New Amphiphilic Ionic Liquids for the Demulsification of Water-in-Heavy Crude Oil Emulsion
Abstract
:1. Introduction
2. Results and Discussion
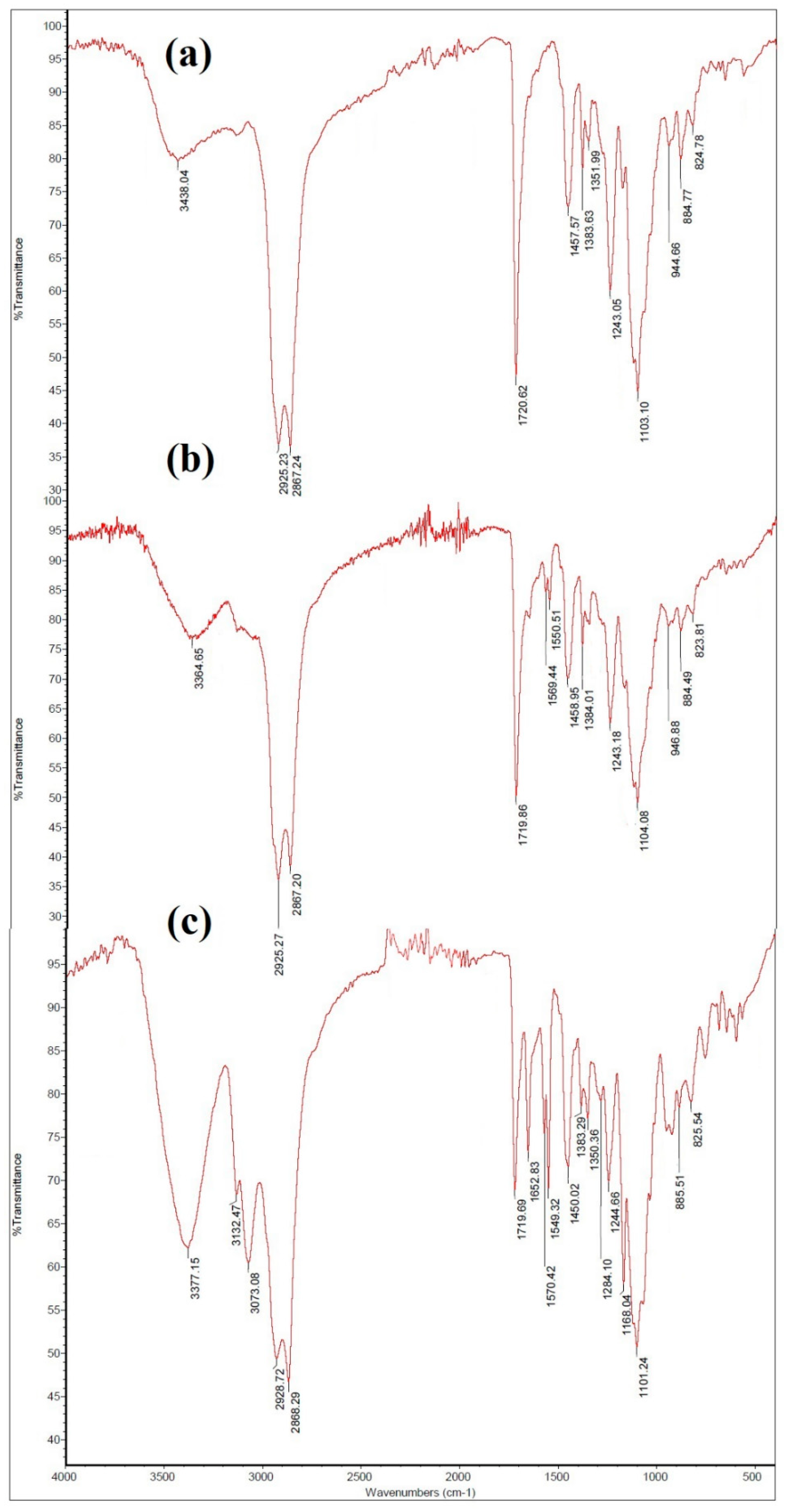
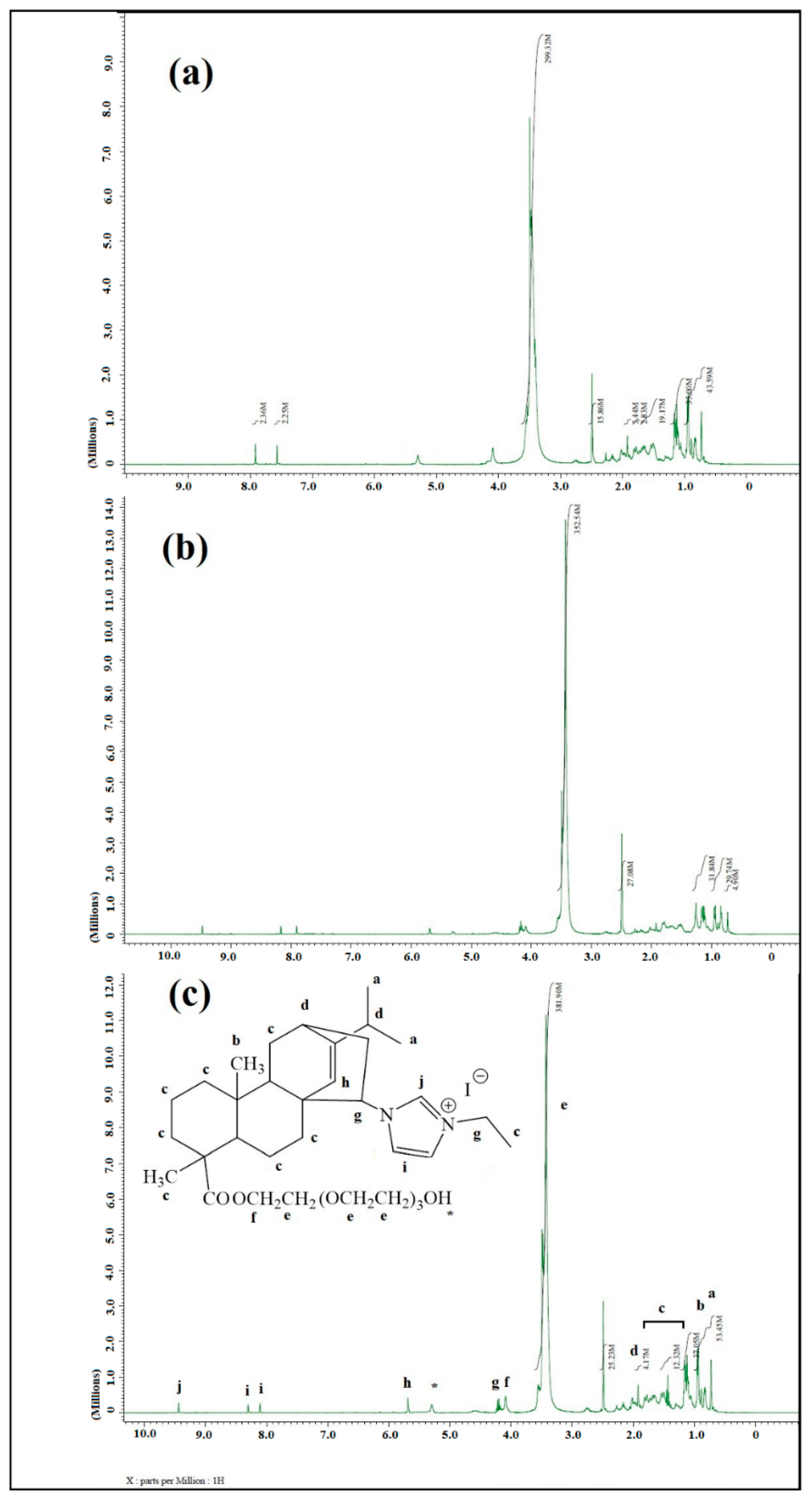
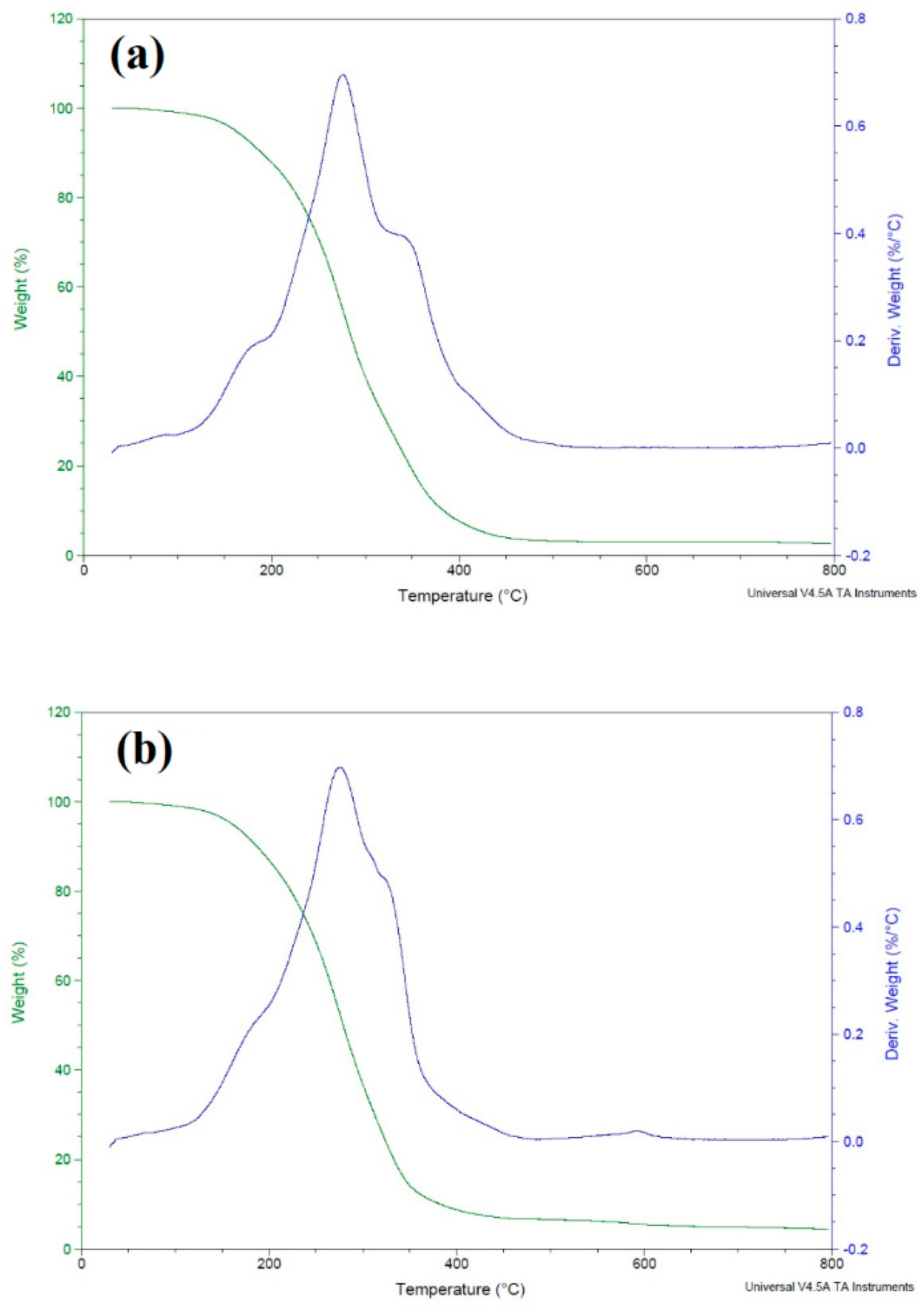
2.1. Characterization of ILs and PILs
2.2. Solubility and Surface Activity of ATHI-IL and ATEI-IL
2.3. Demulsification Efficiency of ATEI-IL and ATHI-IL
3. Materials and Methods
3.1. Materials
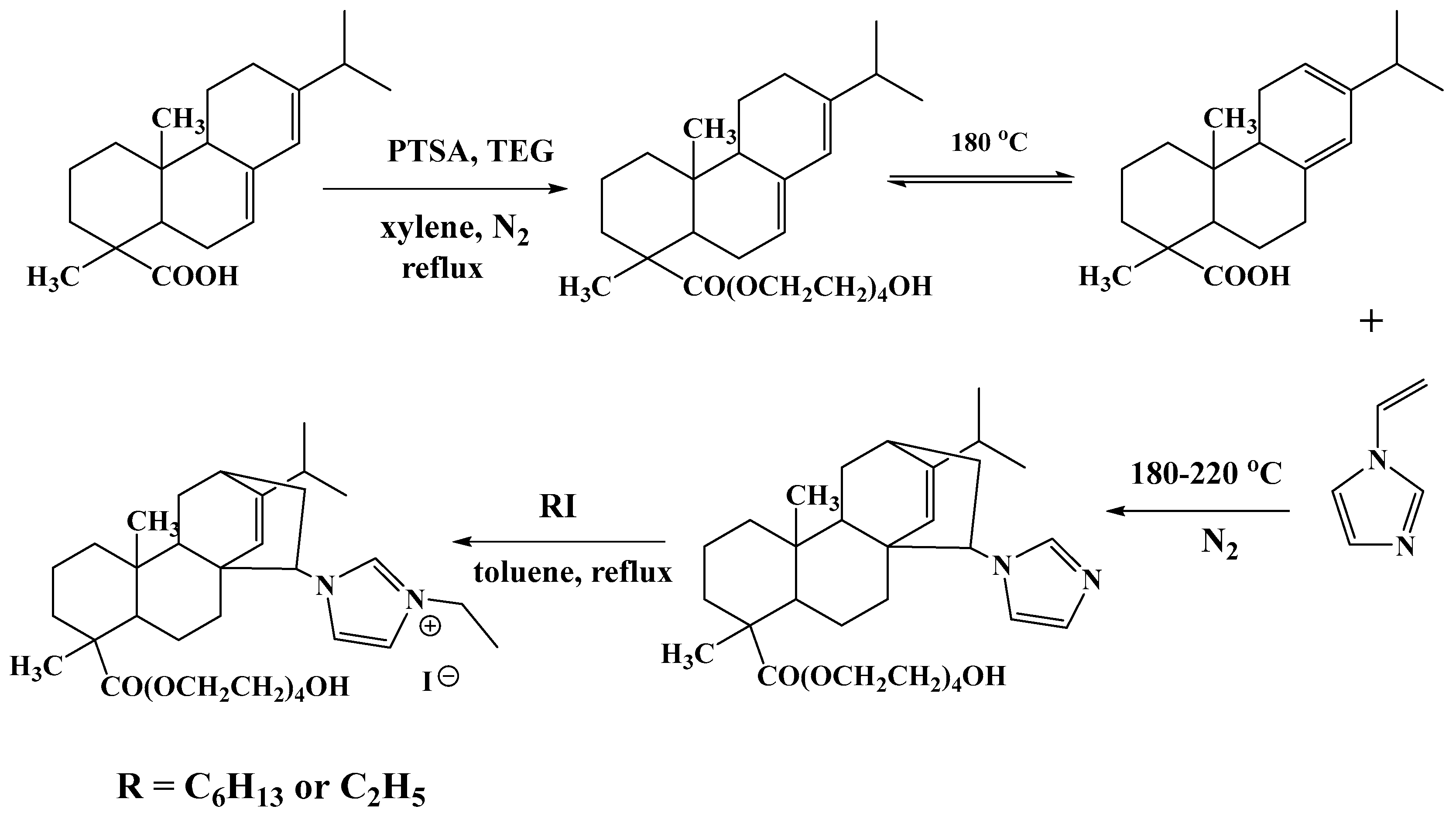
3.2. Synthesis of Amphiphilic Ionic Liquids
3.3. Bottle Test Method
3.4. Characterization
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Abbreviations
| AA | Abietic acid |
| AATG | ester produced from raction of AA with TEG |
| AILs | amphiphilic ionic liquids |
| ATEI-IL | AIL produced from raction of ATI with EI |
| ATHI-IL | AIL produced from raction of ATI with HI |
| ATI | adduct produced from reaction of AATG with VIM |
| DTG | differential thermal analysis |
| EI | ethyl iodide |
| HI | hexyl iodide |
| PTSA | p-toluene sulfonic acid |
| RSN | relative solubility number |
| TEG | tetraethylene glycol |
| TGA | thermal gravimetrical analysis |
| VIM | 1-vinylimidazole |
| W/O | water-in-oil |
References
- Santos, R.L.; Elvio Filho, B.; Dourado, R.S.; Santos, A.F.; Borges, G.R.; Dariva, C.; Santana, C.C.; Franceschi, E.; Santos, D. Study on the use of aprotic ionic liquids as potential additives for crude oil upgrading, emulsion inhibition, and demulsification. Fluid Phase Equilibria 2019, 489, 8–15. [Google Scholar] [CrossRef]
