Current Research of Phytochemical, Medicinal and Non-Medicinal Uses of Uncaria gambir Roxb.: A Review
Abstract
1. Introduction
2. Ethnobotany of Uncaria gambir Roxb.
3. Phytochemical of Uncaria gambir Roxb.
3.1. Flavonoids
3.2. Alkaloids
3.3. Other Compounds
4. Uncaria Gambir Roxb. Uses
4.1. Medicinal Uses
4.1.1. Antioxidant
4.1.2. Antibacteria
4.1.3. Anti-Helmintic
4.1.4. Anticancer
4.1.5. Antifungal
4.1.6. Anti-Inflammatory
4.1.7. Anti-Hyperglycemic
4.1.8. Anti-Hyperuricemia
4.1.9. Anti-Lipid Peroxidation Activity
4.1.10. Antihyperlipidemic and Atherosclerosis
4.2. Non-Medicinal Uses
4.2.1. Adsorbent of Dye
4.2.2. Corrosion Inhibition
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- WHO Traditional Medicine Strategy: 2014–2023. Available online: https://www.who.int/publications/i/item/9789241506096 (accessed on 5 May 2022).
- Vandebroek, I.; Balick, M.J.; Ososki, A.; Kronenberg, F.; Yukes, J.; Wade, C.; Jiménez, F.; Peguero, B.; Castillo, D. The importance of botellas and other plant mixtures in Dominican traditional medicine. J. Ethnopharmacol. 2010, 128, 20–41. [Google Scholar] [CrossRef] [PubMed]
- Talib, W.H.; Mahasneh, A.M. Antiproliferative activity of plant extracts used against cancer in traditional medicine. Sci. Pharm. 2010, 78, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Ginovyan, M.; Petrosyan, M.; Trchounian, A. Antimicrobial activity of some plant materials used in Armenian traditional medicine. BMC Complement. Altern. Med. 2017, 17, 50. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.; Salim, F. Oxindole Alkaloids of Uncaria (Rubiaceae, Subfamily Cinchonoideae): A Review on Its Structure, Properties, and Bioactivities. Stud. Nat. Prod. Chem. 2015, 45, 485–525. [Google Scholar]
- Heitzman, M.E.; Neto, C.C.; Winiarz, E.; Vaisberg, A.J.; Hammond, G.B. Ethnobotany, phytochemistry and pharmacology of Uncaria (Rubiaceae). Phytochemistry 2005, 66, 5–29. [Google Scholar] [CrossRef]
- Rauf, A.; Siregar, A.Z. The condition of Uncaria gambir Roxb. as one of important medicinal plants in North Sumatra Indonesia. Procedia Chem. 2015, 14, 3–10. [Google Scholar] [CrossRef][Green Version]
- Erizon, N.; Jasman, J.; Wakhinuddin, W. Manufacture of Gambir Leaf Press Impac System in Efforts to Increase Productivity of Gambir Farmer. Teknomekanik 2019, 2, 68–75. [Google Scholar] [CrossRef]
- Fauza, H. Gambier: Indonesia leading commodities in the past. Int. J. Adv. Sci. Eng. Inf. Technol. 2014, 4, 455–460. [Google Scholar] [CrossRef][Green Version]
- Statistik Indonesia 2022. Available online: https://www.bps.go.id/publication/2022/02/25/0a2afea4fab72a5d052cb315/statistik-indonesia-2022.html (accessed on 5 May 2022).
- Van Wyk, B.E.; Wink, M. Medicinal Plants of the World; Centre for Agriculture and Bioscience International: Wallingford, UK; Briza Publications: Pretoria, South Africa, 2018. [Google Scholar]
- Andre, N.; Wang, X.; He, Y.; Pan, G.; Kojo, A.; Liu, Y. A review of the occurrence of non-alkaloid constituents in Uncaria species and their structure-activity relationships. Am. J. Biomed. Life Sci. 2013, 1, 79–98. [Google Scholar] [CrossRef]
- Carrick, J.; Chan, K.C.; Cheung, H.T. A new phytochemical survey of Malaya-chemical screening. Chem. Pharm. Bul. 1968, 16, 2436–2441. [Google Scholar] [CrossRef]
- Syarifah, S.; Widyawati, T.; Anggraini, D.R.; Wahyuni, A.S.; Sari, M.I. Anticancer activity of Uncaria gambir roxb on T47D breast cancer cells. J. Phys. Conf. Ser. IOP Publ. 2019, 1317, 012106. [Google Scholar] [CrossRef]
- Kim, T.H. A novel α-glucosidase inhibitory constituent from Uncaria gambir. J. Nat. Med. 2016, 70, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Zebua, E.A.; Silalahi, J.; Julianti, E. Hypoglicemic Activity of Gambier (Uncaria gambir Roxb.) Drinks in Alloxan-Induced Mice. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2018; Volume 122, p. 012088. [Google Scholar]
- Nurliayana, I.; Nurul, Z.M.Y.; Rohaya, A. Quantification of Catechin in Leaves and Stems of Malaysian Uncaria gambir (Hunter) Roxb. By Hplc-Dad. Malaysian J. Anal. Sci. 2016, 20, 567–572. [Google Scholar]
- Saad, M.F.M.; Goh, H.H.; Rajikan, R.; Yusof, T.R.T.; Baharum, S.N.; Bunawan, H. Uncaria gambir (W. Hunter) Roxb: From phytochemical composition to pharmacological importance. Trop. J. Pharm. Res. 2020, 19, 1767–1773. [Google Scholar] [CrossRef]
- Aprely, K.J.; Misfadhila, S.; Asra, R. A Review: The Phytochemistry, Pharmacology and Traditional Use of Gambir (Uncaria gambir (Hunter) Roxb). EAS J. Pharm. Pharmacol. 2021, 3, 21–25. [Google Scholar]
- Zhang, Q.; Zhao, J.J.; Xu, J.; Feng, F.; Qu, W. Medicinal uses, phytochemistry and pharmacology of the genus Uncaria. J. Ethnopharmacol. 2015, 173, 48–80. [Google Scholar] [CrossRef] [PubMed]
- Qin, N.; Lu, X.; Liu, Y.; Qiao, Y.; Qu, W.; Feng, F.; Sun, H. Recent research progress of Uncaria spp. based on alkaloids: Phytochemistry, pharmacology and structural chemistry. Eur. J. Med. Chem. 2021, 210, 112960. [Google Scholar] [CrossRef] [PubMed]
- Putri, S.M. Usaha Gambir Rakyat di Lima Puluh Kota, Sumatera Barat 1833–1930. Lembaran Sejarah 2013, 10, 149–163. [Google Scholar]
- Evalia, N.A.; Sa’id, E.G.; Suryana, R.N. Strategi pengembangan agroindustri dan peningkatan nilai tambah gambir di Kabupaten Lima Puluh kota Sumatera Barat. J. Manaj. Agribis. 2012, 9, 173–182. [Google Scholar]
- Morton, J.F. Widespread tannin intake via stimulants and masticatories, especially guarana, kola nut, betel vine, and accessories. Plant. Polyphenols 1992, 59, 739–765. [Google Scholar]
- Sebayang, L. Budidaya dan Pengolahan Gambir; Balai Pengkajian Teknologi Pertanian Sumatera Utara: Medan, Indonesia, 2013. [Google Scholar]
- Denian, A.; Hadad, M. Karakteristik Pohon Induk Gambir (Uncaria gambir (Hunter) Roxb.) di Sentra Produksi Sumatera Barat dan Riau. Bul. Penelit. Tanam. Rempah Obat 2008, 19, 18–38. [Google Scholar]
- Sebayang, L. Inovasi Teknologi Gambir Di Kabupaten Pakpak Barat; Balai Pengkajian Teknologi Pertanian Sumatera Utara: Medan, Indonesia, 2014. [Google Scholar]
- Sakti, A.S.; Saputri, F.C.; Mun’im, A. Microscopic Characters, phytochemical screening focus on alkaloid and total phenolic content of Uncaria gambir Roxb. and Uncaria sclerophylla Roxb. Leaves. Pharmacogn. J. 2019, 11, 119–123. [Google Scholar] [CrossRef]
