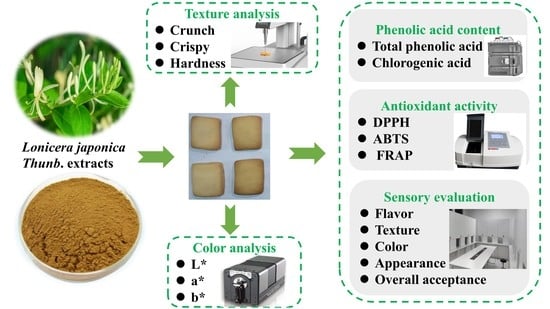
Cookies Fortified with Lonicera japonica Thunb. Extracts: Impact on Phenolic Acid Content, Antioxidant Activity and Physical Properties
Abstract
:Highlights
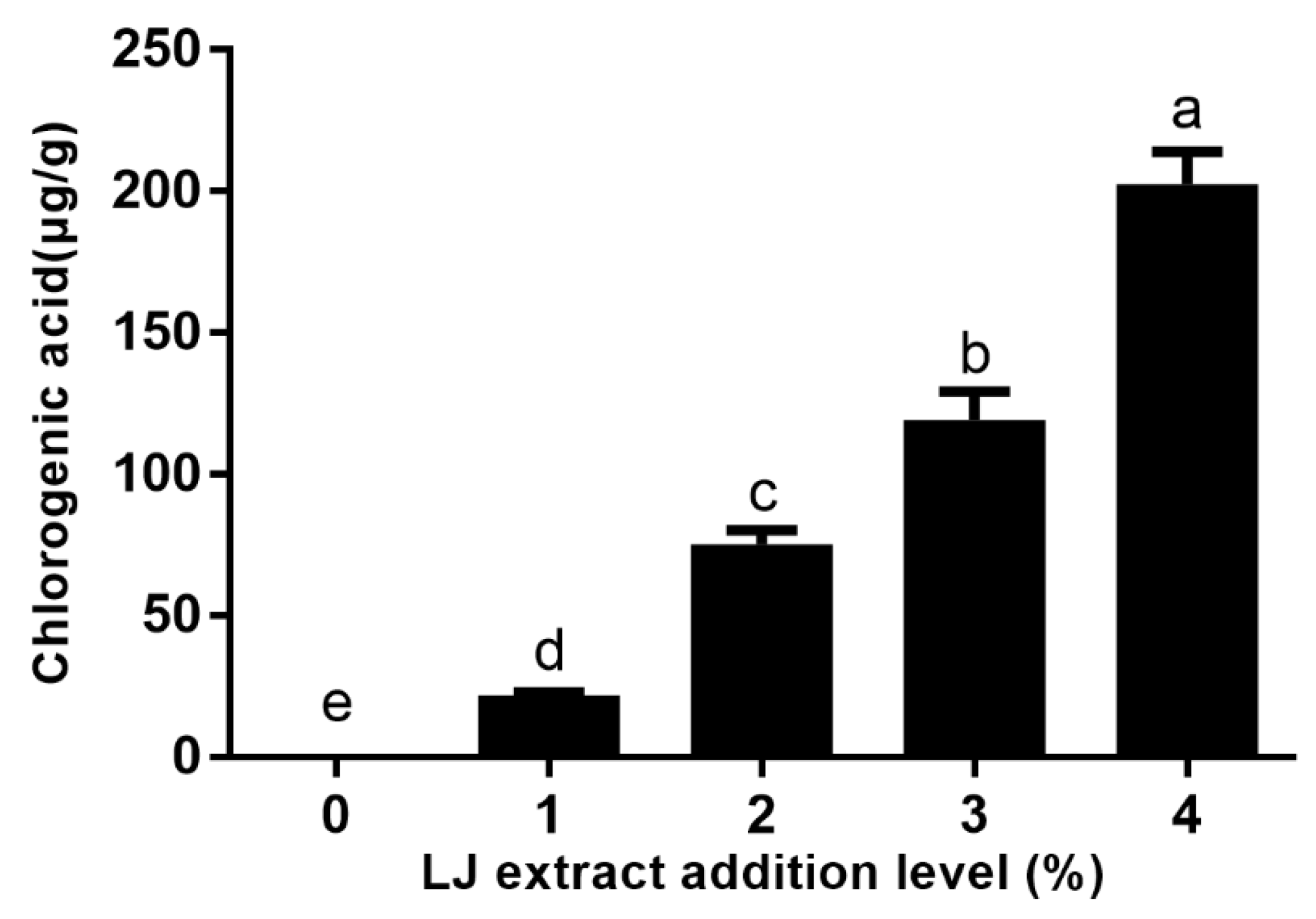
- Cookies with a 4% level of LJ extracts possessed the highest chlorogenic acid content.
