ternes, T.A.; Giger, W. Trace determination of fluoroquinolone antibacterial agents in urban wastewater by solid-phase extraction and liquid chromatography with fluorescence detection. Anal. Chem. 2001, 73, 3632–3638. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; MacNeil, J.D.; Kay, J.F. Chemical Analysis of Antibiotic Residues in Food; Wiley: Hoboken, NJ, USA, 2012. [Google Scholar]
- Hart, C.A.; Kariuki, S. Antimicrobial resistance in developing countries. Br. Med. J. 1998, 317, 647–650. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, R.H.; Wennberg, P.; Johansson, M.I. Screening of human antibiotic substances and determination of weekly mass flows in five sewage treatment plants in Sweden. Environ. Sci. Technol. 2005, 39, 3421–3429. [Google Scholar] [CrossRef]
- Seifrtová, M.; Nováková, L.; Lino, C.; Pena, A.; Solich, P. An overview of analytical methodologies for the determination of antibiotics in environmental waters. Anal. Chim. Acta 2009, 649, 158–179. [Google Scholar] [CrossRef]
- Van Doorslaer, X.; Dewulf, J.; Van Langenhove, H.; Demeestere, K. Fluoroquinolone antibiotics: An emerging class of environmental micropollutants. Sci. Total Environ. 2014, 500–501, 250–269. [Google Scholar] [CrossRef]
- Golet, E.M.; Alder, A.C.; Giger, W. Environmental exposure and risk assessment of fluoroquinolone antibacterial agents in wastewater and river water of the Glatt Valley Watershed, Switzerland. Environ. Sci. Technol. 2002, 36, 3645–3651. [Google Scholar] [CrossRef]
- Mitani, K.; Kataoka, H. Determination of fluoroquinolones in environmental waters by in-tube solid-phase microextraction coupled with liquid chromatography–tandem mass spectrometry. Anal. Chim. Acta 2006, 562, 16–22. [Google Scholar] [CrossRef]
- Graham, D.W.; Olivares-Rieumont, S.; Knapp, C.W.; Lima, L.; Werner, D.; Bowen, E. Antibiotic resistance gene abundances associated with waste discharges to the Almendares River near Havana, Cuba. Environ. Sci. Technol. 2011, 45, 418–424. [Google Scholar] [CrossRef]
- Luaces, M.D.; Urraca, J.L.; Pérez-Conde, M.C.; Martínez Alfonso, N.C.; Valdés-González, A.C.; Gutiérrez, A.M.; Moreno-Bondi, M.C. Chemiluminescence analysis of enrofloxacin in surface water using the tris(1,10-phenantroline)–ruthenium(II)/peroxydisulphate system and extraction with molecularly imprinted polymers. Microchem. J. 2013, 110, 458–464. [Google Scholar] [CrossRef]
- Lombardo-Agüí, M.; Cruces-Blanco, C.; García-Campaña, A.M.; Gámiz-Gracia, L. Multiresidue analysis of quinolones in water by Ultra high performance liquid chromatography tandem mass spectrometry using a simple and effective sample treatment. J. Sep. Sci. 2014, 37, 2145–2152. [Google Scholar] [CrossRef]
- Wagil, M.; Kumirska, J.; Stolte, S.; Puckowski, A.; Maszkowska, J.; Stepnowski, P.; Białk-Bielińska, A. Development of sensitive and reliable LC-MS/MS methods for the determination of three fluoroquinolones in water and fish tissue samples and preliminary environmental risk assessment of their presence in two rivers in northern Poland. Sci. Total Environ. 2014, 493, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Tian, Z.; Qin, W. Quick and sensitive determination of fluoroquinolones by capillary electrophoresis–potential gradient detection. Anal. Lett. 2009, 42, 1057–1069. [Google Scholar] [CrossRef]