- Umar, A.A.; Saaid, I.B.M.; Sulaimon, A.A.; Pilus, R.B.M. A review of petroleum emulsions and recent progress on water-in-crude oil emulsions stabilized by natural surfactants and solids. J. Pet. Sci. Eng. 2018, 165, 673–690. [Google Scholar] [CrossRef]
- Atta, A.M.; Al-Lohedan, H.A.; Ezzat, A.O. Synthesis and application of geminal dicationic ionic liquids and poly (ionic liquids) combined imidazolium and pyridinium cations as demulsifiers for petroleum crude oil saline water emulsions. J. Mol. Liq. 2021, 325, 115264. [Google Scholar] [CrossRef]
- Abdulraheim, A.M. Green polymeric surface active agents for crude oil demulsification. J. Mol. Liq. 2018, 271, 329–341. [Google Scholar] [CrossRef]
- Feitosa, F.X.; Alves, R.S.; de Sant’Ana, H.B. Synthesis and application of additives based on cardanol as demulsifier for water-in-oil emulsions. Fuel 2019, 245, 21–28. [Google Scholar] [CrossRef]
- Srivastava, A.; Karthick, S.; Jayaprakash, K.; Sen, A. Droplet demulsification using ultralow voltage-based electrocoalescence. Langmuir 2018, 34, 1520–1527. [Google Scholar] [CrossRef]
- Zolfaghari, R.; Fakhru’l-Razi, A.; Abdullah, L.C.; Elnashaie, S.S.; Pendashteh, A. Demulsification techniques of water-in-oil and oil-in-water emulsions in petroleum industry. Sep. Purif. Technol. 2016, 170, 377–407. [Google Scholar] [CrossRef]
- Abdullah, M.; Al-Lohedan, H.A.; Atta, A.M. Fabrication of new demulsifiers employing the waste polyethylene terephthalate and their demulsification efficiency for heavy crude oil emulsions. Molecules 2021, 26, 589. [Google Scholar] [CrossRef]
