The Effect of Calcium-Silicate Cements on Reparative Dentinogenesis Following Direct Pulp Capping on Animal Models
Abstract
1. Introduction
1.1. Molecular Mechanisms in Dentinogenesis
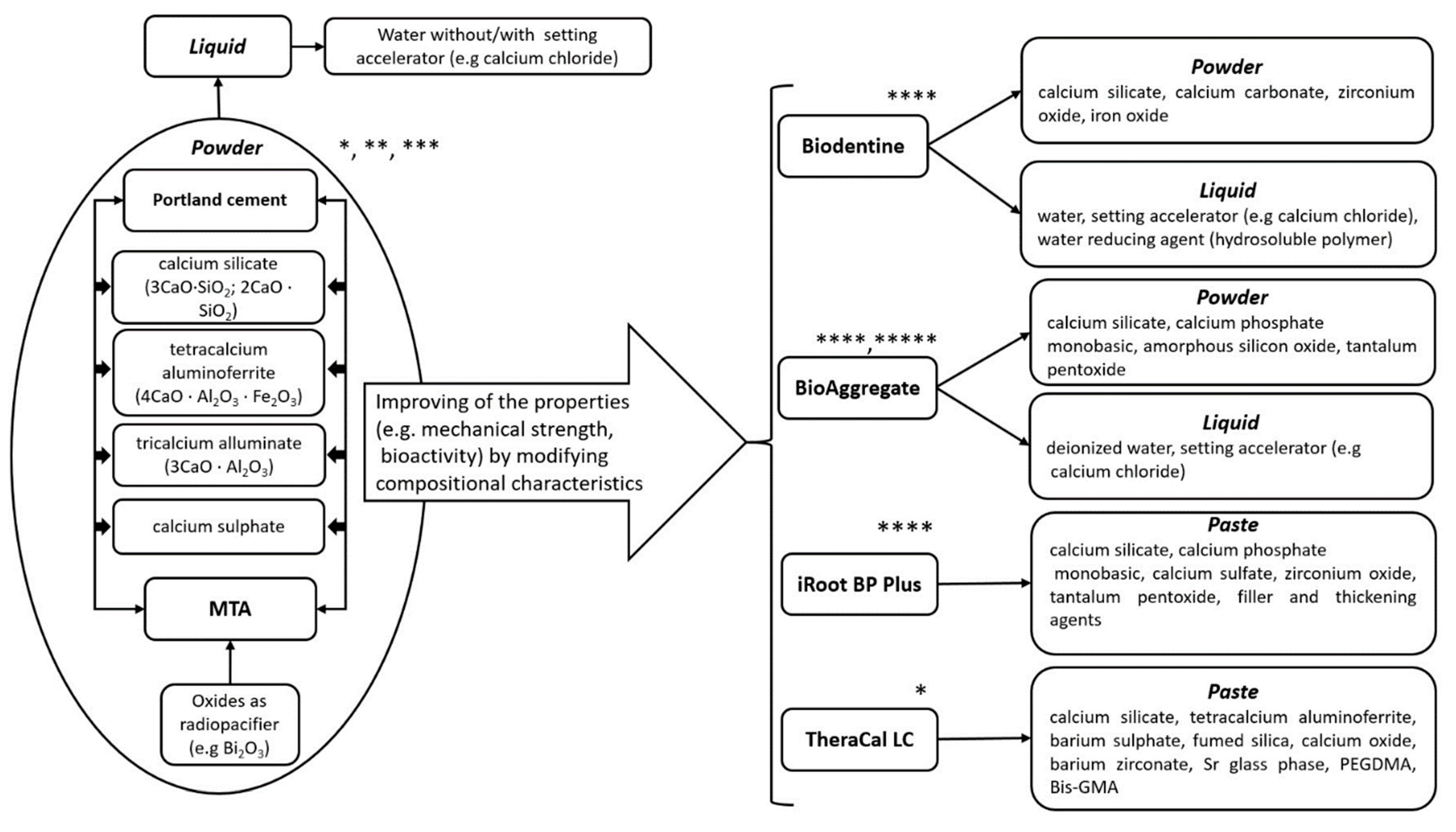
1.2. Calcium-Silicate Biomaterials as Pulp Capping Agents
1.3. Portland Cement (PC)
1.4. Mineral Trioxide Aggregate (MTA)
1.5. TheraCal LC
1.6. Biodentine
1.7. Bioceramic-Based Materials
1.8. Premixed Materials
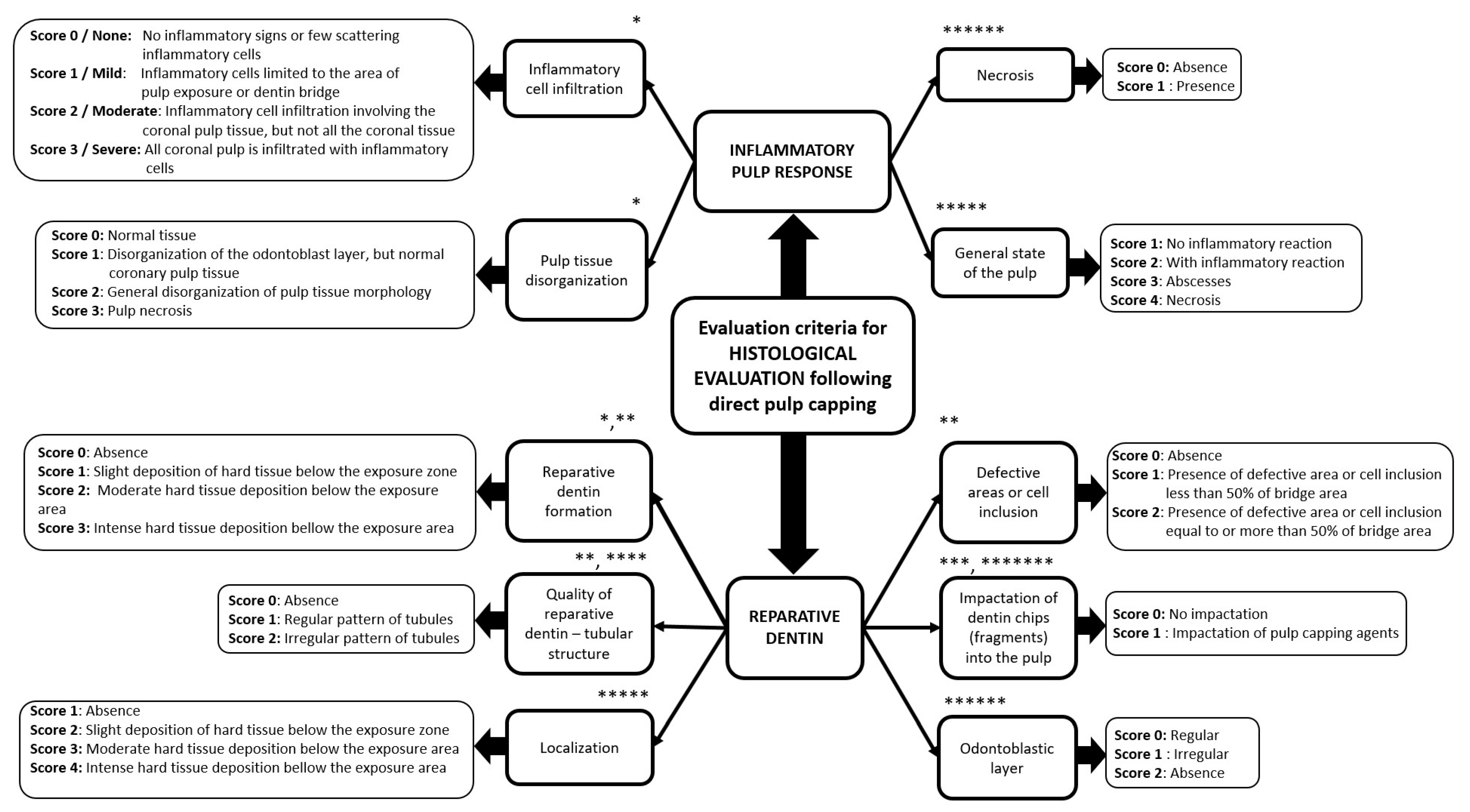
2. Histological Findings in Animal Model Following Direct Pulp Capping Procedure
Methodology
- original scientific studies;
- studies performed on animal subjects;
- studies reporting DPC with hydraulic calcium-silicates cements;
- studies reporting histological assessments, with highlights on inflammatory cell infiltrate, pulp tissue disorganization, reparative dentin formation, quality of reparative dentin, defective area or cell inclusion, the impact of dentin fragments.