- Wang, L.; Yuan, Q.; Liang, G.; Shi, L.; Zhan, Q. Magnetic mixed hemimicelles solid-phase extraction coupled with high-performance liquid chromatography for the extraction and rapid determination of six fluoroquinolones in environmental water samples. J. Sep. Sci. 2015, 38, 996–1001. [Google Scholar] [CrossRef]
- Denadai, M.; Bezerra Cass, Q. Simultaneous determination of fluoroquinolones in environmental water by liquid chromatography–tandem mass spectrometry with direct injection: A green approach. J. Chromatogr. A 2015, 1418, 177–184. [Google Scholar] [CrossRef]
- Mei, M.; Huang, X. Determination of fluoroquinolones in environmental water and milk samples treated with stir cake sorptive extraction based on a boron-rich monolith. J. Sep. Sci. 2016, 39, 1908–1918. [Google Scholar] [CrossRef]
- Speltini, A.; Sturini, M.; Maraschi, F.; Mandelli, E.; Vadivel, D.; Dondi, D.; Profumo, A. Preparation of silica-supported carbon by Kraft lignin pyrolysis, and its use in solid-phase extraction of fluoroquinolones from environmental waters. Microchim. Acta 2016, 183, 2241–2249. [Google Scholar] [CrossRef]
- Rodríguez, E.; Navarro-Villoslada, F.; Benito-Peña, E.; Dolores Marazuela, M.; Cruz Moreno-Bondi, M. Multiresidue determination of ultratrace levels of fluoroquinolone antimicrobials in drinking and aquaculture water samples by automated online molecularly imprinted solid phase extraction and liquid chromatography. Anal. Chem. 2011, 83, 2046–2055. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, X.; Xu, Y.; Du, X.; Sun, X.; Sun, L.; Wang, H.; Zhao, Q.; Yu, A.; Zhang, H.; et al. Determination of fluoroquinolone antibiotics in environmental water samples based on magnetic molecularly imprinted polymer extraction followed by liquid chromatography–tandem mass spectrometry. Anal. Chim. Acta 2010, 662, 31–38. [Google Scholar] [CrossRef]
- Tan, F.; Sun, D.; Gao, J.; Zhao, Q.; Wang, X.; Teng, F.; Quana, X.; Chen, J. Preparation of molecularly imprinted polymer nanoparticles for selective removal of fluoroquinolone antibiotics in aqueous solution. J. Hazard. Mater. 2013, 244–245, 750–757. [Google Scholar] [CrossRef]
- Kou, W.; Zhang, H.; Bibi, A.; Ke, M.; Han, J.; Xiong, J.; Su, R.; Liang, D. Fast quantification of fluoroquinolones in environmental water samples using molecularly imprinted polymers coupled with internal extractive electrospray ionization mass spectrometry. RSC Adv. 2018, 8, 17293–17299. [Google Scholar] [CrossRef] [PubMed]
- Urraca, J.L.; Castellari, M.; Barrios, C.A.; Cruz Moreno-Bondi, M. Multiresidue analysis of fluoroquinolone antimicrobials in chicken meat by molecularly imprinted solid-phase extraction and high performance liquid chromatography. J. Chromatogr. A 2014, 1343, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Maia, A.S.; Paíga, P.; Delerue-Matos, C.; Castro, P.M.L.; Tiritan, M.E. Quantification of fluoroquinolones in wastewaters by liquid chromatography-tandem mass spectrometry. Environ. Pollut. 2020, 259, 113927. [Google Scholar] [CrossRef] [PubMed]
