DDX3 Upregulates Hydrogen Peroxide-Induced Melanogenesis in Sk-Mel-2 Human Melanoma Cells
Abstract
1. Introduction
2. Results
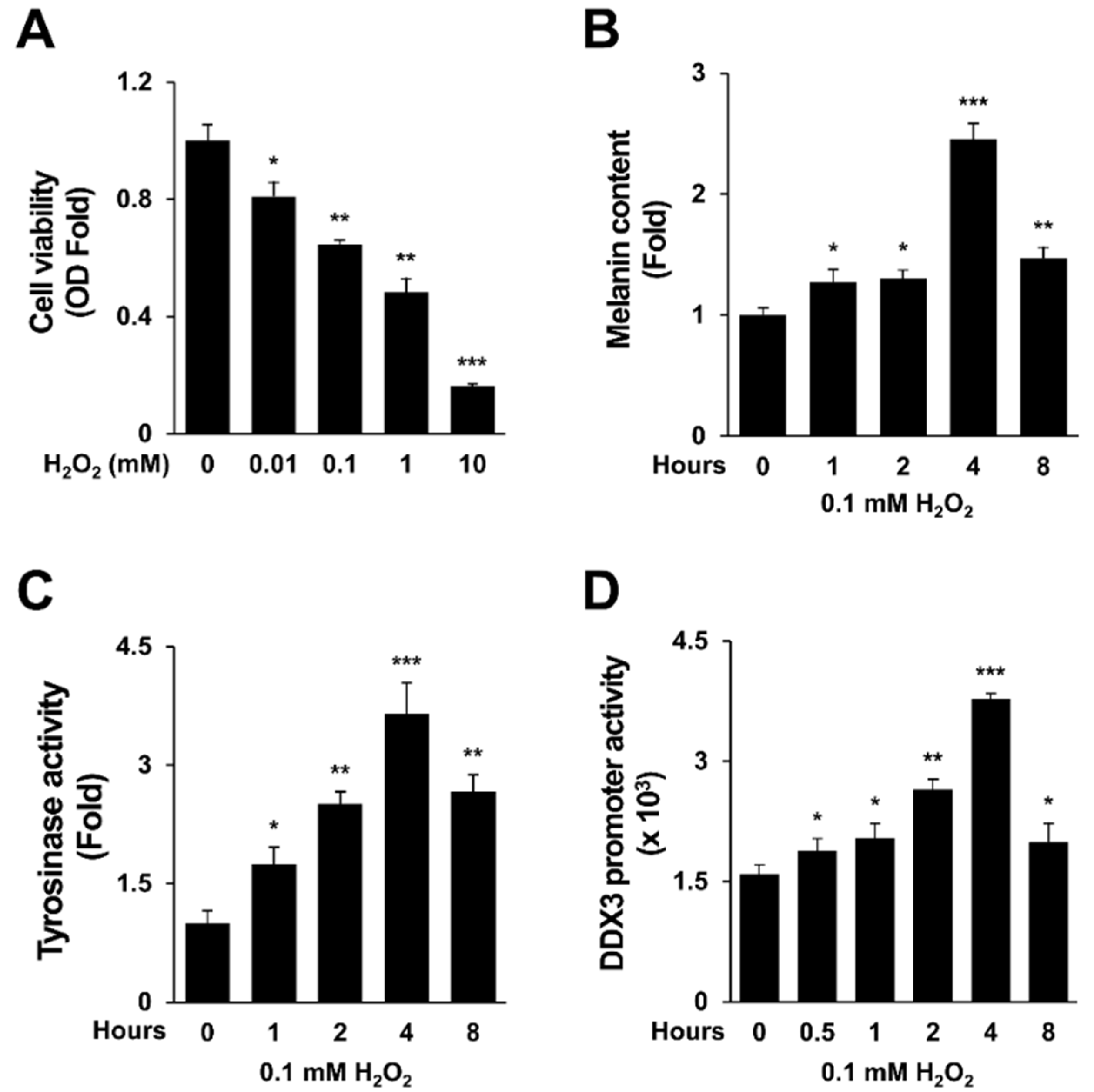
2.1. H2O2 Activates Melanogenesis and DDX3 Promoter Activity
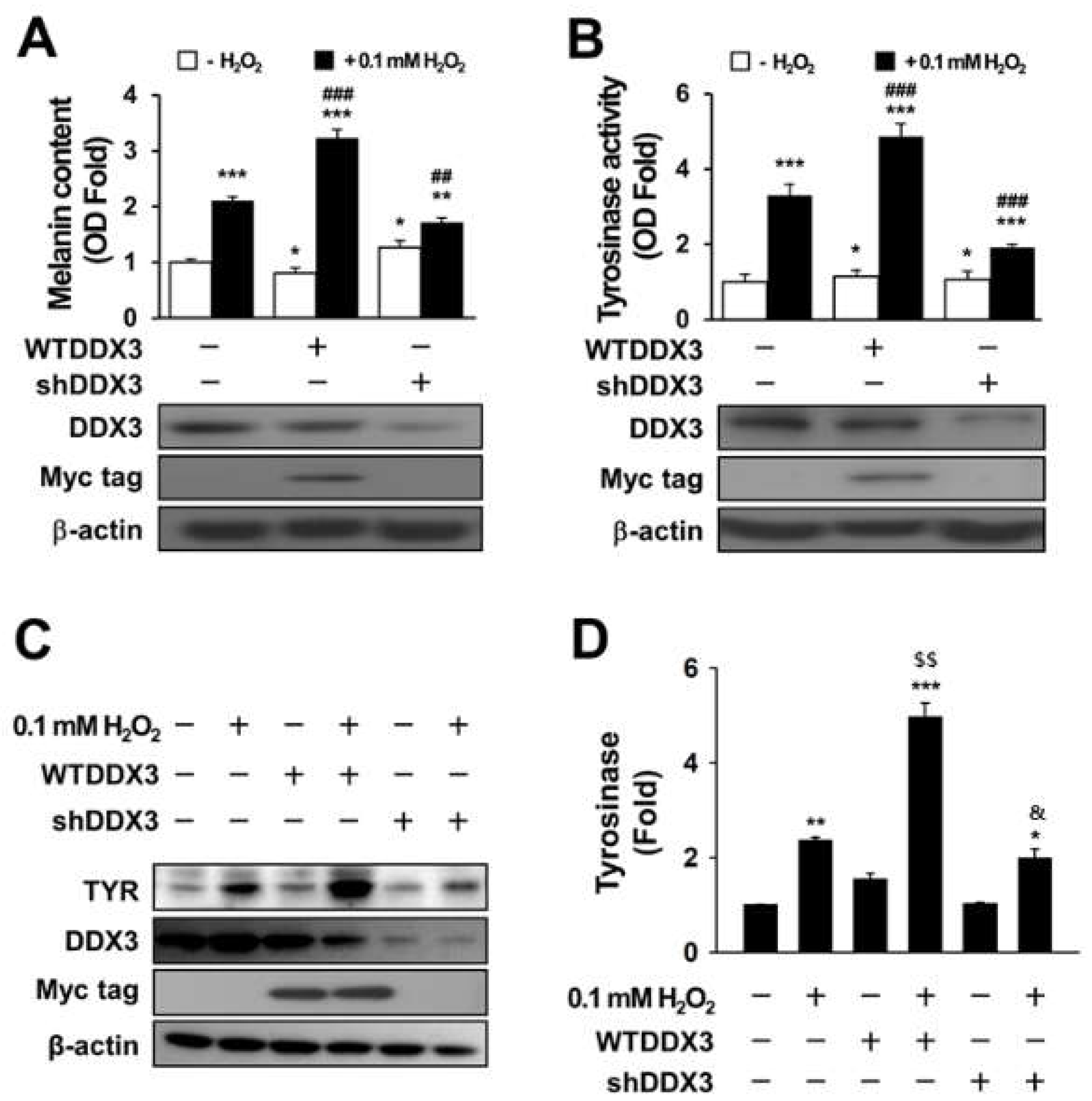
2.2. DDX3 Induces Melanogenesis in H2O2-Treated Condition Cells
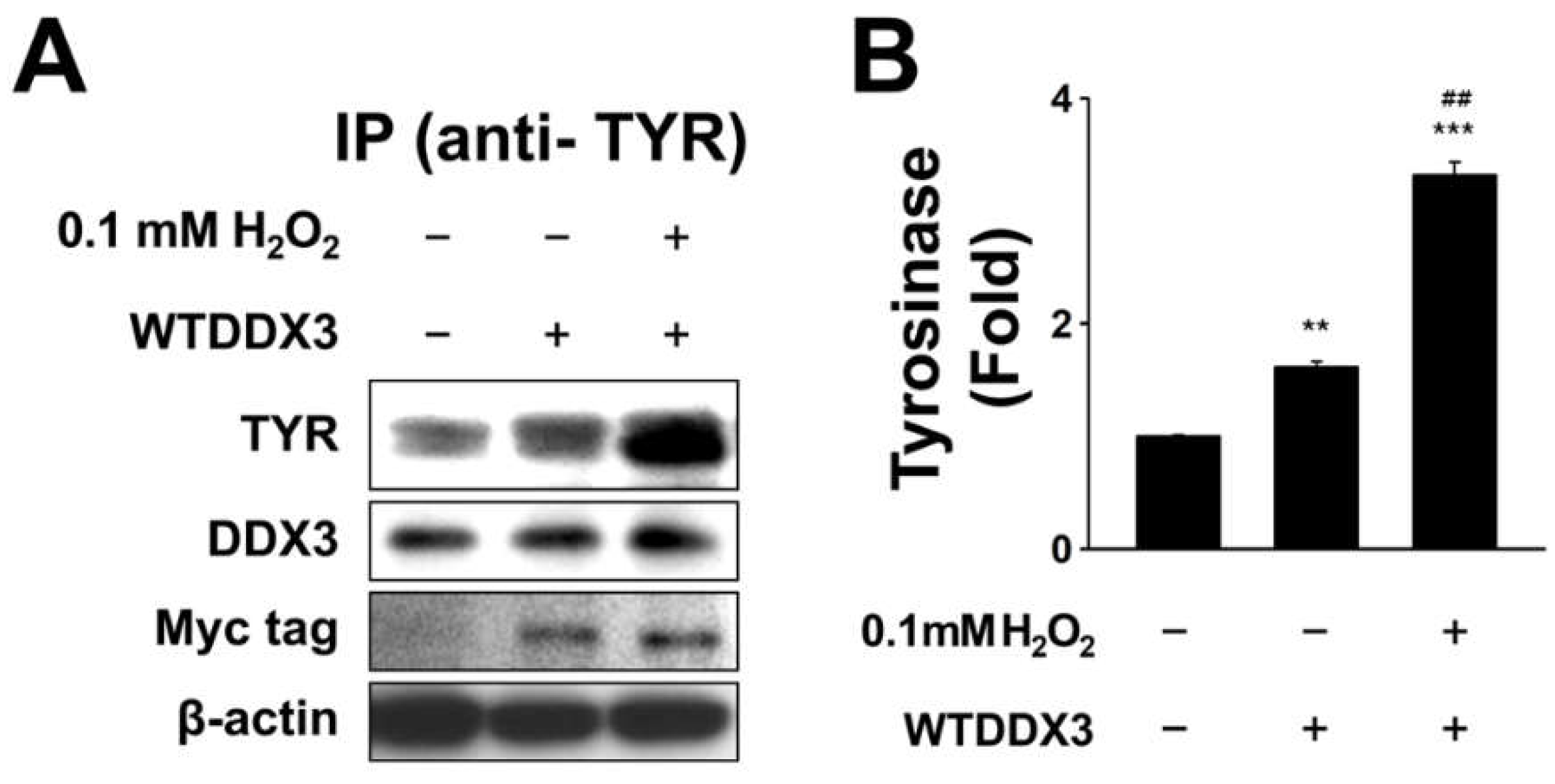
2.3. DDX3 Binds to Tyrosinase in H2O2-Treated Condition
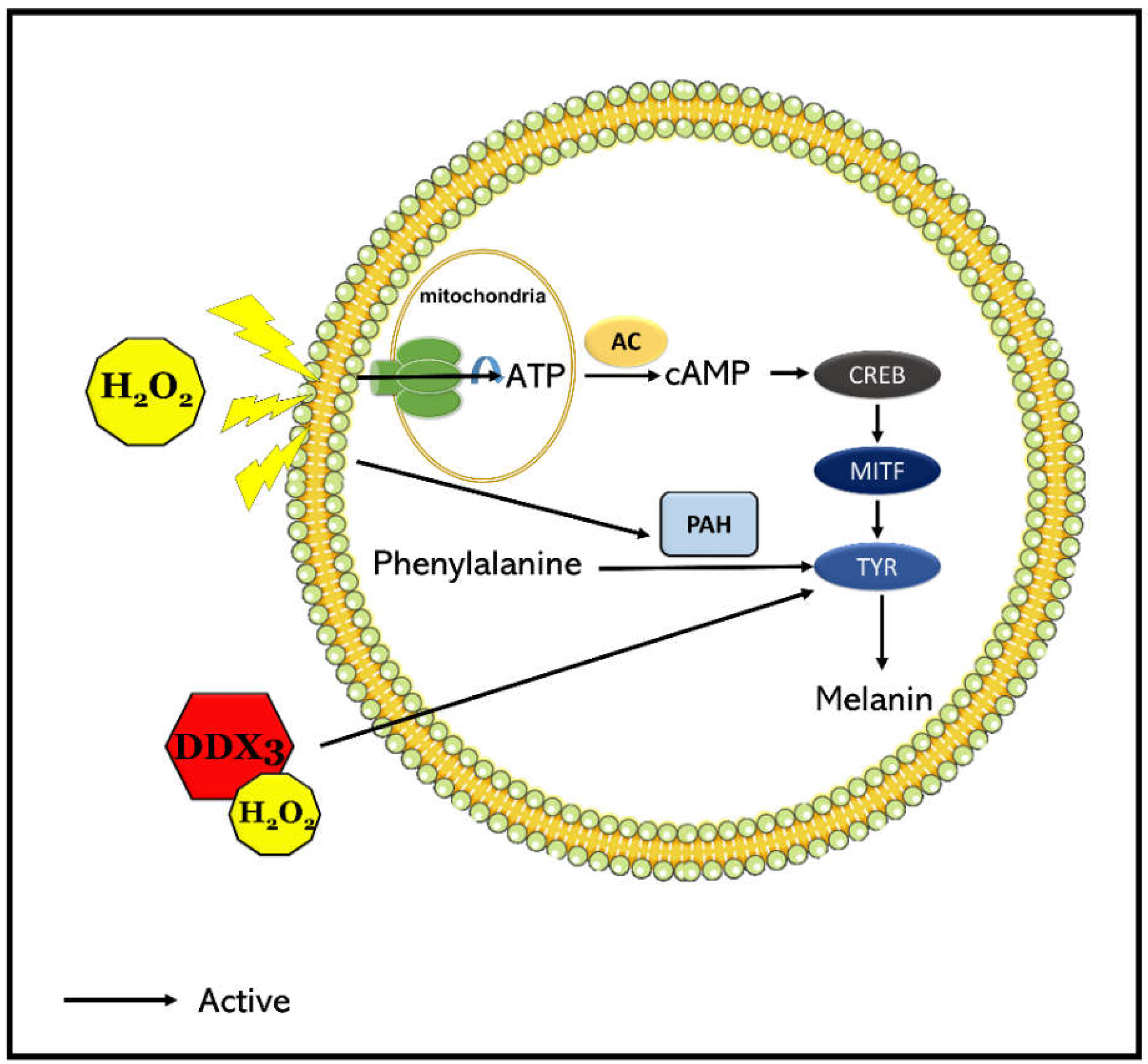
3. Discussion
4. Materials and Methods
4.1. Chemicals and Antibodies
4.2. Cell Culture
4.3. Melanin Content Measurement
4.4. Tyrosinase Activity Assay
4.5. Luciferase Reporter Assay
4.6. Immunoprecipitation (IP)
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cordero, R.J.; Casadevall, A. Melanin. Curr. Biol. 2020, 30, R142–R143. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Kosmadaki, M.; Yaar, M.; Gilchrest, B.A. Cellular mechanisms regulating human melanogenesis. Cell. Mol. Life Sci. 2009, 66, 1493–1506. [Google Scholar] [CrossRef] [PubMed]
- Ariumi, Y. Multiple functions of DDX3 RNA helicase in gene regulation, tumorigenesis, and viral infection. Front. Genet. 2014, 5, 423. [Google Scholar] [CrossRef] [PubMed]
- Rouzaud, F.; Kadekaro, A.L.; Abdel-Malek, Z.A.; Hearing, V.J. MC1R and the response of melanocytes to ultraviolet radiation. Mutat. Res. Mol. Mech. Mutagen. 2005, 571, 133–152. [Google Scholar] [CrossRef] [PubMed]
- Ando, H.; Kondoh, H.; Ichihashi, M.; Hearing, V.J. Approaches to Identify Inhibitors of Melanin Biosynthesis via the Quality Control of Tyrosinase. J. Investig. Dermatol. 2007, 127, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Costin, G.-E.; Hearing, V.J. Human skin pigmentation: Melanocytes modulate skin color in response to stress. FASEB J. 2007, 21, 976–994. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Wakamatsu, K.; Ozeki, H. Chemical Analysis of Melanins and its Application to the Study of the Regulation of Melanogenesis. Pigment Cell Res. 2000, 13, 103–109. [Google Scholar] [CrossRef]
