Synthesis of a Novel Unexpected Cu(II)–Thiazolidine Complex—X-ray Structure, Hirshfeld Surface Analysis, and Biological Studies
Abstract
:1. Introduction
2. Results and Discussion
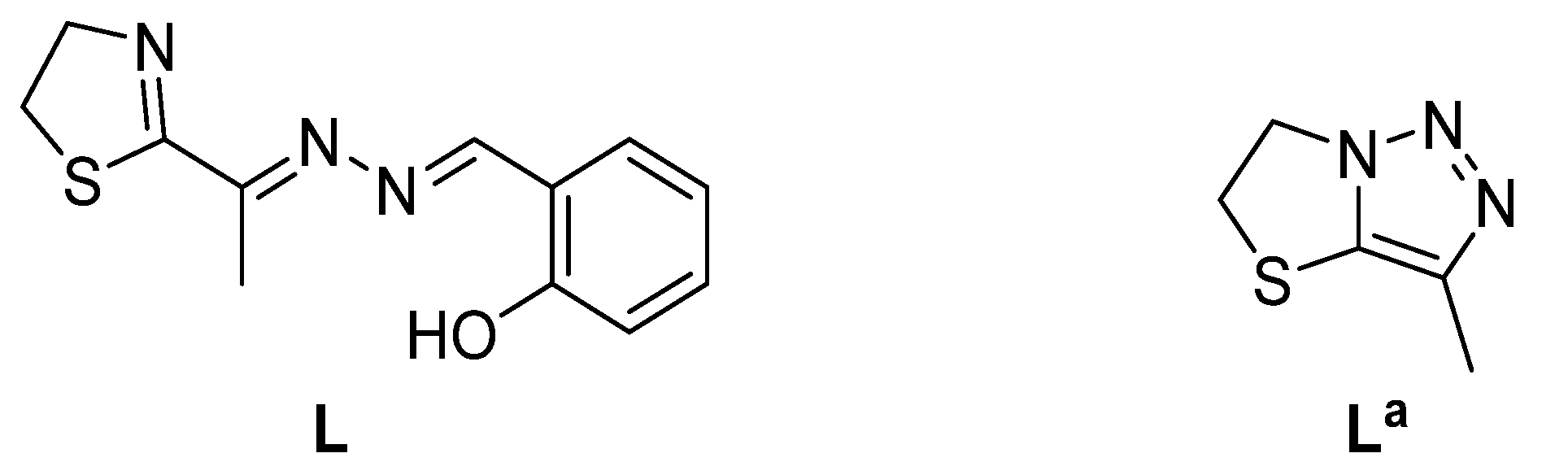
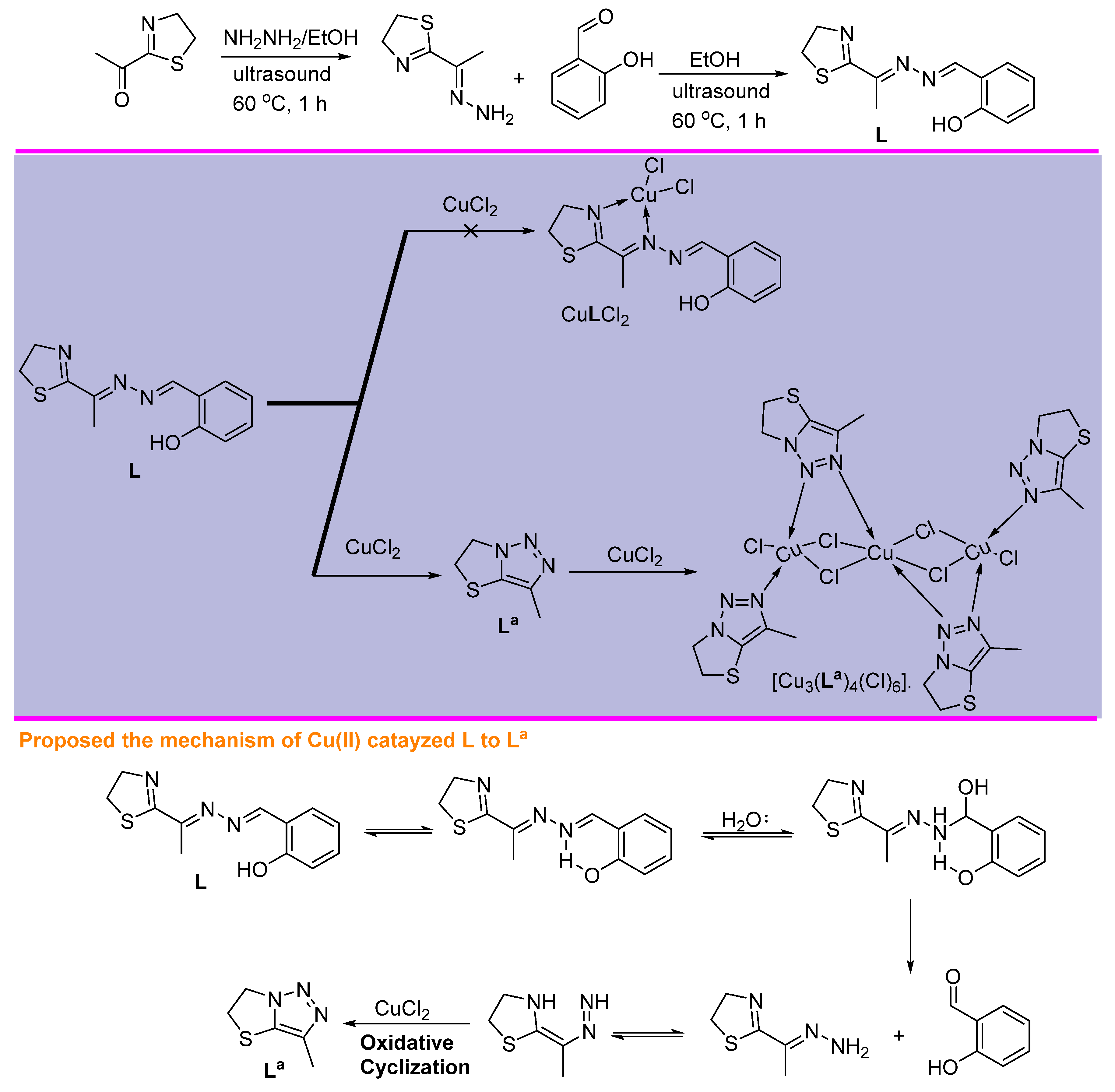
2.1. Chemistry and Characterizations
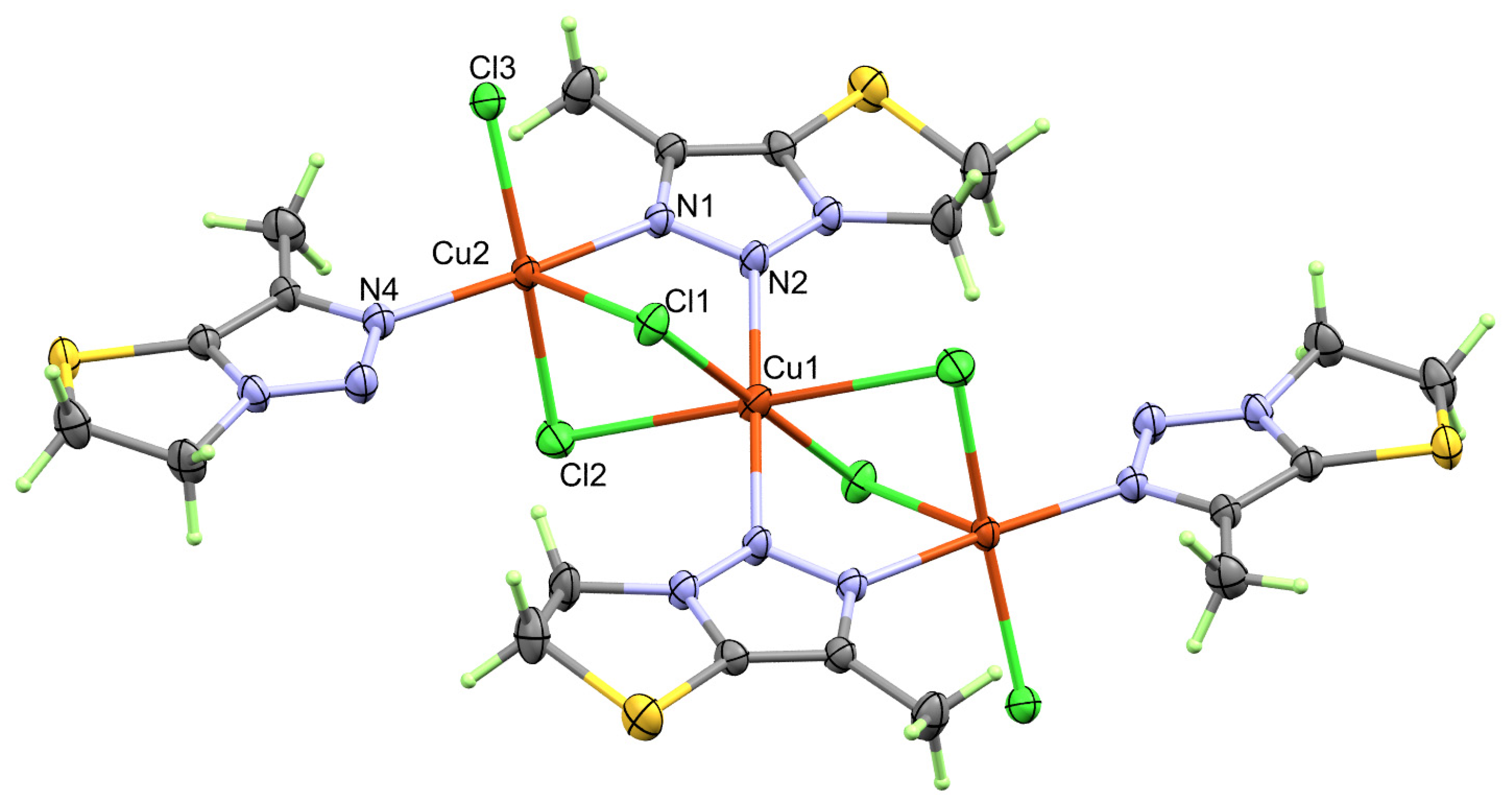
2.2. X-ray Structure Description of [Cu3(La)4(Cl)6] Complex
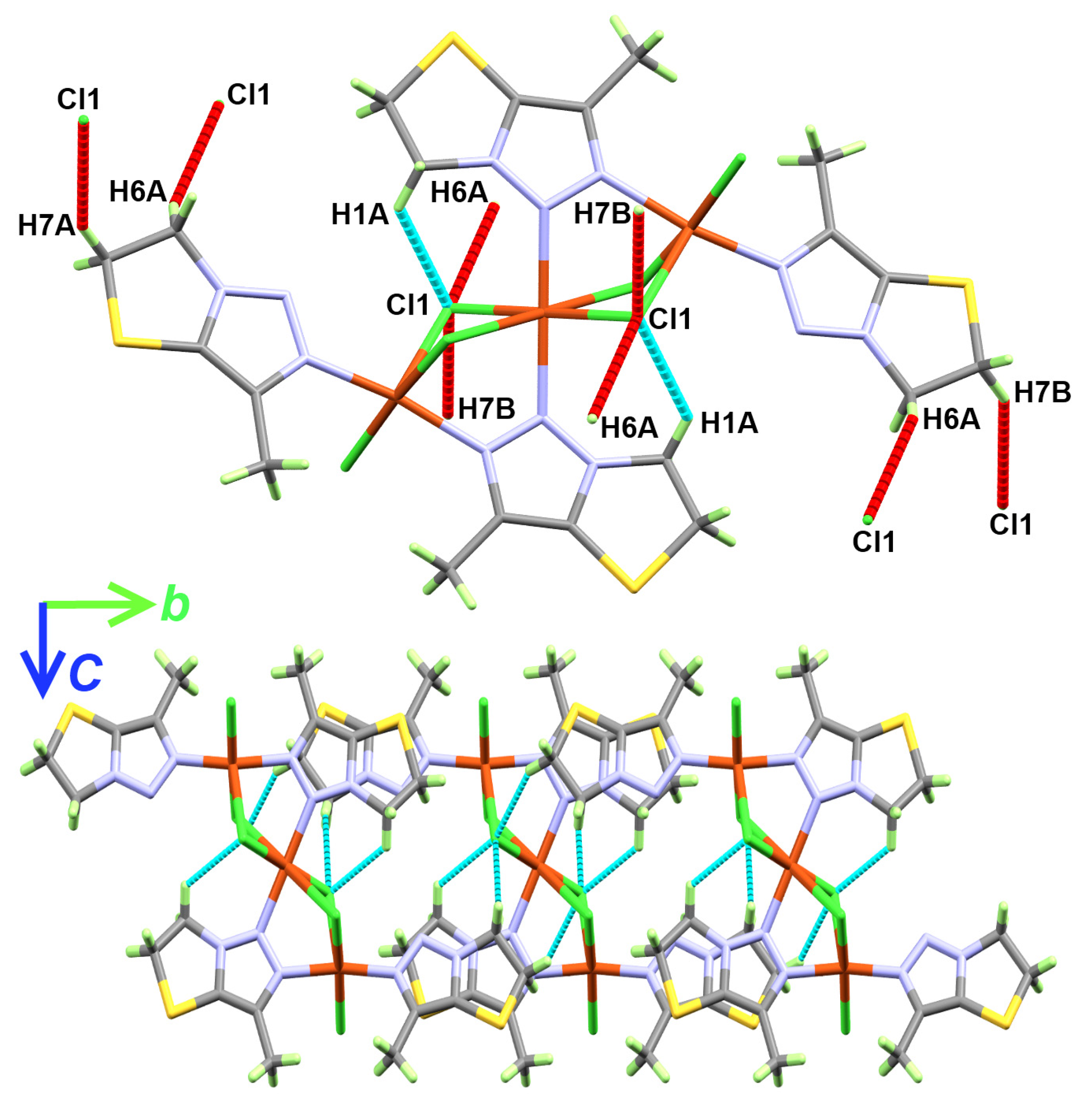
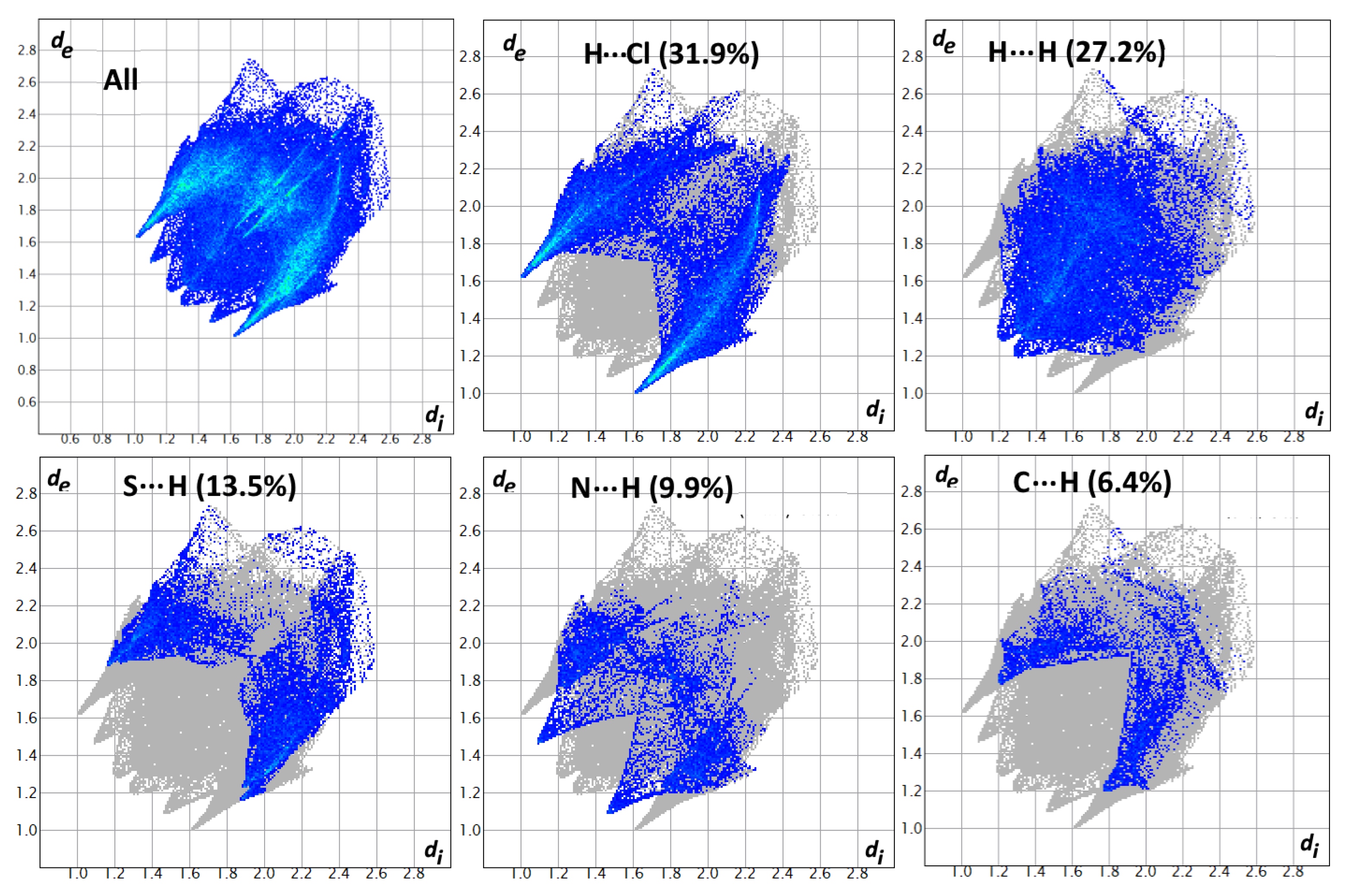
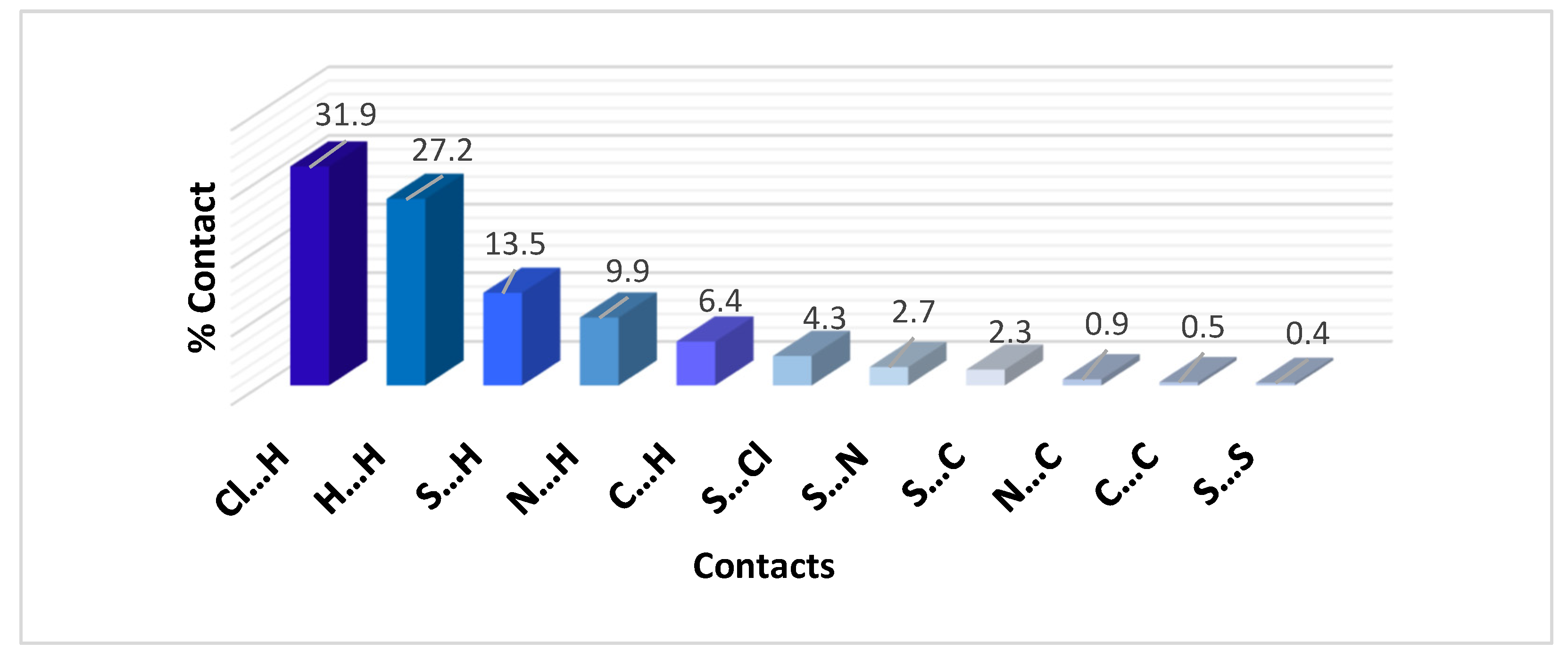
2.3. Hirshfeld Analysis
2.4. XPS Studies
2.5. Biological Studies
2.5.1. Antimicrobial Activity
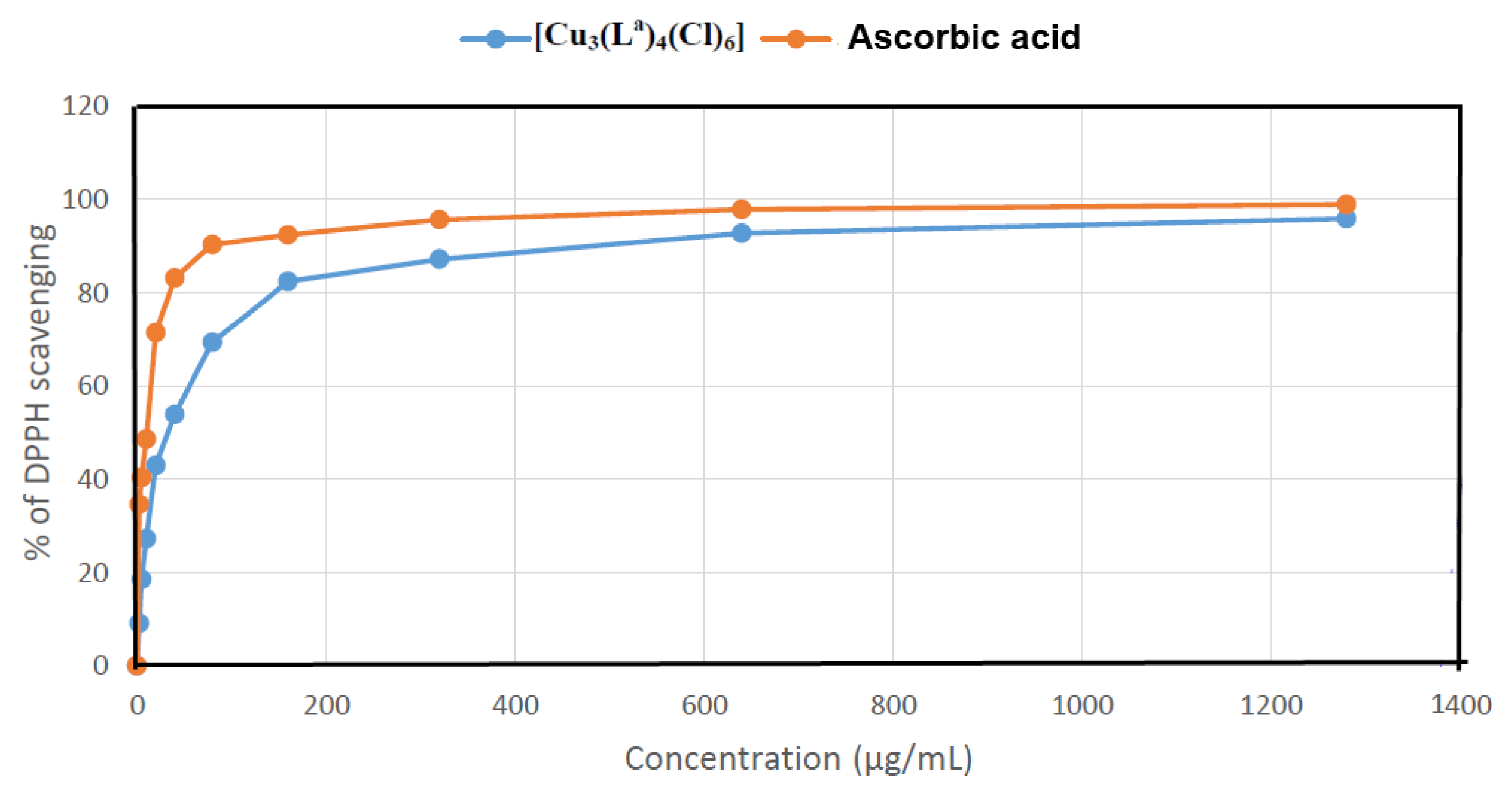
2.5.2. Antioxidant Activity
2.5.3. Cytotoxic Activity against Colon Carcinoma (HCT-116 Cell)
3. Experimental
3.1. Materials
3.2. Instrumentations
3.3. Synthesis of 2-(((1-(4,5-Dihydrothiazol-2-yl)ethylidene)hydrazono)methyl)phenol; L
3.4. Synthesis of [Cu3(La)4(Cl)6] Complex
3.5. X-ray Structure Determination
3.6. Biolological Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Benesperi, I.; Singh, R.; Freitag, M. Copper coordination complexes for energy-relevant applications. Energies 2020, 13, 2198. [Google Scholar] [CrossRef]
- Kardos, J.; Héja, L.; Simon, Á.; Jablonkai, I.; Kovács, R.; Jemnitz, K. Copper signalling: Causes and consequences. Cell Commun. Signal. 2018, 16, 71. [Google Scholar] [CrossRef] [Green Version]
- Georgopoulos, P.G.; Roy, A.; Yonone-Lioy, M.J.; Opiekun, R.E.; Lioy, P.J. Environmental copper: Its dynamics and human exposure issues. J. Toxicol. Environ. Health Part B Crit. Rev. 2001, 4, 341–394. [Google Scholar] [CrossRef] [PubMed]
- Tapia, L.; González-Agüero, M.; Cisternas, M.F.; Suazo, M.; Cambiazo, V.; Uauy, R.; González, M. Metallothionein is crucial for safe intracellular copper storage and cell survival at normal and supra-physiological exposure levels. Biochem. J. 2004, 378, 617–624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blockhuys, S.; Wittung-Stafshede, P. Roles of copper-binding proteins in breast cancer. Int. J. Mol. Sci. 2017, 18, 871. [Google Scholar] [CrossRef] [Green Version]