- Parrilla Vázquez, M.M.; Parrilla Vázquez, P.; Martínez Galera, M.; Gil García, M.D. Determination of eight fluoroquinolones in groundwater samples with ultrasound-assisted ionic liquid dispersive liquid–liquid microextraction prior to high-performance liquid chromatography and fluorescence detection. Anal. Chim. Acta 2012, 748, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Dong, X.; Hou, L.; Wang, X.; Liu, L.; Ma, H.; Zhao, R.S. Room-temperature synthesis of flower-shaped covalent organic frameworks for solid-phase extraction of quinolone antibiotics. Anal. Chim. Acta 2020, 1126, 82–90. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, X.; Xu, J.; Shang, Y.; Wang, H.; Wang, P.; He, X.; Tan, J. Determination of quinolones in environmental water and fish by magnetic metal organic frameworks based magnetic solid-phase extraction followed by high-performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2021, 1651, 462286. [Google Scholar] [CrossRef]
- Li, F.; Wang, M.; Zhou, J.; Yang, M.R.; Wang, T.T. Cyclodextrin-derivatized hybrid nanocomposites as novel magnetic solid-phase extraction adsorbent for preconcentration of trace fluoroquinolones from water samples coupled with HPLC-MS/MS determination. Microchem. J. 2021, 164, 105955. [Google Scholar] [CrossRef]
- Jian, N.G.; Liang, S.H.; Cao, J.K.; Di, Q.N.; Kang, K.; Xu, Q. A nanofiber mat prepared from sulfonated polyaniline for solid-phase extraction of fluoroquinolones from water and biological fluids prior to their quantitation by UPLC-MS/MS. Microchim. Acta 2019, 186, 857. [Google Scholar] [CrossRef]
- Lu, D.; Qin, M.; Liu, C.; Deng, J.; Shi, G.; Zhou, T. Ionic liquid-functionalized magnetic metal−organic framework nanocomposites for efficient extraction and sensitive detection of fluoroquinolone antibiotics in environmental water. ACS Appl. Mater. Interfaces 2021, 13, 5357–5367. [Google Scholar] [CrossRef]
- Heidarian, S.M.T.; Sani, A.T.; Danesh, N.M.; Ramezani, M.; Alibolandi, M.; Gerayelou, G.; Abnous, K.; Taghdisi, S.M. A novel electrochemical approach for the ultrasensitive detection of fluoroquinolones based on a double-labelled aptamer to surpass complementary strands of aptamer lying flat. Sens. Actuators B Chem. 2021, 334, 129632. [Google Scholar] [CrossRef]
| Step | Experimental Condition | V (mL) |
|---|---|---|
| 1 | Preconditioning: HEPES 0.05 M. pH 7.5 | 10 |
| 2 | Loading of the sample in HEPES 0.05 M. pH 7.5 | 150 |
| 3 | Washing 1: water | 3 |
| 4 | Washing 2: ACN/TFA 0.005% in water pH 3.0 (20:80) | 3 |
| 5 | Washing 3: water | 3 |
| 6 | Elution: ACN/TFA 0.5% in water. pH 2.0 (20:80) | 10 |
| Parameter | Code | Level | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Minimum | Central | Maximum | |||||||
| Sample flow rate (mL min−1) | FR | 0.5 | 1.5 | 2.5 | |||||
| Sample volume (mL) | V | 20 | 110 | 220 | |||||
| Experiment | V | FR | Recovery (%) 1 | ||||||
| NOR | LEVO | CIPRO | DANO | LOME | ENRO | SARA | |||
| 1 | 20 | 0.5 | 96 | 61 | 82 | 112 | 83 | 99 | 89 |
| 2 | 20 | 2.5 | 96 | 54 | 75 | 116 | 80 | 90 | 94 |
| 3 | 110 | 1.5 | 97 | 47 | 61 | 112 | 90 | 98 | 84 |
| 4 | 220 | 0.5 | 105 | 78 | 77 | 105 | 118 | 92 | 93 |
| 5 | 220 | 2.5 | 98 | 59 | 67 | 101 | 100 | 70 | 78 |
| FQ | Linear Range (ng L−1) | r2 | LOD (ng L−1) | LOQ (ng L−1) | Recovery (%) 1 |
