The Role of Strontium in CeNiO3 Nano-Crystalline Perovskites for Greenhouse Gas Mitigation to Produce Syngas
Abstract
:1. Introduction
2. Results
2.1. Characterization of Fresh Catalysts
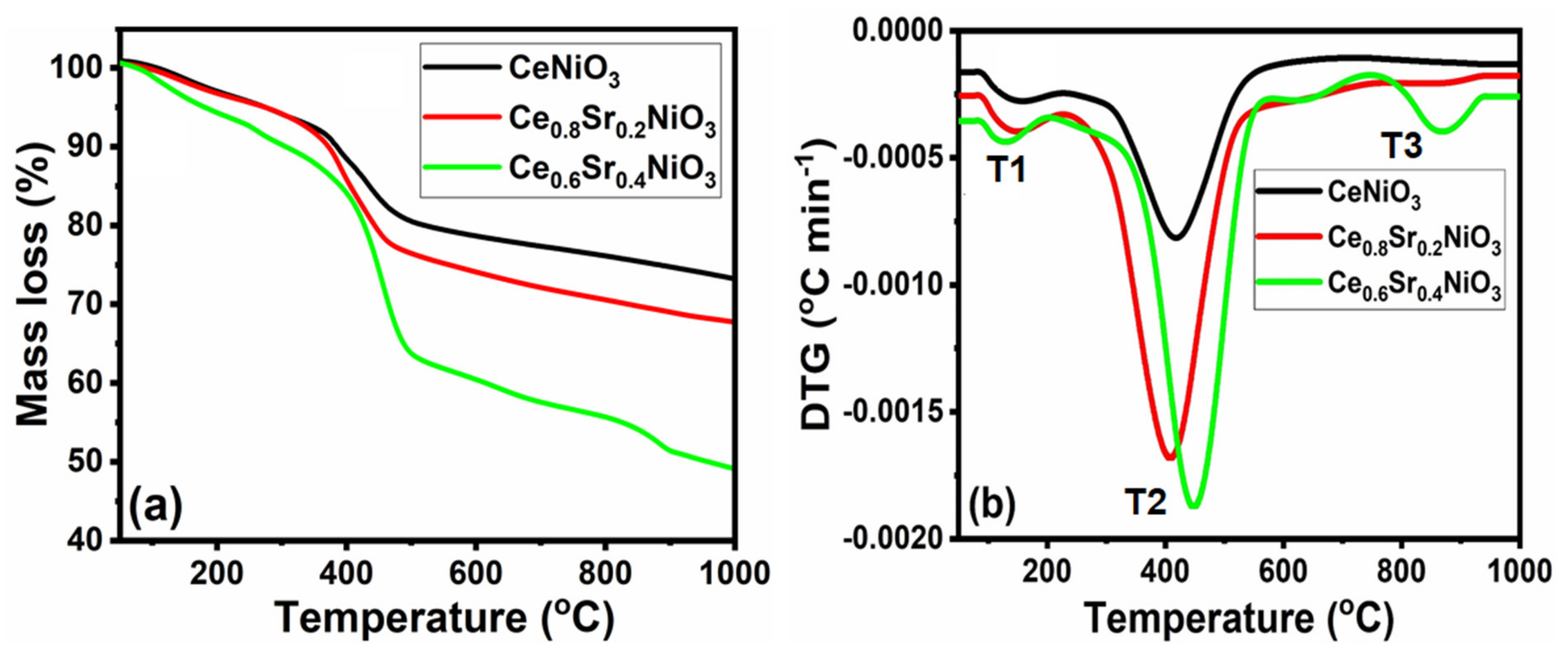
2.1.1. Thermal Decomposition of Precursors
2.1.2. X-ray Diffraction (XRD)
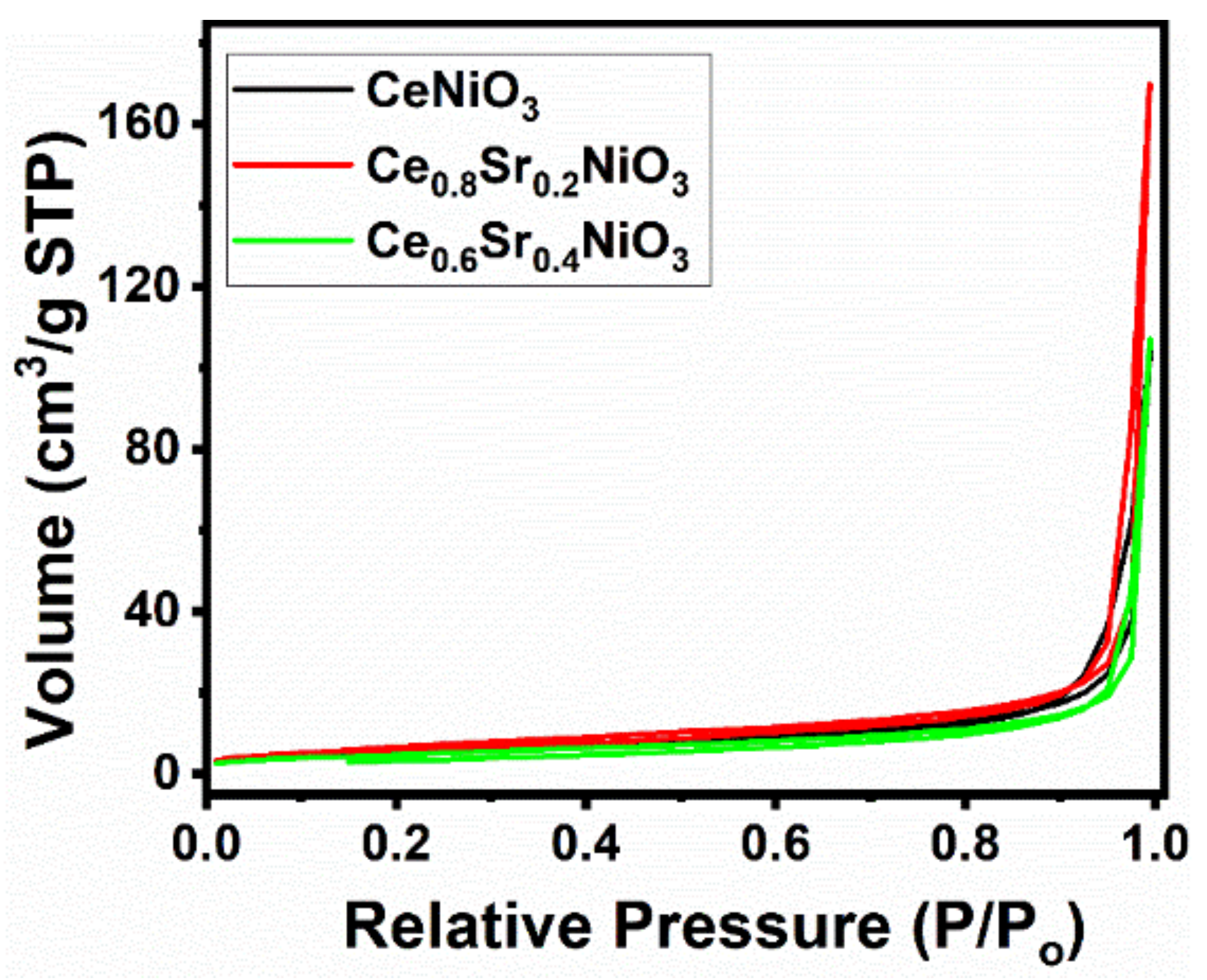
2.1.3. Textural Properties and Elemental Analysis
2.1.4. Morphological Study (TEM) of Fresh and Reduced Perovskites
2.1.5. Temperature-Programmed Reduction (TPR)
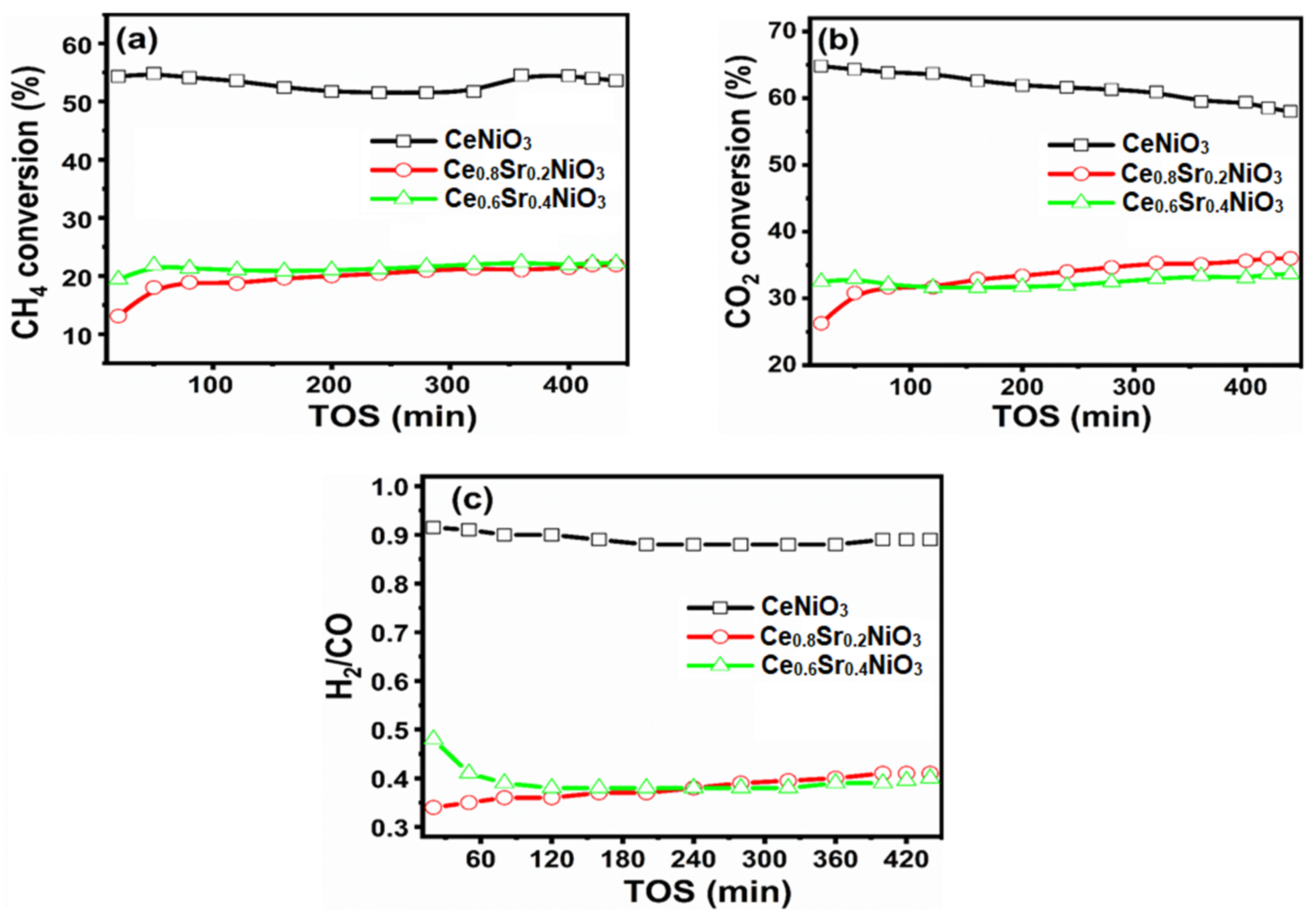
2.2. Catalytic Performances
2.3. Characterization of Spent Perovskites
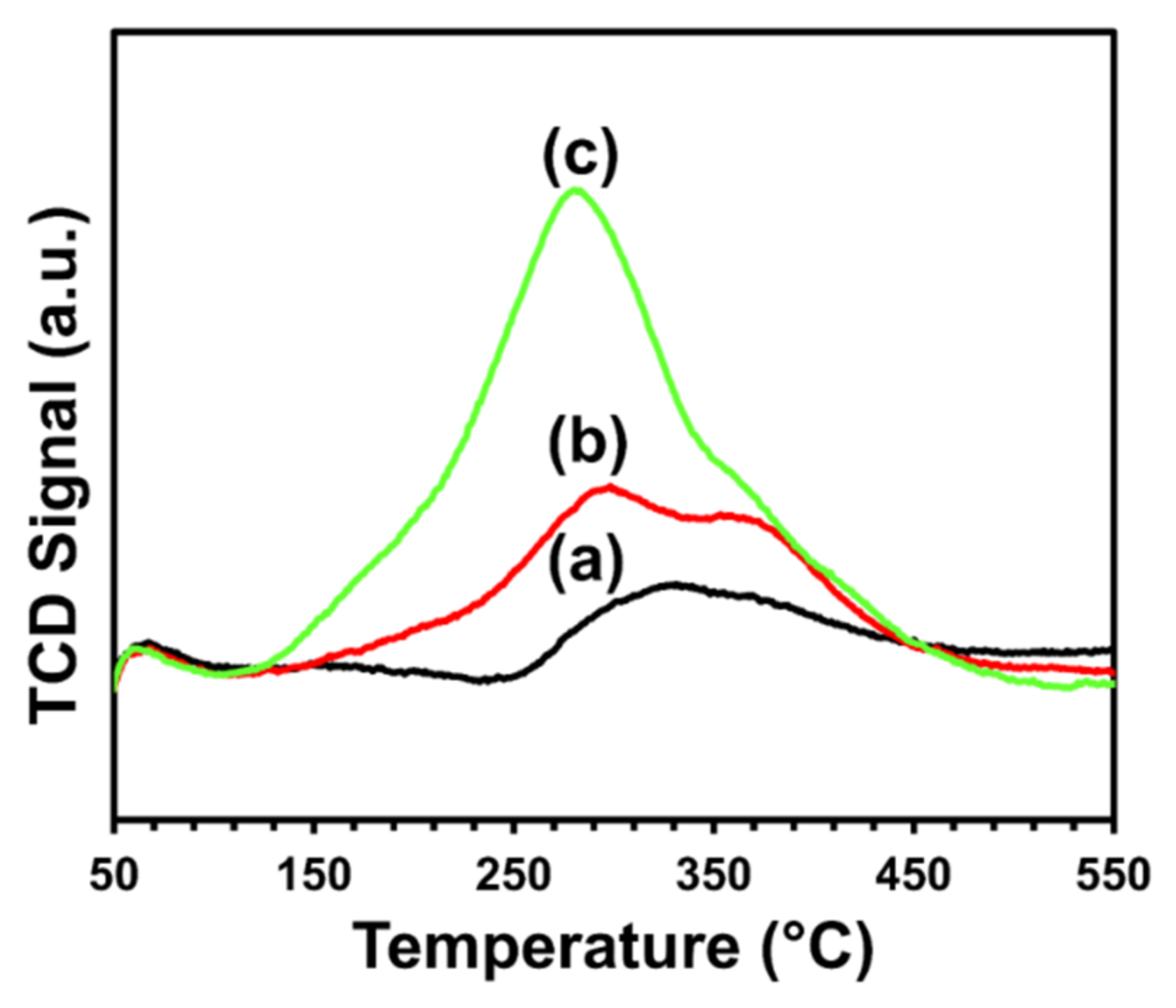
2.3.1. Temperature-Programmed Oxidation (TPO)
2.3.2. Transition Electron Microscopy (TEM) and Elemental Analysis
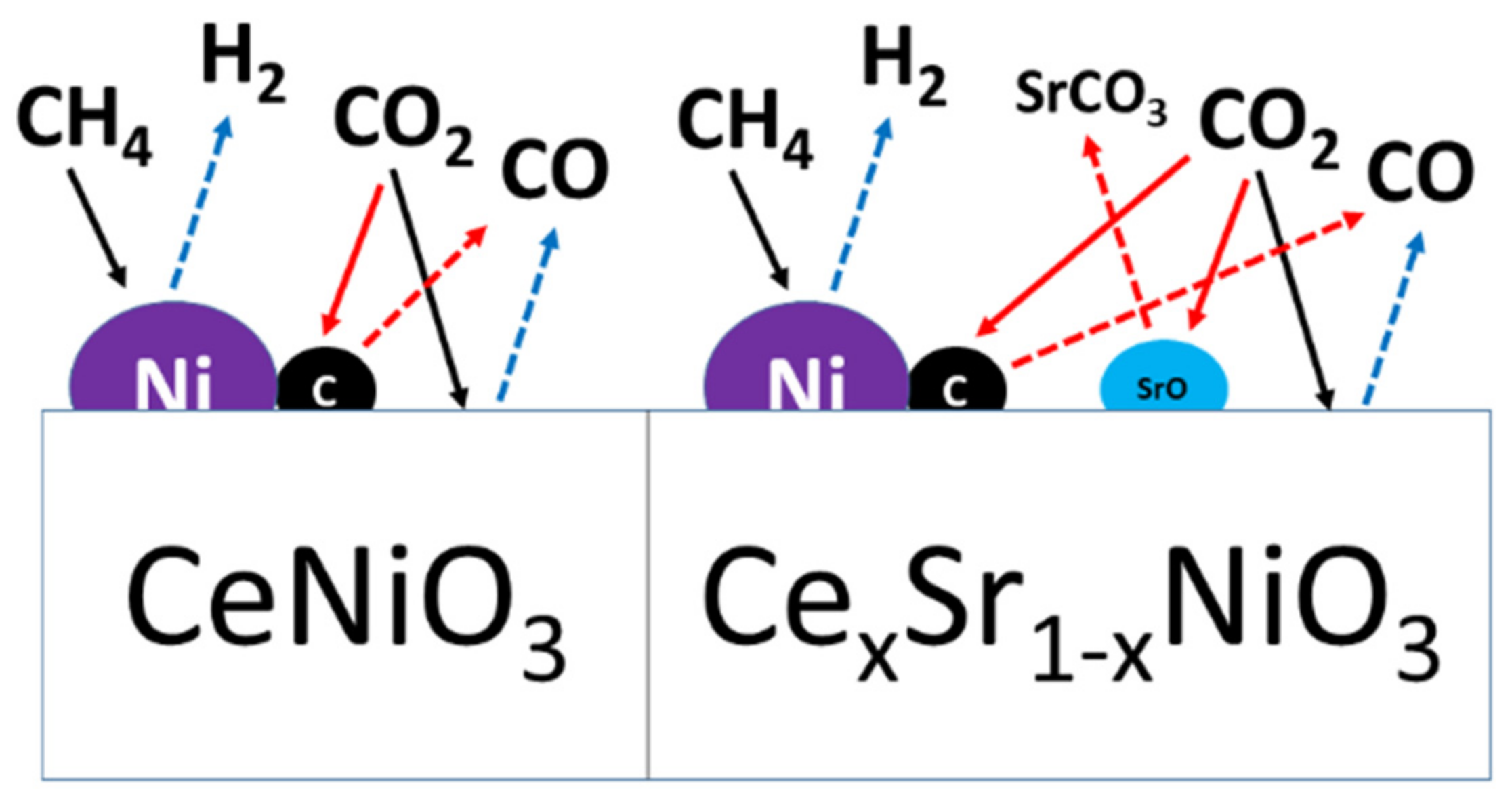
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Wang, Y.; Yao, L.; Wang, S.; Mao, D.; Hu, C. Low-temperature catalytic CO2 dry reforming of methane on Ni-based catalysts: A review. Fuel Process. Technol. 2018, 169, 199–206. [Google Scholar] [CrossRef]
- Li, H.; He, Y.; Shen, D.; Cheng, S.; Wang, J.; Liu, H.; Xing, C.; Shan, S.; Lu, C.; Yang, R. Design an in-situ reduction of Ni/C-SiO2 catalyst and new insight into pretreatment effect for CH4-CO2 reforming reaction. Int. J. Hydrogen Energy 2017, 42, 10844–10853. [Google Scholar] [CrossRef]
- Al-Fatesh, A.S.; Arafat, Y.; Atia, H.; Ibrahim, A.A.; Ha, Q.L.M.; Schneider, M.; M-Pohl, M.; Fakeeha, A.H. CO2-reforming of methane to produce syngas over Co-Ni/SBA-15 catalyst: Effect of support modifiers (Mg, La and Sc) on catalytic stability. J. CO2 Util. 2017, 21, 395–404. [Google Scholar] [CrossRef]
- Ibrahim, A.A.; Fakeeha, A.H.; Al-Fatesh, A.S. Enhancing hydrogen production by dry reforming process with strontium promoter. Int. J. Hydrogen Energy 2014, 39, 1680–1687. [Google Scholar] [CrossRef]
- Li, D.; Lu, M.; Xu, S.; Chen, C.; Zhan, Y.; Jiang, L. Preparation of supported Co catalysts from Co-Mg-Al layered double hydroxides for carbon dioxide reforming of methane. Int. J. Hydrogen Energy 2017, 42, 5063–5071. [Google Scholar] [CrossRef]
