4. Materials and Methods
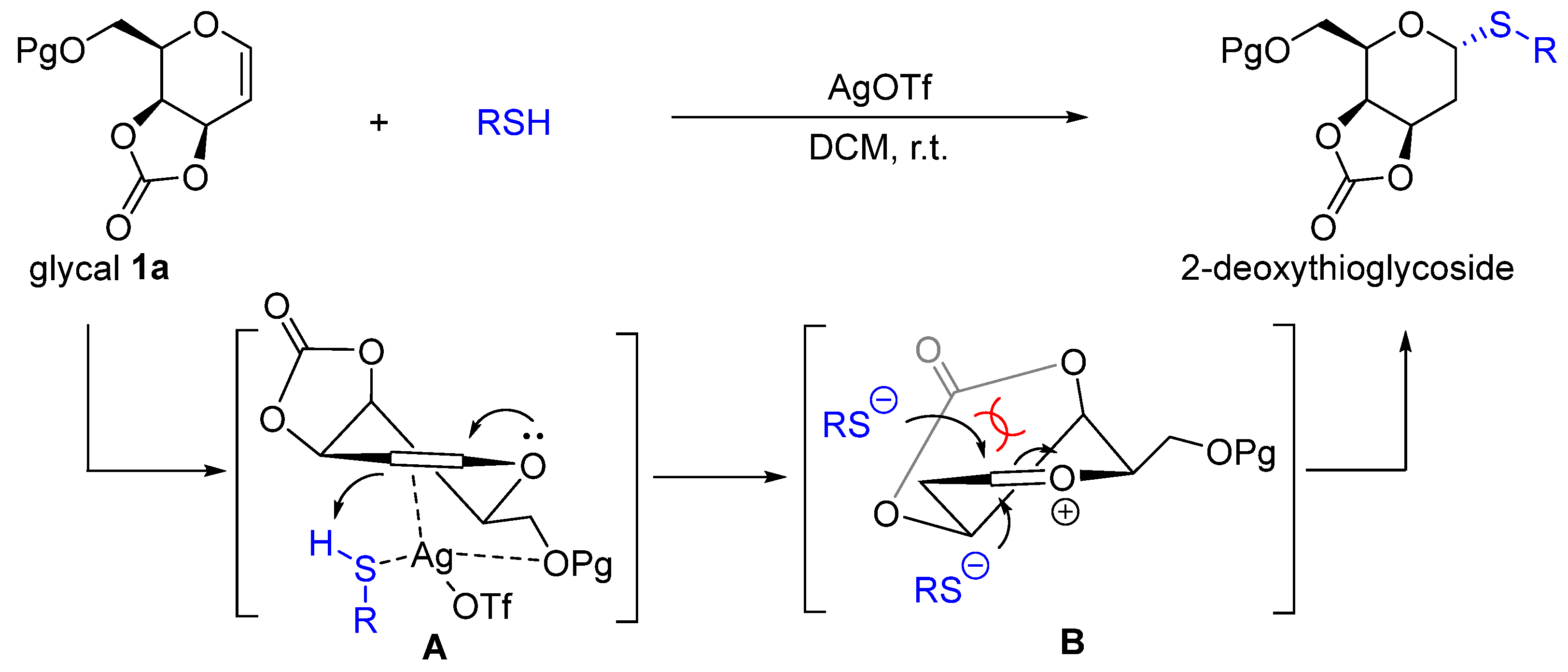
General Procedure. The 3,4-O-carbonate glycal donor (0.100 mmol) and thiol reagent (0.110 mmol) were added to anhydrous dichloromethane (2.00 mL) in a Schlenk tube, followed by adding silver triflate (0.01 mmol) under N2 atmosphere. The reaction mixture was stirred at room temperature and monitored by TLC. Then, aqueous sodium bicarbonate was added to quench the reaction, extracted with dichloromethane, washed by aqueous sodium bicarbonate and dried by sodium sulfate. The organic layer was collected and removed under reduced pressure to afford a crude product which was purified by silica gel flash chromatography with a gradient solvent system (petroleum ether/ethyl acetate as eluent) to yield 2-deoxythioglycosides.
4-Methylphenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside(3a). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 42.7 mg, yield 80%; 1H NMR (400 MHz, CDCl3) δ 7.73–7.63 (m, 4H, Ar-H), 7.47–7.39 (m, 6H, Ar-H), 7.33–7.27 (m, 2H, Ar-H), 7.01–7.03 (m, 2H, Ar-H), 5.45 (dd, J = 9.8, 6.5 Hz, 1H, H-1), 5.05 (ddd, J = 8.8, 4.4, 2.0 Hz, 1H, H-3), 4.90 (dd, J = 8.6, 1.6 Hz, 1H, H-4), 4.16 (ddd, J = 7.7, 6.0, 1.6 Hz, 1H, H-5), 3.84 (dd, J = 10.2, 7.6 Hz, 1H, H-6), 3.77 (dd, J = 10.2, 6.2 Hz, 1H, H-6’), 2.64 (ddd, J = 15.8, 6.5, 3.5 Hz, 1H, H-2), 2.29 (s, 3H, PhCH3), 1.88 (ddd, J = 15.9, 9.8, 3.0 Hz, 1H, H-2’), 1.06 (s, 9H, Si-tBu). 13C NMR (100 MHz, CDCl3) δ 154.2, 138.4, 135.7, 135.6, 133.2, 133.1, 132.8, 130.1, 130.0, 129.9, 129.4, 128.0, 80.8, 73.5, 72.4, 68.5, 61.6, 28.9, 26.9, 21.3, 19.4. HRMS (ESI) m/z: calcd. for C30H34O5SSiNa+ (M + Na)+ 557.1788, found 557.1786; = +45.7 (c = 1.0, CHCl3).