- The addition of higher levels of LJ extracts significantly increased higher antioxidant activity of cookies.
- Cookies with a 1% level of LJ extracts had the highest sensory score.
Abstracts
1. Introduction
2. Results and Discussion
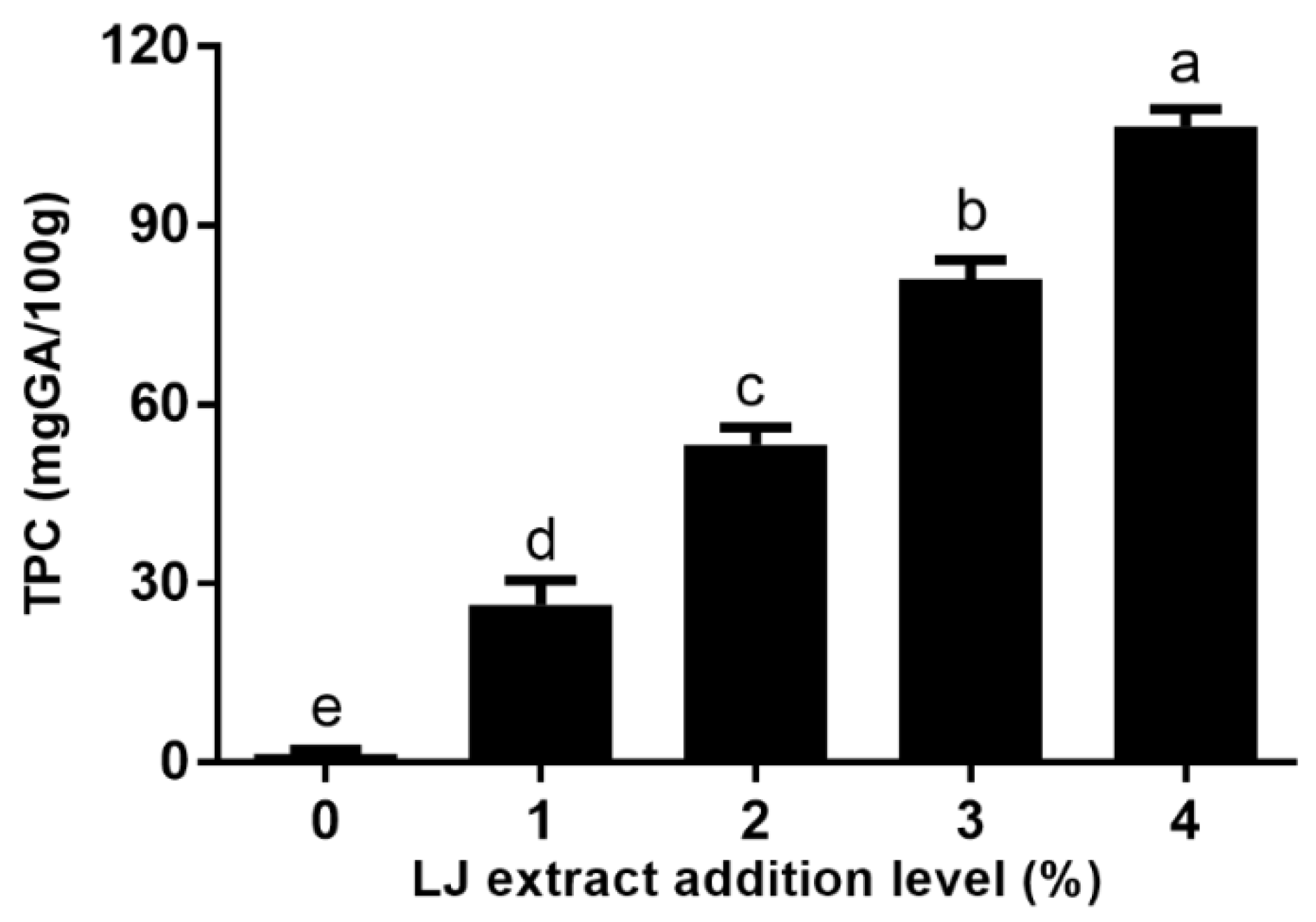
2.1. The TPC of Cookies
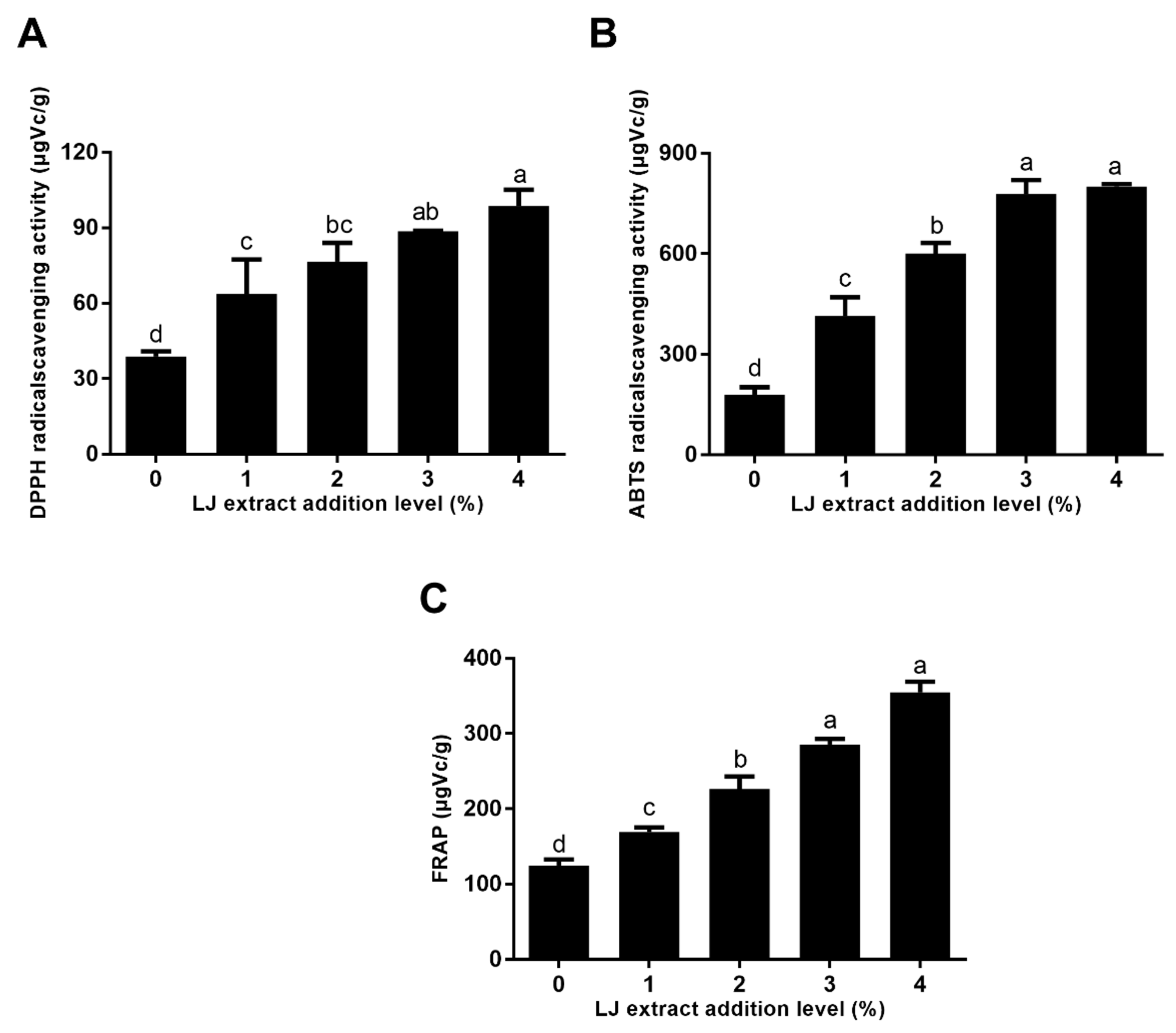
2.2. The Antioxidant Activity of Cookies
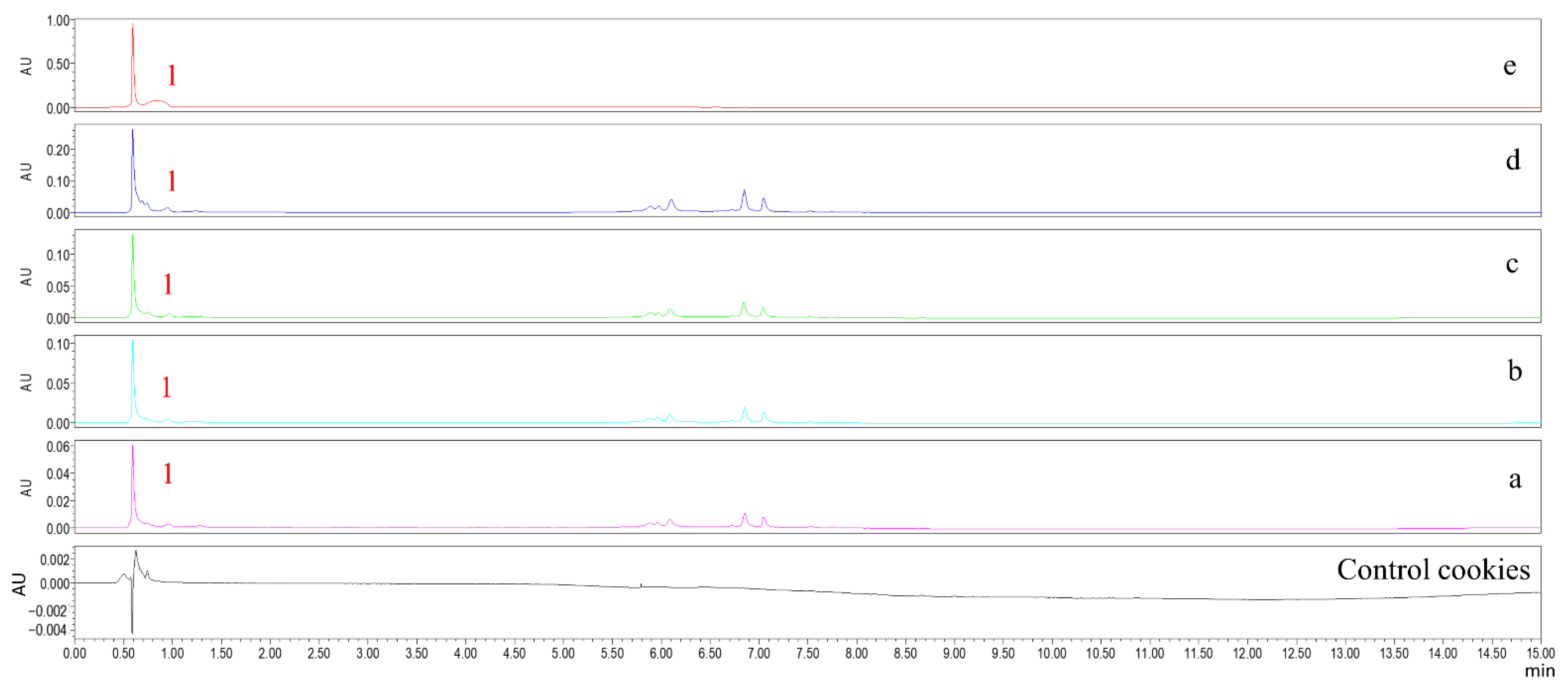
2.3. Chlorogenic Acid Content in Cookies
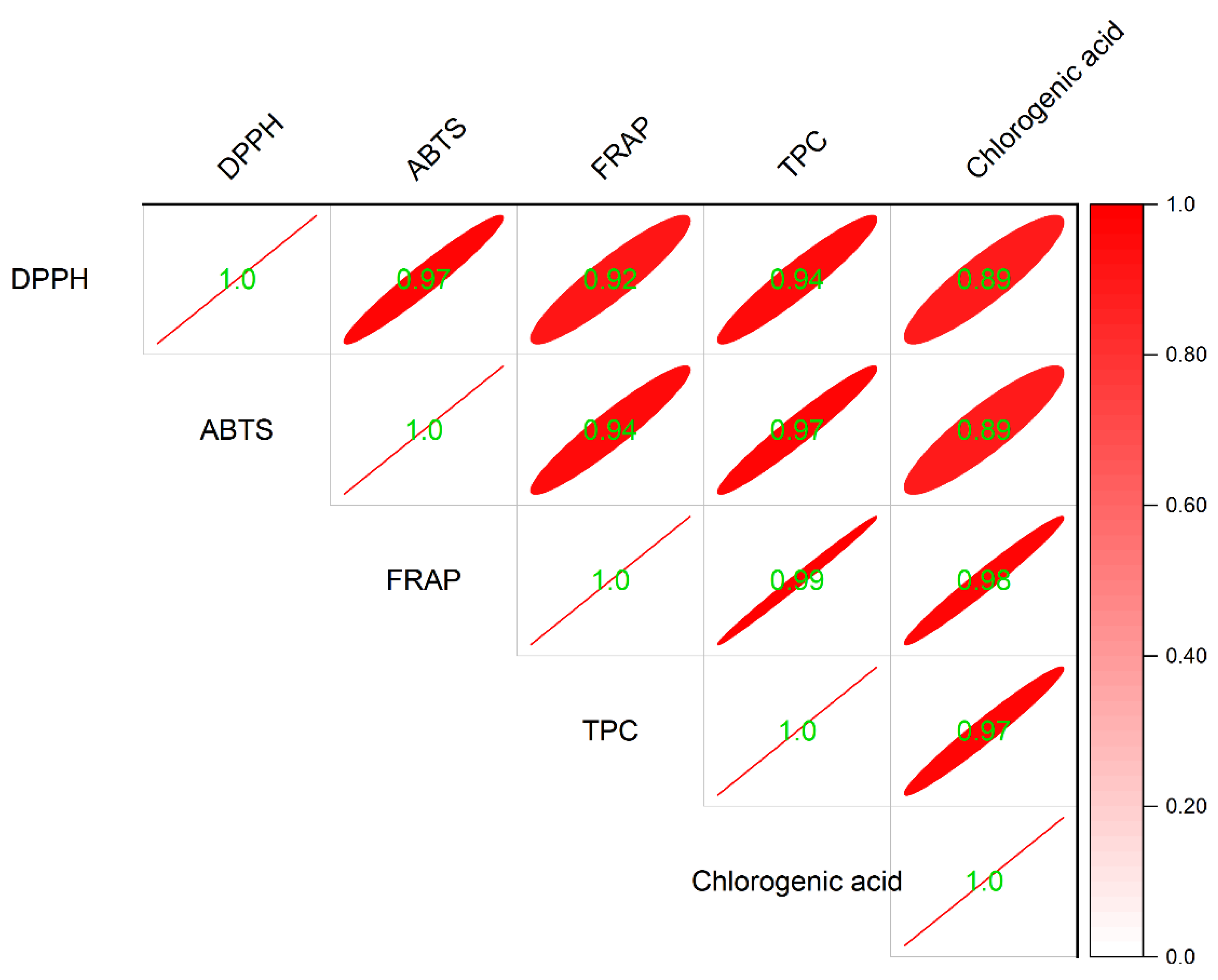
2.4. Correlation Analysis
2.5. Color of Cookies
2.6. Texture Properties of Cookies
2.7. Sensory Evaluation of Cookies
3. Materials and Methods
3.1. Preparation Cookies with LJ Extracts
3.2. Extraction of Phenolic Acids in Cookies
3.3. Determination of the TPC
3.4. Determination of DPPH Free Radical Scavenging Activity
3.5. Determination of ABTS Free Radical Scavenging Activity
3.6. Determination of the FRAP
3.7. UPLC Analysis
3.8. Color of Cookies
3.9. Textural Properties of Cookies
3.10. Sensory Analysis of Cookies
3.11. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Lee, J.; Kang, Y.-R.; Kim, Y.J.; Chang, Y.H. Nutraceuticals and antioxidant properties of Lonicera japonica Thunb. as affected by heating time. Int. J. Food Prop. 2019, 22, 630–645. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Kuang, X.; Wang, W.; Wan, C.; Li, W. Comparison of chemical constitution and bioactivity among different parts of Lonicera japonica Thunb. J. Sci. Food Agric. 2019, 100, 614–622. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhao, X.; Liu, Y. Hypoglycemic and hypolipidemic effects of a polysaccharide from flower buds of Lonicera japonica in streptozotocin-induced diabetic rats. Int. J. Biol. Macromol. 2017, 102, 396–404. [Google Scholar] [CrossRef]