- Neumann, W.; Gulati, A.; Nolan, E.M. Metal homeostasis in infectious disease: Recent advances in bacterial metallophores and the human metal-withholding response. Curr. Opin. Chem. Biol. 2017, 37, 10–18. [Google Scholar] [CrossRef] [Green Version]
- Srivastava, S.; Panda, S.; Li, Z.; Fuhs, S.R.; Hunter, T.; Thiele, D.J.; Hubbard, S.R.; Skolnik, E.Y. Histidine phosphorylation relieves copper inhibition in the mammalian potassium channel KCa3. 1. eLife 2016, 5, e16093. [Google Scholar] [CrossRef] [Green Version]
- Sun, T.-S.; Ju, X.; Gao, H.-L.; Wang, T.; Thiele, D.J.; Li, J.-Y.; Wang, Z.-Y.; Ding, C. Reciprocal functions of Cryptococcus neoformans copper homeostasis machinery during pulmonary infection and meningoencephalitis. Nat. Commun. 2014, 5, 5550. [Google Scholar] [CrossRef] [Green Version]
- Wiemann, P.; Perevitsky, A.; Lim, F.Y.; Shadkchan, Y.; Knox, B.P.; Figueora, J.A.L.; Choera, T.; Niu, M.; Steinberger, A.J.; Wüthrich, M. Aspergillus fumigatus copper export machinery and reactive oxygen intermediate defense counter host copper-mediated oxidative antimicrobial offense. Cell Rep. 2017, 19, 1008–1021. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Geng, X.; McDermott, J.; Shen, J.; Corbin, C.; Xuan, S.; Kim, J.; Zuo, L.; Liu, Z. Copper deficiency in the lungs of TNF-α transgenic mice. Front. Physiol. 2016, 7, 234. [Google Scholar] [CrossRef] [Green Version]
- Cypryk, W.; Lorey, M.; Puustinen, A.; Nyman, T.A.; Matikainen, S. Proteomic and bioinformatic characterization of extracellular vesicles released from human macrophages upon influenza A virus infection. J. Proteome Res. 2017, 16, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Bandmann, O.; Weiss, K.H.; Kaler, S.G. Wilson’s disease and other neurological copper disorders. Lancet Neurol. 2015, 14, 103–113. [Google Scholar] [CrossRef] [Green Version]
- Bansagi, B.; Lewis-Smith, D.; Pal, E.; Duff, J.; Griffin, H.; Pyle, A.; Müller, J.S.; Rudas, G.; Aranyi, Z.; Lochmüller, H. Phenotypic convergence of Menkes and Wilson disease. Neurol. Genet. 2016, 2, e119. [Google Scholar] [CrossRef] [Green Version]
- Manto, M. Abnormal copper homeostasis: Mechanisms and roles in neurodegeneration. Toxics 2014, 2, 327–345. [Google Scholar] [CrossRef]