- Wood, H.C. The Dispensatory of the United States of America; Lippincott: Philadelphia, PA, USA, 1918. [Google Scholar]
- Ferdinal, N.; Nazir, N. Phytochemical screening and fractination of gambier (Uncaria gambir). J. Biomater. Chem. 2013, 1, 23–25. [Google Scholar]
- Viena, V.; Nizar, M. Studi Kandungan Fitokimia Ekstrak Etanol Daun Gambir Asal Aceh Tenggara Sebagai Anti Diabetes. J. Serambi Eng. 2018, 3, 1. [Google Scholar] [CrossRef]
- Das, N.P.; Griffiths, L.A. Studies on flavonoid metabolism. Biosynthesis of (+)-[14C] catechin by the plant Uncaria gambir Roxb. Biochem. J. 1967, 105, 73–77. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Birch, A.J.; Donovan, F.W. Studies in relation to biosynthesis. I. Some possible routes to derivatives of orcinol and phloroglucinol. Aust. J. Chem. 1953, 6, 360. [Google Scholar]
- Das, N.P. Metabolism of [U–14C]-(+)-Catechin in the Plant Uncaria gambir. Planta Med. 1974, 25, 274–280. [Google Scholar] [CrossRef]
- Ferdinal, N. A simple purification method of catechin from gambier. Int. J. Adv. Sci. Eng. Inf. Technol. 2014, 4, 53–55. [Google Scholar] [CrossRef][Green Version]
- Kassim, M.J.; Hussin, M.H.; Achmad, A.; Dahon, N.H.; Suan, T.K.; Hamdan, H.S. Determination of total phenol, condensed tannin and flavonoid contents and antioxidant activity of Uncaria gambir extracts. Indones. J. Pharm. 2011, 22, 50–59. [Google Scholar]
- Anggraini, T.; Tai, A.; Yoshino, T.; Itani, T. Antioxidative activity and catechin content of four kinds of Uncaria gambir extracts from West Sumatra, Indonesia. Afr. J. Biochem. Res. 2011, 5, 33–38. [Google Scholar]
- Hayani, E. Analisis kadar catechin dari gambir dengan berbagai metode. Buletin Teknik Pertan. 2003, 8, 31–33. [Google Scholar]
- Andasuryani, A.; Purwanto, Y.A.; Budiastra, I.W.; Syamsu, K. Determination of catechin content in gambir powder from dried gambir leaves quickly using FT NIR PLS model. Int. J. Adv. Sci. Eng. Inf. Technol. 2014, 4, 303–307. [Google Scholar] [CrossRef][Green Version]
- Taniguchi, S.; Kuroda, K.; Doi, K.I.; Tanabe, M.; Shibata, T.; Yoshida, T.; Hatano, T. Revised structures of gambiriins A1, A2, B1, and B2, chalcane-flavan dimers from gambir (Uncaria gambir extract). Chem. Pharm. Bull. 2007, 55, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Rismana, E.; Ningsih, S.; Fachrudin, F. In vitro study of xanthine oxidase inhibitory of gambir (Uncaria gambir) hunter roxb extracts. Pharmacogn. J. 2017, 9, 862–865. [Google Scholar] [CrossRef]
- Nonaka, G.; Nishioka, I. Novel biflavonoids, chalcan-flavan dimers from Gambir. Chem. Pharm. Bull. 1980, 28, 3145–3149. [Google Scholar] [CrossRef]
- Taniguchi, S.; Kuroda, K.; Yoshikado, N.; Tanabe, M.; Shibata, T.; Yoshida, T.; Hatano, T. New dimeric flavans from gambir, an extract of Uncaria gambir. Heterocycles 2008, 74, 595–605. [Google Scholar]
- Yoshikado, N.; Taniguchi, S.; Kasajima, N.; Ohashi, F.; Doi, K.I.; Shibata, T.; Yoshida, T.; Hatano, T. Uncariagambiriine and gambircatechol: Novel constituents of Uncaria gambir leaves. Heterocycles 2009, 77, 793–800. [Google Scholar]
- Oshima, M.; Aoyama, H.; Shimozu, Y.; Taniguchi, S.; Miura, T.; Hatano, T. Uncariagambiriines B and C, alkaloid-catechin hybrids from Uncaria gambir leaves. Heterocycles 2019, 98, 804–812. [Google Scholar]
- Weinges, K.; Bahr, W.; Ebert, W.; Goritz, K.; Marx, H.D. Progress in the Chemistry of Organic Compounds; Zechmeister, L., Ed.; Springer: Wien, Austria, 1969; Volume 27, pp. 158–260. [Google Scholar]
- Failisnur, F.; Sofyan, S.; Kasim, A.; Angraini, T. Study of cotton fabric dyeing process with some mordant methods by using gambier (Uncaria gambir Roxb) extract. Int. J. Adv. Sci. Eng. Inf. Technol. 2018, 8, 1098–1104. [Google Scholar] [CrossRef]
- Kasim, A.; Malrianti, Y.; Derosya, V.; Syukri, D. GC-MS screening of valuable volatile compounds in the waste of Uncaria gambir. Ann. Biol. 2019, 35, 242–245. [Google Scholar]
- Sazwi, N.N.; Nalina, T.; Rahim, Z.H.A. Antioxidant and cytoprotective activities of Piper betle, Areca catechu, Uncaria gambir and betel quid with and without calcium hydroxide. BMC Complement. Altern. Med. 2013, 13, 351. [Google Scholar]
- Pavolini, T.; Gambarin, F. Montecchio, Curcumina e curcuminoidi, G. Ann. Chim. 1950, 40, 654. [Google Scholar]
- Merlini, L.; Mondelli, R.; Nasini, G.; Hesse, M. Indole alkaloids from gambir structure of bambirtannine, oxogambirtannine and dihydrogambirtannine. Tetrahedron 1967, 23, 3129–3145. [Google Scholar] [CrossRef]
- Chan, K.C. Gambirdine and isogambirdine, the alkaloids from Uncaria gambir (hunt) roxb. Tetrahedron Lett. 1968, 9, 3403–3406. [Google Scholar] [CrossRef]
- Saxton, J.E. Alkaloids of Mitragyna and related genera. In The Alkaloids: Chemistry and Physiology; Academic Press: Cambridge, MA, USA, 1973; Volume 14, pp. 123–156. [Google Scholar]
- Merlini, L.; Mondelli, R.; Nasini, G.; Hesse, M. The structure of roxburghines A–E, new indole alkaloids from an Uncaria sp. Tetrahedron 1970, 26, 2259–2279. [Google Scholar] [CrossRef]
- Taniguchi, S.; Kuroda, K.; Inada, K.; Yoshikado, N.; Yoneda, Y.; Tanabe, M.; Shibata, T.; Yoshida, T.; Hatano, T. Evaluation of gambir quality based on quantitative analysis of polyphenolic constituents. J. Pharm. Soc. Jpn. 2007, 127, 1291–1300. [Google Scholar] [CrossRef] [PubMed]
- Viena, V.; Nizar, M. α-Glucosidase Inhibitory Activity of Ethanolic Extract Gambier (Uncaria gambier Roxb) from Southeast Aceh as Antidiabetes. In Proceedings of the 1st Workshop on Multidisciplinary and Its Applications, Aceh, Indonesia, 20–21 January 2018; European Alliance for Innovation: Ghent, Belgium, 2019. [Google Scholar]
- Paramita, P.P.; Tata, H.L. Phytochemical Compounds Identification of Three Bajakah Species (Salacia sp., Uncaria acida, and Uncaria gambir) using GC-MS Pyrolysis. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2021; Volume 762, p. 012043. [Google Scholar]
- Amir, M.; Mujeeb, M.; Khan, A.; Ashraf, K.; Sharma, D.; Aqil, M. Phytochemical analysis and in vitro antioxidant activity of Uncaria gambir. Int. J. Green Pharm. 2012, 6, 67. [Google Scholar]
- Ferdinal, N.; Sulistyo, J.; Nazir, N. Sintesis Enzimatis Flavonoid-glikosida dari Gambir (Uncaria gambir) menggunakan Enzim CGT-ase dari Bacillus Licheniformis. Pros. SEMIRATA 2013, 1. Available online: https://jurnal.fmipa.unila.ac.id/semirata/article/view/825 (accessed on 24 June 2022).