- Munoz-Munoz, J.L.; García-Molina, F.; Varón, R.; Tudela, J.; García-Cánovas, F.; Rodríguez-López, J.N. Generation of hydrogen peroxide in the melanin biosynthesis pathway. Biochim. et Biophys. Acta (BBA) Proteins Proteom. 2009, 1794, 1017–1029. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Hearing, V.J. Physiological factors that regulate skin pigmentation. BioFactors 2009, 35, 193–199. [Google Scholar] [CrossRef]
- Zhao, L.; Mao, Y.; Zhou, J.; Zhao, Y.; Cao, Y.; Chen, X. Multifunctional DDX3: Dual roles in various cancer development and its related signaling pathways. Am. J. Cancer Res. 2016, 6, 387–402. [Google Scholar]
- Stunnenberg, M.; Geijtenbeek, T.B.; Gringhuis, S.I. DDX3 in HIV-1 infection and sensing: A paradox. Cytokine Growth Factor Rev. 2018, 40, 32–39. [Google Scholar] [CrossRef]
- Zhang, N.; Zhang, P.; Baier, A.; Cova, L.; Hosmane, R.S. Dual inhibition of HCV and HIV by ring-expanded nucleosides containing the 5: 7-fused imidazo [4, 5-e] [1, 3] diazepine ring system. In vitro results and implications. Bioorganic Med. Chem. Lett. 2014, 24, 1154–1157. [Google Scholar] [CrossRef][Green Version]
- Ullah, A.; Aziz, T.; Ullah, N.; Nawaz, T. Molecular mechanisms of Sanguinarine in cancer prevention and treatment. Anti-Cancer Agents Med. Chem. 2022, 22, 3. [Google Scholar] [CrossRef]
- Nguyen, C.N.; Nguyen, K.V.; Eom, S.; Choi, Y.-J.; Kang, L.-J.; Lee, J.; Kim, C.; Lee, S.; Lee, S.-G.; Lee, J.-H. Regulation of p21 expression for anti-apoptotic activity of DDX3 against sanguinarine-induced cell death on intrinsic pathway. Phytomedicine 2019, 65, 153096. [Google Scholar] [CrossRef]
