CO2-Responsive Wormlike Micelles Based on Pseudo-Tetrameric Surfactant
Abstract
1. Introduction
2. Results and Discussion
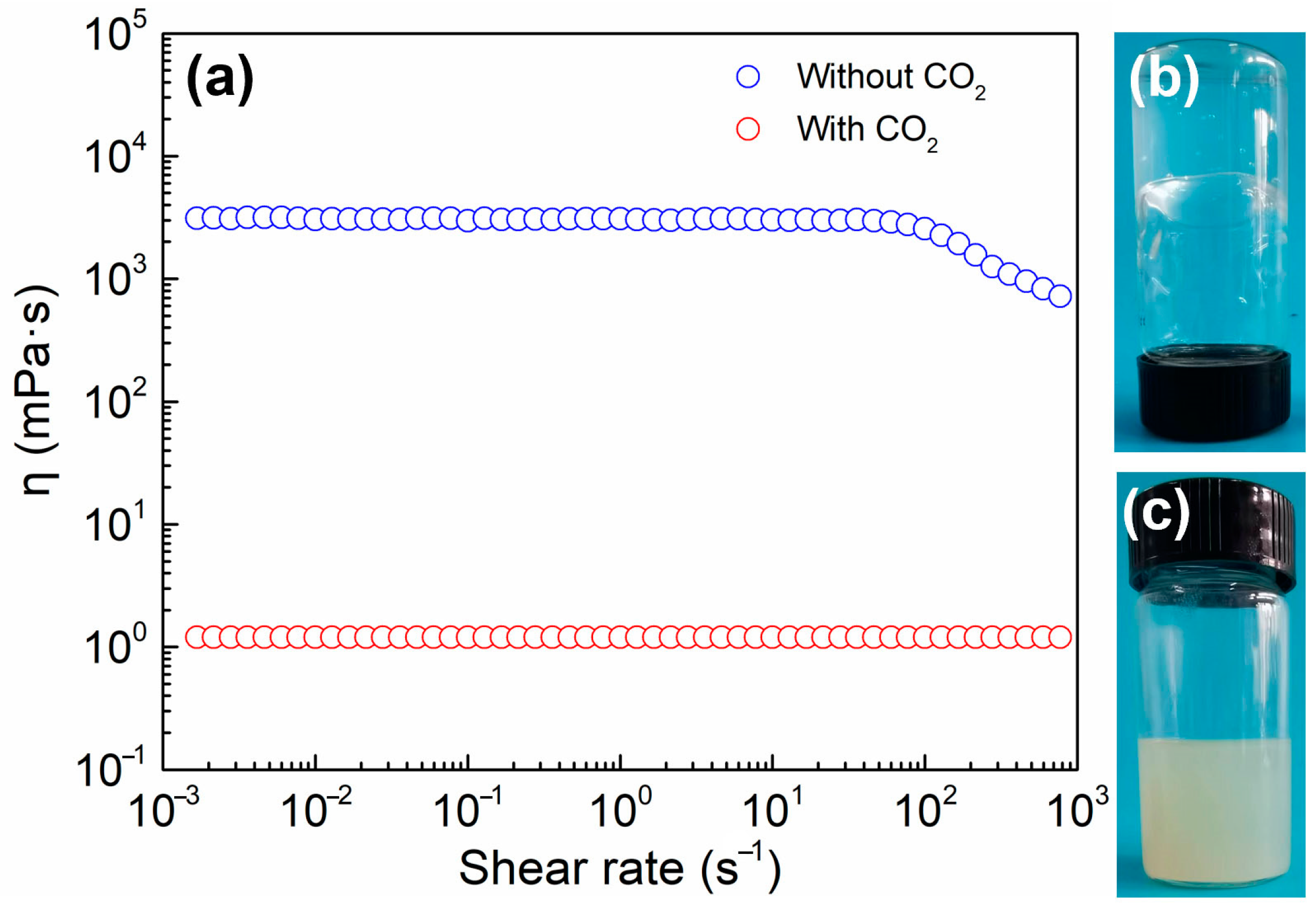
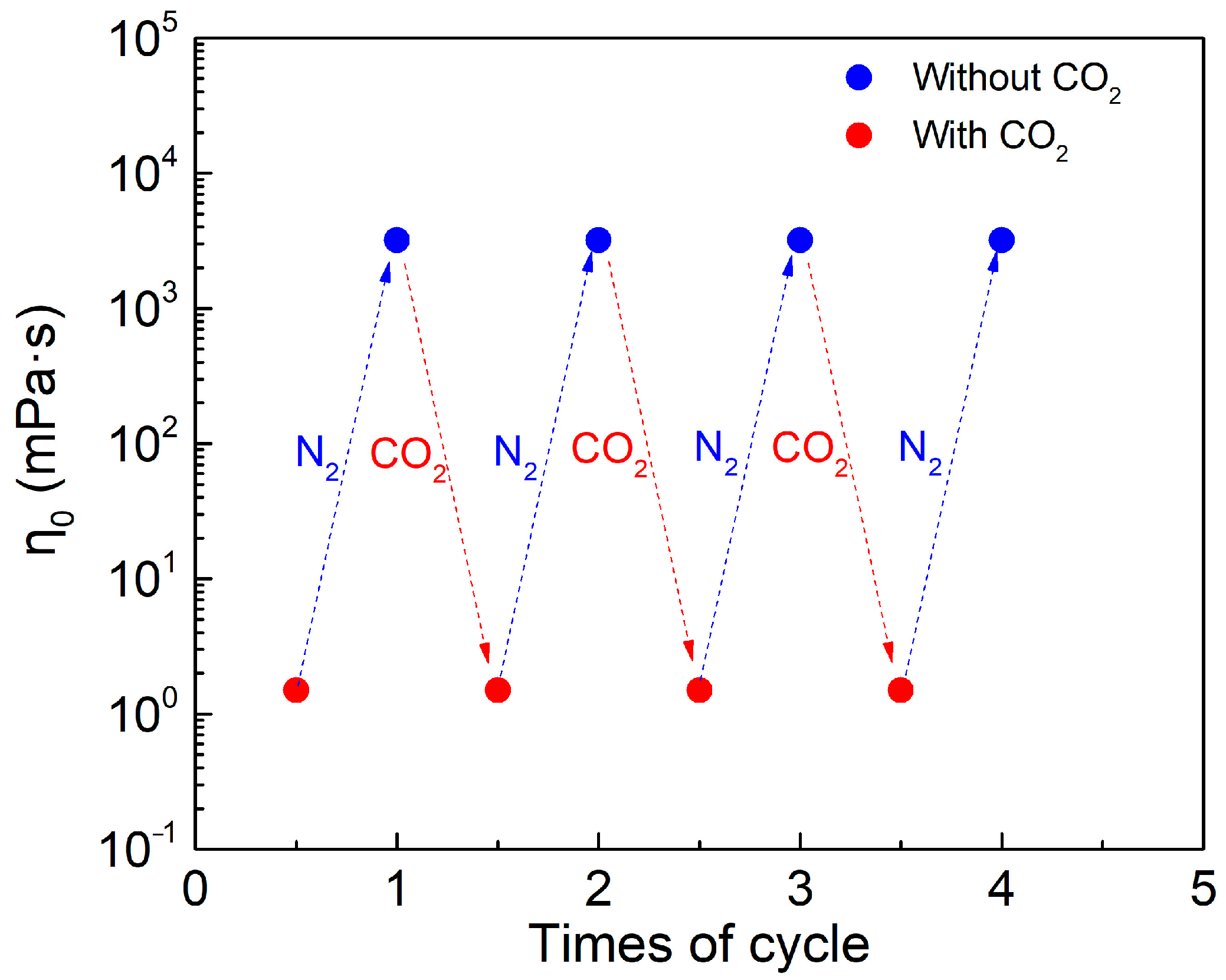
2.1. CO2 Switchable Behavior
2.2. Mechanism of CO2 Switching
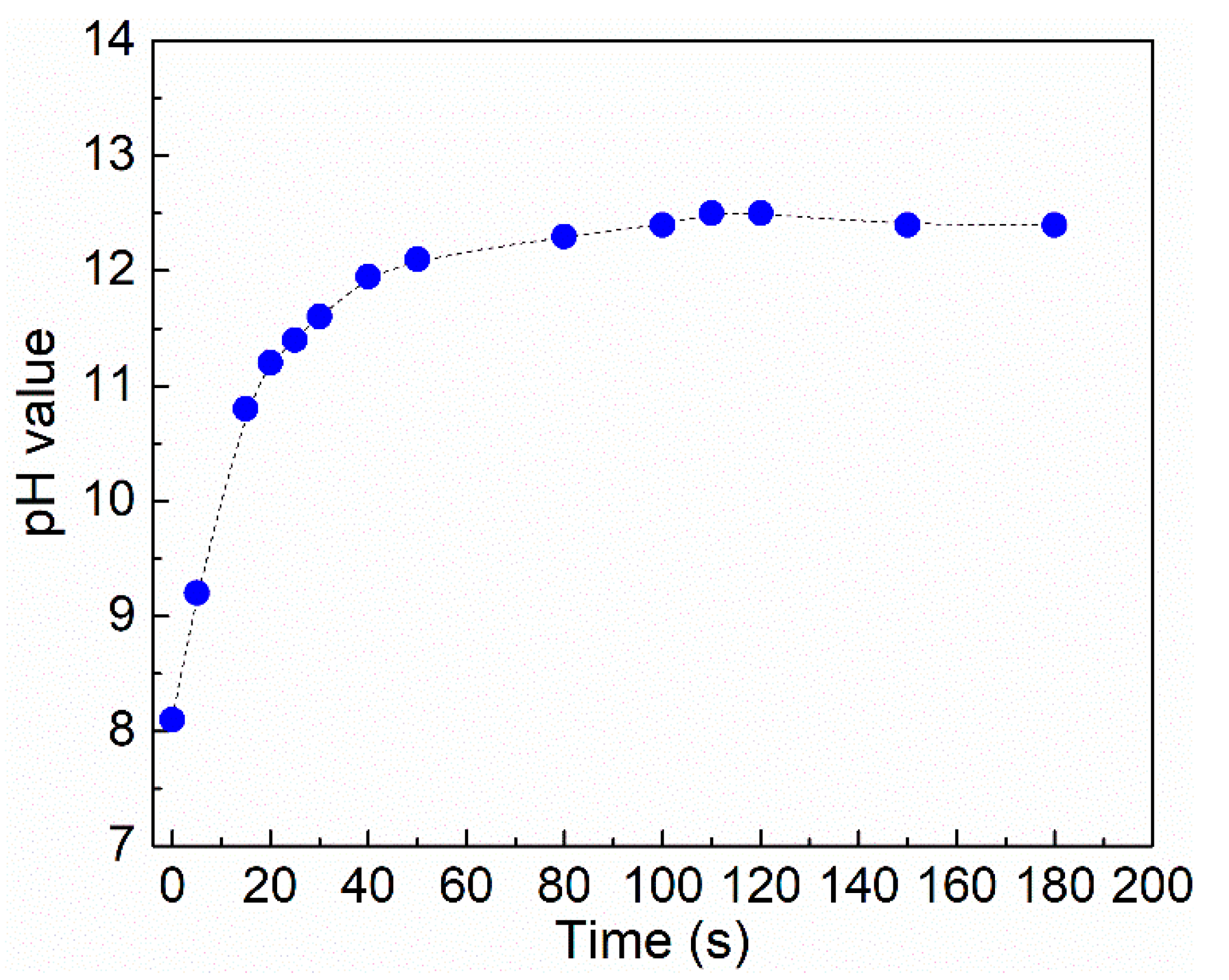
2.2.1. Testing of pH
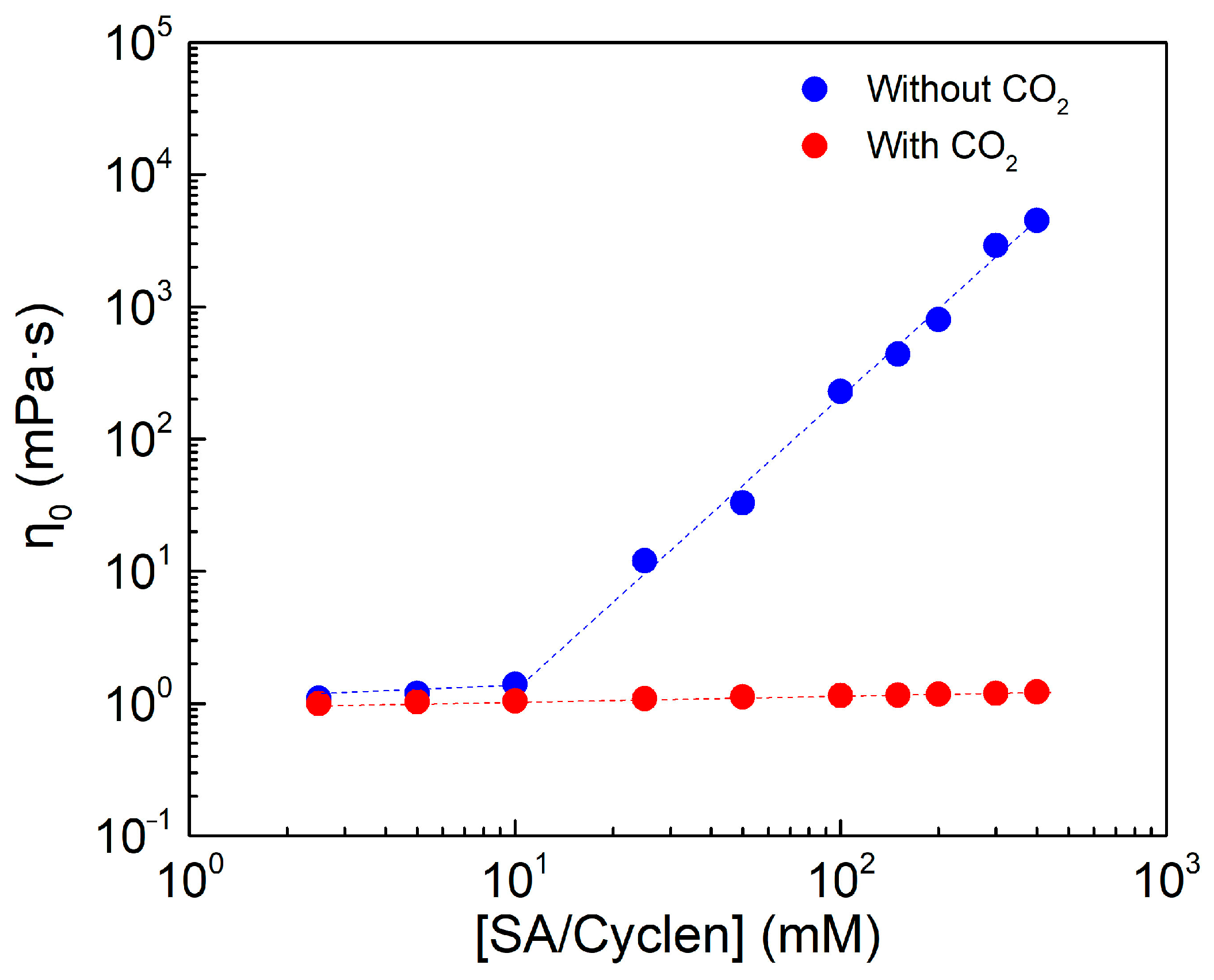
2.2.2. Viscosity of Respective Solutions of SA and Cyclen
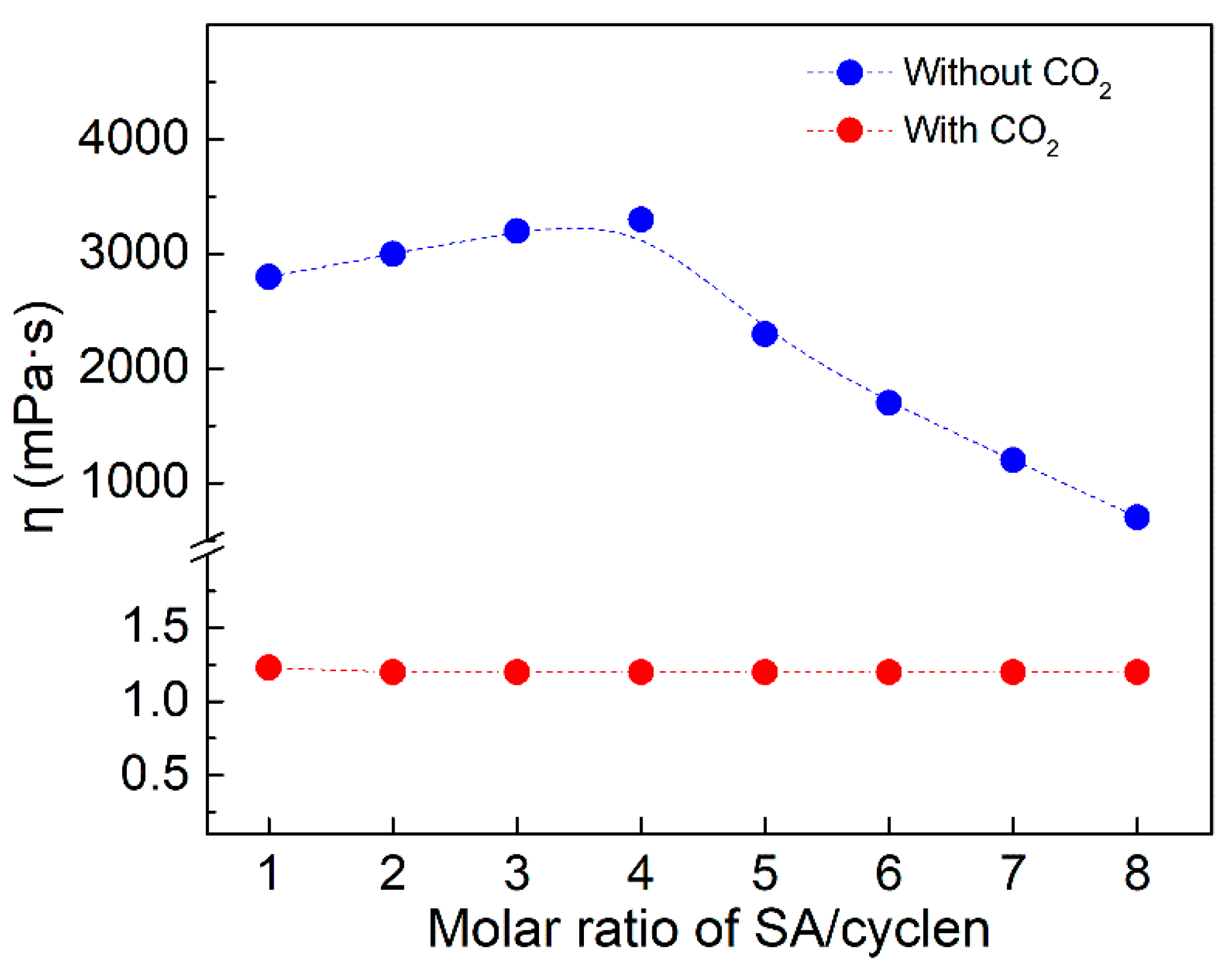
2.2.3. Ratio of SA and Cyclen
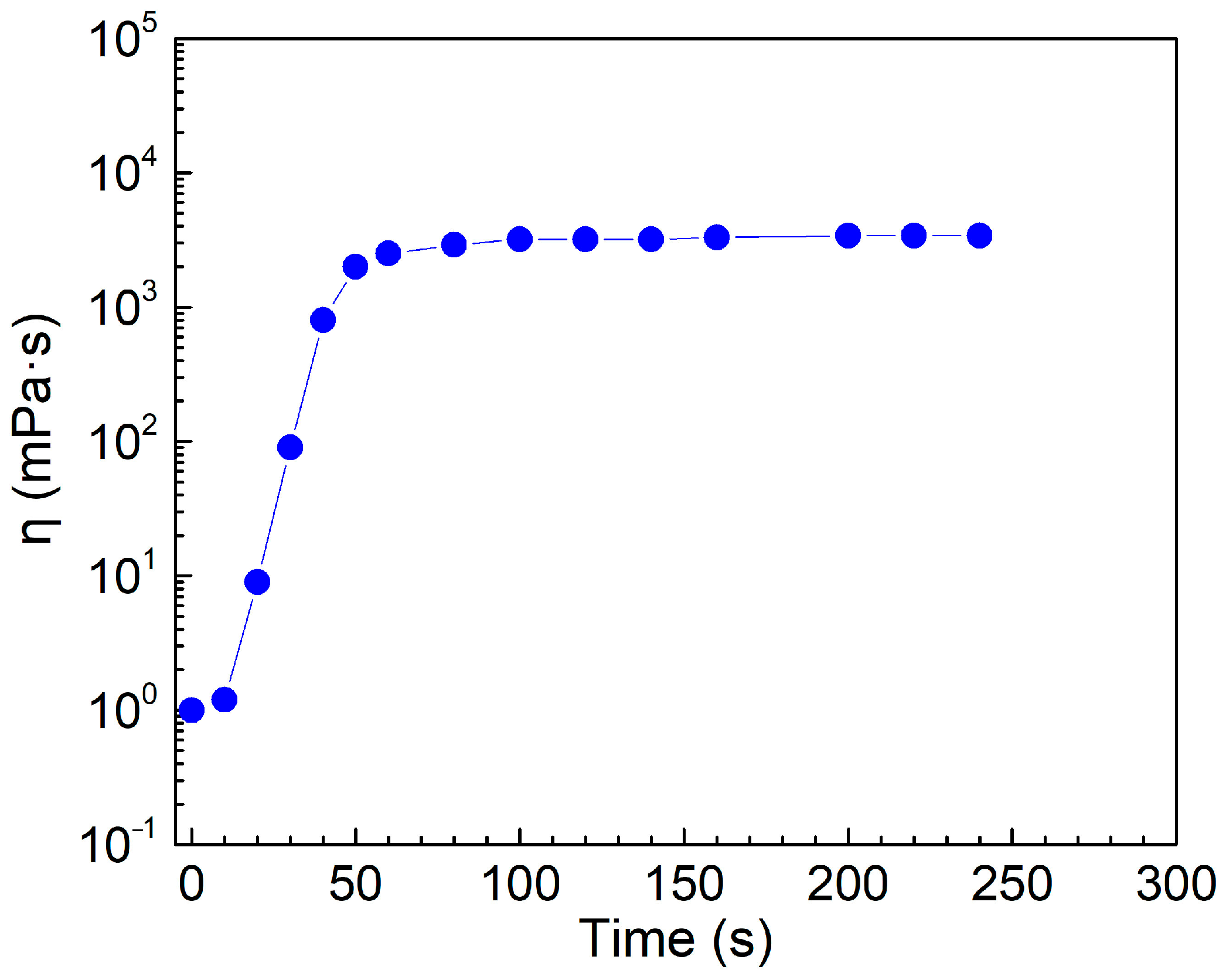
2.3. Effect of Sparging Time for CO2
2.4. Effect of SA/Cyclen Concentration
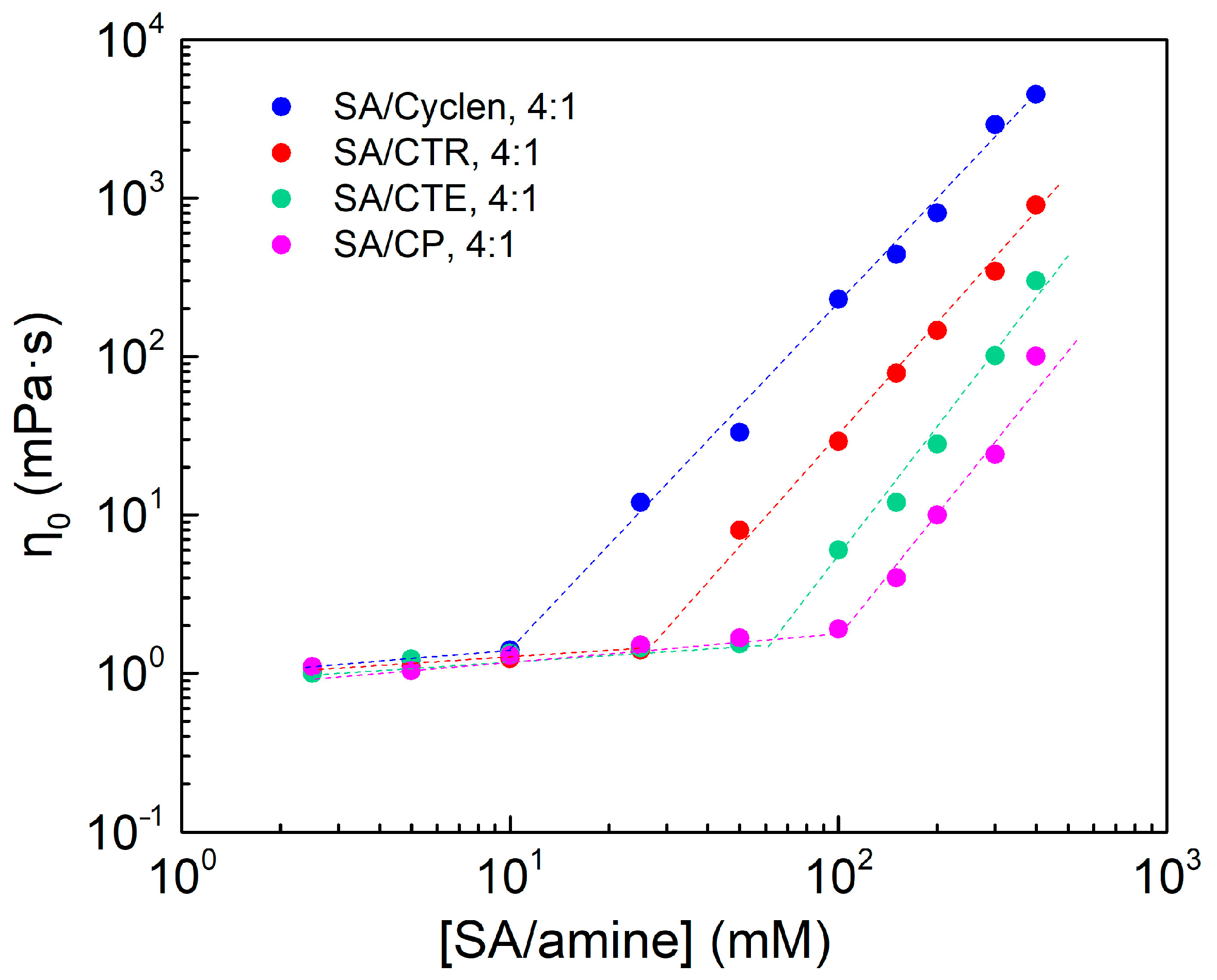
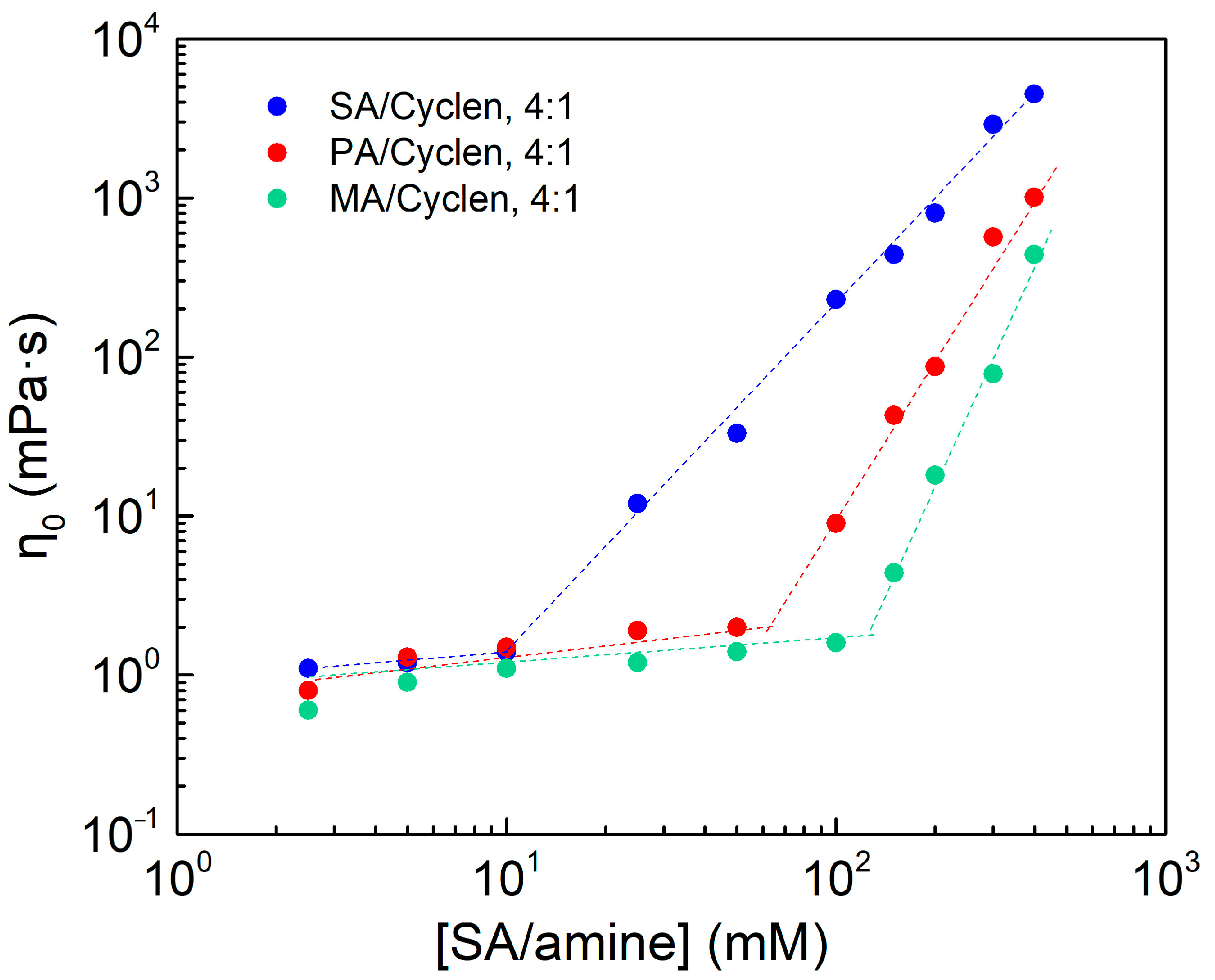
2.5. Effect of Amine Types
2.6. Effect of Hydrophobic Chain Length
3. Experimental Procedures
3.1. Materials
3.2. Preparation of Solution
3.3. pH Testing
3.4. Rheological Performance
3.5. Cryo-TEM
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cates, M.E.; Candau, S.J. Statics and dynamics of worm-like surfactant micelles. J. Phys.-Condes. Matter 1990, 2, 6869–6892. [Google Scholar] [CrossRef]
