1. Introduction
Microsorum scolopendria (
MS) is a fern that grows on the Chilean island of Rapa Nui and is known to the indigenous people as “Matu’a Pua’a.”
MS is native to Polynesian islands, including Fiji, Tahiti, Hawaii, Rapa Nui, and Madagascar [
1], where it is used to treat asthma, inflammatory diseases, and cancer [
2].
MS is known under different names, among them
Phymatosorus scolopendria,
Polypodium scolopendria, and
Microsorum parksii [
3,
4],
Polypodium scolopendria, and
Microsorum parksii [
3,
4]. As a member of the
Polypodiaceae family,
MS has a creeping rhizome 5–7 mm in diameter and leathery-textured leaves with visible veins [
5]. Many of the properties attributed to ferns (and plants in general) have been associated with high contents of polyphenols in plant tissues [
6,
7,
8]. More than eight thousand phenolic compounds have been reported in various plant species [
9,
10]. Polyphenols are widely used in the pharmaceutical, cosmetic, and food industries [
11], mainly due to their properties as antioxidants, antimicrobial agents, and inhibitors of pro-inflammatory mediators [
12]. There is some diversity in the structure of polyphenols, which has led to their subdivision into families centered on phenolic acids, stilbenes, and flavonoids.
MS has not been extensively investigated through phytochemical analyses or characterization of its medicinal properties, despite being widely used by the indigenous people of Rapa Nui. A MEDLINE search using the terms “
Polypodium scolopendria or
Microsorum scolopendria” yielded only three articles in 21 years (between 2000 and 2021). Thus,
MS has been very under-researched so far, only some studies have explored its ecdysteroid content [
3]. Due to its importance in traditional Rapa Nui medicine, where it is used to treat a wide variety of ailments, the number of specimens is declining. In fact, the conservation status of
MS is now classified as “vulnerable” in Chile. It is important to investigate whether it is safe to consume fern material as part of folk medicine, given that certain compounds can be toxic if ingested in large quantities. Thus, the purpose of this study was to perform a functional characterization of Rapa Nui
MS rhizome and leaf extracts with respect to their polyphenolic and flavonoid identity, radical scavenging, antimicrobial, and anti-inflammatory properties. To this end, the reversed-phase high-performance liquid chromatography (RP-HPLC) method coupled with mass spectrometry was used [
13]. The radical scavenging capacity of the
MS extracts was determined by the DPPH (1,1-diphenyl-2-picrylhydrazyl) radical scavenging method and the oxygen radical absorbance capacity (ORAC) method. The antimicrobial activity against
Staphylococcus aureus and
Staphylococcus epidermidis was evaluated in an infection model using the Human Dermal Fibroblast adult (HDFa) cell line. The two bacteria are frequently associated with infections in implants, peripheral venous catheters, and skin [
14,
15].
Bacterial infections, pathologies caused by UV exposure, and cancer share a common inflammatory process, triggered by the activation and downregulation of several signaling pathways, in which reactive oxygen species (ROS) induce certain cellular disorders. Signal transduction in conditions involving ROS-mediated metabolic activity, inflammatory activity, and metabolic dysfunction is, therefore, an important therapeutic focus for the control of various diseases [
16]. In addition, we demonstrated that these extracts have anti-inflammatory, radical scavenging capacity, and sun protection properties.
3. Discussion
There is a wealth of information on the medicinal properties of various plants, including radical scavenging capacity, anti-inflammatory, and antimicrobial effects, which are useful in the treatment of many diseases. These properties are associated with phytochemicals such as polyphenols, flavonoids, and triterpenes. However, there are very few reports on the properties of
MS. In this paper, we studied two
MS extracts (rhizome and leaf) and found that the RAE and HAE extracts had different contents of secondary metabolites. For example, the RAE extract had a very high content of polyphenols and flavonoids per gram of dry sample. Plants are known to contain high concentrations of polyphenols as a defense mechanism against pathogens [
26].
It has been reported in the literature that ethyl acetate extracts are rich in phenolic components [
27,
28,
29,
30,
31]. We found that more than 50% of the phenolic content of the RAE and HAE extracts consisted of flavonoids. Similar results have been described by other authors [
30,
32].