- Al-Fatesh, A.S.; Arafat, Y.; Ibrahim, A.A.; Atia, H.; Fakeeha, A.H.; Armbruster, U.; Abasaeed, A.E.; Frusteri, F. Evaluation of Co-Ni/Sc-SBA–15 as a novel coke resistant catalyst for syngas production via CO2 reforming of methane. Appl. Catal. A Gen. 2018, 567, 102–111. [Google Scholar] [CrossRef]
- Rynkowski, J.; Samulkiewicz, P.; Ladavos, A.K.; Pomonis, P.J. Catalytic performance of reduced La2−xSrxNiO4 perovskite-like oxides for CO2 reforming of CH4. Appl. Catal. A Gen. 2004, 263, 1–9. [Google Scholar] [CrossRef]
- El Hassan, N.; Kaydouh, M.N.; Geagea, H.; El Zein, H.; Jabbour, K.; Casale, S.; El Zakhem, H.; Massiani, P. Low temperature dry reforming of methane on rhodium and cobalt based catalysts: Active phase stabilization by confinement in mesoporous SBA-15. Appl. Catal. A Gen. 2016, 520, 114–121. [Google Scholar] [CrossRef] [Green Version]
- Khan, W.U.; Li, X.; Baharudin, L.; Yip, A.C.K. Copper-promoted cobalt/titaniananorod catalyst for CO hydrogenation to hydrocarbons. Catal. Lett. 2021, 151, 2492–2501. [Google Scholar] [CrossRef]
- Khan, W.U.; Baharudin, L.; Choi, J.; Yip, A.C.K. Recent progress in CO hydrogenation over bimetallic catalysts for higher alcohol synthesis. ChemCatChem 2021, 13, 111–120. [Google Scholar] [CrossRef]
- Khan, W.U.; Chen, S.S.; Tsang, D.C.W.; Hu, X.; Lam, F.L.Y.; Yip, A.C.K. Catalytically active interfaces in titaniananorod-supported copper catalysts for CO oxidation. Nano Res. 2020, 13, 533–542. [Google Scholar] [CrossRef]
- Horn, R.; Schlögl, R. Methane Activation by Heterogeneous Catalysis. Catal. Lett. 2015, 45, 23–39. [Google Scholar] [CrossRef] [Green Version]
- Rostrup-Nielsen, J.R.; Sehested, J.; Nørskov, J.K. Hydrogen and synthesis gas by steam- and CO2 reforming. Adv. Catal. 2002, 47, 65–139. [Google Scholar] [CrossRef]
- Kang, D.; Yu, J.; Ma, W.; Zheng, M.; He, Y.; Li, P. Synthesis of Cu/Ni-La0.7Sr0.3Cr0.5Mn0.5O3–δ and its catalytic performance on dry methane reforming. J. Rare Earth 2019, 37, 585–593. [Google Scholar] [CrossRef]
