Improved Fire Retardancy of Cellulose Fibres via Deposition of Nitrogen-Modified Biopolyphenols
Abstract
:1. Introduction
2. Results and Discussion
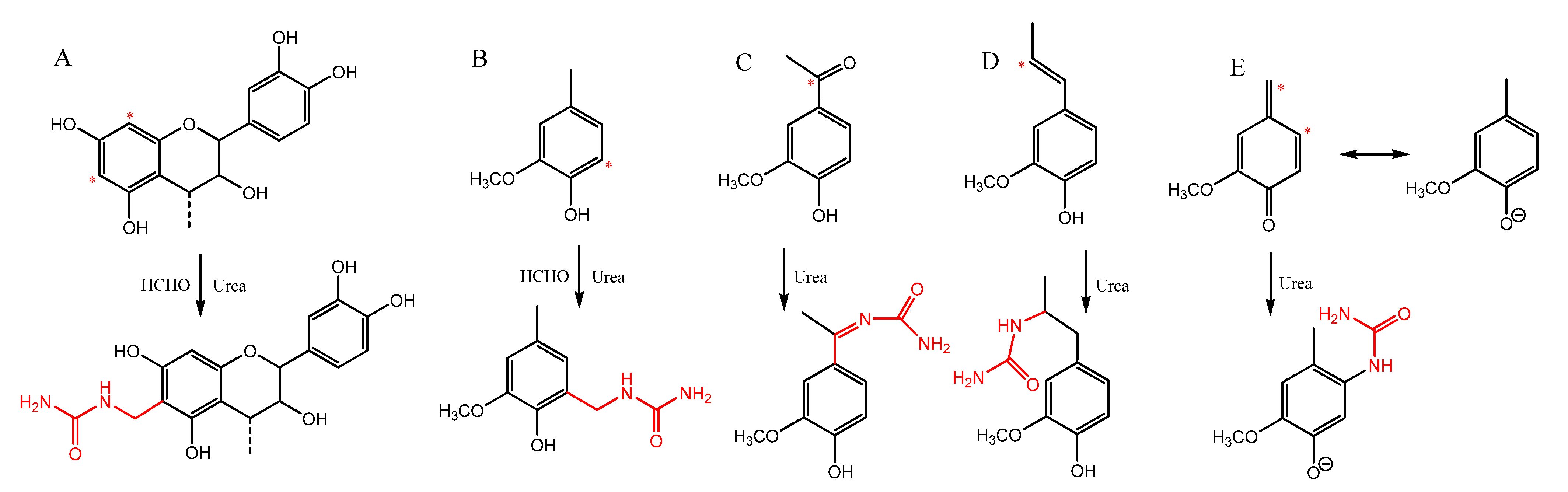
2.1. Nitrogen-Modification of Lignin and Tannin
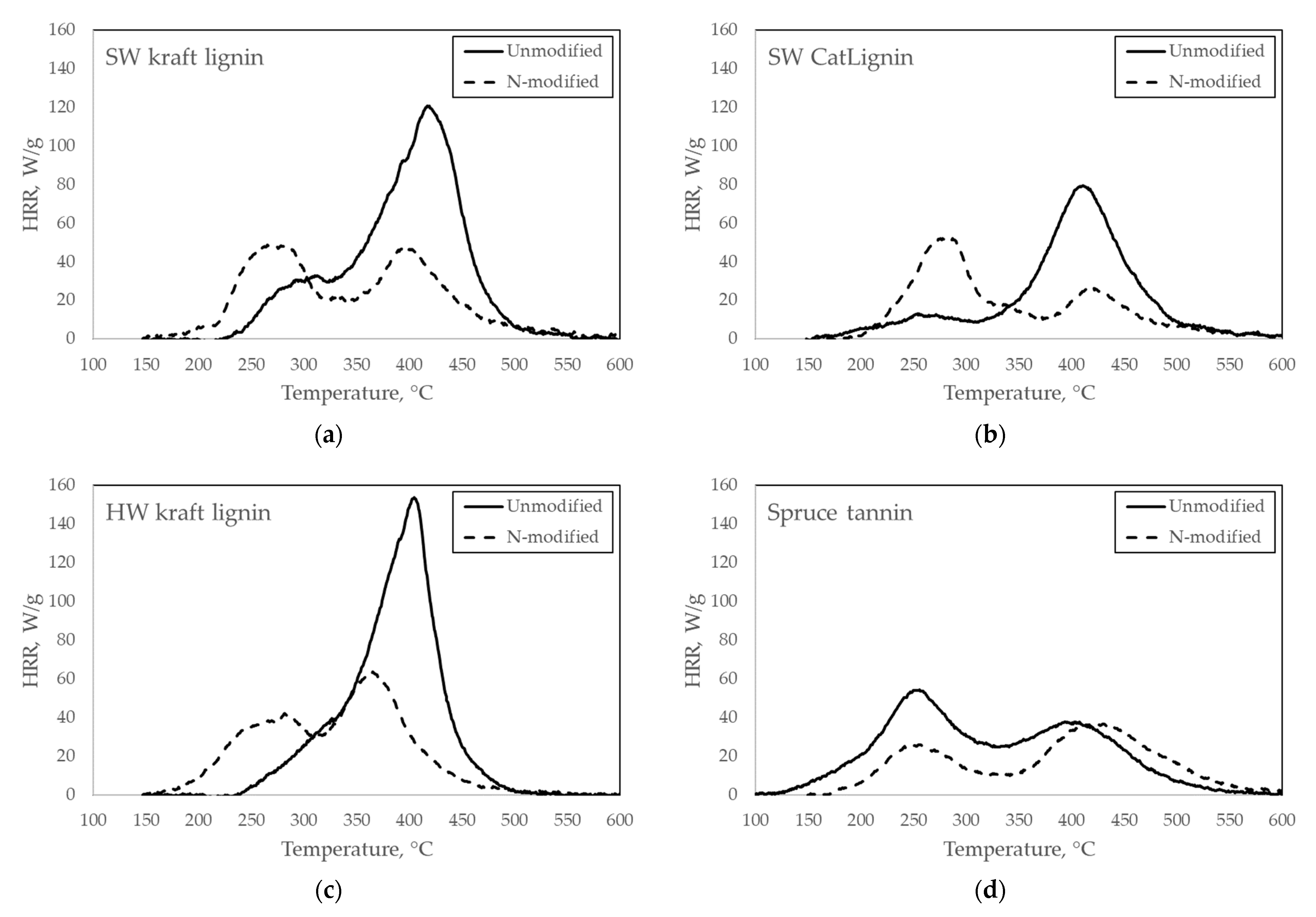
2.2. Heat Release Properties of Unmodified and N-Modified Biopolyphenols
2.3. Deposited Amounts of Biopolyphenols on Cellulose Kraft Pulp Fibres
2.4. Heat Release Properties of Modified Biopolyphenols Deposited on Cellulose Pulp Fibres
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Nitrogen-Modification of Biopolyphenols
3.2.2. Chemical Characterisation of Biopolyphenols
3.2.3. Deposition of Biopolyphenols on Cellulose Fibres
3.2.4. Determination of Pulp Pad Biopolyphenol Content
3.2.5. Scanning Electron Microscopy
3.2.6. Micro-Scale Combustion Calorimetry
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kunič, R. Carbon footprint of thermal insulation materials in building envelopes. Energy Effic. 2017, 10, 1511–1528. [Google Scholar] [CrossRef]
- Orsini, F.; Marrone, P. Approaches for a low-carbon production of building materials: A review. J. Clean. Prod. 2019, 241. [Google Scholar] [CrossRef]
- Tsantaridis, L. Reaction to Fire Performance of Wood and Other Building Products—Cone Calorimeter Results and Analysis. Master’s Thesis, KTH—Royal Institute of Technology, Department of Civil and Architectural Engineering, Division of Building Materials, Stockholm, Sweden, 2003; 65p. [Google Scholar]
- Zhang, R.; Xiao, X.; Tai, Q.; Huang, H.; Hu, Y. Modification of lignin and its application as char agent in intumescent flame-retardant poly(lactic acid). Polym. Eng. Sci. 2012, 52, 2620–2626. [Google Scholar] [CrossRef]
- Zheng, C.; Li, D.; Ek, M. Cellulose-fiber-based insulation materials with improved reaction-to-fire properties. Nord. Pulp Pap. Res. J. 2017, 32, 466–472. [Google Scholar] [CrossRef]
