The Effect of Combining Post-Harvest Calcium Nanoparticles with a Salicylic Acid Treatment on Cucumber Tissue Breakdown via Enzyme Activity during Shelf Life
Abstract
1. Introduction
2. Results
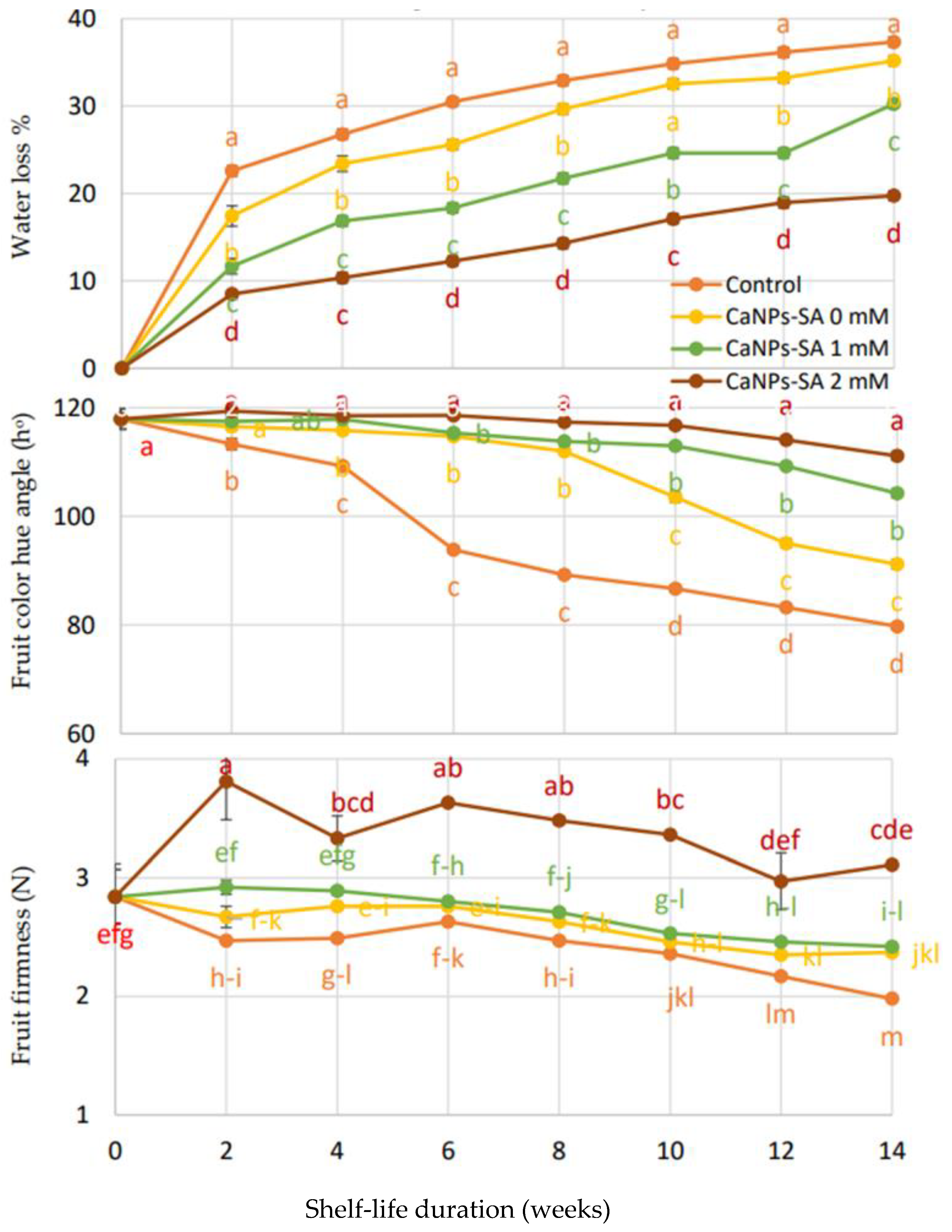
2.1. Physical Properties
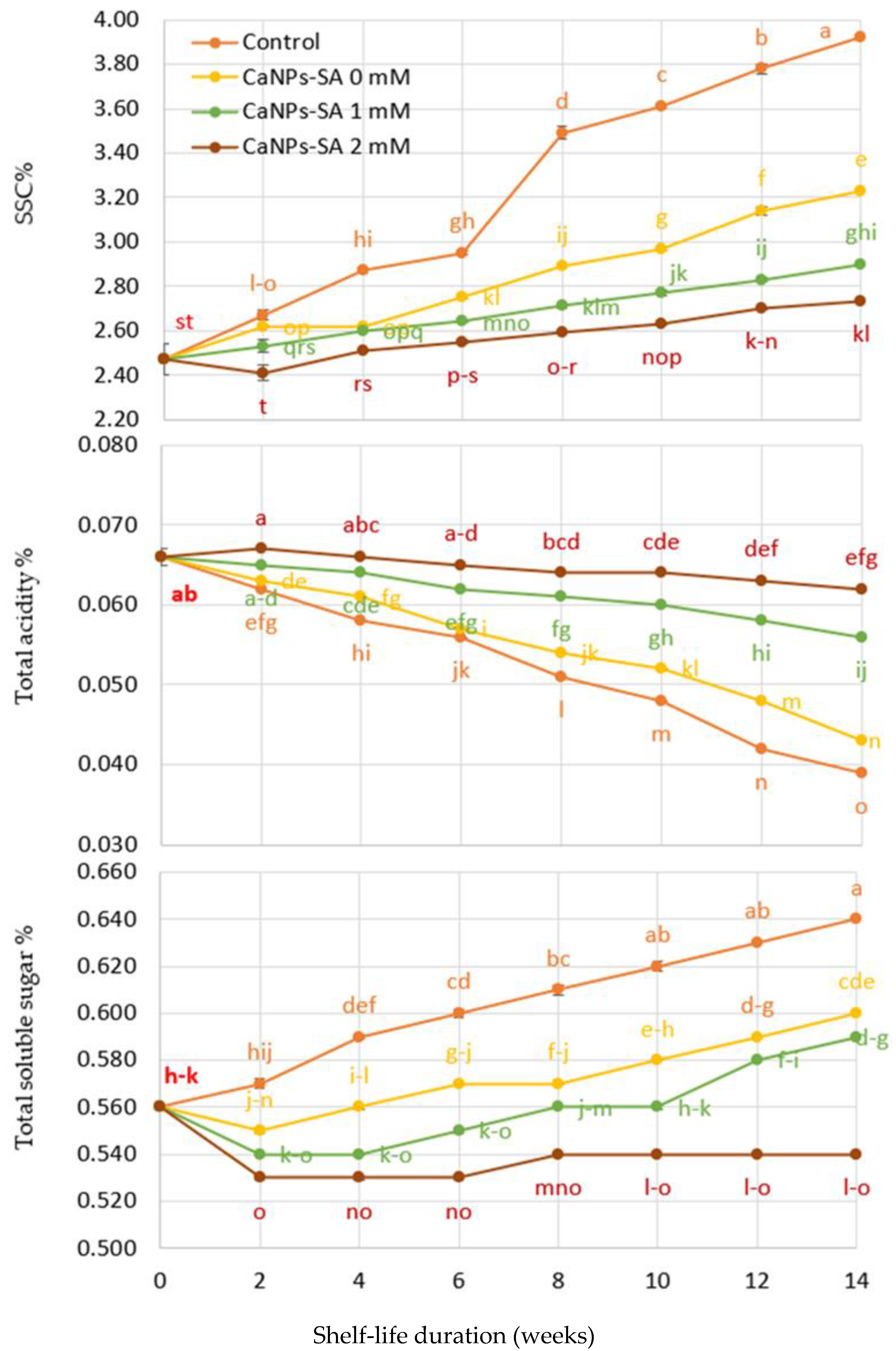
2.2. SSC%, TA%, and SSC/TA Ratio
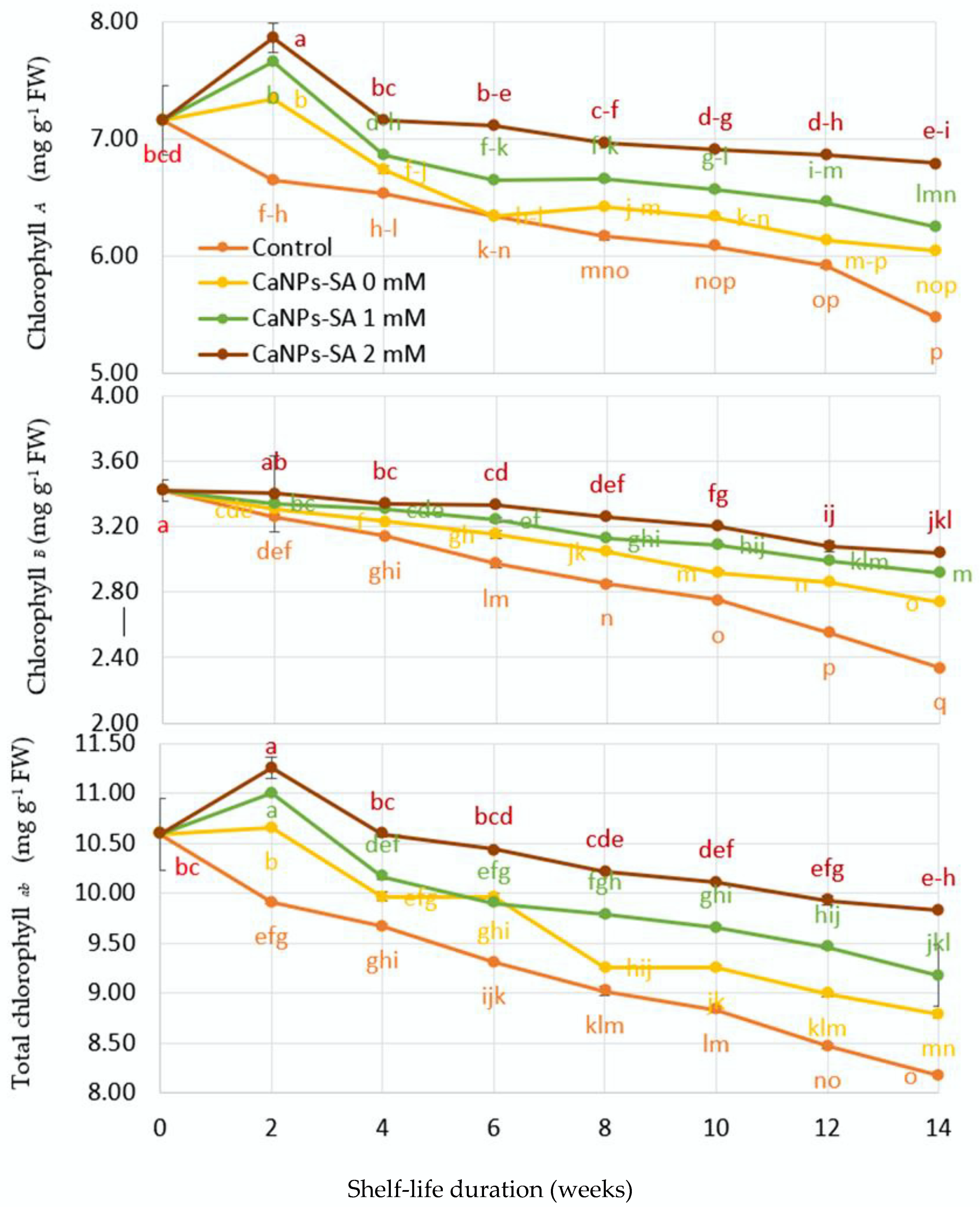
2.3. Cucumber Chlorophyll Pigment Content
2.4. Effect of CaNPs-SA Treatments on Cell Wall Degradation Enzyme Activities
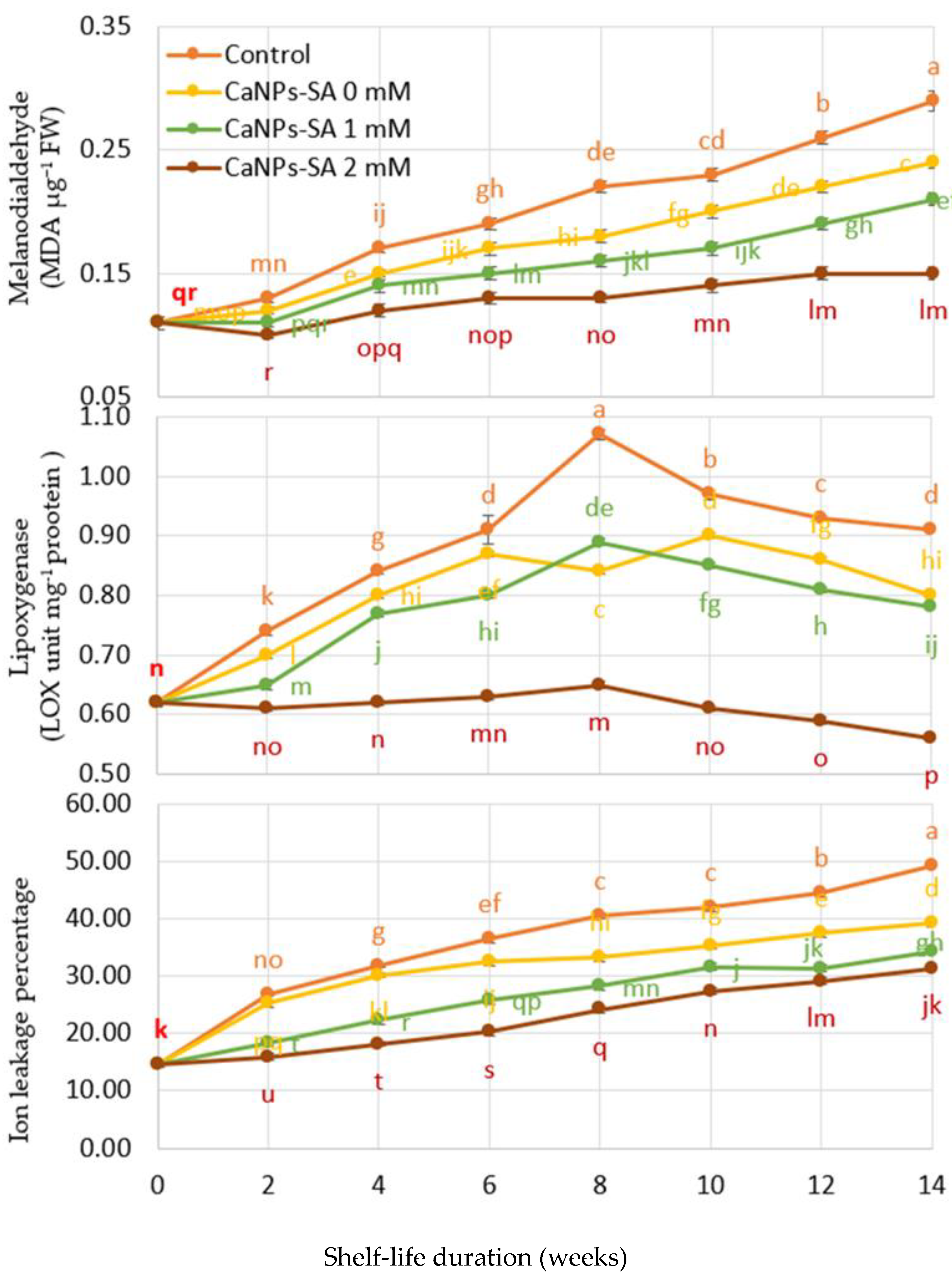
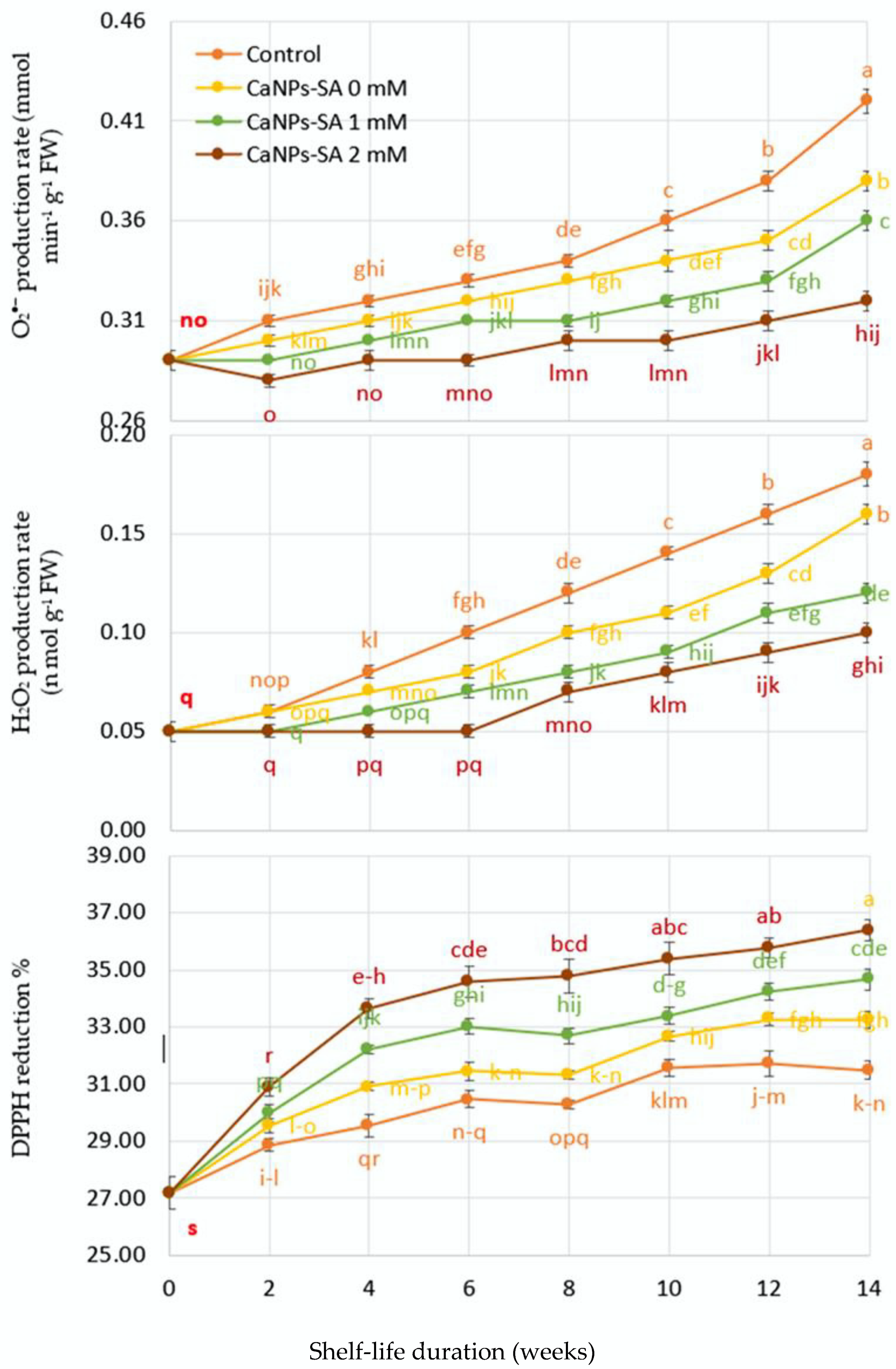
2.5. O2•− and H2O2 Production, DPPH Reduction, LOX, and Percent of Ion Leakage
3. Discussion
4. Materials and Methods
4.1. Fruit Materials
4.2. Use of Salicylic Acid in Calcium Carbonate Nanoparticle (CaNP) Production
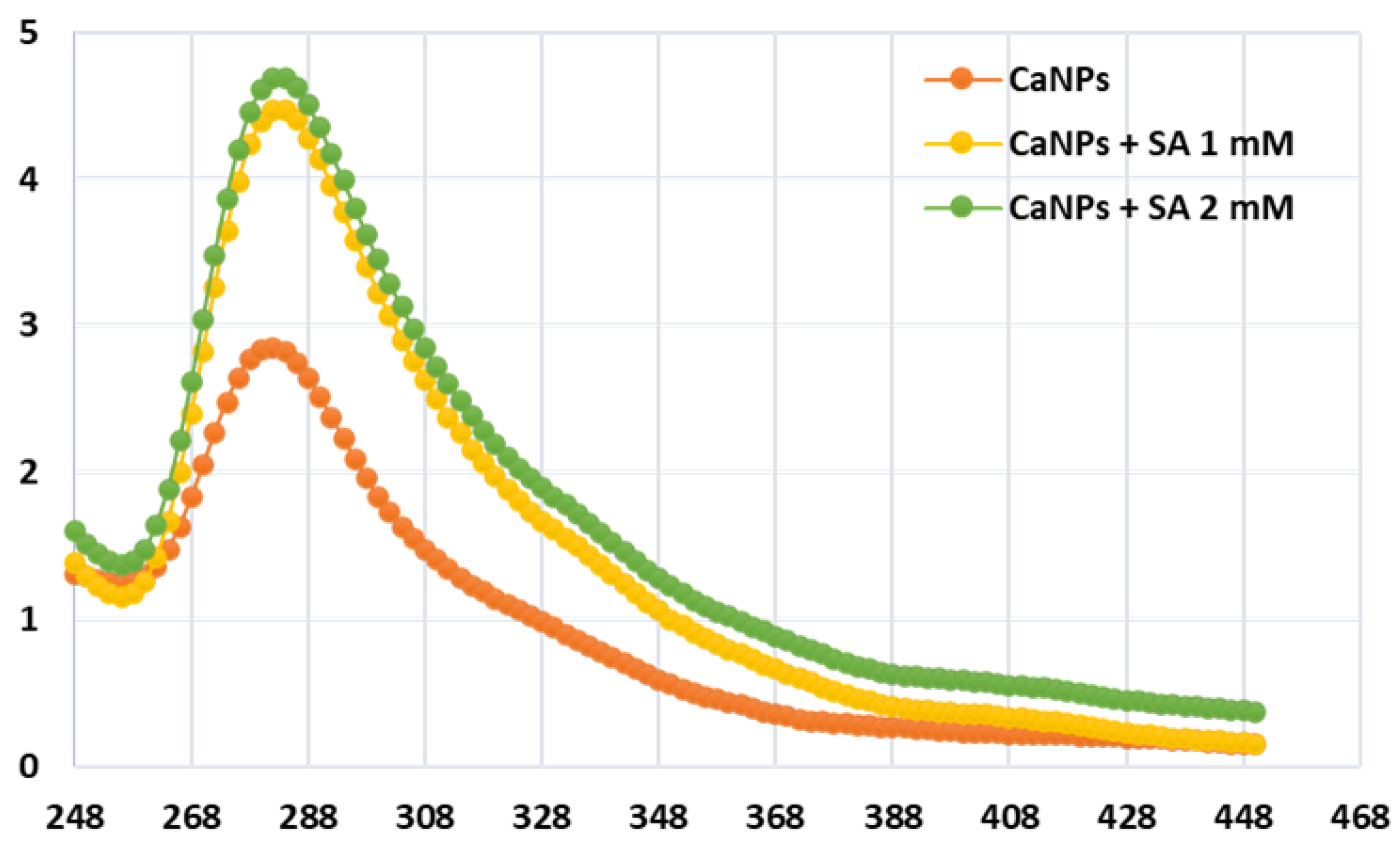
4.3. Nanoparticle Characterization Utilizing UV-Vis Spectroscopy
4.4. CaNP Characterization via Zeta Potential
4.5. Investigation of CaNP Properties via Transmission Electron Microscopy (TEM)
4.6. CaNP Application Routine
4.7. Physical Quality Determination