- case reports, case series, letters to the editor, reviews;
- studies performed in vitro;
- studies that included indirect pulp capping or pulpotomy on animal models;
- non-English language publications.
3. Results
4. Discussions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Murray, P.E.; About, I.; Lumley, P.J.; Smith, G.A.Y.; Franquin, J.C.; Smith, A.J. Postoperative pulpal and repair responses. J. Am. Dent. Assoc. 2000, 131, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.E.; Windsor, L.J.; Smyth, T.W.; Hafez, A.A.; Cox, C.F. Analysis of pulpal reactions to restorative procedures, materials, pulp capping, and future therapies. Crit. Rev. Oral. Biol. Med. 2002, 13, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Didilescu, A.C.; Cristache, C.M.; Andrei, M.; Voicu, G.; Perlea, P. The effect of dental pulp-capping materials on hard-tissue barrier formation: A systematic review and meta-analysis. J. Am. Dent. Assoc. 2018, 149, 903–917.e904. [Google Scholar] [CrossRef]
- Cohenca, N.; Paranjpe, A.; Berg, J. Vital pulp therapy. Dent. Clin. N. Am. 2013, 57, 59–73. [Google Scholar] [CrossRef] [PubMed]
- Parolia, A.; Kundabala, M.; Rao, N.N.; Acharya, S.R.; Agrawal, P.; Mohan, M.; Thomas, M. A comparative histological analysis of human pulp following direct pulp capping with Propolis, mineral trioxide aggregate and Dycal. Aust. Dent. J. 2010, 55, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Aeinehchi, M.; Eslami, B.; Ghanbariha, M.; Saffar, A.S. Mineral trioxide aggregate (MTA) and calcium hydroxide as pulp-capping agents in human teeth: A preliminary report. Int. Endod. J. 2003, 36, 225–231. [Google Scholar] [CrossRef]
- Cho, S.Y.; Seo, D.G.; Lee, S.J.; Lee, J.; Lee, S.J.; Jung, I.Y. Prognostic factors for clinical outcomes according to time after direct pulp capping. J. Endod. 2013, 39, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.S. Orban’s Oral Histology and Embryology; Elsevier: Taramani, India, 2011. [Google Scholar]
- Fehrenbach, M.J.; Popowics, T. Illustrated Dental Embryology, Histology, and Anatomy; Saunders Elsevier: Philadelphia, PA, USA, 2015. [Google Scholar]
- Gronthos, S.; Brahim, J.; Li, W.; Fisher, L.W.; Cherman, N.; Boyde, A.; DenBesten, P.; Robey, P.G.; Shi, S. Stem cell properties of human dental pulp stem cells. J. Dent. Res. 2002, 81, 531–535. [Google Scholar] [CrossRef]
- Holland, G.R. Morphological features of dentine and pulp related to dentine sensitivity. Arch. Oral. Biol. 1994, 39, 3s–11s. [Google Scholar] [CrossRef]
- Yu, C.; Abbott, P.V. An overview of the dental pulp: Its functions and responses to injury. Aust. Dent. J. 2007, 52, S4–S16. [Google Scholar] [CrossRef]
- Smith, A.J.; Smith, J.G.; Shelton, R.M.; Cooper, P.R. Harnessing the Natural Regenerative Potential of the Dental Pulp. Dent. Clin. N. Am. 2012, 56, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Tziafas, D.; Smith, A.J.; Lesot, H. Designing new treatment strategies in vital pulp therapy. J. Dent. 2000, 28, 77–92. [Google Scholar] [CrossRef]
- Smith, A.J.; Cassidy, N.; Perry, H.; Bègue-Kirn, C.; Ruch, J.V.; Lesot, H. Reactionary dentinogenesis. Int. J. Dev. Biol. 1995, 39, 273–280. [Google Scholar]
- Mitsiadis, T.A.; Fried, K.; Goridis, C. Reactivation of Delta–Notch Signaling after Injury: Complementary Expression Patterns of Ligand and Receptor in Dental Pulp. Exp. Cell Res. 1999, 246, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Mitsiadis, T.A.; Rahiotis, C. Parallels between tooth development and repair: Conserved molecular mechanisms following carious and dental injury. J. Dent. Res. 2004, 83, 896–902. [Google Scholar] [CrossRef] [PubMed]
- da Rosa, W.L.O.; Piva, E.; da Silva, A.F. Disclosing the physiology of pulp tissue for vital pulp therapy. Int. Endod. J. 2018, 51, 829–846. [Google Scholar] [CrossRef] [PubMed]
- Simon, S.; Smith, A.J.; Berdal, A.; Lumley, P.J.; Cooper, P.R. The MAP Kinase Pathway Is Involved in Odontoblast Stimulation via p38 Phosphorylation. J. Endod. 2010, 36, 256–259. [Google Scholar] [CrossRef]
- Brizuela, C.; Ormeno, A.; Cabrera, C.; Cabezas, R.; Silva, C.I.; Ramirez, V.; Mercade, M. Direct Pulp Capping with Calcium Hydroxide, Mineral Trioxide Aggregate, and Biodentine in Permanent Young Teeth with Caries: A Randomized Clinical Trial. J. Endod. 2017, 43, 1776–1780. [Google Scholar] [CrossRef]
- Jalan, A.; Warhadpande, M.; Dakshindas, D. A comparison of human dental pulp response to calcium hydroxide and Biodentine as direct pulp-capping agents. J. Conserv. Dent. 2017, 20, 129–133. [Google Scholar] [CrossRef]
- Yildirim, S.; Can, A.; Arican, M.; Embree, M.C.; Mao, J.J. Characterization of dental pulp defect and repair in a canine model. Am. J. Dent. 2011, 24, 331–335. [Google Scholar] [PubMed]
- do Nascimento, A.B.; Fontana, U.F.; Teixeira, H.M.; Costa, C.A. Biocompatibility of a resin-modified glass-ionomer cement applied as pulp capping in human teeth. Am. J. Dent. 2000, 13, 28–34. [Google Scholar] [PubMed]
- Dammaschke, T.; Wolff, P.; Sagheri, D.; Stratmann, U.; Schäfer, E. Mineral trioxide aggregate for direct pulp capping: A histologic comparison with calcium hydroxide in rat molars. Quintessence Int. 2010, 41, e20–e30. [Google Scholar] [PubMed]
- Ford, T.R.P.; Torabinejad, M.; Abedi, H.R.; Bakland, L.K.; Kariyawasam, S.P. Using mineral trioxide aggregate as a pulp-capping material. J. Am. Dent. Assoc. 1996, 127, 1491–1494. [Google Scholar] [CrossRef] [PubMed]
- da Silva, L.A.; de Freitas, A.C.; de Carvalho, F.K.; de Queiroz, A.M.; Nelson-Filho, P.; Porto-Neto, S.T. Direct pulp capping with a self-etching adhesive system: Histopathologic evaluation in dogs’ teeth. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2009, 108, e34–e40. [Google Scholar] [CrossRef] [PubMed]
- Ishizaki, N.T.; Matsumoto, K.; Kimura, Y.; Wang, X.; Yamashita, A. Histopathological study of dental pulp tissue capped with enamel matrix derivative. J. Endod. 2003, 29, 176–179. [Google Scholar] [CrossRef] [PubMed]
- Dick, H.M.; Carmichael, D.J. Reconstituted antigen-poor collagen preparations as potential pulp-capping agents. J. Endod. 1980, 6, 641–644. [Google Scholar] [CrossRef]
- Aminabadi, N.A.; Farahani, R.M.; Oskouei, S.G. Formocresol versus calcium hydroxide direct pulp capping of human primary molars: Two year follow-up. J. Clin. Pediatr Dent. 2010, 34, 317–321. [Google Scholar] [CrossRef] [PubMed]
- Sübay, R.K.; Aşci, S. Human pulpal response to hydroxyapatite and a calcium hydroxide material as direct capping agents. Oral Surg. Oral. Med. Oral. Pathol. 1993, 76, 485–492. [Google Scholar] [CrossRef]