- Hobbs, C.E. Recent advances in bio-based flame retardant additives for synthetic polymeric materials. Polymers 2019, 11, 224. [Google Scholar] [CrossRef] [Green Version]
- Kai, D.; Tan, M.J.; Chee, P.L.; Chua, Y.K.; Yap, Y.L.; Loh, X.J. Towards lignin-based functional materials in a sustainable world. Green Chem. 2016, 18, 1175–1200. [Google Scholar] [CrossRef]
- Basak, S.; Raja, A.S.M.; Saxena, S.; Patil, P.G. Tannin based polyphenolic bio-macromolecules: Creating a new era towards sustainable flame retardancy of polymers. Polym. Degrad. Stab. 2021, 189, 109603. [Google Scholar] [CrossRef]
- Yang, H.; Yu, B.; Xu, X.; Bourbigot, S.; Wang, H.; Song, P. Lignin-derived bio-based flame retardants toward high-performance sustainable polymeric materials. Green Chem. 2020, 22, 2129–2161. [Google Scholar] [CrossRef]
- Widsten, P.; Tamminen, T.; Paajanen, A.; Hakkarainen, T.; Liitiä, T. Modified and unmodified technical lignins as flame retardants for polypropylene. Holzforschung 2021, 75, 584–590. [Google Scholar] [CrossRef]
- Peter, M.G. Chemical Modifications of Biopolymers by Quinones and Quinone Methides. Angew. Chem. Int. Ed. Engl. 1989, 28, 555–570. [Google Scholar] [CrossRef]
- Chatterjee, M.; Ishizaka, T.; Kawanami, H. Reductive amination of furfural to furfurylamine using aqueous ammonia solution and molecular hydrogen: An environmentally friendly approach. Green Chem. 2016, 18, 487–496. [Google Scholar] [CrossRef]
- Handika, S.O.; Lubis, M.A.R.; Sari, R.K.; Laksana, R.P.B.; Antov, P.; Savov, V.; Gajtanska, M.; Iswanto, A.H. Enhancing thermal and mechanical properties of ramie fiber via impregnation by lignin-based polyurethane resin. Materials 2021, 14, 6850. [Google Scholar] [CrossRef]
- Aristri, M.A.; Lubis, M.A.R.; Laksana, R.P.B.; Sari, R.K.; Iswanto, A.H.; Kristak, L.; Antov, P.; Pizzi, A. Thermal and mechanical performance of ramie fibers modified with polyurethane resins derived from acacia mangium bark tannin. J. Mater. Res. Technol. 2022, 18, 2413–2427. [Google Scholar] [CrossRef]
