Regiospecific Hydrogenation of Bromochalcone by Unconventional Yeast Strains
Abstract
:1. Introduction
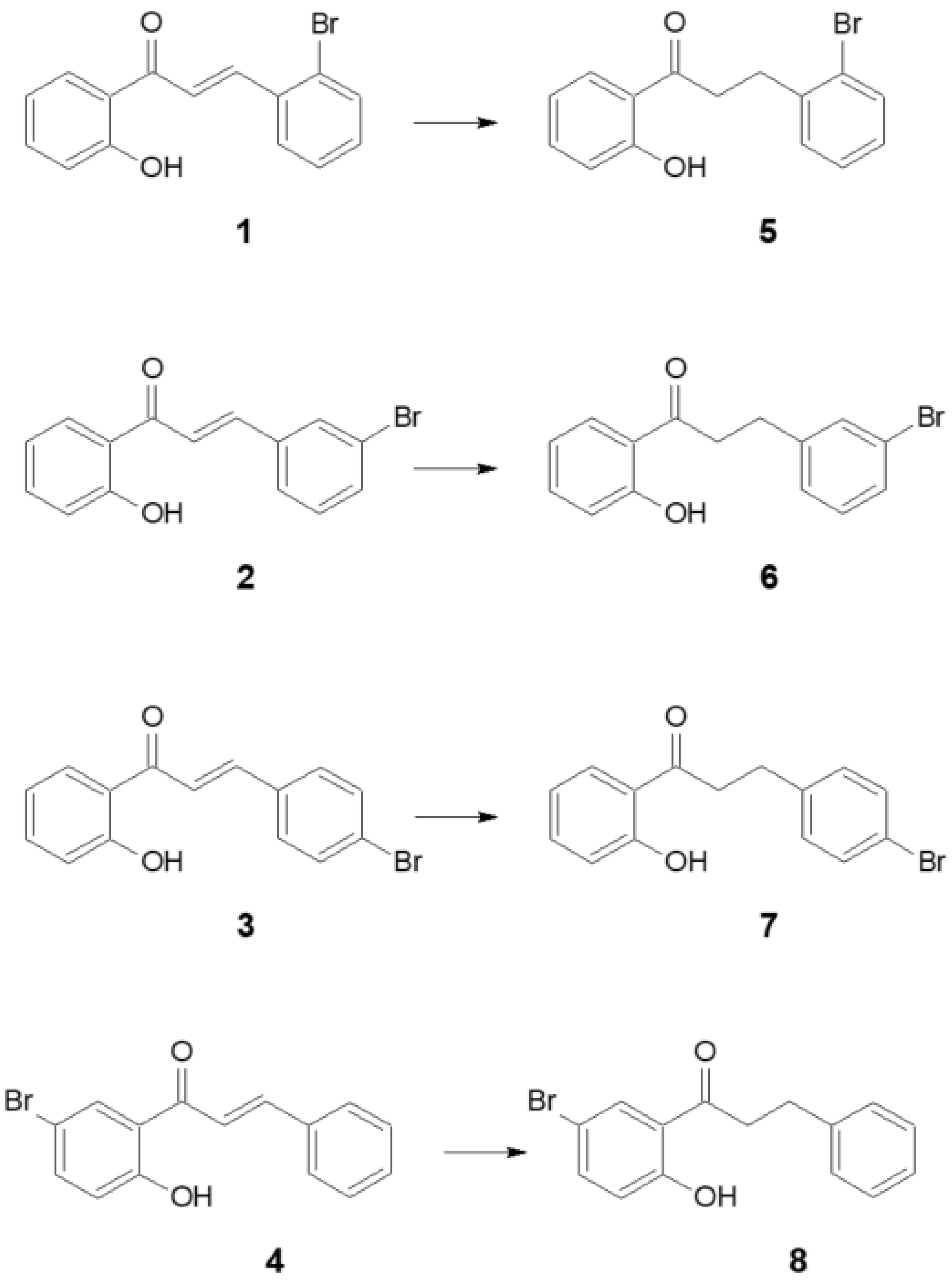
2. Results
3. Discussion
4. Materials and Methods
4.1. Substrates
4.2. Microorganisms
4.3. Screening
4.4. Gas Chromatography
4.5. Preparative Scale
4.6. TLC and NMR Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ohta, H.; Konishi, J.; Tsuchihashi, G. Selective hydrogenation of carbon-carbon double bonds of chalcones by Corynebacterium equi IFO 3730. Agric. Biol. Chem. 1985, 49, 665–669. [Google Scholar] [CrossRef]
- Silva, V.D.; Stambuk, B.U.; Nascimento, M.G. Efficient chemoselective biohydrogenation of 1,3-diaryl-2-propen-1-ones catalysed by Saccharomyces cerevisiae yeasts in biphasic system. J. Mol. Catal. B Enzym. 2010, 63, 157–163. [Google Scholar] [CrossRef]
- Pathan, N.B.; Rahatgaonkar, A.M.; Chorghade, M.S. Stereoselective bioreduction of chalcone and β-diketone by Saccharomyces cerevisiae in biphasic solvent system: A mechanistic study. Indian J. Chem. 2012, 51B, 992–1001. [Google Scholar]
- Corrêa, M.J.C.; Nunes, F.M.; Bitencourt, H.R.; Borges, F.C.; Guilhon, G.M.S.P.; Arruda, M.S.P.; Marinho, A.M.R.; Santos, A.S.; Alves, C.N.; Brasil, D.S.B.; et al. Biotransformation of chalcones by the endophytic fungus Aspergillus flavus isolated from Paspalum maritimum trin. J. Braz. Chem. Soc. 2011, 22, 1333–1338. [Google Scholar] [CrossRef]
- Schoefer, L.; Mohan, R.; Schwiertz, A.; Braune, A.; Blaut, M. Anaerobic degradation of flavonoids by Clostridium orbiscindens. Appl. Environ. Microbiol. 2003, 69, 5849–5854. [Google Scholar] [CrossRef] [Green Version]
- Żyszka-Haberecht, B.; Poliwoda, A.; Lipok, J. Biocatalytic hydrogenation of the C=C bond in the enone unit of hydroxylated chalcones—Process arising from cyanobacterial adaptations. Appl. Microbiol. Biotechnol. 2018, 102, 7097–7111. [Google Scholar] [CrossRef] [Green Version]
- Rosa, G.P.; Seca, A.M.L.; do Carmo Barreto, M.; Pinto, D.C.G.A. Chalcone: A valuable scaffold upgrading by green methods. ACS Sustain. Chem. Eng. 2017, 5, 7467–7480. [Google Scholar] [CrossRef]
- Raimondi, S.; Romano, D.; Amaretti, A.; Molinari, F.; Rossi, M. Enoate reductases from non conventional yeasts: Bioconversion, cloning, and functional expression in Saccharomyces cerevisiae. J. Biotechnol. 2010, 156, 279–285. [Google Scholar] [CrossRef]