3-Methylphenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside(3b). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 43.8 mg, yield 82%; 1H NMR (400 MHz, CDCl3) δ 7.69 (ddd, J = 8.0, 5.2, 1.6 Hz, 4H, Ar-H), 7.58–7.34 (m, 6H, Ar-H), 7.27–7.20 (m, 2H, Ar-H), 7.19–7.03 (m, 2H, Ar-H), 5.56 (dd, J = 9.8, 6.5 Hz, 1H, H-1), 5.08 (ddd, J = 8.4, 4.0, 2.0 Hz, 1H, H-3), 4.93 (dd, J = 8.6, 1.6 Hz, 1H, H-4), 4.19 (ddd, J = 7.7, 6.0, 1.6 Hz, 1H, H-5), 3.87 (dd, J = 10.2, 7.7 Hz, 1H, H-6), 3.80 (dd, J = 10.2, 6.1 Hz, 1H, H-6’), 2.68 (ddd, J = 15.8, 6.5, 3.5 Hz, 1H, H-2), 2.27 (s, 3H, PhCH3), 1.92 (ddd, J = 15.9, 9.8, 3.0 Hz, 1H, H-2’), 1.08 (s, 9H, Si-tBu). 13C NMR (100 MHz, CDCl3) δ 154.2, 138.9, 135.7, 135.6, 133.2, 133.1, 132.9, 132.8, 130.1, 130.0, 129.3, 128.9, 128.9, 128.0, 80.5, 73.4, 72.4, 68.6, 61.5, 29.0, 26.9, 21.4, 19.4. HRMS (ESI) m/z: calcd. for C30H34O5SSiNa+ (M + Na)+ 557.1788, found 557.1790; = +62.2 (c = 1.0, CHCl3).
2-Methylphenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside(3c). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving white solid 43.2 mg, yield 81%; m. p.: 155.7–157.1 °C. 1H NMR (400 MHz, CDCl3) δ 7.64–7.67 (m, 4H, Ar-H), 7.73–7.45 (m, 7H, Ar-H), 7.20–7.09 (m, 2H, Ar-H), 7.03–7.07 (m, 1H, Ar-H), 5.53 (dd, J = 9.8, 6.4 Hz, 1H, H-1), 5.07 (ddd, J = 8.6, 4.2, 2.0 Hz, 1H, H-3), 4.93 (dd, J = 8.7, 1.6 Hz, 1H, H-4), 4.17 (ddd, J = 8.0, 6.0, 1.7 Hz, 1H, H-5), 3.83 (dd, J = 10.1, 8.0 Hz, 1H, H-6), 3.75 (dd, J = 10.1, 6.0 Hz, 1H, H-6’), 2.67 (ddd, J = 15.8, 6.5, 3.5 Hz, 1H, H-2), 2.33(s, 3H, PhCH3), 1.96 (ddd, J = 15.9, 9.8, 2.9 Hz, 1H, H-2’), 1.04 (s, 9H, Si-tBu). 13C NMR (100 MHz, CDCl3) δ 154.2, 139.4, 135.7, 135.6, 133.2, 132.8, 132.8, 130.3, 130.1, 130.0, 128.0, 128.0, 126.8, 79.8, 73.4, 72.4, 68.6, 61.4, 29.2, 26.9, 20.9, 19.4. HRMS (ESI) m/z: calcd. for C30H34O5SSiNa+ (M + Na)+ 557.1788, found 557.1793; = +43.2 (c = 1.0, CHCl3)
2,4-Methoxylphenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-D-galactopyranoside (3d). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 44.9 mg, yield 82%; 1H NMR (400 MHz, CDCl3) δ 7.77–7.64 (m, 4H, Ar-H), 7.52–7.40 (m, 6H, Ar-H), 7.31–7.33 (m, 1H, Ar-H), 7.03–6.99 (m, 1H, Ar-H), 6.85–6.88 (m, 1H, Ar-H), 5.45 (dd, J = 9.8, 6.4 Hz, 1H, H-1), 5.13–5.03 (m, 1H, H-3), 4.96 (dd, J = 8.6, 1.6 Hz, 1H, H-4), 4.18 (ddd, J = 7.8, 5.8, 1.6 Hz, 1H, H-5), 3.86 (dd, J = 10.1, 8.2 Hz, 1H, H-6), 3.76 (dd, J = 10.0, 5.9 Hz, 1H, H-6’), 2.68 (ddd, J = 15.8, 6.4, 3.5 Hz, 1H, H-2), 2.33 (s, 3H, PhCH3), 2.28 (s, 3H, PhCH3), 1.96 (ddd, J = 15.9, 9.9, 2.9 Hz, 1H, H-2’), 1.08 (s, 9H, Si-tBu). 13C NMR (100 MHz, CDCl3) δ 154.3, 140.0, 138.5, 135.7, 135.6, 134.0, 133.2, 132.8, 131.3, 130.1, 130.0, 128.7, 128.0, 127.5, 80.2, 73.4, 72.4, 68.4, 61.4, 29.1, 26.9, 21.2, 21.0, 19.4. HRMS (ESI) m/z: calcd. for C31H36O5SSiNa+ (M + Na)+ 571.1945, found 571.1941; = +68.4 (c = 1.0, CHCl3).