- Magid, L.J. The surfactant-polyelectrolyte analogy. J. Phys. Chem. B 1998, 102, 4064–4074. [Google Scholar] [CrossRef]
- Rehage, H.; Hoffmann, H. Viscoelastic surfactant solutions: Model systems for rheological research. Mol. Phys. 1991, 74, 933–973. [Google Scholar] [CrossRef]
- Yang, J. Viscoelastic wormlike micelles and their applications. Curr. Opin. Colloid Interface Sci. 2002, 7, 276–281. [Google Scholar] [CrossRef]
- Zakin, J.L.; Bewersdorff, H.W. Surfactant drag reduction. Rev. Chem. Eng. 1998, 14, 253–320. [Google Scholar] [CrossRef]
- Shi, H.; Wang, Y.; Fang, B.; Talmon, Y.; Ge, W.; Raghavan, S.R.; Zakin, J.L. Light-responsive threadlike micelles as drag reducing fluids with enhanced heat-transfer capabilities. Langmuir 2011, 27, 5806–5813. [Google Scholar] [CrossRef]
- Dreiss, C.A. Wormlike micelles: Where do we stand? Recent developments, linear rheology and scattering techniques. Soft Matter 2007, 3, 956–970. [Google Scholar] [CrossRef]
- Ezrahi, S.; Tuval, E.; Aserin, A. Properties, main applications and perspectives of worm micelles. Adv. Colloid Interface Sci. 2006, 128, 77–102. [Google Scholar] [CrossRef]
