Gravitational Influence on Human Living Systems and the Evolution of Species on Earth
Abstract
1. Introduction
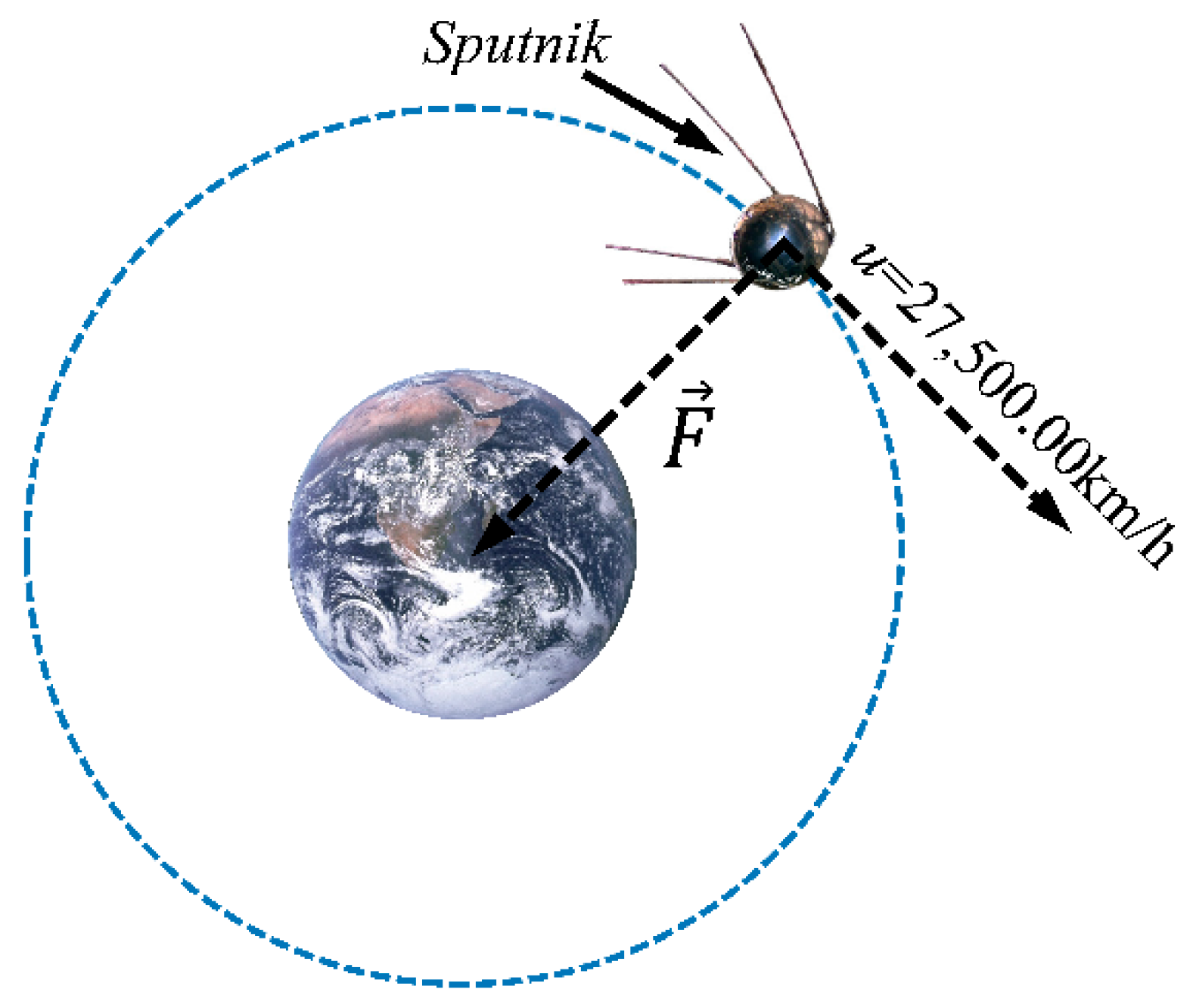
1.1. Using Gravitational Force (g) an Experimental Variable
1.2. Literature Mining
2. Effects of Altered Gravity on Homo sapiens and Other Vertebrate Species
2.1. Gravity and the Thyroid Gland
2.2. Gravity and the Cardiovascular System
2.3. Gravity and the Immune System
2.4. Gravity and the Vestibular System
2.5. Gravity and the Musculoskeletal System
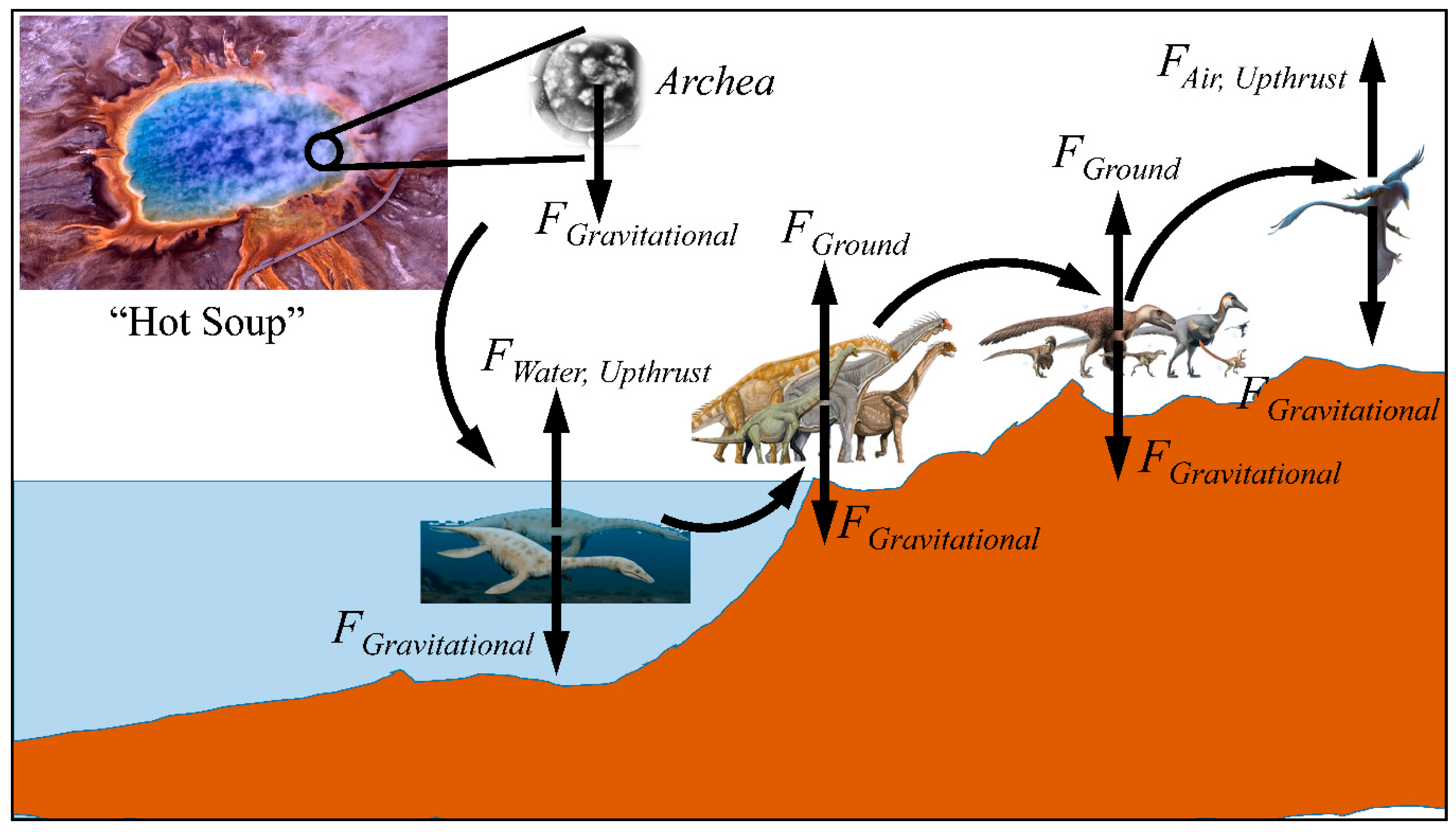
3. Gravity and Evolution
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hansma, H.G. The Power of Crowding for the Origins of Life. Orig. Life Evol. Biosph. 2014, 44, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Fallah-Araghi, A.; Meguellati, K.; Baret, J.-C.; El Harrak, A.; Mangeat, T.; Karplus, M.; Ladame, S.; Marques, C.M.; Griffiths, A.D. Enhanced Chemical Synthesis at Soft Interfaces: A Universal Reaction-Adsorption Mechanism in Microcompartments. Phys. Rev. Lett. 2014, 112, 028301. [Google Scholar] [CrossRef] [PubMed]
- Shapshak, P. Astrobiology—An opposing view. Bioinformation 2018, 14, 346–349. [Google Scholar] [CrossRef]
- Morey-Holton, E.R. The impact of gravity on life. In Evolution on Planet Earth; Elsevier BV: Amsterdam, The Netherlands, 2003; pp. 143–159. [Google Scholar]
- Ross, M. The influence of gravity on structure and function of animals. Adv. Space Res. 1984, 4, 305–314. [Google Scholar] [CrossRef]
- Chandler, D. Weightlessness and microgravity. Phys. Teach. 1991, 29, 312–313. [Google Scholar] [CrossRef]
- Kiss, J.Z.; Wolverton, C.; Wyatt, S.E.; Hasenstein, K.H.; Van Loon, J.J. Comparison of Microgravity Analogs to Spaceflight in Studies of Plant Growth and Development. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.H. From gravity and the organism to gravity and the cell. ASGSB Bull. 1991, 4, 7–18. [Google Scholar]
- Brungs, S.; Egli, M.; Wuest, S.L.; Christianen, P.C.M.; Van Loon, J.J.W.A.; Anh, T.J.N.; Hemmersbach, R. Facilities for Simulation of Microgravity in the ESA Ground-Based Facility Programme. Microgravity Sci. Technol. 2016, 28, 191–203. [Google Scholar] [CrossRef]
- Fujita, S.-I.; Rutter, L.; Ong, Q.; Muratani, M. Integrated RNA-seq Analysis Indicates Asynchrony in Clock Genes between Tissues under Spaceflight. Life 2020, 10, 196. [Google Scholar] [CrossRef]
- Mao, X.W.; Nishiyama, N.C.; Byrum, S.D.; Stanbouly, S.; Jones, T.; Holley, J.; Sridharan, V.; Boerma, M.; Tackett, A.J.; Willey, J.S.; et al. Spaceflight induces oxidative damage to blood-brain barrier integrity in a mouse model. FASEB J. 2020. [Google Scholar] [CrossRef]
