Identification and Quantification, Metabolism and Pharmacokinetics, Pharmacological Activities, and Botanical Preparations of Protopine: A Review
Abstract
1. Introduction
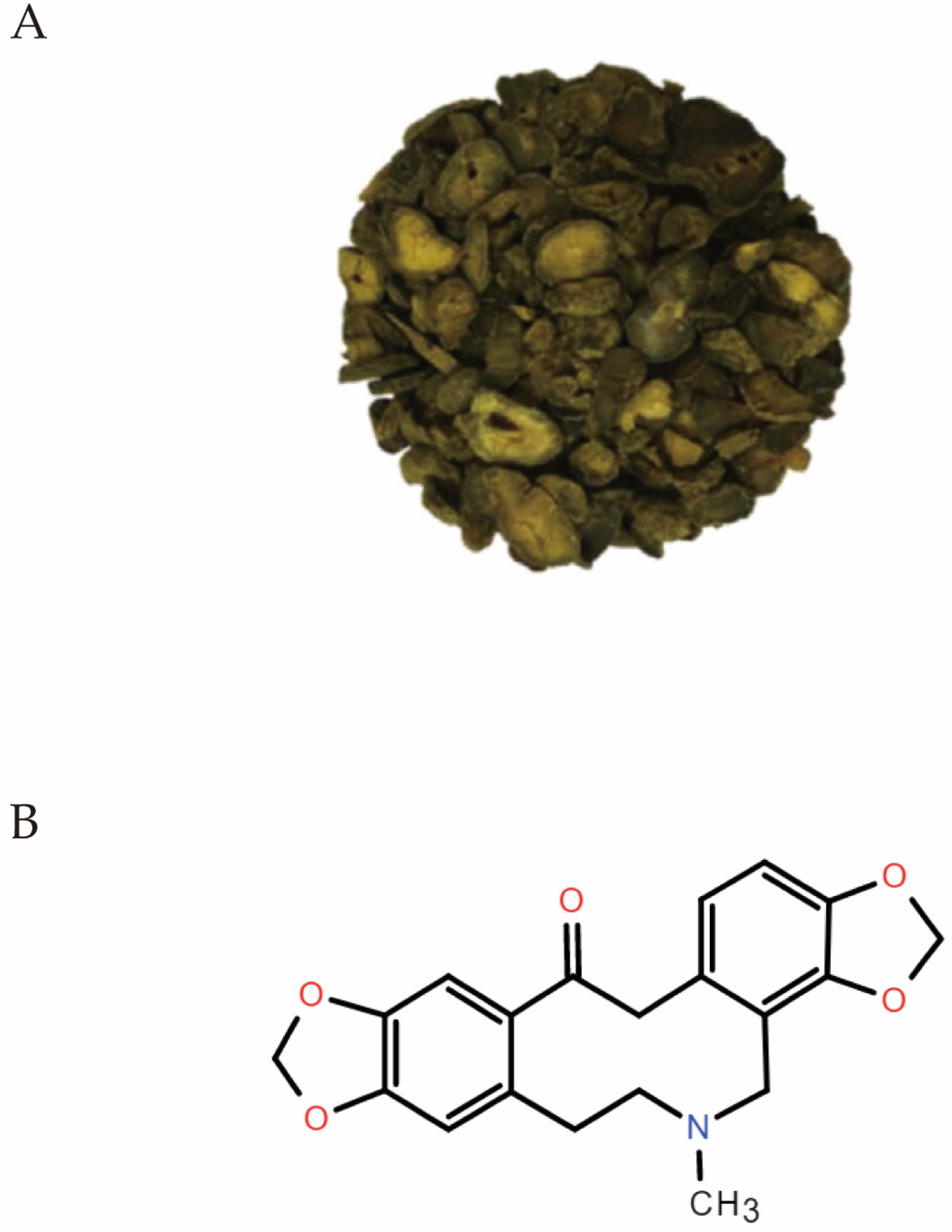
2. Identification and Quantification of Protopine
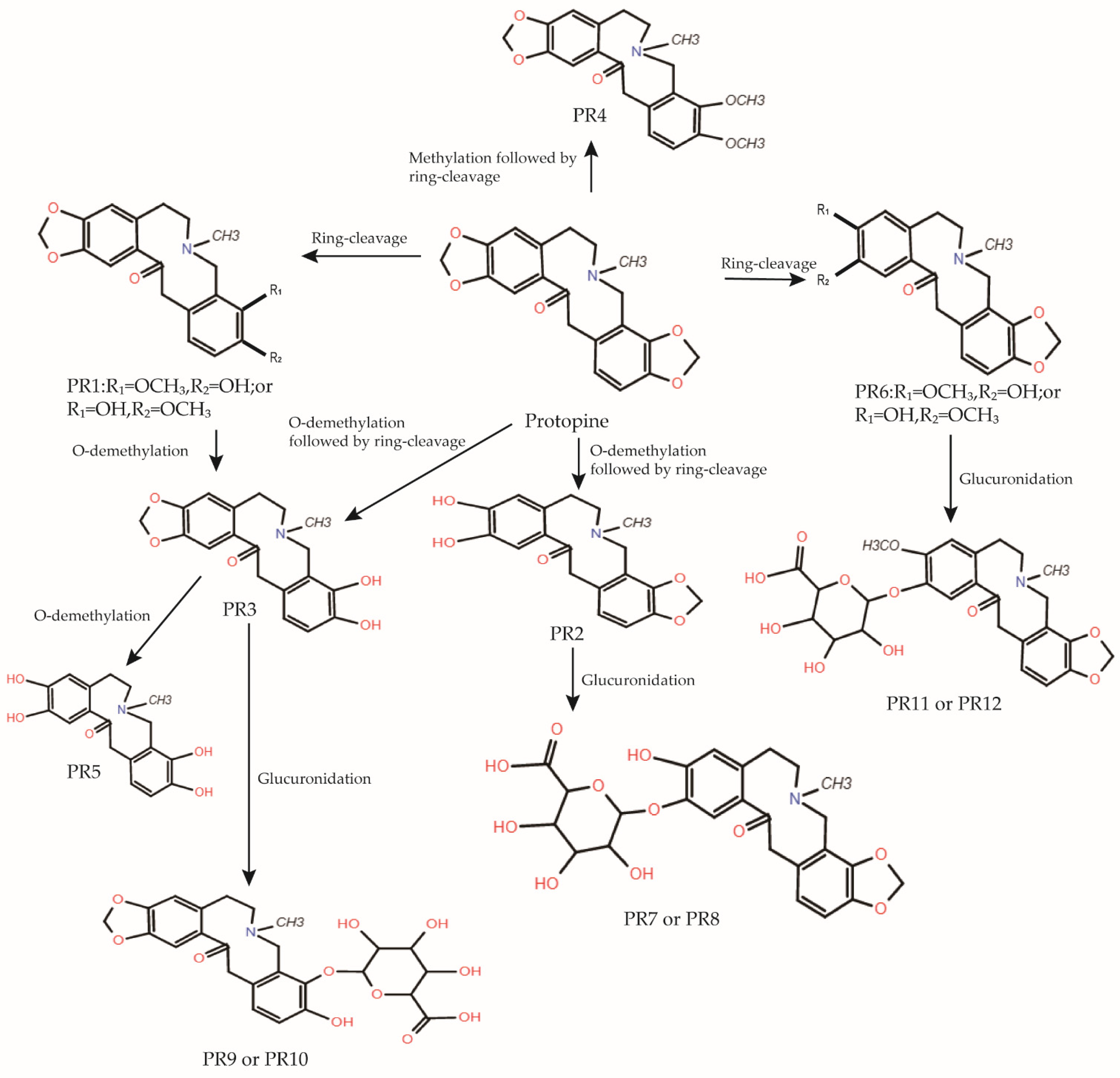
3. Metabolism and Pharmacokinetics of Protopine
4. Pharmacological Activities of Protopine
4.1. Anti-Inflammatory Activities
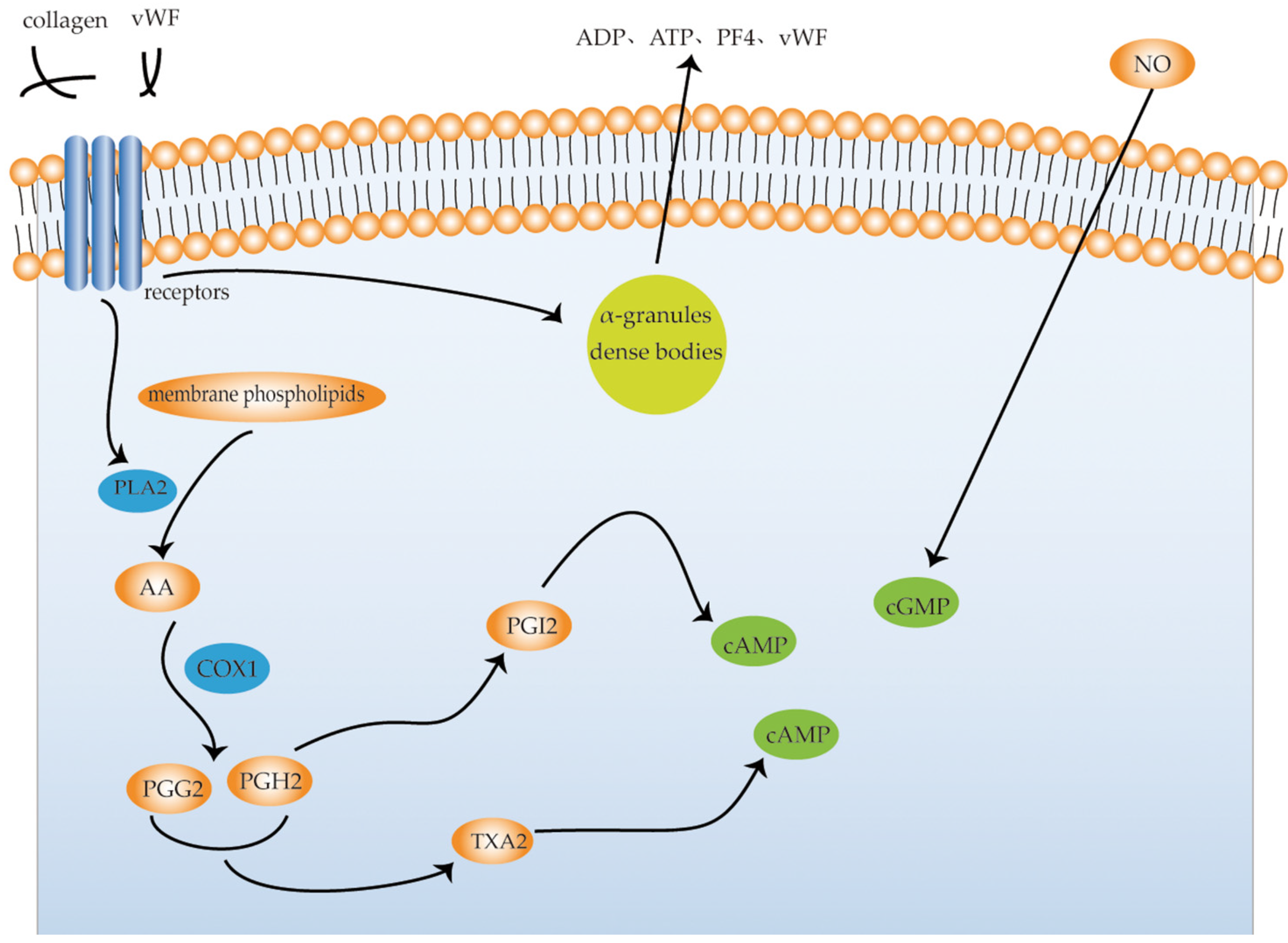
4.2. Anti-Platelet Aggregation Activities
4.3. Anti-Cancer Activity
4.3.1. Liver Cancer
4.3.2. Colon Cancer
4.3.3. Breast Cancer
4.3.4. Pancreatic Cancer
4.3.5. Prostate Cancer
4.3.6. Lung Cancer
4.4. Analgesic Activity
4.5. Vasodilatory Activity
4.6. Anticholinesterase Activity
4.7. Anti-Addictive Activity
4.8. Anticonvulsant Activity
4.9. Antipathogen Activity
4.9.1. Helicobacter Pylori
4.9.2. Parasite
Leishmania
Strongyloides
4.9.3. SARS-CoV-2
4.10. Antioxidation Activity
4.11. Hepatoprotective Activity
4.12. Neuroprotective Activities
4.13. Cytotoxic and Anti-Proliferative Activities of Protopine
5. Botanical Preparations of Protopine
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ekiert, H.M.; Szopa, A. Biological Activities of Natural Products. Molecules 2020, 25, 5769. [Google Scholar] [CrossRef] [PubMed]
- Roy, N.; Parama, D.; Banik, K.; Bordoloi, D.; Devi, A.K.; Thakur, K.K.; Padmavathi, G.; Shakibaei, M.; Fan, L.; Sethi, G.; et al. An Update on Pharmacological Potential of Boswellic Acids against Chronic Diseases. Int. J. Mol. Sci. 2019, 20, 4101. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.B.; Ju, M.K.; Kwon, Y.G.; Lee, S.H. Protopine Attenuates Inflammation Stimulated by Carrageenan and Lps Via the Mapk/Nf-Kappab Pathway. Food Chem. Toxicol. 2019, 131, 110583. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, Z.; Xu, J.; Yang, F.; Dai, C.; Xie, W.; Liang, Z.; Su, S. Optimization of the extraction and purification of Corydalis yanhusuo W.T. Wang based on the Q-marker uniform design method. BMC Chem. 2020, 14, 1–11. [Google Scholar] [CrossRef]
- Huang, Y.-J.; Cheng, P.; Zhang, Z.-Y.; Tian, S.-J.; Sun, Z.-L.; Zeng, J.-G.; Liu, Z.-Y. Biotransformation and tissue distribution of protopine and allocryptopine and effects of Plume Poppy Total Alkaloid on liver drug-metabolizing enzymes. Sci. Rep. 2018, 8, 537. [Google Scholar] [CrossRef]
- He, K.; Gao, J.-L. Protopine Inhibits Heterotypic Celladhesion In Mda-Mb-231 Cells Through Down-Regulation Of Multi-Adhesive Factors. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 415–424. [Google Scholar] [CrossRef]
- Wada, Y.; Kaga, H.; Uchiito, S.; Kumazawa, E.; Tomiki, M.; Onozaki, Y.; Kurono, N.; Tokuda, M.; Ohkuma, A.T.; Orito, K. On the Synthesis of Protopine Alkaloids. J. Org. Chem. 2007, 72, 7301–7306. [Google Scholar] [CrossRef]
- Qing, Z.-X.; Cheng, P.; Liu, X.-B.; Liu, Y.-S.; Zeng, J.-G. Systematic identification of alkaloids in Macleaya microcarpa fruits by liquid chromatography tandem mass spectrometry combined with the isoquinoline alkaloids biosynthetic pathway. J. Pharm. Biomed. Anal. 2015, 103, 26–34. [Google Scholar] [CrossRef]