- Kang, L.; Nguyen, K.V.; Eom, S.; Choi, Y.; Nguyen, C.N.; Lee, J.; Kim, C.; Lee, S.; Lee, S.; Lee, J. Stimulating DDX3 expression by serotonin 5-HT receptor 7 through phosphorylation of p53 via the AC-PKA-ERK signaling pathway. J. Cell. Biochem. 2019, 120, 18193–18208. [Google Scholar] [CrossRef]
- Yang, F.; Fang, E.; Mei, H.; Chen, Y.; Li, H.; Li, D.; Song, H.; Wang, J.; Hong, M.; Xiao, W.; et al. Cis-Acting circ-CTNNB1 Promotes β-Catenin Signaling and Cancer Progression via DDX3-Mediated Transactivation of YY1. Cancer Res. 2019, 79, 557–571. [Google Scholar] [CrossRef]
- Nofsinger, J.; Liu, Y.; Simon, J.D. Aggregation of eumelanin mitigates photogeneration of reactive oxygen species. Free Radic. Biol. Med. 2002, 32, 720–730. [Google Scholar] [CrossRef]
- Islam, M.T. A literature-based phyto-pharmacological review on A. conchigera Griff. Orient. Pharm. Exp. Med. 2019, 19, 379–392. [Google Scholar] [CrossRef]
- Lin, J.Y.; Fisher, D.E. Melanocyte biology and skin pigmentation. Nature 2007, 445, 843–850. [Google Scholar] [CrossRef]
- Mastore, M.; Kohler, L.; Nappi, A.J. Production and utilization of hydrogen peroxide associated with melanogenesis and tyrosinase-mediated oxidations of DOPA and dopamine. FEBS J. 2005, 272, 2407–2415. [Google Scholar] [CrossRef]
- Sarangarajan, R.; Apte, S.P.; Ugwu, S.O. Hypoxia-targeted bioreductive tyrosine kinase inhibitors with glutathione-depleting function. Anti-Cancer Drugs 2006, 17, 21–24. [Google Scholar] [CrossRef]
- Dong, Y.; Cao, J.; Wang, H.; Zhang, J.; Zhu, Z.; Bai, R.; Hao, H.; He, X.; Fan, R.; Dong, C. Nitric oxide enhances the sensitivity of alpaca melanocytes to respond to α-melanocyte-stimulating hormone by up-regulating melanocortin-1 receptor. Biochem. Biophys. Res. Commun. 2010, 396, 849–853. [Google Scholar] [CrossRef]