- Krawczyk-Łebek, A.; Dymarska, M.; Janeczko, T.; Kostrzewa-Susłow, E. New glycosylated dihydrochalcones obtained by biotransformations of 2′-hydroxy-2-methylchalcone in cultures of entomopathogenic filamentous fungi. Int. J. Mol. Sci. 2021, 22, 9619. [Google Scholar] [CrossRef]
- Stompor, M.; Potaniec, B.; Szumny, A.; Zieliński, P.; Zołnierczyk, A.K.; Anioł, M. Microbial synthesis of dihydrochalcones using Rhodococcus and Gordonia species. J. Mol. Catal. B Enzym. 2013, 97, 283–288. [Google Scholar] [CrossRef]
- Stompor, M.; Kałużny, M.; Żarowska, B. Biotechnological methods for chalcone reduction using whole cells of Lactobacillus, Rhodococcus and Rhodotorula strains as a way to produce new derivatives. Appl. Microbiol. Biotechnol. 2016, 100, 8371–8384. [Google Scholar] [CrossRef] [PubMed]
- Klingel, T.; Hadamjetz, M.; Fischer, A.; Wefers, D. Glucosylation of flavonoids and flawonoid glycosides by mutant dextransucrase from Lactobacillus reuteri TMW 1. Carbohydr. Res. 2019, 483, 107741. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Liu, A.; Cao, H.; Luo, Y.; Pezzuto, J.M.; van Breemen, R.B. Biotransformation of the chemopreventive agent 2′,4′,4-trihydroxychalcone (Isoliquiritigenin) by UDP-glucuronosyltransferases. Drug Metab. Dispos. 2008, 36, 2104–2112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krawczyk-Łebek, A.; Dymarska, M.; Janeczko, T.; Kostrzewa-Susłow, E. Glycosylation of methylflavonoids in the cultures of entomopathogenic filamentous fungi as a tool for obtaining new biologically active compounds. Int. J. Mol. Sci. 2022, 23, 5558. [Google Scholar] [CrossRef]
- Stompor, M.; Potaniec, B.; Szumny, A.; Zieliński, P.; Zołnierczyk, A.; Anioł, M. Microbiological reduction of xanthohumol and 4-methoxychalcone. Przem. Chem. 2013, 92, 574–578. [Google Scholar]
- Kozłowska, J.; Potaniec, B.; Żarowska, B.; Anioł, M. Microbial transformations of 4′-methylchalcones as an efficient method of obtaining novel alcohol and dihydrochalcone derivatives with antimicrobial activity. RSC Adv. 2018, 8, 30379–30386. [Google Scholar] [CrossRef] [Green Version]
- Janeczko, T.; Gładkowski, W.; Kostrzewa-Susłow, E. Microbial transformations of chalcones to produce food sweetener derivatives. J. Mol. Catal. B Enzym. 2013, 98, 55–61. [Google Scholar] [CrossRef]