- Bernheim-Groswasser, A.; Wachtel, E.; Talmon, Y. Micellar growth, network formation, and criticality in aqueous solutions of the nonionic surfactant C12E5. Langmuir 2000, 16, 4131–4140. [Google Scholar] [CrossRef]
- Porte, G. Giant micelles in ideal solutions–either rods or vesicles? J. Phys. Chem. 1983, 87, 3541–3550. [Google Scholar] [CrossRef]
- Turner, M.S.; Marques, C.; Cates, M.E. Dynamics of wormlike micelles: The “bond-interchange” reaction scheme. Langmuir 1993, 9, 695–701. [Google Scholar] [CrossRef]
- Reber, A.C.; Khanna, S.N.; Ottenbrie, R. Thermodynamic stability of polyacrylamide and poly(N,N-dimethyl acrylamide). Polym. Adv. Technol. 2007, 18, 978–985. [Google Scholar] [CrossRef]
- Minagawa, K.; Koyama, K. Electro- and magneto-rheological materials: Stimuli-induced rheological functions. Curr. Org. Chem. 2005, 9, 1643–1663. [Google Scholar] [CrossRef]
- Cates, M.E. Reptation of living-polymers-dynamics of entangled polymers in the presence of reversible chain-scission reactions. Macromolecules 1987, 20, 2289–2296. [Google Scholar] [CrossRef]
