From Tradition to Health: Chemical and Bioactive Characterization of Five Traditional Plants
Abstract
1. Introduction
2. Results and Discussion
2.1. Identification of the Main Phenolic Composition
2.1.1. O-Glycosylated Flavonoids
2.1.2. Phenolic Acids
2.1.3. Isoflavonoids
2.1.4. C-Glycosylated Flavonoids
2.1.5. Flavan-3-Ols
2.1.6. Phenylethanoid Glycosides
2.1.7. Iridoid Glycosides
2.2. Biological Properties
2.2.1. Antioxidant Studies
2.2.2. Cytotoxic and Anti-Inflammatory Studies
2.2.3. Antibacterial and Antifungal Studies
3. Materials and Methods
3.1. Samples
3.2. Preparations
3.3. Phenolic Compound Composition
3.4. Bioactive Evaluation
3.4.1. Evaluation of Antioxidant Activity
3.4.2. Evaluation of the Cytotoxic and Hepatotoxic Activity
3.4.3. Evaluation of Anti-Inflammatory Activity
3.4.4. Evaluation of Antimicrobial Activity
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Balunas, M.J.; Kinghorn, A.D. Drug discovery from medicinal plants. Life Sci. 2005, 78, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta Gen. Subj. 2013, 1830, 3670–3695. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z. The modification of natural products for medical use. Acta Pharm. Sin. B 2017, 7, 119–136. [Google Scholar] [CrossRef] [PubMed]
- Martín Ortega, A.M.; Segura Campos, M.R. Medicinal Plants and Their Bioactive Metabolites in Cancer Prevention and Treatment. In Bioactive Compounds; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 85–109. ISBN 9780128147740. [Google Scholar]
- del Villegas-Aguilar, M.C.; Fernández-Ochoa, Á.; Leyva-Jiménez, F.J.; Miranda-Segura, Á.; de la Cádiz-Gurrea, M.L.; Segura-Carretero, A. Phenolic compounds. In Bioactive Food Components Activity in Mechanistic Approach; Cazarin, C., Lemos-Bicas, J., Pastore, G.M., Marostica Junior, M., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 27–53. [Google Scholar]
- Petropoulos, S.A.; Fernandes, Â.; Tzortzakis, N.; Sokovic, M.; Ciric, A.; Barros, L.; Ferreira, I.C.F.R. Bioactive compounds content and antimicrobial activities of wild edible Asteraceae species of the Mediterranean flora under commercial cultivation conditions. Food Res. Int. 2019, 119, 859–868. [Google Scholar] [CrossRef] [PubMed]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef]
- Albuquerque, B.R.; Heleno, S.A.; Oliveira, M.B.P.P.; Barros, L.; Ferreira, I.C.F.R. Phenolic compounds: Current industrial applications, limitations and future challenges. Food Funct. 2021, 12, 14–29. [Google Scholar] [CrossRef]
- Vučić, D.M.; Petković, M.R.; Rodić-Grabovac, B.B.; Stefanović, O.D.; Vasić, S.M.; Čomić, L.R. In vitro activity of heather [Calluna vulgaris (L.) Hull] extracts on selected urinary tract pathogens. Bosn. J. Basic Med. Sci. 2014, 14, 234–238. [Google Scholar] [CrossRef][Green Version]
- Mandim, F.; Barros, L.; Calhelha, R.C.; Abreu, R.M.V.; Pinela, J.; Alves, M.J.; Heleno, S.; Santos, P.F.; Ferreira, I.C.F.R. Calluna vulgaris (L.) Hull: Chemical characterization, evaluation of its bioactive properties and effect on the vaginal microbiota. Food Funct. 2019, 10, 78–89. [Google Scholar] [CrossRef]
- Starchenko, G.; Hrytsyk, A.; Raal, A.; Koshovyi, O. Phytochemical Profile and Pharmacological Activities of Water and Hydroethanolic Dry Extracts of Calluna vulgaris (L.) Hull. Herb. Plant. 2020, 9, 75. [Google Scholar] [CrossRef]
- Rodrigues, F.; Moreira, T.; Pinto, D.; Pimentel, F.B.; Costa, A.S.G.; Nunes, M.A.; Gonçalves Albuquerque, T.; Costaa, H.S.; Palmeira-de-Oliveira, A.; Oliveira, A.I.; et al. The phytochemical and bioactivity profiles of wild Calluna vulgaris L. flowers. Food Res. Int. 2018, 111, 724–731. [Google Scholar] [CrossRef]
- Rivera, D.; Verde, A.; Fajardo, J.; Obón, C.; Consuegra, V.; García-Botía, J.; Ríos, S.; Alcaraz, F.; Valdés, A.; del Moral, A.; et al. Ethnopharmacology in the Upper Guadiana River area (Castile-La Mancha, Spain). J. Ethnopharmacol. 2019, 241, 111968. [Google Scholar] [CrossRef] [PubMed]
- Pinto, D.C.G.A.; Simões, M.A.M.; Silva, A.M.S. Genista tridentata L.: A Rich Source of Flavonoids with Anti-Inflammatory Activity. Medicines 2020, 7, 31. [Google Scholar] [CrossRef] [PubMed]