4-Methoxylphenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranside (3e). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 40.1 mg, yield 73%; 1H NMR (400 MHz, CDCl3) δ 7.69–7.72 (m, 4H, Ar-H), 7.41–7.48 (m, 6H, Ar-H), 7.35–7.38 (m, 2H, Ar-H), 6.74–6.76 (m, 2H, Ar-H), 5.38 (dd, J = 9.8, 6.4 Hz, 1H, H-1), 5.06 (ddd J = 8.6, 4.4, 2.2Hz, 1H, H-3), 4.91 (dd, J = 8.6, 1.6 Hz, 1H, H-4), 4.18 (ddd, J = 7.7, 6.1, 1.6 Hz, 1H, H-5), 3.87 (dd, J = 10.3, 7.5 Hz, 1H, H-6), 3.80 (dd, J = 9.4, 5.3 Hz, 1H, H-6’), 3.77 (s, 3H, PhOCH3), 2.65 (ddd, J = 15.9, 6.5, 3.5 Hz, 1H, H-2), 1.89 (ddd, J = 15.8, 9.8, 3.0 Hz, 1H, H-2’), 1.10 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 160.2, 154.2, 135.7, 135.7, 135.6, 133.2, 132.8, 130.1, 130.0, 128.0, 123.3, 114.7, 81.2, 73.5, 72.5, 68.5, 61.7, 55.4, 28.9, 27.0, 19.4. HRMS (ESI) m/z: calcd. for C30H34O6SSiNa+ (M + Na)+ 573.1738, found 573.1739; = +90.6 (c = 1.0, CHCl3).
4-tert-Butylphenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (3f). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 46.7 mg, yield 81%; 1H NMR (400 MHz, CDCl3) δ 7.69–7.72 (m, 4H, Ar-H), 7.52–7.38 (m, 6H, Ar-H), 7.41–7.34 (m, 2H, Ar-H), 7.30–7.22 (m, 2H, Ar-H), 5.51 (dd, J = 9.7, 6.5 Hz, 1H, H-1), 5.07 (ddd, J = 8.6, 4.2, 2.2 Hz, 1H, H-3), 4.92 (dd, J = 8.7, 1.7 Hz, 1H, H-4), 4.22 (ddd, J = 7.6, 6.2, 1.6 Hz, 1H, H-5), 3.88 (dd, J = 10.2, 7.5 Hz, 1H, H-6), 3.82 (dd, J = 10.2, 6.2 Hz, 1H, H-6’), 2.67 (ddd, J = 15.9, 6.5, 3.5 Hz, 1H, H-2), 1.91 (ddd, J = 15.9, 9.8, 3.0 Hz, 1H, H-2’), 1.29 (s, 9H, C-tBu), 1.09 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 154.2, 151.4, 135.7, 135.6, 133.2, 132.8, 132.6, 130.1, 130.0, 129.6, 128.0, 126.2, 80.7, 73.5, 72.4, 68.5, 61.6, 34.7, 31.3, 29.0, 26.9, 19.4. HRMS (ESI) m/z: calcd. for C33H40O5SSiNa+ (M + Na)+ 599.2258, found 599.2259; = +104.8 (c = 1.0, CHCl3).
Phenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (3g). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 41.6 mg, yield 80%; 1H NMR (400 MHz, CDCl3) δ 7.70 (ddd, J = 8.0, 4.0, 1.7 Hz, 4H, Ar-H), 7.53–7.34 (m, 8H, Ar-H), 7.27–7.21 (m, 3H, Ar-H), 5.57 (dd, J = 9.7, 6.5 Hz, 1H, H-1), 5.08 (ddd, J = 8.4, 4.0, 2.4 Hz, 1H, H-3), 4.92 (dd, J = 8.6, 1.6 Hz, 1H, H-4), 4.20 (ddd, J = 7.6, 6.2, 1.7 Hz, 1H, H-5), 3.87 (dd, J = 10.2, 7.5 Hz, 1H, H-6), 3.81 (dd, J = 10.3, 6.2 Hz, 1H, H-6’), 2.69 (ddd, J = 15.9, 6.5, 3.5 Hz, 1H, H-2), 1.92 (ddd, J = 15.8, 9.8, 3.0 Hz, 1H, H-2’), 1.08 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 154.2, 135.7, 135.6, 133.4, 133.2, 132.8, 132.3, 130.1, 130.0, 129.1, 128.0, 80.5, 73.4, 72.4, 68.6, 61.6, 29.0, 26.9, 19.4. HRMS (ESI) m/z: calcd. for C29H32O5SSiNa+ (M + Na)+ 543.1632, found 543.1642; = +67.9 (c = 1.0, CHCl3).
4-Bromophenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (3h). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 46.0 mg, yield 75%; 1H NMR (400 MHz, CDCl3) δ 7.66–7.70 (m, 4H, Ar-H), 7.51–7.39 (m, 6H, Ar-H), 7.36–7.20 (m, 3H, Ar-H), 7.29 (d, J = 2.4 Hz, 1H, Ar-H), 5.54 (dd, J = 9.8, 6.5 Hz, 1H, H-1), 5.07 (ddd, J = 8.6, 4.4, 2.2 Hz, 1H, H-3), 4.90 (dd, J = 8.6, 1.7 Hz, 1H, H-4), 4.15–4.19 (m, 1H, H-5), 3.87 (dd, J = 10.4, 7.1 Hz, 1H, H-6), 3.79 (dd, J = 10.3, 6.3 Hz, 1H, H-6’), 2.69 (ddd, J = 15.9, 6.5, 3.5 Hz, 1H, H-2), 1.89 (ddd, J = 15.9, 9.9, 3.0 Hz, 1H, H-2’), 1.07 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 154.1, 135.7, 135.6, 133.7, 133.6, 133.1, 132.8, 132.6, 132.3, 132.2, 130.1, 130.0, 128.0, 122.3, 80.5, 73.4, 72.3, 68.8, 61.6, 28.8, 26.9, 19.4. HRMS (ESI) m/z: calcd. for C29H31BrO5SSiNa+ (M + Na)+ 621.0737, found 621.0734; = +45.6 (c = 1.0, CHCl3).