4.8. Cell Wall Enzyme Activities
4.9. Lipid Peroxidation and Ion Leakage Percentage
4.10. O2•− and H2O2 Free Radical Production Rate
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- FAOSTAT. Source FAOSTAT. 2019. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 7 May 2022).
- Lucier, G.; Jerardo, A. Vegetables and Melons Outlook/VGS-319. In Economic Research Service; USDA: Washington, DC, USA, 2007. [Google Scholar]
- Smith, D.L.; Stommel, J.R.; Fung, R.W.M.; Wang, C.Y.; Whitaker, B.D. Influence of cultivar and harvest method on postharvest storage quality of pepper (Capsicum annuum L.) fruit. Post. Biol. Technol. 2006, 42, 243–247. [Google Scholar] [CrossRef]
- Maguire, K.M.; Banks, N.H.; Lang, A. Sources of variation in water vapour permeance of apple fruit. Post. Biol. Technol. 1999, 17, 11–17. [Google Scholar] [CrossRef]
- Cameron, K.D.; Teece, M.A.; Smart, L.B. Increased accumulation of cuticular wax andexpression of lipidtransfer protein in response to periodic drying events in leaves of tree tobacco. Plant Physiol. 2006, 140, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Mamrutha, H.M.; Mogili, T.; Jhansi Lakshmi, K. Leaf cuticular wax amount and crystal morphology regulate postharvest water loss in mulberry (Morus species). Plant Physiol. Biochem. 2010, 48, 690–696. [Google Scholar] [CrossRef]
- Rojas-Argudo, C.; Erez-Gago, M.B.P.; Del R’ıo, M.A. Postharvest quality of coated cherries cv. ’Burlat’ as affected by coating composition and solids content. Food Sci. Technol. Int. 2005, 11, 417–424. [Google Scholar] [CrossRef]
- Hodges, D.M.; Lester, G.E.; Munro, K.D.; Toivonen, P.M.A. Oxidative stress: Importance for postharvest quality. HortScience 2004, 39, 924–929. [Google Scholar] [CrossRef]
- Porat, R.; Weiss, B.; Cohen, L.; Daus, A.; Aharoni, N. Reduction of postharvest rind disorders in citrus fruit by modified atmosphere packaging. Postharvest Biol. Technol. 2004, 33, 35–43. [Google Scholar] [CrossRef]
- Singh, C.; Sharma, H.K.; Sarkar, B.C. Influence of process conditions on the mass transfer during osmotic dehydration of coated pineapple samples. J. Food Process. Preserv. 2010, 34, 700–714. [Google Scholar] [CrossRef]