- Simões, M.A.M.; Pinto, D.C.G.A.; Neves, B.M.R.; Silva, A.M.S. Flavonoid Profile of the Genista tridentata L., a Species Used Traditionally to Treat Inflammatory Processes. Molecules 2020, 25, 812. [Google Scholar] [CrossRef]
- Pinela, J.; Barros, L.; Carvalho, A.M.; Ferreira, I.C.F.R. Influence of the drying method in the antioxidant potential and chemical composition of four shrubby flowering plants from the tribe Genisteae (Fabaceae). Food Chem. Toxicol. 2011, 49, 2983–2989. [Google Scholar] [CrossRef] [PubMed]
- Pereira, O.R.; Silva, A.M.S.; Domingues, M.R.M.; Cardoso, S.M. Identification of phenolic constituents of Cytisus multiflorus. Food Chem. 2012, 131, 652–659. [Google Scholar] [CrossRef]
- Senatore, F.; Rigano, D.; Formisano, C.; Grassia, A.; Basile, A.; Sorbo, S. Phytogrowth-inhibitory and antibacterial activity of Verbascum sinuatum. Fitoterapia 2007, 78, 244–247. [Google Scholar] [CrossRef]
- Jamshidi-Kia, F.; Lorigooini, Z.; Asgari, S.; Saeidi, K. Iranian species of Verbascum: A review of botany, phytochemistry, and pharmacological effects. Toxin Rev. 2019, 38, 255–262. [Google Scholar] [CrossRef]
- Selseleh, M.; Nejad Ebrahimi, S.; Aliahmadi, A.; Sonboli, A.; Mirjalili, M.H. Metabolic profiling, antioxidant, and antibacterial activity of some Iranian Verbascum L. species. Ind. Crop. Prod. 2020, 153, 112609. [Google Scholar] [CrossRef]
- Moein, S.; Moein, M.; Khoshnoud, M.J.; Kalanteri, T. In vitro antioxidant properties evaluation of 10 Iranian medicinal plants by different methods. Iran. Red Crescent Med. J. 2012, 14, 771–775. [Google Scholar] [CrossRef]
- Bazzicalupo, M.; Cornara, L.; Burlando, B.; Cascini, A.; Denaro, M.; Smeriglio, A.; Trombetta, D. Carpobrotus edulis (L.) N.E.Br. extract as a skin preserving agent: From traditional medicine to scientific validation. J. Integr. Med. 2021, 19, 526–536. [Google Scholar] [CrossRef]
- Akinyede, K.A.; Ekpo, O.E.; Oguntibeju, O.O. Ethnopharmacology, therapeutic properties and nutritional potentials of Carpobrotus edulis: A comprehensive review. Sci. Pharm. 2020, 88, 39. [Google Scholar] [CrossRef]
- Hafsa, J.; Hammi, K.M.; Ben Khedher, M.R.; Smach, M.A.; Charfeddine, B.; Limem, K.; Majdoub, H. Inhibition of protein glycation, antioxidant and antiproliferative activities of Carpobrotus edulis extracts. Biomed. Pharmacother. 2016, 84, 1496–1503. [Google Scholar] [CrossRef] [PubMed]
- Omoruyi, B.E.; Ighodaro, D.I.; Afolayan, A.J.; Bradley, G. Inhibition of HIV-1 Protease by Carpobrotus edulis (L.). Evidence-based Complement. Altern. Med. 2020, 2020, 9648056. [Google Scholar] [CrossRef] [PubMed]
- Neves, M.; Antunes, M.; Fernandes, W.; Campos, M.J.; Azevedo, Z.M.; Freitas, V.; Rocha, J.M.; Tecelão, C. Physicochemical and nutritional profile of leaves, flowers, and fruits of the edible halophyte chorão-da-praia (Carpobrotus edulis) on Portuguese west shores. Food Biosci. 2021, 43, 101288. [Google Scholar] [CrossRef]
- Rodrigues da Silva, L.; Campos Chisté, R.; Fernandes, E. Chemical and antioxidant characterization of the portuguese heather honey from Calluna vulgaris. Separations 2021, 8, 177. [Google Scholar] [CrossRef]
- Mandim, F.; Barros, L.; Heleno, S.A.; Pires, T.C.S.P.; Dias, M.I.; Alves, M.J.; Santos, P.F.; Ferreira, I.C.F.R. Phenolic profile and effects of acetone fractions obtained from the inflorescences of: Calluna vulgaris (L.) Hull on vaginal pathogenic and non-pathogenic bacteria. Food Funct. 2019, 10, 2399–2407. [Google Scholar] [CrossRef]
- Caleja, C.; Finimundy, T.C.; Pereira, C.; Barros, L.; Calhelha, R.C.; Sokovic, M.; Ivanov, M.; Carvalho, A.M.; Rosa, E.; Ferreira, I.C.F.R. Challenges of traditional herbal teas: Plant infusions and their mixtures with bioactive properties. Food Funct. 2019, 10, 5939–5951. [Google Scholar] [CrossRef]
- Barros, L.; Dueñas, M.; Carvalho, A.M.; Ferreira, I.C.F.R.; Santos-Buelga, C. Characterization of phenolic compounds in flowers of wild medicinal plants from Northeastern Portugal. Food Chem. Toxicol. 2012, 50, 1576–1582. [Google Scholar] [CrossRef]
- Sabiu, S.; Balogun, F.O.; Amoo, S.O. Phenolics profiling of Carpobrotus edulis (L.) n.e.br. and insights into molecular dynamics of their significance in type 2 diabetes therapy and its retinopathy complication. Molecules 2021, 26, 4867. [Google Scholar] [CrossRef]
