Glycerol-Based Retrievable Heterogeneous Catalysts for Single-Pot Esterification of Palm Fatty Acid Distillate to Biodiesel
Abstract
1. Introduction
2. Results and Discussion
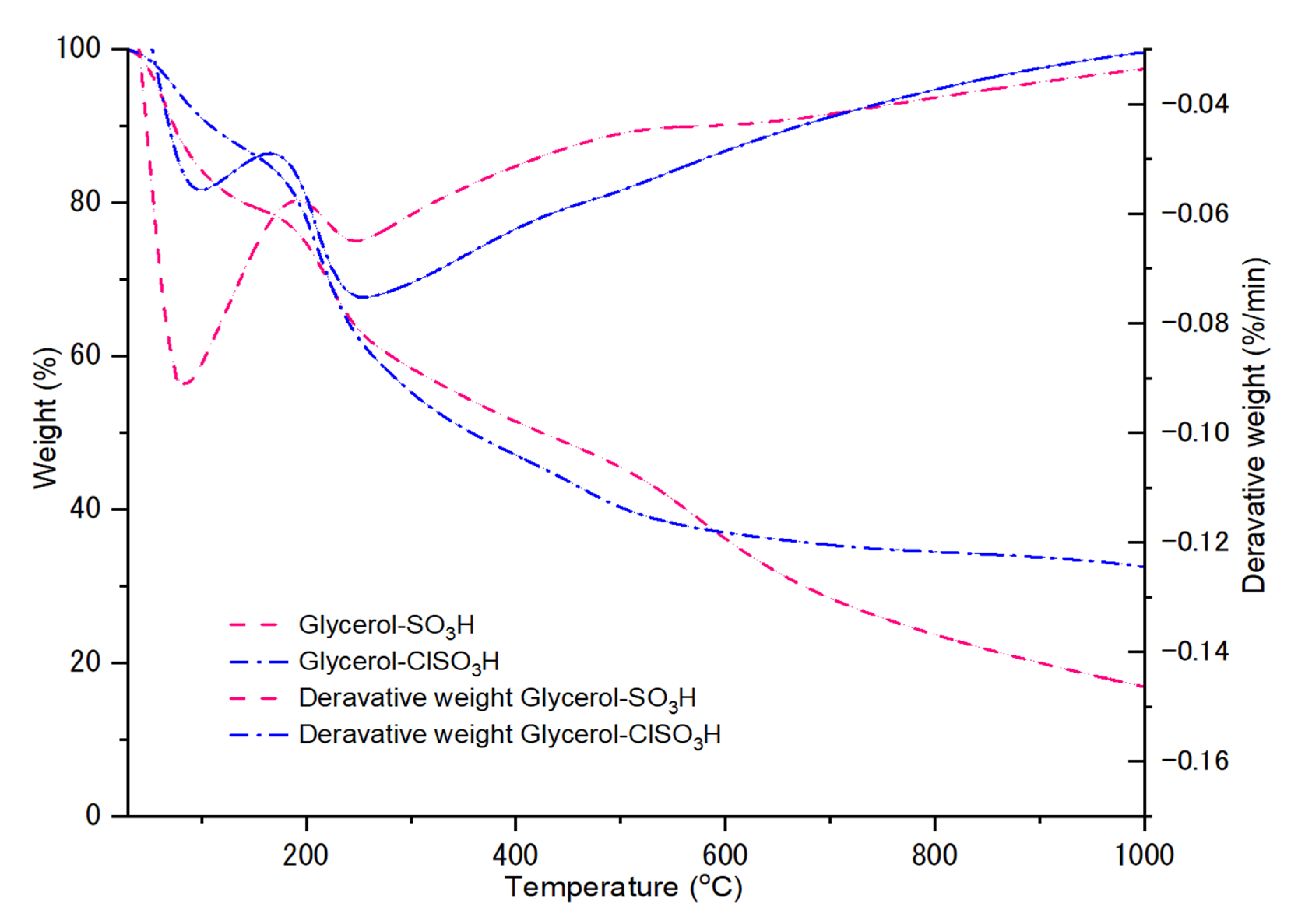
2.1. Thermal Degradation Analysis
2.2. Acid Properties Analysis
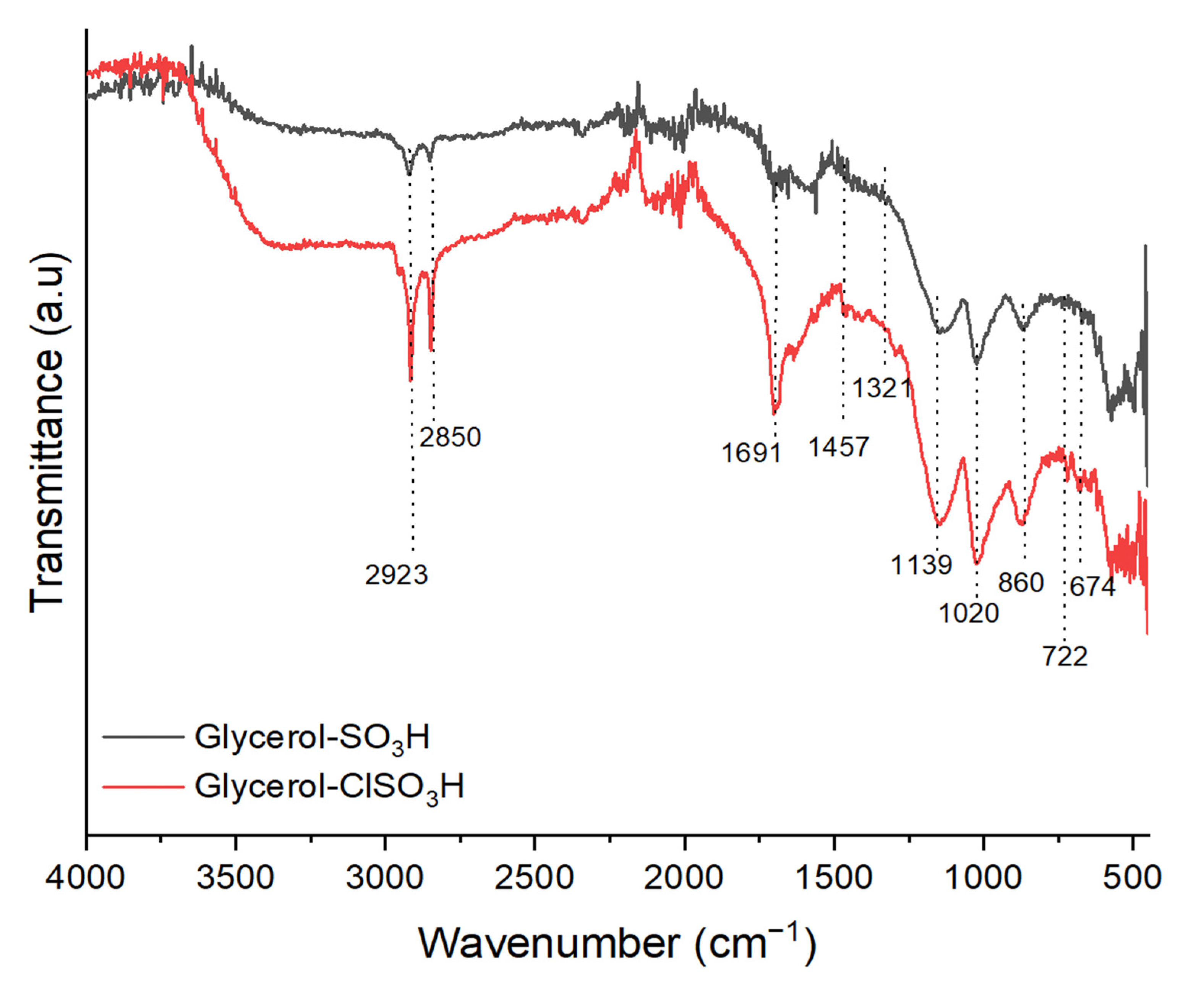
2.3. Functional Groups of Glycerol-SO3H and Glycero-ClSO3H
2.4. Surface Area and Pore Size Analysis
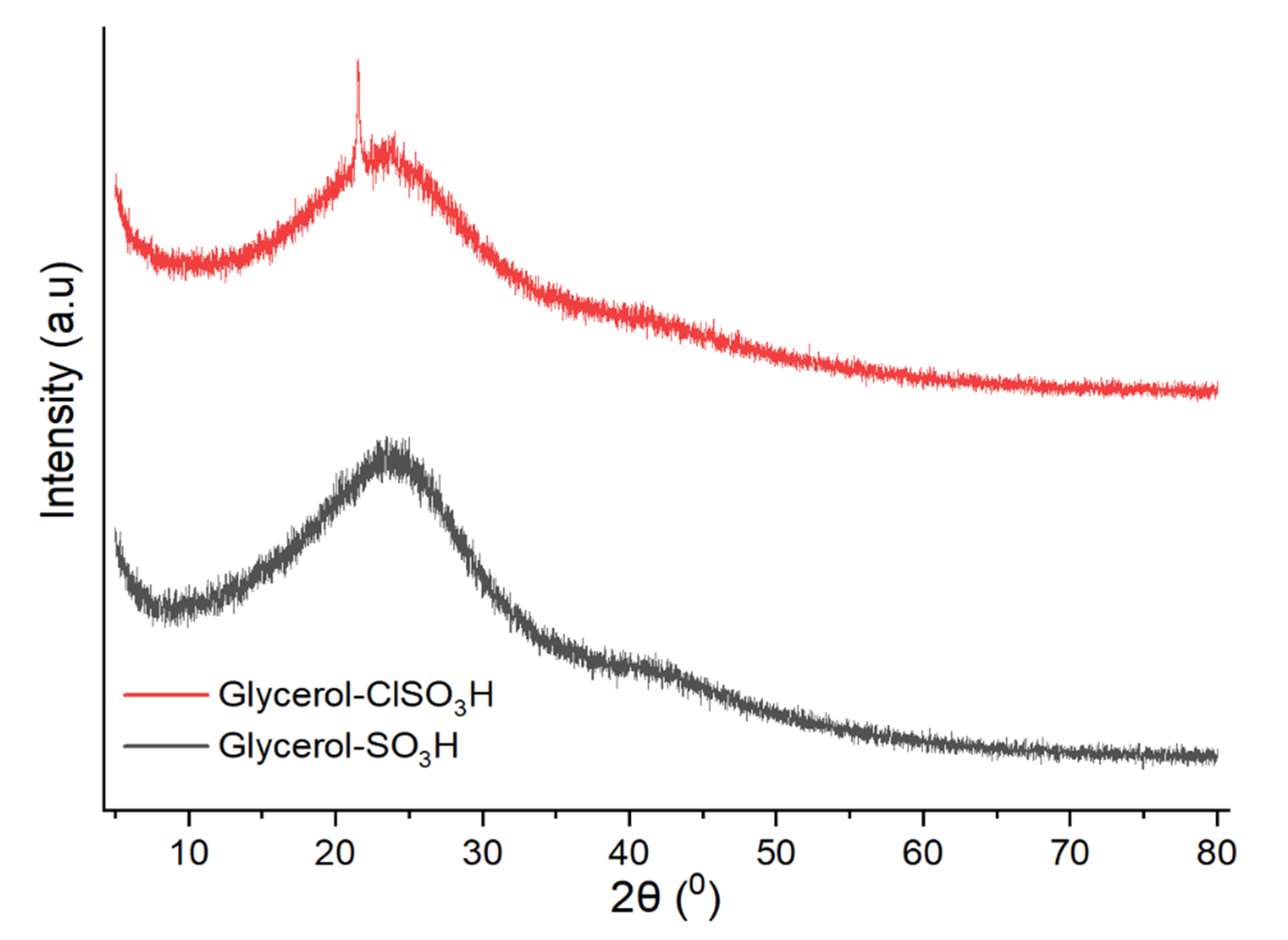
2.5. X-ray Diffraction Analysis
2.6. Microscopic Surface Morphology of Acidic Glycerol Catalysts
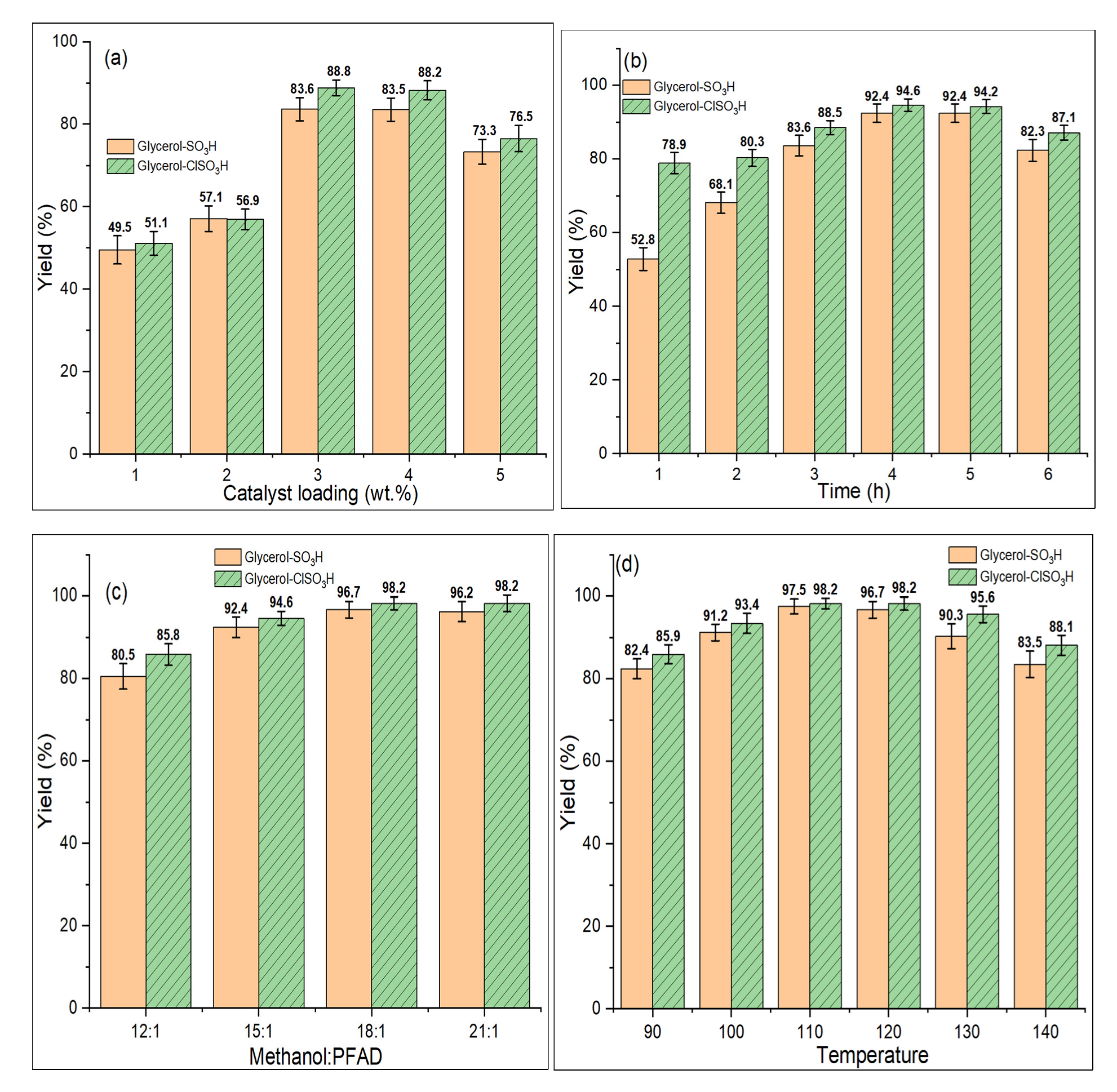
2.7. Catalyst Activity: Experimental Optimization Studies
2.7.1. Catalyst Loading
2.7.2. Reaction Time
2.7.3. Methanol-PFAD Molar Ratio
2.7.4. Reaction Temperature
2.8. Plausible Reaction Mechanism of PFAD Esterification with Produced Acidic Glycerol in Presence of Methanol
2.9. Reusability and Characterization of Spent Catalysts
2.10. Comparison Study of the Esterification by the Sulfonated Catalyst for PFAD Biodiesel Production
3. Materials and Methods
3.1. Chemical Reagents
3.2. Preparation of Acidic Glycerol Catalysts
3.3. Characterizations
3.4. Esterification of Palm Fatty Acid Distillate (PFAD) Using Acidic Glycerol Catalysts
3.5. Biodiesel Yield Analysis

3.6. Catalysts Reusability
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maness, P.C.; Yu, J.; Eckert, C.; Ghirardi, M.L. Photobiological hydrogen production—Prospects and challenges. Microbe 2009, 4, 275–280. [Google Scholar] [CrossRef][Green Version]
- Shuba, E.S.; Kifle, D. Microalgae to biofuels: ‘Promising’ alternative and renewable energy, review. Renew. Sustain. Energy Rev. 2018, 81, 743–755. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Fu, J.; Shi, D. Exploitation of oil-bearing microalgae for biodiesel. Chin. J. Biotechnol. 2008, 24, 341–348. [Google Scholar] [CrossRef]
