Phytochemical Investigation of Cordia africana Lam. Stem Bark: Molecular Simulation Approach
Abstract
1. Introduction
2. Results
2.1. Phytochemical Analysis
2.1.1. Total Phenolic Acid and Flavonoid Content
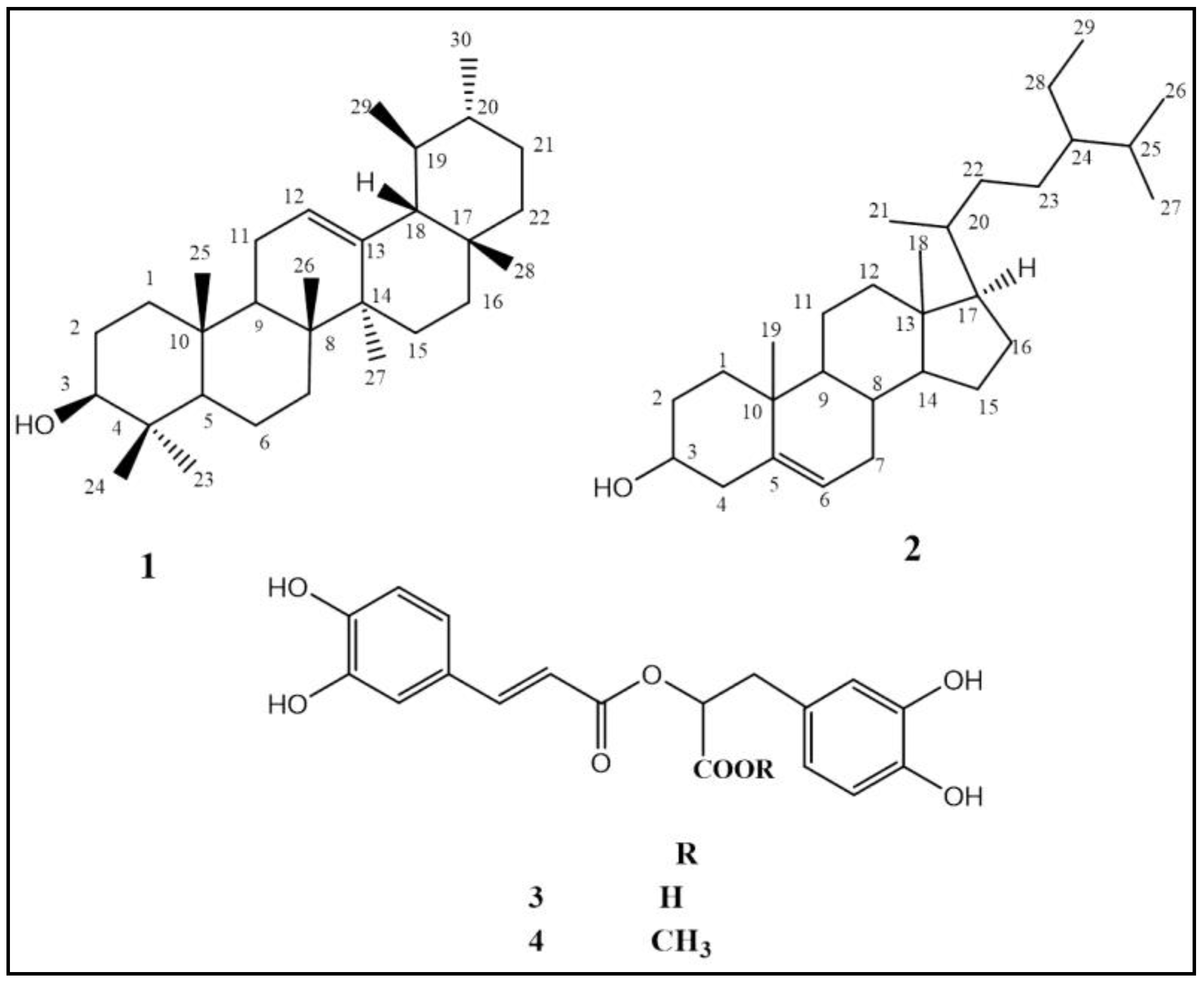
2.1.2. Characterization of Isolated Compounds
2.1.3. Identification of Compounds
2.1.4. HPLC Analysis
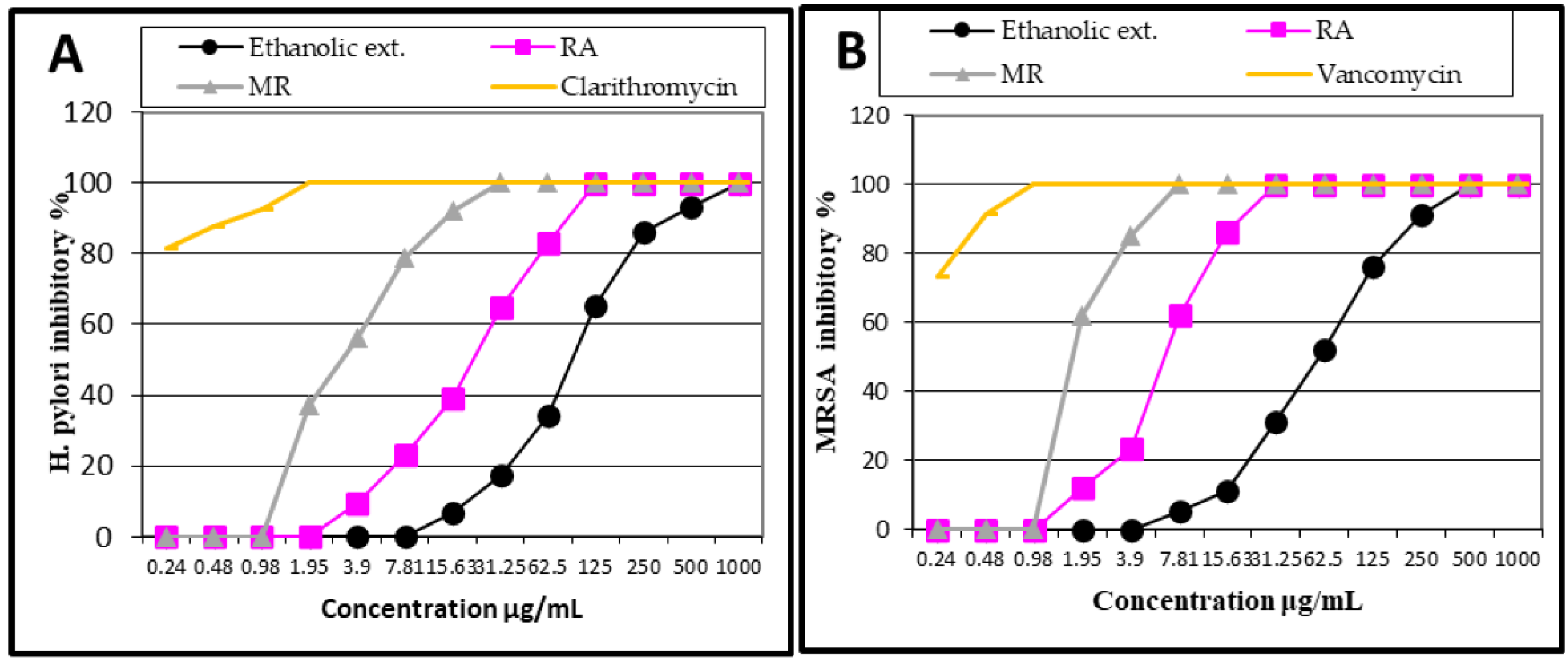
2.2. Antibacterial Activity
2.3. Molecular Docking Analysis
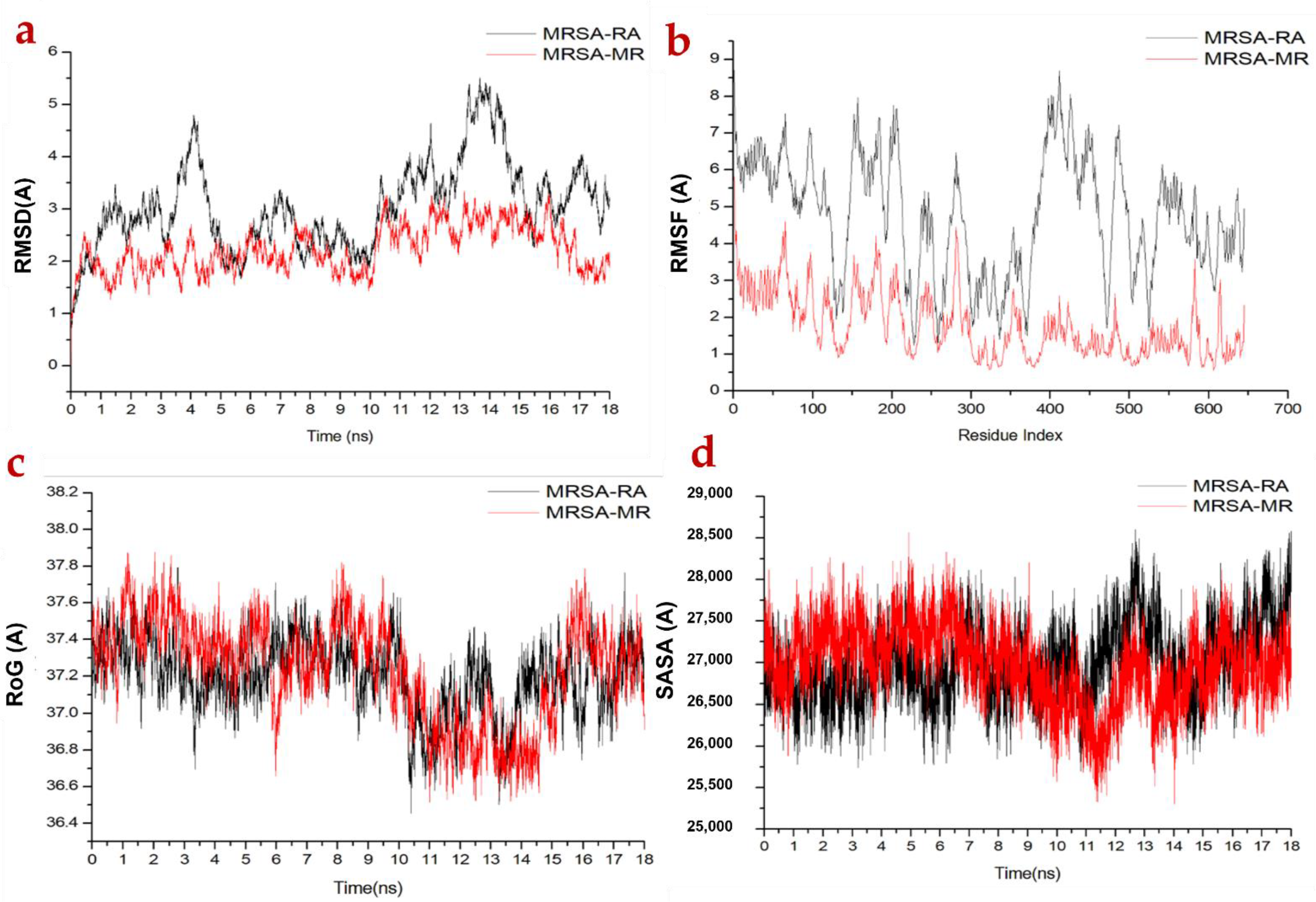
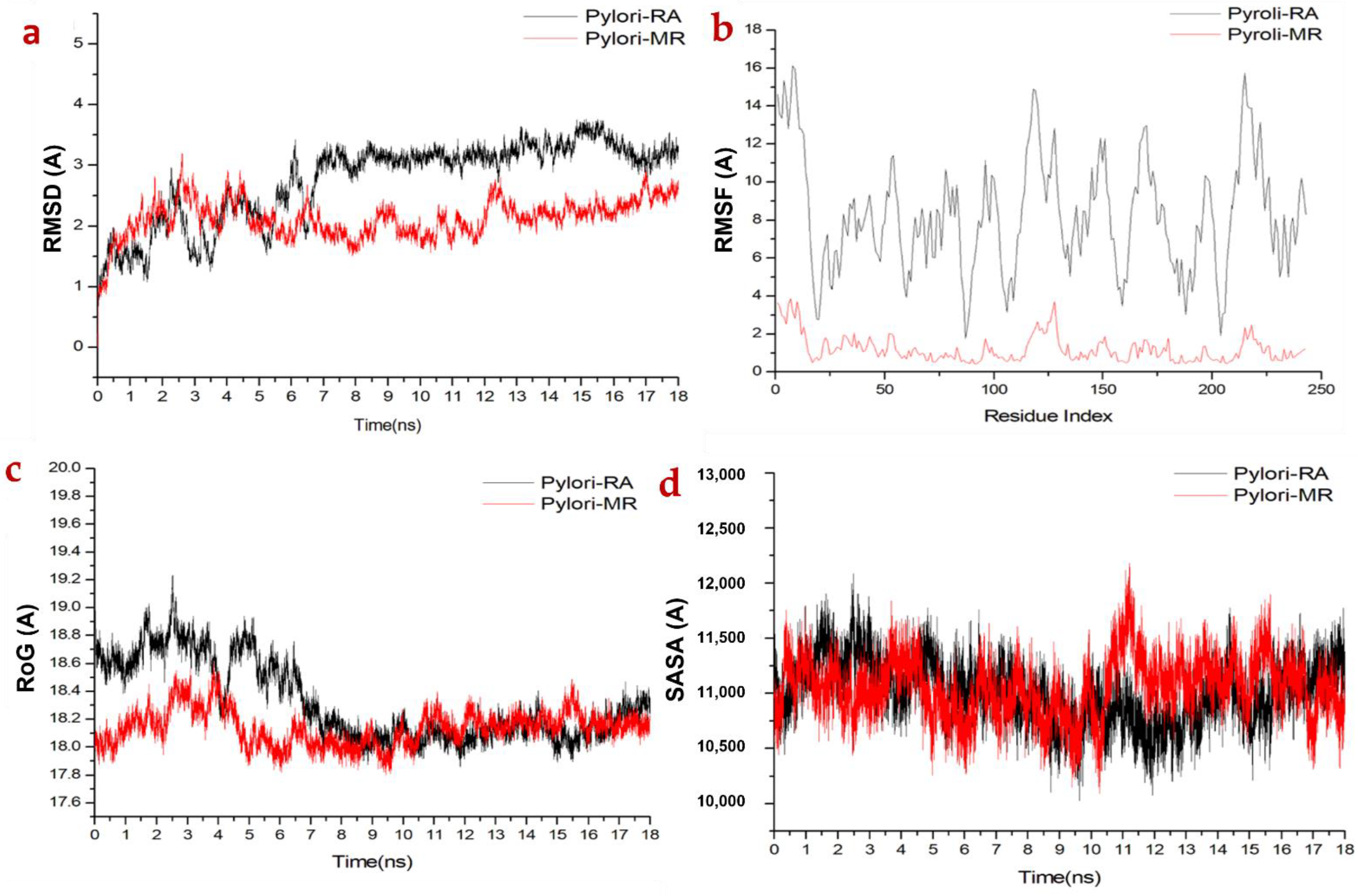
2.3.1. Molecular Dynamic and System Stability
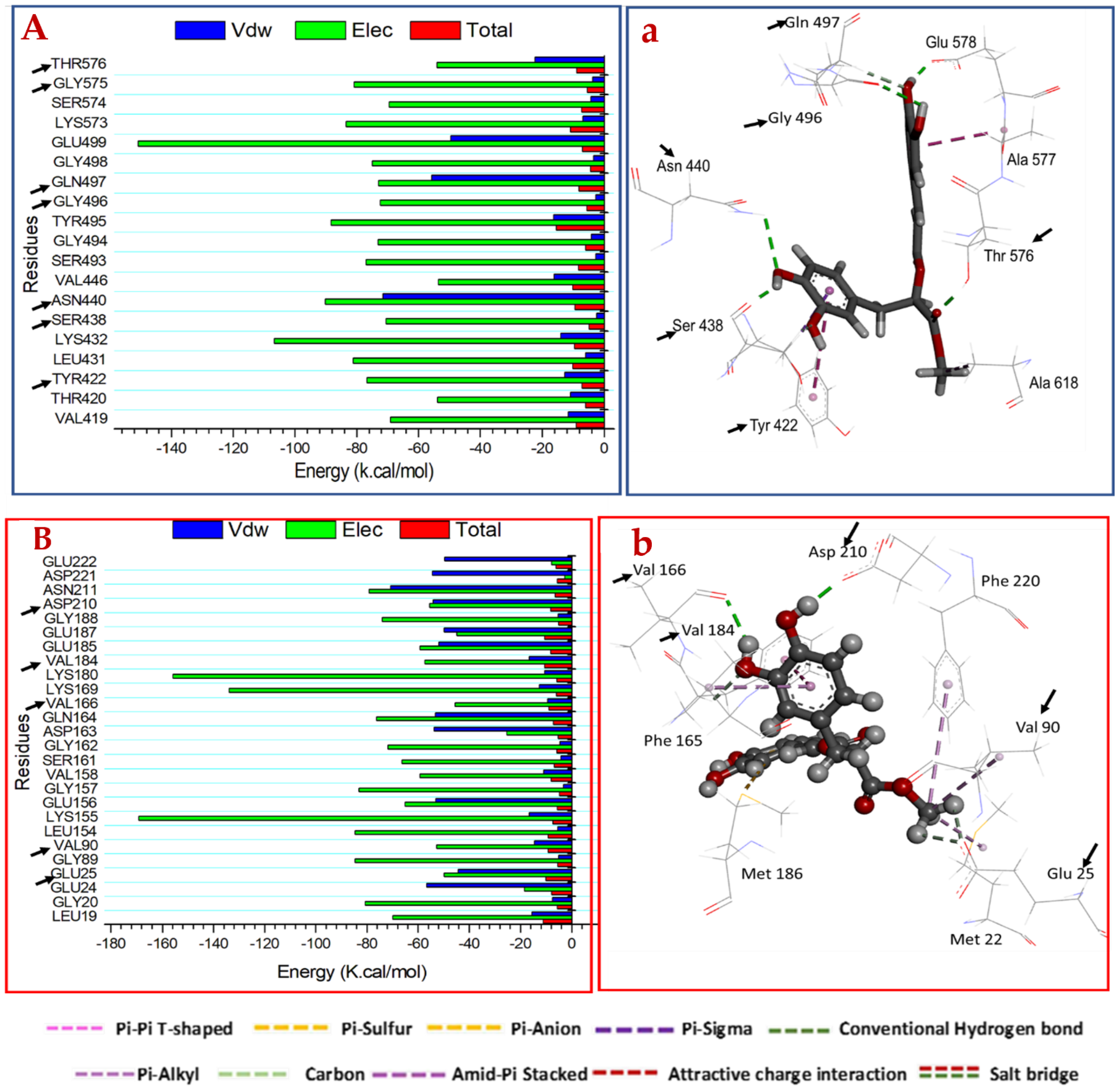
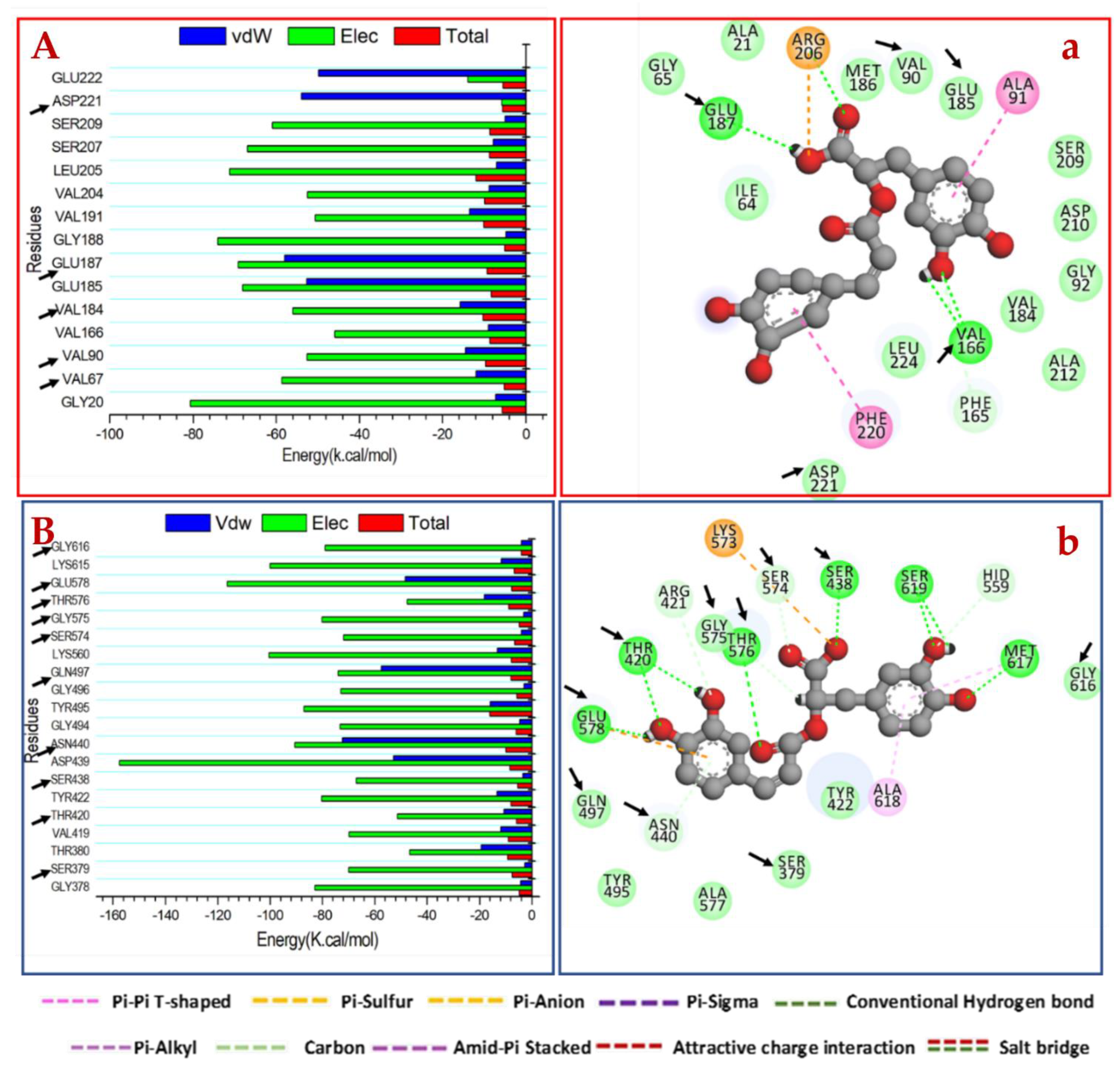
2.3.2. Mechanism of Binding Interactions Based on Binding Free Energy Calculation
2.3.3. Identification of the Critical Residues Responsible for Inhibitor Binding
3. Discussion
4. Materials and Methods
4.1. Plant Material and Extraction
4.2. Chemicals and Reagents
4.3. Determination of Total Phenolic Acid and Flavonoid Contents
4.4. Phytochemical Isolation of Compounds
4.5. HPLC Profiling
4.5.1. Sample Preparation
4.5.2. Standard Solution
4.5.3. Analytical Condition
4.6. Determination of Antibacterial Activities
4.7. Molecular Docking Analysis
4.7.1. System Preparation
4.7.2. Molecular Dynamic (MD) Simulations
4.7.3. Post-MD Analysis
4.7.4. Thermodynamic Calculation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Yirgu, A.; Mohammed, K.; Geldenhuys, C.J. Useful medicinal tree species of Ethiopia: Comprehensive review. S. Afr. J. Bot. 2019, 122, 291–300. [Google Scholar] [CrossRef]
- Giday, M.; Teklehaymanot, T.; Animut, A.; Mekonnen, Y. Medicinal plants of the Shinasha, Agew-awi and Amhara peoples in northwest Ethiopia. J. Ethnopharmacol. 2007, 110, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Alemayehu, G.; Asfaw, Z.; Kelbessa, E. Cordia africana (Boraginaceae) in Ethiopia: A review on its taxonomy, distribution, ethnobotany and conservation status. Int. J. Bot. Stud. 2016, 1, 38–46. [Google Scholar]
- Tijjani, R.G.; Umar, M.L.; Hussaini, I.M.; Shafiu, R. Anti-nociceptive activities of the ethanolic stem bark extract of Cordia africana (Boraginaceae) in rats and mice. Ann. Biol. Sci. 2016, 4, 6–12. [Google Scholar]
- Gordon, R.J.; Lowy, F.D. Pathogenesis of Methicillin-Resistant Staphylococcus aureus Infection. Clin. Infect. Dis. 2008, 46, S350–S359. [Google Scholar] [CrossRef]