- Oka, M.; Kamada, M.; Inoue, R.; Miyamoto, K.; Uheda, E.; Yamazaki, C.; Shimazu, T.; Sano, H.; Kasahara, H.; Suzuki, T.; et al. Altered localisation of ZmPIN1a proteins in plasma membranes responsible for enhanced-polar auxin transport in etiolated maize seedlings under microgravity conditions in space. Funct. Plant Biol. 2020. [Google Scholar] [CrossRef]
- Kaplan, F.; Shapiro-Ilan, D.; Schiller, K.C. Dynamics of entomopathogenic nematode foraging and infectivity in microgravity. NPJ Microgravity 2020, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nay, K.; Koechlin-Ramonatxo, C.; Rochdi, S.; Island, M.; Orfila, L.; Treffel, L.; Bareille, M.; Beck, A.; Gauquelin-Koch, G.; Ropert, M.; et al. Simulated microgravity disturbs iron metabolism and distribution in humans: Lessons from dry immersion, an innovative ground-based human model. FASEB J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Patel, S. The effects of microgravity and space radiation on cardiovascular health: From low-Earth orbit and beyond. IJC Heart Vasc. 2020, 30, 100595. [Google Scholar] [CrossRef]
- Weber, B.; Panzirsch, M.; Stulp, F.; Schneider, S. Sensorimotor performance and haptic support in simulated weightlessness. Exp. Brain Res. 2020, 238, 2373–2384. [Google Scholar] [CrossRef]
- Roda, A.; Mirasoli, M.; Guardigli, M.; Zangheri, M.; Caliceti, C.; Calabria, D.; Simoni, P. Advanced biosensors for monitoring astronauts’ health during long-duration space missions. Biosens. Bioelectron. 2018, 111, 18–26. [Google Scholar] [CrossRef]
- Terai, M.; Nitta, K. The basic health care system for the lunar base crew. Acta Astronaut. 1991, 25, 239–243. [Google Scholar] [CrossRef]
- E Garrett-Bakelman, F.; Darshi, M.; Green, S.J.; Gur, R.C.; Lin, L.; Macias, B.R.; McKenna, M.J.; Meydan, C.; Mishra, T.; Nasrini, J.; et al. The NASA Twins Study: A multidimensional analysis of a year-long human spaceflight. Science 2019, 364, 364. [Google Scholar] [CrossRef]
- White, R.J.; Averner, M. Humans in space. Nature 2001, 409, 1115–1118. [Google Scholar] [CrossRef]
- Maggioni, M.A.; Castiglioni, P.; Merati, G.; Brauns, K.; Gunga, H.-C.; Mendt, S.; Opatz, O.S.; Rundfeldt, L.C.; Steinach, M.; Werner, A.; et al. High-Intensity Exercise Mitigates Cardiovascular Deconditioning During Long-Duration Bed Rest. Front. Physiol. 2018, 9, 9. [Google Scholar] [CrossRef]
- Albi, E.; Krüger, M.; Hemmersbach, R.; Lazzarini, A.; Cataldi, S.; Codini, M.; Beccari, T.; Ambesi-Impiombato, F.S.; Curcio, F. Impact of Gravity on Thyroid Cells. Int. J. Mol. Sci. 2017, 18, 972. [Google Scholar] [CrossRef]