- Lyubchyk, S.; Shapovalova, O.; Lygina, O.; Oliveira, M.C.; Appazov, N.; Lyubchyk, A.; Charmier, A.J.; Lyubchik, S.; Pombeiro, A.J.L. Integrated Green Chemical Approach to the Medicinal Plant Carpobrotus edulis Processing. Sci. Rep. 2019, 9, 18171. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Courts, F.L.; Williamson, G. The occurrence, fate and biological activities of c-glycosyl flavonoids in the human diet. Crit. Rev. Food Sci. Nutr. 2015, 55, 1352–1367. [Google Scholar] [CrossRef] [PubMed]
- Bando, S.I.; Hatano, O.; Takemori, H.; Kubota, N.; Ohnishi, K. Potentiality of syringetin for preferential radiosensitization to cancer cells. Int. J. Radiat. Biol. 2017, 93, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.L.; Liang, H.L.; Hung, C.H.; Kuo, P.L. Syringetin, a flavonoid derivative in grape and wine, induces human osteoblast differentiation through bone morphogenetic protein-2/extracellular signal-regulated kinase 1/2 pathway. Mol. Nutr. Food Res. 2009, 53, 1452–1461. [Google Scholar] [CrossRef]
- Shang, Z.; Xin, Q.; Zhao, W.; Wang, Z.; Li, Q.; Zhang, J.; Cong, W. Rapid profiling and identification of puerarin metabolites in rat urine and plasma after oral administration by UHPLC-LTQ-Orbitrap mass spectrometer. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1068–1069, 180–192. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, G.; Zamilpa, A.; Zavala, M.; Perez, J.; Morales, D.; Tortoriello, J. Chrysoeriol and other polyphenols from Tecoma stans with lipase inhibitory activity. J. Ethnopharmacol. 2016, 185, 1–8. [Google Scholar] [CrossRef]
- Limboonreung, T.; Tuchinda, P.; Chongthammakun, S. Chrysoeriol mediates mitochondrial protection via PI3K/Akt pathway in MPP+ treated SH-SY5Y cells. Neurosci. Lett. 2020, 714, 134545. [Google Scholar] [CrossRef]
- Sova, M.; Saso, L. Natural sources, pharmacokinetics, biological activities and health benefits of hydroxycinnamic acids and their metabolites. Nutrients 2020, 12, 2190. [Google Scholar] [CrossRef]
- Luca, S.V.; Miron, A.; Aprotosoaie, A.C.; Mihai, C.T.; Vochita, G.; Gherghel, D.; Ciocarlan, N.; Skalicka-Woźniak, K. HPLC-DAD-ESI-Q-TOF-MS/MS profiling of Verbascum ovalifolium Donn ex Sims and evaluation of its antioxidant and cytogenotoxic activities. Phytochem. Anal. 2019, 30, 34–45. [Google Scholar] [CrossRef]
- Pires, T.C.S.P.; Dias, M.I.; Barros, L.; Calhelha, R.C.; Alves, M.J.; Oliveira, M.B.P.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Edible flowers as sources of phenolic compounds with bioactive potential. Food Res. Int. 2018, 105, 580–588. [Google Scholar] [CrossRef]
- Yeo, J.D.; Shahidi, F. Identification and quantification of soluble and insoluble-bound phenolics in lentil hulls using HPLC-ESI-MS/MS and their antioxidant potential. Food Chem. 2020, 315, 126202. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N.; Knight, S.; Kuhnert, N. Discriminating between the six isomers of dicaffeoylquinic acid by LC-MSn. J. Agric. Food Chem. 2005, 53, 3821–3832. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N.; Johnston, K.L.; Knight, S.; Kuhnert, N. Hierarchical scheme for LC-MSnidentification of chlorogenic acids. J. Agric. Food Chem. 2003, 51, 2900–2911. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ding, Y. Minireview: Therapeutic potential of myricetin in diabetes mellitus. Food Sci. Hum. Wellness 2012, 1, 19–25. [Google Scholar] [CrossRef]
- Wang, L.; Wu, H.; Yang, F.; Dong, W. The protective effects of myricetin against cardiovascular disease. J. Nutr. Sci. Vitaminol. 2019, 65, 470–476. [Google Scholar] [CrossRef]
- Akindehin, S.; Jung, Y.S.; Kim, S.N.; Son, Y.H.; Lee, I.; Seong, J.K.; Jeong, H.W.; Lee, Y.H. Myricetin Exerts Anti-Obesity Effects through Upregulation of SIRT3 in Adipose Tissue. Nutrients 2018, 10, 1962. [Google Scholar] [CrossRef]
- Niu, Y.; Li, H.; Dong, J.; Wang, H.; Hashi, Y.; Chen, S. Identification of isoflavonoids in Radix Puerariae for quality control using on-line high performance liquid chromatography-diode array detector-electrospray ionization-mass spectrometry coupled with post-column derivatization. Food Res. Int. 2012, 48, 528–537. [Google Scholar] [CrossRef]
