Drug-Inclusive Inorganic–Organic Hybrid Systems for the Controlled Release of the Osteoporosis Drug Zoledronate
Abstract
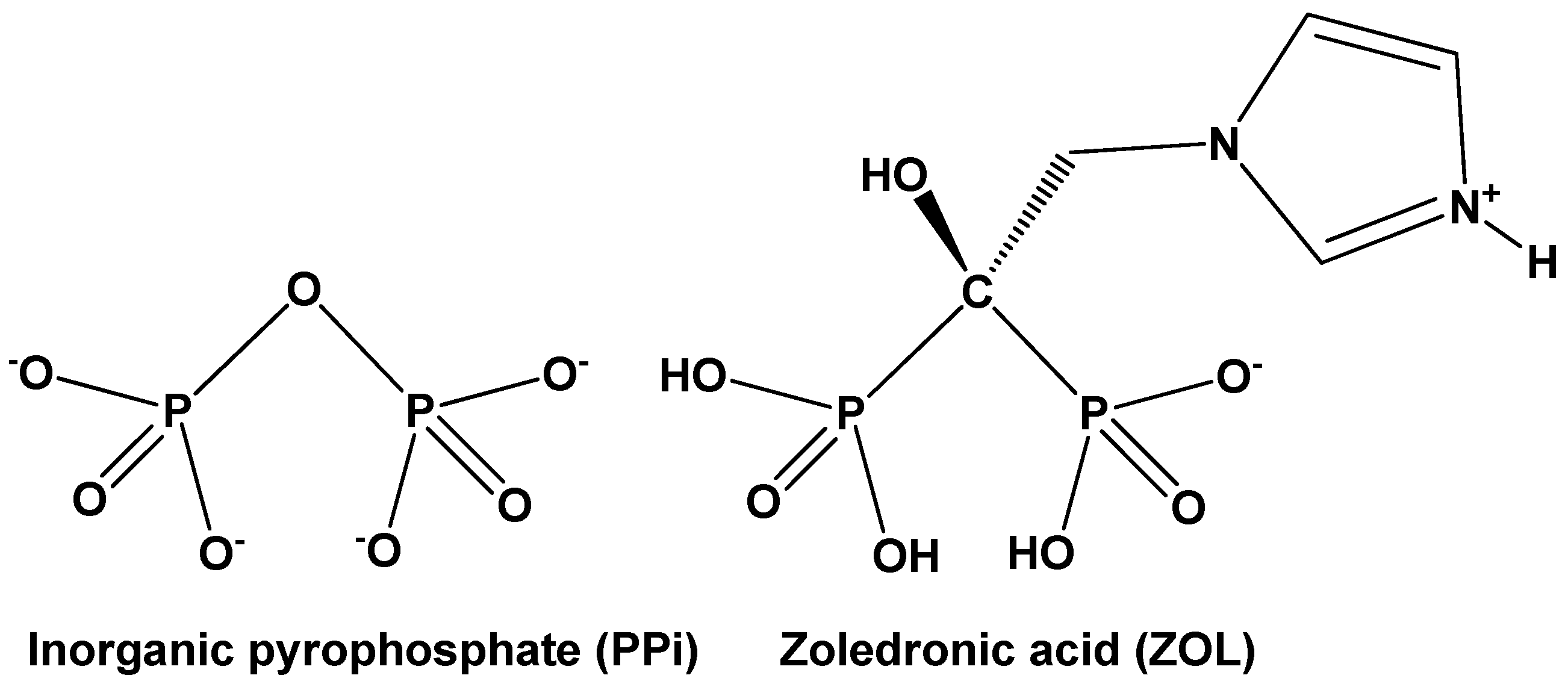
1. Introduction
2. Results
