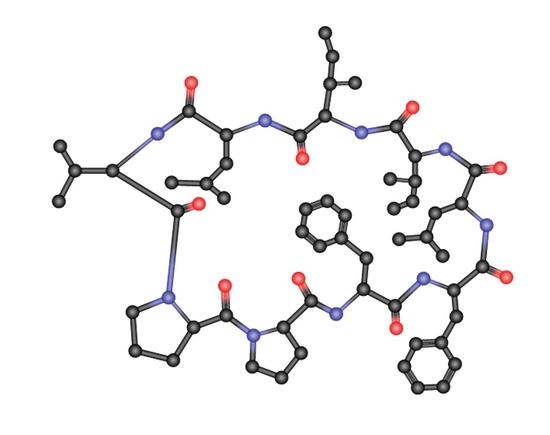
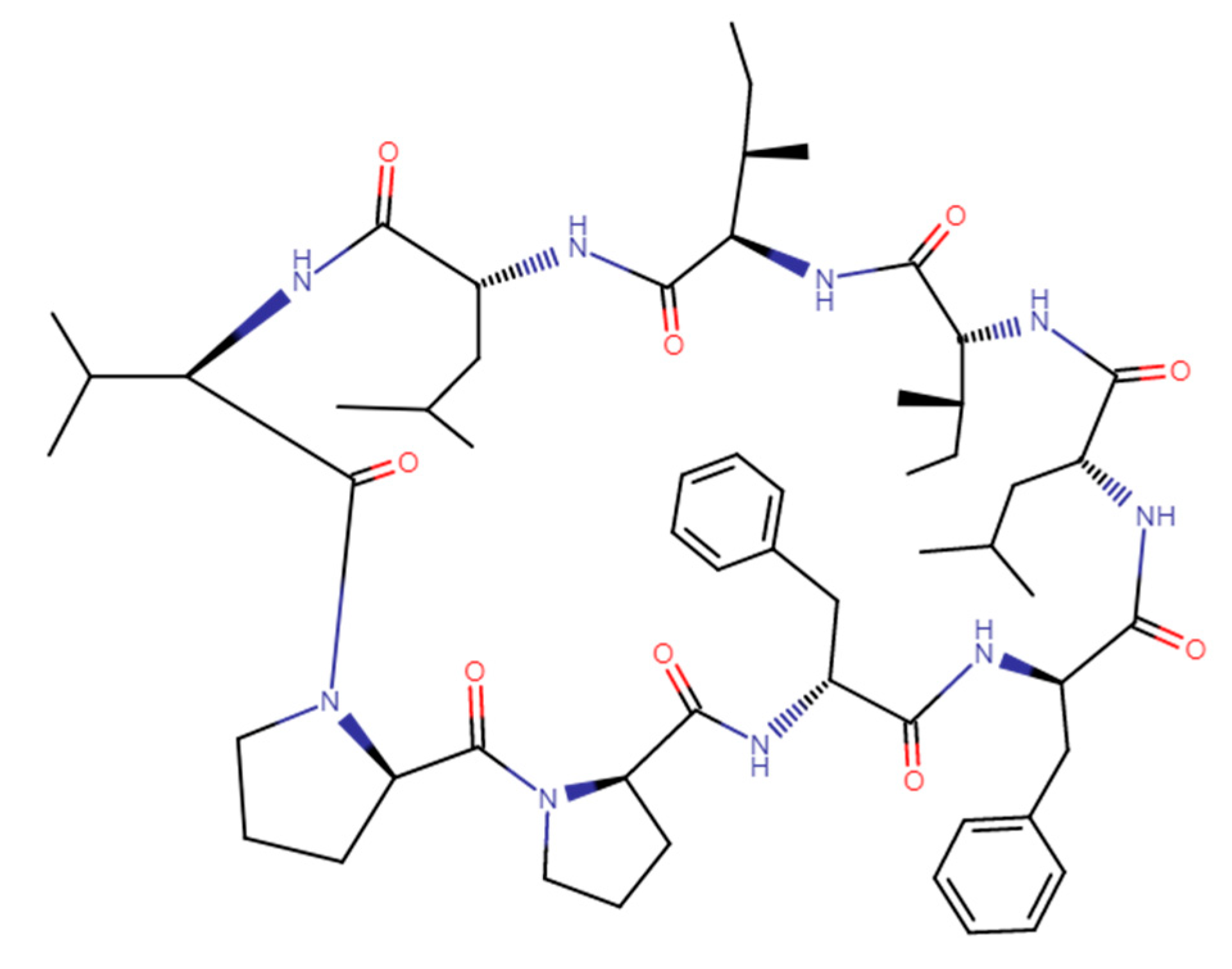
Effects of Modifications on the Immunosuppressive Properties of Cyclolinopeptide A and Its Analogs in Animal Experimental Models
Abstract
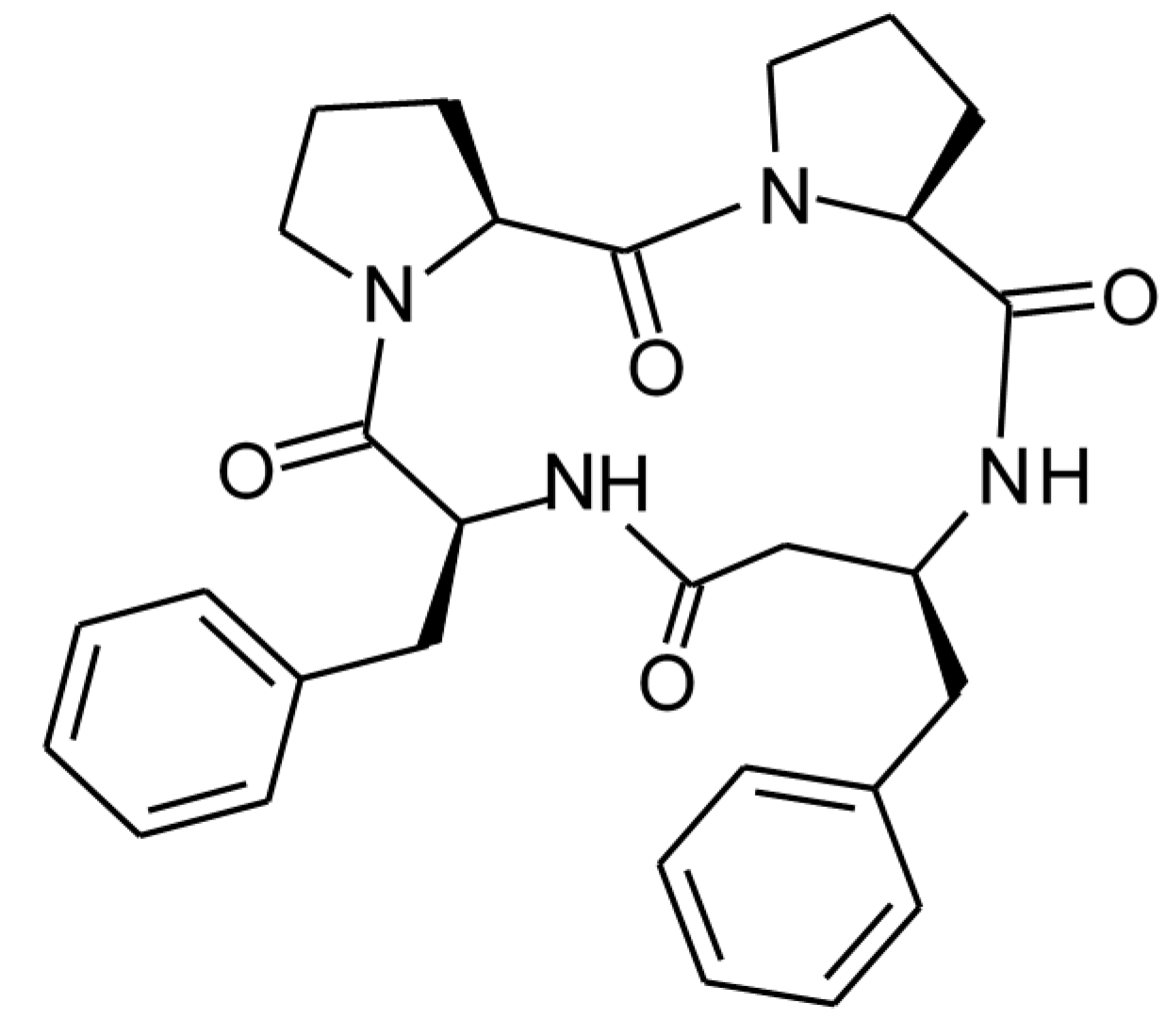
1. Introduction
2. The Effects of CLA on the Humoral and Cellular Immune Responses
3. Role of Single Amino Acids in the Activity of CLA Analogs
4. Effects of Modified Amino Acids on Activity and Establishment of a Key Pro-Pro-Phe-Phe Sequence in CLA Analogs
5. Significance of the Spatial Conformation in the Activity of CLA
6. Modifications of CLA Structure by Unnatural Aminoacids
- absence of Leu1 is not important for CLA activity
- Pro6-Pro7-Phe8-Phe9 sequence is essential for the immunosuppressive activity in native CLA
- Leu, Ileu and Val may be replaced by Thr without loss of CLA activity
- Phe8 or both Phe8 and Phe9 may be replaced by Tyr without loss of CLA activity
- sulfonated Phe or tetrazole derivatives of Val5-Pro6 and Pro6-Pro7 do not alter CLA activity
- substitution with hydroxymethylleucine at position 1 or 4 or with hydroxymethylvaline4 do not significantly affect suppressive activity of CLA
- edge-to-face interaction of the phenylalanine rings is not critical for CLA activity
- CLA modified with β3-homophenylalanine8 enhances its anti-proliferative action
- cyclic peptides containing (S) or (R)-ɣ3-bis-homophenylalanine8 are strongly anti-proliferative but linear ones are inhibitory only in LPS-induced TNF α production
- anti-proliferative actions of CLA analogs modified with (R)- or (S)-4-methylpseudoproline at position 6 or 7 are enhanced and associated with proapoptotic activity
- the cyclic tetrapeptide loses the anti-proliferative activity, but acquires strong anti-inflammatory property associated with its regulatory effect on prostanoid metabolism
7. Modification of Suppressive Action of Methotrexate by CLA Analogs
8. Activity of c(Pro-Pro-β3hPhe-Phe-), the Cyclic Tetrapeptide