- Fan, Y.; Simon, S.; Sjöblom, J. Chemical destabilization of crude oil emulsions: Effect of nonionic surfactants as emulsion inhibitors. Energy Fuels 2009, 23, 4575–4583. [Google Scholar] [CrossRef]
- Manshad, A.K.; Rezaei, M.; Moradi, S.; Nowrouzi, I.; Mohammadi, A.H. Wettability alteration and interfacial tension (IFT) reduction in enhanced oil recovery (EOR) process by ionic liquid flooding. J. Mol. Liq. 2017, 248, 153–162. [Google Scholar] [CrossRef]
- Hezave, A.Z.; Dorostkar, S.; Ayatollahi, S.; Nabipour, M.; Hemmateenejad, B. Investigating the effect of ionic liquid (1-dodecyl-3-methylimidazolium chloride ([C12mim][Cl])) on the water/oil interfacial tension as a novel surfactant. Colloids Surf. A Physicochem. Eng. Asp. 2013, 421, 63–71. [Google Scholar] [CrossRef]
- Liu, J.; Wang, H.; Li, X.; Jia, W.; Zhao, Y.; Ren, S. Recyclable magnetic graphene oxide for rapid and efficient demulsification of crude oil-in-water emulsion. Fuel 2017, 189, 79–87. [Google Scholar] [CrossRef] [Green Version]
- Xu, L.; Chen, Y.; Liu, N.; Zhang, W.; Yang, Y.; Cao, Y.; Lin, X.; Wei, Y.; Feng, L. Breathing demulsification: A three-dimensional (3D) free-standing superhydrophilic sponge. ACS Appl. Mater. Interfaces 2015, 7, 22264–22271. [Google Scholar] [CrossRef]
- González, M.M.; Blanco-Tirado, C.; Combariza, M.Y. Nanocellulose as an inhibitor of water-in-crude oil emulsion formation. Fuel 2020, 264, 116830. [Google Scholar] [CrossRef]
