Synthesis, Biological Evaluation, and Docking Studies of Antagonistic Hydroxylated Arecaidine Esters Targeting mAChRs
Abstract
:1. Introduction
2. Results and Discussion
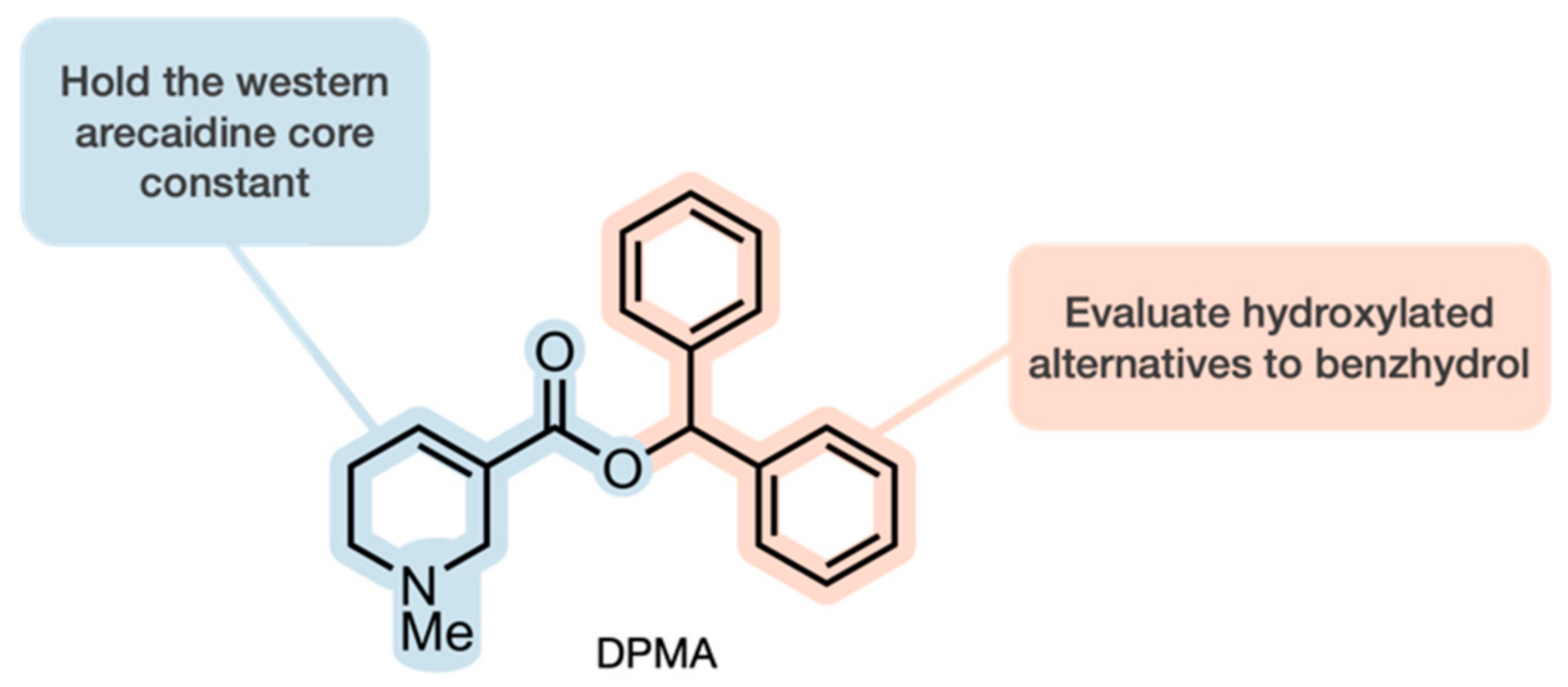
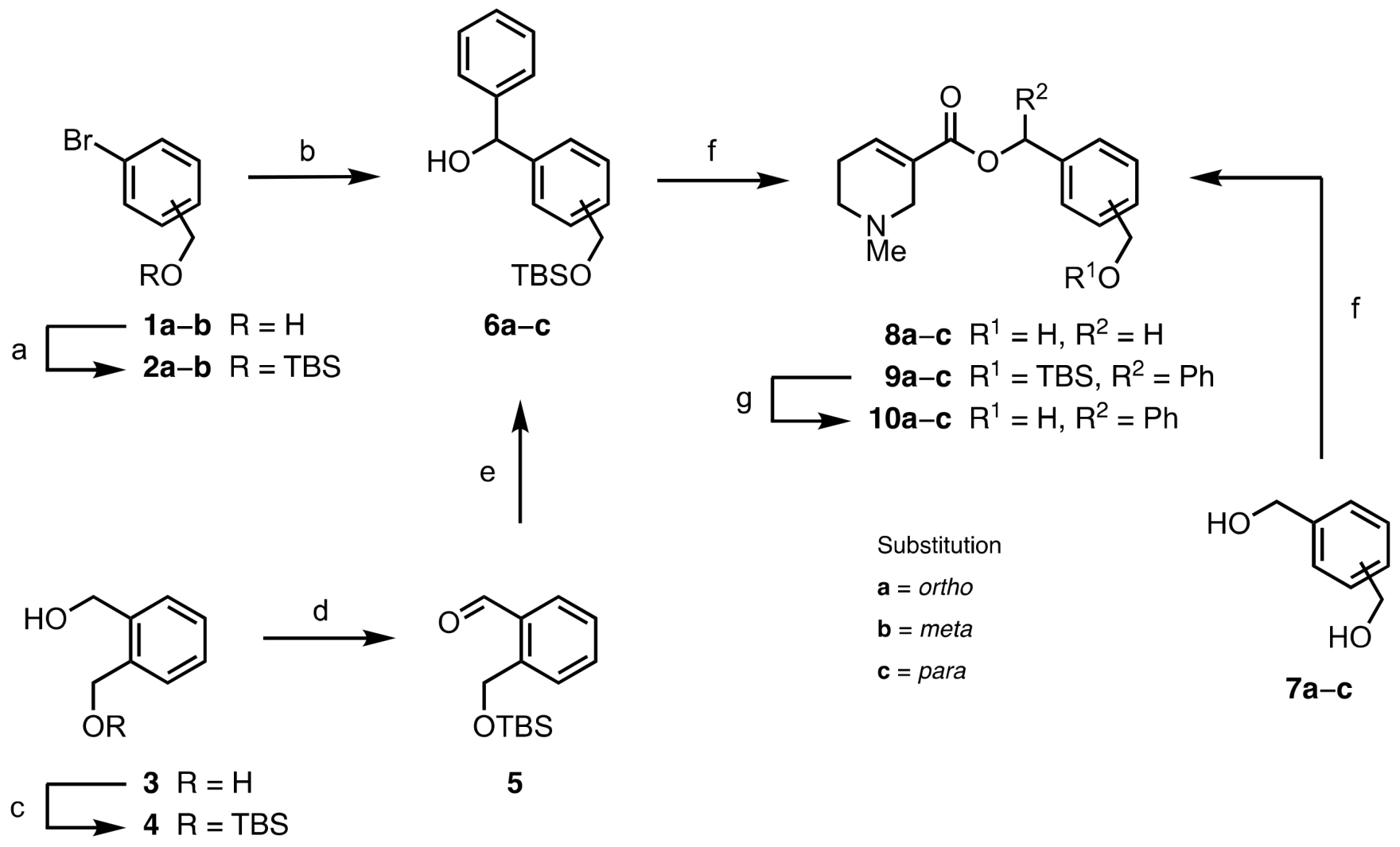
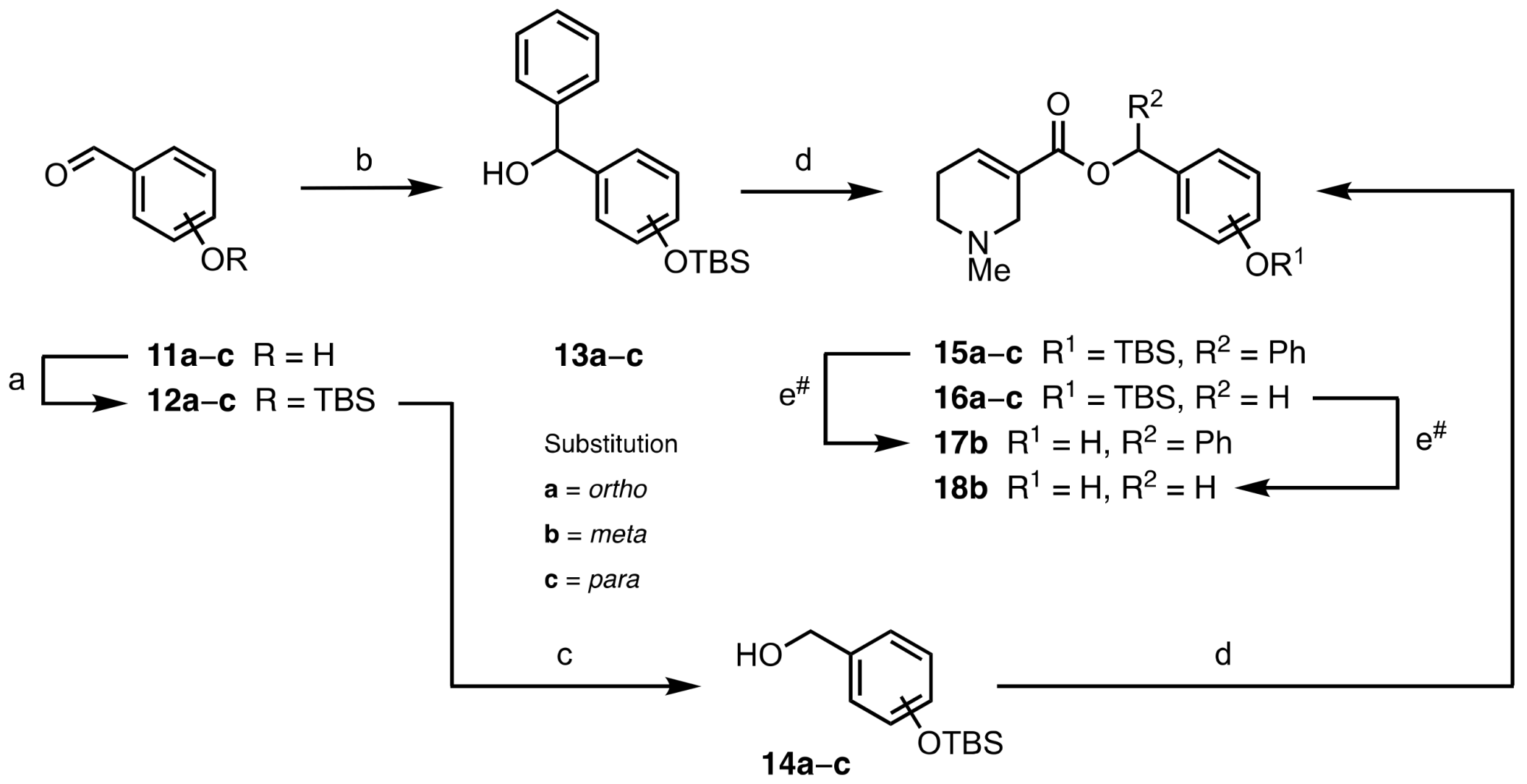
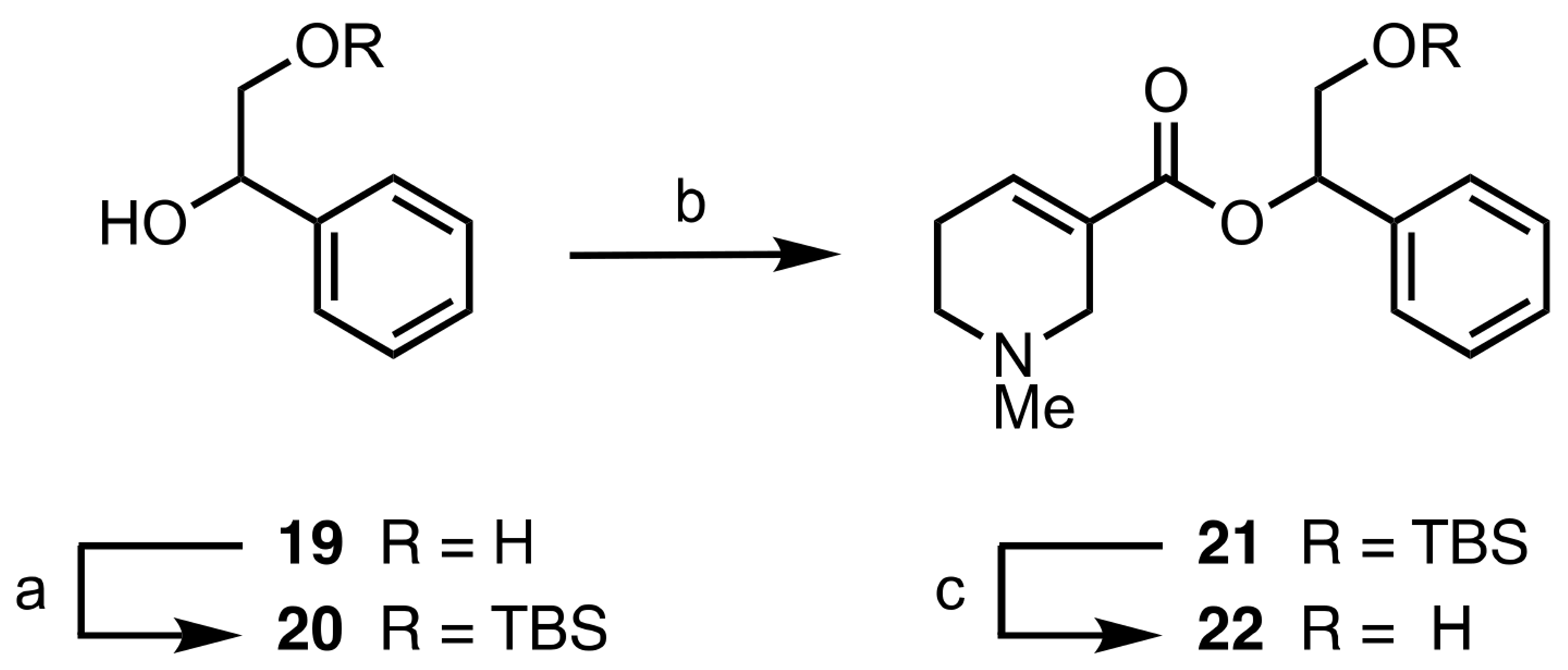
2.1. Chemistry
2.2. Physicochemical Properties
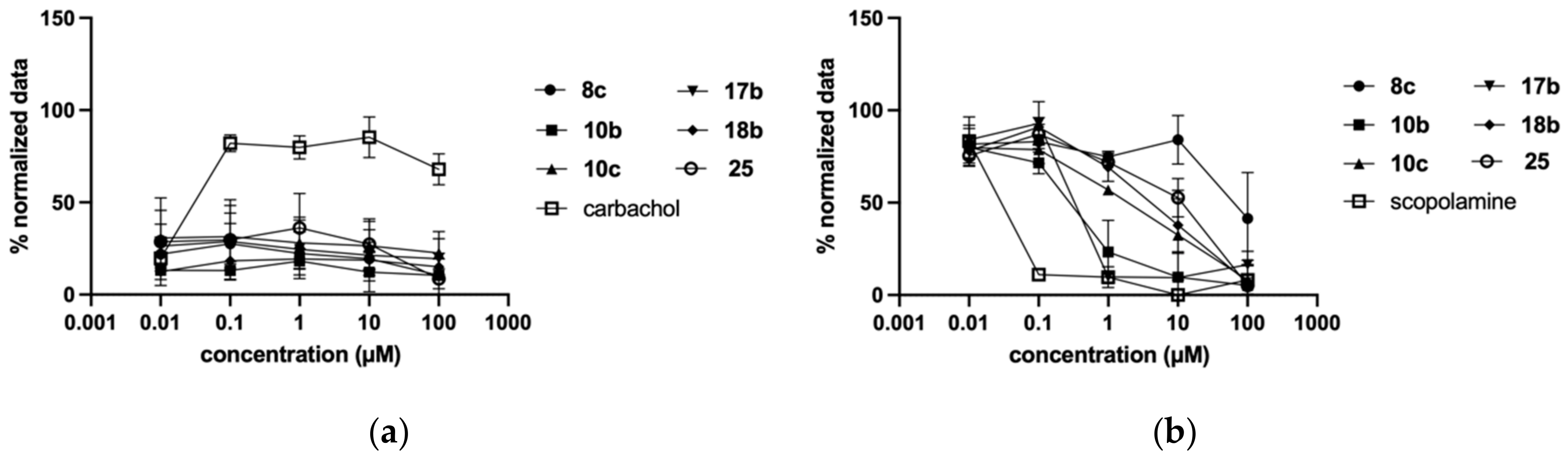
2.3. Biological Evaluation
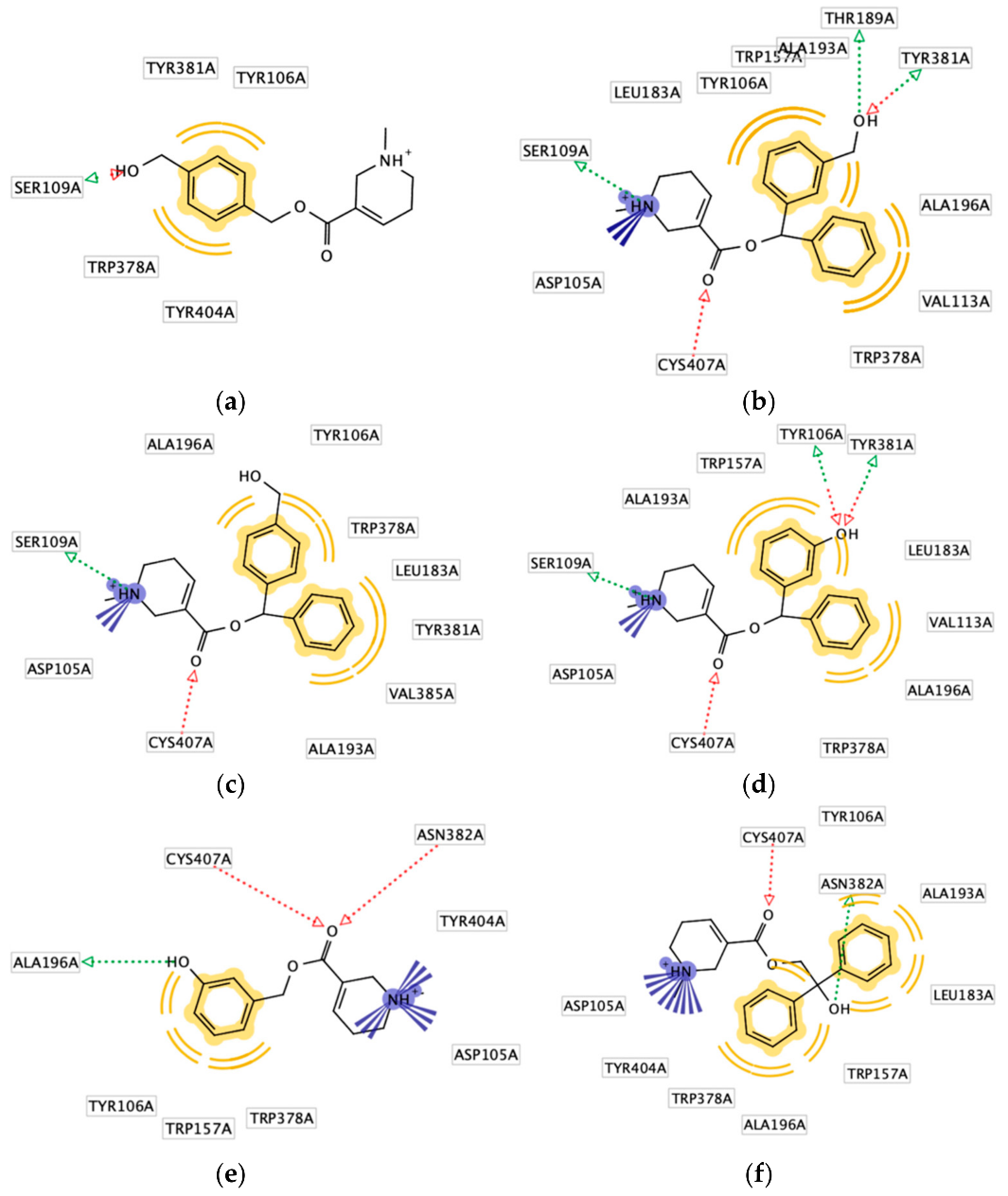
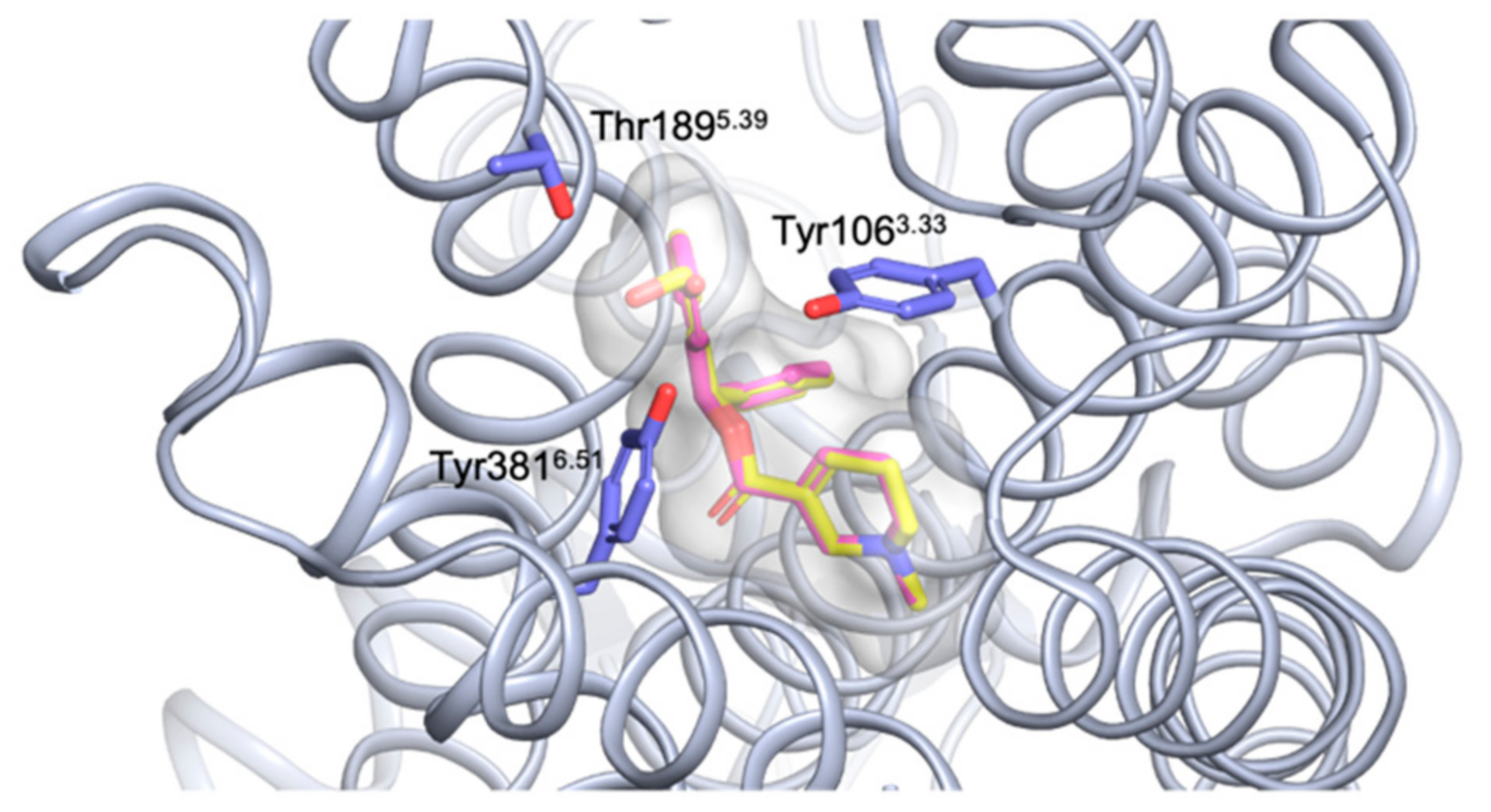
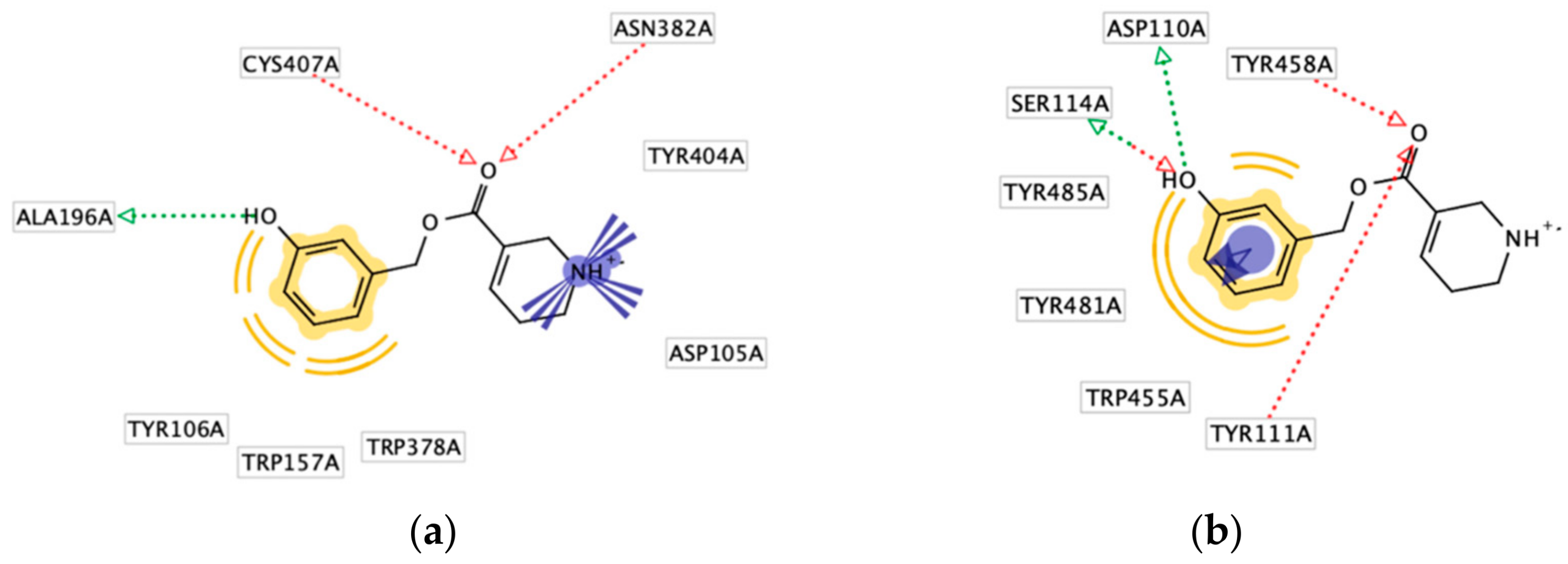
2.4. Molecular Docking Studies
3. Materials and Methods
3.1. Chemistry
3.1.1. General Considerations
3.1.2. Synthetic Procedures for Precursors (6a, 6c, 13b)
3.1.3. General Procedure for the EDC-Mediated Esterification of Arecaidine (8a−c, 9a−c, 15a−c, 16a−c, 21, 25, 27)
3.1.4. General Procedure for the TBAF-Mediated TBS Deprotection of Arecaidine Esters (10a−c, 17b, 18b, 22)
3.2. High-Throughput HPLC-logD