The amounts of phenols and flavonoids have been previously reported in
MS from Tamil Nadu in India [
33], but there are no studies on
MS from Rapa Nui. The main methods used for the quantification and characterization of extracts from plants are the spectrophotometric and chromatographic methods. In this study, the identification of phenols, flavonoids, and other compounds was performed by the RP-HPLC-
MS/
MS method, while concentrations were analyzed by the spectrophotometric method.
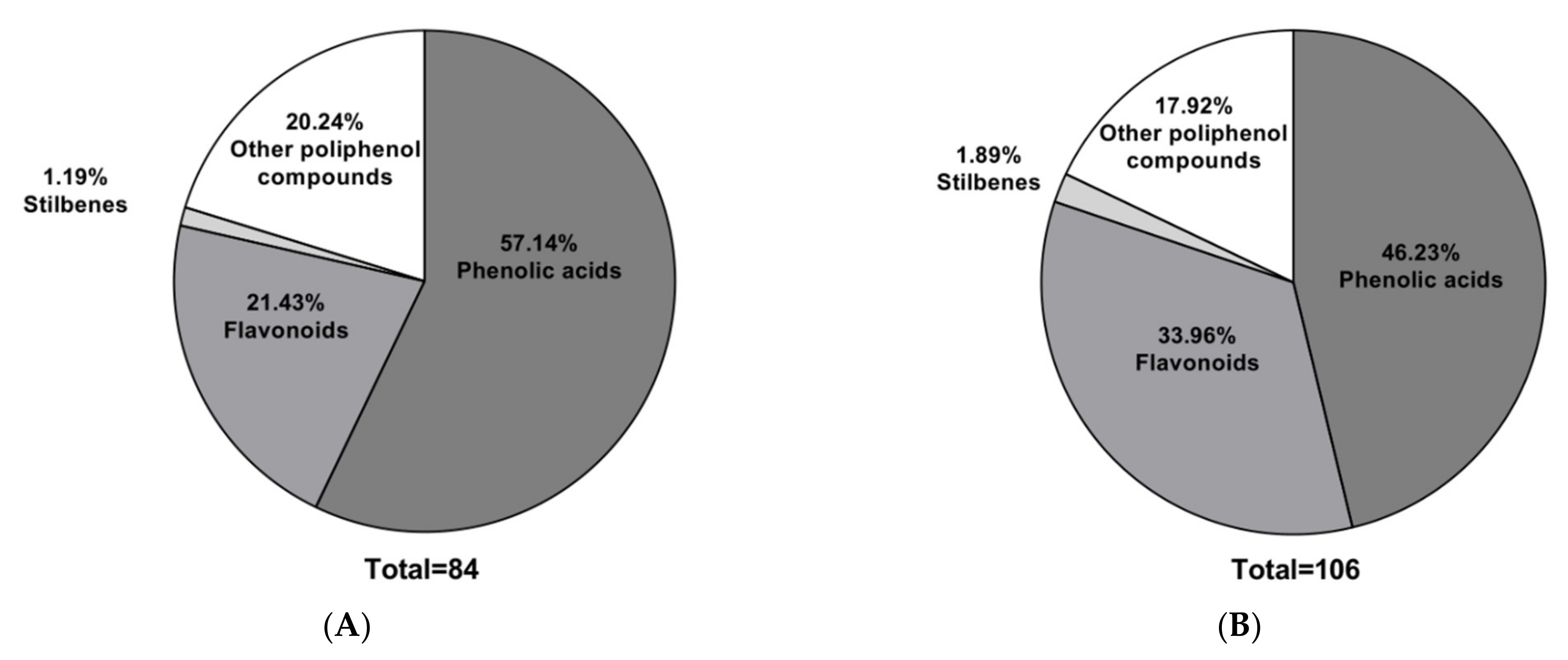
Figure 1 shows that phenolic acids were the predominant class, accounting for over 45% in both extracts. These compounds correspond mainly to the derivatives of hydroxybenzoic and hydroxycinnamic acids, as has been observed in several ferns (
Tables S1 and S2) [
34,
35]. The second class is flavonoids and other types of polyphenols, with an emphasis on coumarin derivatives, as previously reported for
MS extracts from Mahanoro, Madagascar [
1]. Similar results have been reported for the edible fern
Diplazium esculentum from India [
36], whose leaves contain about 40% flavonoids. In addition, a variety of hydroxycinnamic acids were found mostly in the leaf samples of the fern
Stenochlaena palustris from Malaysia [
37].