- Saini, C.S.; Sharma, H.K. Effect of pectin coating on colour and quality of dehydrated pineapple during storage. Asian J. Dairy Food Res. 2016, 35, 120–129. [Google Scholar] [CrossRef][Green Version]
- Jiang, Y.; Li, Y.; Jiang, E. Effects of chitosan on shelf life of cold-stored litchi fruit at ambient temperature. LWT Food. Sci. Technol. Int. 2005, 38, 757–761. [Google Scholar] [CrossRef]
- Babalar, M.; Asghari, M.; Talaei, A.R.; Khosroshahi, A. Effect of pre- and postharvest salicylic acid treatment on ethylene production, fungal decay and overall quality of selva strawberry fruit. Food Chem. 2007, 105, 449–453. [Google Scholar] [CrossRef]
- Kmita, A.; Hutera, B.; Olejnik, E.; Janas, A. Effect of water glass modification with nanoparticles of zinc oxide on selected physical and chemical properties of binder and mechanical properties of sand mixture. Arch. Foundry Eng. 2012, 12, 37–40. [Google Scholar] [CrossRef]
- Ali, I. Nano anti-cancer drugs: Pros and cons and future perspectives. Curr. Cancer Drug Targets 2011, 11, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Lo’ay, A.A.; Ameer, N.M. Performance of calcium nanoparticles blending with ascorbic acid and alleviation internal browning of ‘Hindi Be-Sennara’ mango fruit at a low temperature. Sci. Hortic. 2019, 254, 199–207. [Google Scholar] [CrossRef]
- Zavala, I.F.A.; Wangs, Y.; Wang, C.Y.; Aguilar, G.A.A. Effects of storage temperatures on antioxidant capacity and aroma compounds in strawberry fruit. LWT-Food Sci. Technol. 2004, 37, 687–695. [Google Scholar] [CrossRef]
- Agamy, R.A.; Elsayed, E.H.; Tarek, H.T. Acquired resistant motivated by salicylic acid applications on salt stressed tomato (Lycopersicon esculentum Mill.). Am. -Eurasian J. Agric. Environ. Sci. 2013, 13, 50–57. [Google Scholar] [CrossRef]
- Dat, I.; Vandenabeele, S.; Vra Nova, E.; Vanmontagu, M.; Inze, D.; Vanbreusegem, F. Dual action of the active oxygen species during plant stress responses. Cell Mol. Life Sci. 2000, 57, 779–795. [Google Scholar] [CrossRef]
- Orabi, S.; Dawood, M.; Salman, S. Comparative study between the physiological role of hydrogen peroxide and salicylic acid in alleviating the harmful effect of low temperature. Sci. Agric. 2015, 9, 49–59. [Google Scholar] [CrossRef]
- Khan, W.; Prithiviraj, B.; Smith, D. Photosynthetic responses of corn and soybean to foliar application of salicylates. J. Plant Physiol 2003, 160, 485–492. [Google Scholar] [CrossRef]
- Hayat, Q.; Hayat, S.; Irfan, M.; Ahmad, A. Effect of exogenous salicylic acid under changing environment: A review. Environ. Exper. Bot. 2010, 68, 14–25. [Google Scholar] [CrossRef]
- Srivastava, M.K.; Dwivedi, U.N. Delayed ripening of banana fruit by salicylic acid. Plant Sci. 2000, 158, 87–96. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, K.; Zhang, S.; Ferguson, I. The role of salicylic acid in postharvest ripening of kiwifruit. Postharvest Biol. Technol. 2003, 28, 67–74. [Google Scholar] [CrossRef]
- Fattahi, J.; Fifaii, R.; Babri, M. Postharvest quality of kiwifruit (actinidia deliciosa cv. hayward) affected by pre-storage application of salicylic acid. S. W. J. Hortic. Biol. Environ. 2010, 1, 175–186. Available online: http://anucraiova.3x.ro/swjhbe/index.html (accessed on 7 May 2022).