- Qin, L.; Fang, J.; Wang, P.; Du, Z.; Li, Y.; Wang, S.; Kan, D. Characterization of a pectin from Lonicera japonica Thunb. and its inhibition effect on Aβ42 aggregation and promotion of neuritogenesis. Int. J. Biol. Macromol. 2017, 107, 112–120. [Google Scholar]
- Hu, M.; Hu, Z.; Du, L.; Du, J.; Luo, Q.; Xiong, J. Establishment of cell suspension culture of Lonicera japonica Thunb and analysis its major secondary metabolites. Ind. Crops Prod. 2019, 137, 98–104. [Google Scholar] [CrossRef]
- Hfha, B.; Pch, B.; Tck, B.; Stc, C.; Slc, D.; Sjc, E.; Xhla, F.; Mtl, F.; Wyc, A.; Jyha, F. Antioxidant and anti-inflammatory activities of Lonicera japonica Thunb. var. sempervillosa Hayata flower bud extracts prepared by water, ethanol and supercritical fluid extraction techniques. Ind. Crops Prod. 2016, 89, 543–549. [Google Scholar]
- Ning, X.; Wu, J.; Luo, Z.; Chen, Y.; Mo, Z.; Luo, R.; Bai, C.; Du, W.; Wang, L. Cookies fortified with purple passion fruit epicarp flour: Impact on physical properties, nutrition, in vitro starch digestibility, and antioxidant activity. Cereal Chem. 2020, 98, 328–336. [Google Scholar] [CrossRef]
- Liang, L.; Wu, X.; Zhao, T.; Zhao, J.; Li, F.; Zou, Y.; Mao, G.; Yang, L. In vitro bioaccessibility and antioxidant activity of anthocyanins from mulberry (Morus atropurpurea Roxb.) following simulated gastro-intestinal digestion. Food Res. Int. 2012, 46, 76–82. [Google Scholar] [CrossRef]