- Nasrul, W.; Wardianto, D. Program Kemitraan Masyakat Produksi dan Pemasaran Gambir (Uncaria gambir Roxb). J. Pengabdi. UntukMu NegeRI 2020, 4, 187–191. [Google Scholar] [CrossRef]
- Ariyanti, P.R.; Aditya, M.A. Manfaat gambir (Uncaria gambir Roxb) sebagai antioksidan. Med. J. Lampung Univ. 2016, 5, 129–133. [Google Scholar]
- Firdausni, F.; Hermianti, W.; Diza, Y.H. Aplikasi gambir (Uncaria gambir Roxb) melalui proses pencucian berulang sebagai antioksidan pada pangan berminyak. J. Litbang Industri 2020, 10, 73–81. [Google Scholar] [CrossRef]
- Musdja, M.Y.; Rahman, H.A.; Hasan, D. Antioxidant activity of catechins isolate of Uncaria gambier Roxb in male rats. LIFE Int. J. Health Life-Sci. 2018, 4, 34–46. [Google Scholar] [CrossRef]
- Malrianti, Y.; Kasim, A.; Asben, A.; Syafri, E.; Yeni, G.; Fudholi, A. Catechin Extracted from Uncaria gambier Roxb for Nanocatechin Production: Physical and Chemical Properties. Int. J. Des. Nat. Ecodynamics 2021, 16, 393–399. [Google Scholar] [CrossRef]
- Yeni, G.; Syamsu, K.; Suparno, O.; Mardliyati, E.; Muchtar, H. Repeated extraction process of raw gambiers (Uncaria gambier Roxb.) for the catechin production as an antioxidant. Int. J. Appl. Eng. Res. 2014, 9, 24565–24578. [Google Scholar]
- Rahmawati, N.; Fernando, A. Kandungan Fenolik dan Aktivitas Antioksidan Ekstrak Daun Gambir Kering (Uncaria gambir (Hunter) Roxb). Jurnal ICA Indones. Chem. Acta 2013, 4, 1–6. [Google Scholar]
- Widiyarti, G.; Sundowo, A.; Filailla, E.; Laksmono, J.A. The Mechanically Extraction Process of Gambier (Uncaria gambier Roxb.) from Limapuluh Kota, West Sumatera and Its Antioxidant activity. Pure Appl. Chem. Res. 2020, 9, 8. [Google Scholar] [CrossRef]
- Ismail, A.S.; Rizal, Y.; Armenia, A.; Kasim, A. Determination of the best method for processing gambier liquid by-product [Uncaria gambir (hunter) roxb] as natural antioxidant sources. J. Indones. Trop. Anim. Agric. 2021, 46, 166–172. [Google Scholar] [CrossRef]
- Joycharat, N.; Limsuwan, S.; Subhadhirasakul, S.; Voravuthikunchai, S.P.; Pratumwan, S.; Madahin, I.; Nuankaew, W.; Promsawat, A. Anti-Streptococcus mutans efficacy of Thai herbal formula used as a remedy for dental caries. Pharm. Biol. 2012, 50, 941–947. [Google Scholar] [CrossRef]
- Scalbert, A. Antimicrobial properties of tannins. Phytochemistry 1991, 30, 3875–3883. [Google Scholar] [CrossRef]
- Tsuchiya, H.; Sato, M.; Miyazaki, T.; Fujiwara, S.; Tanigaki, S.; Ohyama, M.; Tanaka, T.; Iinuma, M. Comparative study on the antibacterial activity of phytochemical flavanones against methicillin-resistant Staphylococcus aureus. J. Ethnopharmacol. 1996, 50, 27–34. [Google Scholar] [CrossRef]
- Voravuthikunchai, S.; Lortheeranuwat, A.; Jeeju, W.; Sririrak, T.; Phongpaichit, S.; Supawita, T. Effective medicinal plants against enterohaemorrhagic Escherichia coli O157: H7. J. Ethnopharmacol. 2004, 94, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Kresnawaty, I.; Zainuddin, A. Aktivitas antioksidan dan antibakteri dari derivat metil ekstrak etanol daun gambir (Uncaria gambir). J. Penelit. Tanam. Ind. 2009, 15, 145–151. [Google Scholar] [CrossRef]
- Melia, S.; Novia, D.; Juliyarsi, I. Antioxidant and antimicrobial activities of gambir (Uncaria gambir Roxb) extracts and their application in rendang. Pak. J. Nutr. 2015, 14, 938. [Google Scholar] [CrossRef]
- Magdalena, N.V.; Kusnadi, J. Antibakteri dari Ekstrak Kasar Daun Gambir (Uncaria gambir Var Cubadak) Metode Microwave-Assisted Extraction Terhadap Bakteri Patogen [In Press Januari 2015]. J. Pangan Dan Agroindustri 2015, 3, 124–135. [Google Scholar]
- Musdja, M.Y.; Hapsari, M.A.; Agusta, A. Comparison of activity and inhibitory mechanism between (+)-Catechin and water extract of gambier (Uncaria gambir Roxb.) against some bacteria. Sci. J. PPI-UKM 2017, 4, 2. [Google Scholar]
- Lustigman, S.; Prichard, R.K.; Gazzinelli, A.; Grant, W.N.; Boatin, B.A.; McCarthy, J.S.; Basáñez, M.G. A research agenda for helminth diseases of humans: The problem of helminthiases. PLoS Negl. Trop. Dis. 2012, 6, e1582. [Google Scholar] [CrossRef]
- Hotez, P.J.; Brindley, P.J.; Bethony, J.M.; King, C.H.; Pearce, E.J.; Jacobson, J. Helminth infections: The great neglected tropical diseases. J. Clin. Investig. 2008, 118, 1311–1321. [Google Scholar] [CrossRef]
- Gazzinelli, A.; Correa-Oliveira, R.; Yang, G.J.; Boatin, B.A.; Kloos, H. A research agenda for helminth diseases of humans: Social ecology, environmental determinants, and health systems. PLoS Negl. Trop. Dis. 2012, 6, e1603. [Google Scholar] [CrossRef]
- Ault, S.K. Pan American Health Organization’s Regional Strategic Framework for addressing neglected diseases in neglected populations in Latin America and the Caribbean. Mem. Inst. Oswaldo Cruz 2007, 102, 99–107. [Google Scholar] [CrossRef]
- Patil, S.H.; Deshmukh, P.V.; Sreenivas, S.A.; Sankeertana, V.; Rekha, V.; Anjaiah, B. Evaluation of Anthelmintic activity of Uncaria gambier Roxb. against Pheretima posthuma. Int. J. Drug Dev. Res. 2012, 4, 234–238. [Google Scholar]
- Sonalkar, M.Y.; Nitave, S.A. Comparative anthelmintic activity of Eugenia caryophyllus & Uncaria gambier. World J. Pharm. Pharm. Sci. 2014, 3, 1409–1414. [Google Scholar]
- Niezen, J.H.; Waghorn, T.S.; Charleston, W.A.G.; Waghorn, G.C. Growth and gastrointestinal nematode parasitism in lambs grazing either lucerne (Medicago sativa) or sulla (Hedysarum coronarium) which contains condensed tannins. J. Agric. Sci. 1995, 125, 281–289. [Google Scholar] [CrossRef]
- Athanasiadou, S.; Kyriazakis, I.; Jackson, F.; Coop, R.L. Direct anthelmintic effects of condensed tannins towards different gastrointestinal nematodes of sheep: In vitro and in vivo studies. Vet. Parasitol. 2001, 99, 205–219. [Google Scholar] [CrossRef]
- Shrestha, B.; Basnett, H.; Babu, V.D.; Patel, S.S. Anthelmintic and Antimicrobial activity of the chloroform extract of Pergularia Daemia Forsk Leaves. Adv. Pharmacol. Toxicol. 2009, 10, 13–16. [Google Scholar]
- Cancer. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer#:~:text=Cancer%20is%20a%20leading%20cause,and%20rectum%20and%20prostate%20cancers (accessed on 5 March 2022).