- Yao, H.; Tian, S.H. Effects of pre- and post-harvest application of salicylic acid or methyl jasmonate on inducing disease resistance of sweet cherry fruit in storage. Postharvest Biol. Technol. 2005, 35, 253–262. [Google Scholar] [CrossRef]
- Ding, C.K.; Wang, C.Y.; Gross, K.C.; Smith, D.I. Jasmonate and salicylate induce the expression of pathogenesis-related-protein genes and increase resistance to chilling injury in tomato fruit. Planta 2002, 214, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Lo’ay, A.A.; Taher, M.A. Influence of edible coatings chitosan/PVP blending with salicylic acid on biochemical fruit skin browning incidence and shelf life of guava fruits cv. ‘Banati’. Sci. Hortic. 2018, 235, 424–436. [Google Scholar] [CrossRef]
- Pasanphan, W.; Buettner, G.R.; Chirachanchai, S. Chitosan gallate as a novel potential polysaccharide antioxidant: An EPR study. Carbohydr. Res. 2010, 345, 132–140. [Google Scholar] [CrossRef]
- Tareen, M.J.; Abbasi, N.A.; Hafiz, I.A. Postharvest application of salicylic acid enhanced antioxidant enzymes activity and maintanied quality of peach cv. ‘Flordaking’ fruit during storage. Sci. Hortic. 2012, 142, 221–228. [Google Scholar] [CrossRef]
- El-Ezz, S.F.A.; Al-Harbi, N.A.; Al-Qahtani, S.M.; Allam, H.M.; Abdein, M.A.; Abdelgawad, Z.A. A Comparison of the Effects of Several Foliar Forms of Magnesium Fertilization on ‘Superior Seedless’ (Vitis vinifera L.) in Saline Soils. Coatings 2022, 12, 201. [Google Scholar] [CrossRef]
- Sun, X.; Wang, Z.; Kadouh, H.; Zhou, K. The antimicrobal, mechanical, physical and structureal properies of chitosan-gallic acid film. LWT Food Sci. Techol. 2014, 57, 83–89. [Google Scholar] [CrossRef]
- Foyer, C.H.; Ruban, A.V.; Noctor, G. Viewing oxidative stress through the lens of oxidative signaling rather than damage. Biochem. J. 2017, 474, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Sayyari, M.; Babalar, M.; Kalantari, S.; Martínez-Romero, D.; Guillén, F.; Serrano, M.; Valero, D. Vapour treatments with methyl salicylate or methyl jasmonate alleviated chilling injury and enhanced antioxidant potential during postharvest storage of pomegranates. Food Chem. 2011, 124, 964–970. [Google Scholar] [CrossRef]
- Lo’ay, A.A.; Elgammal, R.E.; Alhaithloul, H.A.S.; Alghanem, S.M.; Fikry, M.; Abdein, M.A.; Hikal, D.M. Enhance Fruit Ripening Uniformity and Accelerate the Rutab Stage by Using ATP in ‘Zaghloul’ Dates during the Shelf Life. Foods 2021, 10, 2641. [Google Scholar] [CrossRef] [PubMed]
- Shoala, T.; Al-Karmalawy, A.A.; Germoush, M.O.; Alshamrani, S.M.; Abdein, M.A.; Awad, N.S. Nanobiotechnological Approaches to Enhance Potato Resistance against Potato Leafroll Virus (PLRV) Using Glycyrrhizic Acid Ammonium Salt and Salicylic Acid Nanoparticles. Horticulturae 2021, 7, 402. [Google Scholar] [CrossRef]
- Youwei, Y.; Yinzhe, R. Grapes preservation using chitosan combined with β−cyclodextrin. Int. J. Agron. 2013, 4, 1–8. [Google Scholar] [CrossRef]
- Lo’ay, A.A.; Mostafa, N.A.; Al-Qahtani, S.M.; Al-Harbi, N.A.; Hassan, S.; Abdein, M.A. Influence of the Position of Mango Fruit on the Tree (Mangifera indica L. CV. ‘Zibda’) on Chilling Sensitivity and Antioxidant Enzyme Activity. Horticulturae 2021, 7, 515. [Google Scholar] [CrossRef]
- Lo’ay, A.A. Chilling Injury in Mangoes. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2005; pp. 1–224, ISBN: 90-8504-309-3. [Google Scholar]
- Purvis, A.C. Regulatin of oxidative stress in horticultural crops. HortiScience 2004, 39, 930–932. [Google Scholar] [CrossRef]
- Taher, M.A.; Lo’ay, A.A.; Gouda, M.; Limam, S.A.; Abdelkader, M.F.M.; Osman, S.O.; Fikry, M.; Ali, E.F.; Mohamed, S.Y.; Khalil, H.A.; et al. Impacts of Gum Arabic and Polyvinylpyrrolidone (PVP) with Salicylic Acid on Peach Fruit (Prunus persica) Shelf Life. Molecules 2022, 27, 2595. [Google Scholar] [CrossRef]
- Champa, W.A.H.; Gill, M.I.S.; Mahajan, B.V.C.; Arora, N.K. Preharvest salicylic acid treatments to improve quality and postharvest life of table grapes (Vitis vinifera L.) cv. Flame Seedless. J. Food Sci. Technol. 2015, 52, 3607–3616. [Google Scholar] [CrossRef]
- Roustan, J.P.; Latché, A.; Fallot, J. Inhibition of ethylene production and stimulation of carrot somatic embryogenesis by salicylic acid. Biol. Plant 1990, 32, 273–276. [Google Scholar] [CrossRef]
- Tang, T.; Huang, D.-W.; Zhou, C.-Q.; Li, X.; Xie, Q.-J.; Liu, F.-S. Molecular cloning and expression patterns of copper/zinc superoxide dismutase and manganese superoxide dismutase in Musca domestica. Gene 2012, 505, 211–220. [Google Scholar] [CrossRef]
- Yang, H.; Wu, F.; Cheng, J. Reduced chilling injury in cucumber by nitricoxide and the antioxidant response. Food Chem. 2011, 127, 1237–1242. [Google Scholar] [CrossRef] [PubMed]
- Yugandhar, P.; Savithramma, N. Green synthesis of calcium carbonate nanoparticles and their effects on seed germination and seedling growth of Vigna mungo (L.). Hepper cv. Int. J. Adv. Res. 2013, 1, 89–103. Available online: http://www.journalijar.com/uploads/2013-11-30_140955_59.pdf (accessed on 7 May 2022).