- Hazmi, B.; Rashid, U.; Kawi, S.; Mokhtar, W.N.A.W.; Ahangar, F.A.; Yaw, T.C.S.; Tsubota, T.; Alharthi, F.A.; Ngamcharussrivichai, C. A heterogeneous bifunctional carbon nanocatalyst from plastic waste to efficiently catalyze waste cooking oil into biodiesel. Catalysts 2022, 12, 874. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, L.L.; Tan, X.; Li, H.; Yang, S. Catalytic high-yield biodiesel production from fatty acids and non-food oils over a magnetically separable acid nanosphere. Ind. Crops Prod. 2021, 173, 114126. [Google Scholar] [CrossRef]
- Mahlia, T.M.I.; Syazmi, Z.A.H.S.; Mofijur, M.; Abas, A.E.P.; Bilad, M.R.; Ong, H.C.; Silitonga, A.S. Patent landscape review on biodiesel production: Technology updates. Renew. Sustain. Energy Rev. 2020, 118, 109526. [Google Scholar] [CrossRef]
- Mansir, N.; Teo, S.H.; Rashid, U.; Saiman, M.I.; Tan, Y.P.; Alsultan, G.A.; Taufiq-Yap, Y.H. Modified waste egg shell derived bifunctional catalyst for biodiesel production from high FFA waste cooking oil. A review. Renew. Sustain. Energy Rev. 2018, 82, 3645–3655. [Google Scholar] [CrossRef]
- Lokman, I.M.; Rashid, U.; Yunus, R.; Taufiq-Yap, Y.H. Carbohydrate-derived solid acid catalysts for biodiesel production from low-cost feedstocks: A review. Catal. Rev. Sci. Eng. 2014, 56, 187–219. [Google Scholar] [CrossRef]
- Sangar, S.K.; Syazwani, O.N.; Farabi, M.S.A.; Razali, S.M.; Shobhana, G.; Teo, S.H.; Taufiq-Yap, Y.H. Effective biodiesel synthesis from palm fatty acid distillate (PFAD) using carbon-based solid acid catalyst derived glycerol. Renew. Energy 2019, 142, 658–667. [Google Scholar] [CrossRef]
- Mardhiah, H.H.; Ong, H.C.; Masjuki, H.H.; Lim, S.; Pang, Y.L. Investigation of carbon-based solid acid catalyst from Jatropha curcas biomass in biodiesel production. Energy Convers. Manag. 2017, 144, 10–17. [Google Scholar] [CrossRef]
- Theam, K.L.; Islam, A.; Lee, H.V.; Taufiq-Yap, Y.H. Sucrose-derived catalytic biodiesel synthesis from low cost palm fatty acid distillate. Process Saf. Environ. Prot. 2015, 95, 126–135. [Google Scholar] [CrossRef]