- Viles, J.H. Metal ions and amyloid fiber formation in neurodegenerative diseases. Copper, zinc and iron in Alzheimer’s, Parkinson’s and prion diseases. Coord. Chem. Rev. 2012, 256, 2271–2284. [Google Scholar] [CrossRef]
- Bella, F.; Galliano, S.; Gerbaldi, C.; Viscardi, G. Cobalt-based electrolytes for dye-sensitized solar cells: Recent advances towards stable devices. Energies 2016, 9, 384. [Google Scholar] [CrossRef] [Green Version]
- Sprengard, R.; Bonrad, K.; Daeubler, T.K.; Frank, T.; Hagemann, V.; Koehler, I.; Pommerehne, J.; Ottermann, C.R.; Voges, F.; Vingerling, B. OLED devices for signage applications: A review of recent advances and remaining challenges. Org. Light-Emit. Mater. Devices VIII 2004, 5519, 173–183. [Google Scholar]
- Liu, Y.; Yiu, S.-C.; Ho, C.-L.; Wong, W.-Y. Recent advances in copper complexes for electrical/light energy conversion. Coord. Chem. Rev. 2018, 375, 514–557. [Google Scholar] [CrossRef]
- Prakash, G.; Nirmala, M.; Ramachandran, R.; Viswanathamurthi, P.; Malecki, J.G.; Sanmartin, J. Heteroleptic binuclear copper (I) complexes bearing bis (salicylidene) hydrazone ligands: Synthesis, crystal structure and application in catalytic N-alkylation of amines. Polyhedron 2015, 89, 62–69. [Google Scholar] [CrossRef]
- Ruben, M.; Lehn, J.-M.; Vaughan, G. Synthesis of ionisable [2×2] grid-type metallo-arrays and reversible protonic modulation of the optical properties of the [CoII4 L4]8+ species. Chem. Commun. 2003, 12, 1338–1339. [Google Scholar] [CrossRef]
- Uppadine, L.H.; Gisselbrecht, J.-P.; Lehn, J.-M. Protonic modulation of redox properties in ionisable [2×2] grid-like metalloarrays. Chem. Commun. 2004, 718–719. [Google Scholar] [CrossRef] [PubMed]
- Wood, A.; Aris, W.; Brook, D.J. Coordinated hydrazone ligands as nucleophiles: Reactions of Fe(papy)2. Inorg. Chem. 2004, 43, 8355–8360. [Google Scholar] [CrossRef]
- Uppadine, L.H.; Lehn, J.M. Three-Level Synthetic Strategy Towards Mixed-Valence and Heterometallic [2×2] Gridlike Arrays. Angew. Chem. 2004, 116, 242–245. [Google Scholar] [CrossRef]
- Qi, Y.; Wang, Z.Y. Dendritic mixed-valence dinuclear ruthenium complexes for optical attenuation at telecommunication wavelengths. Macromolecules 2003, 36, 3146–3151. [Google Scholar] [CrossRef]