- Crawford, S.E.; David, M.Z.; Glikman, D.; King, K.J.; Boyle-Vavra, S.; Daum, R.S. Clinical importance of purulence in methicillin-resistant Staphylococcus aureus skin and soft tissue infections. J. Am. Board Fam. Med. 2009, 22, 647–654. [Google Scholar] [CrossRef]
- Smith, S.; Fowora, M.; Pellicano, R. Infections with Helicobacter pylori and challenges encountered in Africa. World J. Gastroenterol. 2019, 25, 3183–3195. [Google Scholar] [CrossRef]
- Dagnachew, Y.; Asteraye, Y.; Mensigitu, T.; Anza, M. Extraction and Physico-chemical Characterization of Cordia africana Lam Seed Oil. J. Adv. Bot. Zool. 2015, 3, 1–5. [Google Scholar]
- Sarah, T.-B. Ferric reducing antioxidant power and total phenols in Cordia africana fruit. Afr. J. Biochem. Res. 2013, 7, 215–224. [Google Scholar] [CrossRef]
- Mohamed, M.A.A. Phytochemical and Biological Studies of Cordia africana Family Boraginaceae Cultivated in Egypt. CU Theses 2016, 1–29. [Google Scholar]
- Isa, A.I.; Saleh, M.I.A.; Abubakar, A.; Dzoyem, J.P.; Adebayo, S.A.; Musa, I.; Sani, U.F.; Daru, P.A. Evaluation of anti-inflammatory, antibacterial and cytotoxic activities of Cordia africana leaf and stem bark extracts. Bayero J. Pure Appl. Sci. 2016, 9, 228. [Google Scholar] [CrossRef]
- Alhadi, E.A.; Khalid, H.S.; Alhassan, M.S.; Ali, A.A.; Babiker, S.G.; Alabdeen, E.M.Z.; Kabbashi, A.S. Antioxidant and cytotoxicity activity of Cordia africana in Sudan. J. Med. Plant Res. 2015, 3, 29–32. [Google Scholar]
- Mekonnen, A.; Degu, Y.; Carlson, R. Appraisal of solvent system effect on bioactivity profiling of Cordia africana stem bark extracts. Chem. Int. 2020, 6, 1–10. [Google Scholar]
- Eseonu, K.C.; Middleton, S.D.; Eseonu, C.C. A retrospective study of risk factors for poor outcomes in methicillin-resistant Staphylococcus aureus (MRSA) infection in surgical patients. J. Orthop. Surg. Res. 2011, 6, 25. [Google Scholar] [CrossRef]
- Triboulet, S.; Dubée, V.; Lecoq, L.; Bougault, C.; Mainardi, J.L.; Rice, L.B.; Ethève-Quelquejeu, M.; Gutmann, L.; Marie, A.; Dubost, L.; et al. Kinetic Features of L,D-Transpeptidase Inactivation Critical for β-Lactam Antibacterial Activity. PLoS ONE 2013, 8, e67831. [Google Scholar] [CrossRef]
- Foster, T.J. Can β-Lactam Antibiotics Be Resurrected to Combat MRSA? Trends Microbiol. 2019, 27, 26–38. [Google Scholar] [CrossRef]
- Morrisette, T.; Alosaimy, S.; Abdul-mutakabbir, J.C.; Kebriaei, R.; Rybak, M.J. The Evolving Reduction of Vancomycin and Daptomycin Susceptibility in MRSA—Salvaging the Gold Standards with Combination Therapy. Antibiotics 2020, 9, 762. [Google Scholar] [CrossRef]