- Hassanshahi, N.; Hu, G.; Li, J. Application of ionic liquids for chemical demulsification: A review. Molecules 2020, 25, 4915. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, M.M.; Al-Lohedan, H.A.; Faqihi, N.A. Efficacy of Curcumin-based amphiphilic ionic liquids towards the demulsification of water-in-heavy crude oil emulsions. Colloids Surf. A Physicochem. Eng. Asp. 2021, 628, 127320. [Google Scholar] [CrossRef]
- Ezzat, A.O.; Tawfeek, A.M.; Al-Lohedan, H.A. Synthesis and application of novel gemini pyridinium ionic liquids as demulsifiers for arabian heavy crude oil emulsions. Colloids Surf. A Physicochem. Eng. Asp. 2022, 634, 127961. [Google Scholar] [CrossRef]
- Ezzat, A.O.; Atta, A.M.; Al-Lohedan, H.A. Demulsification of stable seawater/Arabian heavy crude oil emulsions using star-like tricationic pyridinium ionic liquids. Fuel 2021, 304, 121436. [Google Scholar] [CrossRef]
- Elmobarak, W.F.; Almomani, F. Evaluation of the efficiency of ionic liquids in the demulsification of oil-in-water emulsions. Environ. Technol. Innov. 2021, 24, 102003. [Google Scholar] [CrossRef]
- Abdullah, M.M.; Al-Lohedan, H.A. Demulsification of Arabian Heavy Crude Oil Emulsions Using Novel Amphiphilic Ionic Liquids Based on Glycidyl 4-Nonylphenyl Ether. Energy Fuels 2019, 33, 12916–12923. [Google Scholar] [CrossRef]
- Kang, S.; Zhang, J.; Yuan, Y. Abietic acid attenuates IL-1β-induced inflammation in human osteoarthritis chondrocytes. Int. Immunopharmacol. 2018, 64, 110–115. [Google Scholar] [CrossRef]