- Lin, Y.; Qiao, Y.; Yan, Y.; Huang, J. Thermo-responsive viscoelastic wormlike micelle to elastic hydrogel transition in dual-component systems. Soft Matter 2009, 5, 3047–3053. [Google Scholar] [CrossRef]
- Abe, M.; Tobita, K.; Sakai, H.; Kamogawa, K.; Momozawa, N.; Kondo, Y.; Yoshino, N. Thermoresponsive viscoelasticity of concentrated solutions with a fluorinated hybrid surfactant. Colloid Surf. A-Physicochem. Eng. Asp. 2000, 167, 47–60. [Google Scholar] [CrossRef]
- Salkar, R.A.; Hassan, P.A.; Samant, S.D.; Valaulikar, B.S.; Kumar, V.V.; Kern, F.; Candau, S.J.; Manohar, C. A thermally reversible vesicle to micelle transition driven by a surface solid-fluid transition. Chem. Commun. 1996, 1223–1224. [Google Scholar] [CrossRef]
- Klijn, J.E.; Stuart, M.C.A.; Scarzello, M.; Wagenaar, A.; Engberts, J.B.F.N. pH-dependent phase behavior of carbohydrate-based gemini surfactants. Effect of the length of the hydrophobic spacer. J. Phys. Chem. B 2006, 110, 21694–21700. [Google Scholar] [CrossRef]
- Johnsson, M.; Wagenaar, A.; Stuart, M.C.A.; Engberts, J. Sugar-based gemini surfactants with pH-dependent aggregation behavior: Vesicle–to–micelle transition, critical micelle concentration, and vesicle surface charge reversal. Langmuir 2003, 2003 19, 4609–4618. [Google Scholar] [CrossRef]