- Estrela, C.; Bammann, L.L.; Estrela, C.R.; Silva, R.S.; Pécora, J.D. Antimicrobial and chemical study of MTA, Portland cement, calcium hydroxide paste, Sealapex and Dycal. Braz. Dent. J. 2000, 11, 3–9. [Google Scholar]
- Guerreiro-Tanomaru, J.M.; Cornélio, A.L.G.; Andolfatto, C.; Salles, L.P.; Tanomaru-Filho, M. pH and Antimicrobial Activity of Portland Cement Associated with Different Radiopacifying Agents. ISRN Dentistry 2012, 2012, 469019. [Google Scholar] [CrossRef]
- Lewis, B. The obsolescence of formocresol. Br. Dent. J. 2009, 207, 525–528. [Google Scholar] [CrossRef] [PubMed]
- Tewari, S.; Tewari, S. Assessment of coronal microleakage in intermediately restored endodontic access cavities. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2002, 93, 716–719. [Google Scholar] [CrossRef] [PubMed]
- Torabinejad, M.; Hong, C.U.; Pitt Ford, T.R.; Kettering, J.D. Cytotoxicity of four root end filling materials. J. Endod. 1995, 21, 489–492. [Google Scholar] [CrossRef]
- Hume, W.R. An analysis of the release and the diffusion through dentin of eugenol from zinc oxide-eugenol mixtures. J. Dent. Res. 1984, 63, 881–884. [Google Scholar] [CrossRef] [PubMed]
- Glass, R.L.; Zander, H.A. Pulp healing. J. Dent. Res. 1949, 28, 97–107. [Google Scholar] [CrossRef]
- Hilton, T.J. Keys to clinical success with pulp capping: A review of the literature. Oper. Dent. 2009, 34, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Andelin, W.E.; Shabahang, S.; Wright, K.; Torabinejad, M. Identification of hard tissue after experimental pulp capping using dentin sialoprotein (DSP) as a marker. J. Endod. 2003, 29, 646–650. [Google Scholar] [CrossRef] [PubMed]
- Cox, C.F.; Sübay, R.K.; Ostro, E.; Suzuki, S.; Suzuki, S.H. Tunnel defects in dentin bridges: Their formation following direct pulp capping. Oper. Dent. 1996, 21, 4–11. [Google Scholar]
- Koike, T.; Polan, M.; Izumikawa, M.; Saito, T. Induction of Reparative Dentin Formation on Exposed Dental Pulp by Dentin Phosphophoryn/Collagen Composite. BioMed Res. Int. 2014, 2014, 745139. [Google Scholar] [CrossRef] [PubMed]
- Paranjpe, A.; Zhang, H.; Johnson, J.D. Effects of mineral trioxide aggregate on human dental pulp cells after pulp-capping procedures. J. Endod. 2010, 36, 1042–1047. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Ma, J.; Shen, Y.; Haapasalo, M.; Ruse, N.D.; Yang, Q.; Troczynski, T. In vitro studies of calcium phosphate silicate bone cements. J. Mater. Sci. Mater. Med. 2013, 24, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Steffen, R.; van Waes, H. Understanding mineral trioxide aggregate/Portland-cement: A review of literature and background factors. Eur. Arch. Paediatry Dent. 2009, 10, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Negm, A.; Hassanien, E.; Abu-Seida, A.; Nagy, M. Physical evaluation of a new pulp capping material developed from portland cement. J. Clin. Exp. Dent. 2016, 8, e278–e283. [Google Scholar] [CrossRef] [PubMed][Green Version]
- França, T.; Silva, R.; Queiroz, M.; Aguiar, C. Arsenic content in Portland cement: A literature review. Indian J. Dent. Res. 2010, 21, 591–595. [Google Scholar] [CrossRef] [PubMed]
- Duarte, M.A.; De Oliveira Demarchi, A.C.; Yamashita, J.C.; Kuga, M.C.; De Campos Fraga, S. Arsenic release provided by MTA and Portland cement. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2005, 99, 648–650. [Google Scholar] [CrossRef] [PubMed]
- Monteiro Bramante, C.; Demarchi, A.C.; de Moraes, I.G.; Bernadineli, N.; Garcia, R.B.; Spångberg, L.S.; Duarte, M.A. Presence of arsenic in different types of MTA and white and gray Portland cement. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2008, 106, 909–913. [Google Scholar] [CrossRef] [PubMed]
- De-Deus, G.; Coutinho-Filho, T. The use of white Portland cement as an apical plug in a tooth with a necrotic pulp and wide-open apex: A case report. Int. Endod. J. 2007, 40, 653–660. [Google Scholar] [CrossRef]
- Camilleri, J.; Montesin, F.E.; Brady, K.; Sweeney, R.; Curtis, R.V.; Ford, T.R. The constitution of mineral trioxide aggregate. Dent. Mater. 2005, 21, 297–303. [Google Scholar] [CrossRef]
- Sipert, C.R.; Hussne, R.P.; Nishiyama, C.K.; Torres, S.A. In vitro antimicrobial activity of Fill Canal, Sealapex, Mineral Trioxide Aggregate, Portland cement and EndoRez. Int. Endod. J. 2005, 38, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Marques, N.; Lourenço Neto, N.; Fernandes, A.P.; Rodini, C.; Hungaro Duarte, M.; Rios, D.; Machado, M.A.; Oliveira, T. Pulp tissue response to Portland cement associated with different radio pacifying agents on pulpotomy of human primary molars. J. Microsc. 2015, 260, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Bortoluzzi, E.A.; Broon, N.J.; Bramante, C.M.; Felippe, W.T.; Tanomaru Filho, M.; Esberard, R.M. The influence of calcium chloride on the setting time, solubility, disintegration, and pH of mineral trioxide aggregate and white Portland cement with a radiopacifier. J. Endod. 2009, 35, 550–554. [Google Scholar] [CrossRef] [PubMed]
- De-Deus, G.; Petruccelli, V.; Gurgel-Filho, E.; Coutinho-Filho, T. MTA versus Portland cement as repair material for furcal perforations: A laboratory study using a polymicrobial leakage model. Int. Endod. J. 2006, 39, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Nagy, M.M.; Tawfik, H.E.; Hashem, A.A.; Abu-Seida, A.M. Regenerative potential of immature permanent teeth with necrotic pulps after different regenerative protocols. J. Endod. 2014, 40, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Torabinejad, M.; Chivian, N. Clinical applications of mineral trioxide aggregate. J. Endod. 1999, 25, 197–205. [Google Scholar] [CrossRef]
- Patel, N.; Patel, K.; Baba, S.M.; Jaiswal, S.; Venkataraghavan, K.; Jani, M. Comparing Gray and White Mineral Trioxide Aggregate as a Repair Material for Furcation Perforation: An in Vitro Dye Extraction Study. J. Clin. Diagn. Res. 2014, 8, ZC70–ZC73. [Google Scholar] [CrossRef] [PubMed]
- Chng, H.K.; Islam, I.; Yap, A.U.; Tong, Y.W.; Koh, E.T. Properties of a new root-end filling material. J. Endod. 2005, 31, 665–668. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Siboni, F.; Prati, C. Chemical–physical properties of TheraCal, a novel light-curable MTA-like material for pulp capping. Int. Endod. J. 2012, 45, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Voicu, G.; Didilescu, A.C.; Stoian, A.B.; Dumitriu, C.; Greabu, M.; Andrei, M. Mineralogical and Microstructural Characteristics of Two Dental Pulp Capping Materials. Materials 2019, 12, 1772. [Google Scholar] [CrossRef]