- Montecucco, C.; Rappuoli, R. Living dangerously: How Helicobacter pylori survives in the human stomach. Nat. Rev. Mol. Cell Biol. 2001, 2, 457–466. [Google Scholar] [CrossRef]
- Uemura, N.; Okamoto, S.; Yamamoto, S.; Matsumura, N.; Yamaguchi, S.; Yamakido, M.; Taniyama, K.; Sasaki, N.; Schlemper, R.J. Helicobacter pylori Infection and the Development of Gastric Cancer. N. Engl. J. Med. 2001, 345, 784–789. [Google Scholar] [CrossRef]
- Shamsdin, S.A.; Alborzi, A.; Ghaderi, A.; Lankrani, K.B.; Pouladfar, G. reza Significance of TC9 and TH9 in Helicobacter pylori-induced gastritis. Helicobacter 2020, 25, e12672. [Google Scholar] [CrossRef]
- Nagata, M.; Toyonaga, K.; Ishikawa, E.; Haji, S.; Okahashi, N.; Takahashi, M.; Izumi, Y.; Imamura, A.; Takato, K.; Hideharu, I.; et al. Helicobacter pylori metabolites exacerbate gastritis through C-type lectin receptors. J. Exp. Med. 2021, 218, e20200815. [Google Scholar] [CrossRef] [PubMed]
- Eloff, J.N.; Famakin, J.O.; Katerere, D.R.P. Isolation of an antibacterial stilbene from Combretum woodii (Combretaceae) leaves. Afr. J. Biotechnol. 2005, 4, 1167–1171. [Google Scholar]
- Valenzuela-Valderrama, M.; Cerda-Opazo, P.; Backert, S.; González, M.F.; Carrasco-Véliz, N.; Jorquera-Cordero, C.; Wehinger, S.; Canales, J.; Bravo, D.; Quest, A.F.G. The Helicobacter pylori urease virulence factor is required for the induction of hypoxia-induced factor-1α in gastric cells. Cancers 2019, 11, 799. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M. High antibiotic resistance rate: A difficult issue for Helicobacter pylori eradication treatment. World J. Gastroenterol. 2015, 21, 13432–13437. [Google Scholar] [CrossRef]
- Malfertheiner, P.; Megraud, F.; O’Morain, C.; Bazzoli, F.; El-Omar, E.; Graham, D.; Hunt, R.; Rokkas, T.; Vakil, N.; Kuipers, E.J.; et al. Current concepts in the management of Helicobacter pylori infection: The maastricht III consensus report. Gut 2007, 56, 772–781. [Google Scholar] [CrossRef]
- Ahn, Y.J.; Park, S.J.; Lee, S.G.; Shin, S.C.; Choi, D.H. Cordycepin: Selective growth inhibitor derived from liquid culture of Cordyceps militaris against Clostridium spp. J. Agric. Food Chem. 2000, 48, 2744–2748. [Google Scholar] [CrossRef]
- Bae, E.A.; Han, M.J.; Kim, D.H. In vitro anti-Helicobacter pylori activity of some flavonoids and their metabolites. Planta Med. 1999, 65, 442–443. [Google Scholar] [CrossRef]
