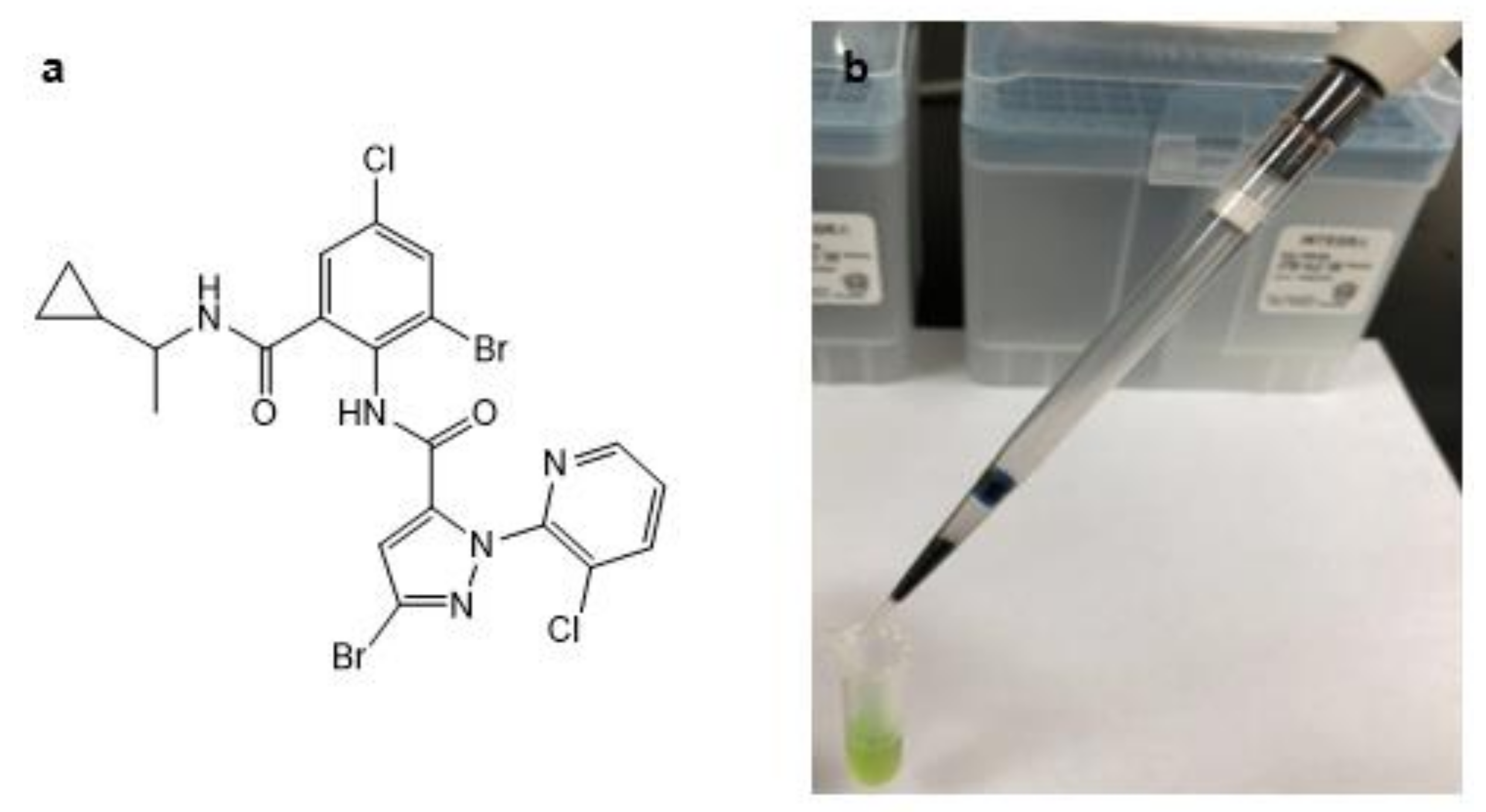
Determination of Cyclaniliprole in Fruits and Vegetables Using Disposable Pipette Extraction Cleanup and Ultrahigh-Performance Liquid