- Albi, E.; Ambesi-Impiombato, F.; Peverini, M.; Damaskopoulou, E.; Fontanini, E.; Lazzarini, R.; Curcio, F.; Perrella, G. Thyrotropin Receptor and Membrane Interactions in FRTL-5 Thyroid Cell Strain in Microgravity. Astrobiology 2011, 11, 57–64. [Google Scholar] [CrossRef]
- Wehland, M.; Warnke, E.; Frett, T.; Hemmersbach, R.; Hauslage, J.; Ma, X.; Aleshcheva, G.; Pietsch, J.; Bauer, J.; Grimm, D. The Impact of Hypergravity and Vibration on Gene and Protein Expression of Thyroid Cells. Microgravity Sci. Technol. 2016, 28, 261–274. [Google Scholar] [CrossRef]
- Fuentes, T.I.; Appleby, N.; Raya, M.; Bailey, L.; Hasaniya, N.; Stodieck, L.; Kearns-Jonker, M. Simulated Microgravity Exerts an Age-Dependent Effect on the Differentiation of Cardiovascular Progenitors Isolated from the Human Heart. PLoS ONE 2015, 10, e0132378. [Google Scholar] [CrossRef] [PubMed]
- White, C.R.; Seymour, R.S. The role of gravity in the evolution of mammalian blood pressure. Evolution 2014, 68, 901–908. [Google Scholar] [CrossRef]
- Jha, R.; Wu, Q.; Singh, M.; Preininger, M.K.; Han, P.; Ding, G.; Cho, H.C.; Jo, H.; Maher, K.O.; Wagner, M.B.; et al. Simulated Microgravity and 3D Culture Enhance Induction, Viability, Proliferation and Differentiation of Cardiac Progenitors from Human Pluripotent Stem Cells. Sci. Rep. 2016, 6, 30956. [Google Scholar] [CrossRef]
- Wnorowski, A.; Sharma, A.; Chen, H.; Wu, H.; Shao, N.-Y.; Sayed, N.; Liu, C.; Countryman, S.; Stodieck, L.S.; Rubins, K.H.; et al. Effects of Spaceflight on Human Induced Pluripotent Stem Cell-Derived Cardiomyocyte Structure and Function. Stem Cell Rep. 2019, 13, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Buchheim, J.-I.; Matzel, S.; Rykova, M.; Vassilieva, G.; Ponomarev, S.; Nichiporuk, I.; Hörl, M.; Moser, D.; Biere, K.; Feuerecker, M.; et al. Stress Related Shift Toward Inflammaging in Cosmonauts After Long-Duration Space Flight. Front. Physiol. 2019, 10, 85. [Google Scholar] [CrossRef]
- Boonyaratanakornkit, J.B.; Cogoli, A.; Li, C.; Schopper, T.; Pippia, P.; Galleri, G.; A Meloni, M.; Hughes-Fulford, M. Key gravity-sensitive signaling pathways drive T-cell activation. FASEB J. 2005, 19, 2020–2022. [Google Scholar] [CrossRef]
- Chang, T.T.; Walther, I.; Li, C.-F.; Boonyaratanakornkit, J.; Galleri, G.; Meloni, M.A.; Pippia, P.; Cogoli, A.; Hughes-Fulford, M. The Rel/NF-κB pathway and transcription of immediate early genes in T cell activation are inhibited by microgravity. J. Leukoc. Biol. 2012, 92, 1133–1145. [Google Scholar] [CrossRef] [PubMed]