- Alihosseini, F. Plant-based compounds for antimicrobial textiles. In Antimicrobial Textiles; Elsevier: Amsterdam, The Netherlands, 2016; pp. 155–195. [Google Scholar]
- Yıldız, A.; Değirmencioğlu, M. Synthesis of silver abietate as an antibacterial agent for textile applications. Bioinorg. Chem. Appl. 2015, 2015, 215354. [Google Scholar] [CrossRef] [PubMed]
- Yao, F.; Zhang, D.; Zhang, C.; Yang, W.; Deng, J. Preparation and application of abietic acid-derived optically active helical polymers and their chiral hydrogels. Bioresour. Technol. 2013, 129, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Al-Sabagh, A.; Khidr, T.; Atta, A. Effect of abietic acid derivative surfactants on calorific value and flow of crude oil and some distillates. Pet. Sci. Technol. 2002, 20, 693–711. [Google Scholar] [CrossRef]
- Atta, A.M.; Al-Lohedan, H.A.; Al-Hussain, S.A. Functionalization of magnetite nanoparticles as oil spill collector. Int. J. Mol. Sci. 2015, 16, 6911–6931. [Google Scholar] [CrossRef] [Green Version]
- Villanueva, M.; Coronas, A.; Garcia, J.; Salgado, J. Thermal stability of ionic liquids for their application as new absorbents. Ind. Eng. Chem. Res. 2013, 52, 15718–15727. [Google Scholar] [CrossRef]
- Wang, H.; Wang, J.; Zhang, S.; Xuan, X. Structural effects of anions and cations on the aggregation behavior of ionic liquids in aqueous solutions. J. Phys. Chem. B 2008, 112, 16682–16689. [Google Scholar] [CrossRef] [PubMed]