- Bermejo, M.R.; Fondo, M.; González, A.M.; Hoyos, O.L.; Sousa, A.; McAuliffe, C.A.; Hussain, W.; Pritchard, R.; Novotorsev, V.M. Electrochemical synthesis and structural characterization of transition metal complexes with 2, 6-bis(1-salicyloylhydrazonoethyl) pyridine, H4daps. J. Chem. Soc. Dalton Trans. 1999, 2211–2218. [Google Scholar] [CrossRef]
- Bermejo, M.R.; Pedrido, R.; González-Noya, A.M.; Romero, M.J.; Vázquez, M.; Sorace, L. Conformational rearrangement of 2, 6-bis (1-salicyloylhydrazonoethyl) pyridine (H4daps) on complexation. Synthesis and X-ray characterization of H4daps and its copper helicate complex [Cu(H2daps)(H2O)]2· 2CH3CN. New J. Chem. 2003, 27, 1753–1759. [Google Scholar] [CrossRef]
- Naskar, S.; Mishra, D.; Chattopadhyay, S.K.; Corbella, M.; Blake, A.J. Versatility of 2, 6-diacetylpyridine (dap) hydrazones in stabilizing uncommon coordination geometries of Mn (II): Synthesis, spectroscopic, magnetic and structural characterization. Dalton Trans. 2005, 2428–2435. [Google Scholar] [CrossRef]
- Naskar, S.; Corbella, M.; Blake, A.J.; Chattopadhyay, S.K. Versatility of 2,6-diacetylpyridine (dap) hydrazones in generating varied molecular architectures: Synthesis and structural characterization of a binuclear double helical Zn (II) complex and a Mn (II) coordination polymer. Dalton Trans. 2007, 1150–1159. [Google Scholar] [CrossRef]
- Rao, S.N.; Mishra, D.; Maurya, R.; Rao, N.N. Oxovanadium binuclear (IV) Schiff base complexes derived from aroyl hydrazones having subnormal magnetic moments. Polyhedron 1997, 16, 1825–1829. [Google Scholar] [CrossRef]
- Fujita, E.; Brunschwig, B.S.; Ogata, T.; Yanagida, S. Toward photochemical carbon dioxide activation by transition metal complexes. Coord. Chem. Rev. 1994, 132, 195–200. [Google Scholar] [CrossRef]
- Kimura, E.; Wada, S.; Shionoya, M.; Okazaki, Y. New series of multifunctionalized nickel (II)-cyclam (cyclam= 1, 4, 8, 11-tetraazacyclotetradecane) complexes. Application to the photoreduction of carbon dioxide. Inorg. Chem. 1994, 33, 770–778. [Google Scholar] [CrossRef]
- Nagasree, K.P.; Kumar, M.M.K.; Prasad, Y.R.; Sriram, D.; Yogeeswari, P. Synthesis and in vitro studies of thiazolidine-4-carboxylic acid hydrazones as potential antitubercular agents. Indian J. Chem. 2018, 87, 538–555. [Google Scholar]