- What Is Cancer. Available online: https://www.cancer.gov/about-cancer/understanding/what-is-cancer (accessed on 5 March 2022).
- Waks, A.G.; Winer, E.P. Breast cancer treatment: A review. J. Am. Med. Assoc. 2019, 321, 288–300. [Google Scholar] [CrossRef]
- Rhodes, A.; Yip, C. Comparison of breast cancer in Indonesia and Malaysia–A clinico-pathological study between Dharmais cancer centre Jakarta and University Malaya Medical Centre, Kuala Lumpur. Asian Pac. J. Cancer Prev. 2011, 12, 2943–2946. [Google Scholar]
- Sonwa, M.M.; König, W.A. Chemical study of the essential oil of Cyperus rotundus. Phytochemistry 2001, 58, 799–810. [Google Scholar] [CrossRef]
- Devasagayam, T.P.A.; Tilak, J.C.; Boloor, K.K.; Sane, K.S.; Ghaskadbi, S.S.; Lele, R.D. Free radicals and antioxidants in human health: Current status and future prospects. J. Assoc. Physicians India 2004, 52, 4. [Google Scholar]
- Patil, S.M.; Kadam, V.J.; Ghosh, R. In vitro antioxidant activity of methanolic extract of stem bark of Gmelina arborea roxb. (verbenaceae). Indian J. Physiother. Occup. Ther. 2009, 1, 1480–1484. [Google Scholar]
- McCoy, C.W.; Samson, R.A.; Boucias, D.G. Entomogenous fungi. In CRC handbook of Natural Pesticides; CRC Press: Boca Raton, FL, USA, 2019; pp. 151–236. [Google Scholar]
- Barkai-Golan, R.; Paster, N. Mycotoxins in Fruits and Vegetables; Elsevier: San Diego, CA, USA, 2008. [Google Scholar]
- Lonsdale, D.; Pautasso, M.; Holdenrieder, O. Wood-decaying fungi in the forest: Conservation needs and management options. Eur. J. For. Res. 2008, 127, 1–22. [Google Scholar] [CrossRef]
- Dai, Y.C.; Cui, B.K.; Yuan, H.S.; Li, B.D. Pathogenic wood-decaying fungi in China. For. Pathol. 2007, 37, 105–120. [Google Scholar] [CrossRef]
- Manion, P.D. Tree Disease Concepts, 2nd ed.; Englewood Cliffs, Prentice-Hall: Hoboken, NJ, USA, 1991; p. 402. [Google Scholar]
- Nandika, D.; Syamsu, K.; Arinana, A.; Kusumawardani, D.T.; Fitriana, Y. Bioactivities of catechin from Gambir (Uncaria gambir Roxb.) against wood-decaying fungi. Bioresources 2019, 14, 5646–5656. [Google Scholar]
- Farkas, G.L.; Kiraaly, Z. Role of phenolic compounds in the physiology of plant diseases and disease resistance. J. Phytopathol. 1962, 44, 105–150. [Google Scholar] [CrossRef]
- Nathan, C. Points of control in inflammation. Nature 2002, 420, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Fullerton, J.N.; Gilroy, D.W. Resolution of inflammation: A new therapeutic frontier. Nat. Rev. Drug Discov. 2016, 15, 551–567. [Google Scholar] [CrossRef] [PubMed]
- Mueller, M.; Hobiger, S.; Jungbauer, A. Anti-inflammatory activity of extracts from fruits, herbs and spices. Food Chem. 2010, 122, 987–996. [Google Scholar] [CrossRef]
- Auliana, F.R.; Fauziah, F. Phytochemical and Anti-Inflammatory of Uncaria gambir: A Review. Asian J. Pharm. Res. Dev. 2022, 10, 79–83. [Google Scholar] [CrossRef]
- Yimam, M.; Lee, Y.C.; Kim, T.W.; Moore, B.; Jiao, P.; Hong, M.; Kim, H.J.; Nam, J.B.; Kim, M.R.; Oh, J.S.; et al. Analgesic and anti-inflammatory effect of UP3005, a botanical composition containing two standardized extracts of Uncaria gambir and Morus alba. Pharmacogn. Res. 2015, 7, S39. [Google Scholar] [CrossRef]