- Arandi, N.Z.; Rabi, T. TheraCal LC: From Biochemical and Bioactive Properties to Clinical Applications. Int. J. Dent. 2018, 2018, 3484653. [Google Scholar] [CrossRef] [PubMed]
- Poggio, C.; Beltrami, R.; Colombo, M.; Ceci, M.; Dagna, A.; Chiesa, M. In vitro antibacterial activity of different pulp capping materials. J. Clin. Exp. Dent. 2015, 7, e584–e588. [Google Scholar] [CrossRef] [PubMed]
- Hebling, J.; Lessa, F.C.; Nogueira, I.; Carvalho, R.M.; Costa, C.A. Cytotoxicity of resin-based light-cured liners. Am. J. Dent. 2009, 22, 137–142. [Google Scholar] [PubMed]
- Bakhtiar, H.; Nekoofar, M.H.; Aminishakib, P.; Abedi, F.; Naghi Moosavi, F.; Esnaashari, E.; Azizi, A.; Esmailian, S.; Ellini, M.R.; Mesgarzadeh, V.; et al. Human Pulp Responses to Partial Pulpotomy Treatment with TheraCal as Compared with Biodentine and ProRoot MTA: A Clinical Trial. J. Endod. 2017, 43, 1786–1791. [Google Scholar] [CrossRef]
- Jeanneau, C.; Laurent, P.; Rombouts, C.; Giraud, T.; About, I. Light-cured Tricalcium Silicate Toxicity to the Dental Pulp. J. Endod. 2017, 43, 2074–2080. [Google Scholar] [CrossRef] [PubMed]
- Priyadarsini, S.; Mukherjee, S.; Mishra, M. Nanoparticles used in dentistry: A review. J. Oral. Biol. Craniofac. Res. 2018, 8, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Parirokh, M.; Torabinejad, M.; Dummer, P.M.H. Mineral trioxide aggregate and other bioactive endodontic cements: An updated overview—part I: Vital pulp therapy. Int. Endod. J. 2018, 51, 177–205. [Google Scholar] [CrossRef] [PubMed]
- Lipski, M.; Nowicka, A.; Kot, K.; Postek-Stefańska, L.; Wysoczańska-Jankowicz, I.; Borkowski, L.; Andersz, P.; Jarząbek, A.; Grocholewicz, K.; Sobolewska, E.; et al. Factors affecting the outcomes of direct pulp capping using Biodentine. Clin. Oral. Investig. 2018, 22, 2021–2029. [Google Scholar] [CrossRef]
- About, I. Biodentine: From biochemical and bioactive properties to clinical applications. G. Ital. Endod. 2016, 30, 81–88. [Google Scholar] [CrossRef]
- Malkondu, Ö.; Kazandağ, M.K.; Kazazoğlu, E. A Review on Biodentine, a Contemporary Dentine Replacement and Repair Material. BioMed Res. Int. 2014, 2014, 160951. [Google Scholar] [CrossRef]
- Loison-Robert, L.S.; Tassin, M.; Bonte, E.; Berbar, T.; Isaac, J.; Berdal, A.; Simon, S.; Fournier, B.P.J. In vitro effects of two silicate-based materials, Biodentine and BioRoot RCS, on dental pulp stem cells in models of reactionary and reparative dentinogenesis. PLoS ONE 2018, 13, e0190014. [Google Scholar] [CrossRef]
- Ree, D.M.; Schwartz, R. Clinical applications of bioceramic materials in endodontics. Endod. Pract. 2015, 7, 1–9. [Google Scholar]
- Jefferies, S. Bioactive and biomimetic restorative materials: A comprehensive review. Part II. J. Esthet. Restor Dent. 2014, 26, 27–39. [Google Scholar] [CrossRef]
- Nekoofar, M.H.; Stone, D.F.; Dummer, P.M. The effect of blood contamination on the compressive strength and surface microstructure of mineral trioxide aggregate. Int. Endod. J. 2010, 43, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Raghavendra, S.S.; Jadhav, G.R.; Gathani, K.M.; Kotadia, P. Bioceramics in endodontics—a review. J. Istanb. Univ. Fac. Dent. 2017, 51, S128–S137. [Google Scholar] [CrossRef]
- Šimundić Munitić, M.; Poklepović Peričić, T.; Utrobičić, A.; Bago, I.; Puljak, L. Antimicrobial efficacy of commercially available endodontic bioceramic root canal sealers: A systematic review. PLoS ONE 2019, 14, e0223575. [Google Scholar] [CrossRef] [PubMed]
- Demirkaya, K. Evaluation of micro surface structure and chemical composition of two different calcium silicate–containing filling materials. Turk. Endod. J. 2018. [Google Scholar] [CrossRef]
- Zhu, L.; Yang, J.; Zhang, J.; Peng, B. A comparative study of BioAggregate and ProRoot MTA on adhesion, migration, and attachment of human dental pulp cells. J. Endod. 2014, 40, 1118–1123. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Yang, X.; Fan, M. BioAggregate and iRoot BP Plus optimize the proliferation and mineralization ability of human dental pulp cells. Int. Endod. J. 2013, 46, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Woo, S.M.; Lee, B.N.; Koh, J.T.; Nör, J.E.; Hwang, Y.C. Effect of Biodentine and Bioaggregate on odontoblastic differentiation via mitogen-activated protein kinase pathway in human dental pulp cells. Int. Endod. J. 2015, 48, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Debelian, G.; Trope, M. The use of premixed bioceramic materials in endodontics. G. Ital. Endod. 2016, 30, 70–80. [Google Scholar] [CrossRef]
- Mahgoub, N.; Alqadasi, B.; Aldhorae, K.; Assiry, A.; Altawili, Z.M.; Tao, H. Comparison between iRoot BP Plus (EndoSequence Root Repair Material) and Mineral Trioxide Aggregate as Pulp-capping Agents: A Systematic Review. J. Int. Soc. Prev. Community Dent. 2019, 9, 542–552. [Google Scholar] [CrossRef] [PubMed]
- Rao, Q.; Kuang, J.; Mao, C.; Dai, J.; Hu, L.; Lei, Z.; Song, G.; Yuan, G. Comparison of iRoot BP Plus and Calcium Hydroxide as Pulpotomy Materials in Permanent Incisors with Complicated Crown Fractures: A Retrospective Study. J. Endod. 2020, 46, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Zamparini, F.; Siboni, F.; Prati, C.; Taddei, P.; Gandolfi, M.G. Properties of calcium silicate-monobasic calcium phosphate materials for endodontics containing tantalum pentoxide and zirconium oxide. Clin. Oral. Investig. 2019, 23, 445–457. [Google Scholar] [CrossRef] [PubMed]
- Lertmalapong, P.; Jantarat, J.; Srisatjaluk, R.L.; Komoltri, C. Bacterial leakage and marginal adaptation of various bioceramics as apical plug in open apex model. J. Investig. Clin. Dent. 2019, 10, e12371. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.J.N.L.; Carvalho, N.K.; Guberman, M.R.d.C.L.; Prado, M.; Senna, P.M.; Souza, E.M.; De-Deus, G. Push-out Bond Strength of Fast-setting Mineral Trioxide Aggregate and Pozzolan-based Cements: ENDOCEM MTA and ENDOCEM Zr. J. Endod. 2017, 43, 801–804. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Park, S.-J.; Lee, S.-H.; Hwang, Y.-C.; Yu, M.-K.; Min, K.-S. Biological Effects and Washout Resistance of a Newly Developed Fast-setting Pozzolan Cement. J. Endod. 2013, 39, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.-H.; Kang, M.; Ahn, S.; Kim, S.; Kim, W.; Kim, Y.; Kim, E. Tooth Discoloration after the Use of New Pozzolan Cement (Endocem) and Mineral Trioxide Aggregate and the Effects of Internal Bleaching. J. Endod. 2013, 39, 1598–1602. [Google Scholar] [CrossRef]
- Kim, M.; Yang, W.; Kim, H.; Ko, H. Comparison of the biological properties of ProRoot MTA, OrthoMTA, and Endocem MTA cements. J. Endod. 2014, 40, 1649–1653. [Google Scholar] [CrossRef]