- Syazwani, O.N.; Rashid, U.; Mastuli, M.S.; Taufiq-Yap, Y.H. Esterification of palm fatty acid distillate (PFAD) to biodiesel using Bi-functional catalyst synthesized from waste angel wing shell (Cyrtopleura costata). Renew. Energy 2019, 131, 187–196. [Google Scholar] [CrossRef]
- Cao, M.; Peng, L.; Xie, Q.; Xing, K.; Lu, M.; Ji, J. Sulfonated Sargassum horneri carbon as solid acid catalyst to produce biodiesel via esterification. Bioresour. Technol. 2021, 324, 124614. [Google Scholar] [CrossRef] [PubMed]
- Ballotin, F.C.; da Silva, M.J.; Lago, R.M.; Teixeira, A.P.d.C. Solid acid catalysts based on sulfonated carbon nanostructures embedded in an amorphous matrix produced from bio-oil: Esterification of oleic acid with methanol. J. Environ. Chem. Eng. 2020, 8, 103674. [Google Scholar] [CrossRef]
- Mo, X.; Lotero, E.; Lu, C.; Liu, Y.; Goodwin, J.G. A Novel Sulfonated Carbon Composite Solid Acid Catalyst for Biodiesel Synthesis. Catal. Lett. 2008, 123, 1–6. [Google Scholar] [CrossRef]
- Wang, J.-X.; Chen, K.-T.; Wen, B.-Z.; Liao, Y.-H.B.; Chen, C.-C. Transesterification of soybean oil to biodiesel using cement as a solid base catalyst. J. Taiwan Inst. Chem. Eng. 2012, 43, 215–219. [Google Scholar] [CrossRef]
- Lokman, I.M.; Goto, M.; Rashid, U.; Taufiq-Yap, Y.H. Sub- and supercritical esterification of palm fatty acid distillate with carbohydrate-derived solid acid catalyst. Chem. Eng. J. 2016, 284, 872–878. [Google Scholar] [CrossRef]
- Coelho, A.; Perrone, O.M.; Gomes, E.; Da-Silva, R.; Thoméo, J.C.; Boscolo, M. Mixed metal oxides from sucrose and cornstarch templated hydrotalcite-like LDHs as catalysts for ethyl biodiesel synthesis. Appl. Catal. A Gen. 2017, 532, 32–39. [Google Scholar] [CrossRef]
- Taufiq-Yap, Y.H.; Lee, H.V.; Hussein, M.Z.; Yunus, R. Calcium-based mixed oxide catalysts for methanolysis of Jatropha curcas oil to biodiesel. Biomass Bioenergy 2011, 35, 827–834. [Google Scholar] [CrossRef]