- Sahiba, N.; Sethiya, A.; Soni, J.; Agarwal, D.K.; Agarwal, S. Saturated Five-Membered Thiazolidines and Their Derivatives: From Synthesis to Biological Applications. Top. Curr. Chem. 2020, 378, 34. [Google Scholar] [CrossRef] [Green Version]
- Havrylyuk, D.; Zimenkovsky, B.; Vasylenko, O.; Lesyk, R. Synthesis and Anticancer and Antiviral Activities of New 2-Pyrazoline-Substituted 4-Thiazolidinones. J. Heterocycl. Chem. 2013, 50, E55–E62. [Google Scholar] [CrossRef]
- Addison, A.W.; Rao, T.N.; Reedijk, J.; van Rijn, J.; Verschoor, G.C. Synthesis, structure, and spectroscopic properties of copper (II) compounds containing nitrogen–sulphur donor ligands; the crystal and molecular structure of aqua [1,7-bis(N-methylbenzimidazol-2′-yl)-2,6-dithiaheptane]copper(II) perchlorate. J. Chem. Soc. Dalton Trans. 1984, 1349–1356. [Google Scholar] [CrossRef]
- Spackman, P.R.; Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer: A program for Hirshfeld surface analysis, visualization and quantitative analysis of molecular crystals. J. Appl. Crystallogr. 2021, 54, 1006–1011. [Google Scholar] [CrossRef] [PubMed]
- Oh, W.-D.; Lee, M.G.-H.; Udayanga, W.C.; Veksha, A.; Bao, Y.; Giannis, A.; Lim, J.-W.; Lisak, G. Insights into the single and binary adsorption of copper (II) and nickel (II) on hexagonal boron nitride: Performance and mechanistic studies. J. Environ. Chem. Eng. 2019, 7, 102872. [Google Scholar] [CrossRef]
- Yuan, H.; Shang, P.; Sun, R.; Yang, J.; Huang, Q.; Song, L.; Wu, C.; Jiang, X.-F. Structural transformation of copper (II)-based complexes driven by N, S cooperative coordination and iodine adsorption behavior. J. Solid State Chem. 2022, 306, 122811. [Google Scholar] [CrossRef]
- Araujo, J.; Archanjo, B.; de Souza, K.; Kwapinski, W.; Falcão, N.; Novotny, E.; Achete, C. Selective extraction of humic acids from an anthropogenic Amazonian dark earth and from a chemically oxidized charcoal. Biol. Fertil. Soils 2014, 50, 1223–1232. [Google Scholar] [CrossRef]
- Winiarski, J.; Tylus, W.; Winiarska, K.; Szczygieł, I.; Szczygieł, B. XPS and FT-IR characterization of selected synthetic corrosion products of zinc expected in neutral environment containing chloride ions. J. Spectrosc. 2018, 2018, 2079278. [Google Scholar] [CrossRef] [Green Version]