- Prasain, J.K.; Reppert, A.; Jones, K.; Moore, D.R.; Barnes, S.; Lila, M.A. Identification of isoflavone glycosides in Pueraria lobata cultures by tandem mass spectrometry. Phytochem. Anal. 2007, 18, 50–59. [Google Scholar] [CrossRef]
- Tala, V.R.S.; Da Silva, V.C.; Rodrigues, C.M.; Nkengfack, A.E.; Dos Santos, L.C.; Vilegas, W. Characterization of proanthocyanidins from Parkia biglobosa (Jacq.) G. Don. (Fabaceae) by flow injection analysis—Electrospray ionization ion trap tandem mass spectrometry and liquid chromatography/electrospray ionization mass spectrometry. Molecules 2013, 18, 2803–2820. [Google Scholar] [CrossRef]
- Li, H.J.; Deinzer, M.L. Tandem mass spectrometry for sequencing proanthocyanidins. Anal. Chem. 2007, 79, 1739–1748. [Google Scholar] [CrossRef]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef] [PubMed]
- Roriz, C.L.; Barros, L.; Carvalho, A.M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Scientific validation of synergistic antioxidant effects in commercialised mixtures of Cymbopogon citratus and Pterospartum tridentatum or Gomphrena globosa for infusions preparation. Food Chem. 2015, 185, 16–24. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Prinsloo, G.; Nogemane, N. The effects of season and water availability on chemical composition, secondary metabolites and biological activity in plants. Phytochem. Rev. 2018, 17, 889–902. [Google Scholar] [CrossRef]
- Tian, X.Y.; Li, M.X.; Lin, T.; Qiu, Y.; Zhu, Y.T.; Li, X.L.; Tao, W.D.; Wang, P.; Ren, X.X.; Chen, L.P. A review on the structure and pharmacological activity of phenylethanoid glycosides. Eur. J. Med. Chem. 2021, 209, 112563. [Google Scholar] [CrossRef]
- Khan, R.A.; Hossain, R.; Roy, P.; Jain, D.; Mohammad Saikat, A.S.; Roy Shuvo, A.P.; Akram, M.; Elbossaty, W.F.; Khan, I.N.; Painuli, S.; et al. Anticancer effects of acteoside: Mechanistic insights and therapeutic status. Eur. J. Pharmacol. 2022, 916, 174699. [Google Scholar] [CrossRef]
- Meddeb, E.; Charni, M.; Ghazouani, T.; Cozzolino, A.; Fratianni, F.; Raboudi, F.; Nazzaro, F.; Fattouch, S. Biochemical and Molecular Study of Carpobrotus edulis Bioactive Properties and Their Effects on Dugesia sicula (Turbellaria, Tricladida) Regeneration. Appl. Biochem. Biotechnol. 2017, 182, 1131–1143. [Google Scholar] [CrossRef]
- Martins, N.; Ferreira, I.C.F.R.; Barros, L.; Carvalho, A.M.; Henriques, M.; Silva, S. Plants used in folk medicine: The potential of their hydromethanolic extracts against Candida species. Ind. Crop. Prod. 2015, 66, 62–67. [Google Scholar] [CrossRef]
- Jin, Y.S. Recent advances in natural antifungal flavonoids and their derivatives. Bioorganic Med. Chem. Lett. 2019, 29, 126589. [Google Scholar] [CrossRef]
- Barros, L.; Pereira, E.; Calhelha, R.C.; Dueñas, M.; Carvalho, A.M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Bioactivity and chemical characterization in hydrophilic and lipophilic compounds of Chenopodium ambrosioides L. J. Funct. Foods 2013, 5, 1732–1740. [Google Scholar] [CrossRef]
- Bessada, S.M.F.; Barreira, J.C.M.; Barros, L.; Ferreira, I.C.F.R.; Oliveira, M.B.P.P. Phenolic profile and antioxidant activity of Coleostephus myconis (L.) Rchb.f.: An underexploited and highly disseminated species. Ind. Crop. Prod. 2016, 89, 45–51. [Google Scholar] [CrossRef]
- Lockowandt, L.; Pinela, J.; Roriz, C.L.; Pereira, C.; Abreu, R.M.V.; Calhelha, R.C.; Alves, M.J.; Barros, L.; Bredol, M.; Ferreira, I.C.F.R. Chemical features and bioactivities of cornflower (Centaurea cyanus L.) capitula: The blue flowers and the unexplored non-edible part. Ind. Crop. Prod. 2019, 128, 496–503. [Google Scholar] [CrossRef]
- Soković, M.; Glamočlija, J.; Marin, P.D.; Brkić, D.; Van Griensven, L.J.L.D. Antibacterial effects of the essential oils of commonly consumed medicinal herbs using an in vitro model. Molecules 2010, 15, 7532–7546. [Google Scholar] [CrossRef] [PubMed]
- Soković, M.; Van Griensven, L.J.L.D. Antimicrobial activity of essential oils and their components against the three major pathogens of the cultivated button mushroom, Agaricus bisporus. Eur. J. Plant Pathol. 2006, 116, 211–224. [Google Scholar] [CrossRef]