- Girardi, C.; De Pittà, C.; Casara, S.; Calura, E.; Romualdi, C.; Celotti, L.; Mognato, M. Integration Analysis of MicroRNA and mRNA Expression Profiles in Human Peripheral Blood Lymphocytes Cultured in Modeled Microgravity. Biomed Res. Int. 2014, 2014, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, B.; Seetharam, A.; Wang, Z.; Liu, Y.; Lossie, A.C.; Thimmapuram, J.; Irudayaraj, J. A Study of Alterations in DNA Epigenetic Modifications (5mC and 5hmC) and Gene Expression Influenced by Simulated Microgravity in Human Lymphoblastoid Cells. PLoS ONE 2016, 11, e0147514. [Google Scholar] [CrossRef]
- Thiel, C.S.; Huge, A.; Hauschild, S.; Tauber, S.; Lauber, B.A.; Polzer, J.; Paulsen, K.; Lier, H.; Engelmann, F.; Schmitz, B.; et al. Stability of gene expression in human T cells in different gravity environments is clustered in chromosomal region 11p15.4. NPJ Microgravity 2017, 3, 22. [Google Scholar] [CrossRef]
- Sundaresan, A.; Mann, V.; Mehta, S.K.; Crucian, B.; Doursout, M.F.; Devakottai, S. Effects of microgravity and other space stressors in immunosuppression and viral reactivation with potential nervous system involvement. Neurol. India 2019, 67, 198–S203. [Google Scholar] [CrossRef] [PubMed]
- Kaur, I.; Simons, E.R.; Castro, V.A.; Ott, C.M.; Pierson, D.L. Changes in monocyte functions of astronauts. Brain Behav. Immun. 2005, 19, 547–554. [Google Scholar] [CrossRef]
- Moser, D.; Sun, S.J.; Li, N.; Biere, K.; Hoerl, M.; Matzel, S.; Feuerecker, M.; Buchheim, J.-I.; Strewe, C.; Thiel, C.S.; et al. Cells’ Flow and Immune Cell Priming under alternating g-forces in Parabolic Flight. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Verhaar, A.; Hoekstra, E.; Tjon, S.; Utomo, W.; Deuring, J.; Bakker, E.R.M.; Muncan, V.; Peppelenbosch, M. Dichotomal effect of space flight-associated microgravity on stress-activated protein kinases in innate immunity. Sci. Rep. 2014, 4, 5468. [Google Scholar] [CrossRef]
- Cogoli-Greuter, M.; Galleri, G.; Pani, G.; Saba, A.; Pippia, P.; Cogoli-Greuter, M. Space flight affects motility and cytoskeletal structures in human monocyte cell line J-111. Cytoskeleton 2011, 68, 125–137. [Google Scholar] [CrossRef]
- Liu, S.; Zhu, L.; Zhang, J.; Yu, J.; Cheng, X.; Peng, B. Anti-osteoclastogenic activity of isoliquiritigenin via inhibition of NF-κB-dependent autophagic pathway. Biochem. Pharmacol. 2016, 106, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Kaur, I.; Simons, E.R.; A Castro, V.; Ott, C.M.; Pierson, D.L. Changes in neutrophil functions in astronauts. Brain Behav. Immun. 2004, 18, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Song, J.; Ling, S.; Niu, S.; Lu, L.; Cui, Z.; Li, Y.; Hao, S.; Zhong, G.; Qi, Z.; et al. Hematopoietic stem cells and lineage cells undergo dynamic alterations under microgravity and recovery conditions. FASEB J. 2019, 33, 6904–6918. [Google Scholar] [CrossRef]