- Guo, C.; Jiang, Y.; Li, L.; Hong, L.; Wang, Y.; Shen, Q.; Lou, Y.; Hu, H.; Zhou, H.; Yu, L.; et al. Application of a liquid chromatography–tandem mass spectrometry method to the pharmacokinetics, tissue distribution and excretion studies of Dactylicapnos scandens in rats. J. Pharm. Biomed. Anal. 2013, 74, 92–100. [Google Scholar] [CrossRef]
- Wang, X.-Y.; Ding, X.; Yuan, Y.-F.; Zheng, L.-Y.; Cao, Y.; Zhu, Z.-Y.; Zhang, G.-Q.; Chai, Y.-F.; Chen, X.-F.; Hong, Z.-Y. Comprehensive two-dimensional APTES-decorated MCF7-cell membrane chromatographic system for characterizing potential anti-breast-cancer components from Yuanhu–Baizhi herbal medicine pair. J. Food Drug Anal. 2018, 26, 823–833. [Google Scholar] [CrossRef]
- Lu, M.; Li, K.; He, H.; Cheng, Y.; Yang, P. Systematic characterization of alkaloids in Eomecon chionantha Hance using ultrahigh-performance liquid chromatography–tandem quadrupole Exactive Orbitrap mass spectrometry with a four-step screening strategy. Rapid Commun. Mass Spectrom. 2020, 34, e8880. [Google Scholar] [CrossRef]
- Bournine, L.; Bensalem, S.; Wauters, J.-N.; Iguer-Ouada, M.; Maiza-Benabdesselam, F.; Bedjou, F.; Castronovo, V.; Bellahcène, A.; Tits, M.; Frédérich, M. Identification and Quantification of the Main Active Anticancer Alkaloids from the Root of Glaucium flavum. Int. J. Mol. Sci. 2013, 14, 23533–23544. [Google Scholar] [CrossRef]
- Ahmad, A.; Amir, M.; Alshadidi, A.A.; Hussain, M.D.; Haq, A.; Kazi, M. Central composite design expert-supported development and validation of HPTLC method: Relevance in quantitative evaluation of protopine in Fumaria indica. Saudi Pharm. J. 2020, 28, 487–494. [Google Scholar] [CrossRef]
- Wu, C.; Wang, X.; Xu, M.; Liu, Y.; Di, X. Intracellular Accumulation as an Indicator of Cytotoxicity to Screen Hepatotoxic Components of Chelidonium majus L. by LC-MS/MS. Molecules 2019, 24, 2410. [Google Scholar] [CrossRef] [PubMed]
- Suau, R.; Cabezudo, B.; Valpuesta, M.; Posadas, N.; Diaz, A.; Torres, G. Identification and quantification of isoquinoline alkaloids in the genus Sarcocapnos by GC-MS. Phytochem. Anal. 2005, 16, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Berkov, S.; Bastida, J.; Nikolova, M.; Viladomat, F.; Codina, C. Rapid TLC/GC-MS identification of acetylcholinesterase inhibitors in alkaloid extracts. Phytochem. Anal. 2008, 19, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Su, S.; Cui, W.; Liu, P.; Duan, J.-A.; Guo, J.; Li, Z.; Shang, E.; Qian, D.; Huang, Z. Simultaneous determination of paeoniflorin, albiflorin, ferulic acid, tetrahydropalmatine, protopine, typhaneoside, senkyunolide I in Beagle dogs plasma by UPLC–MS/MS and its application to a pharmacokinetic study after Oral Administration of Shaofu Zhuyu Decoction. J. Chromatogr. B 2014, 962, 75–81. [Google Scholar] [CrossRef]
- Tao, Y.; Xu, H.; Wang, S.; Wang, B.; Zhang, Y.; Wang, W.; Huang, B.; Wu, H.; Li, D.; Zhang, Y.; et al. Identification of the absorbed constituents after oral administration of Yuanhu Zhitong prescription extract and its pharmacokinetic study by rapid resolution liquid chromatography/quadrupole time-of-flight. J. Chromatogr. B 2013, 935, 1–9. [Google Scholar] [CrossRef]
- Zhou, Q.; Liu, Y.; Wang, X.; Di, X. A sensitive and selective liquid chromatography−tandem mass spectrometry method for simultaneous determination of five isoquinoline alkaloids from Chelidonium majus L. in rat plasma and its application to a pharmacokinetic study. J. Mass Spectrom. 2013, 48, 111–118. [Google Scholar] [CrossRef]