9. Other Biological Activities of CLA
10. The Mechanism of Action
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Kaufmann, H.P.; Tobschirbel, A. Über ein Oligopeptid aus Leinsamen. Chem. Ber. 1959, 92, 2805–2809. [Google Scholar] [CrossRef]
- Ansari, R.; Zarshenas, M.M.; Dadbakhsh, A.H. A Review on Pharmacological and Clinical Aspects of Linum usitatissimum L. Curr. Drug Discov. Technol. 2019, 16, 148–158. [Google Scholar] [CrossRef]
- Shim, Y.Y.; Song, Z.; Jadhav, P.D.; Reaney, M.J. Orbitides from flaxseed (Linum usitatissimum L.): A comprehensive review. Trends Food Sci. Technol. 2019, 93, 197–211. [Google Scholar] [CrossRef]
- Wieczorek, Z.; Bengtsson, B.; Trojnar, J.; Siemion, I.Z. Immunosuppressive activity of cyclolinopeptide A. Pept. Res. 1991, 4, 275–283. [Google Scholar]
- Siemion, I.Z.; Pedyczak, A.; Strug, I.; Wieczorek, Z. Synthesis and biological studies on analogs of cyclolinopeptide A with a shortened peptide chain. Arch. Immunol. Ther. Exp. 1994, 42, 459–465. [Google Scholar]
- Siemion, I.Z.; Cebrat, M.; Lisowski, M.; Zimecki, M.; Wieczorek, Z. Immunosuppressive activity of threonine-containing analogues of cyclolinopeptide A. Arch. Immunol. Ther. Exp. 1992, 40, 257–261. [Google Scholar]
- Wieczorek, Z.; Pedyczak, A.; Bodalski, T.; Lisowski, M.; Trojnar, J.; Zimecki, M.; Siemion, I.Z. Immunosuppressive activity of tyrosine analogues of cyclolinopeptide A. Arch. Immunol. Ther. Exp. 1992, 40, 213–216. [Google Scholar]
- Wieczorek, Z.; Zimecki, M.; Pedyczak, A.; Lisowski, M.; Siemion, I.Z. Immunosuppressive activity of alanine analogs of cyclolinopeptide A. Arch. Immunol. Ther. Exp. 1993, 41, 291–296. [Google Scholar]
- Siemion, I.Z.; Cebrat, M.; Pédyczak, A.; Zimecki, M.; Wieczorek, Z. Synthesis and immunosuppressive activity of glycine containing linear analogs of cyclolinopeptide A. Arch. Immunol. Ther. Exp. 1993, 41, 285–289. [Google Scholar]