- Wang, F.; Han, K.; Yu, W.; Zhao, L.; Wang, Y.; Wang, X.; Yu, H.; Shi, W. Low Temperature CO2 Reforming with Methane Reaction over CeO2-Modified Ni@SiO2 Catalysts. ACS Appl. Mater. Interfaces 2020, 12, 35022–35034. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Zhao, L.; Wang, F.; Xu, L.; Yu, H.; Cui, Y.; Zhang, J.; Shi, W. Effect of calcination temperature on performance of Ni@SiO2 catalyst in methane dry reforming. Ind. Eng. Chem. Res. 2020, 59, 13370–13379. [Google Scholar] [CrossRef]
- Chein, R.-Y.; Fung, W.-Y. Syngas production via dry reforming of methane over CeO2 modified Ni/Al2O3 catalysts. Int. J. Hydrogen Energy 2019, 44, 14303–14315. [Google Scholar] [CrossRef]
- Ma, Q.; Guo, L.; Fang, Y.; Li, H.; Zhang, J.; Zhao, T.-S.; Yang, G.; Yoneyam, Y.; Tsubaki, N. Combined methane dry reforming and methane partial oxidization for syngas production over high dispersion Ni based mesoporous catalyst. Fuel Process. Technol. 2019, 188, 98–104. [Google Scholar] [CrossRef]
- Abdullah, B.; Ghani, N.A.A.; Vo, D.-V.N. Recent advances in dry reforming of methane over Ni-based catalysts. J. Clean. Prod. 2017, 162, 170–185. [Google Scholar] [CrossRef] [Green Version]
- Ali, S.; Khader, M.M.; Almarri, M.J.; Abdelmoneim, A.G. Ni-based nano-catalysts for the dry reforming of methane. Catal. Today 2020, 343, 26–37. [Google Scholar] [CrossRef]
- Shen, J.; Reule, A.A.C.; Semagina, N. Ni/MgAl2O4 catalyst for low-temperature oxidative dry methane reforming with CO2. Int. J. Hydrogen Energy 2019, 44, 4616–4629. [Google Scholar] [CrossRef]
- Wanga, F.; Hana, B.; Zhanga, L.; Xub, L.; Yuc, H.; Shi, W. CO2 reforming with methane over small-sized Ni@SiO2 catalysts with unique features of sintering-free and low carbon. Appl. Catal. B Environ. 2018, 235, 26–35. [Google Scholar] [CrossRef]
- Han, K.; Yu, W.; Xu, L.; Deng, Z.; Yu, H.; Wang, F. Reducing carbon deposition and enhancing reaction stability by ceria for methane dry reforming over Ni@SiO2@CeO2 catalyst. Fuel 2021, 291, 120182. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Siakavelas, G.; Papageridis, K.N.; Baklavaridis, A.; Tzounis, L.; Avraam, D.G.; Goula, M.A. Syngas production via the biogas dry reforming reaction over nickel supported on modified with CeO2 and/or La2O3 alumina catalysts. J. Nat. Gas Sci. Eng. 2016, 31, 164–183. [Google Scholar] [CrossRef]