- Han, I.; Lee, C.-S. Quality properties and bioactivities of American cookies with coffee extract residues. LWT 2021, 151, 112173. [Google Scholar] [CrossRef]
- Canalis, M.B.; Baroni, M.; León, A.; Ribotta, P. Effect of peach puree incorportion on cookie quality and on simulated digestion of polyphenols and antioxidant properties. Food Chem. 2020, 333, 127464. [Google Scholar] [CrossRef]
- Tyagi, P.; Chauhan, A.K.; Singh, S.N. Sensory acceptability of value added cookies incorporated with Tinospora cordifolia (TC) stem powder; improvement in nutritional properties and antioxidant potential. J. Food Sci. Technol. 2020, 57, 2934–2940. [Google Scholar] [CrossRef] [PubMed]
- Dundar, A.N. Total phenolic and antioxidant bioaccessibilities of cookies enriched with bee pollen. J. Food Process. Preserv. 2021, 46, e16085. [Google Scholar] [CrossRef]
- Polat, H.; Capar, T.D.; Inanir, C.; Ekici, L.; Yalcin, H. Formulation of functional crackers enriched with germinated lentil extract: A Response Surface Methodology Box-Behnken Design. LWT 2020, 123, 109065. [Google Scholar] [CrossRef]
- Zhang, Z.; Luo, A.; Zhong, K.; Huang, Y.; Gao, Y.; Zhang, J.; Gao, H.; Xu, Z.; Gao, X. α-Glucosidase inhibitory activity by the flower buds of Lonicera japonica Thunb. J. Funct. Foods 2013, 5, 1253–1259. [Google Scholar] [CrossRef]
- Krystyjan, M.; Gumul, D.; Ziobro, R.; Korus, A. The fortification of biscuits with bee pollen and its effect on physicochemical and antioxidant properties in biscuits. LWT 2015, 63, 640–646. [Google Scholar] [CrossRef]