- Hakkarainen, T.; Mikkola, E.; Östman, B.; Tsantaridis, L.; Brumer, H.; Piispanen, P. Innovative Eco-Efficient High Fire Performance Wood Products for Demanding Applications; State-of-the-Art; VTT Technical Research Centre of Finland: Espoo, Finland; SP Trätek: Stockholm, Sweden; KTH Biotechnology: Stockholm, Sweden, 2005. [Google Scholar]
- Forss, K.G.; Fuhrmann, A.G.M.; Toroi, M. Procedure for Manufacturing Lignocellulosic Material Product. European Patent EP 0355041 B1, 7 March 1988. [Google Scholar]
- Giummarella, N.; Pylypchuk, I.V.; Sevastyanova, O.; Lawoko, M. New Structures in Eucalyptus Kraft Lignin with Complex Mechanistic Implications. ACS Sustain. Chem. Eng. 2020, 8, 10983–10994. [Google Scholar] [CrossRef]
- Li, M.; Yoo, C.G.; Pu, Y.; Ragauskas, A.J. 31P NMR Chemical Shifts of Solvents and Products Impurities in Biomass Pretreatments. ACS Sustain. Chem. Eng. 2018, 6, 1265–1270. [Google Scholar] [CrossRef]
- Bianchi, S.; Kroslakova, I.; Janzon, R.; Mayer, I.; Saake, B.; Pichelin, F. Characterization of condensed tannins and carbohydrates in hot water bark extracts of European softwood species. Phytochemistry 2015, 120, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Varila, T.; Brännström, H.; Kilpeläinen, P.; Hellström, J.; Romar, H.; Nurmi, J.; Lassi, U. From Norway spruce bark to carbon foams: Characterization, and applications. BioResources 2020, 15, 3651–3666. [Google Scholar] [CrossRef]
- Wikberg, H.; Ohra-Aho, T.; Leppävuori, J.; Liitiä, T.; Kanerva, H. Method for Producing Reactive Lignin. WO2018115592A1, 28 June 2018. [Google Scholar]
- Decusati, O.G.; Lamb, L.H. Manufacturing Process for Quaternary Ammonium Tannate, a Vegetable Coagulating/Flocculating Agent. European Patent EP 1078884A2, 27 August 1999. [Google Scholar]