- Schwuger, M.J.M. Rosen: Surfactants and Interfacial Phenomena, Second Edition, John Wiley & Sons Ltd., New York, Chichester, Brisbane, Toronto, Singapore 1989. 431 Seiten, Preis: £ 36.85. Ber. Bunsenges. Phys. Chem. 1990, 94, 796. [Google Scholar]
- Dorbritz, S.; Ruth, W.; Kragl, U. Investigation on aggregate formation of ionic liquids. Adv. Synth. Catal. 2005, 347, 1273–1279. [Google Scholar] [CrossRef]
- Kolbeck, C.; Lehmann, J.; Lovelock, K.; Cremer, T.; Paape, N.; Wasserscheid, P.; Froba, A.; Maier, F.; Steinruck, H.-P. Density and surface tension of ionic liquids. J. Phys. Chem. B 2010, 114, 17025–17036. [Google Scholar] [CrossRef]
- Blesic, M.; Marques, M.H.; Plechkova, N.V.; Seddon, K.R.; Rebelo, L.P.N.; Lopes, A. Self-aggregation of ionic liquids: Micelle formation in aqueous solution. Green Chem. 2007, 9, 481–490. [Google Scholar] [CrossRef]
- Wei, Y.; Wang, F.; Zhang, Z.; Ren, C.; Lin, Y. Micellization and thermodynamic study of 1-alkyl-3-methylimidazolium tetrafluoroborate ionic liquids in aqueous solution. J. Chem. Eng. Data 2014, 59, 1120–1129. [Google Scholar] [CrossRef]
- Atta, A.M.; Abdullah, M.M.; Al-Lohedan, H.A.; Ezzat, A.O. Demulsification of heavy crude oil using new nonionic cardanol surfactants. J. Mol. Liq. 2018, 252, 311–320. [Google Scholar] [CrossRef]
- Belhaj, A.F.; Elraies, K.A.; Mahmood, S.M.; Zulkifli, N.N.; Akbari, S.; Hussien, O.S. The effect of surfactant concentration, salinity, temperature, and pH on surfactant adsorption for chemical enhanced oil recovery: A review. J. Pet. Explor. Prod. Technol. 2020, 10, 125–137. [Google Scholar] [CrossRef] [Green Version]