- Chagas, E.; Vanin, F.M.; Augusto, V.; Garcia, S.; Carvalho, R. Enrichment of antioxidants compounds in cookies produced with camu-camu (Myrciaria dubia) coproducts powders. LWT Food Sci. Technol. 2020, 137, 110472. [Google Scholar] [CrossRef]
- Ramashia, S.E.; Mamadisa, F.M.; Mashau, M.E. Effect of Parinari curatellifolia peel flour on the nutritional, physical and antioxidant properties of biscuits. Processes 2021, 9, 1262. [Google Scholar] [CrossRef]
- Gbenga-Fabusiwa, F.J.; Oladele, E.P.; Oboh, G.; Adefegha, S.A.; Oshodi, A.A. Polyphenol contents and antioxidants activities of biscuits produced from ginger-enriched pigeon pea-wheat composite flour blends. J. Food Biochem. 2018, 42, e12526. [Google Scholar] [CrossRef]
- Papillo, V.A.; Locatelli, M.; Travaglia, F.; Bordiga, M.; Garino, C.; Arlorio, M.; Coïsson, J.D. Spray-dried polyphenolic extract from Italian black rice (Oryza sativa L., var. Artemide) as new ingredient for bakery products. Food Chem. 2018, 269, 603–609. [Google Scholar] [CrossRef]
- Turki, M.; Barbosa-Pereira, L.; Bertolino, M.; Essaidi, I.; Ghirardello, D.; Torri, L.; Bouzouita, N.; Zeppa, G. Physico-Chemical Characterization of Tunisian Canary Palm (Phoenix canariensis Hort. Ex Chabaud) Dates and Evaluation of Their Addition in Biscuits. Foods 2020, 9, 695. [Google Scholar] [CrossRef]
- Lazaridou, A.; Duta, D.; Papageorgiou, M.; Belc, N.; Biliaderis, C.G. Effects of hydrocolloids on dough rheology and bread quality parameters in gluten-free formulations. J. Food Eng. 2007, 79, 1033–1047. [Google Scholar] [CrossRef]