- Sakai, H.; Orihara, Y.; Kodashima, H.; Matsumura, A.; Ohkubo, T.; Tsuchiya, K.; Abe, M. Photoinduced reversible change of fluid viscosity. J. Am. Chem. Soc. 2005, 127, 13454–13455. [Google Scholar] [CrossRef]
- Lin, Y.; Cheng, X.; Qiao, Y.; Yu, C.; Li, Z.; Yan, Y.; Huang, J. Creation of photo-modulated multi-state and multi-scale molecular assemblies via binary-state molecular switch. Soft Matter 2010, 6, 902–908. [Google Scholar] [CrossRef]
- Yang, Y. Novel redox-responsive surfactant. Electrochem. Soc. Interface 2006, 15, 58–59. [Google Scholar] [CrossRef]
- Tsuchiya, K.; Orihara, Y.; Kondo, Y.; Yoshino, N.; Ohkubo, T.; Sakai, H.; Abe, M. Control of viscoelasticity using redox reaction. J. Am. Chem. Soc. 2004, 126, 12282–12283. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jessop, P.G.; Cunningham, M.; Eckert, C.A.; Liotta, C.L. Switchable surfactants. Science 2006, 313, 958–960. [Google Scholar] [CrossRef] [PubMed]
- Schuur, B.; Nijland, M.; Blahušiak, M.; Juan, A. CO2-switchable solvents as entrainer in fluid separations. ACS Sustain. Chem. Eng. 2018, 6, 10429–10435. [Google Scholar] [CrossRef] [PubMed]