- Musdja, M.Y.; Suryani, N.; Pitriyah, P. Anti-Inlammatory Activity of Catechins Isolate of Uncaria gambir Roxb on Carrageenan Induced Paw Edema in Wistar Male Rats. Int. J. Recent Adv. Multidiscip. Res. 2019, 6, 4518–4521. [Google Scholar]
- Yunarto, N.; Intan, P.R.; Kurniatri, A.A.; Sulistyowati, I.; Aini, N. Anti-Inflammatory Activities of Ethyl Acetate Fraction from Uncaria gambir Leaves Through the Inhibition of Edema, COX-2 and iNOS Expression. In Proceedings of the 4th International Symposium on Health Research, Bali, Indonesia, 27–30 November 2019; Atlantis Press: Amsterdam, The Netherlands, 2020; pp. 108–112. [Google Scholar]
- Corry, D.B.; Tuck, M.L. Protection from vascular risk in diabetic hypertension. Curr. Hypertens. Rep. 2000, 2, 154–159. [Google Scholar] [CrossRef]
- Goyal, A.; Mehta, S.R.; Díaz, R.; Gerstein, H.C.; Afzal, R.; Xavier, D.; Liu, L.; Pais, P.; Yusuf, S. Differential clinical outcomes associated with hypoglycemia and hyperglycemia in acute myocardial infarction. Circulation 2009, 120, 2429–2437. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37, S81–S90. [Google Scholar] [CrossRef] [PubMed]
- Hansawasdi, C.; Kawabata, J.; Kasai, T. α-Amylase inhibitors from roselle (Hibiscus sabdariffa Linn.) tea. Biosci. Biotechnol. Biochem. 2000, 64, 1041–1043. [Google Scholar] [CrossRef] [PubMed]
- Pane, Y.S.; Ganie, R.A.; Lindarto, D.; Lelo, A. The effect of gambier extract on the levels of malondialdehyde, superoxide dismutase, and blood glucose in type 2 diabetes mellitus patients. Asian J. Pharm. Clin. Res. 2018, 11, 121–124. [Google Scholar] [CrossRef]
- Yanti, E.; Morika, H.D.; Harmawati; Nur, S.A. Pengaruh Pemberian Gambir (Uncaria gambir) Terhadap Kadar Gula Darah Pada Pasien Diabetes Melitus Tipe II. J. Kesehat. St. Meditory 2020, 2, 27–39. [Google Scholar]
- Widiyarti, G.; Sundowo, A.; Hanafi, M. The free radical scavenging and anti-hyperglycemic activities of various gambiers available in Indonesian market. Makara J. Sci. 2012, 129–134. [Google Scholar] [CrossRef]
- Apea-Bah, F.B.; Hanafi, M.; Dewi, R.T.; Fajriah, S.; Darwaman, A.; Artanti, N.; Lotulung, P.; Ngadymang, P.; Minarti, B. Assessment of the DPPH and-glucosidase inhibitory potential of gambier and qualitative identification of major bioactive compound. J. Med. Plant. Res. 2009, 3, 736–757. [Google Scholar]
- Spanou, C.; Veskoukis, A.S.; Kerasioti, T.; Kontou, M.; Angelis, A.; Aligiannis, N.; Skaltsounis, A.L.; Kouretas, D. Flavonoid glycosides isolated from unique legume plant extracts as novel inhibitors of xanthine oxidase. PLoS ONE 2012, 7, e32214. [Google Scholar] [CrossRef]
- Bardin, T.; Richette, P. Definition of hyperuricemia and gouty conditions. Curr. Opin. Rheumatol. 2014, 26, 186–191. [Google Scholar] [CrossRef]
- Gliozzi, M.; Malara, N.; Muscoli, S.; Mollace, V. The treatment of hyperuricemia. Int. J. Cardiol. 2016, 213, 23–27. [Google Scholar] [CrossRef]