2-Bromophenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (3i). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 43.5 mg, yield 71%; 1H NMR (400 MHz, CDCl3) δ 7.64–7.69 (m, 4H, Ar-H), 7.55 (ddd, J = 7.9, 2.5, 1.5 Hz, 2H, Ar-H), 7.50–7.35 (m, 6H, Ar-H), 7.17–7.20 (m,1H, Ar-H), 7.06–7.11 (m, 1H, Ar-H), 5.71 (dd, J = 9.8, 6.5 Hz, 1H, H-1), 5.12 (ddd, J = 8.6, 4.0, 2.2 Hz, 1H, H-3), 4.95 (dd, J = 8.6, 1.6 Hz, 1H, H-4), 4.21 (ddd, J = 7.8, 6.3, 1.7 Hz, 1H, H-5), 3.85 (dd, J = 10.2, 7.5 Hz, 1H, H-6), 3.79 (dd, J = 10.2, 6.3 Hz, 1H, H-6’), 2.74 (ddd, J = 15.9, 6.6, 3.5 Hz, 1H, H-2), 1.98 (ddd, J = 15.8, 9.8, 2.9 Hz, 1H, H-2’), 1.04 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 154.2, 135.7, 135.6, 135.2, 133.1, 132.8, 132.0, 130.1, 130.0, 128.6, 128.20, 128.0, 125.2, 79.3, 76.8, 72.3, 68.9, 61.3, 28.6, 26.9, 19.3. HRMS (ESI) m/z: calcd. for C29H31BrO5SSiNa+ (M + Na)+ 621.0737, found 621.0735; = +82.4 (c = 1.0, CHCl3).
4-Fluorophenyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (3j). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 39.8 mg, yield 74%; 1H NMR (400 MHz, CDCl3) δ 7.79–7.67 (m, 4H, Ar-H), 7.56–7.40 (m, 8H, Ar-H), 6.90–6.93 (m, 2H, Ar-H), 5.46 (dd, J = 9.9, 6.5 Hz, 1H, H-1), 5.07 (ddd, J = 8.6, 4.2, 2.0 Hz, 1H, H-3), 4.90 (dd, J = 8.7, 1.6 Hz, 1H, H-4), 4.16–4.20 (m, 1H, H-5), 3.87 (dd, J = 10.4, 7.2 Hz, 1H, H-6), 3.80 (dd, J = 10.3, 6.3 Hz, 1H, H-6’), 2.68 (ddd, J = 15.9, 6.5, 3.4 Hz, 1H, H-2), 1.89 (ddd, J = 15.8, 9.8, 2.9 Hz, 1H, H-2’), 1.09 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 163.0 (d, J = 246.7 Hz), 154.1, 135.7, 135.6, 135.1 (d, J = 8.2 Hz), 133.1, 132.8, 130.1, 130.0, 128.0, 116.2 (d, J = 21.6 Hz), 81.0, 73.4, 72.4, 68.7, 61.7, 28.8, 26.9, 19.4. HRMS (ESI) m/z: calcd. for C29H31FO5SSiNa+ (M + Na)+ 561.1538, found 561.1541; = +86.3 (c = 1.0, CHCl3).
Ethyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (5a). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 40.6 mg, yield 86%; 1H NMR (400 MHz, CDCl3) δ 7.67–7.71 (m, 4H, Ar-H), 7.60–7.36 (m, 6H, Ar-H), 5.39 (dd, J = 9.3, 6.5 Hz, 1H, H-1), 5.02 (ddd, J = 8.4, 4.8, 2.4 Hz, 1H, H-3), 4.86 (dd, J = 8.5, 1.7 Hz, 1H, H-4), 4.23–4.01 (m, 1H, H-5), 3.85 (dd, J = 10.3, 7.3 Hz, 1H, H-6), 3.78 (dd, J = 10.3, 6.4 Hz, 1H, H-6’), 2.67–2.75 (m, 1H, H-2), 2.63–2.49 (m, 2H, CH2CH3), 1.79 (ddd, J = 15.8, 9.3, 3.2 Hz, 1H, H-2’), 1.24 (dd, J = 13.4, 6.7 Hz, 3H, CH2CH3), 1.08 (s, 9H, Si-tBu). 13C NMR (100 MHz, CDCl3) δ 154.4, 135.7, 135.6, 133.2, 132.9, 130.1, 130.0, 128.0, 127.9, 76.7, 73.7, 72.5, 68.0, 61.7, 29.2, 26.9, 24.7, 19.3, 15.0. HRMS (ESI) m/z: calcd. For C25H32O5SsiNa+ (M + Na)+ 495.1632, found 495.1634; = +54.2 (c = 1.0, CHCl3).
Octyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (5b). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 43.3 mg, yield 78%; 1H NMR (400 MHz, CDCl3) δ 7.68–7.72 (m, 4H, Ar-H), 7.40–7.50 (m, 6H, Ar-H), 5.35 (dd, J = 9.3, 6.5 Hz, 1H, H-1), 5.02 (ddd, J = 8.4, 4.5, 2.3 Hz, 1H, H-3), 4.87 (dd, J = 8.5, 1.7 Hz, 1H, H-4), 4.13 (ddd, J = 7.4, 6.9, 1.6 Hz, 1H, H-5), 3.85 (dd, J = 10.2, 7.3 Hz, 1H, H-6), 3.78 (dd, J = 10.2, 6.4 Hz, 1H, H-6’), 2.68 (ddd, J = 12.9, 8.1, 6.6 Hz, 1H, H-2), 2.57–2.41 (m, 2H), 1.79 (ddd, J = 15.8, 9.3, 3.1 Hz, 1H, H-2’), 1.59–1.51 (m, 2H, CH2), 1.41–1.12 (m, 10H, CH2), 1.08 (s, 9H, Si-tBu), 0.89 (dd, J = 7.6, 5.7 Hz, 3H, CH3). 13C NMR (100 MHz, CDCl3) δ 154.4, 135.7, 135.6, 133.2, 132.9, 130.1, 130.0, 128.0, 127.9, 77.1, 73.7, 72.5, 67.9, 61.7, 31.9, 30.7, 29.9, 29.3, 29.2, 29.2, 29.0, 26.9, 22.8, 19.3, 14.2. HRMS (ESI) m/z: calcd. for C31H44O5SSiNa+ (M + Na)+ 579.2571, found 579.2556; = +51.4 (c = 1.0, CHCl3).