- Razzaq, A.A.; Yao, Y.; Shah, R.; Qi, P.; Miao, L.; Chen, M.; Zhao, X.; Peng, Y.; Deng, Z. High-performance lithium sulfur batteries enabled by a synergy between sulfur and carbon nanotubes. Energy Storage Mater. 2019, 16, 194–202. [Google Scholar] [CrossRef]
- Anastasiadis, N.C.; Mylonas-Margaritis, I.; Psycharis, V.; Raptopoulou, C.P.; Kalofolias, D.A.; Milios, C.J.; Klouras, N.; Perlepes, S.P. Dinuclear, tetrakis (acetato)-bridged lanthanide (III) complexes from the use of 2-acetylpyridine hydrazone. Inorg. Chem. Commun. 2015, 51, 99–102. [Google Scholar] [CrossRef]
- El-Faham, A.; Soliman, S.M.; Ghabbour, H.A.; Elnakady, Y.A.; Mohaya, T.A.; Siddiqui, M.R.; Albericio, F. Ultrasonic promoted synthesis of novel s-triazine-Schiff base derivatives; molecular structure, spectroscopic studies and their preliminary anti-proliferative activities. J. Mol. Struct. 2016, 1125, 121–135. [Google Scholar] [CrossRef]
- Kluge, T.; Bette, E.; Bette, M.; Schmidt, J.; Steinborn, D. Hydrazone–diacetyl platinum (II) complexes: Substituent effect on intramolecular N–H...O hydrogen-bond strength. J. Organomet. Chem. 2014, 762, 48–57. [Google Scholar] [CrossRef]
- Tong, H.C.; Wang, P.Y.; Kuo, Y.L.; Lin, C.H.; Lo, Y.H. (Benzonitrile-N) chlorido [hydrido-tris (pyrazol-1-yl-N2) borato](triphenyl-phosphine-P) ruthenium (II) ethanol solvate. Acta Crystallogr. Sect. E 2009, E65, m438. [Google Scholar] [CrossRef] [Green Version]
- Barbazán, P.; Carballo, R.; Covelo, B.; Lodeiro, C.; Lima, J.C.; Vázquez-López, E.M. Synthesis, Characterization, and Photophysical Properties of 2-Hydroxybenzaldehyde [(1E)-1-pyridin-2-ylethylidene] hydrazone and Its Rhenium (I) Complexes. Eur. J. Inorg. Chem. 2008, 2008, 2713–2720. [Google Scholar] [CrossRef]
- Barros-García, F.; Bernalte-García, A.; Luna-Giles, F.; Maldonado-Rogado, M.; Viñuelas-Zahínos, E. Preparation, characterization and X-ray structure determinations of the 2-acetyl-2-thiazoline schiff base of hydrazine (ATH) and its cadmium (II) complex [Cd(NO3)2(ATH)2]. Polyhedron 2005, 24, 1125–1132. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT: Integrating space group determination and structure solution. Acta Crystallogr. 2014, 70, C1437. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer17; University of Western Australia: Perth, Australia, 2017; Volume 108, 76730p. [Google Scholar]