- Chou, T.-H.; Ding, H.-Y.; Hung, W.J.; Liang, C.-H. Antioxidative characteristics and inhibition of α-melanocyte-stimulating hormone-stimulated melanogenesis of vanillin and vanillic acid from Origanum vulgare. Exp. Dermatol. 2009, 19, 742–750. [Google Scholar] [CrossRef]
- Takeuchi, S.; Zhang, W.; Wakamatsu, K.; Ito, S.; Hearing, V.J.; Kraemer, K.H.; Brash, D.E. Melanin acts as a potent UVB photosensitizer to cause an atypical mode of cell death in murine skin. Proc. Natl. Acad. Sci. 2004, 101, 15076–15081. [Google Scholar] [CrossRef]
- Xiao, L.; Matsubayashi, K.; Miwa, N. Inhibitory effect of the water-soluble polymer-wrapped derivative of fullerene on UVA-induced melanogenesis via downregulation of tyrosinase expression in human melanocytes and skin tissues. Arch. Dermatol. Res. 2007, 299, 245–257. [Google Scholar] [CrossRef]
- Kim, H.-E.; Lee, S.-G. Induction of ATP synthase β by H2O2 induces melanogenesis by activating PAH and cAMP/CREB/MITF signaling in melanoma cells. Int. J. Biochem. Cell Biol. 2013, 45, 1217–1222. [Google Scholar] [CrossRef]
- van Voss, M.R.H.; Kammers, K.; Vesuna, F.; Brilliant, J.; Bergman, Y.; Tantravedi, S.; Wu, X.; Cole, R.N.; Holland, A.; van Diest, P.J.; et al. Global Effects of DDX3 Inhibition on Cell Cycle Regulation Identified by a Combined Phosphoproteomics and Single Cell Tracking Approach. Transl. Oncol. 2018, 11, 755–763. [Google Scholar] [CrossRef]
- Limtrakul, P.; Yodkeeree, S.; Thippraphan, P.; Punfa, W.; Srisomboon, J. Anti-aging and tyrosinase inhibition effects of Cassia fistula flower butanolic extract. BMC Complement. Altern. Med. 2016, 16, 497. [Google Scholar] [CrossRef]