- Han, L.; Kodama, S.; Okiji, T. Evaluation of calcium-releasing and apatite-forming abilities of fast-setting calcium silicate-based endodontic materials. Int. Endod. J. 2015, 48, 124–130. [Google Scholar] [CrossRef]
- Ashofteh Yazdi, K.; Ghabraei, S.; Bolhari, B.; Kafili, M.; Meraji, N.; Nekoofar, M.H.; Dummer, P.M.H. Microstructure and chemical analysis of four calcium silicate-based cements in different environmental conditions. Clin. Oral. Investig. 2019, 23, 43–52. [Google Scholar] [CrossRef]
- Parirokh, M.; Asgary, S.; Eghbal, M.J.; Kakoei, S.; Samiee, M. A comparative study of using a combination of calcium chloride and mineral trioxide aggregate as the pulp-capping agent on dogs’ teeth. J. Endod. 2011, 37, 786–788. [Google Scholar] [CrossRef]
- Cannon, M.; Gerodias, N.; Viera, A.; Percinoto, C.; Jurado, R. Primate pulpal healing after exposure and TheraCal application. J. Clin. Pediatr. Dent. 2014, 38, 333–337. [Google Scholar] [CrossRef]
- Kuratate, M.; Yoshiba, K.; Shigetani, Y.; Yoshiba, N.; Ohshima, H.; Okiji, T. Immunohistochemical analysis of nestin, osteopontin, and proliferating cells in the reparative process of exposed dental pulp capped with mineral trioxide aggregate. J. Endod. 2008, 34, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Bao, Z.F.; Liu, Y.; Zhang, D.D.; Chen, X.; Jiang, L.M.; Zhong, M. Comparison of in vivo dental pulp responses to capping with iRoot BP Plus and mineral trioxide aggregate. Int. Endod. J. 2016, 49, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Asgary, S.; Eghbal, M.J.; Parirokh, M.; Ghanavati, F.; Rahimi, H. A comparative study of histologic response to different pulp capping materials and a novel endodontic cement. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2008, 106, 609–614. [Google Scholar] [CrossRef]
- Danesh, F.; Vahid, A.; Jahanbani, J.; Mashhadiabbas, F.; Arman, E. Effect of white mineral trioxide aggregate compared with biomimetic carbonated apatite on dentine bridge formation and inflammatory response in a dental pulp model. Int. Endod. J. 2012, 45, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Zaen El-Din, A.M.; Hamama, H.H.; Abo El-Elaa, M.A.; Grawish, M.E.; Mahmoud, S.H.; Neelakantan, P. The effect of four materials on direct pulp capping: An animal study. Aust. Endod. J. 2020, 46, 249–256. [Google Scholar] [CrossRef]
- Akhavan, A.; Arbabzadeh, F.; Bouzari, M.; Razavi, S.M.; Davoudi, A. Pulp Response following Direct Pulp Capping with Dentin Adhesives and Mineral Trioxide Aggregate; An Animal Study. Iran. Endod. J. 2017, 12, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Pedano, M.S.; Camargo, B.; Hauben, E.; De Vleeschauwer, S.; Chen, Z.; De Munck, J.; Vandamme, K.; Van Landuyt, K.; Van Meerbeek, B. Experimental tricalcium silicate cement induces reparative dentinogenesis. Dent. Mater. 2018, 34, 1410–1423. [Google Scholar] [CrossRef]
- Tziafa, C.; Koliniotou-Koumpia, E.; Papadimitriou, S.; Tziafas, D. Dentinogenic responses after direct pulp capping of miniature swine teeth with Biodentine. J. Endod. 2014, 40, 1967–1971. [Google Scholar] [CrossRef]
- Shayegan, A.; Petein, M.; Vanden Abbeele, A. The use of beta-tricalcium phosphate, white MTA, white Portland cement and calcium hydroxide for direct pulp capping of primary pig teeth. Dent. Traumatol. 2009, 25, 413–419. [Google Scholar] [CrossRef]
- Paula, A.B.; Laranjo, M.; Marto, C.M.; Paulo, S.; Abrantes, A.M.; Fernandes, B.; Casalta-Lopes, J.; Marques-Ferreira, M.; Botelho, M.F.; Carrilho, E. Evaluation of dentinogenesis inducer biomaterials: An in vivo study. J. Appl. Oral. Sci. 2020, 28, e20190023. [Google Scholar] [CrossRef] [PubMed]
- Briso, A.L.; Rahal, V.; Mestrener, S.R.; Dezan Junior, E. Biological response of pulps submitted to different capping materials. Braz. Oral. Res. 2006, 20, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Faraco, I.M., Jr.; Holland, R. Response of the pulp of dogs to capping with mineral trioxide aggregate or a calcium hydroxide cement. Dent. Traumatol. 2001, 17, 163–166. [Google Scholar] [CrossRef]
- Negm, A.M.; Hassanien, E.E.; Abu-Seida, A.M.; Nagy, M.M. Biological evaluation of a new pulp capping material developed from Portland cement. Exp. Toxicol. Pathol. 2017, 69, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Gironés, J.; Alcaina-Lorente, A.; Ortiz-Ruiz, C.; Ortiz-Ruiz, E.; Pecci-Lloret, M.P.; Rodríguez-Lozano, F.J.; Martínez, C.M.; Ortiz-Ruiz, A.J. Melatonin as an Agent for Direct Pulp-Capping Treatment. Int. J. Environ. Res. Public Health 2020, 17, 1043. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.A.; Yang, Y.M.; Kwon, Y.S.; Hwang, Y.C.; Yu, M.K.; Min, K.S. Odontogenic effects of a fast-setting calcium-silicate cement containing zirconium oxide. Dent. Mater. J. 2015, 34, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-B.; Park, S.-J.; Kim, H.-H.; Kwon, Y.-S.; Lee, K.-W.; Min, K.-S. Physical properties and biological/odontogenic effects of an experimentally developed fast-setting α-tricalcium phosphate-based pulp capping material. BMC Oral. Health 2014, 14, 87. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Heo, S.M.; Hong, S.O.; Hwang, Y.C.; Lee, K.W.; Min, K.S. Odontogenic effect of a fast-setting pozzolan-based pulp capping material. J. Endod. 2014, 40, 1124–1131. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, S.; Dong, Y. Evaluation of a bioceramic as a pulp capping agent in vitro and in vivo. J. Endod. 2015, 41, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Long, Y.; Liu, S.; Zhu, L.; Liang, Q.; Chen, X.; Dong, Y. Evaluation of Pulp Response to Novel Bioactive Glass Pulp Capping Materials. J. Endod. 2017, 43, 1647–1650. [Google Scholar] [CrossRef]
- Moazzami, F.; Ghahramani, Y.; Tamaddon, A.M.; Dehghani Nazhavani, A.; Adl, A. A histological comparison of a new pulp capping material and mineral trioxide aggregate in rat molars. Iran. Endod. J. 2014, 9, 50–55. [Google Scholar] [PubMed]
- Simon, S.; Cooper, P.; Smith, A.; Picard, B.; Ifi, C.N.; Berdal, A. Evaluation of a new laboratory model for pulp healing: Preliminary study. Int. Endod. J. 2008, 41, 781–790. [Google Scholar] [CrossRef]
- Trongkij, P.; Sutimuntanakul, S.; Lapthanasupkul, P.; Chaimanakarn, C.; Wong, R.; Banomyong, D. Effects of the exposure site on histological pulpal responses after direct capping with 2 calcium-silicate based cements in a rat model. Restor. Dent. Endod. 2018, 43, e36. [Google Scholar] [CrossRef] [PubMed]