- Dai, S.; Kong, F.; Liu, C.; Xiao, F.; Dong, X.; Zhang, Y.; Wang, H. Effect of simulated microgravity conditions of hindlimb unloading on mice hematopoietic and mesenchymal stromal cells. Cell Biol. Int. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tascher, G.; Gerbaix, M.; Maes, P.; Chazarin, B.; Ghislin, S.; Antropova, E.; Vassilieva, G.; Ouzren-Zarhloul, N.; Gauquelin-Koch, G.; Vico, L.; et al. Analysis of femurs from mice embarked on board BION-M1 biosatellite reveals a decrease in immune cell development, including B cells, after 1 wk of recovery on Earth. FASEB J. 2018, 33, 3772–3783. [Google Scholar] [CrossRef]
- Lescale, C.; Schenten, V.; Djeghloul, D.; Bennabi, M.; Gaignier, F.; Vandamme, K.; Strazielle, C.; Kuzniak, I.; Petite, H.; Dosquet, C.; et al. Hind limb unloading, a model of spaceflight conditions, leads to decreased B lymphopoiesis similar to aging. FASEB J. 2014, 29, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Gaignier, F.; Schenten, V.; Bittencourt, M.D.C.; Gauquelin-Koch, G.; Frippiat, J.-P.; Legrand-Frossi, C. Three Weeks of Murine Hindlimb Unloading Induces Shifts from B to T and from Th to Tc Splenic Lymphocytes in Absence of Stress and Differentially Reduces Cell-Specific Mitogenic Responses. PLoS ONE 2014, 9, e92664. [Google Scholar] [CrossRef] [PubMed]
- Morita, H.; Abe, C.; Tanaka, K. Long-term exposure to microgravity impairs vestibulo-cardiovascular reflex. Sci. Rep. 2016, 6, 33405. [Google Scholar] [CrossRef]
- Jamon, M. The development of vestibular system and related functions in mammals: Impact of gravity. Front. Integr. Neurosci. 2014, 8, 11. [Google Scholar] [CrossRef]
- Hallgren, E.; Kornilova, L.; Fransen, E.; Glukhikh, D.; Moore, S.T.; Clément, G.; Van Ombergen, A.; MacDougall, H.G.; Naumov, I.A.; Wuyts, F. Decreased otolith-mediated vestibular response in 25 astronauts induced by long-duration spaceflight. J. Neurophysiol. 2016, 115, 3045–3051. [Google Scholar] [CrossRef]
- Reschke, M.F.; Wood, S.J.; Clément, G. Ocular Counter Rolling in Astronauts After Short- and Long-Duration Spaceflight. Sci. Rep. 2018, 8, 7747. [Google Scholar] [CrossRef]
