Yeast Particles Hyper-Loaded with Terpenes for Biocide Applications
Abstract
:1. Introduction
2. Results and Discussion
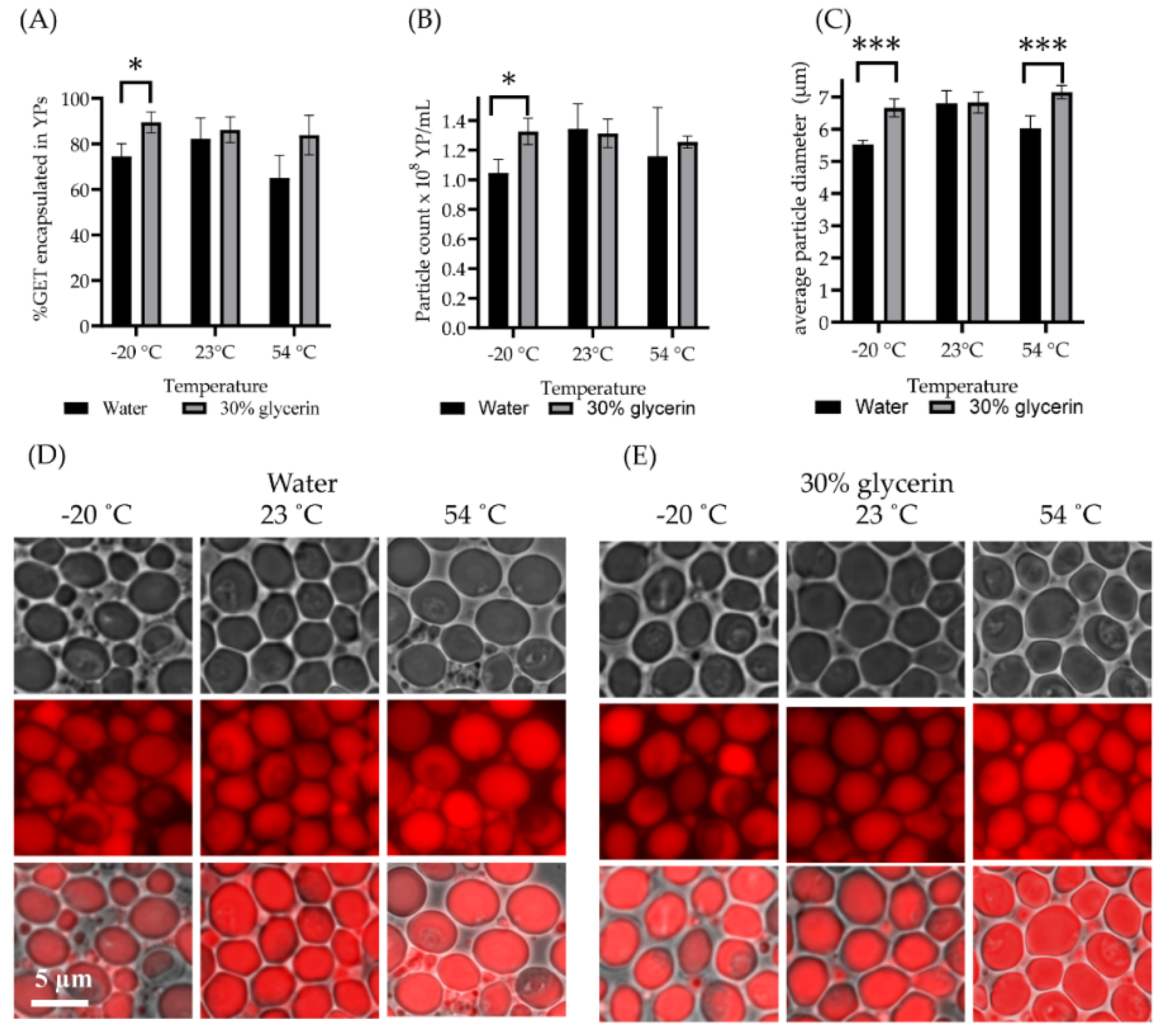
2.1. Preparation and Characterization of Yeast Particles Hyper-Loaded with Terpenes
| Terpene | Log P * | Solubility in Water * (mg/mL) |
|---|---|---|
 Geraniol | 3.56 | 0.686 |
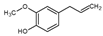 Eugenol | 2.27 | 1.44 |
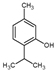 Thymol | 3.3 | 0.9 |
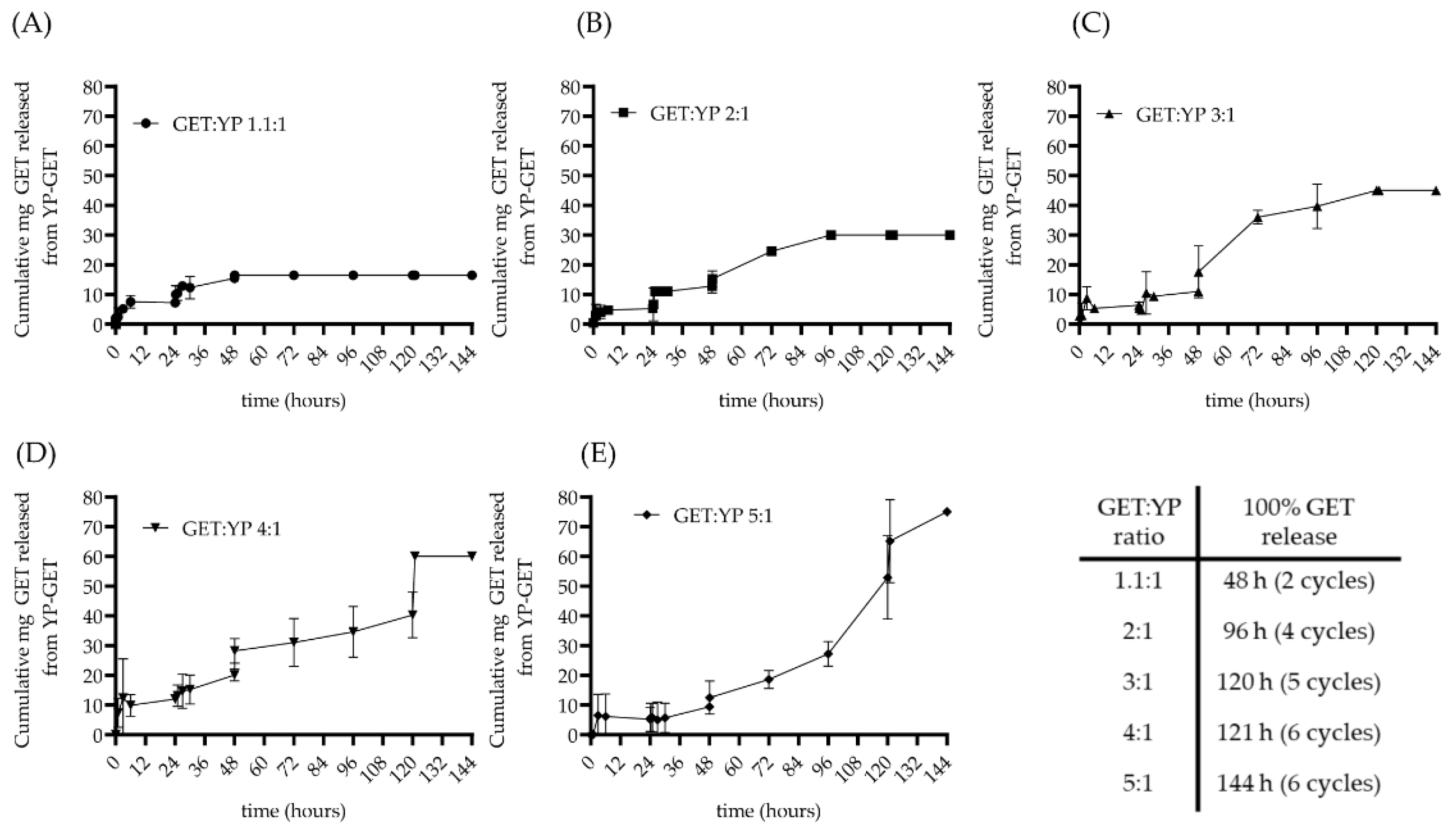
2.2. Terpene Release from Hyper-Loaded YP-GET
2.3. Biological Activity of Hyper-Loaded YP GET
2.4. Optimization of Hyper-Loaded YP-GET Formulations as Dry Granules or Aqueous Suspension Concentrates
3. Materials and Methods
4. Conclusions
5. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Cowan, M.M. Plant Products as Antimicrobial Agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salha, G.B.; Abderrabba, M.; Labidi, J. A Status Review of Terpenes and Their Separation Methods. Rev. Chem. Eng. 2019, 37, 433–447. [Google Scholar] [CrossRef]
- Falleh, H.; Ben Jemaa, M.; Saada, M.; Ksouri, R. Essential Oils: A Promising Eco-Friendly Food Preservative. Food Chem. 2020, 330, 127268. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Kaur, L. Encapsulated Natural Antimicrobials: A Promising Way to Reduce Microbial Growth in Different Food Systems. Food Control 2021, 123, 107678. [Google Scholar] [CrossRef]
- Palaniappan, K.; Holley, R.A. Use of Natural Antimicrobials to Increase Antibiotic Susceptibility of Drug Resistant Bacteria. Int. J. Food Microbiol. 2010, 140, 164–168. [Google Scholar] [CrossRef]