The flavonoids found in this study correspond to both flavonoids and their glycosides, presumably luteolin, kaempferol, isorhamnetin, and their derivatives. The average molecular mass of flavonoids is 345 g/mol, shifting toward 280 g/mol (
Tables S3 and S4). Flavonoids with a basic structure have molecular weight of 222.24 g/mol, similar to phenolic acids, and the majority of the flavonoids found were glycosylated. Members of the stilbene group (
Tables S5 and S6), resveratrol (negative mode in the HAE extract and positive mode in the RAE extract), and pterostilbene (positive mode in HAE) were also detected. Stilbenes are produced in a variety of plant species, including
Vitis vinifera, red berries, and peanuts [
38,
39]. Studies have demonstrated the important role of resveratrol in human health.
We found that 44% of the phenolic acids detected belonged to the hydroxycinnamic acid subclass and 44% to the hydroxybenzoic acid subclass. The average weight of the phenolic acid found was 277 g/mol, shifting toward 200 g/mol. The molecular weights of phenolic acids ranged from 120 to 220 g/mol, and the phenolic acids found in this study were predominantly glycosylated. Glycosylated molecules tend to be more bioavailable upon ingestion due to their altered bioavailability properties, membrane disintegration, bioactivity, and metabolic stability [
40].
The other types of polyphenols were mostly coumarin derivatives, e.g., sculetin and isopimpinellin. This class of polyphenols has a broader range of molecular masses, ranging from low molecular weight molecules such as pyrogallol (126.11 g/mol) to more complex molecules such as 5-heneicosenylresorcinol (402.7 g/mol).
The extract with the best radical scavenging capacity in the DPPH inhibition assay was RAE, with an inhibition rate of 82.96%. The observed values are consistent with the results described by Ding [
41], who analyzed 31 fern extracts from Asia and found that the IC
50 ranged between 5.9 µg/mL and 133.2 µg/mL, with an average of 39.47 ± 35.92 µg/mL. Our results showed that the IC
50 values of the RAE and HAE extracts in the DPPH inhibition assay were 12.05 µg/mL and 20.34 µg/mL, respectively, which is close to the values reported by Ding [
41]. In the ORAC test, the highest radical scavenging capacity was demonstrated by the RAE extract, with an ORAC value of 1.63 (
Table 4).
There is evidence to suggest that
MS extracts have a cytoprotective effect against UV-B ray damage to human epithelial cells [
42]. Thus,
MS may have both an epithelial protective effect at the cellular level and an effect as a sunscreen filter at the dermal level. Indeed, HAE extracts were observed to have an SPF of 20 (medium-high level) and RAE an SPF of 15 (medium level). Therefore, it is quite exciting to consider the possibility that some
MS compounds may find their way into cosmetic products.
Based on the “Performance standards for antimicrobial susceptibility tests” [
43] of the “Clinical and Laboratory Standards Institute” (CLSI),
S. aureus can be resistant to
MS extracts and susceptible to kanamycin and chloramphenicol;
S. epidermidis is resistant to
MS and kanamycin extracts and moderately susceptible to chloramphenicol (
Table 6). Even though the extracts did not show strong antimicrobial activity, they were effective in inhibiting and disrupting biofilms formed by the bacteria
S. aureus and
S. epidermidis (
Figure 2). Bacteria of the genus
Staphylococcus are recognized as the most frequent causes of infections involving biofilm formation. Skin infections in humans are frequently colonized by commensal bacteria such as
S. aureus. These bacteria cause epithelial dysbiosis and increase biofilm formation [
44].