- Dou, Z.; Li, K.; Wang, P.; Cao, L. Effect of Wine and Vinegar Processing of Rhizoma Corydalis on the Tissue Distribution of Tetrahydropalmatine, Protopine and Dehydrocorydaline in Rats. Molecules 2012, 17, 951–970. [Google Scholar] [CrossRef]
- Huang, Y.-J.; Xiao, S.; Sun, Z.-L.; Zeng, J.-G.; Liu, Y.-S.; Liu, Z.-Y. Identification of allocryptopine and protopine metabolites in rat liver S9 by high-performance liquid chromatography/quadrupole-time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2016, 30, 1549–1559. [Google Scholar] [CrossRef]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef]
- Zhang, B.; Zeng, M.; Li, M.; Kan, Y.; Li, B.; Xu, R.; Wu, Y.; Wang, S.; Zheng, X.; Feng, W. Protopine Protects Mice against LPS-Induced Acute Kidney Injury by Inhibiting Apoptosis and Inflammation via the TLR4 Signaling Pathway. Molecules 2019, 25, 15. [Google Scholar] [CrossRef] [PubMed]
- Bae, D.-S.; Kim, Y.-H.; Pan, C.-H.; Nho, C.-W.; Samdan, J.; Yansan, J.; Lee, J.-K. Protopine reduces the inflammatory activity of lipopolysaccharide-stimulated murine macrophages. BMB Rep. 2012, 45, 108–113. [Google Scholar] [CrossRef]
- Saeed, S.; Gilani, A.; Majoo, R.; Shah, B. Anti-thrombotic and anti-inflammatory activities of protopine. Pharmacol. Res. 1997, 36, 1–7. [Google Scholar] [CrossRef]
- Cimmino, G.; Golino, P. Platelet Biology and Receptor Pathways. J. Cardiovasc. Transl. Res. 2013, 6, 299–309. [Google Scholar] [CrossRef]
- Ramasamy, I. Inherited bleeding disorders: Disorders of platelet adhesion and aggregation. Crit. Rev. Oncol. 2003, 49, 1–35. [Google Scholar] [CrossRef]
- van der Meijden, P.E.J.; Heemskerk, J.W.M. Platelet Biology and Functions: New Concepts and Clinical Perspectives. Nat. Rev. Cardiol. 2019, 16, 166–179. [Google Scholar] [CrossRef] [PubMed]
- Ko, F.N.; Wu, T.S.; Lu, S.T.; Wu, Y.C.; Huang, T.F.; Teng, C.M. Antiplatelet Effects of Protopine Isolated from Corydalis Tubers. Thromb. Res. 1989, 56, 289–298. [Google Scholar] [CrossRef]
- Shen, Z.Q.; Chen, Z.H.; Duan, L. Effect of protopine on cytosolic Ca2+ in rabbit platelets. Zhongguo Yao Li Xue Bao 1999, 20, 338–340. [Google Scholar]
- Ma, G.Y.; Zhang, Z.Z.; Chen, Z.H. Effect of protopine on rabbit platelet function. Zhongguo Yao Li Xue Bao 1994, 15, 367–371. [Google Scholar]
- Shiomoto, H.; Matsuda, H.; Kubo, M. Effects of protopine on blood platelet aggregation. II. Effect on metabolic system of adenosine 3′,5′-cyclic monophosphate in platelets. Chem. Pharm. Bull. 1990, 38, 2320–2322. [Google Scholar] [CrossRef]
- Shiomoto, H.; Matsuda, H.; Kubo, M. Effects of protopine on blood platelet aggregation. III. Effect of propopine on the metabolic system of arachidonic acid in platelets. Chem. Pharm. Bull. 1991, 39, 474–477. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Roy, P.S.; Saikia, B.J. Cancer and cure: A critical analysis. Indian J. Cancer 2017, 53, 441–442. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Qi, M.; Li, N.; Lei, Y.; Zhang, D.; Chen, J. Natural products and their derivatives: Promising modulators of tumor immunotherapy. J. Leukoc. Biol. 2020, 108, 493–508. [Google Scholar] [CrossRef]