Chromatography-Tandem Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. MS/MS Optimization
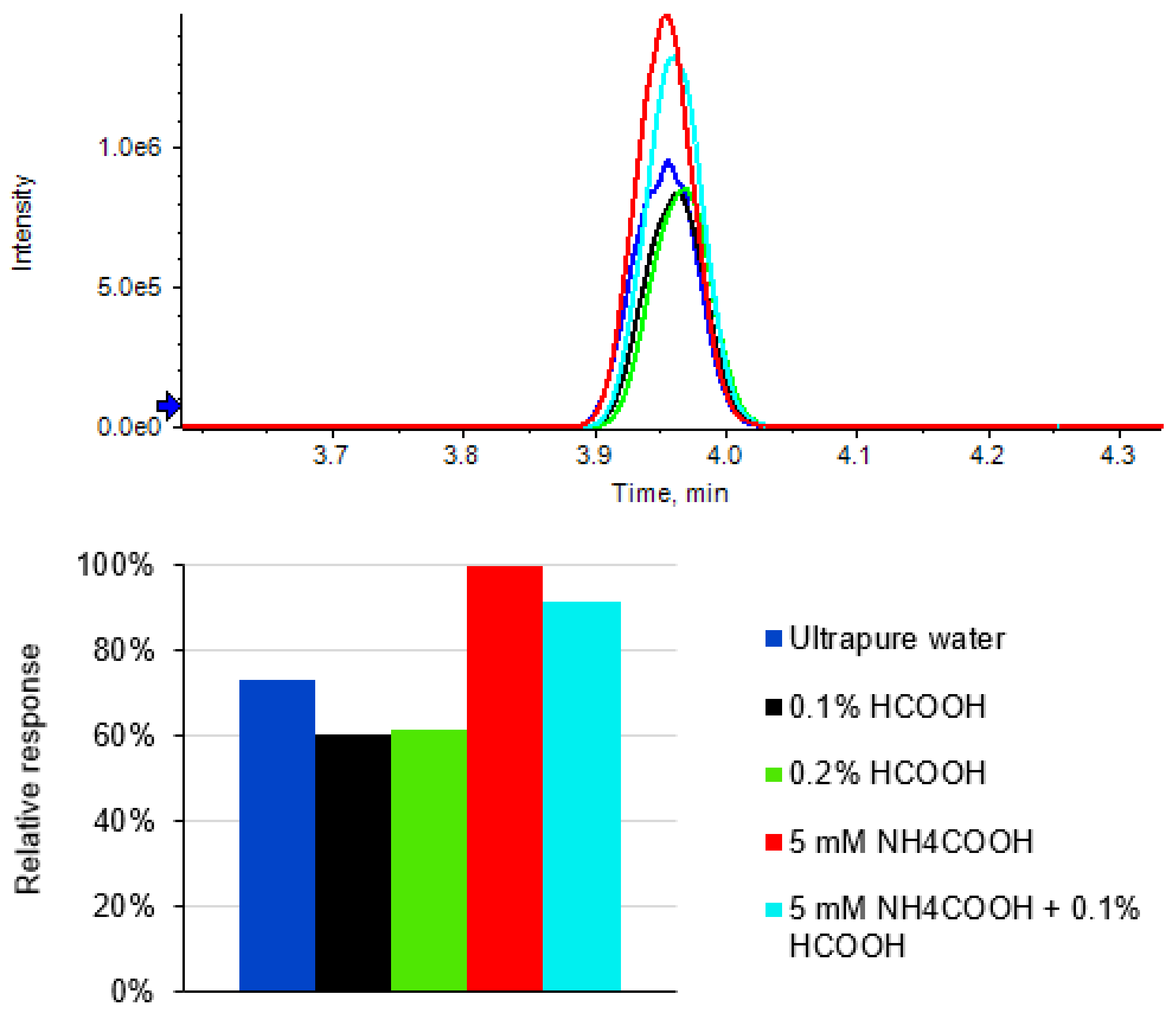
2.2. Chromatography Optimization
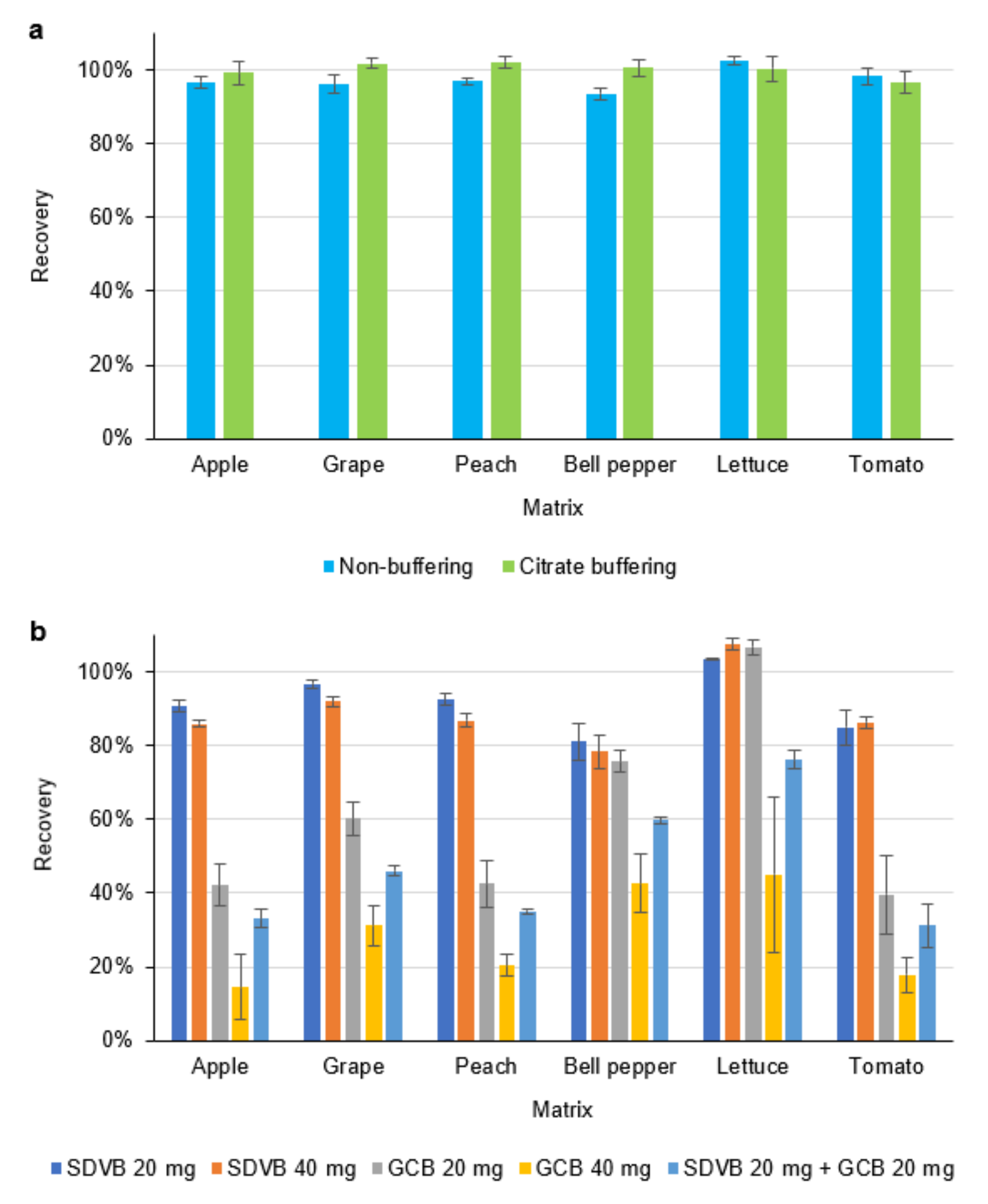