3.3. Biological Evaluation
3.3.1. Materials and Methods
3.3.2. Cell Culture
3.3.3. Cell Viability (MTT Assay)
3.3.4. Radioligand Binding Experiments
3.3.5. Fluo-4 Calcium Assay for Agonist-Antagonist Discrimination
3.3.6. Data Analysis and Statistics
3.4. Molecular Docking
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fryer, A.D.; Christopoulos, A.; Nathanson, N.N. (Eds.) Muscarinic Receptors; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Xiang, Z.; Thompson, A.D.; Jones, C.K.; Lindsley, C.W.; Conn, P.J. Roles of the M1 Muscarinic Acetylcholine Receptor Subtype in the Regulation of Basal Ganglia Function and Implications for the Treatment of Parkinson Disease. J. Pharmacol. Exp. Ther. 2012, 340, 595–603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fisher, A. Cholinergic treatments with emphasis on M1 muscarinic agonists as potential disease-modifying agents for Alzheimer’s disease. Neurotherapeutics 2008, 5, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Bakker, G.; Vingerhoets, C.; Boucherie, D.; Caan, M.; Bloemen, O.; Eersels, J.; Booij, J.; van Amelsvoort, T. Relationship between muscarinic M1 receptor binding and cognition in medication-free subjects with psychosis. NeuroImage Clin. 2018, 18, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Wess, J.; Eglen, R.M.; Gautam, D. Muscarinic acetylcholine receptors: Mutant mice provide new insights for drug development. Nat. Rev. Drug Discov. 2007, 6, 721–733. [Google Scholar] [CrossRef]
- Johnson, C.R.; Kangas, B.D.; Jutkiewicz, E.M.; Bergman, J.; Coop, A. Drug Design Targeting the Muscarinic Receptors and the Implications in Central Nervous System Disorders. Biomedicines 2022, 10, 398. [Google Scholar] [CrossRef]
- Thomsen, M.; Sørensen, G.; Dencker, D. Physiological roles of CNS muscarinic receptors gained from knockout mice. Neuropharmacology 2018, 136, 411–420. [Google Scholar] [CrossRef] [Green Version]