2.1. Synthesis and Characterization of (Sr/Ba)–ZOL Compounds
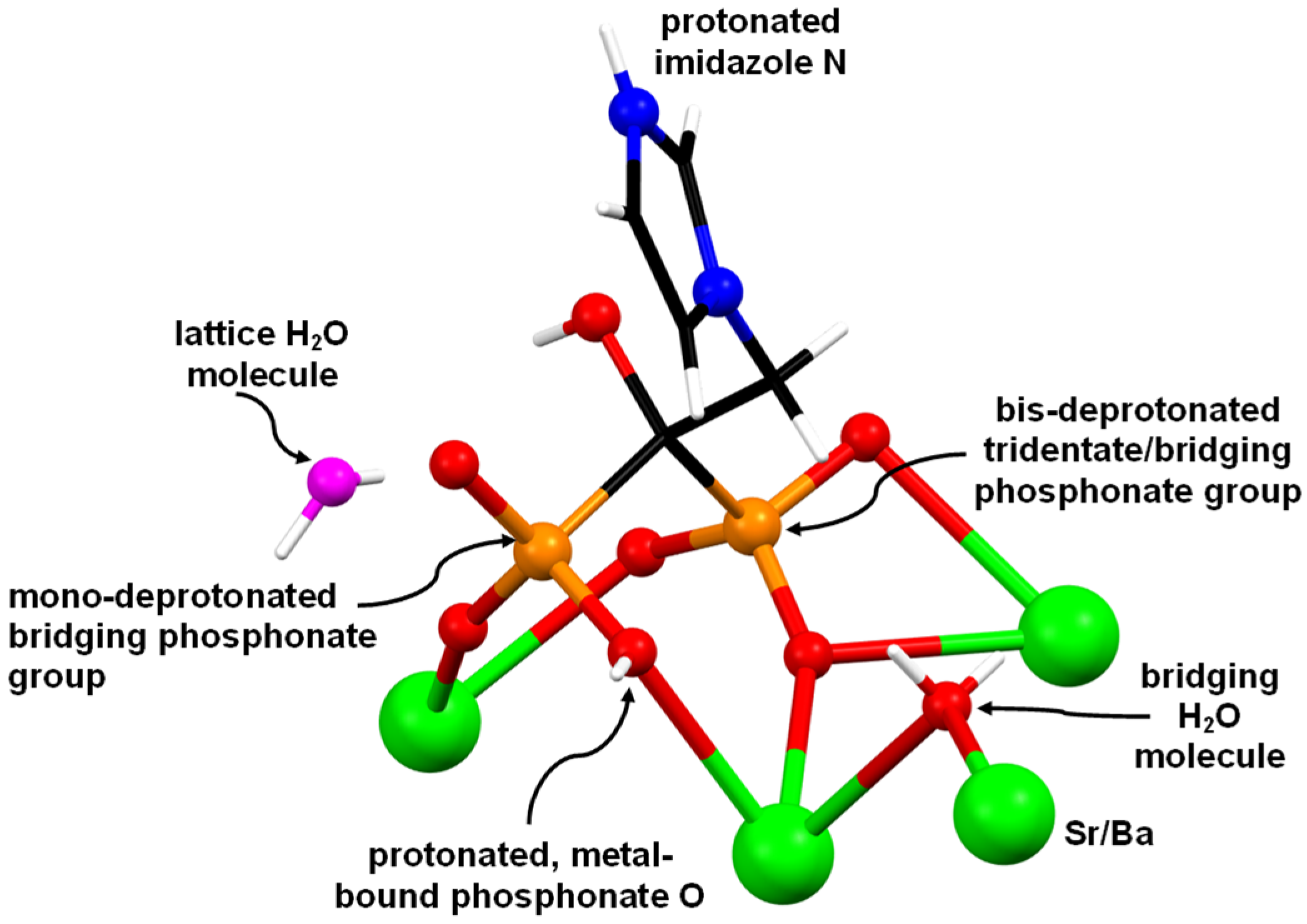
2.2. Powder and Single Crystal X-ray Diffraction Studies of (Sr/Ba)–ZOL Compounds
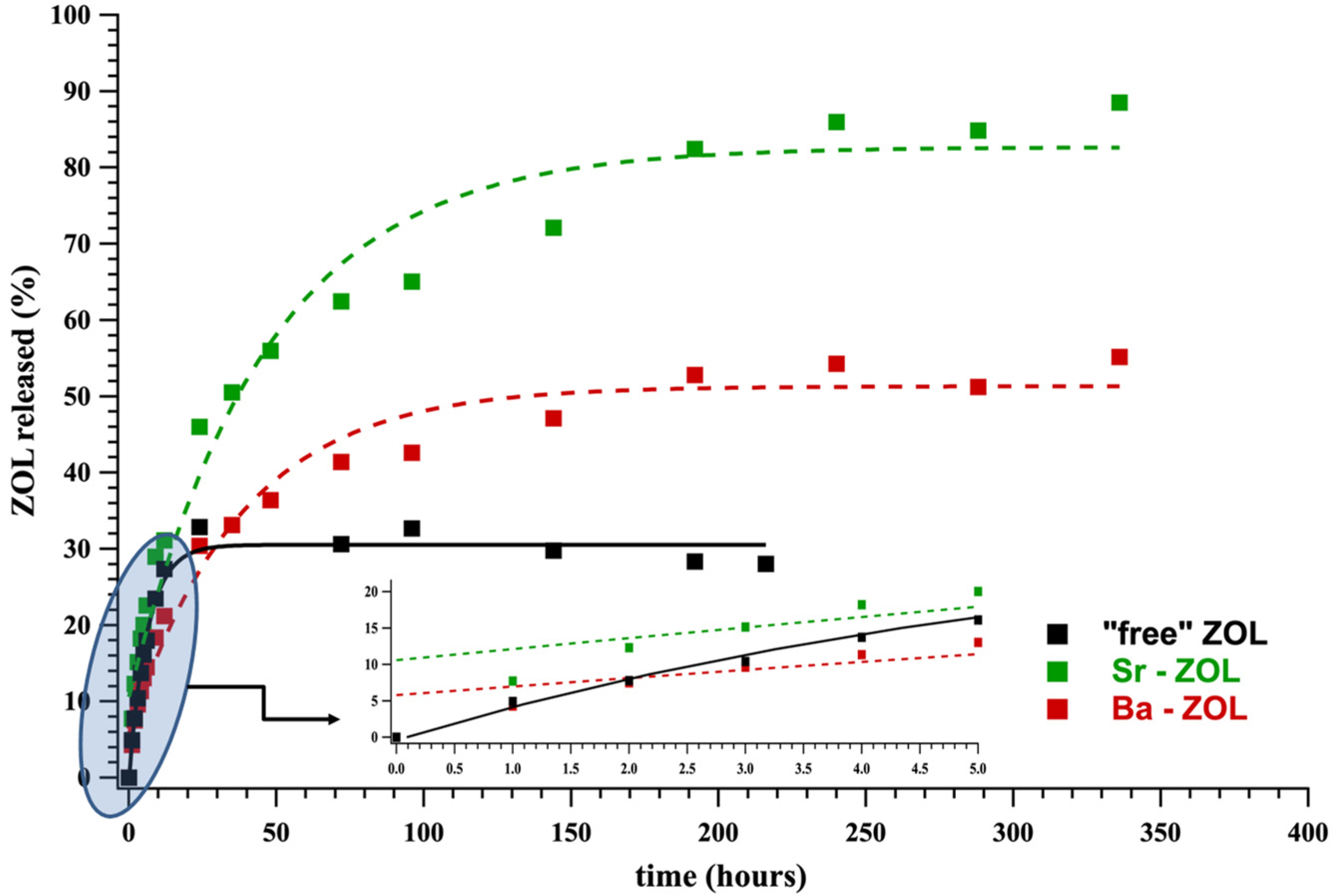
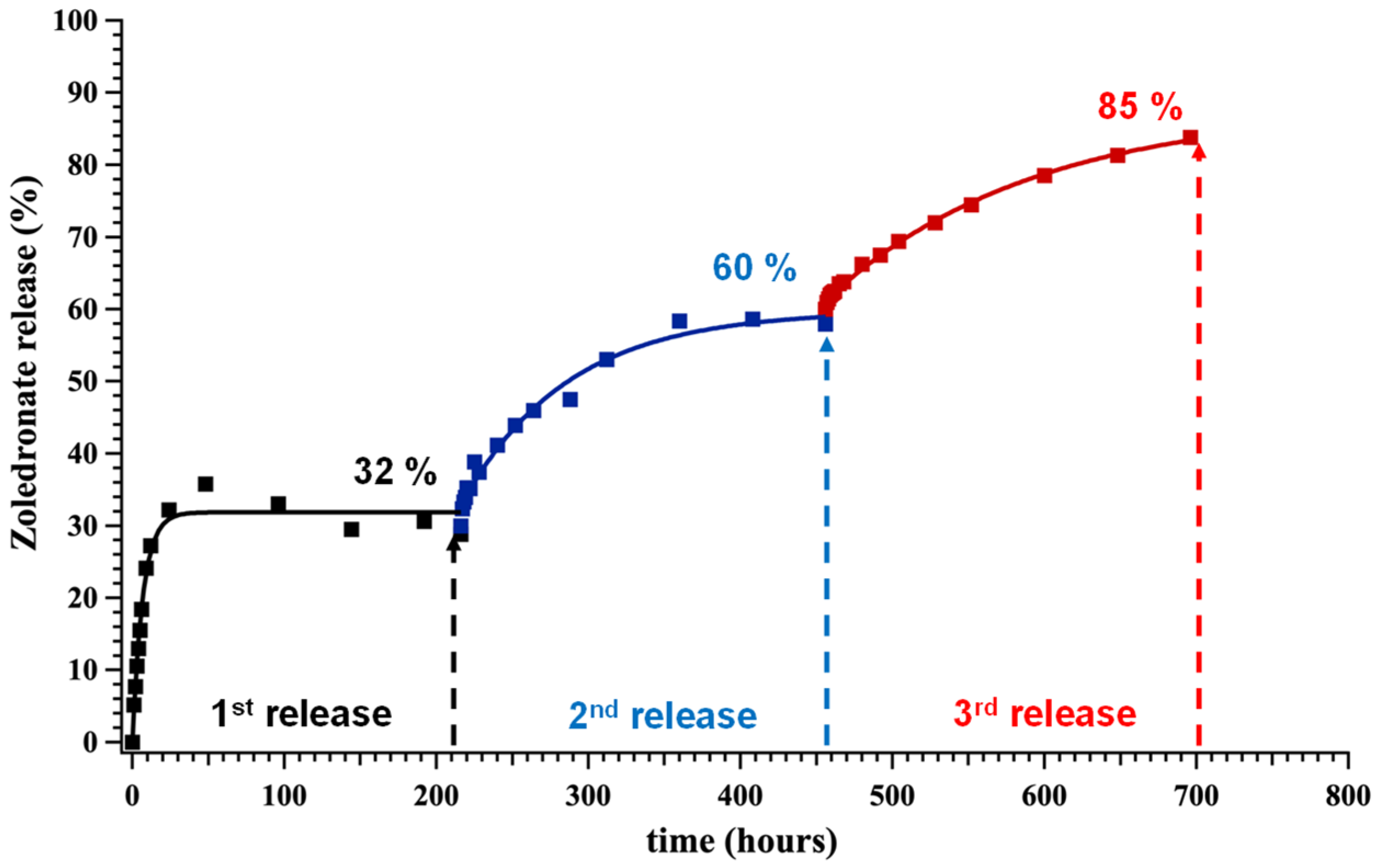
2.3. Controlled Release Study of “Free” ZOL, Sr–ZOL and Ba–ZOL
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Instrumentation
4.3. Synthetic Protocols
4.4. Preparation of Tablets for ZOL Release
4.5. Quantification of ZOL
5. Conclusions
- (1)
- Two novel coordination polymers containing the alkaline-earth metal ions Sr2+ and Ba2+ and the anti-osteoporotic drug ZOL were synthesized and structurally characterized.