n-Buty-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (5c). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 41.5 mg, yield 83%; 1H NMR (400 MHz, CDCl3) δ 7.48–7.71 (m, 4H, Ar-H), 7.55–7.36 (m, 6H, Ar-H), 5.35 (dd, J = 9.3, 6.5 Hz, 1H, H-1), 5.02 (ddd, J = 8.5, 4.4, 2.4 Hz, 1H, H-3), 4.87 (dd, J = 8.5, 1.7 Hz, 1H, H-4), 4.13 (ddd, J = 8.3, 6.4, 1.6 Hz, 1H, H-5), 3.85 (dd, J = 10.2, 7.4 Hz, 1H, H-6), 3.77 (dd, J = 10.2, 6.3 Hz, 1H, H-6’), 2.69 (ddd, J = 14.7, 8.1, 6.7 Hz, 1H, H-2), 2.60–2.46 (m, 2H, CH2), 1.78 (ddd, J = 15.7, 9.3, 3.1 Hz, 1H, H-2’), 1.60–1.49 (m, 2H, CH2), 1.41–1.32 (m, 2H, CH2), 1.08 (s, 9H, Si-tBu), 0.87 (dd, J = 14.6, 6.9 Hz, 3H, CH3). 13C NMR (100 MHz, CDCl3) δ 154.4, 135.7, 135.6, 133.2, 132.9, 130.1, 130.0, 128.0, 127.9, 77.1, 73.7, 72.5, 67.9, 61.6, 31.9, 30.4, 29.2, 26.9, 22.0, 19.4, 13.7. HRMS (ESI) m/z: calcd. for C27H36O5SSiNa+ (M + Na)+ 523.1945, found 523.1945; = +36.4 (c = 1.0, CHCl3).
Isobutyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (5d). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 41.5 mg, yield 83%; 1H NMR (400 MHz, CDCl3) δ 7.67–7.71(m, 4H, Ar-H), 7.54–7.34 (m, 6H, Ar-H), 5.31 (dd, J = 9.3, 6.5 Hz, 1H, H-1), 5.02 (ddd, 8.6, 4.6, 2.4, 1H, H-3), 4.88 (dd, J = 8.5, 1.6 Hz, 1H, H-4), 4.19–4.04 (m, 1H, H-5), 3.85 (dd, J = 10.2, 7.5 Hz, 1H, H-6), 3.77 (dd, J = 10.2, 6.3 Hz, 1H, H-6’), 2.79–2.50 (m, 2H, H-2, CHHCH(CH3)2), 2.42 (dd, J = 12.9, 7.2 Hz, 1H, CHHCH(CH3)2), 1.84–1.74 (m, 2H, H-2’, CH2CH(CH3)2), 1.08 (s, 9H, Si-tBu), 0.94 (dd, J = 6.6, 2.7 Hz, 6H, CH3CHCH3). 13C NMR (100 MHz, CDCl3) δ 154.4, 135.7, 135.6, 135.6, 133.2, 133.0, 130.1, 130.0, 128.0, 127.9, 77.7, 73.7, 72.5, 67.9, 61.7, 39.7, 29.4, 28.8, 26.9, 22.1, 22.0, 19.4. HRMS (ESI) m/z: calcd. for C27H36O5SSiNa+ (M + Na)+ 523.1945, found 523.1942; = +63.3 (c = 1.0, CHCl3).
sec-Butyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (5e). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 41.0 mg, yield 82%; 1H NMR (400 MHz, CDCl3) δ 7.67–7.71 (m, 4H, Ar-H), 7.53–7.37 (m, 6H, Ar-H), 5.43 (dd, J = 9.3, 6.5 Hz, 1H, H-1), 5.00–5.05 (m, 1H, H-3), 4.89 (ddd, J = 8.3, 6.1, 1.7 Hz, 1H, H-4), 4.13–4.17 (m, 1H, H-5), 3.92–3.81 (m, 1H, H-6), 3.75 (ddd, J = 10.1, 6.1, 4.0 Hz, 1H, H-6’), 2.86–2.95 (m, 1H, CHCH2CH3), 2.53 (dddd, J = 15.7, 6.2, 3.8, 2.1 Hz, 1H, H-2), 1.81 (dddd, J = 15.8, 9.3, 4.5, 3.2 Hz, 1H, H-2’), 1.57–1.41 (m, 2H, CHCH2CH3), 1.29 (d, J = 6.9 Hz, 2H, CHCH2H), 1.20 (d, J = 7.0 Hz, 1H, CHCH2H), 1.08 (s, 9H, Si-tBu), 0.91 (dt, J = 23.3, 7.4 Hz, 3H, CHCH2CH3). 13C NMR (100 MHz, CDCl3) δ 154.4, 135.7, 135.7, 135.6, 135.6, 133.2, 130.1, 130.0, 128.0, 127.9, 76.6, 73.6, 72.5, 67.8, 61.6, 41.8, 40.0, 30.2, 26.9, 21.3, 19.4, 11.4. 13C NMR (100 MHz, CDCl3) δ.154.4, 135.7, 135.7, 135.6, 135.6, 133.2, 130.1, 130.0, 128.0, 127.9, 75.8, 73.6, 72.5, 67.8, 61.6, 41.2, 39.3, 29.4, 26.9, 21.3, 19.4, 11.3. HRMS (ESI) m/z: calcd. for C27H36O5SSiNa+ (M + Na)+ 523.1945, found 523.1946; = +35.6 (c = 1.0, CHCl3).