- Nordlund, E.; Lille, M.; Silventoinen, P.; Nygren, H.; Seppänen-Laakso, T.; Mikkelson, A.; Aura, A.-M.; Heiniö, R.-L.; Nohynek, L.; Puupponen-Pimiä, R.; et al. Plant cells as food—A concept taking shape. Food Res. Int. 2018, 107, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Granata, A.; Argyropoulos, D.S. 2-Chloro-4,4,5,5-tetramethyl-1,3,2-dioxaphospholane, a Reagent for the Accurate Determination of the Uncondensed and Condensed Phenolic Moieties in Lignins. J. Agric. Food Chem. 1995, 43, 1538–1544. [Google Scholar] [CrossRef]
- Dence, C.W. The Determination of Lignin. In Methods in Lignin Chemistry; Springer: Berlin/Heidelberg, Germany, 1992; pp. 33–61. [Google Scholar]
- Lyon, R.E.; Walters, R.N. Pyrolysis combustion flow calorimetry. J. Anal. Appl. Pyrolysis 2004, 71, 27–46. [Google Scholar] [CrossRef]
- Lyon, R.E.; Walters, R.N.; Stoliarov, S.I.; Safronava, N. Principles and Practice of Microscale Combustion Calorimetry; DOT/FAA/TC-12/53; Federal Aviation Administration: Atlantic City Airport, NJ, USA, 2013; 75p. [Google Scholar]
- ISO 5660-1:2015; Reaction-to-Fire Tests—Heat Release, Smoke Production and Mass Loss Rate—Part 1: Heat Release Rate (Cone Calorimeter Method) and Smoke Production Rate (Dynamic Measurement). International Organization for Standardization: Geneva, Switzerland, 2015.
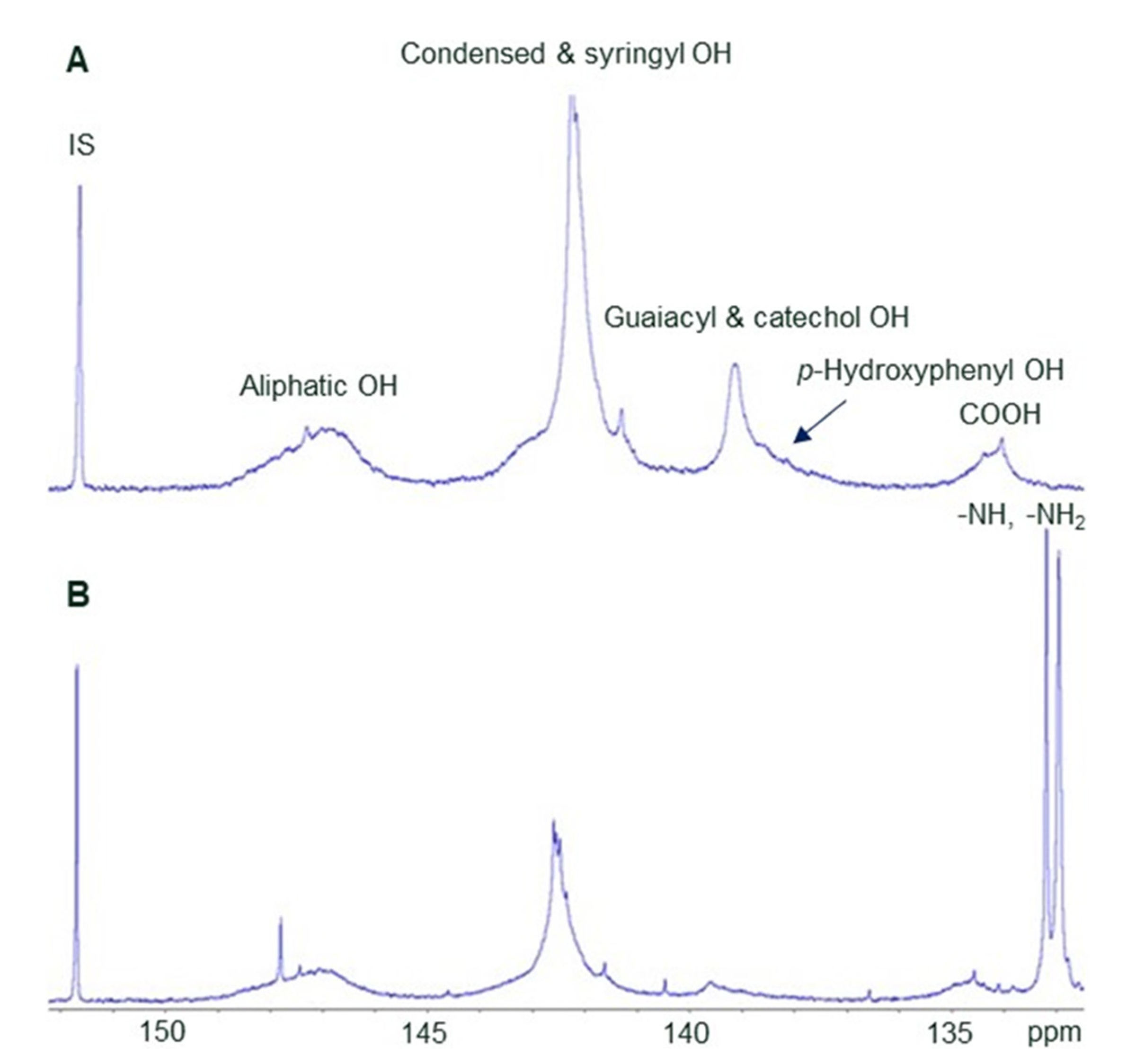




| Lignin/Tannin | Carboxyl, mmol/g | Aliphatic Hydroxyl, mmol/g | Phenolic Hydroxyl by Subtype (Lignins Only), mmol/g | Total Phenolic Hydroxyl, mmol/g | Amino, mmol/g | N, % | ||
|---|---|---|---|---|---|---|---|---|
| Condensedor syringyl | Guaiacyl | p-OH-phenyl | ||||||
| HW kraft lignin | 0.29 ± 0.00 | 0.95 ± 0.02 | 2.62 ± 0.09 | 0.80 ± 0.02 | 0.11 ± 0.09 | 3.53 ± 0.13 | 0.12 ± 0.00 | |
| N-modified HW kraft lignin | 0.30 ± 0.09 | 1.02 ± 0.04 | 2.77 ± 0.02 | 0.29 ± 0.01 | 0.02 ± 0.02 | 2.58 ± 0.05 | 1.47 ± 0.21 | 10.12 ± 0.05 |
| Spruce tannin 3 | 1.13 ± 0.02 | 2.01 ± 0.06 | 3.49 ± 0.15 | 0.57 ± 0.01 | ||||
| N-modified spruce tannin 2 | 10.61 ± 0.02 | |||||||
| Tanfloc SG | 7.60 ± 0.02 | |||||||