- (2)
- Sr–ZOL and Ba–ZOL are isostructural 2D coordination polymers.
- (3)
- Both Sr–ZOL and Ba-ZOL were utilized as controlled release systems (excipient-containing tablets) of the active drug ZOL in conditions that mimic the human stomach (pH = 1.3).
- (4)
- The drug release profiles of Sr–ZOL and Ba–ZOL were compared to that of “free” ZOL (absence of metals). Contrary to the working hypothesis, it was found that the release of ZOL is not delayed compared to the “free” ZOL system. In fact, in the case of Sr–ZOL, it is accelerated.
- (5)
- This behavior was rationalized based on the structural idiosyncrasies of each system. The overall drug release profile for each system was the result of several structural factors, such as presence or absence of π–π interactions between the ZOL imidazole rings, H-bonding interactions and strength of the metal–O(phosphonate) bonds. However, it seems that the governing factor for Sr–ZOL releasing the active drug more rapidly than Ba–ZOL is the particle size and morphology.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Xu, X.-L.; Gou, W.-L.; Wang, A.-Y.; Wang, Y.; Guo, Q.-Y.; Lu, Q.; Lu, S.-B.; Peng, J. Basic research and clinical applications of bisphosphonates in bone disease: What have we learned over the last 40 years? J. Transl. Med. 2013, 11, 303. [Google Scholar] [CrossRef]
- Russell, R.G. Bisphosphonates: From bench to bedside. Ann. N. Y. Acad. Sci. 2006, 1068, 367–401. [Google Scholar] [CrossRef] [PubMed]
- Nancollas, G.H.; Tang, R.; Phipps, R.J.; Henneman, Z.; Gulde, S.; Wu, W.; Mangood, A.; Russell, R.G.G.; Ebetino, F.H. Novel insights into actions of bisphosphonates on bone: Differences in interactions with hydroxyapatite. Bone 2006, 38, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Watts, N.B.; Diab, D.L. Long-Term Use of Bisphosphonates in Osteoporosis. J. Clin. Endocrinol. Metab. 2021, 95, 1555–1565. [Google Scholar] [CrossRef] [PubMed]
- Spinthaki, A.; Matheis, J.; Hater, W.; Demadis, K.D. Antiscalant-driven inhibition and stabilization of “magnesium silicate” under geothermal stresses: The role of magnesium-phosphonate coordination chemistry. Energy Fuels 2018, 32, 11749–11760. [Google Scholar] [CrossRef]
- Barbosa, J.S.; Pinto, M.; Barreiro, S.; Fernandes, C.; Mendes, R.F.; Lavrador, P.; Gaspar, V.M.; Mano, J.F.; Borges, F.; Remião, F.; et al. Coordination Compounds As Multi-Delivery Systems for Osteoporosis. ACS Appl. Mater. Interfaces 2021, 13, 35469–35483. [Google Scholar] [CrossRef] [PubMed]
- Vassaki, M.; Papathanasiou, K.E.; Hadjicharalambous, C.; Chandrinou, D.; Turhanen, P.; Choquesillo-Lazarte, D.; Demadis, K.D. Self-Sacrificial MOFs for Ultra-Long Controlled Release of Bisphosphonate Anti-Osteoporotic Drugs. Chem. Commun. 2020, 56, 5166–5169. [Google Scholar] [CrossRef] [PubMed]