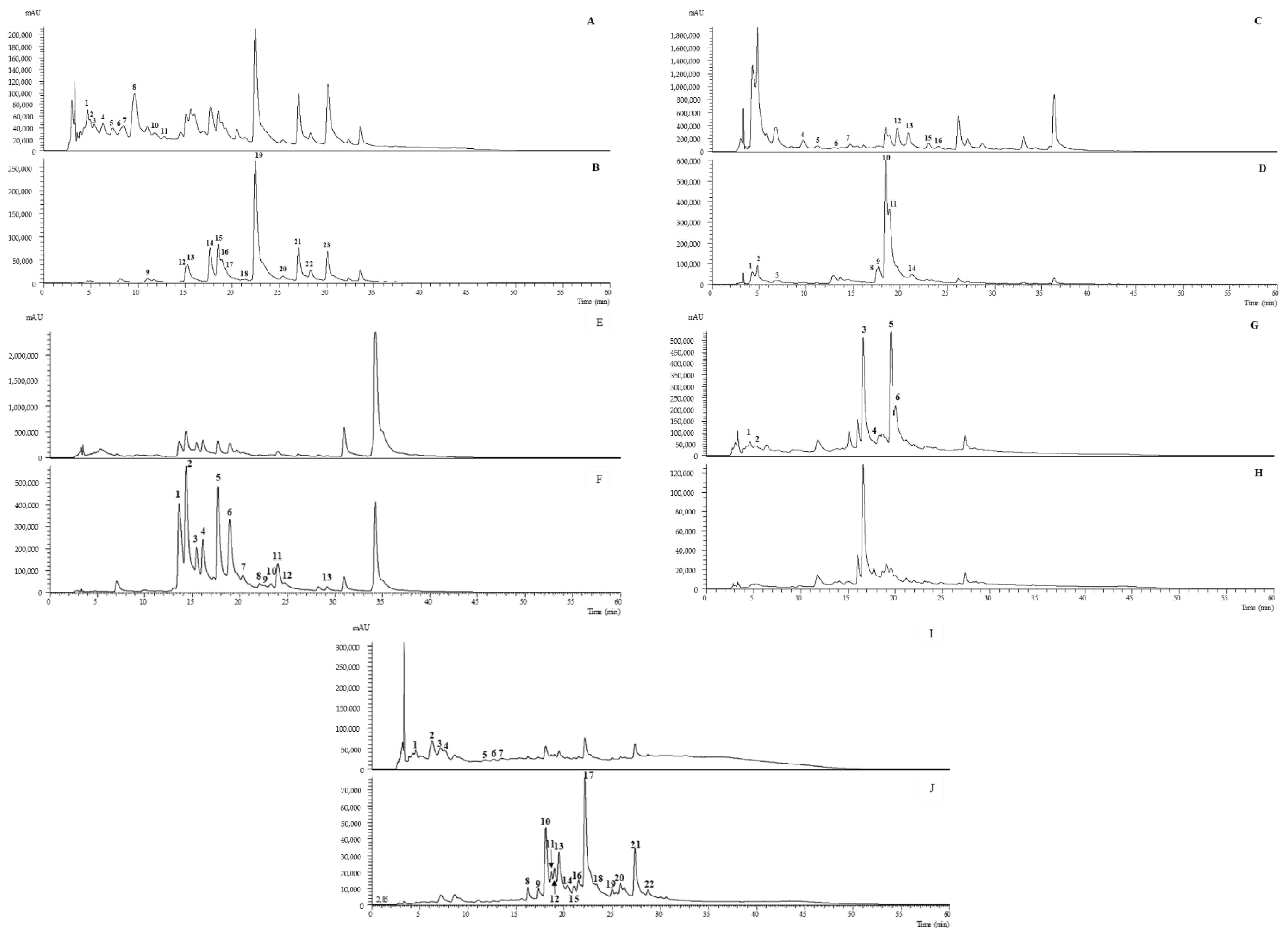
| Peak | Rt (min) | λmax (nm) | [M-H] −(m/z) | MS2 (m/z) | Tentative Identification | Infusion | EtOH:W |
|---|---|---|---|---|---|---|---|
| Calluna vulgaris | |||||||
| 1 cv | 4.64 | 316 | 353 | 191(100),179(45),173(3),135(5) | cis 3-O-Caffeoylquinic acid | 1.01 ± 0.06 * | 0.65 ± 0.01 * |
| 2 cv | 4.91 | 319 | 353 | 191(100),179(38),173(5),135(5) | trans 3-O-Caffeoylquinic acid | 2.04 ± 0.1 * | 0.46 ± 0.01 * |
| 3 cv | 5.34 | 282 | 305 | 219(68),179(100),125(25) | (Epi)gallocatechin | 2.5 ± 0.1 * | 0.516 ± 0.02 * |
| 4 cv | 6.29 | 297 | 337 | 191(7),173(5),163(100),155(5) | 3-O-p-Coumaroylquinic acid | 0.63 ± 0.03 * | 0.27 ± 0.01 * |
| 5 cv | 7.26 | 314 | 325 | 163(100),145(71),119(8) | p-Coumaric acid hexoside | 0.62 ± 0.03 * | 0.17 ± 0.01 * |
| 6 cv | 8.47 | 305 | 337 | 191(7),173(100),163(15),155(5) | cis 4-O-p-Coumaroylquinic acid | 1.08 ± 0.03 * | 0.29 ± 0.01 * |
| 7 cv | 9.52 | 311 | 337 | 191(6),173(100),163(12),155(5) | trans 4-O-p-Coumaroylquinic acid | 2.783 ± 0.001 * | 1.828 ± 0.004 * |
| 8 cv | 10.97 | 279 | 863 | 739(92),713(59),695(100),577(69),575(49),425(14),407(10),289(6),287(12) | β-type (Epi)catechin trimer | 2.9 ± 0.2 * | 1.23 ± 0.03 * |
| 9 cv | 11.77 | 279 | 479 | 317(100) | Myricetin-O-hexoside | 5.3 ± 0.1 * | 5.36 ± 0.03 * |
| 10 cv | 12.82 | 279 | 591 | 573(19),465(62),451(5),439(100),421(33),289(12) | A-Type proanthocyanidin | 2.72 ± 0.01 * | 0.56 ± 0.04 * |
| 11 cv | 14.4 | 281 | 863 | 739(92),713(59),695(100),577(69),575(49),425(14),407(10),289(6),287(12) | β-type (Epi)catechin trimer | 3.2 ± 0.2 * | 0.89 ± 0.01 * |
| 12 cv | 15.16 | 270/310 | 479 | 317(100) | Myricetin-O-hexoside | 5.9 ± 0.1 * | 5.8 ± 0.1 * |
| 13 cv | 15.93 | 287/310 | 449 | 287(100) | Eriodictyol-O-hexoside | 0.9 ± 0.1 * | 0.33 ± 0.03 * |
| 14 cv | 17.63 | 348 | 463 | 317(100) | Myricetin-O-deoxyhexoside | 5.97 ± 0.01 * | 6.7 ± 0.2 * |
| 15 cv | 18.51 | 353 | 463 | 301(100) | Quercetin-3-O-glucoside | 1.34 ± 0.02 * | 1.81 ± 0.02 * |
| 16 cv | 18.87 | 353 | 463 | 301(100) | Quercetin-O-hexoside | 1.09 ± 0.03 * | 1.43 ± 0.05 * |
| 17 cv | 20.48 | 282 | 433 | 271(100) | Naringenin-O-hexoside | 0.316 ± 0.002 | tr |
| 18 cv | 21.33 | 353 | 463 | 301(100) | Quercetin-O-hexoside | 0.937 ± 0.003 * | 1.13 ± 0.01 * |
| 19 cv | 22.41 | 349 | 447 | 301(100) | Quercetin-O-deoxyhexoside | 4.104 ± 0.006 * | 4.9 ± 0.1 * |
| 20 cv | 25.33 | 348 | 505 | 445(34),315(100) | Isorhamnetin derivative | 1.03 ± 0.01 * | 1.13 ± 0.01 * |
| 21 cv | 27.02 | 343 | 431 | 285(100) | Kaempferol-O-deoxyhexoside | 1.64 ± 0.03 * | 1.94 ± 0.02 * |
| 22 cv | 28.31 | 347 | 489 | 447(31),301(100) | Quercetin-acyl-O-deoxyhexoside | 1.25 ± 0.03 * | 1.27 ± 0.02 * |
| 23 cv | 30.04 | 348 | 489 | 447(31),301(100) | Quercetin-acyl-O-deoxyhexoside | 1.4 ± 0.1 * | 1.79 ± 0.05 * |
| Total Phenolic Acids (TPA) | 8.2 ± 0.2 * | 3.67 ± 0.05 * | |||||
| Total Flavan-3-ols (TF3O) | 11.3 ± 0.5 * | 3.20 ± 0.05 * | |||||
| Total Isoflavonoids (TiF) | 18.4 ± 0.2 | 18.2 ± 0.3 | |||||
| Total O-glycosylated Flavonoids (TOF) | 12.79 ± 0.03 * | 15.3 ± 0.3 * | |||||
| Total Phenolic Compounds (TPC) | 51 ± 1 * | 40.4 ± 0.6 * | |||||
| Extraction Yield (η, %) | 9.5 | 27.72 | |||||
| Genista tridentata | |||||||
| 1 g | 4.29 | 291/344sh | 465 | 447(12),375(19),357(5),345(100),327(6),317(5),167(8) | Dihydroquercetin 6-C-hexoside isomer I | 8.5 ± 0.3 * | 10.5 ± 0.1 * |
| 2 g | 4.81 | 292/345sh | 465 | 447(11),375(15),357(10),345(100),327(8),317(5),167(8) | Dihydroquercetin 6-C-hexoside isomer II | 12 ± 1 * | 14.5 ± 0.2 * |
| 3 g | 6.76 | 286 | 479 | 359(100),341(9),221(10),167(9) | Myricetin-C-hexoside | 9.8 ± 0.2 * | 15.04 ± 0.49 * |
| 4 g | 9.66 | 259 | 593 | 431(100),269(13) | Genistein-O-dihexoside | 0.77 ± 0.04 * | 1.004 ± 0.056 * |
| 5 g | 12.85 | 363 | 413 | 311(100),269(5) | Genistein derivative | tr | tr |
| 6 g | 13.57 | 333 | 413 | 311(100),269(5) | Genistein derivative | 0.049 ± 0.002 * | 0.081 ± 0.001 * |
| 7 g | 14.61 | 261/286 | 431 | 311(100),283(32) | Hydroxy-puerarin | 1.2 ± 0.1 * | 1.9 ± 0.1 * |
| 8 g | 17.48 | 347 | 609 | 301(100) | Quercetin-O-deoxyhexosyl-hexoside | 1.12 ± 0.01 * | 1.39 ± 0.02 * |