- Cappiello, F.; Loffredo, M.R.; Del Plato, C.; Cammarone, S.; Casciaro, B.; Quaglio, D.; Mangoni, M.L.; Botta, B.; Ghirga, F. The Revaluation of Plant-Derived Terpenes to Fight Antibiotic-Resistant Infections. Antibiotics 2020, 9, 325. [Google Scholar] [CrossRef]
- Gokoglu, N. Novel Natural Food Preservatives and Applications in Seafood Preservation: A Review. J. Sci. Food Agric. 2019, 99, 2068–2077. [Google Scholar] [CrossRef]
- Calo, J.R.; Crandall, P.G.; O’Bryan, C.A.; Ricke, S.C. Essential Oils as Antimicrobials in Food Systems—A Review. Food Control 2015, 54, 111–119. [Google Scholar] [CrossRef]
- Bakry, A.M.; Abbas, S.; Ali, B.; Majeed, H.; Abouelwafa, M.Y.; Mousa, A.; Liang, L. Microencapsulation of Oils: A Comprehensive Review of Benefits, Techniques, and Applications. Compr. Rev. Food Sci. Food Saf. 2016, 15, 143–182. [Google Scholar] [CrossRef]
- Bhalerao, Y.P.; Wagh, S.J. A Review on Thymol Encapsulation and its Controlled Release through Biodegradable Polymer Shells. Int. J. Pharm. Sci. 2018, 2, 4522–4532. [Google Scholar] [CrossRef]
- Gómez, B.; Barba, F.J.; Domínguez, R.; Putnik, P.; Bursać Kovačević, D.; Pateiro, M.; Toldrá, F.; Lorenzo, J.M. Microencapsulation of Antioxidant Compounds through Innovative Technologies and Its Specific Application in Meat Processing. Trends Food Sci. Technol. 2018, 82, 135–147. [Google Scholar] [CrossRef]
- Saifullah, M.; Shishir, M.; Ferdowsi, R.; Rahman, M.; Van Vuong, Q. Micro and Nano Encapsulation, Retention and Controlled Release of Flavor and Aroma Compounds: A Critical Review. Trends Food Sci. Technol. 2019, 86, 230–251. [Google Scholar] [CrossRef]