- Hezave, A.Z.; Dorostkar, S.; Ayatollahi, S.; Nabipour, M.; Hemmateenejad, B. Dynamic interfacial tension behavior between heavy crude oil and ionic liquid solution (1-dodecyl-3-methylimidazolium chloride ([C12mim][Cl]+ distilled or saline water/heavy crude oil)) as a new surfactant. J. Mol. Liq. 2013, 187, 83–89. [Google Scholar] [CrossRef]
- Al-Sabagh, A.; Elsharaky, E.; El-Tabey, A.E. Demulsification performance and the relative solubility number (RSN) of modified poly (maleic anhydride-alt-1-dodecene) on naturally asphaltenic crude oil emulsion. J. Dispers. Sci. Technol. 2017, 38, 288–295. [Google Scholar] [CrossRef]
- Ezzat, A.O.; Atta, A.M.; Al-Lohedan, H.A.; Aldalbahi, A. New amphiphilic pyridinium ionic liquids for demulsification of water Arabic heavy crude oil emulsions. J. Mol. Liq. 2020, 312, 113407. [Google Scholar] [CrossRef]
- Abdullah, M.M.; Al-Lohedan, H.A. Novel amphiphilic gemini ionic liquids based on consumed polyethylene terephthalate as demulsifiers for Arabian heavy crude oil. Fuel 2020, 266, 117057. [Google Scholar] [CrossRef]
- Das, P.; Manna, S.; Pandey, J.K. Advances in Oil-Water Separation: A Complete Guide for Physical, Chemical, and Biochemical Processes; Elsevier: Amsterdam, The Netherlands, 2022. [Google Scholar]




| Compound | cmc (mM) | (−∂γ/∂ln c)T | γcmc (mN/m) | Γmax × 10−6 (mol/m2) | Amin (nm2/Molecule) | RSN |
|---|---|---|---|---|---|---|
| ATHI-IL | 0.051 | 9.88 | 37.6 | 2.52 | 0.658 | 15.4 |
| ATEI-IL | 0.06 | 9.71 | 40.6 | 2.23 | 0.747 | 16.1 |