| 9 g | 17.64 | 350 | 609 | 301(100) | Quercetin-O-deoxyhexosyl-hexoside | 1.24 ± 0.05 * | 1.56 ± 0.04 * |
| 10 g | 18.48 | 354 | 463 | 301(100) | Quercetin-O-hexoside | 2.5 ± 0.1 * | 7.3 ± 0.1 * |
| 11 g | 18.84 | 354 | 463 | 301(100) | Quercetin-O-hexoside | 1.9 ± 0.1 * | 5.03 ± 0.07 * |
| 12 g | 19.67 | 260/331 | 463 | 301(100), 256(5),185(12) | Ellagic acid hexoside | 5.1 ± 0.4 * | 10.2 ± 0.4 * |
| 13 g | 20.79 | 261/298 | 461 | 299(100) | Chrysoeriol-O-hexoside | 1.14 ± 0.03 * | 1.3 ± 0.1 * |
| 14 g | 21.22 | 335 | 463 | 301(100) | Quercetin-O-hexoside | 1.08 ± 0.01 * | 1.5 ± 0.1 * |
| 15 g | 22.96 | 260/286 | 461 | 299(100) | Chrysoeriol-O-hexoside | 0.97 ± 0.01 * | 1.3 ± 0.1 * |
| 16 g | 23.99 | 260/286 | 473 | 311(13),269(100) | O-acetylgenistein | tr | 0.32 ± 0.02 |
| Total Phenolic Acids (TPA) | 5.1 ± 0.4 * | 10.2 ± 0.4 * | |||||
| Total Isoflavonoids (TiF) | 2.1 ± 0.1 * | 3.3 ± 0.1 * | |||||
| Total O-glycosylated Flavonoids (TOF) | 41 ± 1 * | 59 ± 1 * | |||||
| Total Phenolic Compounds (TPC) | 48 ± 2 * | 73 ± 1 * | |||||
| Extraction Yield (η, %) | 9.09 | 21.90 | |||||
| Cytisus multiflorus | |||||||
| 1 cm | 13.6 | 354 | 625 | 463(100),301(50) | Quercetin-O-dihexoside | 7.2 ± 0.3 | 7.01 ± 0.17 |
| 2 cm | 14.35 | 348 | 579 | 459(23),429(73),357(71),327(100),309(66) | 2’’-O-Pentosyl-8-C-hexoside luteolin | 59 ± 1 * | 62 ± 1 * |
| 3 cm | 15.48 | 340 | 563 | 443(5),413(100),323(6),311(7),293(86) | 2″-O-Pentosyl-8-C-hexoside apigenin isomer I | 8.7 ± 0.2 * | 7.9 ± 0.2 * |
| 4 cm | 16.07 | 336 | 563 | 443(4),413(100),323(5),311(11),293(79) | 2″-O-Pentosyl-8-C-hexoside apigenin isomer II | 9.8 ± 0.4 * | 9.3 ± 0.3 * |
| 5 cm | 17.69 | 353 | 609 | 301(100) | Quercetin-O-deoxyhexosyl-hexoside | 1.9 ± 0.1 * | 7.1 ± 0.4 * |
| 6 cm | 18.94 | 249 | 463 | 301(100) | Quercetin-O-hexoside | 6.0 ± 0.1 | 6.04 ± 0.03 |
| 7 cm | 20.32 | 340 | 707 | 563(34),413(43),293(10) | 6’’-O-(3-hydroxy-3-methylglutaroyl)-2’’-O-pentosyl-C-hexosyl-apigenin | 10.6 ± 0.4 * | 3.6 ± 0.2 * |
| 8 cm | 22.18 | 342 | 623 | 315(100) | Isorhamnetin-O-deoxyhexosyl-hexoside | 1.9 ± 0.1 * | 1.5 ± 0.1 * |
| 9 cm | 23.33 | 335 | 477 | 315(100) | Isorhamnetin-O-hexoside | 2.1 ± 0.1 * | 1.35 ± 0.02 * |
| 10 cm | 23.13 | 334 | 431 | 269(00) | Apigenin-O-hexoside | 2.5 ± 0.1 * | 2.7 ± 0.1 * |
| 11 cm | 23.98 | 346 | 533 | 489(100),285(26) | Luteolin-O-malonyl-hexoside | 1.39 ± 0.05 * | 2.7 ± 0.1 * |
| 12 cm | 24.7 | 347 | 533 | 489(100),285(26) | Luteolin-O-malonyl-hexoside | 2.6 ± 0.1 * | 1.461 ± 0.1 * |
| 13 cm | 28.33 | 333 | 473 | 269(100) | Apigenin-O-acylhexoside | 2.05 ± 0.02 * | 1.9 ± 0.1 * |
| Total O-glycosylated Flavonoids (TOF) | 28 ± 1 * | 32 ± 1 * | |||||
| Total C-glycosylated Flavonoids (TCF) | 88.7 ± 0.4 * | 83 ± 2 * | |||||
| Total Flavonoid Compounds (TFC) | 116 ± 1 | 115 ± 3 | |||||
| Extraction Yield (η, %) | 11.86 | 31.73 | |||||
| Verbascum sinuatum | |||||||
| 1 vs | 4.63 | 275 | 191 | 173(100),111(15) | Quinic acid | 1.02 ± 0.01 * | 1.25 ± 0.03 * |
| 2 vs | 5.21 | 328 | 341 | 179(100),161(14) | Caffeic acid hexoside | 0.112 ± 0.003 * | 0.281 ± 0.004 * |
| 3 vs | 16.58 | 325 | 623 | 461(100),315(34),179(8),161(9),153(10) | Verbascoside | 12.4 ± 0.4 * | 13.1 ± 0.3 * |
| 4 vs | 17.7 | 327 | 623 | 461(100),315(34),179(11),161(8),153(13) | Isoverbascoside | 2.5 ± 0.1 * | 2.41 ± 0.03 * |
| 5 vs | 19.57 | 314 | 637 | 475(100)309(71),205(8) | p-Coumaroyl-6-O-rhamnosyl aucubin isomer I | 2.7 ± 0.1 * | 6.4 ± 0.1 * |
| 6 vs | 20.01 | 312 | 637 | 475(100)309(65),205(10) | p-Coumaroyl-6-O-rhamnosyl aucubin Isomer II | 1.29 ± 0.03 * | 2.3 ± 0.1 * |
| Total Phenolic Acids (TPA) | 1.14 ± 0.01 * | 1.53 ± 0.03 * | |||||
| Total Iridoid Glycosides (TiG) | 4.01 ± 0.11 * | 8.7 ± 0.2 * | |||||
| Total Phenylethanoid Glycosides (TpG) | 14.9 ± 0.4 * | 15.4 ± 0.3 * | |||||
| Total Phenolic Compounds (TPC) | 25.2 ± 0.2 * | 36 ± 1 * | |||||
| Extraction Yield (η, %) | 20.29 | 34.34 | |||||
| Carpobrotus edulis | |||||||
| 1 ce | 6.27 | 308 | 337 | 191(10),163(100),119(13) | 3-O-p-Coumaroylquinic acid | 0.31 ± 0.01 * | 1.49 ± 0.01 * |
| 2 ce | 7.16 | 324 | 353 | 191(100),179(14),135(10) | cis 5-O-Caffeoylquinic acid | 0.57 ± 0.02 * | 1.91 ± 0.01 * |