- Soto, E.R.; Ostroff, G.R. Characterization of Multilayered Nanoparticles Encapsulated in Yeast Cell Wall Particles for DNA. Bioconjug. Chem. 2008, 19, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Aouadi, M.; Tesz, G.J.; Nicoloro, S.M.; Wang, M.; Chouinard, M.; Soto, E.R.; Ostroff, G.R.; Czech, M.P. Orally Delivered siRNA Targeting Macrophage Map4k4 Suppresses Systemic Inflammation. Nature 2009, 458, 1180–1184. [Google Scholar] [CrossRef] [Green Version]
- Soto, E.R.; Ostroff, G.R. Glucan Particles as Carriers of Nanoparticles for Macrophage-Targeted Delivery. In Nanomaterials for Biomecidine; American Chemical Society: Washington, DC, USA, 2012; pp. 57–79. [Google Scholar] [CrossRef]
- Soto, E.R.; Caras, A.C.; Kut, L.; Castle, M.; Ostroff, G.R. Glucan Particles for Macrophage Targeted Delivery of Nanoparticles. J. Drug Deliv. 2012, 2012, 143524. [Google Scholar] [CrossRef] [Green Version]
- Soto, E.R.; O’Connell, O.; Dikengil, F.; Peters, P.J.; Clapham, P.R.; Ostroff, G.R. Targeted Delivery of Glucan Particle Encapsulated Gallium Nanoparticles Inhibits HIV Growth in Human Macrophages. J. Drug Deliv. 2016, 2016, 8520629. [Google Scholar] [CrossRef] [Green Version]
- Mirza, Z.; Soto, E.R.; Dikengil, F.; Levitz, S.M.; Ostroff, G.R. Beta-Glucan Particles as Vaccine Adjuvant Carriers. In Vaccines for Invasive Fungal Infections; Humana Press: New York, NY, USA, 2017; pp. 143–157. [Google Scholar] [CrossRef]
- Soto, E.R.; Rus, F.; Ostroff, G.R. Yeast Cell Wall Particle Encapsulation of Pro-Terpene Payloads. 2019. Available online: https://briefs.techconnect.org/papers/yeast-cell-wall-particle-encapsulation-of-pro-terpene-payloads/ (accessed on 5 November 2021).
- Mirza, Z.; Soto, E.R.; Hu, Y.; Nguyen, T.T.; Koch, D.; Aroian, R.V.; Ostroff, G.R. Anthelmintic Activity of Yeast Particle-Encapsulated Terpenes. Molecules 2020, 25, 2598. [Google Scholar] [CrossRef]