- Clinical and Laboratory Standards Institute. Twentieth Informational Supplement. M100-S22; CLSI: Wayne, PA, USA, 2012. [Google Scholar]
- Yen, G.C.; Duh, P.D. Scavenging effect of methanolic extracts of peanut hulls on free radical and active oxygen species. J. Agric. Food Chem. 1994, 42, 629–632. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]



| CCDC | 2179746 |
|---|---|
| Empirical formula | C20H28Cl6Cu3N12S4 |
| Formula weight | 968.10 g/mol |
| Temperature/K | 296(2) K |
| Wavelength | 1.54178 Å |
| Crystal system | Triclinic |
| Space group | P-1 |
| a/Å | 8.1686(3) |
| b/Å | 9.4973(4) |
| c/Å | 12.7501(5) |
| α/° | 81.796(2) |
| β/° | 75.291(2) |
| γ/° | 65.096(2) |
| Volume | 867.05(6) Å3 |
| Z | 1 |
| Density (calculated) | 1.854 g/cm3 |
| Absorption coefficient | 8.952 mm−1 |
| F(000) | 485 |
| Crystal size | 0.07 × 0.11 × 0.17 mm3 |
| Theta range for data collection | 3.59 to 66.53° |
| Index ranges | −9 ≤ h ≤ 9, −11 ≤ k ≤ 11, −15 ≤ l ≤ 15 |
| Reflections collected | 17364 |
| Independent reflections | 3055 [R(int) = 0.0539] |
| Completeness to theta = 66.67° | 99.70% |
| Absorption correction | Multiscan |
| Max. and min. transmission | 0.5730 and 0.3110 |
| Refinement method | Full-matrix least-squares on F2 |
| Data/restraints/parameters | 3055/0/213 |
| Goodness-of-fit on F2 | 1.067 |
| Final R indices [I > 2sigma(I)] | R1 = 0.0412, wR2 = 0.1123 |
| R indices (all data) | R1 = 0.0442, wR2 = 0.1164 |
| Largest diff. peak and hole | 1.030 and −0.556 |
| Bond | Distance | Bond | Distance |
| Cu1-Cl1 | 2.2633(10) | Cu2-Cl2 | 2.3140(13) |
| Cu1-Cl2 | 2.8810(11) | Cu2-Cl3 | 2.2720(12) |
| Cu1-N2 | 2.041(2) | Cu2-N1 | 2.024(3) |
| Cu2-Cl1 | 2.6178(11) | Cu2-N4 | 1.987(3) |
| Bonds | Angle | Bonds | Angle |
| Cl1-Cu1-Cl2 | 85.63(3) | Cl1-Cu2-Cl2 | 91.05(4) |
| Cl1-Cu1-Cl2 # | 94.37(3) | Cl1-Cu2-N4 | 105.55(9) |
| N2 -Cu1-Cl1 # | 91.33(9) | Cl2-Cu2-N4 | 89.72(10) |
| Cl1-Cu1-N2 | 88.67(9) | N1 -Cu2-N4 | 166.56(13) |
| N2 -Cu1-Cl2 # | 96.87(9) | Cl2-Cu2-Cl3 | 171.35(5) |
| Cl1-Cu2-Cl3 | 97.12(4) | Cl3-Cu2-N1 | 90.63(9) |