tert-Butyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (5f). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 40.0 mg, yield 80%; 1H NMR (400 MHz, CDCl3) δ 7.69–7.72 (m, 4H, Ar-H), 7.56–7.37 (m, 6H, Ar-H), 5.56 (dd, J = 9.4, 6.5 Hz, 1H, H-1), 5.03 (ddd, J = 8.4, 4.8, 2.4 Hz, 1H, H-3), 4.91 (dd, J = 8.5, 1.6 Hz, 1H, H-4), 4.13 (ddd, J = 7.9, 5.7, 1.6 Hz, 1H, H-5), 3.85 (dd, J = 10.0, 8.3 Hz, 1H, H-6), 3.72 (dd, J = 10.0, 5.8 Hz, 1H, H-6’), 2.49 (ddd, J = 15.7, 6.6, 3.8 Hz, 1H, H-2), 1.81 (ddd, J = 15.8, 9.4, 3.1 Hz, 1H, H-2’), 1.31 (s, 9H, C-tBu), 1.07 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 154.6, 135.7, 135.6, 133.2, 132.8, 130.1, 130.0, 128.0, 127.9, 75.4, 73.5, 72.6, 67.6, 61.2, 44.5, 31.8, 29.4, 26.9, 19.4. HRMS (ESI) m/z: calcd. for C27H36O5SSiNa+ (M + Na)+ 523.1945, found 523.1949; = +40.2 (c = 1.0, CHCl3).
Benzyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (5g). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 43.8 mg, yield 82%; 1H NMR (400 MHz, CDCl3) δ 7.70–7.73 (m, 4H, Ar-H), 7.55–7.33 (m, 6H, Ar-H), 7.30–7.10 (m, 5H, Ar-H), 5.21 (dd, J = 9.2, 6.6 Hz, 1H, H-1), 5.00 (ddd, J = 8.4, 4.6, 2,4 Hz, 1H, H-3), 4.86 (dd, J = 8.5, 1.7 Hz, 1H, H-4), 4.14–4.18(m, 1H, H-5), 3.92–3.83 (m, 2H, H-6, PhCHH), 3.76 (dd, J = 10.3, 6.4 Hz, 1H, H-6’), 3.66 (d, J = 13.6 Hz, 1H, PhCHH), 2.46 (ddd, J = 15.7, 6.6, 3.8 Hz, 1H, H-2), 1.77 (ddd, J = 15.8, 9.2, 3.2 Hz, 1H, H-2’), 1.11 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 154.3, 137.9, 135.7, 135.6, 133.2, 132.9, 130.1, 130.0, 129.0, 128.7, 128.0, 127.3, 76.0, 73.6, 72.5, 68.2, 61.7, 34.5, 28.8, 27.0, 19.4. HRMS (ESI) m/z: calcd. for C30H34O5SSiNa+ (M + Na)+ 557.1788, found 557.1777; = +140.3 (c = 1.0, CHCl3).
2-Methoxy-2-oxoethyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranooside (5h). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 41.2 mg, yield 80%; 1H NMR (400 MHz, CDCl3) δ 7.67–7.70 (m, 4H, Ar-H), 7.54–7.37 (m, 6H, Ar-H), 5.50 (dd, J = 9.5, 6.6 Hz, 1H, H-1), 5.05 (ddd, J = 8.6, 4.0, 2.2 Hz, 1H, H-3), 4.88 (dd, J = 8.6, 1.7 Hz, 1H, H-4), 4.14–4.03 (m, 1H, H-5), 3.84 (dd, J = 10.2, 7.2 Hz, 1H, H-6), 3.77 (dd, J = 10.2, 6.4 Hz, 1H, H-6’), 3.67 (s, 3H, CH2COOCH3), 3.52 (d, J = 15.5 Hz, 1H, CHHCOOCH3), 3.17 (d, J = 15.5 Hz, 1H, CHHCOOCH3), 2.61 (ddd, J = 15.8, 6.5, 3.5 Hz, 1H, H-2), 1.76 (ddd, J = 15.8, 9.5, 3.1 Hz, 1H, H-2’), 1.08 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 170.7, 154.2, 135.7, 135.6, 133.1, 132.8, 130.1, 130.0, 128.0, 77.4, 73.5, 72.3, 68.4, 61.5, 52.7, 31.5, 28.6, 26.9, 19.4. HRMS (ESI) m/z: calcd. for C26H32O7SSiNa+ (M + Na)+ 539.1530, found 539.1519; = +28.0 (c = 1.0, CHCl3).
3-S-(6-O-(tert-Butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-a-D-galactopyranosyl)-1,2,3,4-di-O-isopropylidene-β-D-glucofuranoside (5i). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column giving colorless oil 45.9 mg, yield 67%; 1H NMR (400 MHz, CDCl3) δ 7.66 (ddd, J = 7.8, 3.6, 1.7 Hz, 4H, Ar-H), 7.57–7.35 (m, 6H, Ar-H), 5.93 (d, J = 3.6 Hz, 1H, H-1b), 5.05 (dd, J = 9.6, 6.0 Hz, 1H, H-1a), 4.97–5.00 (m, 1H, H-3a), 4.85 (dd, J = 8.3, 1.7 Hz, 1H, H-4a), 4.45 (dd, J = 3.5, 1.8 Hz, 1H, H-2b), 4.30–4.33 (m, 1H, H-5b), 4.04–4.08 (m, 1H, H-5a), 3.92–3.65 (m, 5H, H-6a, 6’a, H-4b, H-7b, 7’b), 3.60 (dd, J = 4.5, 2.0 Hz, 1H, H-3b), 2.41 (ddd, J = 15.7, 5.6, 4.2 Hz, 1H, H-2a), 1.89 (ddd, J = 15.7, 6.4, 3.7 Hz, 1H, H-2’a), 1.57 (s, 3H, CH3), 1.49 (s, 3H, CH3), 1.45 (s, 3H, CH3), 1.31 (s, 3H, CH3), 1.06 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 154.4, 135.7, 135.6, 133.2, 132.9, 130.1, 130.0, 128.0, 112.3, 106.4, 95.21, 83.6, 83.2, 77.9, 73.8, 73.5, 72.1, 67.9, 67.7, 61.7, 47.5, 31.6, 29.7, 27.5, 27.1, 27.0, 26.6, 19.4. HRMS (ESI) m/z: calcd. for C35H47O10SSi+ (M + H)+ 687.2654, found 687.2670; = +27.2 (c = 1.0, CHCl3).