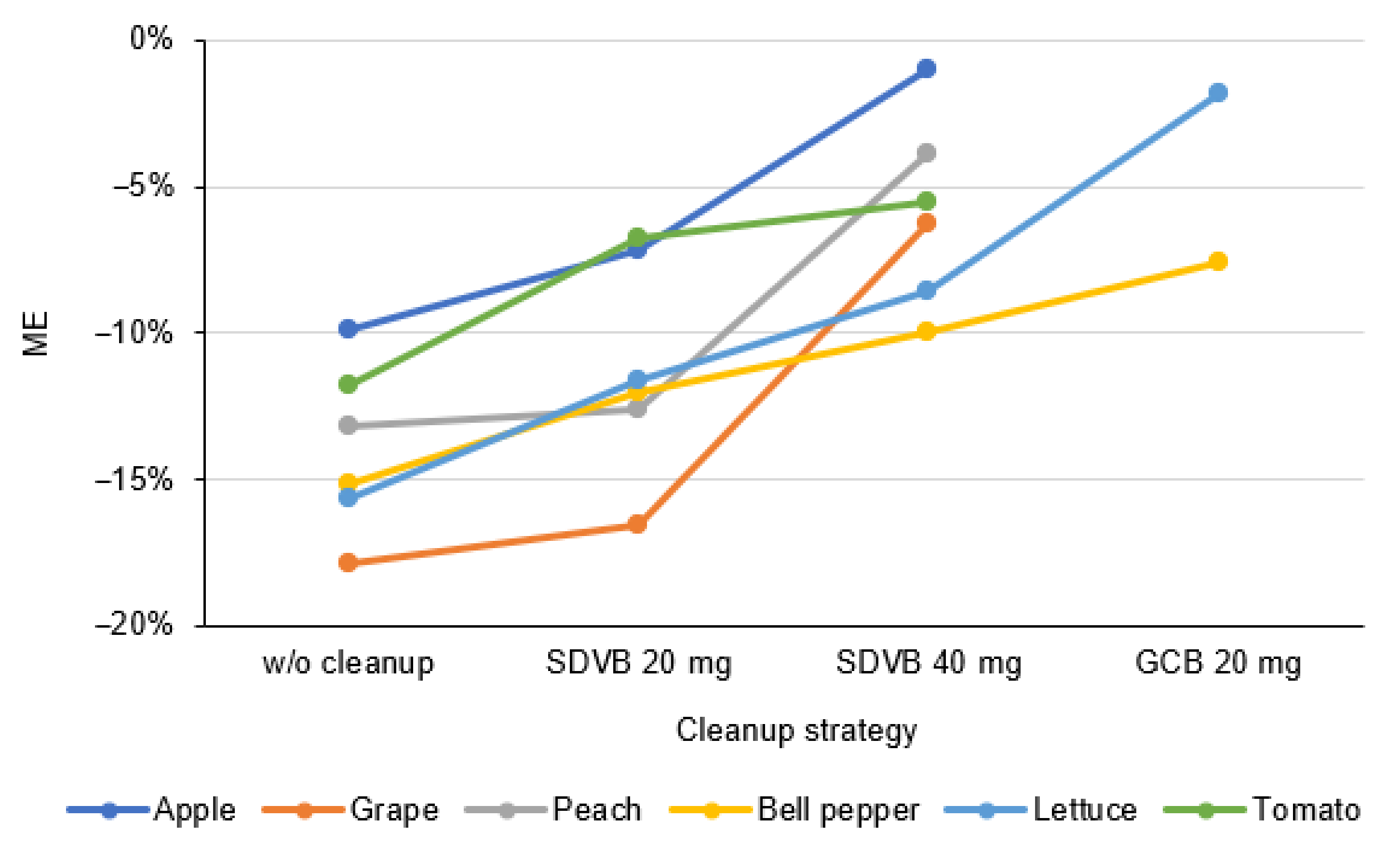
2.3. Sample Preparation Optimization
2.4. Method Validation
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Sample Preparation
3.3. Instrumentation
3.4. Method Validation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- IUPAC Pesticide Properties Database. Available online: http://sitem.herts.ac.uk/aeru/iupac/Reports/2632.htm (accessed on 1 May 2022).