- Cebrat, M.; Lisowski, M.; Siemion, I.Z.; Zimecki, M.; Wieczorek, Z. Sulfonated analogues of cyclolinopeptide A Synthesis, immunosuppressive activity and CD studies. J. Pept. Res. 2009, 49, 415–420. [Google Scholar] [CrossRef]
- Kaczmarek, K.; Jankowski, S.; Siemion, I.Z.; Wieczorek, Z.; Benedetti, E.; Di Lello, P.; Isernia, C.; Saviano, M.; Zabrocki, J. Tetrazole analogues of cyclolinopeptide A: Synthesis, conformation, and biology. Biopolymers 2002, 63, 343–357. [Google Scholar] [CrossRef] [PubMed]
- Zubrzak, P.; Banaś, A.; Kaczmarek, K.; Leplawy, M.T.; Sochacki, M.; Kowalski, M.L.; Szkudlińska, B.; Zabrocki, J.; Di Lello, P.; Isernia, C.; et al. Analogues of cyclolinopeptide a containing?-hydroxymethyl amino acid residues. Biopolymers 2005, 80, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, K.; Farina, B.; Zubrzak, P.; Jankowski, S.; Zimecki, M.; Suder, P.; Benedetti, E.; Fattorusso, R.; Saviano, M.; Zabrocki, J. Synthesis, conformational analysis and immunological activity of β3Phe-substituted Cyclolinopeptide A analogues. J. Pept. Sci. 2009, 15, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Katarzyńska, J.; Mazur, A.; Bilska, M.; Adamek, E.; Zimecki, M.; Jankowski, S.; Zabrocki, J. Synthesis and immunosuppressive activity of new cyclolinopeptide A analogs modified with β-prolines. J. Pept. Sci. 2008, 14, 1283–1294. [Google Scholar] [CrossRef]
- Drygała, P.; Olejnik, J.; Mazur, A.; Kierus, K.; Jankowski, S.; Zimecki, M.; Zabrocki, J. Synthesis and immunosuppressive activity of cyclolinopeptide A analogues containing homophenylalanine. Eur. J. Med. Chem. 2009, 44, 3731–3738. [Google Scholar] [CrossRef] [PubMed]
- Jędrzejczak, K.; Hrynczyszyn, P.; Artym, J.; Kocięba, M.; Zimecki, M.; Zabrocki, J.; Jankowski, S. Synthesis and biological activity of cyclolinopeptide A analogues modified with γ3-bis(homophenylalanine). Eur. J. Med. Chem. 2014, 86, 515–527. [Google Scholar] [CrossRef] [PubMed]