| 3 ce | 7.59 | 324 | 353 | 191(100),179(14),135(10) | trans 5-O-Caffeoylquinic acid | 0.49 ± 0.02 * | 1.8 ± 0.1 * |
| 4 ce | 8.61 | 326 | 355 | 193(100),175(34),149(5) | Feruloyl-hexoside | 0.108 ± 0.003 * | 0.38 ± 0.01 * |
| 5 ce | 11.78 | 308 | 337 | 191(100),163(10),119(13) | cis 5-O-p-Coumaroylquinic acid | 0.11 ± 0.01 * | 0.57 ± 0.03 * |
| 6 ce | 12.66 | 310 | 337 | 191(100),163(10),119(13) | trans 5-O-p-Coumaroylquinic acid | 0.09 ± 0.01 * | 0.56 ± 0.01 * |
| 7 ce | 13.48 | 314 | 367 | 193(5),191(100),173(8) | 5-O-Feruloylquinic acid | 0.097 ± 0.0001 * | 0.357 ± 0.003 * |
| 8 ce | 16.22 | 325 | 771 | 639(92),330(100),315(32),287(8) | Laricitrin-O-pentosyl-O-deoxyhexosyl-hexoside | 0.87 ± 0.01 * | 1.01 ± 0.01 * |
| 9 ce | 17.3 | 322 | 769 | 623(23),315(100) | Isorhamnetin-O-deoxyhexosyl-hexosyl-O-deoxyhexoside | 0.863 ± 0.004 * | 0.99 ± 0.01 * |
| 10 ce | 18.06 | 349 | 639 | 331(100),315(23),287(10) | Laricitrin-O-deoxyhexosyl-hexoside | 0.95 ± 0.01 * | 1.37 ± 0.02 * |
| 11 ce | 18.63 | 349 | 639 | 331(100),315(23),287(10) | Laricitrin-O-deoxyhexosyl-hesxoside | 0.88 ± 0.01 * | 1.109 ± 0.002 * |
| 12ce | 19.02 | 357 | 623 | 315(100) | Isorhamnetin-O-deoxyhexosyl-hexoside | 0.881 ± 0.003 * | 1.12 ± 0.03 * |
| 13 ce | 19.43 | 271/353 | 785 | 653(34),345(100),330(35),287(21) | Syringetin-O-pentosyl-O-deoxyhexosylhexoside | 0.95 ± 0.01 * | 1.22 ± 0.01 * |
| 14 ce | 20.26 | 275/348 | 785 | 653(34),345(100),330(35),287(21) | Syringetin-O-pentosyl-O-deoxyhexosylhexoside | 0.867 ± 0.002 * | 1.08 ± 0.02 * |
| 15 ce | 20.99 | 276/347 | 639 | 345(100),330(34),287(3) | Syringetin-O-pentosyl-hexoside | 0.865 ± 0.002 * | 1.04 ± 0.01 * |
| 16 ce | 21.51 | 272/344 | 623 | 315(100) | Isorhamnetin-O-deoxyhexosyl-hexoside | 0.855 ± 0.004 * | 1.053 ± 0.004 * |
| 17 ce | 22.14 | 275/357 | 653 | 345(100),330(35),287(21) | Syringetin-O-deoxyhexosyl-hexoside | 0.992 ± 0.004 * | 1.81 ± 0.01 * |
| 18 ce | 23.41 | 276/350 | 653 | 345(100),330(35),287(21) | Syringetin-O-deoxyhexosyl-hexoside | 0.859 ± 0.002 * | 1.03 ± 0.01 * |
| 19 ce | 25.02 | 279/339 | 947 | 771(34),639(92),331(100),315(32),287(8) | Laricitrin-O-hexuronosyl-O-pentosyl-O-deoxyhexosyl-hexoside | 0.854 ± 0.001 * | 1.021 ± 0.002 * |
| 20 ce | 25.82 | 272/362 | 549 | 345(100),330(28),287(5) | Syringetin-O-acetyl-hexoside | 0.855 ± 0.001 * | 1.01 ± 0.02 * |
| 21 ce | 27.33 | 275/341 | 961 | 799(34),345(100),330(23),287(10) | Syringetin-O-hexosyl-O-dideoxyhexosyl-hexoside | 0.8811 ± 0.0004 * | 1.28 ± 0.01 * |
| 22 ce | 28.69 | 279/348 | 815 | 653(23),345(100),330(30),287(10) | Syringetin-O-hexosyl-O-deoxyhexosyl-hexoside | 0.846 ± 0.003 * | 1.011 ± 0.002 * |
| Total Phenolic Acids (TPA) | 1.77 ± 0.04 * | 7.1 ± 0.1 * | |||||
| Total O-glycosylated Flavonoids (TOF) | 13.27 ± 0.04 * | 17.2 ± 0.1 * | |||||
| Total Phenolic Compounds (TPC) | 15.04 ± 0.08 * | 24.3 ± 0.3 * | |||||
| Extraction Yield (η, %) | 36.67 | 16.67 | |||||
| C. vulgaris | G. tridentata | C. multiflorus | V. sinuatum | C. edulis | Positive control | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| I | EtOH:W | I | EtOH:W | I | EtOH:W | I | EtOH:W | I | EtOH:W | ||
| Antioxidant (IC50, µg/mL) | Trolox | ||||||||||
| TBARS | 18 ± 1 * | 10.2 ± 0.3 * | 5.3 ± 0.1 * | 3.19 ± 0.02 * | 51 ± 3 * | 3.7 ± 0.1 * | 17.4 ± 0.9 * | 4.2 ± 0.2 * | 24.0 ± 0.8 * | 1.20 ± 0.05 * | 23 ± 2 |
| OxHLIA | n.a | n.a | 78 ± 6 | 76 ± 5 | 109 ± 9 | n.a | n.a | n.a | n.a | 132 ± 6 | 85 ± 2 |
| Cytotoxic and hepatotoxicity activity (GI50, µg/mL) | Ellipticine | ||||||||||
| NCI H460 | 334.6 ± 9.1 * | 219.7 ± 10.8 * | 142.7 ± 5.3 * | 160.5 ± 5.3 * | 246.8 ± 5.6 * | 314.3 ± 8.7 * | 92.1 ± 3.9 * | 140.0 ± 5.8 * | 272.1 ± 24.9 | 256.4 ± 10.9 | 1.0 ± 0.1 |
| Hela | 270.3 ± 8.2 * | 69.6 ± 6.6 * | 83.2 ± 6.5 | 102.9 ± 10.6 | 133.3 ± 9.6 | 147.9 ± 5.1 | 59.1 ± 3.1 * | 101.1 ± 4.9 * | 341.7 ± 15.1 * | 295.5 ± 16.2 | 1.91 ± 0.06 |
| MCF-7 | 322.1 ± 4.3 * | 205.3 ± 9.6 * | 129.1 ± 6.3 * | 146.8 ± 6.5 * | 235.8 ± 8.5 * | 278.7 ± 8.1 * | 74.8 ± 3.7 * | 125.9 ± 6.5 * | 289.7 ± 12.3 * | 260.5 ± 7.9 * | 0.91 ± 0.04 |
| HepG2 | 296.4 ± 22.2 * | 79.4 ± 4.5 * | 123.1 ± 19.1 | 132.4 ± 8.5 | 216.1 ± 11.8 * | 263.4 ± 21.2 * | 65.4 ± 2.9 * | 172.2 ± 22.7 * | 306.3 ± 27.1 * | 210.1 ± 16.7 * | 1.1 ± 0.2 |