- Soto, E.R.; Rus, F.; Li, H.; Garceau, C.; Chicca, J.; Elfawal, M.; Gazzola, D.; Nielsen, M.K.; Urban, J.F., Jr.; Aroian, R.V.; et al. Yeast Particle Encapsulation of Scaffolded Terpene Compounds for Controlled Terpene Release. Foods 2021, 10, 1207. [Google Scholar] [CrossRef]
- Franklin, L.; Ostroff, G.; Harman, G. Terpene-Containing Compositions and Methods of Making and Using Them. U.S. Patent No. 10,667,512, 2 June 2020. [Google Scholar]
- Franklin, L.; Ostroff, G. Compositions and Methods Comprising Terpenes or Terpene Mixtures Selected from Thymol, Eugenol, Geraniol, Citral, and L-Carvone. U.S. Patent No. 9,439,416, 13 September 2016. [Google Scholar]
- Franklin, L.; Ostroff, G. Nematicidal Compositions and Methods of Using Them. U.S. Patent No. 9,655,360, 23 May 2017. [Google Scholar]
- Franklin, L.; Ostroff, G. Nematicidal Compositions and Methods of Using Them. U.S. Patent No. 10,004,229, 26 June 2018. [Google Scholar]
- Franklin, L.; Ostroff, G. Compositions and Methods Comprising Terpenes or Terpene Mixtures Selected from Thymol, Eugenol, Geraniol, Citral and L-Carvone. U.S. Patent No. 10,258,033, 26 April 2019. [Google Scholar]
- Franklin, L.; Ostroff, G. Compositions Containing a Hollow Glucan Particle or a Cell Wall Particle Encapsulating a Terpene Component, Methods of Making and Using Them. U.S. Patent No. 10,638,750, 5 May 2020. [Google Scholar]
- Young, S.H.; Ostroff, G.R.; Zeidler-Erdely, P.C.; Roberts, J.R.; Antonini, J.M.; Castranova, V.A. Comparison of the Pulmonary Inflammatory Potential of Different Components of Yeast Cell Wall. J. Toxicol. Environ. Health Part A 2007, 70, 1116–1124. [Google Scholar] [CrossRef]
- National Library of Medicine. PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/ (accessed on 11 March 2022).