- Cordova, D.; Benner, E.A.; Sacher, M.D.; Rauh, J.J.; Sopa, J.S.; Lahm, G.P.; Selby, T.; Stevenson, T.; Flexner, L.; Gutteridge, S.; et al. Anthranilic diamides: A new class of insecticides with a novel mode of action, ryanodine receptor activation. Pestic. Biochem. Physiol. 2006, 84, 196–214. [Google Scholar] [CrossRef]
- Hamamoto, T. Chemical Structure Advantage of Cyclaniliprole Against the Diamides Resistant Diamondback Moth, Plutella Xylostella. In Proceedings of the International Congress of Entomology, Orlando, FL, USA, 25–30 September 2016. [Google Scholar]
- Pereira, R.M.; Neto, D.A.; Amado, D.; Durigan, M.R.; Franciscatti, R.A.; Mocheti, M.; Omoto, C. Baseline susceptibility and frequency of resistance to diamide insecticides in Helicoverpa armigera (Lepidoptera: Noctuidae) populations in Brazil. Crop Prot. 2020, 137, 105266. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, G.; Zhong, L.; Zhang, F.; Bai, Q.; Zheng, X.; Lu, Z. Resistance monitoring of Chilo suppressalis (Walker) (Lepidoptera: Crambidae) to chlorantraniliprole in eight field populations from east and central China. Crop Prot. 2017, 100, 196–202. [Google Scholar] [CrossRef]
- Wang, X.; Wu, Y. High Levels of Resistance to Chlorantraniliprole Evolved in Field Populations of Plutella xylostella. J. Econ. Entomol. 2012, 105, 1019–1023. [Google Scholar] [CrossRef] [PubMed]
- Oerke, E.C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Peer review of the pesticide risk assessment of the active substance cyclaniliprole. EFSA J. 2016, 14, 4452. [Google Scholar]
- Anastassiades, M.; Lehotay, S.J.; Stajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [CrossRef]
- Lehotay, S.J.; Mastovská, K.; Lightfield, A.R. Use of buffering and other means to improve results of problematic pesticides in a fast and easy method for residue analysis of fruits and vegetables. J. AOAC Int. 2005, 88, 615–629. [Google Scholar] [CrossRef]
- Anastassiades, M.; Scherbaum, E.; Tasdelen, B.; Stajnbaher, D. Recent Developments in QuEChERS Methodology for Pesticide Multiresidue Analysis. In Pesticide Chemistry: Crop Protection, Public Health, Environmental Safety; Ohkawa, H., Miyagawa, H., Lee, P.W., Eds.; Wiley: Weinheim, Germany, 2007; pp. 439–458. [Google Scholar]
- Association of Official Analytical Chemists (AOAC). AOAC Official Method 2007.01 Pesticide Residues in Foods by Acetonitrile Extraction and Partitioning with Magnesium Sulfate. J. AOAC Int. 2007, 90, 485–520. [Google Scholar]
- EN 15662:2008; Foods of Plant Origin−Determination of Pesticide Residues Using GC-MS and/or LC-MS/MS following Acetonitrile Extraction and Partitioning and Clean-Up by Dispersive SPE−QuEChERS-Method. European Committee for Standardization: Brussels, Belgium, 2008.