- Li, Y.; Xu, C.; Zhang, Q.; Liu, J.Y.; Tan, R.X. In vitro anti-Helicobacter pylori action of 30 Chinese herbal medicines used to treat ulcer diseases. J. Ethnopharmacol. 2005, 98, 329–333. [Google Scholar] [CrossRef]
- Ustün, O.; Ozçelik, B.; Akyön, Y.; Abbasoglu, U.; Yesilada, E. Flavonoids with anti-Helicobacter pylori activity from Cistus laurifolius leaves. J. Ethnopharmacol. 2006, 108, 457–461. [Google Scholar] [CrossRef]
- Geller, F.; Schmidt, C.; Göttert, M.; Fronza, M.; Schattel, V.; Heinzmann, B.; Werz, O.; Flores, E.M.M.; Merfort, I.; Laufer, S. Identification of rosmarinic acid as the major active constituent in Cordia americana. J. Ethnopharmacol. 2010, 128, 561–566. [Google Scholar] [CrossRef]
- Al-Musayeib, N.; Perveen, S.; Fatima, I.; Nasir, M.; Hussain, A. Antioxidant, anti-glycation and anti-inflammatory activities of phenolic constituents from cordia sinensis. Molecules 2011, 16, 10214–10226. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Lawania, R.D.; Mishra, A.; Gupta, R. Role of Cordia dichotoma seeds and leaves extract in degenerative disorders. Int. J. Pharm. Sci. Rev. Res. 2010, 2, 21–24. [Google Scholar]
- Nyigo, V.A.; Peter, X.; Mabiki, F.; Malebo, H.M.; Mdegela, R.H.; Fouche, G.E.; Vitus, M.; Nyigo, A.; Abiki, F.M.; Alebo, H.M.M.; et al. Isolation and Identification of Euphol and β-Sitosterol from the Dichloromethane Extracts of Synadenium Glaucescens; Sokoine University of Agriculture: Morogoro, Tanzania, 2016; Volume 5. [Google Scholar]
- Lu, Y.; Foo, L.Y. Rosmarinic acid derivatives from Salvia officinalis. Phytochemistry 1999, 51, 91–94. [Google Scholar] [CrossRef]
- Machaba, K.E.; Mhlongo, N.N.; Soliman, M.E.S. Induced Mutation Proves a Potential Target for TB Therapy: A Molecular Dynamics Study on LprG. Cell Biochem. Biophys. 2018, 76, 345–356. [Google Scholar] [CrossRef]
- Pan, L.; Patterson, J.C. Molecular Dynamics Study of Zn(Aβ) and Zn(Aβ)2. PLoS ONE 2013, 8, e70681. [Google Scholar] [CrossRef]
- Wijffels, G.; Dalrymple, B.; Kongsuwan, K.; Dixon, N.E. Conservation of eubacterial replicases. IUBMB Life 2005, 57, 413–419. [Google Scholar] [CrossRef]
- Richmond, T.J. Solvent accessible surface area and excluded volume in proteins. Analytical equations for overlapping spheres and implications for the hydrophobic effect. J. Mol. Biol. 1984, 178, 63–89. [Google Scholar] [CrossRef]