- Goula, M.A.; Charisiou, N.D.; Siakavelas, G.; Tzounis, L.; Tsiaoussis, I.; Panagiotopoulou, P.; Goula, G.; Yentekakis, I.V. Syngas production via the biogas dry reforming reaction over Ni supported on zirconia modified with CeO2 or La2O3 catalysts. Int. J. Hydrogen Energy 2017, 42, 13724–13740. [Google Scholar] [CrossRef]
- Makri, M.M.; Vasiliades, M.A.; Petallidou, K.C.; Efstathiou, A.M. Effect of support composition on the origin and reactivity of carbon formed during dry reforming of methane over 5 wt% Ni/Ce1−xMxO2−δ (M= Zr4+, Pr3+) catalysts. Catal. Today 2016, 259, 150–164. [Google Scholar] [CrossRef]
- Summa, P.; Swirk, K.; Wierzbicki, D.; Motak, M.; Alxneit, I.; Rønning, M.; Da Costa, P. Co-Precipitated Ni-Mg-Al Hydrotalcite-Derived Catalyst Promoted with Vanadium for CO2 Methanation. Molecules 2021, 26, 6506. [Google Scholar] [CrossRef]
- Świrka, K.; Gálvez, M.E.; Motak, M.; Grzybek, T.; Rønning, M.; Da Costa, P. Dry reforming of methane over Zr- and Y-modified Ni/Mg/Al double-layered Hydroxides. Catal. Commun. 2018, 117, 26–32. [Google Scholar] [CrossRef]
- Świrk, K.; Gálvez, M.E.; Motak, M.; Grzybek, T.; Rønning, M.; Da Costa, P. Yttrium promoted Ni-based double-layered hydroxides for dry methane Reforming. J. CO2 Util. 2018, 27, 247–258. [Google Scholar] [CrossRef]
- Swirk, K.; Rønning, M.; Motak, M.; Beaunier, P.; Da Costa, P.; Grzybek, T. Ce- and Y-Modified Double-Layered Hydroxides as Catalysts for Dry Reforming of Methane: On the Effect of Yttrium Promotion. Catalysts 2019, 9, 56. [Google Scholar] [CrossRef] [Green Version]
- Jacobson, A.J. Materials for Solid Oxide Fuel Cells. Chem. Mater. 2010, 22, 660–674. [Google Scholar] [CrossRef]
- Kim, W.Y.; Jang, J.S.; Ra, E.C.; Kim, K.Y.; Kim, E.H.; Lee, J.S. Reduced perovskite LaNiO3 catalysts modified with Co and Mn for low coke formation in dry reforming of methane. Appl. Catal. A Gen. 2019, 575, 198–203. [Google Scholar] [CrossRef]
- Wei, T.; Jia, L.; Zheng, H.; Chi, B.; Pu, J.; Li, J. LaMnO3-based perovskite with in-situ exsolved Ni nanoparticles: A highly active, performance stable and coking resistant catalyst for CO2 dry reforming of CH4. Appl. Catal. A Gen. 2018, 564, 199–207. [Google Scholar] [CrossRef]