- Xiao, Y.; Han, F.; Lee, I.-S. Biotransformation of the phenolic constituents from licorice and cytotoxicity evaluation of their metabolites. Int. J. Mol. Sci. 2021, 22, 10109. [Google Scholar] [CrossRef]
- de Matos, L.I.; Nitschke, M.; Porto, A.L.M. Hydrogenation of halogenated 2′-Hydroxychalcones by mycelia of marine-derived fungus Penicillium raistrickii. Mar. Biotechnol. 2019, 21, 430–439. [Google Scholar] [CrossRef]
- Łużny, M.; Krzywda, M.; Kozłowska, E.; Kostrzewa-Susłow, E.; Janeczko, T. Effective Hydrogenation of 3-(2″-furyl)- and 3-(2″-thienyl)-1-(2′-hydroxyphenyl)-prop-2-en-1-one in Selected Yeast Cultures. Molecules 2019, 24, 3185. [Google Scholar] [CrossRef] [Green Version]
- Stompor, M.; Broda, D.; Bajek-Bil, A. Dihydrochalcones: Methods of acquisition and pharmacological properties—A first systematic review. Molecules 2019, 24, 4468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwon, M.; Kim, Y.; Lee, J.; Manthey, J.A.; Kim, Y.; Kim, Y. Neohesperidin dihydrochalcone and neohesperidin dihydrochalcone-O-glycoside attenuate subcutaneous fat and lipid accumulation by regulating PI3K/AKT/mTOR pathway in vivo and in vitro. Nutrients 2022, 14, 1087. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, A. Sweeteners permitted in the European Union: Safety aspects. Scand. J. Food Nutr. 2006, 50, 104–116. [Google Scholar] [CrossRef]
- Arokia, M.V.A.; Ramachandran, V.; Vinothkumar, R.; Vijayalakshmi, S.; Sathish, V.; Ernest, D. Pharmacological aspects and potential use of phloretin: A systemic review. Mini Rev. Med. Chem. 2019, 19, 1060–1067. [Google Scholar] [CrossRef]
- Minsat, L.; Peyrot, C.; Brunissen, F.; Renault, J.-H.; Allais, F. Synthesis of biobased phloretin analogues: An access to antioxidant and anti-tyrosinase compounds for cosmetic applications. Antioxidants 2021, 10, 512. [Google Scholar] [CrossRef]
- Casarini, T.P.A.; Frank, L.A.; Pohlmann, A.R.; Guterres, S.S. Dermatological applications of the flavonoid phloretin. Eur. J. Pharmacol. 2020, 889, 173593. [Google Scholar] [CrossRef]
- Han, G.E.; Kang, H.T.; Chung, S.; Lim, C.; Linton, J.A.; Lee, J.H.; Kim, W.; Kim, S.H.; Lee, J.H. Novel neohesperidin dihydrochalcone analogue inhibits adipogenic differentiation of human adipose-derived stem cells through the Nrf2 pathway. Int. J. Mol. Sci. 2018, 19, 2215. [Google Scholar] [CrossRef] [Green Version]