- Salehi, B.; Sharopov, F.; Martorell, M.; Rajkovic, J.; Ademiluyi, A.O.; Sharifi-Rad, M.; Fokou, P.V.T.; Martins, N.; Iriti, M.; Sharifi-Rad, J. Phytochemicals in Helicobacter pylori infections: What are we doing now? Int. J. Mol. Sci. 2018, 19, 2361. [Google Scholar] [CrossRef]
- Kocsmár, É.; Buzás, G.M.; Szirtes, I.; Kocsmár, I.; Kramer, Z.; Szijártó, A.; Fadgyas-Freyler, P.; Szénás, K.; Rugge, M.; Fassan, M.; et al. Primary and secondary clarithromycin resistance in Helicobacter pylori and mathematical modeling of the role of macrolides. Nat. Commun. 2021, 12, 2255. [Google Scholar] [CrossRef]
- Xin, L.Y.; Min, T.H.; Zin, P.N.L.M.; Pulingam, T.; Appaturi, J.N.; Parumasivam, T. Antibacterial potential of Malaysian ethnomedicinal plants against methicillin-susceptible Staphylococcus aureus (MSSA) and methicillin-resistant Staphylococcus aureus (MRSA). Saudi J. Biol. Sci. 2021, 28, 5884–5889. [Google Scholar] [CrossRef]
- Kuete, V. Potential of Cameroonian plants and derived products against microbial infections: A review. Planta Med. 2010, 76, 1479–1491. [Google Scholar] [CrossRef] [PubMed]
- Cos, P.; Maes, L.; Sindambiwe, J.-B.; Vlietinck, A.J.; Berghe, V.D. Bioassays for Antibacterial and Antifungal Activities. In Biological Screening of Plant Constituents: Training Manual; UNIDO-ICS (United Nations Industrial Development Organization and the International Centre for Science and High Technology): Trieste, Italy, 2006. [Google Scholar]
- Mabeku, L.B.K.; Bille, B.E.; Tchouangueu, T.F.; Nguepi, E.; Leundji, H. Treatment of Helicobacter pylori infected mice with bryophyllum pinnatum, a medicinal plant with antioxidant and antimicrobial properties, reduces bacterial load. Pharm. Biol. 2017, 55, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Ríos, J.L.; Recio, M.C. Medicinal plants and antimicrobial activity. J. Ethnopharmacol. 2005, 100, 80–84. [Google Scholar] [CrossRef]
- Livermore, D.M. Antibiotic resistance in staphylococci. Int. J. Antimicrob. Agents 2000, 16, 3–10. [Google Scholar] [CrossRef]
- Lim, D.; Strynadka, N.C.J. Structural basis for the β-lactam resistance of PBP2a from methicillin-resistant Staphylococcus aureus. Nat. Struct. Biol. 2002, 9, 870–876. [Google Scholar] [CrossRef] [PubMed]
- Kȩpa, M.; Miklasińska-Majdanik, M.; Wojtyczka, R.D.; Idzik, D.; Korzeniowski, K.; Smoleń-Dzirba, J.; Wasik, T.J. Antimicrobial potential of caffeic acid against Staphylococcus aureus clinical strains. BioMed Res. Int. 2018, 2018, 7413504. [Google Scholar] [CrossRef]