- Negre-Salvayre, A.; Auge, N.; Ayala, V.; Basaga, H.; Boada, J.; Brenke, R.; Chapple, S.; Cohen, G.; Feher, J.; Grune, T.; et al. Pathological aspects of lipid peroxidation. Free Radic. Res. 2010, 44, 1125–1171. [Google Scholar] [CrossRef]
- Ramana, K.V.; Srivastava, S.; Singhal, S.S. Lipid peroxidation products in human health and disease. Oxid. Med. Cell. Longev. 2013, 2013, 583438. [Google Scholar] [CrossRef] [PubMed]
- Ningsih, S.; Fachrudin, F.; Rismana, E.; Purwaningsih, E.H.; Sumaryono, W.; Jusman, S.W. Evaluation of antilipid peroxidation activity of gambir extract on liver homogenat in vitro. Int. J. Pharmtech Res. 2014, 6, 982–989. [Google Scholar]
- Auclair, S.; Milenkovic, D.; Besson, C.; Chauvet, S.; Gueux, E.; Morand, C.; Mazur, A.; Scalbert, A. Catechin reduces atherosclerotic lesion development in apo E-deficient mice: A transcriptomic study. Atherosclerosis 2009, 204, e21–e27. [Google Scholar] [CrossRef]
- Fedacko, J.; Singh, R.B.; Chaithiraphan, S.; Vargova, V.; Tomlinson, B.; De Meester, F.; Gvozdjakova, A.; Moesgaard, S. Clinical Manifestations of Adverse Effects of Statins, Oxidative Stress and Possible Role of Antioxidants in Prevention? Open Nutraceuticals J. 2010, 3, 154–165. [Google Scholar] [CrossRef][Green Version]
- Lu, T.; Tan, B.; Deng, T.; Hua, Y.; Xi, G. Therapeutic effect of Zhibitai combined with atorvastatin in the treatment of coronary heart disease with hyperlipidemia and its influence on the level of hypersensitive C-reactive protein. Chin. J. Primary Med. Pharm. 2019, 3022–3025. [Google Scholar]
- Yunarto, N.; Aini, N. Effect of purified gambir leaves extract to prevent atherosclerosis in rats. Health Sci. J. Indonesia. 2015, 6, 105–110. [Google Scholar] [CrossRef]
- Yunarto, N.; Sulistyaningrum, N.; Kurniatri, A.A.; Elya, B. Gambir (Uncaria gambir Roxb.) as A Potential Alternative Treatment for Hyperlipidemia. Media Penelit. Dan Pengemb. Kesehat. 2021, 31, 183–192. [Google Scholar] [CrossRef]
- Yunarto, N.; Elya, B.; Konadi, L. Potensi fraksi etil asetat ekstrak daun gambir (Uncaria gambir roxb.) sebagai antihiperlipidemia. J. Kefarmasian Indonesia 2015, 5, 1–10. [Google Scholar] [CrossRef]
- Alioes, Y.; Sukma, R.R.; Sekar, S.L. Effect of Gambir Catechin Isolate (Uncaria gambir Roxb.) Against Rat Triacylglycerol Level (Rattus novergicus). In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2019; Volume 217, p. 012020. [Google Scholar]
- Berry, S.E. Triacylglycerol structure and interesterification of palmitic and stearic acid-rich fats: An overview and implications for cardiovascular disease. Nutr. Res. Rev. 2009, 22, 3–17. [Google Scholar] [CrossRef]
- Bonen, A.; Parolin, M.L.; Steinberg, G.R.; Calles-Escandon, J.; Tandon, N.N.; Glatz, J.F.; Luiken, J.J.; Heigenhauser, G.J.; Dyck, D.J. Triacylglycerol accumulation in human obesity and type 2 diabetes is associated with increased rates of skeletal muscle fatty acid transport and increased sarcolemmal FAT/CD36. FASEB J. 2004, 18, 1144–1146. [Google Scholar] [CrossRef] [PubMed]
- Achmad, A.; Kassim, J.; Suan, T.K.; Amat, R.C.; Seey, T.L. Equilibrium, kinetic and thermodynamic studies on the adsorption of direct dye onto a novel green adsorbent developed from Uncaria gambir extract. J. Phys. Sci. 2012, 23, 1–13. [Google Scholar]