- Guo, S.; Zhang, Q. Paeonol protects melanocytes against hydrogen peroxide-induced oxidative stress through activation of Nrf2 signaling pathway. Drug Dev. Res. 2021, 82, 861–869. [Google Scholar] [CrossRef]
- Ko, H.; Kim, M.-M. H2O2 promotes the aging process of melanogenesis through modulation of MITF and Nrf2. Mol. Biol. Rep. 2019, 46, 2461–2471. [Google Scholar] [CrossRef]
- Tief, K.; Hahne, M.; Schmidt, A.; Beermann, F. Tyrosinase, the key enzyme in melanin synthesis, is expressed in murine brain. Eur. J. Biochem. 1996, 241, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Aisa, H.A. Upregulation of Melanogenesis and Tyrosinase Activity: Potential Agents for Vitiligo. Molecules 2017, 22, 1303. [Google Scholar] [CrossRef] [PubMed]
- Botlagunta, M.; Vesuna, F.; Mironchik, Y.; Raman, A.; Lisok, A.; Winnard, P.; Mukadam, S.; Van Diest, P.; Chen, J.H.; Farabaugh, P.; et al. Oncogenic role of DDX3 in breast cancer biogenesis. Oncogene 2008, 27, 3912–3922. [Google Scholar] [CrossRef] [PubMed]
- Bol, G.; Vesuna, F.; Xie, M.; Zeng, J.; Aziz, K.; Gandhi, N.; Levine, A.; Irving, A.; Korz, D.; Tantravedi, S.; et al. Targeting DDX 3 with a small molecule inhibitor for lung cancer therapy. EMBO Mol. Med. 2015, 7, 648–669. [Google Scholar] [CrossRef] [PubMed]
- Hosmane, R.S. Ring-Expanded (Fat) Nucleosides as Broad-Spectrum Anticancer and Antiviral Agents. Curr. Top. Med. Chem. 2002, 2, 1093–1109. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.-C.; Chi, C.-W.; Chau, G.-Y.; Li, F.-Y.; Tsai, Y.-H.; Wu, J.-C.; Lee, Y.-H.W. DDX3, a DEAD box RNA helicase, is deregulated in hepatitis virus-associated hepatocellular carcinoma and is involved in cell growth control. Oncogene 2005, 25, 1991–2003. [Google Scholar] [CrossRef] [PubMed]