- Soheyla, H.; Foruhe, Z. Effect of Zeta Potential on the Properties of Nano-Drug Delivery Systems—A Review (Part 2). Trop. J. Pharm. Res. 2013, 12, 265–273. [Google Scholar] [CrossRef]
- Naveena, B.; Genitha, I. Effect of calcium chloride, sodium chloride and lime juice on physico-chemical properties of cucumber. Int. J. Agric. Sci. Res. 2017, 7, 765–770. [Google Scholar] [CrossRef]
- Lo’ay, A.A.; Ismail, H.; Kassem, H.S. The Quality of Superior Seedless Bunches during Shelf Life as Determined by Growth on Different Rootstocks. Agriculture 2021, 11, 990. [Google Scholar] [CrossRef]
- Omoba, O.S.; Onyekwere, U. Postharvest physicochemical properties of cucumber fruits (Cucumber sativus L) treated with chitosan-lemon grass extracts under different storage durations. Afr. J. Biotechnol. 2016, 15, 2758–2766. [Google Scholar] [CrossRef]
- Nardello, I.C.; Cantillano, R.F.F.; Seifer, M.; Mello-Farias, P.C.; Malgarim, M.B.; De Oliveira, R.P. Postharvest quality during refrigerated storage of ‘Nadorcott’ mandarin. J. Exper. Agric. Inter. 1994, 19, 1–10. [Google Scholar] [CrossRef]
- Porra, R.J.; Thompson, W.A.; Kriedemann, P.E. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: Verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta 1989, 975, 384–394. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for the determination of reducing suga. Anal. Chem. 1959, 31, 426–429. [Google Scholar] [CrossRef]
- Collmer, A.; Reid, J.L.; Mount, M.S. Assay methods for pectic enzymes. In Methods in Enzymology; Wood, W.A., Kellogg, S.T., Eds.; Academic Press: San Diego, CA, USA, 1988; Volume 161, pp. 329–335. [Google Scholar]
- Payasi, A.; Sanwal, G.G. Pectate lyase activity during ripening of banana fruit. Phytochemistry 2003, 63, 243–248. [Google Scholar] [CrossRef]
- Lo’ay, A.A.; Dawood, H.D. Active chitosan/PVA with ascorbic acid and berry quality of ‘Superior seedless’ grapes. Sci. Hortic. 2017, 224, 286–292. [Google Scholar] [CrossRef]
- Iturbe-Ormaetxe, I.; Escuredo, P.R.; Arrese-Igor, C.; Becana, M. Oxidative damage in pea plants exposed to water deficit or paraquat. Plant Physiol. 1998, 116, 173–181. [Google Scholar] [CrossRef]
- Xu, M.; Dong, J.; Zhang, M.; Xu, X.; Sun, L. Cold-induced endogenous nitric oxide generation plays a role in chilling tolerance of loquat fruit during postharvest storage. Postharvest Biol. Technol. 2012, 65, 5–12. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelkader, M.F.M.; Mahmoud, M.H.; A., L.A.; Abdein, M.A.; Metwally, K.; Ikeno, S.; Doklega, S.M.A. The Effect of Combining Post-Harvest Calcium Nanoparticles with a Salicylic Acid Treatment on Cucumber Tissue Breakdown via Enzyme Activity during Shelf Life. Molecules 2022, 27, 3687. https://doi.org/10.3390/molecules27123687
Abdelkader MFM, Mahmoud MH, A. LA, Abdein MA, Metwally K, Ikeno S, Doklega SMA. The Effect of Combining Post-Harvest Calcium Nanoparticles with a Salicylic Acid Treatment on Cucumber Tissue Breakdown via Enzyme Activity during Shelf Life. Molecules. 2022; 27(12):3687. https://doi.org/10.3390/molecules27123687
Chicago/Turabian StyleAbdelkader, Mohamed F. M., Mohamed H. Mahmoud, Lo’ay A. A., Mohamed A. Abdein, Khaled Metwally, Shinya Ikeno, and Samar M. A. Doklega. 2022. "The Effect of Combining Post-Harvest Calcium Nanoparticles with a Salicylic Acid Treatment on Cucumber Tissue Breakdown via Enzyme Activity during Shelf Life" Molecules 27, no. 12: 3687. https://doi.org/10.3390/molecules27123687
APA StyleAbdelkader, M. F. M., Mahmoud, M. H., A., L. A., Abdein, M. A., Metwally, K., Ikeno, S., & Doklega, S. M. A. (2022). The Effect of Combining Post-Harvest Calcium Nanoparticles with a Salicylic Acid Treatment on Cucumber Tissue Breakdown via Enzyme Activity during Shelf Life. Molecules, 27(12), 3687. https://doi.org/10.3390/molecules27123687