- Yang, Q.; Liu, G.; Yuan Liu, Y. Perovskite-type oxides as the catalyst precursors for preparing supported metallic nanocatalysts: A review. Ind. Eng. Chem. Res. 2018, 57, 1–17. [Google Scholar] [CrossRef]
- Bhattar, S.; Abedin, M.A.; Kanitkar, S.; Spivey, J.J. A review on dry reforming of methane over perovskite derived catalysts. Catal. Today 2021, 365, 2–23. [Google Scholar] [CrossRef]
- Khalesi, A.; Arandiyan, H.R.; Parvari, M. Production of Syngas by CO2 Reforming on MxLa1−xNi0.3Al0.7O3−d (M = Li, Na, K). Ind. Eng. Chem. Res. 2008, 47, 5892–5898. [Google Scholar] [CrossRef]
- Pérez-Camacho, M.N.; Abu-Dahrieh, J.; Goguet, A.; Sun, K.; Rooney, D. Self-cleaning perovskite-type catalysts for the dry reforming of methane. Chin. J. Catal. 2014, 35, 1337–1346. [Google Scholar] [CrossRef]
- Ruocco, C.; Caprariis, B.D.; Palma, V.; Petrullo, A.; Ricca, A.; Scarsella, M.; Filippis, P.D. Methane dry reforming on Ru perovskites, AZrRuO3: Influence of preparation method and substitution of A cation with alkaline earth metals. J. CO2 Util. 2019, 30, 222–231. [Google Scholar] [CrossRef]
- Wang, H.; Dong, X.; Zhao, T.; Yu, H.; Li, M. Dry reforming of methane over bimetallic Ni-Co catalyst prepared from La(CoxNi1-x)0.5Fe0.5O3 perovskite precursor: Catalytic activity and coking resistance. Appl. Catal. B Environ. 2009, 245, 302–313. [Google Scholar] [CrossRef]
- Wang, F.; Xu, L.; Shi, W. Syngas production from CO2 reforming with methane over core-shell Ni@SiO2 catalysts. J. CO2 Util. 2016, 16, 318–327. [Google Scholar] [CrossRef]
- Han, B.; Wang, F.; Zhang, L.; Wang, Y.; Fan, W.; Xu, L.; Yu, H.; Li, Z. Syngas production from methane steam reforming and dry reforming reactions over sintering-resistant Ni@SiO2 catalyst. Res. Chem. Intermed. 2020, 46, 1735–1748. [Google Scholar] [CrossRef]
- Dehghani, F.; Ayatollahi, S.; Bahadorikhalili, S.; Esmaeilpour, M. Synthesis and Characterization of Mixed–Metal Oxide Nanoparticles (CeNiO3, CeZrO4, CeCaO3) and Application in Adsorption and Catalytic Oxidation–Decomposition of Asphaltenes with Different Chemical Structures. Pet. Chem. 2020, 60, 731–743. [Google Scholar] [CrossRef]