- Bol, G.M.; Raman, V.; van der Groep, P.; Vermeulen, J.F.; Patel, A.H.; van der Wall, E.; van Diest, P.J. Expression of the RNA Helicase DDX3 and the Hypoxia Response in Breast Cancer. PLoS ONE 2013, 8, e63548. [Google Scholar] [CrossRef]
- He, T.-Y.; Wu, D.-W.; Lin, P.-L.; Wang, L.; Huang, C.-C.; Chou, M.-C.; Lee, H. DDX3 promotes tumor invasion in colorectal cancer via the CK1ε/Dvl2 axis. Sci. Rep. 2016, 6, 21483. [Google Scholar] [CrossRef]
- Cruciat, C.-M.; Dolde, C.; de Groot, R.E.A.; Ohkawara, B.; Reinhard, C.; Korswagen, H.C.; Niehrs, C. RNA Helicase DDX3 Is a Regulatory Subunit of Casein Kinase 1 in Wnt–β-Catenin Signaling. Science 2013, 339, 1436–1441. [Google Scholar] [CrossRef]
- Chen, H.-H.; Yu, H.-I.; Cho, W.-C.; Tarn, W.-Y. DDX3 modulates cell adhesion and motility and cancer cell metastasis via Rac1-mediated signaling pathway. Oncogene 2014, 34, 2790–2800. [Google Scholar] [CrossRef]
- Stransky, N.; Egloff, A.M.; Tward, A.D.; Kostic, A.D.; Cibulskis, K.; Sivachenko, A.; Kryukov, G.V.; Lawrence, M.S.; Sougnez, C.; McKenna, A.; et al. The Mutational Landscape of Head and Neck Squamous Cell Carcinoma. Science 2011, 333, 1157–1160. [Google Scholar] [CrossRef]
- Chao, C.-H.; Chen, C.-M.; Cheng, P.-L.; Shih, J.-W.; Tsou, A.-P.; Lee, Y.-H.W. DDX3, a DEAD Box RNA Helicase with Tumor Growth–Suppressive Property and Transcriptional Regulation Activity of the p21waf1/cip1 Promoter, Is a Candidate Tumor Suppressor. Cancer Res. 2006, 66, 6579–6588. [Google Scholar] [CrossRef]
- van Voss, M.H.; Vesuna, F.; Bol, G.M.; Meeldijk, J.; Raman, A.; Offerhaus, G.J.; Buerger, H.; Patel, A.H.; van der Wall, E.; van Diest, P.J.; et al. Nuclear DDX3 expression predicts poor outcome in colorectal and breast cancer. OncoTargets Ther. 2017, ume 10, 3501–3513. [Google Scholar] [CrossRef]