- Lokman, I.M.; Rashid, U.; Taufiq-Yap, Y.H. Production of biodiesel from palm fatty acid distillate using sulfonated-glucose solid acid catalyst: Characterization and optimization. Chin. J. Chem. Eng. 2015, 23, 1857–1864. [Google Scholar] [CrossRef]
- Thushari, I.; Babel, S. Sustainable utilization of waste palm oil and sulfonated carbon catalyst derived from coconut meal residue for biodiesel production. Bioresour. Technol. 2018, 248, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Anitha, M.; Kamarudin, S.K.; Kofli, N.T. The potential of glycerol as a value-added commodity. Chem. Eng. J. 2016, 295, 119–130. [Google Scholar] [CrossRef]
- Nomanbhay, S.; Hussein, R.; Ong, M.Y. Sustainability of biodiesel production in Malaysia by production of bio-oil from crude glycerol using microwave pyrolysis: A review. Green Chem. Lett. Rev. 2018, 11, 135–157. [Google Scholar] [CrossRef]
- Ma, T.; Ding, J.; Liu, X.; Chen, G.; Zheng, J. Gas-phase dehydration of glycerol to acrolein over different metal phosphate catalysts. Korean J. Chem. Eng. 2020, 37, 955–960. [Google Scholar] [CrossRef]
- Shatesh, K.; Shamsuddin, M.R.; Farabi, M.A.; Saiman, M.I.; Zainal, Z.; Taufiq-Yap, Y.H. Production of methyl esters from waste cooking oil and chicken fat oil via simultaneous esterification and transesterification using acid catalyst. Energy Convers. Manag. 2020, 226, 113366. [Google Scholar] [CrossRef]
- Ma, T.; Liu, D.; Liu, Z.; Xu, J.; Dong, Y.; Chen, G.; Yun, Z. 12-Tungstophosphoric acid-encapsulated metal-organic framework UiO-66: A promising catalyst for the esterification of acetic acid with n-butanol. J. Taiwan Inst. Chem. Eng. 2022, 133, 104277. [Google Scholar] [CrossRef]
- Soltani, S.; Rashid, U.; Nehdi, I.A.; Al-Resayes, S.I.; Al-Muhtaseb, A.H. Sulfonated mesoporous zinc aluminate catalyst for biodiesel production from high free fatty acid feedstock using microwave heating system. J. Taiwan Inst. Chem. Eng. 2017, 70, 219–228. [Google Scholar] [CrossRef]