- Cannas, M.; Pulina, S.; Conte, P.; Del Caro, A.; Urgeghe, P.P.; Piga, A.; Fadda, C. Effect of Substitution of Rice Flour with Quinoa Flour on the Chemical-Physical, Nutritional, Volatile and Sensory Parameters of Gluten-Free Ladyfinger Biscuits. Foods 2020, 9, 808. [Google Scholar] [CrossRef] [PubMed]
- Faccioli, L.S.; Klein, M.P.; Borges, G.R.; Dalanhol, C.S.; Machado, I.C.K.; Garavaglia, J.; Bosco, S.M.D. Development of crackers with the addition of olive leaf flour (Olea europaea L.): Chemical and sensory characterization. LWT 2021, 141, 110848. [Google Scholar] [CrossRef]
- Zanchett, C.S.; Mignoni, M.; Barro, N.P.R.; Rosa, C.D. Desenvolvimento de chocolate branco com extrato de erva-mate. Braz. J. Food Technol. 2016, 19, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Theagarajan, R.; Malur Narayanaswamy, L.; Dutta, S.; Moses, J.A.; Chinnaswamy, A. Valorisation of grape pomace (cv. Muscat) for development of functional cookies. Int. J. Food Sci. Technol. 2019, 54, 1299–1305. [Google Scholar] [CrossRef]
- Sferrazzo, G.; Palmeri, R.; Restuccia, C.; Parafati, L.; Siracusa, L.; Spampinato, M.; Carota, G.; Distefano, A.; Di Rosa, M.; Tomasello, B.; et al. Mangifera indica L. Leaves as a Potential Food Source of Phenolic Compounds with Biological Activity. Antioxidants 2022, 11, 1313. [Google Scholar] [CrossRef]
- Dunford, N.T.; Gumus, Z.P.; Gur, C.S. Chemical Composition and Antioxidant Properties of Pecan Shell Water Extracts. Antioxidants 2022, 11, 1127. [Google Scholar] [CrossRef]