- Anwanwan, D.; Singh, S.K.; Singh, S.; Saikam, V.; Singh, R. Challenges in liver cancer and possible treatment approaches. Biochim. Biophys. Acta Rev. Cancer 2020, 1873, 188314. [Google Scholar] [CrossRef] [PubMed]
- Nie, C.; Wang, B.; Wang, B.; Lv, N.; Yu, R.; Zhang, E. Protopine triggers apoptosis via the intrinsic pathway and regulation of ROS/PI3K/Akt signalling pathway in liver carcinoma. Cancer Cell Int. 2021, 21, 1–10. [Google Scholar] [CrossRef]
- Vrba, J.; Vrublova, E.; Modriansky, M.; Ulrichova, J. Protopine and allocryptopine increase mRNA levels of cytochromes P450 1A in human hepatocytes and HepG2 cells independently of AhR. Toxicol. Lett. 2011, 203, 135–141. [Google Scholar] [CrossRef]
- Yu, J.-J.; Cong, D.-L.; Jiang, Y.; Zhou, Y.; Wang, Y.; Zhao, C.-F. Study on alkaloids of Corydalis ochotensis and their antitumor bioactivity. Zhong Yao Cai 2014, 37, 1795–1798. [Google Scholar]
- Labianca, R.; Beretta, G.D.; Kildani, B.; Milesi, L.; Merlin, F.; Mosconi, S.; Pessi, M.A.; Prochilo, T.; Quadri, A.; Gatta, G.; et al. Colon Cancer. Crit. Rev. Oncol. Hematol. 2010, 74, 106–133. [Google Scholar] [CrossRef]
- Yaghoubi, A.; Khazaei, M.; Avan, A.; Hasanian, S.M.; Soleimanpour, S. The bacterial instrument as a promising therapy for colon cancer. Int. J. Color. Dis. 2020, 35, 595–606. [Google Scholar] [CrossRef] [PubMed]
- Son, Y.; An, Y.; Jung, J.; Shin, S.; Park, I.; Gwak, J.; Ju, B.G.; Chung, Y.; Na, M.; Oh, S. Protopine isolated from Nandina domestica induces apoptosis and autophagy in colon cancer cells by stabilizing p53. Phytotherapy Res. 2019, 33, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Pearce, L. Breast Cancer. Nurs. Stand. 2016, 30, 15. [Google Scholar] [CrossRef] [PubMed]
- Cano, S.; Klassen, A.F.; Scott, A.; Thoma, A.; Feeny, D.; Pusic, A. Health outcome and economic measurement in breast cancer surgery: Challenges and opportunities. Expert Rev. Pharm. Outcomes Res. 2010, 10, 583–594. [Google Scholar] [CrossRef]
- Fisusi, F.A.; Akala, E.O. Drug Combinations in Breast Cancer Therapy. Pharm. Nanotechnol. 2019, 7, 3–23. [Google Scholar] [CrossRef]
- Goral, V. Pancreatic Cancer: Pathogenesis and Diagnosis. Asian Pac. J. Cancer Prev. 2015, 16, 5619–5624. [Google Scholar] [CrossRef]
- McGuigan, A.; Kelly, P.; Turkington, R.C.; Jones, C.; Coleman, H.G.; McCain, R.S. Pancreatic cancer: A review of clinical diagnosis, epidemiology, treatment and outcomes. World J. Gastroenterol. 2018, 24, 4846–4861. [Google Scholar] [CrossRef]
- Garcia-Gil, M.; Turri, B.; Gabriele, M.; Pucci, L.; Agnarelli, A.; Lai, M.; Freer, G.; Pistello, M.; Vignali, R.; Batistoni, R.; et al. Protopine/Gemcitabine Combination Induces Cytotoxic or Cytoprotective Effects in Cell Type-Specific and Dose-Dependent Manner on Human Cancer and Normal Cells. Pharmaceuticals 2021, 14, 90. [Google Scholar] [CrossRef]
- Wang, G.; Zhao, D.; Spring, D.J.; Depinho, R.A. Genetics and biology of prostate cancer. Genes Dev. 2018, 32, 1105–1140. [Google Scholar] [CrossRef]
- Chen, C.-H.; Liao, C.-H.; Chang, Y.-L.; Guh, J.-H.; Pan, S.-L.; Teng, C.-M. Protopine, a novel microtubule-stabilizing agent, causes mitotic arrest and apoptotic cell death in human hormone-refractory prostate cancer cell lines. Cancer Lett. 2012, 315, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, A.G.; Cote, M.L. Epidemiology of Lung Cancer. Adv. Exp. Med. Biol. 2016, 893, 21–41. [Google Scholar] [PubMed]