- Ramos, F.A.; Takaishi, Y.; Shirotori, M.; Kawaguchi, Y.; Tsuchiya, K.; Shibata, H.; Higuti, T.; Tadokoro, T.; Takeuchi, M. Antibacterial and antioxidant activities of quercetin oxidation products from yellow onion (Allium cepa) skin. J. Agric. Food Chem. 2006, 54, 3551–3557. [Google Scholar] [CrossRef]
- Bylka, W.; Matlawska, I.; Pilewski, N. Natural flavonoids as antimicrobial agents. Jana 2004, 7, 9–16. [Google Scholar]
- Hoskeri, J.H.; Krishna, V.; Jignesh, S.; Sanjay, S.T.; Roshan, A.; Vijay, S. In-silico drug designing using B-sitosterol isolated from flaveria trinervia against peptide deformylase protein to hypothesize bactericidal effect. Int. J. Pharm. Pharm. Sci. 2012, 4, 192–196. [Google Scholar]
- Saboo, S.; Tapadiya, R.; Khadabadi, S.S.; Deokate, U.A. In vitro antioxidant activity and total phenolic, flavonoid contents of the crude extracts of Pterospermum acerifolium wild leaves (Sterculiaceae). J. Chem. Pharm. Res. 2010, 2, 417–423. [Google Scholar]
- Ibrahim, M. Comparison of total flavanoid content of Azadirachta indica root bark extracts prepared by different methods of extraction. Res. J. Pharm. Biol. Chem. Sci. 2011, 2, 254–261. [Google Scholar]
- Krieger, S.; Sonja, K.; Schneider, S.; Krieger, S. Quality Analysis of Extra Virgin Olive Oils–Part 7: Nutritive Benefits–Determination of Phenolic Compounds in Virgin Olive Oil Using the Agilent 1290 Infinity 2D-LC Solution; Agilent Technologies: Santa Clara, CA, USA, 2014; pp. 1–10. [Google Scholar]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Clinical and Laboratory Standards Institute: Malvern, PA, USA, 2017; ISBN 1562388045. [Google Scholar]
- Liu, X.; Ouyang, S.; Yu, B.; Liu, Y.; Huang, K.; Gong, J.; Zheng, S.; Li, Z.; Li, H.; Jiang, H. PharmMapper server: A web server for potential drug target identification using pharmacophore mapping approach. Nucleic Acids Res. 2010, 38, W609–W614. [Google Scholar] [CrossRef] [PubMed]
- Ha, N.C.; Oh, S.T.; Sung, J.Y.; Cha, K.A.; Lee, M.H.; Oh, B.H. Supramolecular assembly and acid resistance of Helicobacter pylori urease. Nat. Struct. Biol. 2001, 8, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Lovering, A.L.; Gretes, M.C.; Safadi, S.S.; Danel, F.; De Castro, L.; Page, M.G.P.; Strynadka, N.C.J. Structural Insights into the Anti-methicillin-resistant Staphylococcus aureus (MRSA) Activity of Ceftobiprole. J. Biol. Chem. 2012, 287, 32096–32102. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera-A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Hospital, A.; Goñi, J.R.; Orozco, M.; Gelpí, J.L. Molecular dynamics simulations: Advances and applications. Adv. Appl. Bioinform. Chem. 2015, 8, 37–47. [Google Scholar]