| Sample | PHRR (W/g) | TPHRR (°C) | THR (J/g) | Char Yield (wt-%) | |
|---|---|---|---|---|---|
| SW kraft lignin | Test 1 Test 2 Average | 121 124 123 | 419 416 417 | 9440 9860 9650 | 38.7 37.7 38.2 |
| N-modified SW kraft lignin 1 | Test 1 Test 2 Average | 49/48 49/50 49/49 | 269/395 277/402 273/399 | 6340 6200 6270 | 41.6 40.5 41.0 |
| SW CatLignin | Test 1 Test 2 Average | 79 77 78 | 411 413 412 | 6360 6330 6340 | 48.9 49.5 49.2 |
| N-modified SW CatLignin | Test 1 Test 2 Average | 53 52 52 | 285 288 287 | 4860 4920 4890 | 43.8 44.2 44.0 |
| HW kraft lignin | Test 1 Test 2 Average | 154 151 153 | 405 405 405 | 9230 9580 9400 | 36.3 36.2 36.3 |
| N-modified HW kraft lignin | Test 1 Test 2 Average | 64 64 64 | 366 363 365 | 6420 6330 6370 | 39.8 40.0 39.9 |
| Spruce tannin | Test 1 Test 2 Average | 54 55 55 | 256 254 255 | 7280 7300 7290 | 46.1 45.5 45.8 |
| N-modified spruce tannin | Test 1 Test 2 Average | 37 38 37 | 430 413 422 | 5140 5150 5140 | 44.4 44.2 44.3 |
| Retention Aids | Klason Lignin, % 1 | Acid-Soluble Lignin, % 1 | Total Lignin, % 1 | Retention, % 2 |
|---|---|---|---|---|
| None | 6.5 | 0.3 | 6.8 | 85.9 |
| Tanfloc SG 2.5% | 9.7 | 0.9 | 10.7 | 91.0 |
| Tanfloc SG 2.5%, A-PAM 0.15% | 20.0 ± 0.3 | 3.1 ± 0.1 | 23.0 ± 0.2 | 98.4 |
| Tanfloc SG 2.5%, A-PAM 0.30% | 22.0 ± 3.2 | 2.9 ± 0.0 | 25.0 ± 3.2 | 103.0 |
| Tanfloc SG 2.5%, A-PAM 0.45% | 22.8 ± 0.8 | 3.3 ± 0.1 | 26.1 ± 0.7 | 99.4 |
| Sample | Klason Lignin, % | Acid-Soluble Lignin, % | Total Lignin, % | N, % |
|---|---|---|---|---|
| Bleached SW kraft pulp | 1.2 ± 0.0 | 0.0 ± 0.0 | 1.2 | 0.00 ± 0.00 |
| +N-modified SW kraft lignin | 12.5 ± 1.1 | 1.1 ± 0.0 | 13.6 | 1.15 ± 0.13 |
| +N-modified SW CatLignin | 11.5 ± 0.6 | 0.6 ± 0.0 | 12.1 | 1.20 ± 0.22 |
| +N-modified HW kraft lignin | 14.5 ± 1.0 | 1.4 ± 0.1 | 15.9 | 0.52 ± 0.01 |
| +N-modified spruce tannin | 9.8 ± 1.0 | 0.7 ± 0.0 | 10.5 | 0.57 ± 0.02 |
| Sample | PHRR (W/g) | TPHRR (°C) | THR (J/g) | Char Yield (wt-%) | |