- Jędrzejczak, K.; Hrynczyszyn, P.; Szczesio, M.; Artym, J.; Jastrząbek, T.; Kocięba, M.; Główka, M.; Huben, K.; Kochanowska, I.; Zimecki, M.; et al. Synthesis and biological activity of cyclolinopeptide A analogues modified with γ 4-bis(homo-phenylalanine). Bioorganic Med. Chem. 2017, 25, 4265–4276. [Google Scholar] [CrossRef]
- Katarzyńska, J.; Artym, J.; Kochanowska, I.; Jędrzejczak, K.; Zimecki, M.; Lisowski, M.; Wieczorek, R.; Piotrowski, Ł.; Marcinek, A.; Zabrocki, J.; et al. 4-Methylpseudoproline analogues of cyclolinopeptide A: Synthesis, structural analysis and evaluation of their suppressive effects in selected immunological assays. Peptides 2020, 132, 170365. [Google Scholar] [CrossRef] [PubMed]
- Katarzyńska, J.; Mazur, A.; Rudzińska, E.; Artym, J.; Zimecki, M.; Jankowski, S.; Zabrocki, J. Cyclolinopeptide derivatives modify methotrexate-induced suppression of the humoral immune response in mice. Eur. J. Med. Chem. 2011, 46, 4608–4617. [Google Scholar] [CrossRef]
- Zimecki, M.; Artym, J.; Kałas, W.; Strządała, L.; Kaleta-Kuratewicz, K.; Kuryszko, J.; Kaszuba, A.; Kaczmarek, K.; Zabrocki, J. Anti-Inflammatory Activity of a Cyclic Tetrapeptide in Mouse and Human Experimental Models. Pharmaceutics 2020, 12, 1030. [Google Scholar] [CrossRef]
- Bell, A.; McSteen, P.M.; Cebrat, M.; Picur, B.; Siemion, I.Z. Antimalarial activity of cyclolinopeptide A and its analogues. Acta Pol. Pharm. Drug Res. 2000, 57, 57. [Google Scholar]
- Kaneda, T.; Yoshida, H.; Nakajima, Y.; Toishi, M.; Nugroho, A.E.; Morita, H. Cyclolinopeptides, cyclic peptides from flaxseed with osteoclast differentiation inhibitory activity. Bioorganic Med. Chem. Lett. 2016, 26, 1760–1761. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, Y.; Yamamoto, N.; Hayashi, K.; Takeuchi, A.; Miwa, S.; Igarashi, K.; Higuchi, T.; Abe, K.; Yonezawa, H.; Araki, Y.; et al. Anti-tumor Effects of Cyclolinopeptide on Giant-cell Tumor of the Bone. Anticancer. Res. 2019, 39, 6145–6153. [Google Scholar] [CrossRef] [PubMed]