| Cl2-Cu1-Cl2 # | 180 | Cl1-Cu2-N1 | 87.53(9) |
| Cl1-Cu1-Cl1 # | 180 | Cl2-Cu2-N1 | 86.88(9) |
| Cl2-Cu1-N2 | 83.13(9) | Cl3-Cu2-N4 | 90.82(9) |
| N2 -Cu1-N2 # | 180 |
| D-H...A | d(D-H) | d(H...A) | d(D...A) | <(DHA) | Symm. Code |
|---|---|---|---|---|---|
| C1-H1A...Cl1 | 0.97 | 2.72 | 3.427(5) | 130 | |
| C6-H6A...Cl1 | 0.97 | 2.74 | 3.609(5) | 150 | −x,−y,1 − z |
| C7-H7B...Cl1 | 0.97 | 2.81 | 3.692(5) | 150 | 1 + x, −1 + y, z |
| Microbe | [Cu3(La)4(Cl)6] | Control |
|---|---|---|
| A. fungimatus | 15 | 17 a |
| C. albicans | 20 | 20 a |
| S. aureus | 14 | 24 b |
| B. subtilis | 30 | 26 b |
| E. coli | 18 | 30 b |
| P. vulgaris | 13 | 25 b |
| Microbe | [Cu3(La)4(Cl)6] | Control |
|---|---|---|
| A. fumigatus | 1250 | 125.25 a |
| C. albicans | 312.5 | 312.5 a |
| S. aureus | 625 | 9.7 b |
| B. subtilis | 9.7 | 4.8 b |
| E. coli | 312.5 | 4.8 b |
| P. vulgaris | 1250 | 4.8 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Altowyan, M.S.; Khalil, S.M.S.M.; Al-Wahaib, D.; Barakat, A.; Soliman, S.M.; Ali, A.E.; Elbadawy, H.A. Synthesis of a Novel Unexpected Cu(II)–Thiazolidine Complex—X-ray Structure, Hirshfeld Surface Analysis, and Biological Studies. Molecules 2022, 27, 4583. https://doi.org/10.3390/molecules27144583
Altowyan MS, Khalil SMSM, Al-Wahaib D, Barakat A, Soliman SM, Ali AE, Elbadawy HA. Synthesis of a Novel Unexpected Cu(II)–Thiazolidine Complex—X-ray Structure, Hirshfeld Surface Analysis, and Biological Studies. Molecules. 2022; 27(14):4583. https://doi.org/10.3390/molecules27144583
Chicago/Turabian StyleAltowyan, Mezna Saleh, Samar M. S. M. Khalil, Dhuha Al-Wahaib, Assem Barakat, Saied M. Soliman, Ali Eldissouky Ali, and Hemmat A. Elbadawy. 2022. "Synthesis of a Novel Unexpected Cu(II)–Thiazolidine Complex—X-ray Structure, Hirshfeld Surface Analysis, and Biological Studies" Molecules 27, no. 14: 4583. https://doi.org/10.3390/molecules27144583
APA StyleAltowyan, M. S., Khalil, S. M. S. M., Al-Wahaib, D., Barakat, A., Soliman, S. M., Ali, A. E., & Elbadawy, H. A. (2022). Synthesis of a Novel Unexpected Cu(II)–Thiazolidine Complex—X-ray Structure, Hirshfeld Surface Analysis, and Biological Studies. Molecules, 27(14), 4583. https://doi.org/10.3390/molecules27144583