- Collins, L.G.; Haines, C.; Perkel, R.; E Enck, R. Lung cancer: Diagnosis and management. Am. Fam. Physician 2007, 75, 56–63. [Google Scholar] [PubMed]
- Lemjabbar-Alaoui, H.; Hassan, O.U.; Yang, Y.-W.; Buchanan, P. Lung cancer: Biology and treatment options. Biochim. Biophys. Acta 2015, 1856, 189–210. [Google Scholar] [CrossRef]
- Loeser, J.D.; Melzack, R. Pain: An Overview. Lancet 1999, 353, 1607–1609. [Google Scholar] [CrossRef]
- Henschke, N.; Kamper, S.; Maher, C. The Epidemiology and Economic Consequences of Pain. Mayo Clin. Proc. 2015, 90, 139–147. [Google Scholar] [CrossRef]
- Xu, Y.; Sun, J.; Li, W.; Zhang, S.; Yang, L.; Teng, Y.; Lv, K.; Liu, Y.; Su, Y.; Zhang, J.; et al. Analgesic effect of the main components of Corydalis yanhusuo (yanhusuo in Chinese) is caused by inhibition of voltage gated sodium channels. J. Ethnopharmacol. 2021, 280, 114457. [Google Scholar] [CrossRef]
- Xu, Q.; Jin, R.L.; Wu, Y.Y. Opioid, calcium, and adrenergic receptor involvement in protopine analgesia. Zhongguo Yao Li Xue Bao 1993, 14, 495–500. [Google Scholar]
- Ko, F.N.; Wu, T.S.; Lu, S.T.; Wu, Y.C.; Huang, T.F.; Teng, C.M. Ca(2+)-Channel Blockade in Rat Thoracic Aorta by Protopine Isolated from Corydalis Tubers. Jpn. J. Pharmacol. 1992, 58, 1–9. [Google Scholar] [CrossRef]
- Huang, Y.H.; Zhang, Z.Z.; Jiang, J.X. Relaxant effects of protopine on smooth muscles. Zhongguo Yao Li Xue Bao 1991, 12, 16–19. [Google Scholar]
- Li, B.; Wu, Q.; Shi, J.-S.; Sun, A.-S.; Huang, X.-N. Effects of protopine on intracellular calcium and the PKC activity of rat aorta smooth muscle. Sheng Li Xue Bao 2005, 57, 240–246. [Google Scholar]
- Pohanka, M. Diagnoses of Pathological States Based on Acetylcholinesterase and Butyrylcholinesterase. Curr. Med. Chem. 2020, 27, 2994–3011. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K. Cholinesterase Inhibitors as Alzheimer’s Therapeutics (Review). Mol. Med. Rep. 2019, 20, 1479–1487. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Lee, K.T.; Baek, N.-I.; Kim, S.-H.; Park, H.W.; Lim, J.P.; Shin, T.Y.; Eom, D.O.; Yang, J.H.; Eun, J.S. Acetylcholinesterase inhibitors from the aerial parts of Corydalis speciosa. Arch. Pharmacal Res. 2004, 27, 1127–1131. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Hwang, S.Y.; Jang, Y.P.; Park, M.J.; Markelonis, G.J.; Oh, T.H.; Kim, Y.C. Protopine from Corydalis ternata has Anticholinesterase and Antiamnesic Activities. Planta Medica 1999, 65, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Langlois, L.D.; Nugent, F.S. Opiates and Plasticity in the Ventral Tegmental Area. ACS Chem. Neurosci. 2017, 8, 1830–1838. [Google Scholar] [CrossRef] [PubMed]
- Capasso, A.; Piacente, S.; Pizza, C.; de Tommasi, N.; Jativa, C.; Sorrentino, L. Isoquinoline Alkaloids from Argemone Mexicana Reduce Morphine Withdrawal in Guinea Pig Isolated Ileum. Planta Med. 1997, 63, 326–328. [Google Scholar] [CrossRef]
- Capasso, A.; De Tommasi, N.; Rastrelli, L.; De Simone, F. New Protopine Alkaloids from Aristolochia Constricta Reduce Morphine Withdrawal in Vitro. Phytother. Res. 2000, 14, 653–655. [Google Scholar] [CrossRef]