- Gallo, P.; Saviano, M.; Rossi, F.; Pavone, V.; Pedone, C.; Ragone, R.; Stiuso, P.; Colonna, G. Specific interaction between cyclophilin and cyclic peptides. Biopolymers 1995, 36, 273–281. [Google Scholar] [CrossRef]
- Gallo, P.; Rossi, F.; Saviano, M.; Pedone, C.; Colonna, G.; Ragone, R. Specific interaction between bovine cyclophilin A and synthetic analogues of cyclolinopeptide A. J. Biochem. 1998, 124, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Gaymes, T.J.; Cebrat, M.; Siemion, I.Z.; Kay, E.J. Cyclolinopeptide A (CLA) mediates its immunosuppressive activity through cyclophilin-dependent calcineurin inactivation. FEBS Lett. 1997, 418, 224–227. [Google Scholar] [CrossRef]
- Thomson, A.W.; Bonham, C.A.; Zeevi, A. Mode of Action of Tacrolimus (FK506): Molecular and Cellular Mechanisms. Ther. Drug Monit. 1995, 17, 584–591. [Google Scholar] [CrossRef]
- Mukerjee, N.; McGinnis, K.M.; Gnegy, M.E.; Wang, K.K. Caspase-Mediated Calcineurin Activation Contributes to IL-2 Release during T Cell Activation. Biochem. Biophys. Res. Commun. 2001, 285, 1192–1199. [Google Scholar] [CrossRef]
- Strasser, A.; Harris, A.W.; Huang, D.C.; Krammer, P.H.; Cory, S. Bcl-2 and Fas/APO-1 regulate distinct pathways to lymphocyte apoptosis. EMBO J. 1995, 14, 6136–6147. [Google Scholar] [CrossRef] [PubMed]
- Zimecki, M. Potential therapeutic interventions via EP2/EP4 prostaglandin receptors. Postępy Hig. Med. Doświadczalnej 2012, 66, 287–294. [Google Scholar] [CrossRef]
- Bishop-Bailey, D.; Burke-Gaffney, A.; Hellewell, P.G.; Pepper, J.R.; Mitchell, J.A. Cyclo-oxygenase-2 Regulates Inducible ICAM-1 and VCAM-1 Expression in Human Vascular Smooth Muscle Cells. Biochem. Biophys. Res. Commun. 1998, 249, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Kanda, N.; Koike, S.; Watanabe, S. IL-17 suppresses TNF-α–induced CCL27 production through induction of COX-2 in human keratinocytes. J. Allergy Clin. Immunol. 2005, 116, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
| Peptide | Action | Ref. |
|---|---|---|