|---|---|---|---|---|---|
| NOR | 3.3–333 | 0.9974 | 0.3 | 0.9 | 99 (2.8) |
| LEVO | 6.6–333 | 0.9977 | 0.3 | 1.0 | 66 (5.3) |
| CIPRO | 6.6–333 | 0.9855 | 0.7 | 2.2 | 66 (8.4) |
| DANO | 0.7–133 | 0.9843 | 0.3 | 0.7 | 101 (2.9) |
| LOME | 6.6–666 | 0.9993 | 0.2 | 0.5 | 74 (3.8) |
| ENRO | 3.3–333 | 0.9967 | 0.3 | 1.1 | 88 (2.8) |
| SARA | 6.6–333 | 0.9995 | 0.1 | 0.4 | 70 (7.8) |
| Analytical Method | Analyte | LOD (ng L−1) | LOQ (ng L−1) | References |
|---|---|---|---|---|
| LC-MS/MS | NOR | 36.3 | 121 | [26] |
| CIPRO | 7.0 | 23.4 | ||
| LOME | 59 | 197 | ||
| ENRO | 55.1 | 184 | ||
| UA-IL-DLLME-LC-FLD | NOR | 0.8 | 3 | [27] |
| CIPRO | 4 | 13 | ||
| DANO | 0.8 | 3 | ||
| LOME | 13 | 43 | ||
| ENRO | 10 | 33 | ||
| SPE-LC-MS/MS | NOR | 3.4 | 10.2 | [15] |
| CIPRO | 3.3 | 10.1 | ||
| ENRO | 3.3 | 10.1 | ||
| SPE-LC-MS/MS | NOR | 0.11 | 0.38 | [28] |
| CIPRO | 0.09 | 0.29 | ||
| ENRO | 0.02 | 0.06 | ||
| MSPE-HPLC-MS/MS | NOR | 1 | - | [29] |
| CIPRO | 3 | - | ||
| ENRO | 3 | - | ||
| SARA | 2 | - | ||
| LOME | 3 | - | ||
| MSPE-HPLC-MS/MS | NOR | 8.5 | 28 | [30] |
| CIPRO | 23 | 78 | ||
| ENRO | 3.9 | 13 | ||
| SARA | 6 | 20 | ||
| LOME | 2.4 | 8.2 | ||
| DANO | 15 | 49 | ||
| SPE-HPLC/MS/MS | NOR | 0.5 | 1.5 | [31] |
| CIPRO | 1 | 3 | ||
| ENRO | 1 | 3 | ||
| LOME | 1 | 3 | ||
| DANO | 0.5 | 1.5 | ||
| MSPE/HPLC-DAD | CIPRO | 20 | - | [32] |
| ENRO | 20 | - | ||
| DANO | 10 | - | ||
| Electrochemical biosensor | CIPRO | 29 | - | [33] |
| Present work | NOR | 0.3 | 0.9 | |
| LEVO | 0.3 | 1.0 | ||
| CIPRO | 0.7 | 2.2 | ||
| DANO | 0.3 | 0.9 | ||
| LOME | 0.2 | 0.5 | ||
| ENRO | 0.3 | 1.1 | ||
| SARA | 0.1 | 0.4 |
| Sampling Point | FQ | Estimated Concentration (ng L−1) |
|---|---|---|
| A | NOR | 21.3 |
| DANO | 3.2 | |
| LOME | 3.5 | |
| B | NOR | 21.7 |
| DANO | 2.2 | |
| LOME | 2.3 | |
| C | Not found | |
| D | Not found |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baeza, A.N.; Urriza-Arsuaga, I.; Navarro-Villoslada, F.; Urraca, J.L. The Ultratrace Determination of Fluoroquinolones in River Water Samples by an Online Solid-Phase Extraction Method Using a Molecularly Imprinted Polymer as a Selective Sorbent. Molecules 2022, 27, 8120. https://doi.org/10.3390/molecules27238120
Baeza AN, Urriza-Arsuaga I, Navarro-Villoslada F, Urraca JL. The Ultratrace Determination of Fluoroquinolones in River Water Samples by an Online Solid-Phase Extraction Method Using a Molecularly Imprinted Polymer as a Selective Sorbent. Molecules. 2022; 27(23):8120. https://doi.org/10.3390/molecules27238120
Chicago/Turabian StyleBaeza, A. N., Idoia Urriza-Arsuaga, F. Navarro-Villoslada, and Javier L. Urraca. 2022. "The Ultratrace Determination of Fluoroquinolones in River Water Samples by an Online Solid-Phase Extraction Method Using a Molecularly Imprinted Polymer as a Selective Sorbent" Molecules 27, no. 23: 8120. https://doi.org/10.3390/molecules27238120
APA StyleBaeza, A. N., Urriza-Arsuaga, I., Navarro-Villoslada, F., & Urraca, J. L. (2022). The Ultratrace Determination of Fluoroquinolones in River Water Samples by an Online Solid-Phase Extraction Method Using a Molecularly Imprinted Polymer as a Selective Sorbent. Molecules, 27(23), 8120. https://doi.org/10.3390/molecules27238120