- Trongkij, P.; Sutimuntanakul, S.; Lapthanasupkul, P.; Chaimanakarn, C.; Wong, R.H.; Banomyong, D. Pulpal responses after direct pulp capping with two calcium-silicate cements in a rat model. Dent. Mater. J. 2019, 38, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Hanada, K.; Morotomi, T.; Washio, A.; Yada, N.; Matsuo, K.; Teshima, H.; Yokota, K.; Kitamura, C. In vitro and in vivo effects of a novel bioactive glass-based cement used as a direct pulp capping agent. J. Biomed. Mater. Res. B Appl. Biomater. 2019, 107, 161–168. [Google Scholar] [CrossRef]
- Mjör, I.A.; Dahl, E.; Cox, C.F. Healing of pulp exposures: An ultrastructural study. J. Oral. Pathol. Med. 1991, 20, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Călin, C.; Sajin, M.; Moldovan, V.T.; Coman, C.; Stratul, S.I.; Didilescu, A.C. Immunohistochemical expression of non-collagenous extracellular matrix molecules involved in tertiary dentinogenesis following direct pulp capping: A systematic review. Ann. Anat. Anat. Anz. 2021, 235, 151674. [Google Scholar] [CrossRef] [PubMed]
- Duarte, M.A.; Demarchi, A.C.; Yamashita, J.C.; Kuga, M.C.; Fraga Sde, C. pH and calcium ion release of 2 root-end filling materials. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2003, 95, 345–347. [Google Scholar] [CrossRef] [PubMed]
- Banava, S.; Fazlyab, M.; Heshmat, H.; Mojtahedzadeh, F.; Motahhary, P. Histological Evaluation of Single and Double-visit Direct Pulp Capping with Different Materials on Sound Human Premolars: A Randomized Controlled Clinical Trial. Iran. Endod. J. 2015, 10, 82–88. [Google Scholar] [PubMed]
- Takita, T.; Hayashi, M.; Takeichi, O.; Ogiso, B.; Suzuki, N.; Otsuka, K.; Ito, K. Effect of mineral trioxide aggregate on proliferation of cultured human dental pulp cells. Int. Endod. J. 2006, 39, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Stuart, K.G.; Miller, C.H.; Brown, C.E.; Newton, C.W. The comparative antimicrobial effect of calcium hydroxide. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 1991, 72, 101–104. [Google Scholar] [CrossRef]
- Robertson, A.; Andreasen, F.M.; Andreasen, J.O.; Norén, J.G. Long-term prognosis of crown-fractured permanent incisors. The effect of stage of root development and associated luxation injury. Int. J. Paediatry Dent. 2000, 10, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Petrou, M.A.; Alhamoui, F.A.; Welk, A.; Altarabulsi, M.B.; Alkilzy, M.; Splieth, C.H. A randomized clinical trial on the use of medical Portland cement, MTA and calcium hydroxide in indirect pulp treatment. Clin. Oral. Investig. 2014, 18, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Cao, L.; Fan, M.; Xu, Q. Direct Pulp Capping with Calcium Hydroxide or Mineral Trioxide Aggregate: A Meta-analysis. J. Endod. 2015, 41, 1412–1417. [Google Scholar] [CrossRef] [PubMed]
- Ulmansky, M.; Sela, J.; Sela, M. Scanning electron microscopy of calcium hydroxide induced bridges. J. Oral. Pathol. 1972, 1, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Straffon, L.H.; Corpron, R.L.; Bruner, F.W.; Daprai, F. Twenty-four-month clinical trial of visible-light-activated cavity liner in young permanent teeth. ASDC J. Dent. Child. 1991, 58, 124–128. [Google Scholar] [PubMed]
- Tran, X.V.; Gorin, C.; Willig, C.; Baroukh, B.; Pellat, B.; Decup, F.; Opsahl Vital, S.; Chaussain, C.; Boukpessi, T. Effect of a calcium-silicate-based restorative cement on pulp repair. J. Dent. Res. 2012, 91, 1166–1171. [Google Scholar] [CrossRef]
- Koc Vural, U.; Kiremitci, A.; Gokalp, S. Randomized Clinical Trial to Evaluate MTA Indirect Pulp Capping in Deep Caries Lesions After 24-Months. Oper. Dent. 2017, 42, 470–477. [Google Scholar] [CrossRef]
- Graham, L.; Cooper, P.R.; Cassidy, N.; Nor, J.E.; Sloan, A.J.; Smith, A.J. The effect of calcium hydroxide on solubilisation of bio-active dentine matrix components. Biomaterials 2006, 27, 2865–2873. [Google Scholar] [CrossRef]
- Nakashima, M. Bone morphogenetic proteins in dentin regeneration for potential use in endodontic therapy. Cytokine Growth Factor Rev. 2005, 16, 369–376. [Google Scholar] [CrossRef]
- Holland, R.; de Souza, V.; Nery, M.J.; Faraco Júnior, I.M.; Bernabé, P.F.; Otoboni Filho, J.A.; Dezan Júnior, E. Reaction of rat connective tissue to implanted dentin tube filled with mineral trioxide aggregate, Portland cement or calcium hydroxide. Braz. Dent. J. 2001, 12, 3–8. [Google Scholar] [PubMed]
- Holland, R.; de Souza, V.; Nery, M.J.; Otoboni Filho, J.A.; Bernabé, P.F.; Dezan Júnior, E. Reaction of rat connective tissue to implanted dentin tubes filled with mineral trioxide aggregate or calcium hydroxide. J. Endod. 1999, 25, 161–166. [Google Scholar] [CrossRef]
- Duque, C.; Hebling, J.; Smith, A.J.; Giro, E.M.; Oliveira, M.F.; de Souza Costa, C.A. Reactionary dentinogenesis after applying restorative materials and bioactive dentin matrix molecules as liners in deep cavities prepared in nonhuman primate teeth. J. Oral. Rehabil. 2006, 33, 452–461. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J. Vitality of the dentin-pulp complex in health and disease: Growth factors as key mediators. J. Dent. Educ. 2003, 67, 678–689. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Walboomers, X.F.; Jansen, J.A. The formation of tertiary dentin after pulp capping with a calcium phosphate cement, loaded with PLGA microparticles containing TGF-beta1. J. Biomed. Mater. Res. A 2008, 85, 439–444. [Google Scholar] [CrossRef]
- Schwendicke, F.; Brouwer, F.; Stolpe, M. Calcium Hydroxide versus Mineral Trioxide Aggregate for Direct Pulp Capping: A Cost-effectiveness Analysis. J. Endod. 2015, 41, 1969–1974. [Google Scholar] [CrossRef] [PubMed]
- Farrugia, C.; Baca, P.; Camilleri, J.; Arias Moliz, M.T. Antimicrobial activity of ProRoot MTA in contact with blood. Sci. Rep. 2017, 7, 41359. [Google Scholar] [CrossRef]
- Lee, B.-N.; Chun, S.-J.; Chang, H.-S.; Hwang, Y.-C.; Hwang, I.-N.; Oh, W.-M. Physical properties and biological effects of mineral trioxide aggregate mixed with methylcellulose and calcium chloride. J. Appl. Oral. Sci. 2017, 25, 680–688. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Perez-Heredia, M.; Clavero-González, J.; Marchena-Rodríguez, L. Use of melatonin in oral health and as dental premedication. J. Biol. Res. (Thessalon) 2015, 22, 13. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Asgary, S.; Parirokh, M.; Eghbal, M.J.; Brink, F. A comparative study of white mineral trioxide aggregate and white Portland cements using X-ray microanalysis. Aust. Endod. J. 2004, 30, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Funteas, U.; Wallace, J.A.; Fochtman, E. A comparative analysis of Mineral Trioxide Aggregate and Portland cement. Aust. Endod. J. 2003, 29, 43–44. [Google Scholar] [CrossRef] [PubMed]