- Cho, H.; Kim, O.; Lee, Y.; Kang, L.-J.; Nguyen, C.N.; Ishihara, A.; Kim, H.-E. Feruloylserotonin inhibits hydrogen peroxide-induced melanogenesis and apoptosis in B16F10 and SK-Mel-2 melanoma cells. Biochem. Biophys. Res. Commun. 2017, 491, 973–979. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, I.S.; Dong, Y.; Lee, I.-S.; Kim, J.S.; Kim, J.-S.; Woo, J.-T.; Cha, B.-Y. Melanogenesis-Inducing Effect of Cirsimaritin through Increases in Microphthalmia-Associated Transcription Factor and Tyrosinase Expression. Int. J. Mol. Sci. 2015, 16, 8772–8788. [Google Scholar] [CrossRef]
- Miao, X.; Yang, Z.-L.; Xiong, L.; Zou, Q.; Yuan, Y.; Li, J.; Liang, L.; Chen, M.; Chen, S. Nectin-2 and DDX3 are biomarkers for metastasis and poor prognosis of squamous cell/adenosquamous carcinomas and adenocarcinoma of gallbladder. Int. J. Clin. Exp. Pathol. 2013, 6, 179–190. [Google Scholar]
- Robinson, G.; Parker, M.; Kranenburg, T.A.; Lu, C.; Chen, X.; Ding, L.; Phoenix, T.N.; Hedlund, E.; Wei, L.; Zhu, X.; et al. Novel mutations target distinct subgroups of medulloblastoma. Nature 2012, 488, 43–48. [Google Scholar] [CrossRef]
- Ojha, J.; Secreto, C.R.; Rabe, K.G.; Van Dyke, D.L.; Kortum, K.M.; Slager, S.L.; Shanafelt, T.D.; Fonseca, R.; Kay, N.E.; Braggio, E. Identification of recurrent truncated DDX3X mutations in chronic lymphocytic leukaemia. Br. J. Haematol. 2014, 169, 445–448. [Google Scholar] [CrossRef]
- Chen, W.-J.; Wang, W.-T.; Tsai, T.-Y.; Li, H.-K.; Lee, Y.-H.W. DDX3 localizes to the centrosome and prevents multipolar mitosis by epigenetically and translationally modulating p53 expression. Sci. Rep. 2017, 7, 9411. [Google Scholar] [CrossRef]
- Lai, M.-C.; Chang, W.-C.; Shieh, S.-Y.; Tarn, W.-Y. DDX3 Regulates Cell Growth through Translational Control of Cyclin E1. Mol. Cell. Biol. 2010, 30, 5444–5453. [Google Scholar] [CrossRef]