- Klis, F.M.; Boorsma, A.; De Groot, P.W.J. Cell Wall Construction in Saccharomyces cerevisiae. Yeast 2006, 23, 185–202. [Google Scholar] [CrossRef]
- Shaw, J.A.; Mol, P.C.; Bowers, B.; Silverman, S.J.; Valdivieso, M.H.; Duran, A.; Cabib, E. The Function of Chitin Synthases 2 and 3 in the Saccharomyces cerevisiae Cell Cycle. J. Cell Biol. 1991, 114, 111–123. [Google Scholar] [CrossRef] [Green Version]
- Touhami, A.; Nysten, B.; Dufrêne, Y.F. Nanoscale Mapping of the Elasticity of Microbial Cells by Atomic Force Microscopy. Langmuir 2003, 19, 4539–4543. [Google Scholar] [CrossRef]
- Stenson, J.D.; Hartley, P.; Wang, C.; Thomas, C.R. Determining the Mechanical Properties of Yeast Cell Walls. Biotechnol. Prog. 2011, 27, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Ivanovska, I.L.; De Pablo, P.J.; Ibarra, B.; Sgalari, G.; MacKintosh, F.C.; Carrascosa, J.L.; Schmidt, C.F.; Wuite, G.J.L. Bacteriophage Capsids: Tough Nanoshells with Complex Elastic Properties. Proc. Natl. Acad. Sci. USA 2004, 101, 7600–7605. [Google Scholar] [CrossRef] [Green Version]
- Miron, D.; Battisti, F.; Silva, F.K.; Lana, A.D.; Pippi, B.; Casanova, B.; Gnoatto, S.; Fuentefria, A.; Mayorga, P.; Schapoval, E. Antifungal Activity and Mechanism of Action of Monoterpenes against Dermatophytes and Yeasts. Rev. Bras. Farmacogn. 2014, 24, 660–667. [Google Scholar] [CrossRef]
- Oliveira, E.; Nogueira, J.; Aguiar Campolina, G.; Batista, L.R.; Alves, E.; Rodrigues Silva Caetano, A.; Magalhães Brandão, R.; Nelson, D.L.; Das Graças Cardoso, M. Mechanism of Action of Various Terpenes and Phenylpropanoids against Escherichia coli and Staphylococcus aureus. FEMS Microbiol. Lett. 2021, 368, fnab052. [Google Scholar] [CrossRef]
- Kunicka, A.; Kalemba, D. Antibacterial and Antifungal Properties of Essential Oils. Curr. Med. Chem. 2003, 10, 813–829. [Google Scholar] [CrossRef]
- Saad, N.Y.; Muller, C.D.; Lobstein, A. Major Bioactivities and Mechanism of Action of Essential Oils and Their Components. Flavour Fragr. J. 2013, 28, 269–279. [Google Scholar] [CrossRef]
- Engineering Toolbox: Glycerine Boiling and Freezing Points. 2010. Available online: https://www.engineeringtoolbox.com/glycerine-boiling-freezing-points-d_1590.html (accessed on 2 August 2021).