- Dadwal, U.C.; Maupin, K.A.; Zamarioli, A.; Tucker, A.; Harris, J.S.; Fischer, J.P.; Rytlewski, J.D.; Scofield, D.C.; Wininger, A.E.; Bhatti, F.U.R.; et al. The effects of spaceflight and fracture healing on distant skeletal sites. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Vico, L.; Hargens, A. Skeletal changes during and after spaceflight. Nat. Rev. Rheumatol. 2018, 14, 229–245. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, G.; Dong, D.; Shang, P. Effects of Iron Overload and Oxidative Damage on the Musculoskeletal System in the Space Environment: Data from Spaceflights and Ground-Based Simulation Models. Int. J. Mol. Sci. 2018, 19, 2608. [Google Scholar] [CrossRef]
- Ciofani, G.; Ricotti, L.; Rigosa, J.; Menciassi, A.; Mattoli, V.; Monici, M. Hypergravity effects on myoblast proliferation and differentiation. J. Biosci. Bioeng. 2012, 113, 258–261. [Google Scholar] [CrossRef]
- Ikawa, T.; Kawaguchi, A.; Okabe, T.; Ninomiya, T.; Nakamichi, Y.; Nakamura, M.; Uehara, S.; Udagawa, N.; Takahashi, N.; Nakamura, H.; et al. Hypergravity suppresses bone resorption in ovariectomized rats. Adv. Space Res. 2011, 47, 1214–1224. [Google Scholar] [CrossRef]
- Vorselen, D.; Roos, W.H.; Mackintosh, F.C.; Wuite, G.J.L.; A Van Loon, J.J.W. The role of the cytoskeleton in sensing changes in gravity by nonspecialized cells. FASEB J. 2013, 28, 536–547. [Google Scholar] [CrossRef]
- Bizzarri, M.; Monici, M.; Van Loon, J.J.W.A. How Microgravity Affects the Biology of Living Systems. BioMed Res. Int. 2015, 2015, 1–4. [Google Scholar] [CrossRef]
- Kacena, M.A.; Todd, P.; Gerstenfeld, L.C.; Landis, W.J. Experiments with osteoblasts cultured under varying orientations with respect to the gravity vector. Cytotechnology 2002, 39, 147–154. [Google Scholar] [CrossRef]
- Maier, J.A.; Cialdai, F.; Monici, M.; Morbidelli, L. The Impact of Microgravity and Hypergravity on Endothelial Cells. BioMed Res. Int. 2015, 2015, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Costa-Almeida, R.; Carvalho, D.T.O.; Ferreira, M.J.S.; Pesqueira, T.; Monici, M.; Van Loon, J.J.; Granja, P.L.; Gomes, M.E. Continuous Exposure to Simulated Hypergravity-Induced Changes in Proliferation, Morphology, and Gene Expression of Human Tendon Cells. Stem Cells Dev. 2018, 27, 858–869. [Google Scholar] [CrossRef]
- Bradamante, S.; Rivero, D.; Barenghi, L.; Balsamo, M.; Minardi, S.P.; Vitali, F.; Cavalieri, D. SCD – Stem Cell Differentiation Toward Osteoblast Onboard the International Space Station. Microgravity Sci. Technol. 2018, 30, 713–729. [Google Scholar] [CrossRef]
- Blaber, E.A.; Finkelstein, H.; Dvorochkin, N.; Sato, K.Y.; Yousuf, R.; Burns, B.P.; Globus, R.K.; Almeida, E.A. Microgravity Reduces the Differentiation and Regenerative Potential of Embryonic Stem Cells. Stem Cells Dev. 2015, 24, 2605–2621. [Google Scholar] [CrossRef] [PubMed]