6-S-(6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-a-D-galactopyranosyl)-,2,3,4,6-tetra-O-acetyl-β-D-glucopyranoside (5j). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving white solid 53.0 mg, yield 69%; m.p.: 144.6–146.3 °C. 1H NMR (400 MHz, CDCl3) δ 7.84–7.56 (m, 4H, Ar-H), 7.65–7.35 (m, 6H, Ar-H), 5.56 (dd, J = 9.8, 6.4 Hz, 1H, H-1a), 5.12–5.17(m, 1H, H-3b), 5.09–5.02 (m, 2H, H-3a, H-2b), 5.02–4.97 (m, 2H, H-4a, H-4b), 4.61 (d, J = 10.1 Hz, 1H, H-1b), 4.11 (ddd, J = 8.0, 6.8, 1.5 Hz, 1H, H-5), 4.00 (dd, J = 12.5, 4.8 HZ, 1H, H-6a), 3.90 (dd, J = 12.5, 2.2 Hz, 1H, H-6’a), 3.90–3.79 (m, 2H, H-6, 6’b), 3.60 (ddd, J = 10.1, 4.7, 2.2 Hz, 1H, H-5b), 2.56 (ddd, J = 15.8, 6.4, 3.3 Hz, 1H, H-2a), 2.20–1.97 (m, 9H, OAc), 1.89–1.82 (m, 4H, H-2’a, OAc), 1.07 (s, 9H, Si-tBu). 13C NMR (100 MHz, CDCl3) δ 170.6, 170.3, 169.5, 169.2, 154.1, 135.7, 135.6, 133.1, 132.7, 130.1, 130.0, 128.0, 83.3, 77.6, 76.4, 73.9, 73.1, 72.02, 71.1, 68.2, 67.9, 61.8, 60.7, 29.0, 26.9, 20.8, 20.7, 19.4. HRMS (ESI) m/z: calcd. for C37H46O14SSiNa+ (M + Na)+ 797.2270, found 797.2265; = +21.1 (c = 1.0, CHCl3).
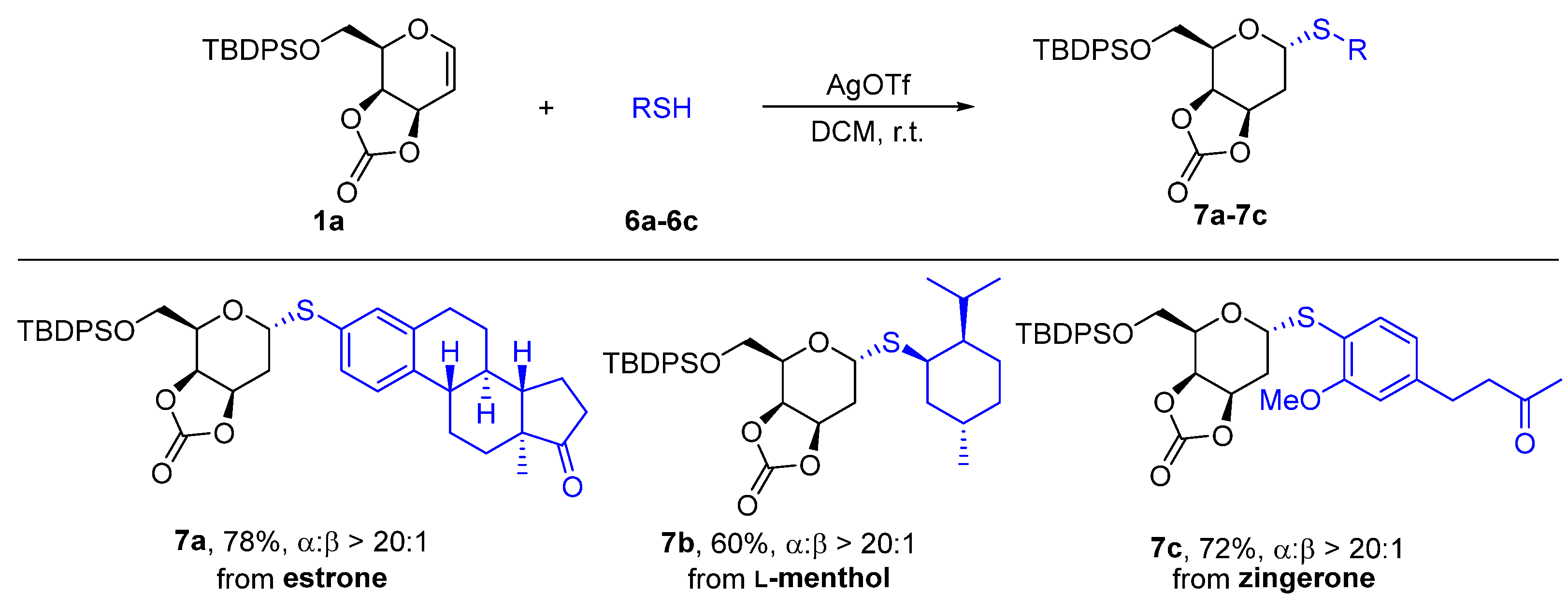
Estronyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (7a). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 54.3 mg, yield 78%; 1H NMR (400 MHz, CDCl3) δ 7.80–7.64 (m, 4H, Ar-H), 7.52–7.36 (m, 6H, Ar-H), 7.26–7.10 (m, 3H, Ar-H), 5.51 (dd, J = 9.8, 6.5 Hz, 1H, H-1), 5.08 (ddd, J = 8.6, 4.1, 2.4 Hz, 1H, H-3), 4.94 (dd, J = 8.6, 1.6 Hz, 1H, H-4), 4.20 (ddd, J = 7.6, 5.9, 1.6 Hz, 1H, H-5), 3.88 (dd, J = 10.2, 7.8 Hz, 1H, H-6), 3.79 (dd, J = 10.2, 6.0 Hz, 1H, H-6’), 2.83–2.74 (m, 2H, PhCH2), 2.68 (ddd, J = 15.9, 6.5, 3.4 Hz, 1H, H-2), 2.52 (dd, J = 18.9, 8.6 Hz, 1H, PhCH), 2.34–2.38 (m, 1H, COCHH), 2.22–2.23 (m, 1H, COCHH), 2.23–2.04 (m, 3H, CH2, CHH), 2.04–1.86 (m, 3H, H-2’, CH2), 1.71–1.53 (m, 3H, CH2, CHH), 1.50–1.37 (m, 2H, CHCH), 1.09 (s, 9H, Si-tBu), 0.91 (s, 3H, CH3). 13C NMR (100 MHz, CDCl3) δ 220.9, 154.2, 140.1, 137.5, 135.7, 135.6, 133.3, 133.2, 132.8, 130.3, 130.1, 130.0, 129.9, 128.0, 126.2, 80.7, 73.4, 72.4, 68.5, 61.5, 50.6, 48.1, 44.4, 38.0, 36.0, 31.7, 29.3, 28.9, 26.9, 26.4, 25.7, 21.7, 19.4, 13.9. HRMS (ESI) m/z: calcd. For C41H48O5SsiNa+ (M + Na)+ 719.2833, found 719.2830; = +114.6 (c = 1.0, CHCl3).