| PLP2 | >400 | >400 | >400 | >400 | >400 | >400 | 223.1 ± 15.4 | >400 | >400 | >400 | 3.2 ± 0.7 |
| Anti-inflammatory activity (IC50, µg/mL) | DX | ||||||||||
| 229.2 ± 7.9 | >400 | 144.4 ± 2.2 * | 207.4 ± 15.5 * | 293.2 ± 11.8 | >400 | 121.1 ± 3.9 * | 130.1 ± 2.8 * | >400 | 237.9 ± 5.8 | 6 ± 1 | |
| C. edulis | G. tridentata | V. sinuatum | C. multiflorus | C. vulgaris | Controls | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| I | EtOH:W | I | EtOH:W | I | EtOH:W | I | EtOH:W | I | EtOH:W | St | Kt | |||||||||||||
| Bacteria | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC |
| Staphylococcus aureus | 0.25 | 0.50 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.50 | 0.25 | 0.50 | 0.25 | 0.25 | 0.25 | 0.50 | 0.25 | 0.25 | 0.25 | 0.50 | 0.50 | 0.50 | 0.1 | 0.2 | - | - |
| Bacillus cereus | 1.00 | 1.00 | 0.50 | 0.50 | 1.00 | 2.00 | 0.50 | 1.00 | 1.00 | 1.00 | 0.50 | 0.50 | 2.00 | 2.00 | 1.00 | 2.00 | 1.00 | 1.00 | 1.00 | 1.00 | 0.025 | 0.05 | - | - |
| Micrococcus flavus | 1.00 | 2.00 | 0.50 | 1.00 | 2.00 | 4.00 | 1.00 | 2.00 | 1.00 | 2.00 | 1.00 | 2.00 | 2.00 | 4.00 | 1.00 | 1.00 | 4.00 | 4.00 | 1.00 | 2.00 | 0.05 | 0.1 | - | - |
| Listeria monocytogenes | 0.50 | 1.00 | 0.50 | 0.50 | 1.00 | 2.00 | 0.50 | 0.50 | 1.00 | 1.00 | 1.00 | 2.00 | 0.50 | 1.00 | 0.50 | 1.00 | 2.00 | 4.00 | 1.00 | 1.00 | 0.2 | 0.3 | - | - |
| Enterobacter cloacae | 0.50 | 1.00 | 1.00 | 1.00 | 1.00 | 2.00 | 1.00 | 2.00 | 0.50 | 1.00 | 1.00 | 2.00 | 1.00 | 2.00 | 2.00 | 2.00 | 1.00 | 1.00 | 2.00 | 2.00 | 0.025 | 0.05 | - | - |
| Salmonella Typhimurium | 0.50 | 1.00 | 0.50 | 0.50 | 1.00 | 2.00 | 0.50 | 1.00 | 1.00 | 2.00 | 1.00 | 2.00 | 0.50 | 1.00 | 1.00 | 1.00 | 2.00 | 2.00 | 0.50 | 1.00 | 0.1 | 0.2 | - | - |
| Fungi | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC |
| Aspergillus fumigatus | 0.50 | 1.00 | 0.50 | 1.00 | 1.00 | 1.00 | 0.25 | 0.50 | 0.50 | 1.00 | 0.25 | 0.50 | 0.50 | 0.50 | 0.25 | 0.25 | 1.00 | 1.00 | 0.50 | 0.50 | - | - | 0.2 | 0.5 |
| Aspergillus versicolor | 0.50 | 1.00 | 0.50 | 1.00 | 0.50 | 1.00 | 0.25 | 0.50 | 0.50 | 1.00 | 0.25 | 0.50 | 0.25 | 0.50 | 0.25 | 0.25 | 0.50 | 1.00 | 0.50 | 1.00 | - | - | 0.2 | 0.5 |
| Aspergillus niger | 0.50 | 1.00 | 0.50 | 1.00 | 0.50 | 1.00 | 0.25 | 0.50 | 0.50 | 1.00 | 0.50 | 1.00 | 0.25 | 0.50 | 0.25 | 0.25 | 0.50 | 1.00 | 1.00 | 2.00 | - | - | 0.2 | 0.5 |
| Penicillium funiculosum | 0.50 | 0.50 | 0.50 | 1.00 | 0.50 | 1.00 | 0.25 | 0.50 | 0.50 | 1.00 | 0.12 | 0.50 | 0.25 | 0.50 | 0.50 | 0.50 | 0.50 | 1.00 | 1.00 | 2.00 | - | - | 0.2 | 0.5 |
| Penicillium aurantiogriseum | 0.50 | 1.00 | 1.00 | 2.00 | 0.50 | 1.00 | 0.50 | 1.00 | 0.50 | 1.00 | 0.50 | 1.00 | 0.25 | 0.50 | 0.50 | 1.00 | 0.50 | 1.00 | 1.00 | 2.00 | - | - | 0.2 | 0.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Oliveira, P.; Carreira-Casais, A.; Pereira, E.; Dias, M.I.; Pereira, C.; Calhelha, R.C.; Stojković, D.; Sokovic, M.; Simal-Gandara, J.; Prieto, M.A.; et al. From Tradition to Health: Chemical and Bioactive Characterization of Five Traditional Plants. Molecules 2022, 27, 6495. https://doi.org/10.3390/molecules27196495
Garcia-Oliveira P, Carreira-Casais A, Pereira E, Dias MI, Pereira C, Calhelha RC, Stojković D, Sokovic M, Simal-Gandara J, Prieto MA, et al. From Tradition to Health: Chemical and Bioactive Characterization of Five Traditional Plants. Molecules. 2022; 27(19):6495. https://doi.org/10.3390/molecules27196495
Chicago/Turabian StyleGarcia-Oliveira, Paula, Anxo Carreira-Casais, Eliana Pereira, Maria Inês Dias, Carla Pereira, Ricardo C. Calhelha, Dejan Stojković, Marina Sokovic, Jesus Simal-Gandara, Miguel A. Prieto, and et al. 2022. "From Tradition to Health: Chemical and Bioactive Characterization of Five Traditional Plants" Molecules 27, no. 19: 6495. https://doi.org/10.3390/molecules27196495
APA StyleGarcia-Oliveira, P., Carreira-Casais, A., Pereira, E., Dias, M. I., Pereira, C., Calhelha, R. C., Stojković, D., Sokovic, M., Simal-Gandara, J., Prieto, M. A., Caleja, C., & Barros, L. (2022). From Tradition to Health: Chemical and Bioactive Characterization of Five Traditional Plants. Molecules, 27(19), 6495. https://doi.org/10.3390/molecules27196495