- Díaz-Muñoz, L.L.; Reynel-Ávila, H.E.; Mendoza-Castillo, D.I.; Bonilla-Petriciolet, A.; Jáuregui-Rincón, J. Preparation and Characterization of Alkaline and Acidic Heterogeneous Carbon-Based Catalysts and Their Application in Vegetable Oil Transesterification to Obtain Biodiesel. Int. J. Chem. Eng. 2022, 2022, 7056220. [Google Scholar] [CrossRef]
- Zhang, F.; Tian, X.F.; Fang, Z.; Shah, M.; Wang, Y.T.; Jiang, W.; Yao, M. Catalytic production of Jatropha biodiesel and hydrogen with magnetic carbonaceous acid and base synthesized from Jatropha hulls. Energy Convers. Manag. 2017, 142, 107–116. [Google Scholar] [CrossRef]
- Naeem, M.M.; Al-Sakkari, E.G.; Boffito, D.C.; Gadalla, M.A.; Ashour, F.H. One-pot conversion of highly acidic waste cooking oil into biodiesel over a novel bio-based bi-functional catalyst. Fuel 2021, 283, 118914. [Google Scholar] [CrossRef]
- Soltani, S.; Rashid, U.; Yunus, R.; Taufiq-Yap, Y.H. Biodiesel production in the presence of sulfonated mesoporous ZnAl2O4 catalyst via esterification of palm fatty acid distillate (PFAD). Fuel 2016, 178, 253–262. [Google Scholar] [CrossRef]
- Hazmi, B.; Rashid, U.; Kawi, S.; Mokhtar, W.N.A.W.; Yaw, T.C.S.; Moser, B.R.; Alsalme, A. Palm fatty acid distillate esterification using synthesized heterogeneous sulfonated carbon catalyst from plastic waste: Characterization, catalytic efficacy and stability, and fuel properties. Process Saf. Environ. Prot. 2022, 162, 1139–1151. [Google Scholar] [CrossRef]
- Jenie, S.N.A.; Kristiani, A.; Sudiyarmanto; Khaerudini, D.S.; Takeishi, K. Sulfonated magnetic nanobiochar as heterogeneous acid catalyst for esterification reaction. J. Environ. Chem. Eng. 2020, 8, 103912. [Google Scholar] [CrossRef]