- Tong, K.S.; Kassim, M.J.; Azraa, A. Adsorption of copper ion from its aqueous solution by a novel biosorbent Uncaria gambir: Equilibrium, kinetics, and thermodynamic studies. Chem. Eng. J. 2011, 170, 145–153. [Google Scholar] [CrossRef]
- Hussin, M.H.; Kassim, M.J. The corrosion inhibition and adsorption behavior of Uncaria gambir extract on mild steel in 1 M HCl. Mater. Chem. Phys. 2011, 125, 461–468. [Google Scholar] [CrossRef]
- Ogunleye, O.O.; Arinkoola, A.O.; Eletta, O.A.; Agbede, O.O.; Osho, Y.A.; Morakinyo, A.F.; Hamed, J.O. Green corrosion inhibition and adsorption characteristics of Luffa cylindrica leaf extract on mild steel in hydrochloric acid environment. Heliyon 2020, 6, e03205. [Google Scholar] [CrossRef]

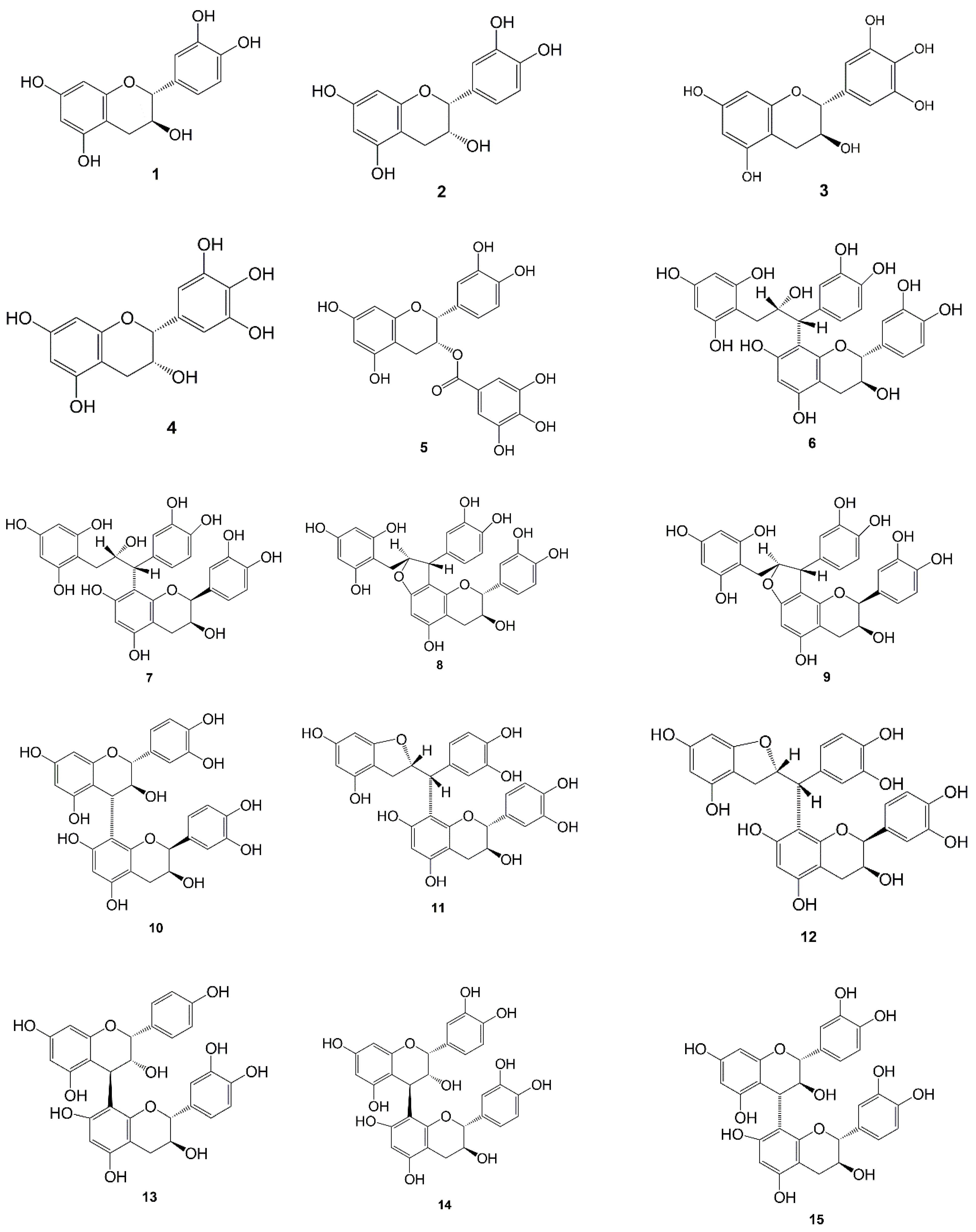
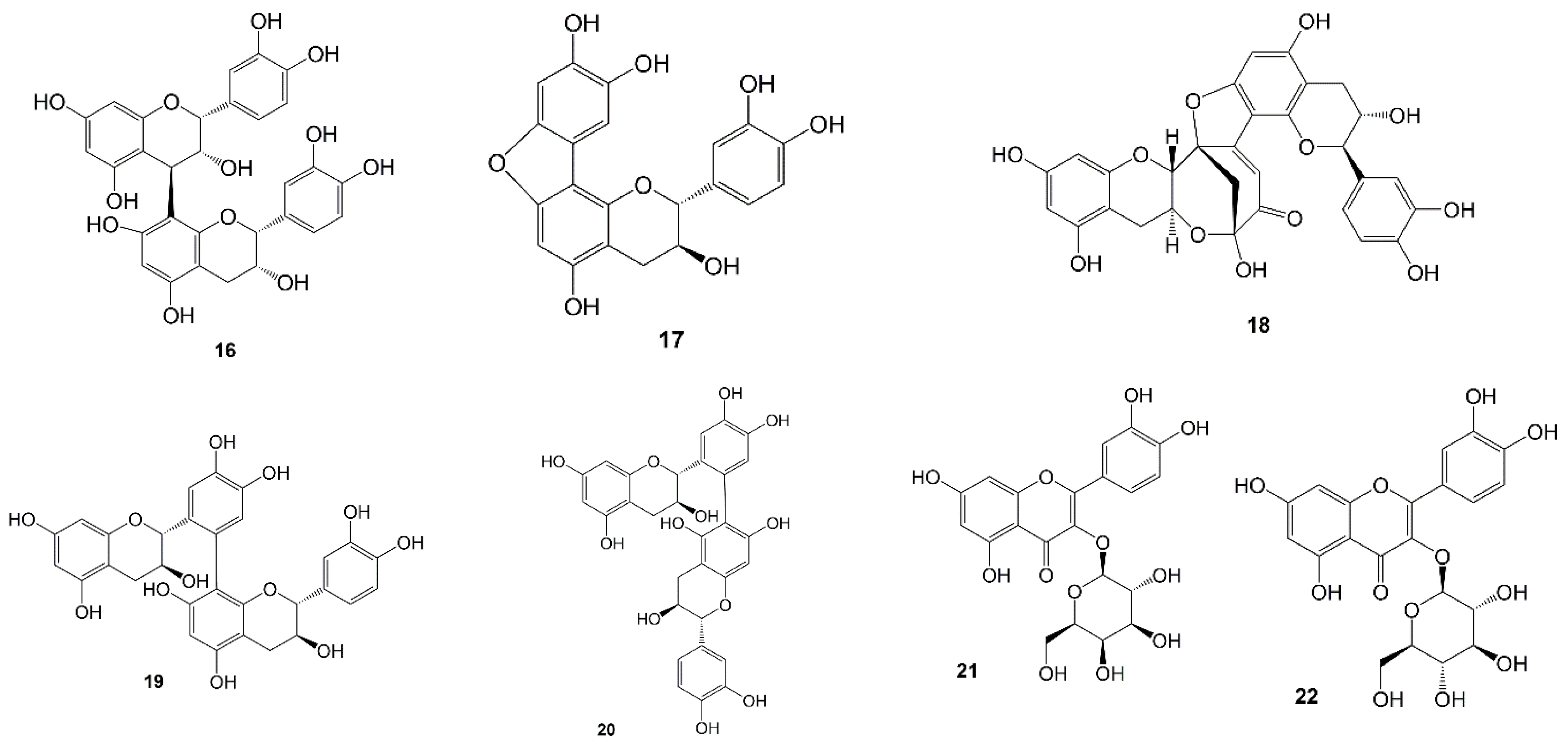
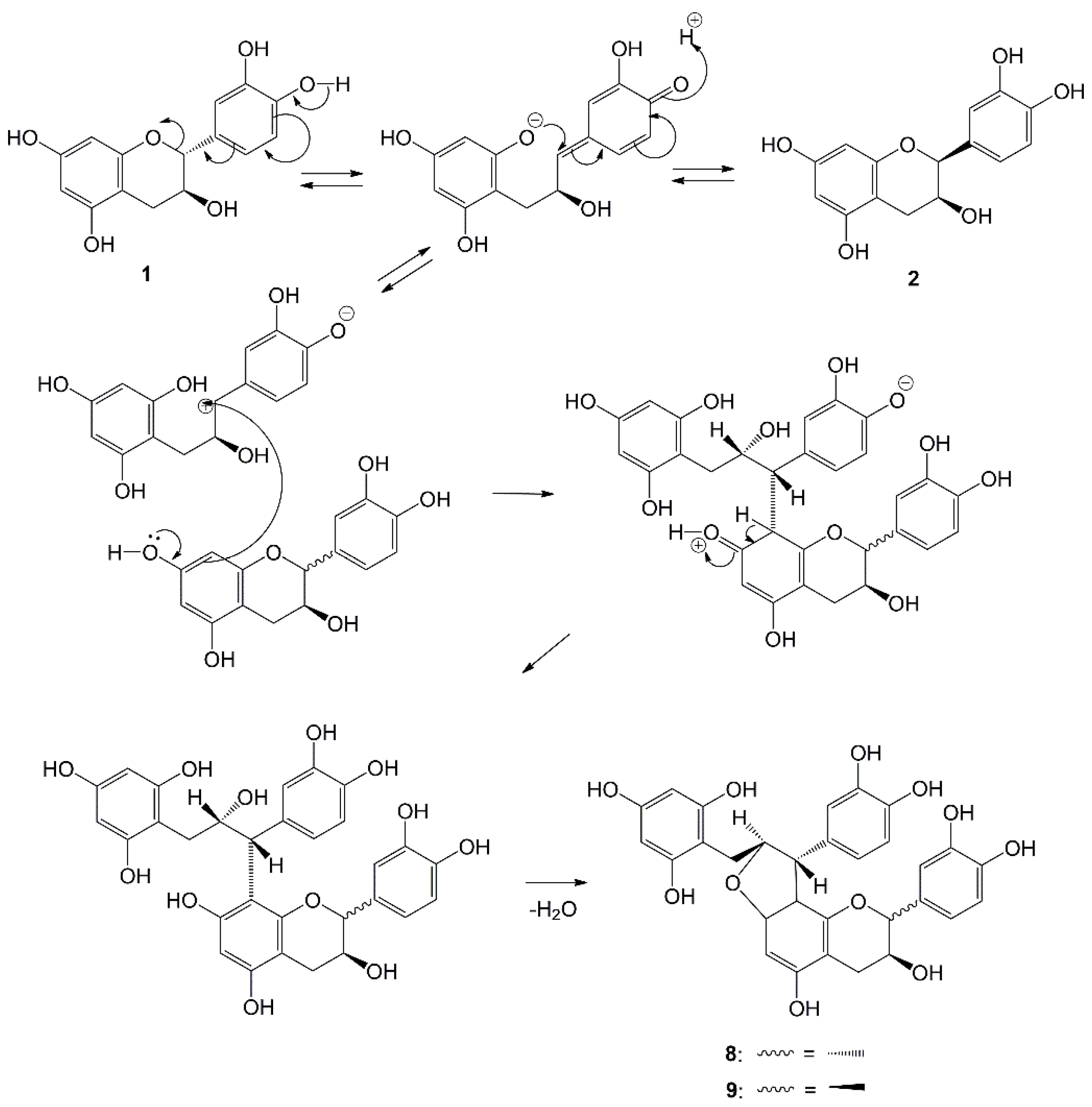

| No | Compound | Type | Extract | Plant Part | Ref. |
|---|---|---|---|---|---|
| 1 | Catechin | Flavonoid | Water | Leaves | [37] |
| Water | Leaves and young twigs | [47,57] | |||
| Methanol | Leaves and young twigs | [40] | |||
| Ethanol | Leaves | [41] | |||
| Ethyl acetate | Leaves and young twigs | [36] | |||
| 2 | Epicatechin | Flavonoid | Water | Leaves | [37] |
| Methanol | Leaves and young twigs | [40] | |||
| Ethyl acetate | Leaves and young twigs | [36] | |||
| 3 | Gallocatechin | Flavonoid | Ethyl acetate | Leaves and young twigs | [36] |
| 4 | Epigallocatechin | Flavonoid | Ethyl acetate | Leaves and young twigs | [36] |
| 5 | Epicatechin gallate | Flavonoid | Ethyl acetate | Leaves and young twigs | [36] |
| 6 | Gambiriin A1 | Dimeric flavonoid | Methanol | Leaves and young twigs | [40] |
| 7 | Gambiriin A2 | Dimeric flavonoid | Methanol | Leaves and young twigs | [40] |
| 8 | Gambiriin B1 | Dimeric flavonoid | Methanol | Leaves and young twigs | [40] |
| 9 | Gambiriin B2 | Dimeric flavonoid | Methanol | Leaves and young twigs | [40] |
| 10 | Catechin-(4α→8)-ent-epicatechin | Flavonoid | Methanol | Leaves and young twigs | [43] |
| 11 | Gambiriin C | Dimeric flavonoid | Methanol | Leaves and young twigs | [40] |
| 12 | Procyanidin B1 | Dimeric flavonoid | Methanol | Leaves and young twigs | [40] |
| 13 | Procyanidin B3 | Dimeric flavonoid | Methanol | Leaves and young twigs | [40] |
| 14 | Dimeric proanthocyanidin | Dimeric flavonoid | Methanol | Leaves and young twigs | [40] |
| 15 | Gambirflavan D1 | Dimeric flavonoid | Methanol | Leaves and young twigs | [43] |
| 16 | Gambirflavan D2 | Dimeric flavonoid | Methanol | Leaves and young twigs | [43] |
| 17 | Gambircatechol | Dimeric flavonoid | Acetone | Leaves | [44] |
| 18 | Dehydrodicatechin A | Dimeric flavonoid | Acetone | Leaves | [44] |
| 19 | Catechin-(8→6′)-catechin | Dimeric flavonoid | Acetone | Leaves | [44] |
| 20 | Catechin-(6→6′)-catechin | Dimeric flavonoid | Acetone | Leaves | [44] |
| 21 | Hyperoside | Flavonoid-glycosides | Water | Leaves and twigs | [45] |
| 22 | Isoquercitrin | Flavonoid-glycosides | Water | Leaves and twigs | [45] |