- Abdelmoaty, H.; Hammond, T.G.; Wilson, B.L.; Birdsall, H.H.; Clement, J.Q. Identification of Putative Major Space Genes Using Genome-Wide Literature Data. Biotechnology 2015. [Google Scholar] [CrossRef]
- Lillywhite, H.B. Snakes, Blood Circulation and Gravity. Sci. Am. 1988, 259, 92–98. [Google Scholar] [CrossRef]
- Perez, D.; Sheehy, C.M.; Lillywhite, S.M. Variation of organ position in snakes. J. Morphol. 2019, 280, 1798–1807. [Google Scholar] [CrossRef]
- Wright, P.A.; Turko, A.J. Amphibious fishes: Evolution and phenotypic plasticity. J. Exp. Biol. 2016, 219, 2245–2259. [Google Scholar] [CrossRef]
- Brunt, E.M.; Turko, A.J.; Scott, G.R.; Wright, P.A. Amphibious fish jump better on land after acclimation to a terrestrial environment. J. Exp. Biol. 2016, 219, 3204–3207. [Google Scholar] [CrossRef]
- Albrecht-Buehler, G. Possible Mechanisms of Indirect Gravity Sensing by Cells; ASGSB bulletin: Publication of the American Society for Gravitational and Space Biology; National Center for Biotechnology Information: Bethesda, MD, USA, July 1991; Volume 4, pp. 25–34. [Google Scholar] [PubMed]
- Todd, P. Physical effects at the cellular level under altered gravity conditions. Adv. Space Res. 1992, 12, 43–49. [Google Scholar] [CrossRef]
- Klaus, D.M.; Benoit, M.R.; Nelson, E.S.; Hammond, T.G. Extracellular mass transport considerations for space flight research concerning suspended and adherent in vitro cell cultures. J. Gravitat. Physiol. 2004, 11, 17–27. [Google Scholar]
- Volkmann, D.; Baluška, F. Gravity: One of the driving forces for evolution. Protoplasma 2006, 229, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Alpatov, A.M.; I’Lin E, A.; Antipov, V.V.; Tairbekov, M.G. Biological experiments on “Kosmos-1887”. Kosm Biol Aviakosm Med. 1989, 23, 26–32. [Google Scholar]
- Nishida, Y.; Maruyama, S.; Shouji, I.; Kemuriyama, T.; Tashiro, A.; Ohta, H.; Hagisawa, K.; Hiruma, M.; Yokoe, H. Effects and biological limitations of +Gz acceleration on the autonomic functions-related circulation in rats. J. Physiol. Sci. 2016, 66, 447–462. [Google Scholar] [CrossRef] [PubMed]
- Ohira, T.; Kawano, F.; Ohira, T.; Goto, K.; Ohira, Y. Responses of skeletal muscles to gravitational unloading and/or reloading. J. Physiol. Sci. 2015, 65, 293–310. [Google Scholar] [CrossRef] [PubMed]
- Stupak, H.D.; Park, S.Y. Gravitational forces, negative pressure and facial structure in the genesis of airway dysfunction during sleep: A review of the paradigm. Sleep Med. 2018, 51, 125–132. [Google Scholar] [CrossRef]