- Khandan, M.; Saffarzadeh-Matin, S.; Shalmashi, A. Green hydrophobization of fume silica: Tailoring of heterogeneous basic catalyst for biodiesel production. J. Clean. Prod. 2020, 260, 121066. [Google Scholar] [CrossRef]
- Krishnamurthy, K.N.; Sridhara, S.N.; Ananda Kumar, C.S. Optimization and kinetic study of biodiesel production from Hydnocarpus wightiana oil and dairy waste scum using snail shell CaO nano catalyst. Renew. Energy 2019, 146, 280–296. [Google Scholar] [CrossRef]
- Lin, Q.-X.; Zhang, C.-H.; Wang, X.-H.; Cheng, B.-G.; Mai, N.; Ren, J.-L. Impact of activation on properties of carbon-based solid acid catalysts for the hydrothermal conversion of xylose and hemicelluloses. Catal. Today 2019, 319, 31–40. [Google Scholar] [CrossRef]
- Pan, H.; Sun, J.; Liu, J.; Zhang, Y.; Zhou, S. Preparation of sulfonated carbon derived from orange peel and its application in esterification. Chem. Phys. Lett. 2021, 770, 138395. [Google Scholar] [CrossRef]
- Arumugamurthy, S.S.; Sivanandi, P.; Pandian, S.; Choksi, H.; Subramanian, D. Conversion of a low value industrial waste into biodiesel using a catalyst derived from brewery waste: An activation and deactivation kinetic study. Waste Manag. 2019, 100, 318–326. [Google Scholar] [CrossRef]
- Dehghan, L.; Golmakani, M.T.; Hosseini, S.M.H. Optimization of microwave-assisted accelerated transesterification of inedible olive oil for biodiesel production. Renew. Energy 2019, 138, 915–922. [Google Scholar] [CrossRef]
- Abdullah, R.F.; Rashid, U.; Taufiq-Yap, Y.H.; Ibrahim, M.L.; Ngamcharussrivichai, C.; Azam, M. Synthesis of bifunctional nanocatalyst from waste palm kernel shell and its application for biodiesel production. RSC Adv. 2020, 10, 27183–27193. [Google Scholar] [CrossRef] [PubMed]
- Fadhil, A.B.; Al-Tikrity, E.T.B.; Albadree, M.A. Biodiesel production from mixed non-edible oils, castor seed oil and waste fish oil. Fuel 2017, 210, 721–728. [Google Scholar] [CrossRef]