- Lee, T.S.; Cerutti, D.S.; Mermelstein, D.; Lin, C.; Legrand, S.; Giese, T.J.; Roitberg, A.; Case, D.A.; Walker, R.C.; York, D.M. GPU-Accelerated Molecular Dynamics and Free Energy Methods in Amber18: Performance Enhancements and New Features. J. Chem. Inf. Model. 2018, 58, 2043–2050. [Google Scholar] [CrossRef]
- Wang, J.; Wang, W.; Kollman, P.A.; Case, D.A. Automatic atom type and bond type perception in molecular mechanical calculations. J. Mol. Graph. Model. 2006, 25, 247–260. [Google Scholar] [CrossRef]
- Berendsen, H.J.C.; Postma, J.P.M.; Van Gunsteren, W.F.; Dinola, A.; Haak, J.R. Molecular dynamics with coupling to an external bath. J. Chem. Phys. 1984, 81, 3684–3690. [Google Scholar] [CrossRef]
- Roe, D.R.; Cheatham, T.E. PTRAJ and CPPTRAJ: Software for processing and analysis of molecular dynamics trajectory data. J. Chem. Theory Comput. 2013, 9, 3084–3095. [Google Scholar] [CrossRef]
- Seifert, E. OriginPro 9.1: Scientific data analysis and graphing software-Software review. J. Chem. Inf. Model. 2014, 54, 1552. [Google Scholar] [CrossRef] [PubMed]
- Genheden, S.; Ryde, U. The MM/PBSA and MM/GBSA methods to estimate ligand-binding affinities. Expert Opin. Drug Discov. 2015, 10, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Drissi, M.; Benhalima, N.; Megrouss, Y.; Rachida, R.; Chouaih, A.; Hamzaoui, F. Theoretical and experimental electrostatic potential around the m-nitrophenol molecule. Molecules 2015, 20, 4042–4054. [Google Scholar] [CrossRef] [PubMed]
- Hou, T.; Wang, J.; Li, Y.; Wang, W. Assessing the performance of the MM/PBSA and MM/GBSA methods. 1. The accuracy of binding free energy calculations based on molecular dynamics simulations. J. Chem. Inf. Model. 2011, 51, 69–82. [Google Scholar] [CrossRef]
- Sitkoff, D.; Sharp, K.A.; Honig, B. Accurate calculation of hydration free energies using macroscopic solvent models. J. Phys. Chem. 1994, 98, 1978–1988. [Google Scholar] [CrossRef]
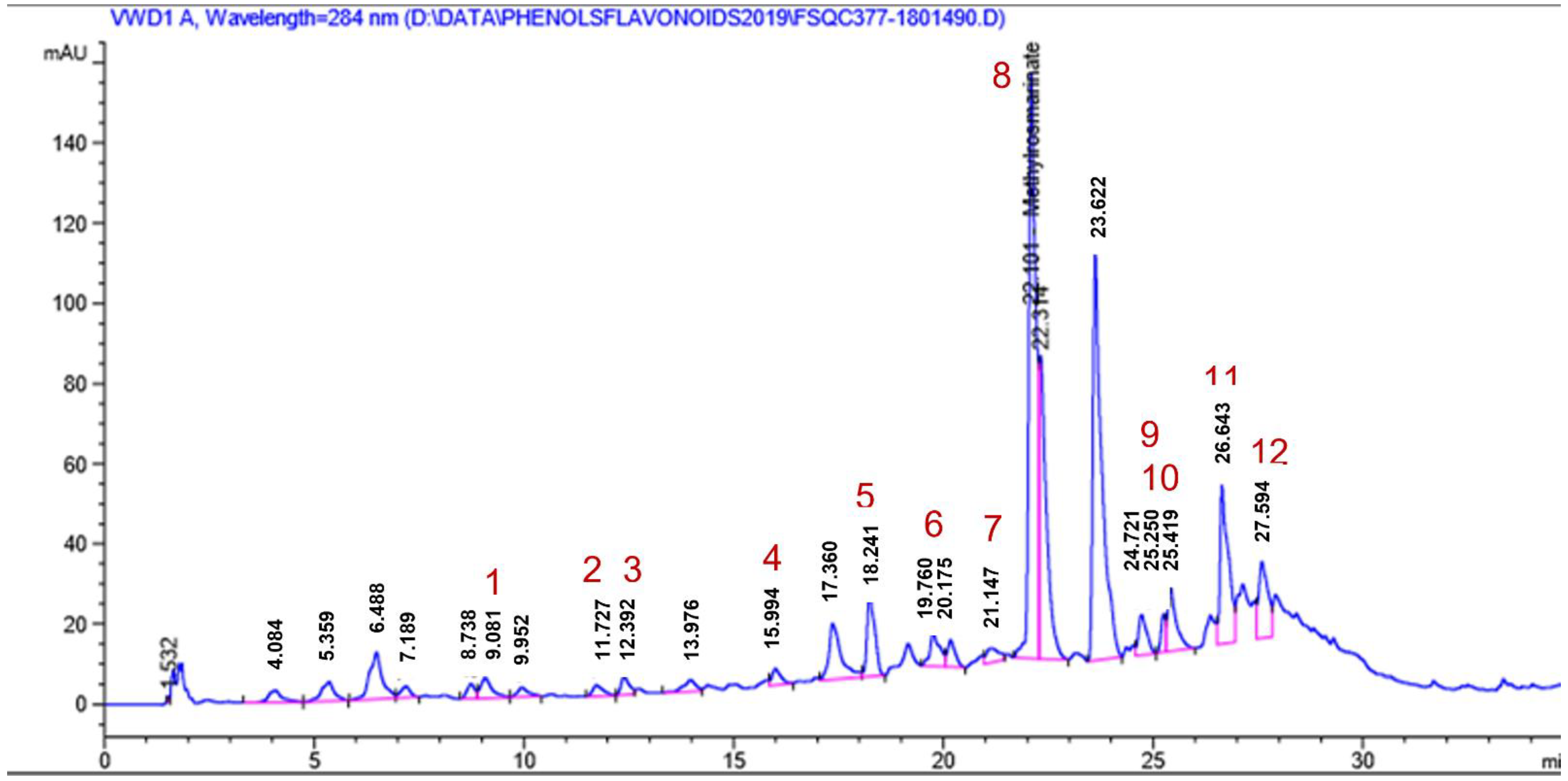
| Peak No. | Identified Phenolic Compounds | Rt | Concentration (ppm) |
|---|---|---|---|
| 1. | p-hydroxy benzoic acid | 9.08 | 124.68 |