- Suridjan, I.; Comley, R.A.; Rabiner, E.A. The application of positron emission tomography (PET) imaging in CNS drug development. Brain Imaging Behav. 2019, 13, 354–365. [Google Scholar] [CrossRef]
- McCluskey, S.P.; Plisson, C.; Rabiner, E.A.; Howes, O. Advances in CNS PET: The state-of-the-art for new imaging targets for pathophysiology and drug development. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 451–489. [Google Scholar] [CrossRef] [Green Version]
- Ozenil, M.; Pacher, K.; Balber, T.; Vraka, C.; Roller, A.; Holzer, W.; Spreitzer, H.; Mitterhauser, M.; Wadsak, W.; Hacker, M.; et al. Enhanced arecoline derivatives as muscarinic acetylcholine receptor M1 ligands for potential application as PET radiotracers. Eur. J. Med. Chem. 2020, 204, 112623. [Google Scholar] [CrossRef]
- Ozenil, M.; Aronow, J.; Piljak, D.; Vraka, C.; Holzer, W.; Spreitzer, H.; Wadsak, W.; Hacker, M.; Pichler, V. Synthesis, Biological, and Computational Evaluation of Antagonistic, Chiral Hydrobenzoin Esters of Arecaidine Targeting mAChR. Pharmaceuticals 2020, 13, 437. [Google Scholar] [CrossRef]