- Harikrishnan, M.P.; Mary, A.J.C.; Bose, A.C. Electrochemical performance of ANiO3 (A= La, Ce) perovskite oxide material and its device performance for supercapattery application. Electrochim. Acta 2020, 362, 137095. [Google Scholar] [CrossRef]
- Chen, J.; He, Z.; Li, G.; An, T.; Shi, H.; Li, Y. Visible-light-enhanced photothermocatalytic activity of ABO3-type perovskites for the decontamination of gaseous styrene. Appl. Catal. B Environ. 2017, 209, 146–154. [Google Scholar] [CrossRef] [Green Version]
- Hu, Q.; Yue, B.; Shao, H.; Yang, F.; Wang, J.; Wang, Y.; Liu, J. Facile syntheses of cerium-based CeMO3 (M = Co, Ni, Cu) perovskite nanomaterials for high-performance supercapacitor electrodes. J. Mater. Sci. 2020, 55, 8421–8434. [Google Scholar] [CrossRef]
- García de la Cruz, R.M.; Falcón, H.; Peña, M.A.; Fierro, J.L.G. Role of bulk and surface structures of La1−xSrxNiO3 perovskite-type oxides in methane combustion. Appl. Catal. B Environ. 2001, 33, 45–55. [Google Scholar] [CrossRef]
- Wang, Y.; Cui, X.; Li, Y.; Shu, Z.; Chen, H.; Shi, J. A simple co-nanocasting method to synthesize high surface area mesoporous LaCoO3 oxides for CO and NO oxidations. Microporous Mesoporous Mater. 2013, 176, 8–15. [Google Scholar] [CrossRef]
- Wei, Y.; Zhao, Z.; Jiao, J.; Liu, J.; Duan, A.; Jiang, G. Facile synthesis of three-dimensionally ordered macroporous LaFeO3-supported gold nanoparticle catalysts with high catalytic activity and stability for soot combustion. Catal. Today 2015, 245, 37–45. [Google Scholar] [CrossRef]
- Sudhakaran, M.S.P.; Hossain, M.M.; Gnanasekaran, G.; Mok, Y.S. Dry Reforming of Propane over γ-Al2O3 and Nickel Foam Supported Novel SrNiO3 Perovskite Catalyst. Catalysts 2019, 9, 68. [Google Scholar]
- Pino, L.; Italiano, C.; Lagana, M.; Vita, A.; Recupero, V. Kinetic study of the methane dry (CO2) reforming reaction over the Ce0.70La0.20Ni0.10O2−δ catalyst. Catal. Sci. Technol. 2020, 10, 2652–2662. [Google Scholar] [CrossRef]
- Ostrovskii, N.M. Problems in the Study of Catalyst Deactivation Kinetics. Kinet. Catal. 2005, 46, 693–704. [Google Scholar] [CrossRef]
- Moral, A.; Reyero, I.; Alfaro, C.; Bimbela, F.; Gandía, L.M. Syngas production by means of biogas catalytic partial oxidation and dry reforming using Rh-based catalysts. Catal. Today 2018, 299, 280–288. [Google Scholar] [CrossRef]