- Ber, B.S.; Hatton, J.F.; Stewart, G.P. Chemical Modification of ProRoot MTA to Improve Handling Characteristics and Decrease Setting Time. J. Endod. 2007, 33, 1231–1234. [Google Scholar] [CrossRef] [PubMed]
- Min, K.-S.; Kim, H.-I.; Park, H.-J.; Pi, S.-H.; Hong, C.-U.; Kim, E.-C. Human Pulp Cells Response to Portland Cement In Vitro. J. Endod. 2007, 33, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.G.d.; Xavier, C.B.; Demarco, F.F.; Pinheiro, A.L.B.; Costa, A.T.; Pozza, D.H. Comparative chemical study of MTA and portland cements. Braz. Dent. J. 2007, 18, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, J. The chemical composition of mineral trioxide aggregate. J. Conserv. Dent. 2008, 11, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Zakerzadeh, A.; Esnaashari, E.; Dadfar, S. In Vitro Comparison of Cytotoxicity and Genotoxicity of Three Vital Pulp Capping Materials. Iran. Endod. J. 2017, 12, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Lee, D.; Song, D.; Kim, H.M.; Kim, S.Y. Biocompatibility and Bioactivity of Set Direct Pulp Capping Materials on Human Dental Pulp Stem Cells. Materials 2020, 13, 3925. [Google Scholar] [CrossRef]
- Poggio, C.; Ceci, M.; Dagna, A.; Beltrami, R.; Colombo, M.; Chiesa, M. In vitro cytotoxicity evaluation of different pulp capping materials: A comparative study. Arh. Hig. Rada Toksikol. 2015, 66, 181–188. [Google Scholar] [CrossRef]
- Nowicka, A.; Lipski, M.; Parafiniuk, M.; Sporniak-Tutak, K.; Lichota, D.; Kosierkiewicz, A.; Kaczmarek, W.; Buczkowska-Radlińska, J. Response of Human Dental Pulp Capped with Biodentine and Mineral Trioxide Aggregate. J. Endod. 2013, 39, 743–747. [Google Scholar] [CrossRef]
- Luo, Z.; Kohli, M.R.; Yu, Q.; Kim, S.; Qu, T.; He, W.X. Biodentine induces human dental pulp stem cell differentiation through mitogen-activated protein kinase and calcium-/calmodulin-dependent protein kinase II pathways. J. Endod. 2014, 40, 937–942. [Google Scholar] [CrossRef]
- Collado-González, M.; García-Bernal, D.; Oñate-Sánchez, R.E.; Ortolani-Seltenerich, P.S.; Álvarez-Muro, T.; Lozano, A.; Forner, L.; Llena, C.; Moraleda, J.M.; Rodríguez-Lozano, F.J. Cytotoxicity and bioactivity of various pulpotomy materials on stem cells from human exfoliated primary teeth. Int. Endod. J. 2017, 50 (Suppl. S2), e19–e30. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Ikeda, H.; Suda, H. Sympathetic Regulation of Tertiary Dentinogenesis via Beta-2 Adrenergic Receptor on Rat Odontoblasts. J. Endod. 2015, 41, 1056–1060. [Google Scholar] [CrossRef] [PubMed]
- Tziafa, C.; Koliniotou-Koumpia, E.; Papadimitriou, S.; Tziafas, D. Dentinogenic Activity of Biodentine in Deep Cavities of Miniature Swine Teeth. J. Endod. 2015, 41, 1161–1166. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, R.V.; Chisini, L.A.; Ferruá, C.P.; Guiraldo, R.D.; Gonini-Júnior, A.; Moura, S.K.; Tarquínio, S.B.; Demarco, F.F. The influence of concentration of HEMA on degree of conversion and cytotoxicity of a dental bonding resin. Minerva Stomatol. 2016, 65, 65–71. [Google Scholar] [PubMed]
- Górecka, V.; Suliborski, S.; Biskupski, T. Direct pulp capping with a dentin adhesive resin system in children’s permanent teeth after traumatic injuries: Case reports. Quintessence Int. 2000, 31, 241–248. [Google Scholar]
- Heitmann, T.; Unterbrink, G. Direct pulp capping with a dentinal adhesive resin system: A pilot study. Quintessence Int. 1995, 26, 765–770. [Google Scholar] [PubMed]
- Goldberg, M.; Njeh, A.; Uzunoglu, E. Is Pulp Inflammation a Prerequisite for Pulp Healing and Regeneration? Mediat. Inflamm. 2015, 2015, 347649. [Google Scholar] [CrossRef] [PubMed]
| Search Strategy Pubmed Database | |
|---|---|
| #1 | ((((“Animals, Laboratory”[Mesh]) OR “Animals, Laboratory/drug effects”[Mesh]) OR (animal AND testing)) OR (laboratory animal)) OR (animal AND laboratory) |
| #2 | (((“Dental Pulp Capping”[Mesh]) OR (Dental Pulp Capping)) OR (“Dental Pulp Capping/adverse effects”[Mesh])) OR ((((“Dental Pulp Exposure”[Mesh]) OR “Dental Pulp Exposure/drug therapy”[Mesh]) OR “Dental Pulp Exposure/physiopathology”[Mesh]) OR “Dental Pulp Exposure/adverse effects”[Mesh]) OR (dental pulp) |
| #3 | (((((“Dentinogenesis”[Mesh]) OR “Dentinogenesis/drug effects”[Mesh]) OR “Dentinogenesis/physiology”[Mesh]) OR (dentinogenesis)) OR (tertiary dentin)) OR (dentin) |
| #4 | (((((“Histological Techniques/analysis”[Mesh] OR “Histological Techniques/diagnosis”[Mesh] OR “Histological Techniques/drug effects”[Mesh])) OR (histologic)) OR (histology)) OR (histocytologic)) |
| #5 | ((((((((pulp-capping agent[MeSH Terms]) OR ((((“mineral trioxide aggregate” [Supplementary Concept]) OR “Calcium Compounds”[Mesh]) OR “Calcium Compounds/adverse effects”[Mesh]) OR “Calcium Compounds/therapeutic use”[Mesh])) OR (MTA cement)) OR (Aggregate ProRoot)) OR (Tricalcium Silicate)) OR (Biodentine)) OR (“accelerated Portland cement” [Supplementary Concept])) OR (Portland cement)) OR (MTA) |
| #6 | #1 AND #2 AND (#3 OR #4) AND #5 AND (English(Filter)) |
| Publication. | DPC Agent | Animal Type | Teeth Type | Histological Evaluation |
|---|---|---|---|---|
| Guerrero-Gironés, et al., 2020 [107] | Melatonin (5 mg, Sigma-Aldrich, St. Louis, MA, USA) MTA (ProRoot MTA, Dentsply Maillefer, Ballagues, Switzerland) MTA and melatonin Melatonin + melatonin | Sprague Dawley rats | First and secondary maxillary molars | 30 days follow-up
|
| Paula, et al., 2020 [103] | White ProRoot MTA (Dentsply Tulsa Dental Specialties, Tulsa, OK, USA) Biodentine (Septodont, France) Positive control group: Glass ionomer cement (Ketac Fil Plus Aplicab, 3M Espe, USA) Negative control groups (No intervention performed) | Wistar Hun rats | First mandibular molars | 3 days follow-up
|
| Hanada, et al., 2019 [117] | Bioactive glass cement * (Nishika Canal Sealer BG/NCS-BG; Nippon Shika Yakunin, Yamaguchi, Japan), NSY-222-S—modified from NCS-BG, WMTA (WMTA ProRoot Dentsply, Tulsa Dental, OK, USA) CH (Dycal, Dentsply Caulk Milford, DE) | Wistar Rats | Maxillary first molars | 1st day follow-up
|
| Trongkij, et al., 2019 [116] | White ProRoot MTA (Dentsply Tulsa Dental Specialties, Tulsa, OK, USA) Bio-MA (M-Dent/SCG, Bangkok, Thailand) Positive control group (uncapped pulp exposure) Negative control groups (intact teeth) | Wistar rats | Maxillary first molars | First-day follow-up
|
| Trongkij, et al., 2018 [115] | White ProRoot MTA (Dentsply Tulsa Dental Specialties, Tulsa, OK, USA) Bio-MA ** (M-Dent/SCG, Bangkok, Thailand) Positive control group (pulp exposure without capping material) Negative control group (intact teeth with no preparation) | Wistar rats | Maxillary first molars | First-day follow-up