- Mercer, S.M.; Jessop, P.G. ”Switchable water”: Aqueous solutions of switchable ionic strength. ChemSusChem 2010, 3, 467–470. [Google Scholar] [CrossRef]
- Cunningham, M.C.; Jessop, P.G. Carbon dioxide-switchable polymers: Where are the future opportunities? Macromolecules 2019, 52, 6801–6816. [Google Scholar] [CrossRef]
- Zhang, Y.; Yin, H.; Feng, Y. CO2-responsive anionic wormlike micelles based on natural erucic acid. Green Mat. 2014, 2, 95–103. [Google Scholar] [CrossRef]
- Zhao, Z.; Lu, G. Visco-elastic properties of VES diverting acid for carbonate reservoirs. Chin. J. Chem. Eng. 2010, 18, 511–514. [Google Scholar] [CrossRef]
- Verma, G.; Aswal, V.K.; Hassan, P. pH-Responsive self-assembly in an aqueous mixture of surfactant and hydrophobic amino acid mimic. Soft Matter 2009, 5, 2919–2927. [Google Scholar] [CrossRef]
- Chu, Z.; Feng, Y. pH Switchable wormlike micelles. Chem. Commun. 2010, 46, 9028–9030. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; He, S.; Tang, L.; Yang, S.; Ma, T.; Su, X. Application of CO2-switchable oleic-acid-based surfactant for reducing viscosity of heavy oil. Molecules 2021, 26, 6273. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Han, Y.; Chu, Z.; He, S.; Zhang, J.; Feng, Y. Thermally induced structural transitions from fluids to hydrogels with pH-switchable anionic wormlike micelles. J. Colloid Interface Sci. 2013, 394, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Feng, Y.; Wang, Y.; Li, X. CO2-switchable viscoelastic fluids based on a pseudogemini surfactant. Langmuir 2013, 29, 4187–4192. [Google Scholar] [CrossRef]
- Hossain, M.S.; Berg, S.; Bergström, C.A.S.; Larsson, P. Aggregation behavior of medium chain fatty acids studied by coarse-grained molecular dynamics simulation. AAPS PhamSciTech 2019, 20, 61. [Google Scholar] [CrossRef]


| Amines | IUPAC Name | Molecular Structure | Number of Carbon Atoms of Spacer Group | Viscosity a at 100 mM (mPa·s) |
|---|---|---|---|---|
| Cyclen | 1,4,7,10-tetraazacyclododecane |  | 2, 2, 2, 2 | 231 |
| CTR | 1,4,7,10-tetraazacyclotridecane | 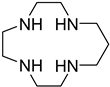 | 3, 2, 2, 2 | 29.2 |
| CTE | 1,4,8,11-tetraazacyclotetradecane |  | 3, 2, 3, 2 | 6.1 |