- Wu, D.-W.; Lin, P.-L.; Cheng, Y.-W.; Huang, C.-C.; Wang, L.; Lee, H. DDX3 enhances oncogenic KRAS-induced tumor invasion in colorectal cancer via the β-catenin/ZEB1 axis. Oncotarget 2016, 7, 22687–22699. [Google Scholar] [CrossRef] [PubMed]
- Pugh, T.J.; Weeraratne, S.D.; Archer, T.C.; Krummel, D.A.P.; Auclair, D.; Bochicchio, J.; Carneiro, M.O.; Carter, S.L.; Cibulskis, K.; Erlich, R.L.; et al. Medulloblastoma exome sequencing uncovers subtype-specific somatic mutations. Nature 2012, 488, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.T.W.; Jaeger, N.; Kool, M.; Zichner, T.; Hutter, B.; Sultan, M.; Cho, Y.-J.; Pugh, T.; Hovestadt, V.; Stütz, A.M.; et al. Dissecting the genomic complexity underlying medulloblastoma. Nature 2012, 488, 100–105. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eom, S.; Lee, S.; Lee, J.; Yeom, H.D.; Lee, S.-G.; Lee, J. DDX3 Upregulates Hydrogen Peroxide-Induced Melanogenesis in Sk-Mel-2 Human Melanoma Cells. Molecules 2022, 27, 7010. https://doi.org/10.3390/molecules27207010
Eom S, Lee S, Lee J, Yeom HD, Lee S-G, Lee J. DDX3 Upregulates Hydrogen Peroxide-Induced Melanogenesis in Sk-Mel-2 Human Melanoma Cells. Molecules. 2022; 27(20):7010. https://doi.org/10.3390/molecules27207010
Chicago/Turabian StyleEom, Sanung, Shinhui Lee, Jiwon Lee, Hye Duck Yeom, Seong-Gene Lee, and Junho Lee. 2022. "DDX3 Upregulates Hydrogen Peroxide-Induced Melanogenesis in Sk-Mel-2 Human Melanoma Cells" Molecules 27, no. 20: 7010. https://doi.org/10.3390/molecules27207010
APA StyleEom, S., Lee, S., Lee, J., Yeom, H. D., Lee, S.-G., & Lee, J. (2022). DDX3 Upregulates Hydrogen Peroxide-Induced Melanogenesis in Sk-Mel-2 Human Melanoma Cells. Molecules, 27(20), 7010. https://doi.org/10.3390/molecules27207010