- Figueroa, J.G.; Borrás-Linares, I.; del Pino-García, R.; Curiel, J.A.; Lozano-Sánchez, J.; Segura-Carretero, A. Functional ingredient from avocado peel: Microwave-assisted extraction, characterization and potential applications for the food industry. Food Chem. 2021, 352, 129300. [Google Scholar] [CrossRef]

| LJ Extract Level | L* (Lightness) | a* (Redness) | b* (Yellowness) |
|---|---|---|---|
| 0% | 73.22 ± 1.13 d | 4.60 ± 0.32 a | 30.91 ± 0.91 a |
| 1% | 69.24 ± 0.39 c | 5.49 ± 0.21 b | 35.19 ± 1.10 b |
| 2% | 64.46 ± 1.21 b | 5.50 ± 0.13 b | 36.81 ± 0.42 c |
| 3% | 64.24 ± 0.64 b | 5.63 ± 0.12 b | 36.87 ± 0.09 c |
| 4% | 62.30 ± 0.26 a | 5.68 ± 0.12 b | 37.48 ± 0.22 c |
| LJ Extract Level | Crunch | Crispy | Hardness |
|---|---|---|---|
| 0 | 3.00 ± 1.52 a | 8.75 ± 0.96 a | 2061.80 ± 196.77 c |
| 1% | 3.50 ± 1.92 a | 10.00 ± 1.41 ab | 1995.75 ± 129.81 b |
| 2% | 3.75 ± 1.26 a | 10.25 ± 1.50 ab | 1968.08 ± 138.69 ab |
| 3% | 3.75 ± 1.50 a | 11.00 ± 2.45 ab | 1786.67 ± 58.16 a |
| 4% | 4.25 ± 0.50 a | 12.00 ± 1.63 b | 1540.32 ± 72.06 a |
| LJ Extract Level | Flavor | Texture | Color | Appearance | Overall Acceptance |
|---|---|---|---|---|---|
| 0% | 27.67 ± 0.58 a | 22.67 ± 2.08 ab | 15.67 ± 0.58 a | 18.33 ± 0.58 a | 84.33 ± 1.53 a |
| 1% | 27.67 ± 0.58 a | 26.00 ± 1.00 a | 15.67 ± 1.53 a | 16.33 ± 1.15 ab | 85.67 ± 3.21 a |
| 2% | 21.67 ± 2.89 b | 25.00 ± 1.00 a | 13.33 ± 0.58 b | 15.33 ± 0.58 b | 75.33 ± 3.21 b |
| 3% | 20.33 ± 1.53 b | 23.33 ± 2.89 ab | 11.67 ± 0.58 b | 15.67 ± 2.31 b | 71.00 ± 4.36 b |
| 4% | 11.67 ± 3.79 c | 20.33 ± 2.52 b | 9.33 ± 1.15 c | 11.33 ± 1.53 c | 52.67 ± 3.79 c |