- Verykios, X. Mechanistic aspects of the reaction of CO2 reforming of methane over Rh/Al2O3 catalyst. Appl. Catal. A Gen. 2003, 255, 101–111. [Google Scholar] [CrossRef]
- Choudhary, V.R.; Uphade, B.S.; Belhekar, A.A. Oxidative Conversion of Methane to Syngas over LaNiO3 Perovskite with or without Simultaneous Steam and CO2 Reforming Reactions: Influence of Partial Substitution of La and Ni. J. Catal. 1996, 163, 312–318. [Google Scholar] [CrossRef]
- Lima, S.M.; Assaf, J.M.; Peña, M.A.; Fierro, J.L.G. Structural features of La1−xCexNiO3 mixed oxides and performance for the dry reforming of methane. Appl. Catal. A Gen. 2006, 311, 94–104. [Google Scholar] [CrossRef]
- Valderrama, G.; Goldwasser, M.R.; de Navarro, C.U.; Tatibouët, J.M.; Barrault, J.; Batiot-Dupeyrat, C.; Martínez, F. Dry reforming of methane over Ni perovskite type oxides. Catal. Today 2005, 107–108, 785–791. [Google Scholar] [CrossRef]
- Das, S.; Bhattar, S.; Liu, L.; Wang, Z.; Xi, S.; Spivey, J.J.; Kawi, S. Effect of Partial Fe Substitution in La0.9Sr0.1NiO3 Perovskite-derived Catalysts on Reaction Mechanism of Methane Dry Reforming. ACS Catal. 2020, 10, 12466–12486. [Google Scholar] [CrossRef]
- Komarala, E.P.; Komissarov, I.; Rosen, B.A. Effect of Fe and Mn Substitution in LaNiO3 on Exsolution, Activity, and Stability for Methane Dry Reforming. Catalysts 2020, 10, 27. [Google Scholar] [CrossRef] [Green Version]
- Mousavi, M.; Pour, A.N.; Gholizadeh, M.; Mohammadi, A.; Shahri, S.M.K. Dry Reforming of Methane by La0.5Sr0.5NiO3 Perovskite Oxides: Influence of Preparation Method on Performance and Structural Features of the Catalysts. J. Chem. Technol. Biotechnol. 2020, 95, 2911–2920. [Google Scholar] [CrossRef]
- Voorhoeve, R.J.H. 5—Perovskite-Related Oxides as Oxidation—Reduction Catalysts. In Advanced Materials in Catalysis; Burton, J.J., Garten, R.L., Eds.; Academic Press: New York, NY, USA, 1977; pp. 129–180. [Google Scholar]
- Messaoudi, H.; Thomas, S.; Djaidja, A.; Slyemi, S.; Barama, A. Study of LaxNiOy and LaxNiOy/MgAl2O4 catalysts in dry reforming of methane. J. CO2 Util. 2018, 24, 40–49. [Google Scholar] [CrossRef]