Assays with 512 µg/mL pyrogallol show an MIC value against
S. aureus [
43] without compromising the integrity of the MO membrane [
45,
46], an effect also observed with resveratrol [
47]. Chin [
48] reported that daidzein and daidzin have lower MIC values than pyrogallol against
S. aureus, with daidzein being more effective than daidzin. Daidzeinhad an MIC concentration of 64 µg/mL and the daidzin of 128 µg/mL [
48]. These results suggest that the total polyphenol concentration in the samples is not relevant for the antimicrobial activity, indicating that the activity may have decreased due to the presence of other molecules in the extracts. The results also show that
S. epidermidis is more sensitive to
MS extracts than
S. aureus, due to the presence of other components in the extracts, such as cirsimaritin [
49] and resveratrol [
50]. Our own results were similar, as both extracts had an effect on
S. epidermidis but not on
S. aureus.
The assays performed to evaluate biofilm inhibition and disruption depended on the MIC concentration of each extract tested on the bacteria. In this study, MIC values and three serial dilutions were used for the assay. If the extract did not exhibit an MIC value, a concentration of 512 µg/mL of the extract was used.
Figure 2A,B show graphs of bacterial biofilm inhibition and disruption by the RAE extract against
S. aureus and
S. epidermidis. In this case, the RAE extract showed significant inhibition of biofilm formation (close to 50% in the assay with 512 µg/mL) against
S. aureus. These results may be attributed to the relative abundance of polyphenols such as isoxanthohumol, resveratrol, and kaempferide [
51,
52]. These types of compounds have been shown to have the ability to inhibit biofilm formation by downregulating the expression of genes such as rsbU and spa, which are genes responsible for bacterial adhesion, communication, and bacterial protection [
53,
54,
55]. The extract that showed more significant inhibition of biofilm formation was RAE at 128 µg/mL against
S. epidermidis.
Both extracts analyzed showed a dose-dependent effect in biofilm disruption. The results suggest that in assays against S. aureus, these extracts may inhibit bacterial biofilm formation by: (i) increasing susceptibility to antibiotics; (ii) creating instability in the bacterium. Therefore, in the future, it might be interesting to evaluate the effect of extracts on different concentrations of antibiotics in biofilm studies and to assess the possibility of reducing the antibiotic concentrations currently used to treat infections caused by these bacteria, as well as to consider the possibility of synergistic effects of the two compounds.
The inhibition assay showed a greater reduction in the percentage of biofilm. Biofilm formation consists of four steps: i.—bacteria attach to a surface; ii.—microcolonies are formed; iii.—biofilm maturation; and finally, iv.—bacteria spread to colonize other surfaces [
53]. In the biofilm inhibition test, the bacteria are in steps i and ii, while in the biofilm disintegration test, the bacteria are in step iii. In this step, the barriers that constitute a defense mechanism are already present, requiring a more complex process of elimination. Assays with
S. epidermidis (
Figure 2B,D) confirmed that the rhizome extract inhibited biofilm formation more efficiently than the leaf extract. The HAE extract was effective in disrupting biofilms rather than inhibiting their formation. This extract is known to contain resveratrol, and it has been reported that this compound inhibits bacterial quorum sensing and disrupts bacterial biofilms of the genus
Staphylococcus [
54].
The different responses of the two bacteria to the evaluated extracts can be mainly attributed to multidrug resistance and the presence of virulence genes in both pathogens.
S. epidermidis exhibits multidrug resistance [
55,
56,
57], whereas
S. aureus has major resistance mechanisms, making it a highly virulent bacterium tolerant to various antibiotics due to increased activation of these genes [
58].
The
MS extracts had no effect on
S. aureus viability, but they had an effect on the formation and disruption of bacterial biofilm. It has been reported that phenolic compounds do not play a predominant role in the viability of many bacteria, but they may play a role in reducing virulence, deactivating quorum sensing, and reducing the production of α- family proteins. It has been observed that the main inhibitors that can inactivate the metabolism of bacteria [
59], their adhesion to surfaces [
52,
60] and inhibit biofilm formation, are phenolic compounds [
61,
62].
The results of the inhibition of COX enzymes were complemented by the DPPH radical scavenging assays and ORAC assays, because both extracts not only have the ability to act on radical molecules, but also to inhibit their production. Although not all extracts exhibited a low IC
50 for COX-2 enzyme (the results were compared with the Celecoxib drug used as positive control), they also did not show a selectivity index of less than 1. The HAE extract exhibited an IC
50 of 3.52 µg/mL for COX-2 enzyme and a selectivity index of 6.84. This extract was the second-best in terms of radical scavenging ability. The RAE extract showed a IC
50 of 3.14 for COX-2 and a selectivity index of 9.96. In addition, it had the highest radical scavenging capacity in the DPPH and ORAC assays (
Table 4). These results are consistent with the low percentages of ROS production observed when the HDFa cell line was exposed to both bacteria. Therefore, both extracts had a protective effect on the cell line (
Figure 4).