- Vassaki, M.; Kotoula, C.; Turhanen, P.; Choquesillo-Lazarte, D.; Demadis, K.D. Calcium and Strontium Coordination Polymers as Controlled Delivery Systems of the Anti-Osteoporosis Drug Risedronate and the Augmenting Effect of Solubilizers. Appl. Sci. 2021, 11, 11383. [Google Scholar] [CrossRef]
- Abood, N.A.; AL-Askari, M.; Saeed, B.A. Structures and Vibrational Frequencies of Imidazole, Benzimidazole and its 2-Alkyl Derivatives Determined by DFT Calculations. Basrah J. Sci. 2012, 30, 119–131. [Google Scholar]
- Zenobi, M.C.; Luengo, C.V.; Avena, M.J.; Rueda, E.H. An ATR-FTIR Study of Different Phosphonic Acids in Aqueous Solution. Spectrochim. Acta Part A 2008, 70, 270–276. [Google Scholar] [CrossRef]
- Povoroznyuk, V.V.; Grygorieva, V.N.; Pekhnyo, V.I.; Kozachkova, O.M.; Tsaryk, N.V. Zoledronic Acid and Its Calcium-contain Complexes in Treatment of Experimental Osteoporosis in Wistar Female Rats. Biochem. Anal. Biochem. 2017, 6, 340. [Google Scholar]
- Chernyshev, V.V.; Shkavrov, S.V.; Paseshnichenko, K.A.; Puryaeva, T.P.; Velikodny, Y.A. Zoledronic acid: Monoclinic and triclinic polymorphs from powder diffraction data. Acta Cryst. 2013, C69, 263–266. [Google Scholar] [CrossRef]
- Papathanasiou, K.E.; Turhanen, P.; Brückner, S.I.; Brunner, E.; Demadis, K.D. Smart, programmable and responsive injectable hydrogels for controlled release of cargo osteoporosis drugs. Sci. Rep. 2017, 7, 4743. [Google Scholar] [CrossRef]
- Fronczek CCDC 1562048: Experimental Crystal Structure Determination. Available online: https://doi.org/10.5517/ccdc.csd.cc1pffn6. (accessed on 19 September 2022).
- Fronczek, F.R. CCDC 1562049: Experimental Crystal Structure Determination. Available online: https://doi.org/10.5517/ccdc.csd.cc1pffp7 (accessed on 19 September 2022).
- Ruscika, R.; Bianchi, M.; Quintero, M.; Martinez, A.; Vega, D.R. Solid-State Forms of Zoledronic Acid: Polymorphism in Hydrates. J. Pharm. Sci. 2010, 99, 4962–4972. [Google Scholar] [CrossRef]
- Sridhar, B.; Ravikumar, K. Multiple hydrogen bonds in cytosinium zoledronate trihydrate. Acta Cryst. 2011, C67, o115–o119. [Google Scholar] [CrossRef]
- Sarkar, A.; Cukrowski, I. Tris(dicyclohexylammonium) hydrogen [1-hydroxy-2-(1H-imidazol-1-yl)-1-phosphonatoethane]phosphonate ethanol monosolvate monohydrate. Acta Cryst. 2011, E67, o2980. [Google Scholar] [CrossRef]
- Sikorska, M.; Chojnacki, J. Bis(ammonium) Zoledronate Dihydrate. J. Crystallogr. 2013, 2013, 741483. [Google Scholar] [CrossRef]
- Freire, E.; Vega, D.R.; Baggio, R. Zoledronate complexes. I. Poly[[μ2-aqua[μ3-1-hydroxy-2-(1H,3H-imidazol-3-ium-1-yl)ethylidenediphosphonato]potassium(I)] monohydrate]. Acta Cryst. 2010, C66, m13–m16. [Google Scholar] [CrossRef]
- Freire, E.; Vega, D.R. Aquabis[1-hydroxy-2-(imidazol-3-ium-1-yl)-1,10-ethylidenediphosphonato-k2O,O’]-zinc(II) dehydrate. Acta Cryst. 2009, E65, m1430–m1431. [Google Scholar]