- Sinjman, P.W.; Joubert, E.; Ferreira, D.; Li, X.C.; Ding, Y.; Green, I.R.; Gelderblom, W.C.A. Antioxidant activity of the dihydrochalcones aspalathin and nothofagin and their corresponding flavones in relation to other rooibos (Aspalathus linearis) flavonoids, epigallocatechin gallate, and Trolox. J. Agric. Food Chem. 2009, 57, 6678–6684. [Google Scholar] [CrossRef]
- Łużny, M.; Kozłowska, E.; Kostrzewa-Susłow, E.; Janeczko, T. Highly effective, regiospecific hydrogenation of methoxychalcone by Yarrowia lipolytica leads to the food sweeteners production. Catalysts 2020, 10, 1135. [Google Scholar] [CrossRef]
- Kwan, E.E.; Zeng, Y.; Besser, H.A.; Jacobsen, E.N. Concerted nucleophilic aromatic substitutions. Nat. Chem. 2018, 10, 917–923. [Google Scholar] [CrossRef]
- Fager, D.C.; Lee, K.A.; Hoveyda, A.H. Catalytic Enantioselective Addition of an Allyl Group to Ketones Containing a Tri-, a Di-, or a Monohalomethyl Moiety. Stereochemical Control Based on Distinctive Electronic and Steric Attributes of C–Cl, C–Br, and C–F Bonds. J. Am. Chem. Soc. 2019, 141, 16125–16138. [Google Scholar] [CrossRef] [PubMed]
- Loughlin, W.A. Biotransformations in organic synthetis. Bioresour. Technol. 2000, 74, 49–62. [Google Scholar] [CrossRef]
- Ferreira, I.M.; Rocha, L.C.; Yoshioka, S.A.; Nitschke, M.; Jeller, A.H.; Pizzuti, L.; Seleghim, M.H.R.; Porto, A.L.M. Chemoselective reduction of chalcones by whole hyphae of marine fungus Penicillium citrinum 1186, free and immobilised on bioplymers. Biocatal. Agric. Biotechnol. 2014, 3, 358–364. [Google Scholar] [CrossRef]
- Żyszka-Haberecht, B.; Poliwoda, A.; Lipok, J. Structural constraints in cyanobacteria-mediated whole-cell biotransformation of methoxylated and methylated derivatives of 2′-hydroxychalcone. J. Biotechnol. 2019, 293, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Filippucci, S.; Tasselli, G.; Labbani, F.-Z.K.; Turchetti, B.; Cramarossa, M.R.; Buzzini, P.; Forti, L. Non-conventional yeasts as sources of ene-reductases for the bioreduction of chalcones. Fermentation 2020, 6, 29. [Google Scholar] [CrossRef] [Green Version]
- de Matos, L.I.; Nitschke, M.; Porto, A.L.M. Regioselective and chemoselective biotransformation of 2′-hydroxychalcone derivatives by marine-derived fungi. Biocatal. Biotransform. 2021, 1–11. [Google Scholar] [CrossRef]