- Beghi, E.; Giussani, G.; Sander, J.W. The natural history and prognosis of epilepsy. Epileptic Disord. 2015, 17, 243–253. [Google Scholar] [CrossRef]
- Cheke, R.S.; Firke, S.D.; Patil, R.R.; Bari, S. ISATIN: New Hope Against Convulsion. Central Nerv. Syst. Agents Med. Chem. 2018, 18, 76–101. [Google Scholar] [CrossRef]
- Prokopenko, Y.; Tsyvunin, V.; Shtrygol’, S.; Georgiyants, V. In Vivo Anticonvulsant Activity of Extracts and Protopine from the Fumaria schleicheri Herb. Sci. Pharm. 2015, 84, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Kardos, J.; Blaskó, G.; Simonyi, M. Enhancement of gamma-aminobutyric acid receptor binding by protopine-type alkaloids. Arzneimittelforschung 1986, 36, 939–940. [Google Scholar] [PubMed]
- Camilo, V.; Sugiyama, T.; Touati, E. Pathogenesis of Helicobacter Pylori Infection. Helicobacter 2017, 22 (Suppl. 1), e12405. [Google Scholar] [CrossRef] [PubMed]
- Mahady, G.B.; Pendland, S.L.; Stoia, A.; Chadwick, L.R. In Vitro Susceptibility of Helicobacter Pylori to Isoquinoline Alkaloids from Sanguinaria Canadensis and Hydrastis Canadensis. Phytother. Res. 2003, 17, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Bi, K.; Chen, Y.; Zhao, S.; Kuang, Y.; Wu, C.-H.J. Current Visceral Leishmaniasis Research: A Research Review to Inspire Future Study. BioMed Res. Int. 2018, 2018, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Chandra, D. Alkaloids and Leishmania donovani UDP-Galactopyarnosemutase: A Novel Approach in Drug Designing Against Visceral Leishmaniasis. Infect. Disord. Drug Targets 2018, 18, 145–155. [Google Scholar] [CrossRef]
- Krolewiecki, A.; Nutman, T.B. Strongyloidiasis: A Neglected Tropical Disease. Infect. Dis. Clin. N. Am. 2019, 33, 135–151. [Google Scholar] [CrossRef]
- Wikman-Jorgensen, P.; Requena-Méndez, A.; Llenas-García, J. A Review on Strongyloidiasis in Pregnant Women. Res. Rep. Trop. Med. 2021, 12, 219–225. [Google Scholar] [CrossRef]
- Satou, T.; Koga, M.; Matsuhashi, R.; Koike, K.; Tada, I.; Nikaido, T. Assay of nematocidal activity of isoquinoline alkaloids using third-stage larvae of Strongyloides ratti and S. venezuelensis. Veter. Parasitol. 2002, 104, 131–138. [Google Scholar] [CrossRef]
- Hosseini, E.S.; Kashani, N.R.; Nikzad, H.; Azadbakht, J.; Bafrani, H.H.; Kashani, H.H. The novel coronavirus Disease-2019 (COVID-19): Mechanism of action, detection and recent therapeutic strategies. Virology 2020, 551, 1–9. [Google Scholar] [CrossRef]
- Hu, B.; Guo, H.; Zhou, P.; Shi, Z.L. Characteristics of Sars-Cov-2 and Covid-19. Nat. Rev. Microbiol. 2021, 19, 141–154. [Google Scholar] [CrossRef]
- Ita, K. Coronavirus Disease (COVID-19): Current Status and Prospects for Drug and Vaccine Development. Arch. Med. Res. 2021, 52, 15–24. [Google Scholar] [CrossRef]
- Pandeya, K.; Ganeshpurkar, A.; Mishra, M.K. Natural RNA dependent RNA polymerase inhibitors: Molecular docking studies of some biologically active alkaloids of Argemone mexicana. Med. Hypotheses 2020, 144, 109905. [Google Scholar] [CrossRef]
- Kücükakin, B.; Gögenur, I.; Reiter, R.J.; Rosenberg, J. Oxidative Stress in Relation to Surgery: Is There a Role for the Antioxidant Melatonin? J. Surg. Res. 2009, 152, 338–347. [Google Scholar] [CrossRef]