L-Menthyl-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside(7b). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 34.9 mg, yield 60%; 1H NMR (400 MHz, CDCl3) δ 7.83–7.55 (m, 4H, Ar-H), 7.55–7.34 (m, 6H, Ar-H), 5.32 (dd, J = 9.7, 6.4 Hz, 1H, H-1), 5.06 (ddd, J = 8.7, 3.1 Hz, 1H, H-3), 5.01–5.08 (m, 1H, H-4), 4.14 (ddd, J = 9.5, 5.5, 1.5 Hz, 1H, H-5), 3.84–3.88 (m, 1H, H-6), 3.74 (dd, J = 9.6, 5.5 Hz, 1H, H-6’), 3.36–3,37 (m, 1H, SCH), 2.55 (ddd, J = 15.8, 6.4, 3.5 Hz, 1H, H-2), 1.88–1.95 (m, 2H, CH, CH), 1.83 (ddd, J = 15.8, 9.7, 2.8 Hz, 1H, H-2’), 1.77–1.69 (m, 2H, CH2), 1.52–1.43 (m, 1H, CHH), 1.25–1.13 (m, 1H, CHH), 1.08 (s, 9H, Si-tBu), 1.05–0.99 (m, 2H, CH, CHH), 0.88–0.92 (m, 4H, CH3, CHH), 0.83 (d, J = 6.6 Hz, 3H, CH3), 0.67 (d, J = 6.5 Hz, 3H, CH3).13C NMR (100 MHz, CDCl3) δ 154.5, 135.6, 135.5, 133.2, 132.8, 130.1, 130.0, 128.0, 127.9, 75.4, 73.5, 72.6, 67.5, 61.0, 48.3, 44.5, 40.3, 35.4, 29.9, 29.5, 26.9, 26.6, 26.4, 22.2, 21.0, 20.4, 19.4. HRMS (ESI) m/z: calcd. for C33H46O5SSiNa+ (M + Na)+ 605.2727, found 605.2728; = +56.7 (c = 1.0, CHCl3).
Zingerone-1-thio-6-O-(tert-butyldiphenylsilyl)-3,4-O-carbonate-2-deoxy-α-D-galactopyranoside (7c). The title compound was prepared according to the general procedure of 2-deoxythioglycosides synthesis and purified by flash column chromatography giving colorless oil 41.2 mg, yield 72%; 1H NMR (400 MHz, CDCl3) δ 7.81–7.63 (m, 4H), 7.52–7.34 (m, 6H, Ar-H), 7.33–7.14 (m, 1H, Ar-H), 6.69 (d, J = 1.6 Hz, 1H, Ar-H), 6.60 (dd, J = 7.8, 1.7 Hz, 1H, Ar-H), 5.61 (dd, J = 9.6, 6.5 Hz, 1H, H-1), 5.08–5.10 (m, 1H, H-3), 4.95 (dd, J = 8.7, 1.6 Hz, 1H, H-4), 4.22 (ddd, J = 7.9, 5.9, 1.6 Hz, 1H, H-5), 3.94–3.78 (m, 4H, H-6, OMe), 3.70 (dd, J = 10.0, 6.0 Hz, 1H, H-6’), 2.82–2.86 (m, 2H, CH2CH2), 2.77–2.63 (m, 3H, H-2, CH2CH2), 2.14 (s, 3H, CH2COCH3), 1.92 (ddd, J = 15.8, 9.6, 3.0 Hz, 1H, H-2’), 1.07 (s, 9H, Si-tBu).13C NMR (100 MHz, CDCl3) δ 207.7, 158.4, 154.3, 143.3, 135.7, 135.6, 134.5, 133.2, 132.8, 130.0, 129.9, 128.0, 121.0, 118.1, 111.3, 78.6, 73.5, 72.5, 68.23, 61.4, 55.9, 45.0, 30.2, 29.8, 28.9, 26.9, 19.3. HRMS (ESI) m/z: calcd. for C34H40O7SSiNa+ (M + Na)+ 643.2165, found 643.2154; = +99.8 (c = 1.0, CHCl3).