- Shin, D.; Kim, J.; Kang, H.S. Simultaneous determination of multi-pesticide residues in fish and shrimp using dispersive-solid phase extraction with liquid chromatography-tandem mass spectrometry. Food Control 2020, 120, 107552. [Google Scholar] [CrossRef]
- Acosta-Dacal, A.; Rial-Berriel, C.; Diaz-Diaz, R.; Bernal-Suarez, M.D.; Luzardo, O.P. Optimization and validation of a QuEChERS-based method for the simultaneous environmental monitoring of 218 pesticide residues in clay loam soil. Sci. Total Environ. 2021, 753, 142015. [Google Scholar] [CrossRef]
- Gawel, M.; Kiljanek, T.; Niewiadowska, A.; Semeniuk, S.; Goliszek, M.; Burek, O.; Posyniak, A. Determination of neonicotinoids and 199 other pesticide residues in honey by liquid and gas chromatography coupled with tandem mass spectrometry. Food Chem. 2019, 282, 36–47. [Google Scholar] [CrossRef]
- Cao, J.L.; Zheng, Y.X.; Kaium, A.; Liu, X.G.; Xu, J.; Dong, F.S.; Wu, X.H.; Zheng, Y.Q. A comparative study of biochar, multiwalled carbon nanotubes and graphitized carbon black as QuEChERS absorbents for the rapid determination of six triazole fungicides by UPLC-MS/MS. Int. J. Environ. Anal. Chem. 2019, 99, 209–223. [Google Scholar] [CrossRef]
- Xu, X.; Xu, X.Y.; Han, M.; Qiu, S.T.; Hou, X. Development of a modified QuEChERS method based on magnetic multiwalled carbon nanotubes for the simultaneous determination of veterinary drugs, pesticides and mycotoxins in eggs by UPLC-MS/MS. Food Chem. 2019, 276, 419–426. [Google Scholar] [CrossRef]
- Bordin, D.C.M.; Alves, M.N.R.; de Campos, E.G.; De Martinis, B.S. Disposable pipette tips extraction: Fundamentals, applications and state of the art. J. Sep. Sci. 2016, 39, 1168–1172. [Google Scholar] [CrossRef]
- Lu, Z.; Fang, N.; Zhang, Z.; Hou, Z.; Lu, Z.; Li, Y. Residue analysis of fungicides fenpicoxamid, isofetamid, and mandestrobin in cereals using zirconium oxide disposable pipette extraction clean-up and ultrahigh-performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2020, 1620, 461004. [Google Scholar] [CrossRef]
- Teixeira, R.A.; Flores, D.H.; da Silva, R.C.S.; Dutra, F.V.A.; Borges, K.B. Pipette-tip solid-phase extraction using poly(1-vinylimidazole-co-trimethylolpropane trimethacrylate) as a new molecularly imprinted polymer in the determination of avermectins and milbemycins in fruit juice and water samples. Food Chem. 2018, 262, 86–93. [Google Scholar]
- Mastrianni, K.R.; Metavarayuth, K.; Brewer, W.E.; Wang, Q. Analysis of 10 β-agonists in pork meat using automated dispersive pipette extraction and LC-MS/MS. J. Chromatogr. B 2018, 1084, 64–68. [Google Scholar] [CrossRef]
- Li, Z.; Li, Y.; Liu, X.; Li, X.; Zhou, L.; Pan, C. Multiresidue Analysis of 58 Pesticides in Bean Products by Disposable Pipet Extraction (DPX) Cleanup and Gas Chromatography–Mass Spectrometry Determination. J. Agric. Food Chem. 2012, 60, 4788–4798. [Google Scholar] [CrossRef]