- Kozłowska, J.; Potaniec, B.; Anioł, M. Biotransformation of Hydroxychalcones as a Method of Obtaining Novel and Unpredictable Products Using Whole Cells of Bacteria. Catalysts 2020, 10, 1167. [Google Scholar] [CrossRef]
- Janeczko, T.; Popłoński, J.; Kozłowska, E.; Dymarska, M.; Huszcza, E.; Kostrzewa-Susłow, E. Application of α- and β-naphthoflavones as monooxygenase inhibitors of Absidia coerulea KCh 93, Syncephalastrum racemosum KCh 105 and Chaetomium sp. KCh 6651 in transformation of 17α-methyltestosterone. Bioorganic Chem. 2018, 78, 178–184. [Google Scholar] [CrossRef]
- Łużny, M.; Tronina, T.; Kozłowska, E.; Dymarska, M.; Popłoński, J.; Łyczko, J.; Kostrzewa-Susłow, E.; Janeczko, T. Biotransformation of metoxyflavones by selected entomopathogenic filamentous fungi. Int. J. Mol. Sci. 2020, 21, 6121. [Google Scholar] [CrossRef]
- Pawlak, A.; Henklewska, M.; Hernández Suárez, B.; Motykiewicz-Pers, K.; Łużny, M.; Kozłowska, E.; Obmińska-Mrukowicz, B.; Janeczko, T. Chalcone methoxy derivatives exhibit antiproliferative and proapoptotic activity on canine lymphoma and leukemia cells. Molecules 2020, 25, 4362. [Google Scholar] [CrossRef]
- Janeczko, T.; Dymarska, M.; Siepka, M.; Gniłka, R.; Leśniak, A.; Popłoński, J.; Kostrzewa-Susłow, E. Enantioselective reduction of flavanone and oxidation of cis- and trans-flavan-4-ol by selected yeast cultures. J. Mol. Catal. B Enzym. 2014, 109, 47–52. [Google Scholar] [CrossRef]
- Cho, C.S.; Shim, S.C. A ruthenium-catalysed one-pot method for α-alkylation of ketones with aldehydes. J. Organomet. Chem. 2006, 691, 4329–4332. [Google Scholar] [CrossRef]

| Strain | Substrate | Degree of Conversion after a Specified Incubation Time | |||
|---|---|---|---|---|---|
| 1 Day | 3 Days | 7 Days | 10 Days | ||
| Rhodotorula rubra KCh 4 | 1 | 7 | 22 | 71 | 75 |
| 2 | 10 | 13 | 14 | 21 | |
| 3 | 34 | 83 | 99 | >99 | |
| 4 | 67 | 87 | 90 | 92 | |
| Rhodotorula rubra KCh 82 | 1 | 16 | 24 | 72 | 82 |
| 2 | 10 | 12 | 21 | 54 | |
| 3 | 34 | 68 | 99 | >99 | |
| 4 | 66 | 87 | 88 | 91 | |
| Rhodotorula marina KCh 77 | 1 | 3 | 11 | 63 | 72 |
| 2 | 53 | 72 | 90 | 97 | |
| 3 | 23 | 47 | 92 | 99 | |
| 4 | 57 | 81 | 86 | 90 | |
| Rhodotorula glutinis KCh 242 | 1 | 59 | 93 | 97 | 98 |
| 2 | 93 | 95 | 98 | 99 | |
| 3 | 15 | 38 | 67 | 89 | |
| 4 | 4 | 9 | 11 | 15 | |