| 2. | Caffeic acid | 11.72 | 8.95 |
| 3. | Syringic acid | 12.39 | 22.44 |
| 4. | Ferulic acid | 15.99 | 14.47 |
| 5. | Rutin | 18.24 | 499.63 |
| 6. | o-Coumaric acid | 19.76 | 23.07 |
| 7. | Myricetin | 21.14 | 452.86 |
| 8. | Methyl rosmarinate | 22.10 | 727.66 |
| 9. | Quercetin | 25.25 | 239.75 |
| 10. | Rosmarinic acid | 25.42 | 138.18 |
| 11. | Naringenin | 26.64 | 1971.68 |
| 12. | Kaempferol | 27.59 | 943.38 |
| H. pylori | MRSA | |||
|---|---|---|---|---|
| MIC90 | MIC | MIC90 | MIC | |
| Ethanolic extract | 382.8 ± 0.96 | 1000 ± 0.55 | 238 ± 1.9 | 500 ± 0.8 |
| Rosmarinic acid (RA) | 87.7 ± 0.85 | 125 ± 0.58 | 15.63 ± 0.96 | 31.25 ± 0.6 |
| Methyl rosmarinate (MR) | 14.4 ± 1.7 | 31.25 ± 0.9 | 5.14 ± 2.1 | 7.81 ± 1.7 |
| Vancomycin | - | - | 0.46 ± 1.8 | 0.98 ± 1.6 |
| Clarithromycin | 0.7 ± 1.3 | 1.95 ± 0.8 | - | - |
| Energy Components (Kcal/mol) | ||||||
|---|---|---|---|---|---|---|
| MRSA | ||||||
| Complex | MIC | ΔEvdW | ΔEelec | ΔGgas | ΔGsolv | ΔGbind |
| RA | 31.25 | −27.21 ± 0.22 | −19.97 ± 1.49 | −47.19 ± 1.40 | 35.96 ± 1.04 | −11.22 ± 0.45 |
| MR | 7.81 | −46.03 ± 0.06 | −26.78 ± 0.08 | −72.81 ± 0.09 | 31.35 ± 0.05 | −40.53 ± 0.06 |
| H. pylori | ||||||
| RA | 125 | −27.20 ± 0.10 | −29.12 ± 1.08 | −64.31 ± 1.04 | 33.15 ± 1.05 | −23.36 ± 0.10 |
| MR | 31.25 | −36.97± 0.08 | −43.01 ±0.19 | −79.98 ± 0.14 | 38.44 ± 0.15 | −41.54 ± 0.10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabry, M.M.; El-Fishawy, A.M.; El-Rashedy, A.A.; El Gedaily, R.A. Phytochemical Investigation of Cordia africana Lam. Stem Bark: Molecular Simulation Approach. Molecules 2022, 27, 4039. https://doi.org/10.3390/molecules27134039
Sabry MM, El-Fishawy AM, El-Rashedy AA, El Gedaily RA. Phytochemical Investigation of Cordia africana Lam. Stem Bark: Molecular Simulation Approach. Molecules. 2022; 27(13):4039. https://doi.org/10.3390/molecules27134039
Chicago/Turabian StyleSabry, Manal M., Ahlam M. El-Fishawy, Ahmed A. El-Rashedy, and Rania A. El Gedaily. 2022. "Phytochemical Investigation of Cordia africana Lam. Stem Bark: Molecular Simulation Approach" Molecules 27, no. 13: 4039. https://doi.org/10.3390/molecules27134039
APA StyleSabry, M. M., El-Fishawy, A. M., El-Rashedy, A. A., & El Gedaily, R. A. (2022). Phytochemical Investigation of Cordia africana Lam. Stem Bark: Molecular Simulation Approach. Molecules, 27(13), 4039. https://doi.org/10.3390/molecules27134039