- Freire, E.; Quintero, M.; Vega, D.R.; Baggio, R. Crystal structure and magnetic properties of two new zoledronate complexes: A Mn dimer [Mn(II)(H3Zol)2·(H2O)2] and a Fe15 molecular cluster [Fe(III)15(HZol)10(H2Zol)2 (H2O)12(Cl4:(H2O)2)·Cl7·(H2O)65] (where H4Zol: C5H10N2O7P2 is zoledronic acid). Inorg. Chim. Acta 2013, 394, 229–236. [Google Scholar] [CrossRef]
- Cao, D.-K.; Li, Y.-Z.; Zheng, L.-M. Layered Cobalt(II) and Nickel(II) Diphosphonates Showing Canted Antiferromagnetism and Slow Relaxation Behavior. Inorg. Chem. 2007, 46, 7571–7578. [Google Scholar] [CrossRef]
- Qiu, L.; Lv, G.; Guo, L.; Chen, L.; Luo, S.; Zou, M.; Lin, J. Synthesis, crystal structure and antitumor effect of a novel copper(II) complex bearing zoledronic acid derivative. Eur. J. Med. Chem. 2015, 89, 42–50. [Google Scholar] [CrossRef]
- Bruker APEX3. APEX3 V2019.1; Bruker-AXS: Madison, WI, USA, 2019. [Google Scholar]
- Sheldrick, G.M. SHELXT—Integrated Space-Group and Crystal-Structure Determination. Acta Crystallogr. Sect. A Found. Crystallogr. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Macrae, C.F.; Bruno, I.J.; Chisholm, J.A.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Rodriguez-Monge, L.; Taylor, R.; van de Streek, J.; Wood, P.A. Mercury CSD 2.0—New Features for the Visualization and Investigation of Crystal Structures. J. Appl. Crystallogr. 2008, 41, 466–470. [Google Scholar] [CrossRef]
- Martin, M.B.; Grimley, J.S.; Lewis, J.C.; Heath, H.T.; Bailey, B.N.; Kendrick, H.; Yardley, V.; Caldera, A.; Lira, R.; Urbina, S.J.A.; et al. Bisphosphonates Inhibit the Growth of Trypanosoma brucei, Trypanosoma cruzi, Leishmania donovani, Toxoplasma gondii, and Plasmodium falciparum: A Potential Route to Chemotherapy. J. Med. Chem. 2001, 44, 909–916. [Google Scholar] [CrossRef]
- Papathanasiou, K.E.; Vassaki, M.; Spinthaki, A.; Alatzoglou, F.-E.G.; Tripodianos, E.; Turhanen, P.; Demadis, K.D. Phosphorus chemistry: From small molecules to polymers to pharmaceutical and industrial applications. Pure Appl. Chem. 2019, 91, 421–441. [Google Scholar] [CrossRef]
- Papathanasiou, K.E.; Demadis, K.D. Polymeric Matrices for the Controlled Release of Phosphonate Active Agents for Medicinal Applications. In Handbook of Polymers for Pharmaceutical Technologies, Bioactive and Compatible Synthetic/Hybrid Polymers; Thakur, V.K., Thakur, M.K., Eds.; Wiley-Scrivener Publishing LLC: Salem, MA, USA, 2015; Chapter 4; Volume 4, pp. 87–122. [Google Scholar]
- Papathanasiou, K.E.; Moschona, A.; Spinthaki, A.; Vassaki, M.; Demadis, K.D. Silica-Based Polymeric Gels as Platforms for Delivery of Phosphonate Pharmaceutics. In Polymer Gels: Synthesis and Characterization; Thakur, V.K., Thakur, M.K., Eds.; Springer: Berlin/Heidelberg, Germany, 2018; Chapter 5; pp. 127–140. [Google Scholar]