| Saccharomyces cerevisiae KCh 464 | 1 | 77 | 82 | 98 | 99 |
| 2 | 2 | 3 | 3 | 4 | |
| 3 | >99 | >99 | >99 | >99 | |
| 4 | 63 | 77 | 84 | 92 | |
| Candida viswanathii KCh 120 | 1 | 94 | 96 | 98 | 99 |
| 2 | 32 | 52 | 94 | 96 | |
| 3 | 98 | 99 | >99 | >99 | |
| 4 | 6 | 30 | 34 | 75 | |
| Candida parapsilosis KCh 909 | 1 | 92 | 95 | 97 | 98 |
| 2 | 80 | 85 | 94 | 95 | |
| 3 | 46 | 92 | >99 | >99 | |
| 4 | 3 | 15 | 18 | 20 | |
| Yarrowia lipolytica KCh 71 | 1 | 98 | 99 | >99 | >99 |
| 2 | 90 | 95 | 97 | 98 | |
| 3 | 74 | 95 | 98 | 92 | |
| 4 | 91 | 93 | 95 | 96 | |
| Strain | Substrate | Degree of Conversion after Specified Incubation Time | |||
|---|---|---|---|---|---|
| 1 h | 3 h | 6 h | 12 h | ||
| Rhodotorula rubra KCh 4 | 4 | 31 | 38 | 45 | 50 |
| Rhodotorula glutinis KCh 242 | 2 | 21 | 25 | 28 | 31 |
| Saccharomyces cerevisiae KCh 464 | 1 | 31 | 40 | 56 | 71 |
| 3 | 58 | 81 | 94 | 99 | |
| 4 | 26 | 44 | 57 | 78 | |
| Candida viswanathii KCh 120 | 1 | 81 | 99 | >99 | >99 |
| 3 | 23 | 40 | 68 | 94 | |
| Candida parapsilosis KCh 909 | 1 | 75 | 92 | 94 | 96 |
| 2 | 29 | 58 | 82 | 88 | |
| Yarrowia lipolytica KCh 71 | 1 | 96 | 98 | 98 | 99 |
| 2 | 91 | 95 | 96 | 97 | |
| 3 | 10 | 18 | 28 | 42 | |
| 4 | 80 | 93 | 96 | 97 | |
| Proton | Compound | |
|---|---|---|
| 1 | 5 | |
| H-2 | 8.26 (d, 1H, J = 15.4 Hz) | 3.30–3.36 (m, 2 H) |
| H-3 | 7.59 (d, 1 H, J = 15.5 Hz) | 3.14–3.20 (m, 2 H) |
| H-3′ | 7.04 (dd, 1 H, J = 8.4, 0.9 Hz) | 6.98 (dd, 1 H, J = 8.4, 1.2, 0.4 Hz) |
| H-4′ | 7.52 (ddd, 1 H, J = 8.2, 7.2, 1.4 Hz) | 7.45 (ddd, 1 H, J = 8.4, 7.1, 1.6 Hz) |
| H-5′ | 6.95 (ddd, 1 H, J = 8.1, 7.1, 1.0 Hz) | 6.87 (ddd, 1 H, J = 8.2, 7.1, 1.1 Hz) |
| H-6′ | 7.91 (dd, 1 H, J = 8.1, 1.4 Hz) | 7.76 (dd, 1 H, J = 8.1, 1.6 Hz) |
| H-3″ | 7.75 (dd, 1 H, J = 7.8, 1.4 Hz) | 7.55 (dd, 1 H, J = 8.0, 1.2 Hz) |
| H-4″ | 7.38 (t, 1 H, J = 7.5 Hz) | 7.24 (td, 1 H, J = 7.4, 1.3 Hz) |
| H-5″ | 7.28 (td, 1 H, J = 7.9, 1.6 Hz) | 7.09 (ddd, 1 H, J = 7.9, 7.3, 1.9 Hz) |
| H-6″ | 7.66 (dd, 1 H, J = 8.0, 1.0 Hz) | 7.30 (dd, 1 H, J = 8.0, 1.2 Hz) |
| -OH | 12.71 (s, 1 H) | 12.28 (s, 1 H) |
| Carbon | Compound | |||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 5 | 2 | 6 | 3 | 7 | 4 | 8 | |
| C-1 | 193.59 | 205.21 | 193.49 | 204.86 | 193.59 | 205.03 | 192.87 | 205.69 |
| C-2 | 143.86 | 38.36 | 143.70 | 39.75 | 144.12 | 39.82 | 139.10 | 40.23 |