| 23 | Pyrocatechol | Phenolic | Water | Leaves | [37] |
| Water | Leaves and young twigs | [47] | |||
| Acetone | Leaves and young twigs | [48] | |||
| 24 | Phloroglucinol | Phenolic | Acetic acid | Leaves and young twigs | [48] |
| 25 | 3-methylphenol | Phenolic | Methanol | Wood | [58] |
| 26 | 2-methoxyphenol | Phenolic | Methanol | Wood and bark | [58] |
| 27 | 4-methoxyphenol | Phenolic | Methanol | Wood and bark | [58] |
| 28 | 2,6-dimethoxyphenol | Phenolic | Methanol | Wood and bark | [58] |
| 29 | 2-methoxy-4-methylphenol | Phenolic | Methanol | Wood and bark | [58] |
| 30 | 4-ethyl-2-methoxyphenol | Phenolic | Methanol | Wood and bark | [58] |
| 31 | 2-methoxy-4-vinylphenol | Phenolic | Methanol | Wood and bark | [58] |
| 32 | 4-allyl-2-methoxyphenol | Phenolic | Methanol | Wood and bark | [58] |
| 33 | 4-allyl-2,6-dimethoxyphenol | Phenolic | Methanol | Wood and bark | [58] |
| 34 | Quinic acid | Cyclic polyol | Water | All part | [49] |
| 35 | Gambirine | Alkaloid | Methanol | Leaves | [50] |
| 36 | Gambirtannine | Alkaloid | Methanol | Leaves | [51] |
| 37 | Dihydrogambirtannine | Alkaloid | Methanol | Leaves | [51] |
| 38 | Oogambirtannine | Alkaloid | Methanol | Leaves | [51] |
| 39 | Neooxygambirtannine | Alkaloid | Methanol | Leaves | [51] |
| 40 | Isogambirine | Alkaloid | Methanol | Leaves | [52] |
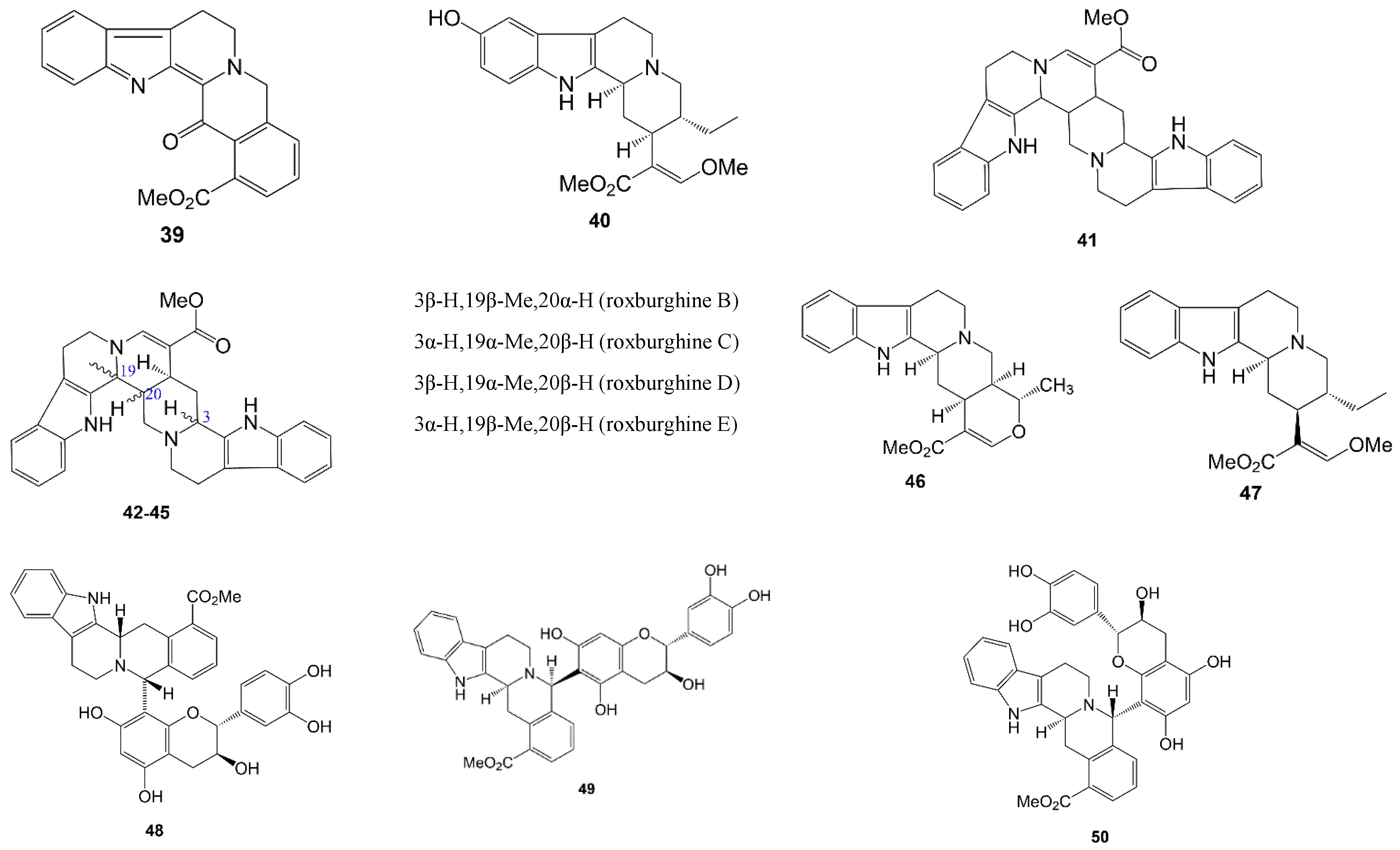
| 41 | Roxburghines A | Alkaloid | Methanol | Leaves | [54] |
| 42 | Roxburghines B | Alkaloid | Methanol | Leaves | [54] |
| 43 | Roxburghines C | Alkaloid | Methanol | Leaves | [54] |
| 44 | Roxburghines D | Alkaloid | Methanol | Leaves and stems | [54] |
| 45 | Roxburghines E | Alkaloid | Methanol | Leaves and stems | [54] |
| 46 | Tetrahydroalstonine | Alkaloid | Methanol | Leaves and stems | [54] |
| 47 | Dihydrocorynantheine | Alkaloid | Methanol | Leaves and stems | [54] |
| 48 | Uncariagambiriine | Catechin-alkaloid | Acetone | Leaves | [44] |
| 49 | Uncariagambiriine B | Catechin-alkaloid | Water | Leaves and twigs | [45] |
| 50 | Uncariagambiriine C | Catechin-alkaloid | Water | Leaves and twigs | [45] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Munggari, I.P.; Kurnia, D.; Deawati, Y.; Julaeha, E. Current Research of Phytochemical, Medicinal and Non-Medicinal Uses of Uncaria gambir Roxb.: A Review. Molecules 2022, 27, 6551. https://doi.org/10.3390/molecules27196551
Munggari IP, Kurnia D, Deawati Y, Julaeha E. Current Research of Phytochemical, Medicinal and Non-Medicinal Uses of Uncaria gambir Roxb.: A Review. Molecules. 2022; 27(19):6551. https://doi.org/10.3390/molecules27196551
Chicago/Turabian StyleMunggari, Indah Putri, Dikdik Kurnia, Yusi Deawati, and Euis Julaeha. 2022. "Current Research of Phytochemical, Medicinal and Non-Medicinal Uses of Uncaria gambir Roxb.: A Review" Molecules 27, no. 19: 6551. https://doi.org/10.3390/molecules27196551
APA StyleMunggari, I. P., Kurnia, D., Deawati, Y., & Julaeha, E. (2022). Current Research of Phytochemical, Medicinal and Non-Medicinal Uses of Uncaria gambir Roxb.: A Review. Molecules, 27(19), 6551. https://doi.org/10.3390/molecules27196551