- Rasband, W.S. ImageJ (1997–2014); U.S. National Institutes of Health: Bethesda, MD, USA. Available online: https://imagej.nih.gov/ij/ (accessed on 16 August 2021).
- Sultanbawa, Y.; Cusack, A.; Currie, M.; Davis, C. An Innovative Microplate Assay to Facilitate the Detection of Antimicrobial Activity in Plant Extracts. J. Rapid Methods Autom. Microbiol. 2009, 17, 519–534. [Google Scholar] [CrossRef]
- Gillum, A.M.; Tsay, E.Y.H.; Kirsch, D.R. Isolation of the Candida albicans Gene for Orotidine-5’-Phosphate Decarboxylase by Complementation of S. Cerevisiae Ura3 and E. Coli PyrF Mutations. Mol. Gen. Genet. 1984, 198, 179–182. [Google Scholar] [CrossRef]




| Sample | Minimum Inhibitory Concentration (MIC 75%) | ||
|---|---|---|---|
| E. coli | S. aureus | C. albicans | |
| Empty YPs | Not active | Not active | Not active |
| YP-GET 1.1:1 | 0.156 ± 0.05 | 0.313 ± 0 | 0.156 ± 0.026 |
| YP-GET 2:1 | 0.156 ± 0.06 | 0.313 ± 0 | 0.156 ± 0.026 |
| YP-GET 3:1 | 0.156 ± 0.05 | 0.313 ± 0 | 0.156 ± 0.026 |
| YP-GET 4:1 | 0.156 ± 0.03 | 0.313 ± 0 | 0.156 ± 0.039 |
| YP-GET 5:1 | 0.156 ± 0.05 | 0.313 ± 0 | 0.156 ± 0.030 |
| Unencapsulated GET | 0.625 ± 0 **** | 1.250 ± 0.318 * | 0.625 ± 0 *** |
| Ampicillin | 0.008 **** | <0.00025 **** | - |
| Fluconazole | - | - | <2 **** |
| GET:YP Ratio | Materials to Produce 100 g YP-GET | Final Composition | |||
|---|---|---|---|---|---|
| g YP | mL (g) Water | g GET | %YP | %GET | |
| 1.1:1 | 15 | 68.5 | 16.5 | 15 | 16.5 |
| 2:1 | 15 | 55 | 30 | 15 | 30 |
| 3:1 | 15 | 40 | 45 | 15 | 45 |
| 4:1 | 15 | 25 | 60 | 15 | 60 |
| 5:1 | 15 | 10 | 75 | 15 | 75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soto, E.R.; Rus, F.; Ostroff, G.R. Yeast Particles Hyper-Loaded with Terpenes for Biocide Applications. Molecules 2022, 27, 3580. https://doi.org/10.3390/molecules27113580
Soto ER, Rus F, Ostroff GR. Yeast Particles Hyper-Loaded with Terpenes for Biocide Applications. Molecules. 2022; 27(11):3580. https://doi.org/10.3390/molecules27113580
Chicago/Turabian StyleSoto, Ernesto R., Florentina Rus, and Gary R. Ostroff. 2022. "Yeast Particles Hyper-Loaded with Terpenes for Biocide Applications" Molecules 27, no. 11: 3580. https://doi.org/10.3390/molecules27113580
APA StyleSoto, E. R., Rus, F., & Ostroff, G. R. (2022). Yeast Particles Hyper-Loaded with Terpenes for Biocide Applications. Molecules, 27(11), 3580. https://doi.org/10.3390/molecules27113580