- Cabello-Verrugio, C.; Simon, F.; Trollet, C.; Santibañez, J.F. Oxidative Stress in Disease and Aging: Mechanisms and Therapies 2016. Oxidative Med. Cell. Longev. 2017, 2017, 1–2. [Google Scholar] [CrossRef]
- Xiao, X.; Liu, J.; Hu, J.; Li, T.; Zhang, Y. Protective Effect of Protopine on the Focal Cerebral Ischaemic Injury in Rats. Basic Clin. Pharmacol. Toxicol. 2007, 101, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Liu, J.; Hu, J.; Zhu, X.; Yang, H.; Wang, C.; Zhang, Y. Protective effects of protopine on hydrogen peroxide-induced oxidative injury of PC12 cells via Ca2+ antagonism and antioxidant mechanisms. Eur. J. Pharmacol. 2008, 591, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Mohi-Ud-Din, R.; Mir, R.H.; Sawhney, G.; Dar, M.A.; Bhat, Z.A. Possible Pathways of Hepatotoxicity Caused by Chemical Agents. Curr. Drug Metab. 2019, 20, 867–879. [Google Scholar] [CrossRef] [PubMed]
- Janbaz, K.; Saeed, S.; Gilani, A. An assessment of the potential of protopine to inhibit microsomal drug metabolising enzymes and prevent chemical-induced hepatotoxicity in rodents. Pharmacol. Res. 1998, 38, 215–219. [Google Scholar] [CrossRef]
- Sveinsson, O.A.; Kjartansson, O.; Valdimarsson, E.M. Cerebral Ischemia/Infarction—Epidemiology, Causes and Symptoms. Laeknabladid 2014, 100, 271–279. [Google Scholar]
- Sai, C.; Wang, J.; Li, B.; Ding, L.; Wang, H.; Wang, Q.; Hua, H.; Zhang, F.; Ren, Q. Isolation and identification of alkaloids from Macleaya microcarpa by UHPLC–Q-TOF-MS and their cytotoxic activity in vitro, antiangiogenic activity in vivo. BMC Chem. 2020, 14, 5. [Google Scholar] [CrossRef]
- Wang, X.; Tanaka, M.; Krstin, S.; Peixoto, H.S.; Wink, M. The Interference of Selected Cytotoxic Alkaloids with the Cytoskeleton: An Insight into Their Modes of Action. Molecules 2016, 21, 906. [Google Scholar] [CrossRef]
- Maggini, V.; Lombardi, N.; Crescioli, G.; Gallo, E.; Sivelli, F.; Gensini, G.F.; Vannacci, A.; Firenzuoli, F. Chelidonium majus: Relevant safety aspects of a hepatotoxic plant, trawling the web. Phytother. Res. 2019, 33, 2465–2469. [Google Scholar] [CrossRef]
- Zhang, R.; Guo, Q.; Kennelly, E.J.; Long, C.; Chai, X. Diverse alkaloids and biological activities of Fumaria (Papaveraceae): An ethnomedicinal group. Fitoterapia 2020, 146, 104697. [Google Scholar] [CrossRef]
- Croaker, A.; King, G.J.; Pyne, J.H.; Anoopkumar-Dukie, S.; Liu, L. Sanguinaria canadensis: Traditional Medicine, Phytochemical Composition, Biological Activities and Current Uses. Int. J. Mol. Sci. 2016, 17, 1414. [Google Scholar] [CrossRef] [PubMed]
- Wangchuk, P.; Bremner, J.B.; Samten; Rattanajak, R.; Kamchonwongpaisan, S. Antiplasmodial agents from the Bhutanese medicinal plant Corydalis calliantha. Phytotherapy Res. 2010, 24, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Vrba, J.; Orolinova, E.; Ulrichova, J. Induction of heme oxygenase-1 by Macleaya cordata extract and its constituent sanguinarine in RAW264.7 cells. Fitoterapia 2012, 83, 329–335. [Google Scholar] [CrossRef]
- Guo, Z.-Y.; Zhang, Z.-Y.; Xiao, J.-Q.; Qin, J.-H.; Zhao, W. Antibacterial Effects of Leaf Extract of Nandina domestica and the Underlined Mechanism. Evid. Based Complement. Altern. Med. 2018, 2018, 1–9. [Google Scholar] [CrossRef] [PubMed]