|---|---|---|---|---|---|
| Bleached SW kraft pulp | Test 1 Test 2 Average | 315 351 333 | 380 379 379 | 11,360 11,550 11,460 | 6.5 6.6 6.6 |
| Bleached SW kraft pulp + retention aid system Tanfloc SG + A-PAM | Test 1 Test 2 Average | 361 337 349 | 372 371 372 | 12,240 11,870 12,050 | 6.4 7.5 6.9 |
| +N-modified SW kraft lignin | Test 1 Test 2 Average | 268 249 258 | 380 374 377 | 9520 9120 9320 | 15.4 14.4 14.9 |
| +N-modified SW CatLignin | Test 1 Test 2 Average | 265 253 259 | 380 380 380 | 9610 9910 9760 | 14.8 14.5 14.6 |
| +N-modified HW kraft lignin | Test 1 Test 2 Average | 272 242 257 | 386 375 381 | 10,070 9870 9970 | 14.5 12.9 13.7 |
| +N-modified spruce tannin | Test 1 Test 2 Average | 247 303 275 | 371 382 376 | 9280 10,170 9730 | 17.8 14.5 16.2 |
| Dry Weight or Volume | % of Pulp Amount | |
|---|---|---|
| Bleached SW kraft pulp | 1.95 g | 100 |
| N-modified biopolyphenol | 0.40 g | 20 |
| 0.5 M NaOH | 5 mL | |
| Tanfloc SG | 50 mg | 2.5 |
| A-PAM | 9 mg | 0.45 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pöhler, T.; Widsten, P.; Hakkarainen, T. Improved Fire Retardancy of Cellulose Fibres via Deposition of Nitrogen-Modified Biopolyphenols. Molecules 2022, 27, 3741. https://doi.org/10.3390/molecules27123741
Pöhler T, Widsten P, Hakkarainen T. Improved Fire Retardancy of Cellulose Fibres via Deposition of Nitrogen-Modified Biopolyphenols. Molecules. 2022; 27(12):3741. https://doi.org/10.3390/molecules27123741
Chicago/Turabian StylePöhler, Tiina, Petri Widsten, and Tuula Hakkarainen. 2022. "Improved Fire Retardancy of Cellulose Fibres via Deposition of Nitrogen-Modified Biopolyphenols" Molecules 27, no. 12: 3741. https://doi.org/10.3390/molecules27123741
APA StylePöhler, T., Widsten, P., & Hakkarainen, T. (2022). Improved Fire Retardancy of Cellulose Fibres via Deposition of Nitrogen-Modified Biopolyphenols. Molecules, 27(12), 3741. https://doi.org/10.3390/molecules27123741