- Roy, T.; Ágarwal, A.K.; Sharma, Y.C. A cleaner route of biodiesel production from waste frying oil using novel potassium tin oxide catalyst: A smart liquid-waste management. Waste Manag. 2021, 135, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, J.; Rashid, U.; Patuzzi, F.; Baratieri, M.; Taufiq-Yap, Y.H. Synthesis of char-based acidic catalyst for methanolysis of waste cooking oil: An insight into a possible valorization pathway for the solid by-product of gasification. Energy Convers. Manag. 2018, 158, 186–192. [Google Scholar] [CrossRef]
- Elias, S.; Rabiu, A.M.; Okeleye, B.I.; Okudoh, V.; Oyekola, O. Bifunctional heterogeneous catalyst for biodiesel production from waste vegetable oil. Appl. Sci. 2020, 10, 3153. [Google Scholar] [CrossRef]
- Nabihah-Fauzi, N.; Asikin-Mijan, N.; Ibrahim, M.L.; Hashim, H.; Yusup, S.; Taufiq-Yap, Y.H.; Mastuli, M.S. Sulfonated SnO2nanocatalysts: Via a self-propagating combustion method for esterification of palm fatty acid distillate. RSC Adv. 2020, 10, 29187–29201. [Google Scholar] [CrossRef]
- Deris, N.H.; Rashid, U.; Soltani, S.; Choong, T.S.Y.; Nehdi, I.A. Study the effect of various sulfonation methods on catalytic activity of carbohydrate-derived catalysts for ester production. Catalysts 2020, 10, 638. [Google Scholar] [CrossRef]
- Lim, S.; Yap, C.Y.; Pang, Y.L.; Wong, K.H. Biodiesel synthesis from oil palm empty fruit bunch biochar derived heterogeneous solid catalyst using 4-benzenediazonium sulfonate. J. Hazard. Mater. 2020, 390, 121532. [Google Scholar] [CrossRef]






| Catalyst | BET Specific Surface Area (m2/g) | Pore Volume (cm3/g) | Average Pore Size (nm) | Total Acidity (mmol/g) | Element (wt.%) | |||
|---|---|---|---|---|---|---|---|---|
| C | O | S | Cl | |||||
| Glycerol-SO3H | 8.85 (1.01) * | 0.07 (0.02) * | 5.28 (5.02) * | 3.43 (1.05) * | 56.3 (66.1) * | 25.7 (25.4) * | 18.0 (8.5) * | - - |
| Glycerol-ClSO3H | 4.71 (1.04) * | 0.02 (0.01) * | 5.28 (5.20) * | 3.96 (1.37) * | 48.9 (56.0) * | 32.4 (32.1) * | 16.6 (10.6) * | 2.1 (1.2) * |