| cyclolinopeptide A | suppression of: graft-versus-host reaction, allogeneic graft rejection, post-adjuvant polyarthritis, humoral and cellular immune response and reduction of hemolytic anemia in New Zealand Black mice | [4] |
| cyclolinopeptide A | inhibition of human Plasmodium falciparum growth in culture | [21] |
| cyclolinopeptide A | inhibition of osteoclast differentiation | [22] |
| cyclolinopeptide A | inhibition of bone giant cell tumor growth | [23] |
| H-Ile-Ile-Leu-Val-Pro-Pro- (R)-ɣ3hhPhe-(R)-ɣ3hhPhe-Leu-OH c(Leu-Ile-Ile-Leu-Val-Pro-Pro-Phe-(S)-ɣ3hhPhe-) | suppression of: carrageenan-induced footpad inflammation, LPS-induced TNF α production in whole blood cell cultures and effectual phase of the cellular immune response | [16] |
| c(Pro-Pro-β3hPhe-Phe-) | suppression of: contact sensitivity to oxazolone, toluene diisocyanate, nonspecific skin irritation, carrageenan inflammation in air poach, ovalbumin-induced pleurisy and dextran sulfate-induced colitis | [20] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zimecki, M.; Kaczmarek, K. Effects of Modifications on the Immunosuppressive Properties of Cyclolinopeptide A and Its Analogs in Animal Experimental Models. Molecules 2021, 26, 2538. https://doi.org/10.3390/molecules26092538
Zimecki M, Kaczmarek K. Effects of Modifications on the Immunosuppressive Properties of Cyclolinopeptide A and Its Analogs in Animal Experimental Models. Molecules. 2021; 26(9):2538. https://doi.org/10.3390/molecules26092538
Chicago/Turabian StyleZimecki, Michał, and Krzysztof Kaczmarek. 2021. "Effects of Modifications on the Immunosuppressive Properties of Cyclolinopeptide A and Its Analogs in Animal Experimental Models" Molecules 26, no. 9: 2538. https://doi.org/10.3390/molecules26092538
APA StyleZimecki, M., & Kaczmarek, K. (2021). Effects of Modifications on the Immunosuppressive Properties of Cyclolinopeptide A and Its Analogs in Animal Experimental Models. Molecules, 26(9), 2538. https://doi.org/10.3390/molecules26092538