- Chojnacka, K.; Santoro, S.; Awartani, R.; Richards, N.G.J.; Himo, F.; Aponick, A. Synthetic studies on the solanacol ABC ring system by cation-initiated cascade cyclization: Implications for strigolactone biosynthesis. Org. Biomol. Chem. 2011, 9, 5350–5353. [Google Scholar] [CrossRef] [PubMed]
- Fang, Z.; Li, Y.; Xie, H. A 4-OTBS benzyl-based protective group for carboxylic acids. Tetrahedron Lett. 2019, 60, 1658–1662. [Google Scholar] [CrossRef]
- Pike, V.W. Considerations in the Development of Reversibly Binding PET Radioligands for Brain Imaging. Curr. Med. Chem. 2016, 23, 1818–1869. [Google Scholar] [CrossRef] [PubMed]
- Gobbi, L.; Mercier, J.; Bang, A.B.; Nicolas, J.M.; Reilly, J.; Wagner, B.; Whitehead, D.; Briard, E.; Maguire, R.P.; Borroni, E.; et al. A Comparative Study of in vitro Assays for Predicting the Nonspecific Binding of PET Imaging Agents in vivo. ChemMedChem 2020, 15, 585–592. [Google Scholar] [CrossRef]
- Tong, L.; Li, W.; Lo, M.M.C.; Gao, X.; Wai, J.M.C.; Rudd, M.; Tellers, D.; Joshi, A.; Zeng, Z.; Miller, P.; et al. Discovery of [11C]MK-6884: A Positron Emission Tomography (PET) Imaging Agent for the Study of M4Muscarinic Receptor Positive Allosteric Modulators (PAMs) in Neurodegenerative Diseases. J. Med. Chem. 2020, 63, 2411–2425. [Google Scholar] [CrossRef] [Green Version]
- Wager, T.T.; Hou, X.; Verhoest, P.R.; Villalobos, A. Moving beyond Rules: The Development of a Central Nervous System Multiparameter Optimization (CNS MPO) Approach To Enable Alignment of Druglike Properties. ACS Chem. Neurosci. 2010, 1, 435–449. [Google Scholar] [CrossRef] [Green Version]
- Rankovic, Z. CNS Drug Design: Balancing Physicochemical Properties for Optimal Brain Exposure. J. Med. Chem. 2015, 58, 2584–2608. [Google Scholar] [CrossRef]
- Marvin 22.2. Chemaxon: Budapest, Hungary. Available online: https://www.chemaxon.com (accessed on 27 March 2022).