- Akparibo, I.Y.; Chumbley, E. Aerospace, Gravitational Effects, High Performance; StatPearls Publishing. LLC.: Treasure Island, FL, USA, 2020. [Google Scholar]
- Guettler, N.; Bron, D.; Manen, O.; Gray, G.; Syburra, T.; Rienks, R.; D’Arcy, J.; Davenport, E.D.; Nicol, E.D. Management of cardiac conduction abnormalities and arrhythmia in aircrew. Heart 2018, 105, s38–s49. [Google Scholar] [CrossRef]
- Shen, M.; Frishman, W.H. Effects of Spaceflight on Cardiovascular Physiology and Health. Cardiol. Rev. 2019, 27, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Najrana, T.; Sanchez-Esteban, J. Mechanotransduction as an Adaptation to Gravity. Front. Pediatr. 2016, 4, 140. [Google Scholar] [CrossRef]
- Nickerson, C.A.; Ott, C.M.; Wilson, J.W.; Ramamurthy, R.; Pierson, D.L. Microbial Responses to Microgravity and Other Low-Shear Environments. Microbiol. Mol. Biol. Rev. 2004, 68, 345–361. [Google Scholar] [CrossRef]
- Sonnenfeld, G. Immune Responses in Space Flight. Int. J. Sports Med. 1998, 19, S195–S204. [Google Scholar] [CrossRef]
- Egroover, A. Gravitropisms and reaction woods of forest trees—evolution, functions and mechanisms. New Phytol. 2016, 211, 790–802. [Google Scholar] [CrossRef]
- Moulia, B.; Bastien, R.; Chauvet, H.; Leblanc-Fournier, N. Posture control in land plants: Growth, position sensing, proprioception, balance, and elasticity. J. Exp. Bot. 2019, 70, 3467–3494. [Google Scholar] [CrossRef]
- Shevchenko, G.V. Putative gravisensors among microtubule associated proteins. Cell Biol. Int. 2017, 43, 983–990. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adamopoulos, K.; Koutsouris, D.; Zaravinos, A.; Lambrou, G.I. Gravitational Influence on Human Living Systems and the Evolution of Species on Earth. Molecules 2021, 26, 2784. https://doi.org/10.3390/molecules26092784
Adamopoulos K, Koutsouris D, Zaravinos A, Lambrou GI. Gravitational Influence on Human Living Systems and the Evolution of Species on Earth. Molecules. 2021; 26(9):2784. https://doi.org/10.3390/molecules26092784
Chicago/Turabian StyleAdamopoulos, Konstantinos, Dimitrios Koutsouris, Apostolos Zaravinos, and George I. Lambrou. 2021. "Gravitational Influence on Human Living Systems and the Evolution of Species on Earth" Molecules 26, no. 9: 2784. https://doi.org/10.3390/molecules26092784
APA StyleAdamopoulos, K., Koutsouris, D., Zaravinos, A., & Lambrou, G. I. (2021). Gravitational Influence on Human Living Systems and the Evolution of Species on Earth. Molecules, 26(9), 2784. https://doi.org/10.3390/molecules26092784