|
| Long, et al., 2017 [112] | MTA (ProRoot MTA, Dentsply, Sirona, Tulsa, OK) Novel bioactive glass: *** phosphate buffer solution solely (BG-PB) and phosphate buffer solution in addition with 1 wt.% sodium alginate (BG-PB-SA) Negative control group No DPC was applied, the cavity was sealed with a glass-ionomer cement (Fuji IX, GC International, Tokyo, Japan) | Wistar rats | Maxillary first molars | One week
|
| Liu, et al., 2015 [111] | iRoot BP Plus Innovative BioCeramix, Vancouver, BC, Canada) MTA (Dentsply, Tulsa Dental, Tulsa, OK, USA) Glass ionomer cement (Control group) (Fuji IX, GC International, Tokyo, Japan) | Wistar rats | Maxillary first molars | 1 week follow-up
|
| Kim, et al., 2015 [108] | Endocem Zr (Maruchi, Wonju, Korea) MTA (ProRoot, Dentsply, Tulsa Dental, Tulsa, OK, USA) Light-cured glass-ionomer cement (Control group) (Fuji II LC, GC, Tokyo, Japan) | Wistar rats | Maxillary first molars | 4 weeks follow-up
|
| Lee, et al., 2014 [109] | α-tricalcium phosphate-based (α-TCP; Mediclus, Cheongju, Korea) MTA (ProRoot MTA, Dentsply, Tulsa Dental, Tulsa, OK, USA) Light-cured glass-ionomer cement (control group) (Fuji II LC; GC, Tokyo, Japan) | Wistar rats | Maxillary first molars | 4 weeks follow-up
|
| Moazzami, et al., 2014 [113] | Odontoblastic differentiating material (ODM) **** MTA (ProRoot MTA, Dentsply, Tulsa Dental, Tulsa, OK, USA) 2 control groups: teeth DPC with ODM without active ingredient and with a light-cured glass-ionomer cement (GC International, Tokyo, Japan) | Sprague Dawley rats | Maxillary molars | 2 weeks follow-up
|
| Park, et al., 2014 [110] | Endocem (Maruchi, Wonju, Korea) MTA (ProRoot, Dentsply, Tulsa Dental, Tulsa, OK, USA) Light-cured glass-ionomer cement Control group (Fuji II LC, GC, Tokyo, Japan) | Rats | Maxillary first molars | 4 weeks follow-up
|
| Kuratate, et al., 2008 [94] | WMTA (white ProRoot MTA, Dentsply Tulsa Dental, Tulsa, OK) Negative control group | Rats | Maxillary first molars | 1st day follow-up
|
| Simon, et al., 2008 [114] | MTA (ProRoot MTA, Dentsply, Tulsa Dental, Tulsa, OK, USA) Light-cured resin Control group (Point4, Kerr Hawe, Bioggio, Switzerland) | Mice | Maxillary first molars | 2 weeks follow-up
|
| Publication. | DPC Agent | Animal Type | Teeth Type | Histological Evaluation |
|---|---|---|---|---|
| Zaen El-Din, et al., 2020 [98] | MTA (ProRoot White MTA, Dentsply, Sirona) Biodentine (Septodont, Saint-Maur-des-Fossés, France) Nano-hydroxyapatite (nHAP) * CH (Dycal, Dentply Sirona Endodontics) | Dogs | Anterior teeth | 7 days follow-up
|
| Akhavan, et al., 2017 [99] | MTA (ProRoot MTA, Dentsply, Sirona, Tulsa, OK), Dentin adhesives: Clearfil S3/CS3, Bond (Kuraray, Osaka, Japan); Optibond (FL/OBF, Kerr, Orange, CA, USA); Single Bond/SB, (3M, ESPE, MN, USA); Clearfil SE/CSE Bond (Kuraray, Tokyo, Japan) CH (Dycal, Dentsply, Germany) | Dogs | Premolars, canines, first, second and third incisors | 7 days follow-up
|
| Negm, et al., 2017 [106] | MTA (Endocem Maruchi, Korea) Port Cal ** PC with 20% bismuth oxide | Dogs | N/A | 3 weeks follow-up
|
| Shi, et al., 2016 [95] | iRoot BP Plus (Innovative BioCeramix, Vancouver, BC, Canada) MTA (ProRoot MTA, Dentsply, Tulsa Dental, Tulsa, OK, USA) | Beagle dogs | Maxillary and mandibular incisors | 3 months follow-up
|
| Danesh, et al., 2012 [97] | BCAp *** (biomimetic carbonated apatite) MTA (ProRoot MTA, Dentsply, Tulsa Dental, Tulsa, OK, USA) | Beagle dogs | Canines | 7 days follow-up
|
| Parirokh, et al., 2011 [92] | MTA (ProRoot MTA, Dentsply, Tulsa Dental, Tulsa, OK, USA) MTA With addition of calcium chloride | Dogs | Lateral incisor, canines and premolars | 2 months follow-up
|
| Asgary, et al., 2008 [96] | MTA (ProRoot MTA, Dentsply, Tulsa Dental, Tulsa, OK, USA) CH (Dycal, LD Caulk, Milford, DE) NEC **** (Novel endodontic cement) | Beagle dogs | Canines | 8 weeks follow-up
|
| Briso, et al., 2006 [104] | MTA (ProRoot MTA, Dentsply, Tulsa Dental, Tulsa, OK, USA) CH (Reagen, Quimibras, Rio de Janeiro, Brazil) | Mongrel dogs | N/A | 60 days follow-up
|
| Faraco and Holland, 2001 [105] | MTA (Dentsply Tulsa, Tulsa, OK, USA) CH (Dycal, L.D. Caulk, Milford, DE) | Dogs | N/A | 2 months follow-up
|
| Publication. | DPC Agent | Animal Type | Teeth Type | Histological Evaluation |
| Li, et al., 2018 [100] | MTA (ProRoot MTA, Dentsply, Sirona, Tulsa, OK, USA), TheraCal LC (Bisco, Schaumburg, IL, USA) TCS 50 * | Gottingen minipigs | Incisors, canines, premolars and molars | 7 days follow-up
|
| Tziafa, et al., 2014 [101] | Biodentine Experimental group (Septodont, Saint-Maur-des-Fossés, France) MTA Control group (Angelus, Londrina, PR, Brazil) | Miniature swine pigs | Incisors, canines, premolars, molars | 3 weeks follow-up
|
| Shayegan, et al., 2009 [102] | Beta-tricalcium phosphate (beta-TCP) (RTR, Septodont) White MTA (Dentsply, DeTrey GmbH) White PC (Cantillana, Belgium) CH (Dentsply, DeTrey GmbH) | Pigs | Incisors, maxillary and mandibular molars | 3 weeks follow-up
|
| Publication. | DPC Agent | Animal Type | Teeth Type | Histological Evaluation |
|---|---|---|---|---|
| Cannon, et al., 2014 [93] | TheraCal LC (Bisco, Schaumburg, IL, USA) Pure PC with 2% Chlorhexidine solution, Glass ionomer cement (Triage, Fuji VII, GC, USA) CH (Dycal, Dentsply) | Primate (Capucin Cebus Opella) | 3 teeth in each quadrant | 4 weeks follow-up
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andrei, M.; Vacaru, R.P.; Coricovac, A.; Ilinca, R.; Didilescu, A.C.; Demetrescu, I. The Effect of Calcium-Silicate Cements on Reparative Dentinogenesis Following Direct Pulp Capping on Animal Models. Molecules 2021, 26, 2725. https://doi.org/10.3390/molecules26092725
Andrei M, Vacaru RP, Coricovac A, Ilinca R, Didilescu AC, Demetrescu I. The Effect of Calcium-Silicate Cements on Reparative Dentinogenesis Following Direct Pulp Capping on Animal Models. Molecules. 2021; 26(9):2725. https://doi.org/10.3390/molecules26092725
Chicago/Turabian StyleAndrei, Mihai, Raluca Paula Vacaru, Anca Coricovac, Radu Ilinca, Andreea Cristiana Didilescu, and Ioana Demetrescu. 2021. "The Effect of Calcium-Silicate Cements on Reparative Dentinogenesis Following Direct Pulp Capping on Animal Models" Molecules 26, no. 9: 2725. https://doi.org/10.3390/molecules26092725
APA StyleAndrei, M., Vacaru, R. P., Coricovac, A., Ilinca, R., Didilescu, A. C., & Demetrescu, I. (2021). The Effect of Calcium-Silicate Cements on Reparative Dentinogenesis Following Direct Pulp Capping on Animal Models. Molecules, 26(9), 2725. https://doi.org/10.3390/molecules26092725