- Zhang, L.; Villalobos, A. Strategies to facilitate the discovery of novel CNS PET ligands. EJNMMI Radiopharm. Chem. 2016, 1, 13. [Google Scholar] [CrossRef] [Green Version]
- Donovan, S.F.; Pescatore, M.C. Method for measuring the logarithm of the octanol–water partition coefficient by using short octadecyl–poly(vinyl alcohol) high-performance liquid chromatography columns. J. Chromatogr. A 2002, 952, 47–61. [Google Scholar] [CrossRef]
- Vallat, P.; Fan, W.; Tayar, N.E.; Carrupt, P.A.; Testa, B. Solvatochromic Analysis of the Retention Mechanism of two Novel Stationary Phases Used for Measuring Lipophilicity by RP-HPLC. J. Liq. Chromatogr. 1992, 15, 2133–2151. [Google Scholar] [CrossRef]
- Liang, C.; Qiao, J.; Lian, H. Determination of reversed-phase high performance liquid chromatography based octanol-water partition coefficients for neutral and ionizable compounds: Methodology evaluation. J. Chromatogr. A 2017, 1528, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Ghose, A.K.; Herbertz, T.; Hudkins, R.L.; Dorsey, B.D.; Mallamo, J.P. Knowledge-Based, Central Nervous System (CNS) Lead Selection and Lead Optimization for CNS Drug Discovery. ACS Chem. Neurosci. 2012, 3, 50–68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kilian, J.; Ozenil, M.; Millard, M.; Fürtös, D.; Maisetschläger, V.; Holzer, W.; Wadsak, W.; Hacker, M.; Langer, T.; Pichler, V. Design, Synthesis, and Biological Evaluation of 4: Difluorobenzhydrol Carbamates as Selective M1 Antagonists. Pharmaceuticals 2022, 15, 248. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, J.A.; Weinstein, H. [19] Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein-coupled receptors. In Receptor Molecular Biology; Sealfon, S.C., Ed.; Academic Press: Cambridge, MA, USA, 1995; Volume 25, pp. 366–428. ISBN 1043-9471. [Google Scholar]
- Van Rhee, A.M.; Jacobson, K.A. Molecular architecture of G protein-coupled receptors. Drug Dev. Res. 1996, 37, 1–38. [Google Scholar] [CrossRef] [Green Version]
- Thal, D.M.; Sun, B.; Feng, D.; Nawaratne, V.; Leach, K.; Felder, C.C.; Bures, M.G.; Evans, D.A.; Weis, W.I.; Bachhawat, P.; et al. Crystal structures of the M1 and M4 muscarinic acetylcholine receptors. Nature 2016, 531, 335. [Google Scholar] [CrossRef] [Green Version]
- Chien, H.C.; Colas, C.; Finke, K.; Springer, S.; Stoner, L.; Zur, A.A.; Venteicher, B.; Campbell, J.; Hall, C.; Flint, A.; et al. Reevaluating the Substrate Specificity of the L-Type Amino Acid Transporter (LAT1). J. Med. Chem. 2018, 61, 7358–7373. [Google Scholar] [CrossRef]
- Van Zyl, V.L.; Muller, A.; Williams, D.B.G. Charge-tagged polar phosphine ligands in Pd-catalysed reactions in aqueous and ionic media. Tetrahedron Lett. 2018, 59, 918–921. [Google Scholar] [CrossRef]
- Yang, M.H.; Hunt, J.R.; Sharifi, N.; Altman, R.A. Palladium Catalysis Enables Benzylation of α,α-Difluoroketone Enolates. Angew. Chem. Int. Ed. 2016, 55, 9080–9083. [Google Scholar] [CrossRef] [Green Version]
- Lewis, R.S.; Garza, C.J.; Dang, A.T.; Pedro, T.K.A.; Chain, W.J. Michael Additions of Highly Basic Enolates to ortho-Quinone Methides. Org. Lett. 2015, 17, 2278–2281. [Google Scholar] [CrossRef] [Green Version]
- Englert, C.; Nischang, I.; Bader, C.; Borchers, P.; Alex, J.; Pröhl, M.; Hentschel, M.; Hartlieb, M.; Traeger, A.; Pohnert, G.; et al. Photocontrolled Release of Chemicals from Nano- and Microparticle Containers. Angew. Chem. Int. Ed. 2018, 57, 2479–2482. [Google Scholar] [CrossRef]
- Wei, P.; Xue, F.; Shi, Y.; Strand, R.; Chen, H.; Yi, T. A fluoride activated methylene blue releasing platform for imaging and antimicrobial photodynamic therapy of human dental plaque. Chem. Commun. 2018, 54, 13115–13118. [Google Scholar] [CrossRef] [PubMed]
- Bouffard, J.; Itami, K. A Nickel Catalyst for the Addition of Organoboronate Esters to Ketones and Aldehydes. Org. Lett. 2009, 11, 4410–4413. [Google Scholar] [CrossRef] [PubMed]
- Huy, P.H.; Motsch, S.; Kappler, S.M. Formamides as Lewis Base Catalysts in SN Reactions—Efficient Transformation of Alcohols into Chlorides, Amines, and Ethers. Angew. Chem. Int. Ed. 2016, 55, 10145–10149. [Google Scholar] [CrossRef] [PubMed]
- Gharpure, S.J.; Shukla, M.K.; Vijayasree, U. Stereoselective Synthesis of Donor−Acceptor Substituted Cyclopropafuranones by Intramolecular Cyclopropanation of Vinylogous Carbonates: Divergent Synthesis of Tetrahydrofuran-3-one, Tetrahydropyran-3-one, and Lactones. Org. Lett. 2009, 11, 5466–5469. [Google Scholar] [CrossRef] [PubMed]
- Giumanini, A.G.; Tubaro, F. Reduction of Long Chain and Bulky Carboxylic Esters by sodium tetrahydroborate. J. Für Prakt. Chem. 1990, 332, 755–761. [Google Scholar] [CrossRef]
- Song, S.; Zhu, S.F.; Pu, L.Y.; Zhou, Q.L. Iridium-Catalyzed Enantioselective Hydrogenation of Unsaturated Heterocyclic Acids. Angew. Chem. Int. Ed. 2013, 52, 6072–6075. [Google Scholar] [CrossRef]
- Vraka, C.; Nics, L.; Wagner, K.H.; Hacker, M.; Wadsak, W.; Mitterhauser, M.P. A yesterday’s value? Nucl. Med. Biol. 2017, 50, 1–10. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [Green Version]
- Vuckovic, Z.; Gentry, P.R.; Berizzi, A.E.; Hirata, K.; Varghese, S.; Thompson, G.; van der Westhuizen, E.T.; Burger, W.A.; Raphaël, R.; Valant, C.; et al. Crystal structure of the M5 muscarinic acetylcholine receptor. Proc. Natl. Acad. Sci. USA 2019, 116, 26001–26007. [Google Scholar] [CrossRef] [Green Version]
- Bell, E.W.; Zhang, Y. DockRMSD: An open-source tool for atom mapping and RMSD calculation of symmetric molecules through graph isomorphism. J. Cheminform. 2019, 11, 40. [Google Scholar] [CrossRef] [Green Version]
- QUACPAC 2.0.1.2. OpenEye Scientific Software. Available online: http://www.eyesopen.com (accessed on 27 March 2022).