- Guan, H.; Brewer, W.E.; Garris, S.T.; Morgan, S.L. Disposable pipette extraction for the analysis of pesticides in fruit and vegetables using gas chromatography/mass spectrometry. J. Chromatogr. A 2010, 1217, 1867–1874. [Google Scholar] [CrossRef]
- Patring, J.D.; Jastrebova, J.A. Application of liquid chromatography–electrospray ionisation mass spectrometry for determination of dietary folates: Effects of buffer nature and mobile phase composition on sensitivity and selectivity. J. Chromatogr. A 2007, 1143, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M. The effect of the mobile phase additives on sensitivity in the analysis of peptides and proteins by high-performance liquid chromatography–electrospray mass spectrometry. J. Chromatogr. B 2005, 825, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Hennion, M.-C. Solid-phase extraction: Method development, sorbents, and coupling with liquid chromatography. J. Chromatogr. A 1999, 856, 3–54. [Google Scholar] [CrossRef]
- Perestrelo, R.; Silva, P.; Porto-Figueira, P.; Pereira, J.A.M.; Silva, C.; Medina, S.; Camara, J.S. QuEChERS-Fundamentals, relevant improvements, applications and future trends. Anal. Chim. Acta 2019, 1070, 1–28. [Google Scholar] [CrossRef]
- Lehotay, S.J.; Majors, R.E.; Anastassiades, M. The QuEChERS revolution. LCGC Asia Pac. 2010, 13, 30–36. [Google Scholar]
- European Commission SANTE/11312/2022. Guidance Document on Analytical Quality Control and Method Validation Procedures for Pesticide Residues and Analysis in Food and Feed. 2022. Available online: https://ec.europa.eu/food/system/files/2022-02/pesticides_mrl_guidelines_wrkdoc_2021-11312.pdf (accessed on 1 May 2022).
- China Pesticide Information Network. Available online: http://www.chinapesticide.org.cn/hysj/index.jhtml (accessed on 1 May 2022).
- Niessen, W.M.A.; Manini, P.; Andreoli, R. Matrix effects in quantitative pesticide analysis using liquid chromatography-mass spectrometry. Mass Spectrom. Rev. 2006, 25, 881–899. [Google Scholar] [CrossRef]
- Stahnke, H.; Kittlaus, S.; Kempe, G.; Alder, L. Reduction of matrix effects in liquid chromatography-electrospray ionization-mass spectrometry by dilution of the sample extracts: How much dilution is needed? Anal. Chem. 2012, 84, 1474–1482. [Google Scholar] [CrossRef]

| Matrix | Calibration Equation | r | ME (%) | LOQ (mg kg−1) | MRL (mg kg−1) (EU/US) |
|---|---|---|---|---|---|
| Acetonitrile | y = 6472x − 148 | 0.9987 | - | 0.01 | - |
| Apple | y = 6407x + 384 | 0.9980 | −1.0 | 0.01 | 0.01/0.3 |
| Grape | y = 6062x + 335 | 0.9947 | −6.3 | 0.01 | 0.01/0.8 |
| Peach | y = 6218x + 923 | 0.9934 | −3.9 | 0.01 | 0.01/1 |
| Bell pepper | y = 5980x + 347 | 0.9972 | −7.6 | 0.01 | 0.01/0.2 |
| Lettuce | y = 6356x + 965 | 0.9921 | −1.8 | 0.01 | 0.01/15 |