| AIL | Crude Oil: Brine Volumetric Ratio | ||||||
|---|---|---|---|---|---|---|---|
| Dose (ppm) | 90:10 | 70:30 | 50:50 | ||||
| DE (%) | Time (h) | DE (%) | Time (h) | DE (%) | Time (h) | ||
| ATEI-IL | 250 | 100 | 4.5 | 100 | 6 | 36 | 7 |
| 500 | 100 | 4 | 100 | 5 | 40 | 6 | |
| 1000 | 100 | 3.5 | 100 | 4.5 | 88 | 6 | |
| ATHI-IL | 250 | 100 | 5 | 93.33 | 6 | 28 | 6 |
| 500 | 100 | 5 | 100 | 5.5 | 40 | 5.5 | |
| 1000 | 100 | 4 | 93.33 | 5.5 | 72 | 5 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdullah, M.M.S.; Ezzat, A.O.; Al-Lohedan, H.A.; Aldalbahi, A.; Atta, A.M. New Amphiphilic Ionic Liquids for the Demulsification of Water-in-Heavy Crude Oil Emulsion. Molecules 2022, 27, 3238. https://doi.org/10.3390/molecules27103238
Abdullah MMS, Ezzat AO, Al-Lohedan HA, Aldalbahi A, Atta AM. New Amphiphilic Ionic Liquids for the Demulsification of Water-in-Heavy Crude Oil Emulsion. Molecules. 2022; 27(10):3238. https://doi.org/10.3390/molecules27103238
Chicago/Turabian StyleAbdullah, Mahmood M. S., Abdelrahman O. Ezzat, Hamad A. Al-Lohedan, Ali Aldalbahi, and Ayman M. Atta. 2022. "New Amphiphilic Ionic Liquids for the Demulsification of Water-in-Heavy Crude Oil Emulsion" Molecules 27, no. 10: 3238. https://doi.org/10.3390/molecules27103238
APA StyleAbdullah, M. M. S., Ezzat, A. O., Al-Lohedan, H. A., Aldalbahi, A., & Atta, A. M. (2022). New Amphiphilic Ionic Liquids for the Demulsification of Water-in-Heavy Crude Oil Emulsion. Molecules, 27(10), 3238. https://doi.org/10.3390/molecules27103238