- Shearan, S.; Stock, N.; Emmerling, F.; Demel, J.; Wright, P.A.; Demadis, K.D.; Vassaki, M.; Costantino, F.; Vivani, R.; Sallard, S.; et al. New Directions in Metal Phosphonate and Phosphinate Chemistry. Crystals 2019, 9, 270. [Google Scholar] [CrossRef]


| Sr–ZOL | Ba–ZOL | |
|---|---|---|
| Empirical Formula | C5H12SrN2O9P2 | C5H12BaN2O9P2 |
| Mr | 393.73 | 443.43 |
| Crystal System | triclinic | triclinic |
| Space Group | ||
| a (Å) | 6.3648(2) | 6.479(2) |
| b (Å) | 6.6900(2) | 6.837(2) |
| c(Å) | 13.9296(5) | 14.173(5) |
| α (°) | 102.5690(10) | 77.532(12) |
| β (°) | 91.4870(10) | 88.748(11) |
| γ (°) | 90.1080(10) | 89.021(10) |
| V (Å3) | 578.703 | 612.819 |
| Z | 2 | 2 |
| R factor (%) | 2.98 | 3.80 |
| CCDC code | 2195677 | 2195679 |
| Compound. | PA | PB | N | OH | Total H-Bonds 1 | π-π Interactions | M-O Bonds (PO3/H2O/OH) 2 | Total Interactions | Lattice H2O | M2+ Cations |
|---|---|---|---|---|---|---|---|---|---|---|
| ZOLMolecule 1 | 5 | 4 | 1 | 1 | 11 | 1 | 0 | 12 | 1 | 0 |
| ZOLMolecule 2 | 3 | 6 | 1 | 1 | ||||||
| Sr-ZOL | 5 | 4 | 1 | 1 | 11 | 2 | 6/2/0 | 21 | 1 | 1 |
| Ba-ZOL | 4 | 3 | 1 | 1 | 9 | 2 | 6/2/0 | 19 | 1 | 1 |
| Initial Rate (μmol/min) 1 | Plateau BP (%) | tp (h) 2 | t½ (h) 3 | |
|---|---|---|---|---|
| “free” ZOL | 0.39 | 30 | 25.5 | 4.3 |
| Sr–ZOL | 0.50 | 80 | 153.2 | 24.4 |
| Ba–ZOL | 0.33 | 50 | 134.1 | 20.8 |
| Tablet | ZOL acid·H2O | Sr–ZOL * | Ba–ZOL * |
|---|---|---|---|
| MW (g/mol) | 290.11 | 393.73 | 443.43 |
| Drug system (g) | 0.247 | 0.335 | 0.377 |
| Lactose (g) | 0.251 | 0.222 | 0.208 |
| Cellulose (g) | 0.251 | 0.222 | 0.208 |
| Silica (g) | 0.251 | 0.222 | 0.208 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vassaki, M.; Lazarou, S.; Turhanen, P.; Choquesillo-Lazarte, D.; Demadis, K.D. Drug-Inclusive Inorganic–Organic Hybrid Systems for the Controlled Release of the Osteoporosis Drug Zoledronate. Molecules 2022, 27, 6212. https://doi.org/10.3390/molecules27196212
Vassaki M, Lazarou S, Turhanen P, Choquesillo-Lazarte D, Demadis KD. Drug-Inclusive Inorganic–Organic Hybrid Systems for the Controlled Release of the Osteoporosis Drug Zoledronate. Molecules. 2022; 27(19):6212. https://doi.org/10.3390/molecules27196212
Chicago/Turabian StyleVassaki, Maria, Savvina Lazarou, Petri Turhanen, Duane Choquesillo-Lazarte, and Konstantinos D. Demadis. 2022. "Drug-Inclusive Inorganic–Organic Hybrid Systems for the Controlled Release of the Osteoporosis Drug Zoledronate" Molecules 27, no. 19: 6212. https://doi.org/10.3390/molecules27196212
APA StyleVassaki, M., Lazarou, S., Turhanen, P., Choquesillo-Lazarte, D., & Demadis, K. D. (2022). Drug-Inclusive Inorganic–Organic Hybrid Systems for the Controlled Release of the Osteoporosis Drug Zoledronate. Molecules, 27(19), 6212. https://doi.org/10.3390/molecules27196212