| Tomato | y = 6118x + 291 | 0.9974 | −5.5 | 0.01 | 0.01/0.2 |
| Matrix | Spike Level (mg kg−1) | Day 1 | Day 2 | Day 3 | RSDr (%) | |||
|---|---|---|---|---|---|---|---|---|
| Rec. (%) | RSDa (%) | Rec. (%) | RSDa (%) | Rec. (%) | RSDa (%) | |||
| Apple | 0.01 | 95.2 | 5.1 | 98.2 | 4.0 | 97.3 | 7.9 | 5.6 |
| 0.1 | 85.8 | 0.9 | 102.6 | 1.7 | 102.1 | 1.3 | 7.5 | |
| 1 | 93.6 | 1.8 | 97.9 | 2.6 | 97.6 | 3.7 | 3.4 | |
| Grape | 0.01 | 91.2 | 2.7 | 98.0 | 4.9 | 98.6 | 4.2 | 5.2 |
| 0.1 | 91.9 | 1.3 | 107.5 | 0.8 | 105.8 | 0.4 | 6.4 | |
| 1 | 90.5 | 2.4 | 98.1 | 2.2 | 98.3 | 1.2 | 4.3 | |
| Peach | 0.01 | 90.9 | 3.0 | 100.4 | 1.7 | 102.1 | 1.6 | 5.6 |
| 0.1 | 86.7 | 1.9 | 110.8 | 4.5 | 102.7 | 2.6 | 9.9 | |
| 1 | 93.1 | 2.2 | 98.4 | 1.5 | 99.1 | 1.1 | 3.2 | |
| Bell pepper | 0.01 | 86.2 | 2.0 | 102.2 | 0.8 | 101.1 | 1.9 | 7.9 |
| 0.1 | 75.7 | 2.8 | 102.9 | 1.4 | 101.0 | 1.4 | 12.1 | |
| 1 | 88.2 | 1.6 | 102.3 | 1.1 | 103.8 | 0.5 | 7.5 | |
| Lettuce | 0.01 | 88.4 | 2.3 | 108.4 | 1.0 | 107.2 | 2.1 | 9.5 |
| 0.1 | 106.6 | 2.0 | 111.5 | 1.4 | 110.0 | 1.5 | 2.3 | |
| 1 | 88.7 | 2.3 | 105.2 | 0.9 | 106.3 | 0.9 | 8.4 | |
| Tomato | 0.01 | 83.8 | 3.3 | 95.9 | 2.1 | 95.9 | 4.0 | 7.1 |
| 0.1 | 86.2 | 1.7 | 100.4 | 1.5 | 100.0 | 1.7 | 6.5 | |
| 1 | 90.4 | 1.8 | 94.5 | 1.5 | 94.9 | 2.0 | 2.8 | |
| Compound | Molecular Formula | Retention Time (min) | Ion Transition (m/z) | DP (V) | CE (V) | CXP (V) | MRM Ratio |
|---|---|---|---|---|---|---|---|
| CYCP | C21H17Br2Cl2N5O2 | 3.94 | 599.9 > 283.8 a,b; 599d.9 > 514.8 a | 90; 90 | 23; 32 | 9; 21 | 0.28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, Z.; Yue, W.; Ren, W.; Wang, Y.; Li, Y. Determination of Cyclaniliprole in Fruits and Vegetables Using Disposable Pipette Extraction Cleanup and Ultrahigh-Performance Liquid Chromatography-Tandem Mass Spectrometry. Molecules 2022, 27, 6464. https://doi.org/10.3390/molecules27196464
Lu Z, Yue W, Ren W, Wang Y, Li Y. Determination of Cyclaniliprole in Fruits and Vegetables Using Disposable Pipette Extraction Cleanup and Ultrahigh-Performance Liquid Chromatography-Tandem Mass Spectrometry. Molecules. 2022; 27(19):6464. https://doi.org/10.3390/molecules27196464
Chicago/Turabian StyleLu, Zhou, Weiqian Yue, Weiming Ren, Yanhong Wang, and Yueru Li. 2022. "Determination of Cyclaniliprole in Fruits and Vegetables Using Disposable Pipette Extraction Cleanup and Ultrahigh-Performance Liquid Chromatography-Tandem Mass Spectrometry" Molecules 27, no. 19: 6464. https://doi.org/10.3390/molecules27196464
APA StyleLu, Z., Yue, W., Ren, W., Wang, Y., & Li, Y. (2022). Determination of Cyclaniliprole in Fruits and Vegetables Using Disposable Pipette Extraction Cleanup and Ultrahigh-Performance Liquid Chromatography-Tandem Mass Spectrometry. Molecules, 27(19), 6464. https://doi.org/10.3390/molecules27196464