| Perovskites | Ni/Ce/Sr Content (%) a | SBET (m2/g) | Pore Volume (cm3/g) | Pore size (nm) | Deactivation Factor (%) | Coke (wt%) d | |
|---|---|---|---|---|---|---|---|
| Fresh | Used | ||||||
| CeNiO3 | 49.8/50.1/- | 50.2/49.6/- | 20.7 | 0.162 | 30.1 | 7.7 b (0.88) c | 9.1 |
| Ce0.8Sr0.2NiO3 | 51.1/44.1/4.7 | 50.9/43.8/5.3 | 25.6 | 0.261 | 40.8 | −62.6 b (1.71) c | 4.7 |
| Ce0.6Sr0.4NiO3 | 50.7/32.1/17.2 | 51/31.1/17.8 | 17.3 | 0.164 | 38.1 | −13.4 b (1.15) c | 2.1 |
| Catalyst | Reaction Temp. (°C)/GHSV (L/h/gcat) | Highest CH4 Conversion (%) | %DF a | TOS (h) | Ref. |
|---|---|---|---|---|---|
| CeNiO3 | 750/72 | 32 | 12.9 | ~26 | [55] |
| La0.6Sr0.4NiO3 | 700/- | 85 | - | 20 | [56] |
| La0.9Sr0.1NiO3 | 700/18 | 70 | 3.6 | 8 | [57] |
| LaNi0.8Fe0.2O3 | 800/13.7 | 65 | 7.7 | 35 | [58] |
| La0.5Sr0.5NiO3 | 750/18 | 69 | 47.8 | 24 | [59] |
| CeNiO3 | 700/84 | 55 | 7.7 | 8 | This work |
| Ce0.8Sr0.2NiO3 | 22.5 | −62.6 | |||
| Ce0.6Sr0.4NiO3 | 22.5 | −13.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, N.; Wahab, R.; Manoharadas, S.; Alrayes, B.F.; Alam, M.; Alharthi, F.A. The Role of Strontium in CeNiO3 Nano-Crystalline Perovskites for Greenhouse Gas Mitigation to Produce Syngas. Molecules 2022, 27, 356. https://doi.org/10.3390/molecules27020356
Ahmad N, Wahab R, Manoharadas S, Alrayes BF, Alam M, Alharthi FA. The Role of Strontium in CeNiO3 Nano-Crystalline Perovskites for Greenhouse Gas Mitigation to Produce Syngas. Molecules. 2022; 27(2):356. https://doi.org/10.3390/molecules27020356
Chicago/Turabian StyleAhmad, Naushad, Rizwan Wahab, Salim Manoharadas, Basel F. Alrayes, Manawwer Alam, and Fahad A. Alharthi. 2022. "The Role of Strontium in CeNiO3 Nano-Crystalline Perovskites for Greenhouse Gas Mitigation to Produce Syngas" Molecules 27, no. 2: 356. https://doi.org/10.3390/molecules27020356
APA StyleAhmad, N., Wahab, R., Manoharadas, S., Alrayes, B. F., Alam, M., & Alharthi, F. A. (2022). The Role of Strontium in CeNiO3 Nano-Crystalline Perovskites for Greenhouse Gas Mitigation to Produce Syngas. Molecules, 27(2), 356. https://doi.org/10.3390/molecules27020356