| Ingredients | Formulations | ||||
|---|---|---|---|---|---|
| Control | 1% | 2% | 3% | 4% | |
| Wheat flour | 100 | 99 | 98 | 97 | 96 |
| LJ extracts | 0 | 1 | 2 | 3 | 4 |
| Sugar | 25 | 25 | 25 | 25 | 25 |
| Butter | 30 | 30 | 30 | 30 | 30 |
| Baking soda | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, W.; Chen, J.; Li, L.; Ren, G.; Duan, X.; Zhou, Q.; Zhang, M.; Gao, D.; Zhang, S.; Liu, X. Cookies Fortified with Lonicera japonica Thunb. Extracts: Impact on Phenolic Acid Content, Antioxidant Activity and Physical Properties. Molecules 2022, 27, 5033. https://doi.org/10.3390/molecules27155033
Cao W, Chen J, Li L, Ren G, Duan X, Zhou Q, Zhang M, Gao D, Zhang S, Liu X. Cookies Fortified with Lonicera japonica Thunb. Extracts: Impact on Phenolic Acid Content, Antioxidant Activity and Physical Properties. Molecules. 2022; 27(15):5033. https://doi.org/10.3390/molecules27155033
Chicago/Turabian StyleCao, Weiwei, Junliang Chen, Linlin Li, Guangyue Ren, Xu Duan, Qian Zhou, Mengli Zhang, Danping Gao, Shanshan Zhang, and Xu Liu. 2022. "Cookies Fortified with Lonicera japonica Thunb. Extracts: Impact on Phenolic Acid Content, Antioxidant Activity and Physical Properties" Molecules 27, no. 15: 5033. https://doi.org/10.3390/molecules27155033
APA StyleCao, W., Chen, J., Li, L., Ren, G., Duan, X., Zhou, Q., Zhang, M., Gao, D., Zhang, S., & Liu, X. (2022). Cookies Fortified with Lonicera japonica Thunb. Extracts: Impact on Phenolic Acid Content, Antioxidant Activity and Physical Properties. Molecules, 27(15), 5033. https://doi.org/10.3390/molecules27155033