Molecules such as resveratrol, present in
MS extracts, inhibit not only COX-2 expression, but also its activity [
63]. Diadzein, which selectively inhibits COX-1 and COX-2 enzyme expression [
64], is a compound mainly present in HAE extracts and provides excellent selectivity (
Table 7). Flavonoids such as kaempferol, luteolin, and apigenin have the ability to selectively inhibit COX-1 and COX-2 [
36,
65]. Extracts with a higher concentration of flavonoids were found to exhibit greater selectivity against COX enzymes (
Table 7).
None of the extracts evaluated had any detrimental effect on cell viability, as shown in
Figure 3. Studies performed with
MS ethanolic extracts from Tahiti showed that they do not induce cytotoxicity on epithelial cell lines in the range of 1 to 250 µg/mL [
42]. Phenolic compounds of the flavonoid family, such as cirsimaritin [
66] and kaempferol [
67] have been reported to increase cell viability. These compounds are present in the
MS extracts studied.
The results of the infection assays indicate the possibility that the molecules present in the extracts affect the proliferation of
S. epidermidis and create a molecular cascade that helps to fight the infection, either by reducing biofilm formation, biofilm adherence to the cell, and the production of virulent proteins from this pathogen [
68], or by increasing the viability of HDFa cells, as shown in
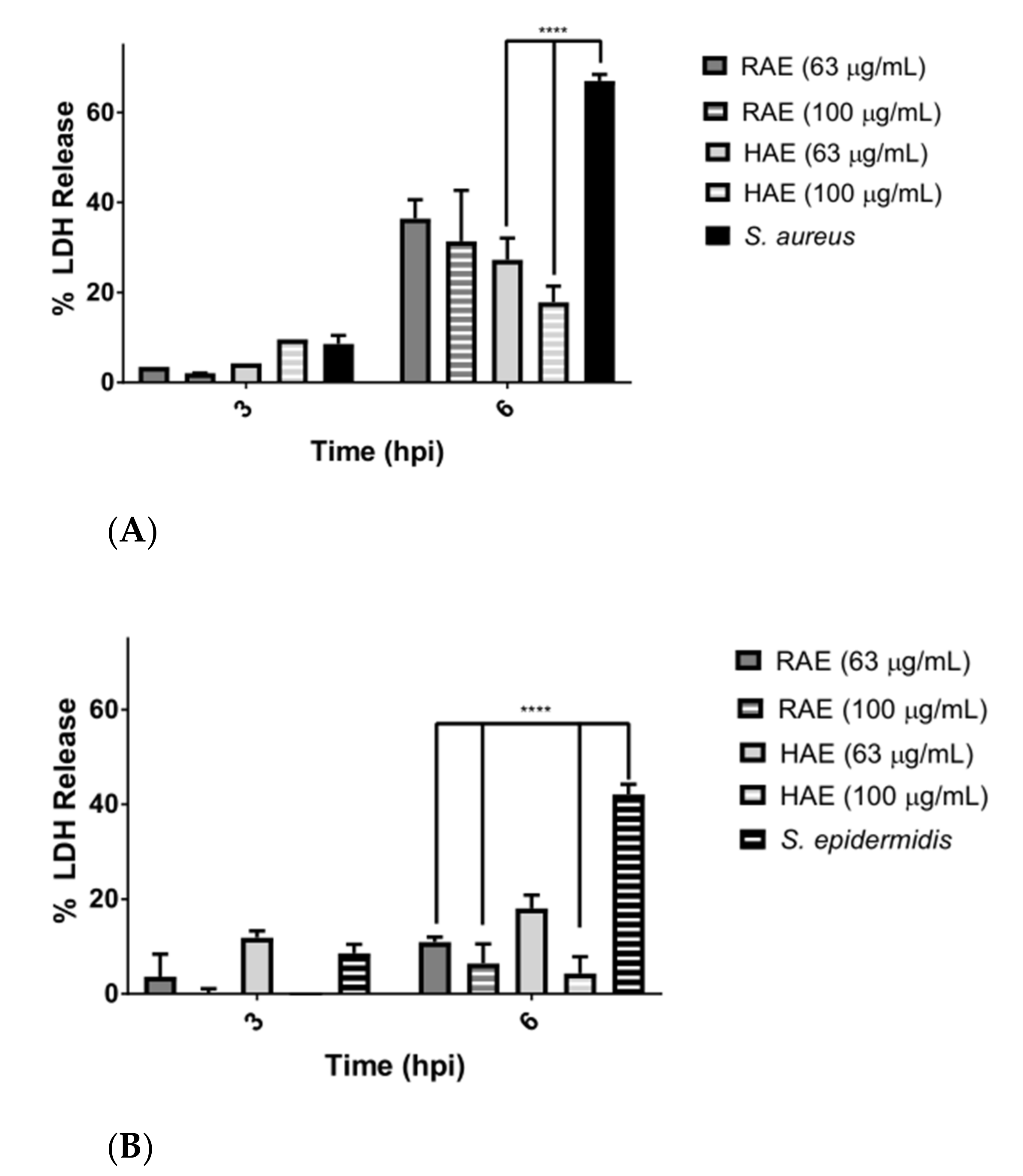
Figure 3. A large difference was observed in the LDH release among the cells infected with the bacteria, but without the presence of the extracts (
Figure 4A,B). These results confirmed the protective effect of the extracts on cells damaged by pathogens.
Previous reports showed that daidzein inhibits the expression of caspases 3 and 9 in viral infections in vitro [
69], while pyrogallol inhibits bacterial infections in vivo [
70]. In addition, a decreased release of LDH and a decreased expression of IL-1β were observed in bacterial infections, suggesting that these compounds prevent an increase in the oxidative stress produced by infections [
71].
The differences between
S. aureus and
S. epidermidis in the rate of LDH release may be due to the fact that
S. aureus interacts more aggressively with the host cell and releases exoenzymes into the intracellular medium (SpIF proteases were detected), thus disrupting the metabolism of the eukaryotic cell [