| C-3 | 123.14 | 30.82 | 121.60 | 29.57 | 120.82 | 29.44 | 146.72 | 29.81 |
| C-1′ | 120.03 | 119.37 | 120.02 | 119.30 | 120.05 | 119.34 | 121.39 | 120.60 |
| C-2′ | 163.79 | 162.55 | 163,79 | 162.56 | 163.77 | 162.59 | 162.64 | 161.42 |
| C-3′ | 118.85 | 118.65 | 118.85 | 118.74 | 118.84 | 118.77 | 128.80 | 120.71 |
| C-4′ | 136.75 | 136.52 | 136.80 | 136.60 | 136.71 | 136.62 | 119.55 | 139.08 |
| C-5′ | 119.06 | 119.08 | 119.10 | 119.12 | 119.05 | 119.12 | 110.59 | 110.61 |
| C-6′ | 129.89 | 130.01 | 129.82 | 129.85 | 129.75 | 129.86 | 131.98 | 132.19 |
| C-1″ | 134.90 | 140.11 | 136.84 | 143.21 | 133.64 | 139.83 | 134.44 | 140.50 |
| C-2″ | 126.28 | 124.46 | 131.09 | 127.27 | 132.45 | 131.47 | 129.01 | 128.52 |
| C-3″ | 128.15 | 133.07 | 123.31 | 122.72 | 130.10 | 130.34 | 129.25 | 128.78 |
| C-4″ | 127.90 | 127.83 | 127.63 | 129.58 | 125.40 | 120.24 | 131.43 | 126.56 |
| C-5″ | 131.80 | 128.30 | 130.86 | 130.28 | 140.10 | 130.34 | 129.25 | 128.78 |
| C-6″ | 133.83 | 130.88 | 133.74 | 131.61 | 132.45 | 131.47 | 129.01 | 128.52 |
| Proton | Compound | |
|---|---|---|
| 2 | 6 | |
| H-2 | 7.79–7.85 (m, 1 H) | 3.29–3.35 (m, 2 H) |
| H-3 | 7.64 (d, 1 H, J = 15.5 Hz) | 3.00–3.08 (m, 2 H) |
| H-3′ | 7.04 (d, 1 H, J = 8.1 Hz) | 6.99 (ddd, 1 H, J = 8.4, 1.1, 0.4 Hz) |
| H-4′ | 7.52 (t, 1 H, J = 7.1) | 7.48 (ddd,1 H, J = 8.4, 7.2, 1.7 Hz) |
| H-5′ | 6.96 (t, 1 H, J = 7.2 Hz) | 6.89 (dd, 1 H, J = 8.2, 7.1, 1.1. Hz) |
| H-6′ | 7.92 (d, 1 H, J = 7.1 Hz) | 7.74 (dd, 1 H, J = 8.1, 1.6 Hz) |
| H-2″ | 7.79–7.85 (m, 1 H) | 7.14–7.20 (m, 1 H) |
| H-4″ | 7.54–7.58 (m, 1 H) | 7.33–7.37 (m, 1 H) |
| H-5″ | 7.04 (d, 1 H, J = 8.1 Hz) | 7.14–7.20 (m, 1 H) |
| H-6″ | 7.54–7.58 (m, 1 H) | 7.41–7.42 (m, 1 H) |
| -OH | 12.70 (s, 1 H) | 12.23 (s, 1 H) |
| Proton | Compound | |
|---|---|---|
| 3 | 7 | |
| H-2 | 7.84 (d, 1 H, J = 15.5 Hz) | 3.28–3.34 (m, 2 H) |
| H-3 | 7.64 (d, 1 H, J = 5.5 Hz) | 3.00–3.06 (m, 2 H) |
| H-3′ | 7.03 (dd, 1 H, J = 8.3, 0.8 Hz) | 6.99 (dd, 1 H, J = 8.3, 1.1 Hz) |
| H-4′ | 7.49–7.53 (m, 1 H) | 7.47 (dddd, 1 H, J = 8.6, 7.0,1.7, 0.4 Hz) |
| H-5′ | 6.95 (ddd, 1 H, J = 8.1, 7.1, 1.0 Hz) | 6.87 (ddd, 1 H, J = 8.2, 7.1, 1.1 Hz) |
| H-6′ | 7.90 (dd, 1 H, J = 8.1, 1.4 Hz) | 7.73 (dd, 1 H, J = 8.1, 1.5 Hz) |
| H-2″ | 7.56–7.59 (m, 1 H) | 7.40–7.44 (m, 1 H) |
| H-3″ | 7.49–7.53 (m, 1 H) | 7.11–7.15 (m, 1 H) |