- Wolber, G.; Langer, T. LigandScout: 3-D Pharmacophores Derived from Protein-Bound Ligands and Their Use as Virtual Screening Filters. J. Chem. Inf. Model. 2005, 45, 160–169. [Google Scholar] [CrossRef] [PubMed]
- The PyMol Molecular Graphics System, Version 2.3. Schrödinger, LLC.: New York, NY, USA, 2022. Available online: https://www.pymol.org (accessed on 27 March 2022).
- Matano, Y.; Nomura, H.; Hisanaga, T.; Nakano, H.; Shiro, M.; Imahori, H. Diverse Structures and Remarkable Oxidizing Ability of Triarylbismuthane Oxides. Comparative Study on the Structure and Reactivity of a Series of Triarylpnictogen Oxides. Organometallics 2004, 23, 5471–5480. [Google Scholar] [CrossRef]
- Yang, S.; Tang, W.; Yang, Z.; Xu, J. Iridium-Catalyzed Highly Efficient and Site-Selective Deoxygenation of Alcohols. ACS Catal. 2018, 8, 9320–9326. [Google Scholar] [CrossRef]
- Zheng, Y.; Clarkson, G.J.; Wills, M. Asymmetric Transfer Hydrogenation of o-Hydroxyphenyl Ketones: Utilizing Directing Effects That Optimize the Asymmetric Synthesis of Challenging Alcohols. Org. Lett. 2020, 22, 3717–3721. [Google Scholar] [CrossRef]


| Cmpd. | HPLC-logD | CNS MPO Score 1 |
|---|---|---|
| 8a | 0.79 ± 0.01 | 5.71 |
| 8b | 0.87 ± 0.01 | 5.70 |
| 8c | 1.03 ± 0.04 | 5.70 |
| 10b | 2.6 ± 0.1 | 5.20 |
| 10c | 2.8 ± 0.2 | 5.20 |
| 17b | 2.69 ± 0.01 | 4.83 |
| 18b | 1.57 ± 0.04 | 5.72 |
| 22 | 0.92 ± 0.08 | 5.75 |
| 25 | 2.69 ± 0.01 | 5.34 |
| 27 | 2.69 ± 0.01 | 5.03 |
| DPMA | 3.32 ± 0.04 2 | 4.16 |
| Affinity: Ki ± SD (nM) | x-Fold Selectivity for hM1 vs. hMx 1 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Cmpd. | hM1 | hM2 | hM3 | hM4 | hM5 | hM2 | hM3 | hM4 | hM5 |
| 8c | 525 ± 36 | >1000 2 | 663 ± 56 | 697 ± 132 | 417 ± 73 | >1.9 | 1.3 | 1.3 | 0.8 |
| 10b | 12.5 ± 0.8 | 198 ± 35 | 70.4 ± 1.8 | 32.3 ± 3.8 | 49.1 ± 8.5 | 15.8 | 5.6 | 2.6 | 3.9 |
| 10c | 74.0 ± 3.2 | 980 ± 32 | 150 ± 26 | 93 ± 23 | 72 ± 13 | 13.2 | 2.0 | 1.3 | 1.0 |
| 17b | 5.5 ± 0.5 | 172 ± 20 | 20.4 ± 1.2 | 18.4 ± 5.8 | 11.0 ± 1.5 | 31.3 | 3.7 | 3.3 | 2.0 |
| 18b | 259 ± 75 | >1000 3 | 188 ± 18 | 634 ± 90 | 90 ± 27 | >3.7 | 0.7 | 2.4 | 0.35 |
| 25 | 231 ± 60 | >1000 3 | 283 ± 20 | 217 ± 23 | 284 ± 62 | >4.3 | 1.2 | 0.9 | 1.2 |
| DPMA | 3.1 ± 0.5 4 | 110 ± 39 4 | 56 ± 22 4 | 16 ± 4 4 | 14 ± 1 4 | 35.5 | 18.1 | 5.2 | 4.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kilian, J.; Millard, M.; Ozenil, M.; Krause, D.; Ghaderi, K.; Holzer, W.; Urban, E.; Spreitzer, H.; Wadsak, W.; Hacker, M.; et al. Synthesis, Biological Evaluation, and Docking Studies of Antagonistic Hydroxylated Arecaidine Esters Targeting mAChRs. Molecules 2022, 27, 3173. https://doi.org/10.3390/molecules27103173
Kilian J, Millard M, Ozenil M, Krause D, Ghaderi K, Holzer W, Urban E, Spreitzer H, Wadsak W, Hacker M, et al. Synthesis, Biological Evaluation, and Docking Studies of Antagonistic Hydroxylated Arecaidine Esters Targeting mAChRs. Molecules. 2022; 27(10):3173. https://doi.org/10.3390/molecules27103173
Chicago/Turabian StyleKilian, Jonas, Marlon Millard, Marius Ozenil, Dominik Krause, Khadija Ghaderi, Wolfgang Holzer, Ernst Urban, Helmut Spreitzer, Wolfgang Wadsak, Marcus Hacker, and et al. 2022. "Synthesis, Biological Evaluation, and Docking Studies of Antagonistic Hydroxylated Arecaidine Esters Targeting mAChRs" Molecules 27, no. 10: 3173. https://doi.org/10.3390/molecules27103173
APA StyleKilian, J., Millard, M., Ozenil, M., Krause, D., Ghaderi, K., Holzer, W., Urban, E., Spreitzer, H., Wadsak, W., Hacker, M., Langer, T., & Pichler, V. (2022). Synthesis, Biological Evaluation, and Docking Studies of Antagonistic Hydroxylated Arecaidine Esters Targeting mAChRs. Molecules, 27(10), 3173. https://doi.org/10.3390/molecules27103173