58]. Meanwhile,
S. epidermidis is characterized by accompanying
S. aureus as an opportunistic pathogen, generating a bacterial biofilm more rapidly, and being a reservoir of multidrug-resistance genes [
72,
73].
The reduction in the percentage of ROS may be a product of infection inhibition or the extracts may have exerted an intracellular effect, reduced the production of ROS or decreasing their concentration.
S. aureus produces Dps proteins, which are responsible for resistance to the nitric oxide and hydrogen peroxide produced by the infected cell [
74,
75], and are a mechanism of DNA protection. The subsequent reduction in ROS forces the infected cells to produce higher concentrations of ROS to defend themselves against pathogens.
As shown in
Figure 5E, preincubation of cells with
MS extracts produced a protective effect on the cells, possibly helping to avoid infection by
S. epidermidis. The RAE extract (87.35% less ROS in a concentration of 100 µg/mL) had the lowest rate of ROS formation in the presence of bacteria. These results suggest either a higher expression of antioxidant proteins, such as the enzymes glutathione peroxidase and superoxide dismutase [
76], or a suppression of pro-oxidative proteins, such as COX-2.
The ability of a wide variety of flavonoids and stilbenes to inhibit pro-oxidative pathways and suppress the COX-2 enzyme has been previously reported [
77].
MS extracts have also been reported to protect against oxidative stress by activating molecular cascades involved in signal transduction, stress, and extracellular matrix synthesis and repair [
42]. It has been reported in our laboratory and in the literature that polyphenols in
MS extracts reduce the concentration of ROS produced by oxidative stress, as is the case with resveratrol [
78] and pyrogallol [
79]. Daidzein and diadzin can reduce ROS concentrations and LDH release in response to oxidative damage, raising the activity of the enzyme superoxide dismutase and lowering the expression of COX-2 and NF-κβ [
64,
80,
81,
82]. Thus, when HDFa cells were pre-incubating with 100 µg/mL RAE and then infected with
S. aureus, a 64% reduction in ROS formation was observed relative to infected controls (
Figure 5B,C).
Coumarin derivatives such as protocatechuic acid, psoralen, and catechol in
MS extracts have been reported to inhibit NF-κβ, reduce ROS, and improve cell survival [
12,
71].