| CP | 1,4,8,12-tetraazacyclopentadecane | 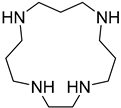 | 3, 3, 3, 2 | 1.9 |
| Long-Chain Carboxylic Acid | Reagent Name | Number of Carbon Atoms Contained in Hydrophobic Chain | Viscosity a at 100 mM (mPa·s) |
|---|---|---|---|
| SA | Stearic acid | 18 | 231 |
| PA | Palmitic acid | 16 | 8.9 |
| MA | Myristic acid | 14 | 1.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, X.; He, X.; Zhang, D.; Su, X. CO2-Responsive Wormlike Micelles Based on Pseudo-Tetrameric Surfactant. Molecules 2022, 27, 7922. https://doi.org/10.3390/molecules27227922
Wei X, He X, Zhang D, Su X. CO2-Responsive Wormlike Micelles Based on Pseudo-Tetrameric Surfactant. Molecules. 2022; 27(22):7922. https://doi.org/10.3390/molecules27227922
Chicago/Turabian StyleWei, Xia, Xiran He, Dongmei Zhang, and Xin Su. 2022. "CO2-Responsive Wormlike Micelles Based on Pseudo-Tetrameric Surfactant" Molecules 27, no. 22: 7922. https://doi.org/10.3390/molecules27227922
APA StyleWei, X., He, X., Zhang, D., & Su, X. (2022). CO2-Responsive Wormlike Micelles Based on Pseudo-Tetrameric Surfactant. Molecules, 27(22), 7922. https://doi.org/10.3390/molecules27227922