| H-5″ | 7.49–7.53 (m, 1 H) | 7.11–7.15 (m, 1 H) |
| H-6″ | 7.56–7.59 (m, 1 H) | 7.40–7.44 (m, 1 H) |
| -OH | 12.74 (s, 1 H) | 12.23 (s, 1 H) |
| Proton | Compound | |
|---|---|---|
| 4 | 8 | |
| H-2 | 7.56 (d, 1 H, J = 15.5 Hz) | 3.26–3.32 (m, 2 H) |
| H-3 | 7.95 (d, 1 H, J = 15.4 Hz) | 3.02–3.09 (m, 2 H) |
| H-3′ | 6.94 (d, 1 H, J = 8.9 Hz) | 6.88 (d, 1 H, J = 8.9 Hz) |
| H-4′ | 7.57 (dd, 1 H, J = 8.0, 2.3 Hz) | 7.52 (dd, 1 H, J = 8.9,2.4 Hz) |
| H-6′ | 8.01 (d, 1 H, J = 2.3 Hz) | 7.82 (d, 1 H, J = 2.4 Hz) |
| H-2″ | 7.67–7.70 (m, 1 H) | 7.20–7.26 (m, 1 H) |
| H-3″ | 7.43–7.48 (m, 1 H) | 7.29–7.34 (m, 1 H) |
| H-4″ | 7.43–7.48 (m, 1 H) | 7.20–7.26 (m, 1 H) |
| H-5″ | 7.43–7.48 (m, 1 H) | 7.29–7.34 (m, 1 H) |
| H-6″ | 7.67–7.70 (m, 1 H) | 7.20–7.26 (m, 1 H) |
| -OH | 12.74 (s, 1 H) | 12.19 (s, 1 H) |
| Retention Times of Substrates (1–4) and Products (5–8) [min] | |||||||
|---|---|---|---|---|---|---|---|
| 1 | 5 | 2 | 6 | 3 | 7 | 4 | 8 |
| 12.26 | 11.27 | 12.42 | 11.53 | 12.54 | 11.65 | 12.09 | 11.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Łużny, M.; Kaczanowska, D.; Gawdzik, B.; Wzorek, A.; Pawlak, A.; Obmińska-Mrukowicz, B.; Dymarska, M.; Kozłowska, E.; Kostrzewa-Susłow, E.; Janeczko, T. Regiospecific Hydrogenation of Bromochalcone by Unconventional Yeast Strains. Molecules 2022, 27, 3681. https://doi.org/10.3390/molecules27123681
Łużny M, Kaczanowska D, Gawdzik B, Wzorek A, Pawlak A, Obmińska-Mrukowicz B, Dymarska M, Kozłowska E, Kostrzewa-Susłow E, Janeczko T. Regiospecific Hydrogenation of Bromochalcone by Unconventional Yeast Strains. Molecules. 2022; 27(12):3681. https://doi.org/10.3390/molecules27123681
Chicago/Turabian StyleŁużny, Mateusz, Dagmara Kaczanowska, Barbara Gawdzik, Alicja Wzorek, Aleksandra Pawlak, Bożena Obmińska-Mrukowicz, Monika Dymarska, Ewa Kozłowska, Edyta Kostrzewa-Susłow, and Tomasz Janeczko. 2022. "Regiospecific Hydrogenation of Bromochalcone by Unconventional Yeast Strains" Molecules 27, no. 12: 3681. https://doi.org/10.3390/molecules27123681
APA StyleŁużny, M., Kaczanowska, D., Gawdzik, B., Wzorek, A., Pawlak, A., Obmińska-Mrukowicz, B., Dymarska, M., Kozłowska, E., Kostrzewa-Susłow, E., & Janeczko, T. (2022). Regiospecific Hydrogenation of Bromochalcone by Unconventional Yeast Strains. Molecules, 27(12), 3681. https://doi.org/10.3390/molecules27123681