| In Vivo/In Vivo | Signaling Pathway | Effects | References |
|---|---|---|---|
| LPS-induced AKI mouse model | apoptosis and TLR4 | reduces levels of BUN, Scr, inflammatory cells, and inflammatory factors (IFN-γ, TNF, and IL-2) | [7] |
| PS-stimulated BV2 cell model | NF-κB andMAPK | inhibits the production of iNOS, COX-2, and inflammatory factors (TNF-α, IL-1 β, and IL-6) | [3] |
| CA-induced mouse model | NF-κB | inhibits the expression of iNOS, COX-2, and alleviated paw edema | [3] |
| LPS-stimulated RAW264.7 cell model | NF-κB andMAPK | reduces the production of NO, COX-2 and PGE2 | [8] |
| CA-induced rat model | inhibits paw edema | [3] |
| Cancer Type | Cell Line | Mechanism | References |
|---|---|---|---|
| Liver Cancer | HepG2 | [23] | |
| HepG2 and Huh-7 | es regulates ROS/PI3K/Akt signaling pathway and the intrinsic pathway | [21] | |
| Colon Cancer | HCT116 | induces apoptosis and autophagy by stabilizing p53 | [26] |
| SW480 | [23] | ||
| Breast Cancer | MDA-MB-231 | anti-adhesion and anti-invasive | [30] |
| Pancreatic Cancer | MIA Paca-2 and PANC-1 | [33] | |
| Prostate Cancer | HRPC | regulates Cdk1 activity and Bcl-2 family of proteins. | [35] |
| Lung Cancer | A549_ | [23] |
| Pathogen | Mechanism | References | |
|---|---|---|---|
| Bacteria | Helicobacter pylori | [58] | |
| Parasite | Leishmania | inhibits the function of UDP- galactosidase (UGM) | [60] |
| Strongyloides | [63] | ||
| Virus | SARS-CoV-2 | inhibits the activity of RNA-dependent RNA polymerase | [67] |
| Application in Diseases | References | |
|---|---|---|
| Chelidonium majus L. | Liver Dysfunction and Skin Diseases | [93] |
| Fumaria L. | Hypertension, Rash and Arthritis | [94] |
| Sanguinaria canadensis | Asthma, Tuberculosis and Dysentery | [95] |
| Corydalis Calliantha Long | Malaria | [96] |
| Dactylicapnos scandens | Hypertension, Inflammation, and Pain | [9] |
| Corydalis yanhusuo W.T. Wang | Backache, Arthralgia and Trauma | [3] |
| Hypecoum erectum L. | Inflammation | [24] |
| Macleaya cordata | Inflammation | [97] |
| Nandina domestica | Chronic Bronchitis | [98] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, W.; Kong, L.; Cao, Y.; Yan, L. Identification and Quantification, Metabolism and Pharmacokinetics, Pharmacological Activities, and Botanical Preparations of Protopine: A Review. Molecules 2022, 27, 215. https://doi.org/10.3390/molecules27010215
Huang W, Kong L, Cao Y, Yan L. Identification and Quantification, Metabolism and Pharmacokinetics, Pharmacological Activities, and Botanical Preparations of Protopine: A Review. Molecules. 2022; 27(1):215. https://doi.org/10.3390/molecules27010215
Chicago/Turabian StyleHuang, Wangli, Lingbo Kong, Yang Cao, and Liang Yan. 2022. "Identification and Quantification, Metabolism and Pharmacokinetics, Pharmacological Activities, and Botanical Preparations of Protopine: A Review" Molecules 27, no. 1: 215. https://doi.org/10.3390/molecules27010215
APA StyleHuang, W., Kong, L., Cao, Y., & Yan, L. (2022). Identification and Quantification, Metabolism and Pharmacokinetics, Pharmacological Activities, and Botanical Preparations of Protopine: A Review. Molecules, 27(1), 215. https://doi.org/10.3390/molecules27010215