| Catalyst | BET Specific Surface Area (m2/g) | Acidity (mmol/g) | Reaction Parameters | ReusabilityCycle | Yield (%) | Ref. |
|---|---|---|---|---|---|---|
| HSO3/SnO2 | 7.50 | 5.30 | 4 wt.%, 100 °C, 3 h, 9:1 | 5 | 96.4 | [46] |
| BSY-SO3H | 889 | 0.58 | 8 wt.%, 65 °C, 3 h, 21:1 | 4 | 87.8 | [39] |
| PSS-ICG | 8.70 | 14.64 | 2.5 wt.%, 80 °C, 4 h, 10:1 | - | 96.3 | [47] |
| EFB-4BDS | 2.85 | 3.93 | 20 wt.%, 7 h | - | 98.1 | [48] |
| Glycerol-SO3H Glycerol-ClSO3H | 8.85 4.71 | 3.43 3.96 | 3 wt.%, 120 °C, 4 h, 18:13 wt.%, 120 °C, 4 h, 18:1 | 3 3 | 96.7 98.2 | This study This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hazmi, B.; Beygisangchin, M.; Rashid, U.; Mokhtar, W.N.A.W.; Tsubota, T.; Alsalme, A.; Ngamcharussrivichai, C. Glycerol-Based Retrievable Heterogeneous Catalysts for Single-Pot Esterification of Palm Fatty Acid Distillate to Biodiesel. Molecules 2022, 27, 7142. https://doi.org/10.3390/molecules27207142
Hazmi B, Beygisangchin M, Rashid U, Mokhtar WNAW, Tsubota T, Alsalme A, Ngamcharussrivichai C. Glycerol-Based Retrievable Heterogeneous Catalysts for Single-Pot Esterification of Palm Fatty Acid Distillate to Biodiesel. Molecules. 2022; 27(20):7142. https://doi.org/10.3390/molecules27207142
Chicago/Turabian StyleHazmi, Balkis, Mahnoush Beygisangchin, Umer Rashid, Wan Nur Aini Wan Mokhtar, Toshiki Tsubota, Ali Alsalme, and Chawalit Ngamcharussrivichai. 2022. "Glycerol-Based Retrievable Heterogeneous Catalysts for Single-Pot Esterification of Palm Fatty Acid Distillate to Biodiesel" Molecules 27, no. 20: 7142. https://doi.org/10.3390/molecules27207142
APA StyleHazmi, B., Beygisangchin, M., Rashid, U., Mokhtar, W. N. A. W., Tsubota, T., Alsalme, A., & Ngamcharussrivichai, C. (2022). Glycerol-Based Retrievable Heterogeneous Catalysts for Single-Pot Esterification of Palm Fatty Acid Distillate to Biodiesel. Molecules, 27(20), 7142. https://doi.org/10.3390/molecules27207142








